Epileptic Seizure Detection Using Machine Learning: Taxonomy, Opportunities, and Challenges
Abstract
1. Introduction
2. Background
3. Research Methodology
3.1. Research Objectives (ROs)
3.2. Research Questions (RQs)
3.3. Search Scheme
3.3.1. Search String
3.3.2. Literature Resources
3.3.3. Inclusion and Exclusion Criteria
- Parameters defined for inclusion criteria (IC) are:
- IC (1)
- Include studies that were primarily conducted for epileptic seizure prediction using machine learning techniques.
- IC (2)
- Feature extraction methods targeting wavelet transform methods for the decomposition of EGG signals.
- IC (3)
- Studies that encompass machine learning classifiers that included RF, SVM, ANN, and KNN.
- The exclusion criteria applied to all the articles to exclude out-of-scope articles such as:
- EC (1)
- If the study did not involve any feature extraction techniques that involve wavelet transform techniques.
- EC (2)
- The studies did not involve the classifiers such as RF, SVM, ANN, and KNN.
3.4. Selection of Relevant Papers
3.5. Abstract-Based Keywording
4. Data Analysis
4.1. Search Results
4.2. Assessment of Research Questions
4.2.1. Assessment of Question 1: What Machine Learning Classifiers Are Used in the Majority of the Research for the Diagnosis of Epileptic Seizure?
Random Forest (RF)
Support Vector Machine (SVM)
K-Nearest Neighbor (KNN)
Artificial Neural Network (ANN)
4.2.2. Assessment of Question 2: What Kind of Features Extracting Methods Are Being Used, and What Features Are Being Extracted from the Eeg Signal?
4.2.3. Assessment of Question 3: What Are the Gaps and Challenges in the Detection of Epileptic Seizures?
4.2.4. Assessment of Question 4: What Datasets Have Been Used in the Majority of the Research for Epileptic Seizure Detection?
5. Discussions
5.1. Hierarchal Representation of Epileptic Seizure Detection Techniques
5.2. Challenges and Gaps
- Researchers have the opportunity to predict epileptic seizure with feature extraction techniques by studying the non-linear features thoroughly and understanding their results on different classifiers. Furthermore, a variety of datasets is used, but due to different parameters for feature extraction methods, it becomes difficult to gain insight into a larger dataset with a combination of feature extraction techniques.
- Furthermore, this review research has been conducted to understand the basis of how epileptic seizure detection methods are being used in the domain of machine learning. Hence, for future work, researchers can work specifically on petit mal, known as absence seizure. Their detection is somewhat challenging because of its minimal duration and negligible visual symptoms. Whereas the occurrence of these seizures is more frequent and can psychologically affect a child’s life since it only occurs in kids from age 4 to 14.
- To the best of our knowledge, there has not been much literature regarding the ML techniques being applied solely for the detection of absence seizures. The major opportunity could be in the generation of a device that is user-friendly for a home environment and not as terrifying as an electroencephalogram for children.
- Normally, children with absence seizures show mild signs or symptoms of absence or abnormal behavior, such as staring blankly at the wall or not understanding a word someone says. A device should be made after testing the algorithms for the detection process with less chance of false positives so that the absence seizures can be detected and monitored at home by parents or guardians.
