Mammaglobin-A Expression Is Highly Specific for Tumors Derived from the Breast, the Female Genital Tract, and the Salivary Gland
Abstract
1. Introduction
2. Materials and Methods
2.1. Tissue Microarrays (TMAs)
2.2. Immunohistochemistry (IHC)
2.3. Statistics
3. Results
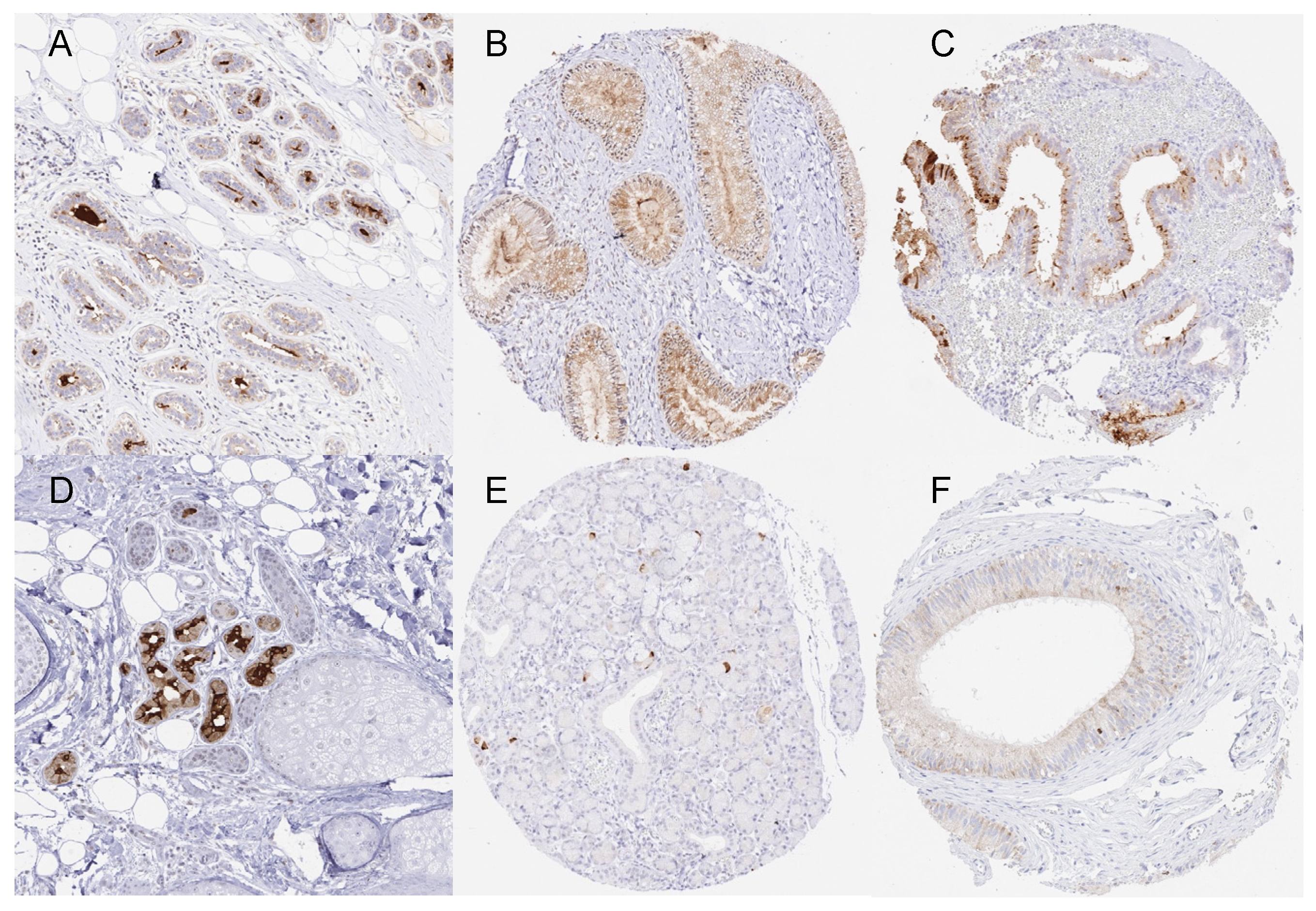
3.1. Technical Issues
3.2. Mammaglobin-A in Normal Tissue
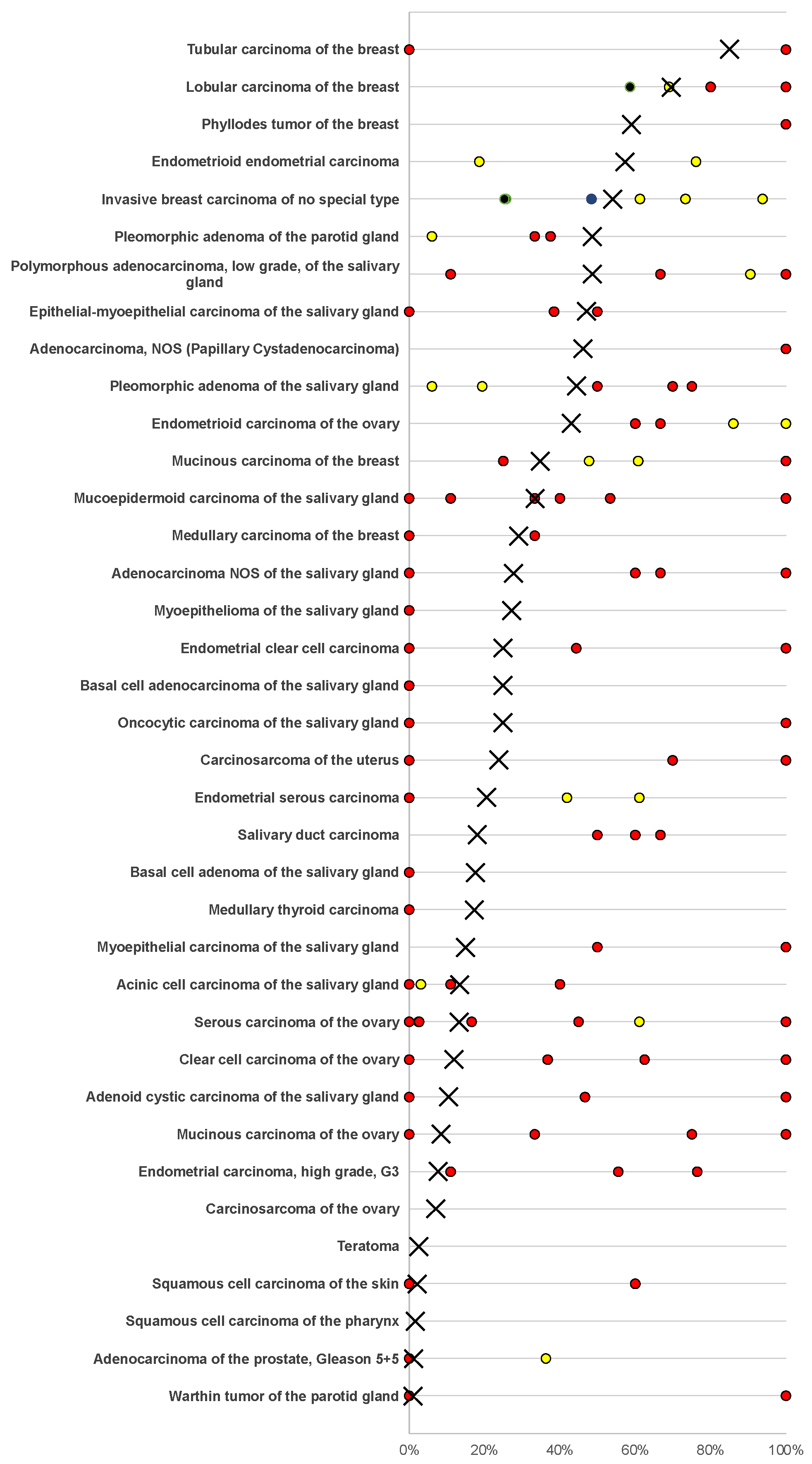
3.3. Mammaglobin-A in Neoplastic Tissues
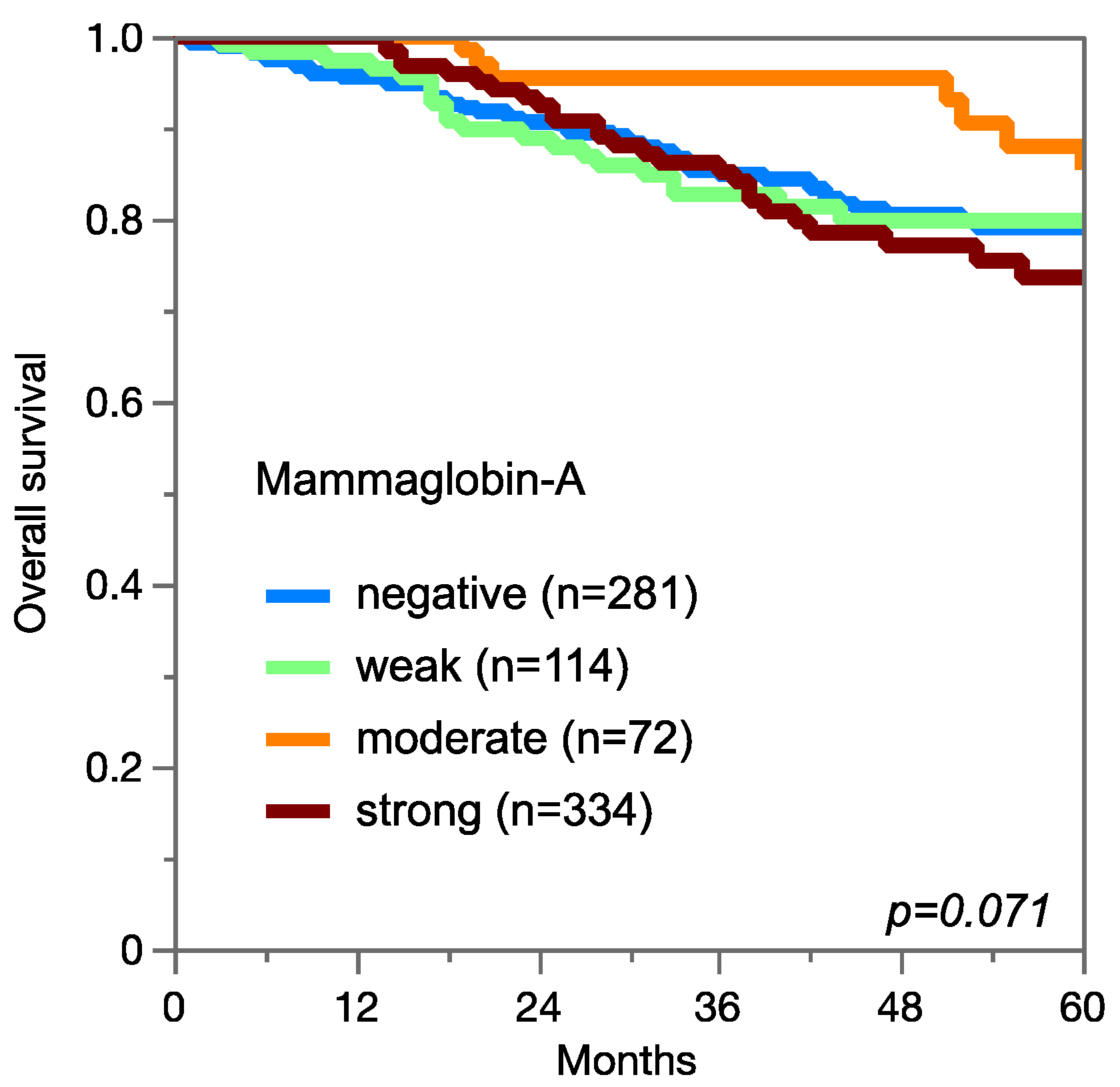
3.4. Mammaglobin-A Expression, Tumor Phenotype, and Prognosis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Watson, M.A.; Darrow, C.; Zimonjic, D.B.; Popescu, N.C.; Fleming, T.P. Structure and transcriptional regulation of the human mammaglobin gene, a breast cancer associated member of the uteroglobin gene family localized to chromosome 11q13. Oncogene 1998, 16, 817–824. [Google Scholar] [CrossRef] [PubMed]
- Watson, M.A.; Watson, M.A.; Fleming, T.P. Mammaglobin, a mammary-specific member of the uteroglobin gene family, is overexpressed in human breast cancer. Cancer. Res. 1996, 56, 860–865. [Google Scholar] [PubMed]
- Ni, J.; Kalff-Suske, M.; Gentz, R.; Schageman, J.; Beato, M.; Klug, J. All human genes of the uteroglobin family are localized on chromosome 11q12.2 and form a dense cluster. Ann. N. Y. Acad. Sci. 2000, 923, 25–42. [Google Scholar] [CrossRef] [PubMed]
- Becker, R.M.; Darrow, C.; Zimonjic, D.B.; Popescu, N.C.; Watson, M.A.; Fleming, T.P. Identification of mammaglobin B, a novel member of the uteroglobin gene family. Genomics 1998, 54, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Brown, N.M.; Stenzel, T.T.; Friedman, P.N.; Henslee, J.; Huper, G.; Marks, J.R. Evaluation of expression based markers for the detection of breast cancer cells. Breast Cancer Res. Treat. 2006, 97, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, A.B.; Kundu, G.C.; Mantile-Selvaggi, G.; Yuan, C.J.; Mandal, A.K.; Chattopadhyay, S.; Zheng, F.; Pattabiraman, N.; Zhang, Z. Uteroglobin: A novel cytokine? Cell Mol. Life Sci. 1999, 55, 771–787. [Google Scholar] [CrossRef]
- Mukherjee, A.B.; Kundu, G.C.; Mandal, A.K.; Pattabiraman, N.; Yuan, C.J.; Zhang, Z. Uteroglobin: Physiological role in normal glomerular function uncovered by targeted disruption of the uteroglobin gene in mice. Am. J. Kidney. Dis. 1998, 32, 1106–1120. [Google Scholar] [CrossRef]
- Miele, L. Antiflammins Bioactive peptides derived from uteroglobin. Ann. N. Y. Acad. Sci. 2000, 923, 128–140. [Google Scholar] [CrossRef]
- Miele, L.; Cordella-Miele, E.; Mukherjee, A.B. Uteroglobin: Structure, molecular biology, and new perspectives on its function as a phospholipase A2 inhibitor. Endocr. Rev. 1987, 8, 474–490. [Google Scholar] [CrossRef]
- Vasanthakumar, G.; Manjunath, R.; Mukherjee, A.B.; Warabi, H.; Schiffmann, E. Inhibition of phagocyte chemotaxis by uteroglobin, an inhibitor of blastocyst rejection. Biochem. Pharmacol. 1988, 37, 389–394. [Google Scholar] [CrossRef]
- Levin, S.W.; Butler, J.D.; Schumacher, U.K.; Wightman, P.D.; Mukherjee, A.B. Uteroglobin inhibits phospholipase A2 activity. Life Sci. 1986, 38, 1813–1819. [Google Scholar] [CrossRef] [PubMed]
- Al Joudi, F.S. Human mammaglobin in breast cancer: A brief review of its clinical utility. Indian J. Med. Res. 2014, 139, 675–685. [Google Scholar] [PubMed]
- Carter, D.; Douglass, J.F.; Cornellison, C.D.; Retter, M.W.; Johnson, J.C.; Bennington, A.A.; Fleming, T.P.; Reed, S.G.; Houghton, R.L.; Diamond, D.L.; et al. Purification and characterization of the mammaglobin/lipophilin B complex, a promising diagnostic marker for breast cancer. Biochemistry 2002, 41, 6714–6722. [Google Scholar] [CrossRef] [PubMed]
