The Accuracy of Point-of-Care Ultrasound (POCUS) in Acute Gallbladder Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Protocol
2.2. Inclusion Criteria
2.3. Exclusion Criteria
2.4. Point-of-Care Ultrasound Procedure
2.5. POCUS Findings and Diagnosis
2.6. Clinical Decision after Surgeon POCUS
2.7. Data Recording
2.8. Ethics Approval and Consent
2.9. Statistical Analysis
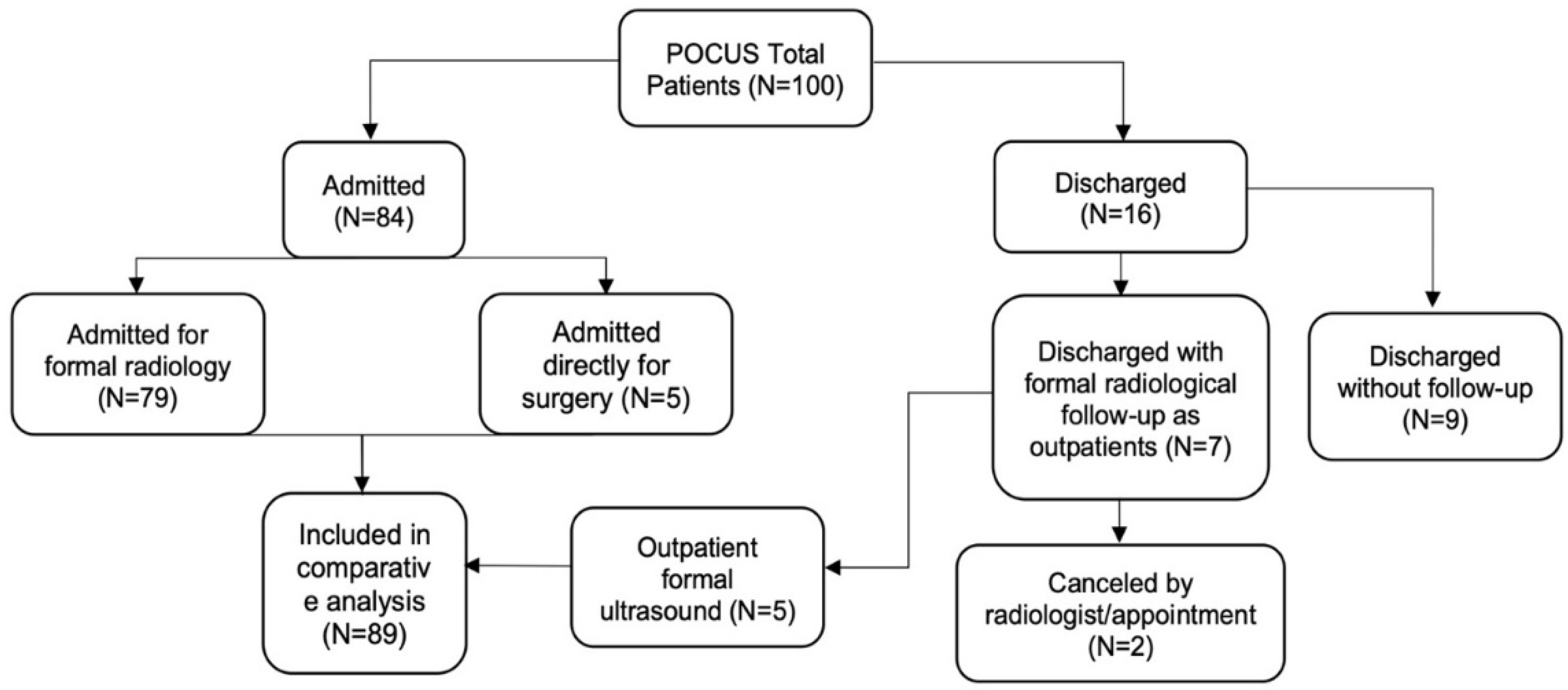
3. Results
3.1. Demographic Data
3.2. Clinical Presentation
3.3. Surgeon POCUS Diagnosis and Parameters
3.4. Accuracy of POCUS
3.5. Accuracy in Diagnosing Acute Cholecystitis
3.6. Common Bile Duct Assessment
3.7. The Timeframe of POCUS to Radiology or Surgery
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hilsden, R.; Leeper, R.; Koichopolos, J.; Vandelinde, J.D.; Parry, N.; Thompson, D.; Myslik, F. Point-of-care biliary ultrasound in the emergency department (BUSED): Implications for surgical referral and emergency department wait times. Trauma Surg. Acute Care Open 2018, 3, e000164. [Google Scholar] [CrossRef]
- Eskelinen, M.; Meklin, J.; Syrjänen, K.; Eskelinen, M. Performance of a Diagnostic Score in Confirming Acute Cholecystitis Among Patients with Acute Abdominal Pain. Anticancer. Res. 2020, 40, 6947–6956. [Google Scholar] [CrossRef] [PubMed]
- Wehrle, C.; Talukder, A.; Tien, L.; Parikh, S.; Devarakonda, A.; Holsten, S.; Fox, E.; Lawson, A. The Accuracy of Point-of-Care Ultrasound in the Diagnosis of Acute Cholecystitis. Am. Surg. 2021, 88, 267–272. [Google Scholar] [CrossRef]
- Freitas, M.L.; Frangos, S.G.; Frankel, H.L. The status of ultrasonography training and use in general surgery residency programs. J. Am. Coll. Surg. 2006, 202, 453–458. [Google Scholar] [CrossRef] [PubMed]
- Jarwan, W.; Alshamrani, A.A.; Alghamdi, A.; Mahmood, N.; Kharal, Y.M.; Rajendram, R.; Hussain, A. Point-of-Care ultrasound training: An Assessment of Interns’ needs and barriers to training. Cureus 2020, 12, e11209. [Google Scholar] [CrossRef]
- Yokoe, M.; Takada, T.; Strasberg, S.M.; Solomkin, J.S.; Mayumi, T.; Gomi, H.; Pitt, H.A.; Garden, O.J.; Kiriyama, S.; Hata, J.; et al. TG13 diagnostic criteria and severity grading of acute cholecystitis (with videos). J. Hepato-Biliary-Pancreat. Sci. 2013, 20, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Zago, M.; Martinez Casas, I.; Pereira, J.; Mariani, D.; Silva, A.R.; Casamassima, A.; Barbosa, E.; Ferreira, F.; Ruesseler, M.; Bass, G.A.; et al. Tailored Ultrasound Learning for Acute Care Surgeons. A Review of The Musec (Modular Ultrasound Estes Course) Project. Eur. J. Trauma Emerg. Surg. 2016, 42, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef]