6. Conclusions
Funding
Data Availability Statement
Conflicts of Interest
References
- Amin, H.U.; Yusoff, M.Z.; Ahmad, R.F. A novel approach based on wavelet analysis and arithmetic coding for automated detection and diagnosis of epileptic seizure in EEG signals using machine learning techniques. Biomed. Signal Process. Control 2020, 56, 101707. [Google Scholar] [CrossRef]
- Sharmila, A.; Geethanjali, P. DWT based detection of epileptic seizure from EEG signals using naive Bayes and k-NN classifiers. IEEE Access 2016, 4, 7716–7727. [Google Scholar] [CrossRef]
- Sharmila, A.; Geethanjali, P. A review on the pattern detection methods for epilepsy seizure detection from EEG signals. Biomed. Eng. Biomed. Tech. 2019, 64, 507–517. [Google Scholar] [CrossRef] [PubMed]
- Harpale, V.; Bairagi, V. An adaptive method for feature selection and extraction for classification of epileptic EEG signal in significant states. J. King Saud Univ.-Comput. Inf. Sci. 2021, 33, 668–676. [Google Scholar] [CrossRef]
- Thara, D.K.; PremaSudha, B.G.; Xiong, F. Auto-detection of epileptic seizure events using deep neural network with different feature scaling techniques. Pattern Recognit. Lett. 2019, 128, 544–550. [Google Scholar]
- Boonyakitanont, P.; Lek-Uthai, A.; Chomtho, K.; Songsiri, J. A review of feature extraction and performance evaluation in epileptic seizure detection using EEG. Biomed. Signal Process. Control 2020, 57, 101702. [Google Scholar] [CrossRef]
- Acharya, U.R.; Hagiwara, Y.; Adeli, H. Automated seizure prediction. Epilepsy Behav. 2018, 88, 251–261. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Chen, W.; Zhang, T. Automatic epilepsy detection using wavelet-based nonlinear analysis and optimized SVM. Biocybern. Biomed. Eng. 2016, 36, 708–718. [Google Scholar] [CrossRef]
- Pattnaik, S.; Rout, N.; Sabut, S. Machine learning approach for epileptic seizure detection using the tunable-Q wavelet transform based time–frequency features. Int. J. Inf. Technol. 2022, 14, 3495–3505. [Google Scholar] [CrossRef]
- Orhan, U.; Hekim, M.; Ozer, M. EEG signals classification using the K-means clustering and a multilayer perceptron neural network model. Expert Syst. Appl. 2011, 38, 13475–13481. [Google Scholar] [CrossRef]
- Naderi, M.A.; Mahdavi-Nasab, H. Analysis and classification of EEG signals using spectral analysis and recurrent neural networks. In Proceedings of the 2010 17th Iranian Conference of Biomedical Engineering (ICBME), Isfahan, Iran, 3–4 November 2010; pp. 3–4. [Google Scholar]
- Subasi, A.; Gursoy, M.I. EEG signal classification using PCA, ICA, LDA and support vector machines. Expert Syst. Appl. 2010, 37, 8659–8666. [Google Scholar] [CrossRef]
- Kumar, Y.; Dewal, M.; Anand, R. Epileptic seizure detection using DWT based fuzzy approximate entropy and support vector machine. Neurocomputing 2014, 133, 271–279. [Google Scholar] [CrossRef]
- Nicolaou, N.; Georgiou, J. Detection of epileptic electroencephalogram based on Permutation Entropy and Support Vector Machines. Expert Syst. Appl. 2012, 39, 202–209. [Google Scholar] [CrossRef]
- Potapov, P. On the loss of information in PCA of spectrum-images. Ultramicroscopy 2017, 182, 191–194. [Google Scholar] [CrossRef] [PubMed]
- Harikumar, R.; Narayanan, B.S. Fuzzy techniques for classification of epilepsy risk level from EEG signals. In Proceedings of the TENCON 2003. Conference on Convergent Technologies for Asia-Pacific Region, Bangalore, India, 15–17 October 2003; Volume 1, pp. 209–213. [Google Scholar]
- Hassan, A.R.; Siuly, S.; Zhang, Y. Epileptic seizure detection in EEG signals using tunable-Q factor wavelet transform and bootstrap aggregating. Comput. Methods Programs Biomed. 2016, 137, 247–259. [Google Scholar] [CrossRef]