- Zafrakas, M.; Petschke, B.; Donner, A.; Fritzsche, F.; Kristiansen, G.; Knuchel, R.; Dahl, E. Expression analysis of mammaglobin A (SCGB2A2) and lipophilin B (SCGB1D2) in more than 300 human tumors and matching normal tissues reveals their co-expression in gynecologic malignancies. BMC Cancer 2006, 6, 88. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Yang, X.; Duan, C.; Li, J.; Tong, R.; Fan, Y.; Feng, J.; Cao, R.; Zhong, W.; Feng, X.; et al. Identification and characterization of mammaglobin-A epitope in heterogenous breast cancers for enhancing tumor-targeting therapy. Signal. Transduct. Target. 2020, 5, 82. [Google Scholar] [CrossRef]
- Picot, N.; Guerrette, R.; Beauregard, A.P.; Jean, S.; Michaud, P.; Harquail, J.; Benzina, S.; Robichaud, G.A. Mammaglobin 1 promotes breast cancer malignancy and confers sensitivity to anticancer drugs. Mol. Carcinog. 2016, 55, 1150–1162. [Google Scholar] [CrossRef]
- Koh, E.H.; Cho, Y.W.; Mun, Y.J.; Ryu, J.H.; Kim, E.J.; Choi, D.S.; Maeng, K.Y.; Han, J.; Kang, D. Upregulation of human mammaglobin reduces migration and invasion of breast cancer cells. Cancer. Investig. 2014, 32, 22–29. [Google Scholar] [CrossRef]
- Sjodin, A.; Ljuslinder, I.; Henriksson, R.; Hedman, H. Mammaglobin and lipophilin B expression in breast tumors and their lack of effect on breast cancer cell proliferation. Anticancer Res. 2008, 28, 1493–1498. [Google Scholar]
- De Lara, S.; Parris, T.Z.; Werner, R.E.; Helou, K.; Kovacs, A. GATA3 as a putative marker of breast cancer metastasis-A retrospective immunohistochemical study. Breast J. 2018, 24, 184–188. [Google Scholar] [CrossRef]
- Talaat, I.M.; Hachim, M.Y.; Hachim, I.Y.; Ibrahim, R.A.E.; Ahmed, M.; Tayel, H.Y. Bone marrow mammaglobin-1 (SCGB2A2) immunohistochemistry expression as a breast cancer specific marker for early detection of bone marrow micrometastases. Sci. Rep. 2020, 10, 13061. [Google Scholar] [CrossRef]
- Shaoxian, T.; Baohua, Y.; Xiaoli, X.; Yufan, C.; Xiaoyu, T.; Hongfen, L.; Rui, B.; Xiangjie, S.; Ruohong, S.; Wentao, Y. Characterisation of GATA3 expression in invasive breast cancer: Differences in histological subtypes and immunohistochemically defined molecular subtypes. J. Clin. Pathol. 2017, 70, 926–934. [Google Scholar] [CrossRef] [PubMed]
- Dyhdalo, K.S.; Booth, C.N.; Brainard, J.A.; Croyle, M.C.; Kolosiwsky, A.M.; Goyal, A.; Gildea, T.R.; Almeida, F.A.; Nassar, A.; Reynolds, J.P. Utility of GATA3, mammaglobin, GCDFP-15, and ER in the detection of intrathoracic metastatic breast carcinoma. J. Am. Soc. Cytopathol. 2015, 4, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Huo, L.; Zhang, J.; Gilcrease, M.Z.; Gong, Y.; Wu, Y.; Zhang, H.; Resetkova, E.; Hunt, K.K.; Deavers, M.T. Gross cystic disease fluid protein-15 and mammaglobin A expression determined by immunohistochemistry is of limited utility in triple-negative breast cancer. Histopathology 2013, 62, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Cimino, P.J., Jr.; Perrin, R.J. Mammaglobin-A immunohistochemistry in primary central nervous system neoplasms and intracranial metastatic breast carcinoma. Appl. Immunohistochem. Mol. Morphol. 2014, 22, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, E.; Tsunoda, N.; Hatanaka, Y.; Mori, N.; Iwata, H.; Yatabe, Y. Breast-specific expression of MGB1/mammaglobin: An examination of 480 tumors from various organs and clinicopathological analysis of MGB1-positive breast cancers. Mod. Pathol. 2007, 20, 208–214. [Google Scholar] [CrossRef]
- Hagemann, I.S.; Pfeifer, J.D.; Cao, D. Mammaglobin expression in gynecologic adenocarcinomas. Hum. Pathol. 2013, 44, 628–635. [Google Scholar] [CrossRef]
- Bellone, S.; Tassi, R.; Betti, M.; English, D.; Cocco, E.; Gasparrini, S.; Bortolomai, I.; Black, J.D.; Todeschini, P.; Romani, C.; et al. Mammaglobin B (SCGB2A1) is a novel tumour antigen highly differentially expressed in all major histological types of ovarian cancer: Implications for ovarian cancer immunotherapy. Br. J. Cancer 2013, 109, 462–471. [Google Scholar] [CrossRef]