- The Royal College of Radiologists. Standards for Patient Consent Particular to Radiology, 2nd ed.; The Royal College of Radiologists: London, UK, 2012. [Google Scholar]
- Society and College of Radiographers; British Medical Ultrasound Society. Guidelines for Professional Ultrasound Practice, 4th ed.; Society and College of Radiographers: London, UK, 2019. [Google Scholar]
- Altman, D.; Machin, D.; Bryant, T.; Gardner, M. Statistics with Confidence, 2nd ed.; BMJ Books: Hoboken, NJ, USA, 2000. [Google Scholar]
- Pereira, J.; Bass, G.A.; Mariani, D.; Dumbrava, B.D.; Casamassima, A.; Da Silva, A.R.; Pinheiro, L.; Martinez-Casas, I.; Zago, M. Surgeon-performed point-of-care ultrasound for acute cholecystitis: Indications and limitations: A European Society for Trauma and Emergency Surgery (ESTES) consensus statement. Eur. J. Trauma Emerg. Surg. 2020, 46, 173–183. [Google Scholar] [CrossRef]
- Williams, R.J.; Windsor, A.C.; Rosin, R.D.; Mann, D.V.; Crofton, M. Ultrasound scanning of the acute abdomen by surgeons in training. Ann. R. Coll. Surg. Engl. 1994, 76, 228. [Google Scholar]
- Villar, J.; Summers, S.M.; Menchine, M.D.; Fox, J.C.; Wang, R. The absence of gallstones on point-of-care ultrasound rules out acute cholecystitis. J. Emerg. Med. 2015, 49, 475–480. [Google Scholar] [CrossRef]
- Miura, F.; Takada, T.; Strasberg, S.M.; Solomkin, J.S.; Pitt, H.A.; Gouma, D.J.; Garden, O.J.; Büchler, M.W.; Yoshida, M.; Mayumi, T.; et al. TG13 flowchart for the management of acute cholangitis and cholecystitis. J. Hepato-Biliary-Pancreat. Sci. 2013, 20, 47–54. [Google Scholar] [CrossRef]
- O’Connor, O.J.; Maher, M.M. Imaging of cholecystitis. Am. J. Roentgenol. 2011, 196, W367–W374. [Google Scholar] [CrossRef]
- Kendall, J.L.; Shimp, R.J. Performance and interpretation of focused right upper quadrant ultrasound by emergency physicians. J. Emerg. Med. 2001, 21, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Rosen, C.L.; Brown, D.F.; Chang, Y.; Moore, C.; Averill, N.J.; Arkoff, L.J.; McCabe, C.J.; Wolfe, R.E. Ultrasonography by emergency physicians in patients with suspected cholecystitis. Am. J. Emerg. Med. 2001, 19, 32–36. [Google Scholar] [CrossRef]
- Gustafsson, C.; Lindelius, A.; Törngren, S.; Järnbert-Pettersson, H.; Sondén, A. Surgeon-performed ultrasound in diagnosing acute cholecystitis and appendicitis. World J. Surg. 2018, 42, 3551–3559. [Google Scholar] [CrossRef] [PubMed]
- Summers, S.M.; Scruggs, W.; Menchine, M.D.; Lahham, S.; Anderson, C.; Amr, O.; Lotfipour, S.; Cusick, S.S.; Fox, J.C. A prospective evaluation of emergency department bedside ultrasonography for the detection of acute cholecystitis. Ann. Emerg. Med. 2010, 56, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Borzellino, G.; Massimiliano Motton, A.P.; Minniti, F.; Montemezzi, S.; Tomezzoli, A.; Genna, M. Sonographic diagnosis of acute cholecystitis in patients with symptomatic gallstones. J. Clin. Ultrasound 2016, 44, 152–158. [Google Scholar] [CrossRef]