- Mursalin, M.; Zhang, Y.; Chen, Y.; Chawla, N.V. Automated epileptic seizure detection using improved correlation-based feature selection with random forest classifier. Neurocomputing 2017, 241, 204–214. [Google Scholar] [CrossRef]
- Hussain, W.; Qbal, M.S.I.; Xiang, J.; Wang, B.; Niu, Y.; Gao, Y.; Wang, X.; Sun, J.; Zhan, Q.; Cao, R.; et al. Epileptic Seizure Detection With Permutation Fuzzy Entropy Using Robust Machine Learning Techniques. IEEE Access 2019, 7, 182238–182258. [Google Scholar] [CrossRef]
- Zhou, P.-Y.; Chan, K.C.C. Fuzzy Feature Extraction for Multichannel EEG Classification. IEEE Trans. Cogn. Dev. Syst. 2016, 10, 267–279. [Google Scholar] [CrossRef]
- Tzimourta, K.D.; Tzallas, A.T.; Giannakeas, N.; Astrakas, L.G.; Tsalikakis, D.G.; Angelidis, P.; Tsipouras, M.G. A robust methodology for classification of epileptic seizures in EEG signals. Health Technol. 2018, 9, 135–142. [Google Scholar] [CrossRef]
- Alickovic, E.; Kevric, J.; Subasi, A. Performance evaluation of empirical mode decomposition, discrete wavelet transform, and wavelet packed decomposition for automated epileptic seizure detection and prediction. Biomed. Signal Process. Control 2018, 39, 94–102. [Google Scholar] [CrossRef]
- Subasi, A.; Kevric, J.; Abdullah Canbaz, M. Epileptic seizure detection using hybrid machine learning methods. Neural Comput. Appl. 2019, 31, 317–325. [Google Scholar] [CrossRef]
- Raghu, S.; Sriraam, N.; Vasudeva Rao, S.; Hegde, A.S.; Kubben, P.L. Automated detection of epileptic seizures using successive decomposition index and support vector machine classifier in long-term EEG. Neural Comput. Appl. 2020, 32, 8965–8984. [Google Scholar] [CrossRef]
- Omidvar, M.; Zahedi, A.; Bakhshi, H. EEG signal processing for epilepsy seizure detection using 5-level Db4 discrete wavelet transform, GA-based feature selection and ANN/SVM classifiers. J. Ambient. Intell. Humaniz. Comput. 2021, 12, 10395–10403. [Google Scholar] [CrossRef]
- Hussain, L. Detecting epileptic seizure with different feature extracting strategies using robust machine learning classification techniques by applying advance parameter optimization approach. Cogn. Neurodynamics 2018, 12, 271–294. [Google Scholar] [CrossRef] [PubMed]
- Savadkoohi, M.; Oladunni, T.; Thompson, L. A machine learning approach to epileptic seizure prediction using Electroencephalogram (EEG) Signal. Biocybern. Biomed. Eng. 2020, 40, 1328–1341. [Google Scholar] [CrossRef] [PubMed]
- Mardini, W.; Yassein, M.M.B.; Al-Rawashdeh, R.; Aljawarneh, S.; Khamayseh, Y.; Meqdadi, O. Enhanced Detection of Epileptic Seizure Using EEG Signals in Combination With Machine Learning Classifiers. IEEE Access 2020, 8, 24046–24055. [Google Scholar] [CrossRef]
- Sarić, R.; Jokić, D.; Beganović, N.; Pokvić, L.G.; Badnjević, A. FPGA-based real-time epileptic seizure classification using Artificial Neural Network. Biomed. Signal Process. Control 2020, 62, 102106. [Google Scholar] [CrossRef]
- Usman, S.M.; Khalid, S.; Aslam, M.H. Epileptic Seizures Prediction Using Deep Learning Techniques. IEEE Access 2020, 8, 39998–40007. [Google Scholar] [CrossRef]
- Zhang, T.; Chen, W.; Li, M. Generalized Stockwell transform and SVD-based epileptic seizure detection in EEG using random forest. Biocybern. Biomed. Eng. 2018, 38, 519–534. [Google Scholar] [CrossRef]
- Ramos-Aguilar, R.; Olvera-López, J.A.; Olmos-Pineda, I.; Sánchez-Urrieta, S. Feature extraction from EEG spectrograms for epileptic seizure detection. Pattern Recognit. Lett. 2020, 133, 202–209. [Google Scholar] [CrossRef]