- Kandalaft, P.L.; Simon, R.A.; Isacson, C.; Gown, A.M. Comparative Sensitivities and Specificities of Antibodies to Breast Markers GCDFP-15, Mammaglobin A, and Different Clones of Antibodies to GATA-3: A Study of 338 Tumors Using Whole Sections. Appl. Immunohistochem. Mol. Morphol. 2016, 24, 609–614. [Google Scholar] [CrossRef]
- Kononen, J.; Bubendorf, L.; Kallioniemi, A.; Barlund, M.; Schraml, P.; Leighton, S.; Torhorst, J.; Mihatsch, M.J.; Sauter, G.; Kallioniemi, O.P. Tissue microarrays for high-throughput molecular profiling of tumor specimens. Nat. Med. 1998, 4, 844–847. [Google Scholar] [CrossRef]
- Mirlacher, M.; Simon, R. Recipient block TMA technique. Methods Mol. Biol. 2010, 664, 37–44. [Google Scholar]
- Simon, R.; Mirlacher, M.; Sauter, G. Immunohistochemical analysis of tissue microarrays. Methods Mol. Biol. 2010, 664, 113–126. [Google Scholar] [PubMed]
- Uhlen, M.; Bandrowski, A.; Carr, S.; Edwards, A.; Ellenberg, J.; Lundberg, E.; Rimm, D.L.; Rodriguez, H.; Hiltke, T.; Snyder, M.; et al. A proposal for validation of antibodies. Nat. Methods 2016, 13, 823–827. [Google Scholar] [CrossRef]
- Thul, P.J.; Akesson, L.; Wiking, M.; Mahdessian, D.; Geladaki, A.; Ait Blal, H.; Alm, T.; Asplund, A.; Bjork, L.; Breckels, L.M.; et al. A subcellular map of the human proteome. Science 2017, 356, 6340. [Google Scholar] [CrossRef] [PubMed]
- Lizio, M.; Harshbarger, J.; Shimoji, H.; Severin, J.; Kasukawa, T.; Sahin, S.; Abugessaisa, I.; Fukuda, S.; Hori, F.; Ishikawa-Kato, S.; et al. Gateways to the FANTOM5 promoter level mammalian expression atlas. Genome. Biol. 2015, 16, 22. [Google Scholar] [CrossRef] [PubMed]
- Lizio, M.; Abugessaisa, I.; Noguchi, S.; Kondo, A.; Hasegawa, A.; Hon, C.C.; de Hoon, M.; Severin, J.; Oki, S.; Hayashizaki, Y.; et al. Update of the FANTOM web resource: Expansion to provide additional transcriptome atlases. Nucleic. Acids Res. 2019, 47, D752–D758. [Google Scholar] [CrossRef] [PubMed]
- Consortium, G.T. The Genotype-Tissue Expression (GTEx) project. Nat. Genet. 2013, 45, 580–585. [Google Scholar]
- Tassi, R.A.; Bignotti, E.; Falchetti, M.; Calza, S.; Ravaggi, A.; Rossi, E.; Martinelli, F.; Bandiera, E.; Pecorelli, S.; Santin, A.D. Mammaglobin B expression in human endometrial cancer. Int. J. Gynecol. Cancer. 2008, 18, 1090–1096. [Google Scholar] [CrossRef]
- Soares, C.D.; de Lima Morais, T.M.; Carlos, R.; Martins, M.D.; de Almeida, O.P.; Mariano, F.V.; Altemani, A. Immunohistochemical expression of mammaglobin in salivary duct carcinomas de novo and salivary duct carcinoma ex pleomorphic adenoma. Hum. Pathol. 2019, 92, 59–66. [Google Scholar] [CrossRef]
- Sauter, G.; Simon, R.; Hillan, K. Tissue microarrays in drug discovery. Nat. Rev. Drug Discov. 2003, 2, 962–972. [Google Scholar] [CrossRef]
- Miettinen, M.; McCue, P.A.; Sarlomo-Rikala, M.; Rys, J.; Czapiewski, P.; Wazny, K.; Langfort, R.; Waloszczyk, P.; Biernat, W.; Lasota, J.; et al. GATA3: A multispecific but potentially useful marker in surgical pathology: A systematic analysis of 2500 epithelial and nonepithelial tumors. Am. J. Surg. Pathol. 2014, 38, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Zhang, T. Human mammaglobin: A specific marker for breast cancer prognosis. J. BUON 2016, 21, 35–41. [Google Scholar] [PubMed]
- Lee, G.W.; Kim, J.Y.; Koh, E.H.; Kang, D.; Choi, D.S.; Maeng, K.Y.; Lee, J.S. Plasma human mammaglobin mRNA associated with poor outcome in patients with breast cancer. Genet. Mol. Res. 2012, 11, 4034–4042. [Google Scholar] [CrossRef] [PubMed]
- Nunez-Villar, M.J.; Martinez-Arribas, F.; Pollan, M.; Lucas, A.R.; Sanchez, J.; Tejerina, A.; Schneider, J. Elevated mammaglobin (h-MAM) expression in breast cancer is associated with clinical and biological features defining a less aggressive tumour phenotype. Breast Cancer Res. 2003, 5, R65–R70. [Google Scholar] [CrossRef]