- Carmody, E.; Arenson, A.M.; Hanna, S. Failed or difficult laparoscopic cholecystectomy: Can preoperative ultrasonography identify potential problems? J. Clin. Ultrasound 1994, 22, 391–396. [Google Scholar] [CrossRef]
- Scruggs, W.; Fox, J.C.; Potts, B.; Zlidenny, A.; McDonough, J.; Anderson, C.L.; Larson, J.; Barajas, G.; Langdorf, M.I. Accuracy of ED bedside ultrasound for identification of gallstones: Retrospective analysis of 575 studies. West. J. Emerg. Med. 2008, 9, 1. [Google Scholar]
- Fang, R.; Pilcher, J.A.; Putnam, A.T.; Smith, T.; Smith, D.L. Accuracy of surgeon-performed gallbladder ultrasound. Am. J. Surg. 1999, 178, 475–478. [Google Scholar] [CrossRef] [PubMed]
- Rozycki, G.S. Surgeon-performed ultrasound: Its use in clinical practice. Ann. Surg. 1998, 228, 16. [Google Scholar] [CrossRef]
- Gaspari, R.J.; Dickman, E.; Blehar, D. Learning curve of bedside ultrasound of the gallbladder. J. Emerg. Med. 2009, 37, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Carnell, J.; Fischer, J.W.; Stone, M.B.; Nagdev, A.D. Emergency ultrasound fellowship program compliance with ACEP guidelines in 2010: A web- based survey. Acad. Emerg. Med. 2011, 18, S4–S249. [Google Scholar]
- Mohamed, M.S.; Mufti, G.R. The surgical assessment unit—Effective strategy for improvement of the emergency surgical pathway? J. R. Soc. Med. 2005, 98, 14–17. [Google Scholar] [PubMed]
- Allemann, F.; Cassina, P.; Röthlin, M.; Largiadèr, F. Ultrasound scans done by surgeons for patients with acute abdominal pain: A prospective study. Eur. J. Surg. 1999, 165, 966–970. [Google Scholar] [CrossRef]
- Healy, P.; McCrone, L.; Tully, R.; Flannery, E.; Flynn, A.; Cahir, C.; Arumugasamy, M.; Walsh, T. Virtual outpatient clinic as an alternative to an actual clinic visit after surgical discharge: A randomised controlled trial. BMJ Qual. Saf. 2019, 28, 24–31. [Google Scholar] [CrossRef]
- Wu, X.D.; Tian, X.; Liu, M.M.; Wu, L.; Zhao, S.; Zhao, L. Meta-analysis comparing early versus delayed laparoscopic cholecystectomy for acute cholecystitis. J. Br. Surg. 2015, 102, 1302–1313. [Google Scholar] [CrossRef]


| Gender | |
|---|---|
| Male | 34 |
| Female | 66 |
| Age | |
| Mean age | 50.5 (18–93) |
| Presentation | |
| RUQ pain | 96 (96%) |
| RP elevation | 43 (43%) |
| Leukocytosis (WCC > 11 × 109/L) | 34 (34%) |
| Murphy’s sign positive | 29 (29%) |
| Abnormal LFTs | 27 (27%) |
| Fever (temperature > 37.5 °C) | 15 (15%) |
| Jaundice | 11 (11%) |