- Karabiber Cura, O.; Kocaaslan Atli, S.; Türe, H.S.; Akan, A. Epileptic seizure classifications using empirical mode decomposition and its derivative. Biomed. Eng. Online 2020, 19, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Cao, J.; Lai, X.; Hu, D. Epileptic state classification for seizure prediction with wavelet packet features and random forest. In Proceedings of the 2019 Chinese Control And Decision Conference (CCDC), Nanchang, China, 3–5 June 2019; pp. 3983–3987. [Google Scholar]
- Rabby, M.K.M.; Islam, A.K.; Belkasim, S.; Bikdash, M.U. Wavelet transform-based feature extraction approach for epileptic seizure classification. In Proceedings of the 2021 ACM Southeast Conference, Oxford, AL, USA, 15–17 April 2021; pp. 164–169. [Google Scholar]
- Hu, Z.; Han, C.; Guo, F.; Qin, Q.; Li, S.; Qin, Y. Epileptic Seizure Prediction from the Scalp EEG Signals by using Random Forest Algorithm. In Proceedings of the 2020 13th International Congress on Image and Signal Processing, BioMedical Engineering and Informatics (CISP-BMEI), Chengdu, China, 17–19 October 2020; pp. 669–674. [Google Scholar]
- Jacobs, D.; Hilton, T.; del Campo, M.; Carlen, P.L.; Bardakjian, B.L. Classification of Pre-Clinical Seizure States Using Scalp EEG Cross-Frequency Coupling Features. IEEE Trans. Biomed. Eng. 2018, 65, 2440–2449. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Xun, G.; Jia, K.; Zhang, A. A multi-view deep learning method for epileptic seizure detection using short-time fourier transform. In Proceedings of the 8th ACM International Conference on Bioinformatics, Computational Biology, and Health Informatics, Boston, MA, USA, 20–23 August 2017; pp. 213–222. [Google Scholar]
- Rabcan, J.; Levashenko, V.; Zaitseva, E.; Kvassay, M. EEG Signal Classification Based On Fuzzy Classifiers. IEEE Trans. Ind. Inform. 2021, 18, 757–766. [Google Scholar] [CrossRef]
- Hosseini, M.-P.; Pompili, D.; Elisevich, K.; Soltanian-Zadeh, H. Random ensemble learning for EEG classification. Artif. Intell. Med. 2018, 84, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Gong, G.; Li, N.; Qiu, S. Detection Analysis of Epileptic EEG Using a Novel Random Forest Model Combined With Grid Search Optimization. Front. Hum. Neurosci. 2019, 13, 52. [Google Scholar] [CrossRef]
- Sharma, M.; Sharma, P.; Pachori, R.B.; Acharya, U.R. Dual-Tree Complex Wavelet Transform-Based Features for Automated Alcoholism Identification. Int. J. Fuzzy Syst. 2018, 20, 1297–1308. [Google Scholar] [CrossRef]
- Lee, H.; Kim, S. Black-Box Classifier Interpretation Using Decision Tree and Fuzzy Logic-Based Classifier Implementation. Int. J. Fuzzy Log. Intell. Syst. 2016, 16, 27–35. [Google Scholar] [CrossRef]
- Donos, C.; Dümpelmann, M.; Schulze-Bonhage, A. Early Seizure Detection Algorithm Based on Intracranial EEG and Random Forest Classification. Int. J. Neural Syst. 2015, 25, 1550023. [Google Scholar] [CrossRef]
- Siddiqui, M.K.; Islam, Z.; Kabir, M.A. A novel quick seizure detection and localization through brain data mining on ECoG dataset. Neural Comput. Appl. 2018, 31, 5595–5608. [Google Scholar] [CrossRef]
- Selvakumari, R.S.; Mahalakshmi, M.; Prashalee, P. Patient-Specific Seizure Detection Method using Hybrid Classifier with Optimized Electrodes. J. Med Syst. 2019, 43, 121. [Google Scholar] [CrossRef]
- Fasil, O.K.; Rajesh, R. Time-domain exponential energy for epileptic EEG signal classification. Neurosci. Lett. 2019, 694, 1–8. [Google Scholar]
- Hassan, A.R.; Subasi, A. Automatic identification of epileptic seizures from EEG signals using linear programming boosting. Comput. Methods Programs Biomed. 2016, 136, 65–77. [Google Scholar] [CrossRef]