- Raica, M.; Cimpean, A.M.; Meche, A.; Alexa, A.; Suciu, C.; Muresan, A. Analysis of the immunohistochemical expression of mammaglobin A in primary breast carcinoma and lymph node metastasis. Rom. J. Morphol. Embryol. 2009, 50, 341–347. [Google Scholar]
- Span, P.N.; Waanders, E.; Manders, P.; Heuvel, J.J.; Foekens, J.A.; Watson, M.A.; Beex, L.V.; Sweep, F.C. Mammaglobin is associated with low-grade, steroid receptor-positive breast tumors from postmenopausal patients, and has independent prognostic value for relapse-free survival time. J. Clin. Oncol. 2004, 22, 691–698. [Google Scholar] [CrossRef] [PubMed]
- Guan, X.F.; Hamedani, M.K.; Adeyinka, A.; Walker, C.; Kemp, A.; Murphy, L.C.; Watson, P.H.; Leygue, E. Relationship between mammaglobin expression and estrogen receptor status in breast tumors. Endocrine 2003, 21, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Baker, E.; Whiteoak, N.; Hall, L.; France, J.; Wilson, D.; Bhaskar, P. Mammaglobin-A, VEGFR3, and Ki67 in Human Breast Cancer Pathology and Five Year Survival. Breast Cancer Auckl. 2019, 13, 1178223419858957. [Google Scholar] [CrossRef] [PubMed]
- Ni, Y.B.; Tsang, J.Y.; Chan, S.K.; Tse, G.M. GATA-binding protein 3, gross cystic disease fluid protein-15 and mammaglobin have distinct prognostic implications in different invasive breast carcinoma subgroups. Histopathology 2015, 67, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Fritzsche, F.R.; Thomas, A.; Winzer, K.J.; Beyer, B.; Dankof, A.; Bellach, J.; Dahl, E.; Dietel, M.; Kristiansen, G. Co-expression and prognostic value of gross cystic disease fluid protein 15 and mammaglobin in primary breast cancer. Histol. Histopathol. 2007, 22, 1221–1230. [Google Scholar] [PubMed]
- Friedmann-Morvinski, D.; Verma, I.M. Dedifferentiation and reprogramming: Origins of cancer stem cells. EMBO Rep. 2014, 15, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Jopling, C.; Boue, S.; Izpisua Belmonte, J.C. Dedifferentiation, transdifferentiation and reprogramming: Three routes to regeneration. Nat. Rev. Mol. Cell Biol. 2011, 12, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Lerret, N.M.; Rogozinska, M.; Jaramillo, A.; Marzo, A.L. Adoptive transfer of Mammaglobin-A epitope specific CD8 T cells combined with a single low dose of total body irradiation eradicates breast tumors. PLoS ONE 2012, 7, e41240. [Google Scholar] [CrossRef]
- Cui, H.; Zhang, W.; Hu, W.; Liu, K.; Wang, T.; Ma, N.; Liu, X.; Liu, Y.; Jiang, Y. Recombinant mammaglobin A adenovirus-infected dendritic cells induce mammaglobin A-specific CD8+ cytotoxic T lymphocytes against breast cancer cells in vitro. PLoS ONE 2013, 8, e63055. [Google Scholar] [CrossRef] [PubMed]
- Viehl, C.T.; Frey, D.M.; Phommaly, C.; Chen, T.; Fleming, T.P.; Gillanders, W.E.; Eberlein, T.J.; Goedegebuure, P.S. Generation of mammaglobin-A-specific CD4 T cells and identification of candidate CD4 epitopes for breast cancer vaccine strategies. Breast Cancer Res. Treat. 2008, 109, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Tiriveedhi, V.; Sarma, N.J.; Subramanian, V.; Fleming, T.P.; Gillanders, W.E.; Mohanakumar, T. Identification of HLA-A24-restricted CD8(+) cytotoxic T-cell epitopes derived from mammaglobin-A, a human breast cancer-associated antigen. Hum. Immunol. 2012, 73, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Ilias Basha, H.; Tiriveedhi, V.; Fleming, T.P.; Gillanders, W.E.; Mohanakumar, T. Identification of immunodominant HLA-B7-restricted CD8+ cytotoxic T cell epitopes derived from mammaglobin-A expressed on human breast cancers. Breast Cancer Res. Treat. 2011, 127, 81–89. [Google Scholar] [CrossRef]
- Soysal, S.D.; Muenst, S.; Kan-Mitchell, J.; Huarte, E.; Zhang, X.; Wilkinson-Ryan, I.; Fleming, T.; Tiriveedhi, V.; Mohanakumar, T.; Li, L.; et al. Identification and translational validation of novel mammaglobin-A CD8 T cell epitopes. Breast Cancer Res. Treat. 2014, 147, 527–537. [Google Scholar] [CrossRef]