| Final POCUS Diagnosis | 100 (%) |
|---|---|
| Normal findings | 38 (42.7%) |
| Symptomatic gallstones/sludge without inflammation | 23 (25.8%) |
| Acute gallstone cholecystitis | 24 (27%) |
| Acalculous cholecystitis | 5 (5.6%) |
| Complicated acute cholecystitis | 3 (3.4%) |
| Isolated CBD dilatation | 2 (2.2%) |
| Liver pathology | 2 (2.2%) |
| Patient Number | Explanation |
|---|---|
| 1 | Inconclusive diagnosis for both POCUS and radiology. |
| 2 | POCUS diagnosed gallstones. CT scan also diagnosed splenic infarction. This patient would have required CT because of atypical presentation. |
| 3 | POCUS reported no gallstones. Radiologist diagnosed a gallstone. |
| 4 | POCUS was unable to visualize the gallbladder content due to gas. Radiologist reported a difficult examination, inconclusive for acute cholecystitis. |
| 5 | POCUS diagnosed acute cholecystitis. Radiologist identified gallstones but not acute cholecystitis. |
| 6 | POCUS diagnosed acute calculous cholecystitis. Radiologist reported no acute cholecystitis. |
| 7 | POCUS diagnosed acute acalculous cholecystitis. Radiology reported no cholecystitis after 21 h of antibiotic therapy. |
| 8 | POCUS diagnosed acute cholecystitis. CT revealed a superior mesenteric artery thrombus. |
| 9 | POCUS was unable to visualize the gallbladder content due to gas. Radiologist detected gallstones after the patient had been fasting. |
| 10 | POCUS detected a gallstone in the gallbladder neck. Radiologist did not see gallstones at 48 h. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dumbrava, B.-D.; Bass, G.A.; Jumean, A.; Birido, N.; Corbally, M.; Pereira, J.; Biloslavo, A.; Zago, M.; Walsh, T.N. The Accuracy of Point-of-Care Ultrasound (POCUS) in Acute Gallbladder Disease. Diagnostics 2023, 13, 1248. https://doi.org/10.3390/diagnostics13071248
Dumbrava B-D, Bass GA, Jumean A, Birido N, Corbally M, Pereira J, Biloslavo A, Zago M, Walsh TN. The Accuracy of Point-of-Care Ultrasound (POCUS) in Acute Gallbladder Disease. Diagnostics. 2023; 13(7):1248. https://doi.org/10.3390/diagnostics13071248
Chicago/Turabian StyleDumbrava, Bogdan-Daniel, Gary Alan Bass, Amro Jumean, Nuha Birido, Martin Corbally, Jorge Pereira, Alan Biloslavo, Mauro Zago, and Thomas Noel Walsh. 2023. "The Accuracy of Point-of-Care Ultrasound (POCUS) in Acute Gallbladder Disease" Diagnostics 13, no. 7: 1248. https://doi.org/10.3390/diagnostics13071248
APA StyleDumbrava, B.-D., Bass, G. A., Jumean, A., Birido, N., Corbally, M., Pereira, J., Biloslavo, A., Zago, M., & Walsh, T. N. (2023). The Accuracy of Point-of-Care Ultrasound (POCUS) in Acute Gallbladder Disease. Diagnostics, 13(7), 1248. https://doi.org/10.3390/diagnostics13071248