- Kim, S.W.; Goedegebuure, P.; Gillanders, W.E. Mammaglobin-A is a target for breast cancer vaccination. Oncoimmunology 2016, 5, e1069940. [Google Scholar] [CrossRef]


| Mammaglobin-A Immunostaining | |||||||
|---|---|---|---|---|---|---|---|
| Tumor Entity | on TMA (n) | Anal. (n) | Neg. (%) | Weak (%) | Mod. (%) | Str. (%) | |
| Tumors of the skin | Pilomatricoma | 35 | 32 | 100.0 | 0.0 | 0.0 | 0.0 |
| Basal cell carcinoma | 88 | 83 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Benign nevus | 29 | 29 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Squamous cell carcinoma of the skin | 90 | 90 | 97.8 | 2.2 | 0.0 | 0.0 | |
| Malignant melanoma | 48 | 45 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Merkel cell carcinoma | 46 | 41 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Tumors of the head and neck | Squamous cell carcinoma of the larynx | 110 | 105 | 100.0 | 0.0 | 0.0 | 0.0 |
| Squamous cell carcinoma of the pharynx | 60 | 59 | 98.3 | 1.7 | 0.0 | 0.0 | |
| Oral squamous cell carcinoma (floor of the mouth) | 130 | 129 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Pleomorphic adenoma of the parotid gland | 50 | 39 | 51.3 | 10.3 | 15.4 | 23.1 | |
| Warthin tumor of the parotid gland | 104 | 98 | 99.0 | 0.0 | 1.0 | 0.0 | |
| Adenocarcinoma, NOS (papillary cystadenocarcinoma) | 14 | 13 | 53.8 | 0.0 | 0.0 | 46.2 | |
| Salivary duct carcinoma | 15 | 11 | 81.8 | 9.1 | 0.0 | 9.1 | |
| Acinic cell carcinoma of the salivary gland | 181 | 148 | 86.5 | 1.4 | 1.4 | 10.8 | |
| Adenocarcinoma NOS of the salivary gland | 109 | 83 | 72.3 | 6.0 | 6.0 | 15.7 | |
| Adenoid cystic carcinoma of the salivary gland | 180 | 114 | 89.5 | 7.9 | 0.0 | 2.6 | |
| Basal cell adenocarcinoma of the salivary gland | 25 | 24 | 75.0 | 20.8 | 0.0 | 4.2 | |
| Basal cell adenoma of the salivary gland | 101 | 91 | 82.4 | 9.9 | 5.5 | 2.2 | |
| Epithelial–myoepithelial carcinoma of the salivary gland | 53 | 51 | 52.9 | 23.5 | 7.8 | 15.7 | |
| Mucoepidermoid carcinoma of the salivary gland | 343 | 259 | 66.4 | 9.7 | 6.9 | 17.0 | |
| Myoepithelial carcinoma of the salivary gland | 21 | 20 | 85.0 | 5.0 | 10.0 | 0.0 | |
| Myoepithelioma of the salivary gland | 11 | 11 | 72.7 | 18.2 | 0.0 | 9.1 | |
| Oncocytic carcinoma of the salivary gland | 12 | 12 | 75.0 | 16.7 | 0.0 | 8.3 | |
| Polymorphous adenocarcinoma, low grade, of the salivary gland | 41 | 35 | 51.4 | 34.3 | 11.4 | 2.9 | |
| Pleomorphic adenoma of the salivary gland | 53 | 36 | 55.6 | 13.9 | 13.9 | 16.7 | |
| Tumors of the lung, pleura, and thymus | Adenocarcinoma of the lung | 246 | 176 | 100.0 | 0.0 | 0.0 | 0.0 |
| Squamous cell carcinoma of the lung | 130 | 69 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Small cell carcinoma of the lung | 20 | 16 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Mesothelioma, epithelioid | 39 | 28 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Mesothelioma, biphasic | 76 | 63 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Thymoma | 29 | 29 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Tumors of the female genital tract | Squamous cell carcinoma of the vagina | 78 | 73 | 100.0 | 0.0 | 0.0 | 0.0 |
| Squamous cell carcinoma of the vulva | 130 | 124 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Squamous cell carcinoma of the cervix | 130 | 125 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Endometrioid endometrial carcinoma | 236 | 225 | 42.7 | 43.6 | 10.7 | 3.1 | |
| Endometrial serous carcinoma | 82 | 73 | 79.5 | 17.8 | 0.0 | 2.7 | |
| Carcinosarcoma of the uterus | 48 | 46 | 76.1 | 21.7 | 0.0 | 2.2 | |
| Endometrial carcinoma, high grade, G3 | 13 | 13 | 92.3 | 7.7 | 0.0 | 0.0 | |
| Endometrial clear cell carcinoma | 8 | 8 | 75.0 | 25.0 | 0.0 | 0.0 | |
| Endometrioid carcinoma of the ovary | 110 | 93 | 57.0 | 25.8 | 6.5 | 10.8 | |
| Serous carcinoma of the ovary, high grade | 559 | 479 | 86.6 | 13.2 | 0.2 | 0.0 | |
| Mucinous carcinoma of the ovary | 96 | 71 | 91.5 | 5.6 | 1.4 | 1.4 | |
| Clear cell carcinoma of the ovary | 50 | 42 | 88.1 | 11.9 | 0.0 | 0.0 | |
| Carcinosarcoma of the ovary | 47 | 42 | 92.9 | 4.8 | 2.4 | 0.0 | |
| Brenner tumor | 9 | 9 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Tumors of the breast | Invasive breast carcinoma of no special type | 1391 | 1214 | 45.9 | 20.6 | 12.4 | 21.2 |
| Lobular carcinoma of the breast | 294 | 260 | 30.4 | 21.2 | 8.8 | 39.6 | |
| Medullary carcinoma of the breast | 26 | 24 | 70.8 | 12.5 | 8.3 | 8.3 | |
| Tubular carcinoma of the breast | 27 | 20 | 15.0 | 35.0 | 15.0 | 35.0 | |
| Mucinous carcinoma of the breast | 58 | 43 | 65.1 | 9.3 | 7.0 | 18.6 | |
| Phyllodes tumor of the breast | 50 | 44 | 40.9 | 34.1 | 13.6 | 11.4 | |
| Tumors of the digestive system | Adenomatous polyp, low-grade dysplasia | 50 | 50 | 100.0 | 0.0 | 0.0 | 0.0 |
| Adenomatous polyp, high-grade dysplasia | 50 | 49 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Adenocarcinoma of the colon | 1932 | 1808 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Gastric adenocarcinoma, diffuse type | 226 | 161 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Gastric adenocarcinoma, intestinal type | 224 | 167 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Gastric adenocarcinoma, mixed type | 62 | 56 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Adenocarcinoma of the esophagus | 133 | 82 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Squamous cell carcinoma of the esophagus | 124 | 71 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Squamous cell carcinoma of the anal canal | 91 | 85 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Cholangiocarcinoma | 50 | 49 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Hepatocellular carcinoma | 50 | 50 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Ductal adenocarcinoma of the pancreas | 662 | 593 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Pancreatic/ampullary adenocarcinoma | 119 | 86 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Acinar cell carcinoma of the pancreas | 14 | 13 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Gastrointestinal stromal tumor (GIST) | 50 | 49 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Tumors of the urinary system | Non-invasive papillary urothelial carcinoma, pTa G2 low grade | 177 | 141 | 100.0 | 0.0 | 0.0 | 0.0 |
| Non-invasive papillary urothelial carcinoma, pTa G2 high grade | 141 | 117 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Non-invasive papillary urothelial carcinoma, pTa G3 | 187 | 113 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Urothelial carcinoma, pT2-4 G3 | 1207 | 825 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Small cell neuroendocrine carcinoma of the bladder | 18 | 18 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Sarcomatoid urothelial carcinoma | 25 | 21 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Clear cell renal cell carcinoma | 858 | 824 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Papillary renal cell carcinoma | 255 | 232 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Clear cell (tubulo) papillary renal cell carcinoma | 21 | 20 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Chromophobe renal cell carcinoma | 131 | 122 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Oncocytoma | 177 | 162 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Tumors of the male genital organs | Adenocarcinoma of the prostate, Gleason 3 + 3 | 83 | 83 | 100.0 | 0.0 | 0.0 | 0.0 |
| Adenocarcinoma of the prostate, Gleason 4 + 4 | 80 | 80 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Adenocarcinoma of the prostate, Gleason 5 + 5 | 85 | 85 | 98.8 | 0.0 | 1.2 | 0.0 | |
| Adenocarcinoma of the prostate (recurrence) | 261 | 254 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Small cell neuroendocrine carcinoma of the prostate | 17 | 15 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Seminoma | 621 | 611 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Embryonal carcinoma of the testis | 50 | 44 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Yolk sac tumor | 50 | 43 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Teratoma | 50 | 38 | 97.4 | 0.0 | 0.0 | 2.6 | |
| Squamous cell carcinoma of the penis | 80 | 79 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Tumors of endocrine organs | Adenoma of the thyroid gland | 50 | 47 | 100.0 | 0.0 | 0.0 | 0.0 |
| Papillary thyroid carcinoma | 50 | 48 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Follicular thyroid carcinoma | 49 | 49 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Medullary thyroid carcinoma | 50 | 46 | 82.6 | 13.0 | 4.3 | 0.0 | |
| Anaplastic thyroid carcinoma | 26 | 24 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Adrenal cortical adenoma | 50 | 44 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Adrenal cortical carcinoma | 26 | 25 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Phaeochromocytoma | 50 | 48 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Pancreas, neuroendocrine tumor (NET) | 46 | 42 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Pancreas, neuroendocrine carcinoma (NEC) | 3 | 3 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Tumors of hematopoietic and lymphoid tissues | Hodgkin lymphoma | 103 | 100 | 100.0 | 0.0 | 0.0 | 0.0 |
| Non-Hodgkin lymphoma | 62 | 61 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Small lymphocytic lymphoma, B-cell type (B-SLL/B-CLL) | 50 | 50 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Diffuse large B-cell lymphoma (DLBCL) | 114 | 114 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Follicular lymphoma | 88 | 88 | 100.0 | 0.0 | 0.0 | 0.0 | |
| T-cell non-Hodgkin lymphoma | 24 | 24 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Mantle cell lymphoma | 18 | 18 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Marginal zone lymphoma | 16 | 16 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Diffuse large B-cell lymphoma (DLBCL) in the testis | 16 | 16 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Burkitt lymphoma | 5 | 3 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Tumors of soft tissue and bone | Tendosynovial giant cell tumor | 45 | 43 | 100.0 | 0.0 | 0.0 | 0.0 |
| Granular cell tumor | 53 | 43 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Leiomyoma | 50 | 47 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Leiomyosarcoma | 87 | 87 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Liposarcoma | 132 | 121 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Malignant peripheral nerve sheath tumor (MPNST) | 13 | 11 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Myofibrosarcoma | 26 | 26 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Angiosarcoma | 73 | 67 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Angiomyolipoma | 91 | 88 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Dermatofibrosarcoma protuberans | 21 | 18 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Ganglioneuroma | 14 | 14 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Kaposi sarcoma | 8 | 5 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Neurofibroma | 117 | 116 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Sarcoma, not otherwise specified (NOS) | 75 | 71 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Paraganglioma | 41 | 41 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Primitive neuroectodermal tumor (PNET) | 23 | 16 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Rhabdomyosarcoma | 7 | 7 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Schwannoma | 121 | 118 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Synovial sarcoma | 12 | 11 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Osteosarcoma | 43 | 36 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Chondrosarcoma | 38 | 17 | 100.0 | 0.0 | 0.0 | 0.0 | |
| Mammaglobin-A IHC | ||||||||
|---|---|---|---|---|---|---|---|---|
| n | Negative (%) | Weak (%) | Moderate (%) | Strong (%) | p | |||
| Breast cancer of no special type (NST) | Tumor stage | pT1 | 601 | 44.3 | 21.5 | 14.1 | 20.1 | 0.1421 |
| pT2 | 411 | 46.5 | 18.5 | 11.7 | 23.4 | |||
| pT3–4 | 84 | 54.8 | 16.7 | 6.0 | 22.6 | |||
| Grade | G1 | 176 | 37.5 | 21.6 | 13.6 | 27.3 | 0.0011 | |
| G2 | 590 | 41.9 | 21.7 | 13.7 | 22.7 | |||
| G3 | 372 | 54.3 | 19.4 | 10.8 | 15.6 | |||
| Nodal stage | pN0 | 515 | 44.1 | 21.6 | 14.6 | 19.8 | 0.3017 | |
| pN1 | 223 | 44.8 | 19.7 | 13.0 | 22.4 | |||
| pN2 | 69 | 53.6 | 15.9 | 5.8 | 24.6 | |||
| pN3 | 56 | 50.0 | 14.3 | 8.9 | 26.8 | |||
| HER2 status | neg | 851 | 47.1 | 20.8 | 11.2 | 20.9 | 0.4501 | |
| pos | 118 | 43.2 | 22.0 | 16.1 | 18.6 | |||
| ER status | neg | 200 | 63.5 | 14.5 | 9.5 | 12.5 | <0.0001 | |
| pos | 717 | 41.6 | 23.0 | 12.8 | 22.6 | |||
| PR status | neg | 390 | 55.4 | 17.7 | 10.5 | 16.4 | <0.0001 | |
| pos | 568 | 40.0 | 23.2 | 13.2 | 23.6 | |||
| Triple negative | no | 756 | 41.7 | 22.6 | 13.4 | 22.4 | <0.0001 | |
| yes | 134 | 75.4 | 10.4 | 5.2 | 9.0 | |||
| Endometrioid endometrial cancers | Tumor stage | pT1 | 113 | 33.6 | 48.7 | 14.2 | 3.5 | 0.0198 |
| pT2 | 24 | 50.0 | 37.5 | 12.5 | 0.0 | |||
| pT3–4 | 35 | 48.6 | 51.4 | 0.0 | 0.0 | |||
| pN0 | 50 | 36.0 | 56.0 | 6.0 | 2.0 | 0.0986 | ||
| Nodal stage | pN+ | 30 | 63.3 | 33.3 | 3.3 | 0.0 | ||
| Endometrioid ovarian cancer | Tumor stage | pT1 | 22 | 40.9 | 31.8 | 9.1 | 18.2 | 0.1686 |
| pT2 | 4 | 100.0 | 0.0 | 0.0 | 0.0 | |||
| pT3 | 5 | 80.0 | 20.0 | 0.0 | 0.0 | |||
| Nodal stage | pN0 | 20 | 40.0 | 35.0 | 10.0 | 15.0 | 0.3835 | |
| pN1 | 7 | 71.4 | 14.3 | 0.0 | 14.3 | |||
| Serous ovarian cancers | Tumor stage | pT1 | 31 | 77.4 | 22.6 | 0.0 | 0.0 | 0.2 |
| pT2 | 40 | 92.5 | 7.5 | 0.0 | 0.0 | |||
| pT3 | 245 | 88.2 | 11.8 | 0.0 | 0.0 | |||
| Nodal stage | pN0 | 78 | 83.3 | 16.7 | 0.0 | 0.0 | 0.3767 | |
| pN1 | 153 | 87.6 | 12.4 | 0.0 | 0.0 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gorbokon, N.; Timm, P.; Dum, D.; Menz, A.; Büscheck, F.; Völkel, C.; Hinsch, A.; Lennartz, M.; Luebke, A.M.; Hube-Magg, C.; et al. Mammaglobin-A Expression Is Highly Specific for Tumors Derived from the Breast, the Female Genital Tract, and the Salivary Gland. Diagnostics 2023, 13, 1202. https://doi.org/10.3390/diagnostics13061202
Gorbokon N, Timm P, Dum D, Menz A, Büscheck F, Völkel C, Hinsch A, Lennartz M, Luebke AM, Hube-Magg C, et al. Mammaglobin-A Expression Is Highly Specific for Tumors Derived from the Breast, the Female Genital Tract, and the Salivary Gland. Diagnostics. 2023; 13(6):1202. https://doi.org/10.3390/diagnostics13061202
Chicago/Turabian StyleGorbokon, Natalia, Patrick Timm, David Dum, Anne Menz, Franziska Büscheck, Cosima Völkel, Andrea Hinsch, Maximilian Lennartz, Andreas M Luebke, Claudia Hube-Magg, and et al. 2023. "Mammaglobin-A Expression Is Highly Specific for Tumors Derived from the Breast, the Female Genital Tract, and the Salivary Gland" Diagnostics 13, no. 6: 1202. https://doi.org/10.3390/diagnostics13061202
APA StyleGorbokon, N., Timm, P., Dum, D., Menz, A., Büscheck, F., Völkel, C., Hinsch, A., Lennartz, M., Luebke, A. M., Hube-Magg, C., Fraune, C., Krech, T., Lebok, P., Clauditz, T. S., Jacobsen, F., Sauter, G., Uhlig, R., Steurer, S., Minner, S., ... Höflmayer, D. (2023). Mammaglobin-A Expression Is Highly Specific for Tumors Derived from the Breast, the Female Genital Tract, and the Salivary Gland. Diagnostics, 13(6), 1202. https://doi.org/10.3390/diagnostics13061202