- Zabihi, M.; Kiranyaz, S.; Ince, T.; Gabbouj, M. Patient-specific epileptic seizure detection in long-term EEG recording in paediatric patients with intractable seizures. In Proceedings of the IET Intelligent Signal Processing Conference 2013 (ISP 2013), London, UK, 2–3 December 2013. [Google Scholar]
- Shoeb, A.H.; Guttag, J.V. Application of machine learning to epileptic seizure detection. In Proceedings of the 27th International Conference on International Conference on Machine Learning, Haifa, Israel, 21–24 June 2010. [Google Scholar]
- Ahmad, M.A.; Khan, N.A.; Majeed, W. Computer assisted analysis system of electroencephalogram for diagnosing epilepsy. In Proceedings of the 2014 22nd International Conference on Pattern Recognition (ICPR), Stockholm, Sweden, 24–28 August 2014; pp. 3386–3391. [Google Scholar]
- Chen, D.; Wan, S.; Xiang, J.; Bao, F.S. A high-performance seizure detection algorithm based on Discrete Wavelet Transform (DWT) and EEG. PLoS ONE 2017, 12, e0173138. [Google Scholar] [CrossRef]
- Lahmiri, S.; Shmuel, A. Accurate Classification of Seizure and Seizure-Free Intervals of Intracranial EEG Signals From Epileptic Patients. IEEE Trans. Instrum. Meas. 2018, 68, 791–796. [Google Scholar] [CrossRef]
- Amin, H.U.; Malik, A.S.; Ahmad, R.F.; Badruddin, N.; Kamel, N.; Hussain, M.; Chooi, W.-T. Feature extraction and classification for EEG signals using wavelet transform and machine learning techniques. Australas. Phys. Eng. Sci. Med. 2015, 38, 139–149. [Google Scholar] [CrossRef]
- Raghu, S.; Sriraam, N.; Temel, Y.; Rao, S.V.; Kubben, P.L. EEG based multi-class seizure type classification using convolutional neural network and transfer learning. Neural Netw. 2020, 124, 202–212. [Google Scholar] [CrossRef] [PubMed]
- Mursalin, M.; Islam, S.S.; Noman, M.K.; Al-Jumaily, A.A. Epileptic seizure classification using statistical sampling and a novel feature selection algorithm. arXiv 2019, arXiv:1902.09962. [Google Scholar]
- Guo, S.; DiPietro, L.A. Factors Affecting Wound Healing. J. Dent. Res. 2010, 89, 219–229. [Google Scholar] [CrossRef]
- Zeiler, M.D.; Fergus, R. Visualizing and understanding convolutional networks. In Computer Vision—ECCV 2014, Proceedings of the European Conference on Computer Vision, Zurich, Switzerland, 6–12 September 2014; Springer: Cham, Switzerland, 2014; pp. 818–833. [Google Scholar]
- Bhattacharyya, A.; Sharma, M.; Pachori, R.B.; Sircar, P.; Acharya, U.R. A novel approach for automated detection of focal EEG signals using empirical wavelet transform. Neural Comput. Appl. 2016, 29, 47–57. [Google Scholar] [CrossRef]
- Moeller, F.; LeVan, P.; Muhle, H.; Stephani, U.; Dubeau, F.; Siniatchkin, M.; Gotman, J. Absence seizures: Individual patterns revealed by EEG-fMRI. Epilepsia 2010, 51, 2000–2010. [Google Scholar] [CrossRef]
- Tzallas, A.T.; Tsipouras, M.G.; Fotiadis, D.I. Epileptic Seizure Detection in EEGs Using Time–Frequency Analysis. IEEE Trans. Inf. Technol. Biomed. 2009, 13, 703–710. [Google Scholar] [CrossRef] [PubMed]
- Harender, B.; Sharma, R.K. DWT based epileptic seizure detection from EEG signal using k-NN classifier. In Proceedings of the 2017 International Conference on Trends in Electronics and Informatics (ICEI), Tirunelveli, India, 11–12 May 2017; pp. 762–765. [Google Scholar]
- Kumar, M.; Pachori, R.B.; Acharya, U.R. Use of Accumulated Entropies for Automated Detection of Congestive Heart Failure in Flexible Analytic Wavelet Transform Framework Based on Short-Term HRV Signals. Entropy 2017, 19, 92. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, M.; Wang, D.; Jiang, Y. Kernel PCA feature extraction and the SVM classification algorithm for multiple-status, through-wall, human being detection. EURASIP J. Wirel. Commun. Netw. 2017, 151, 151–157. [Google Scholar] [CrossRef]


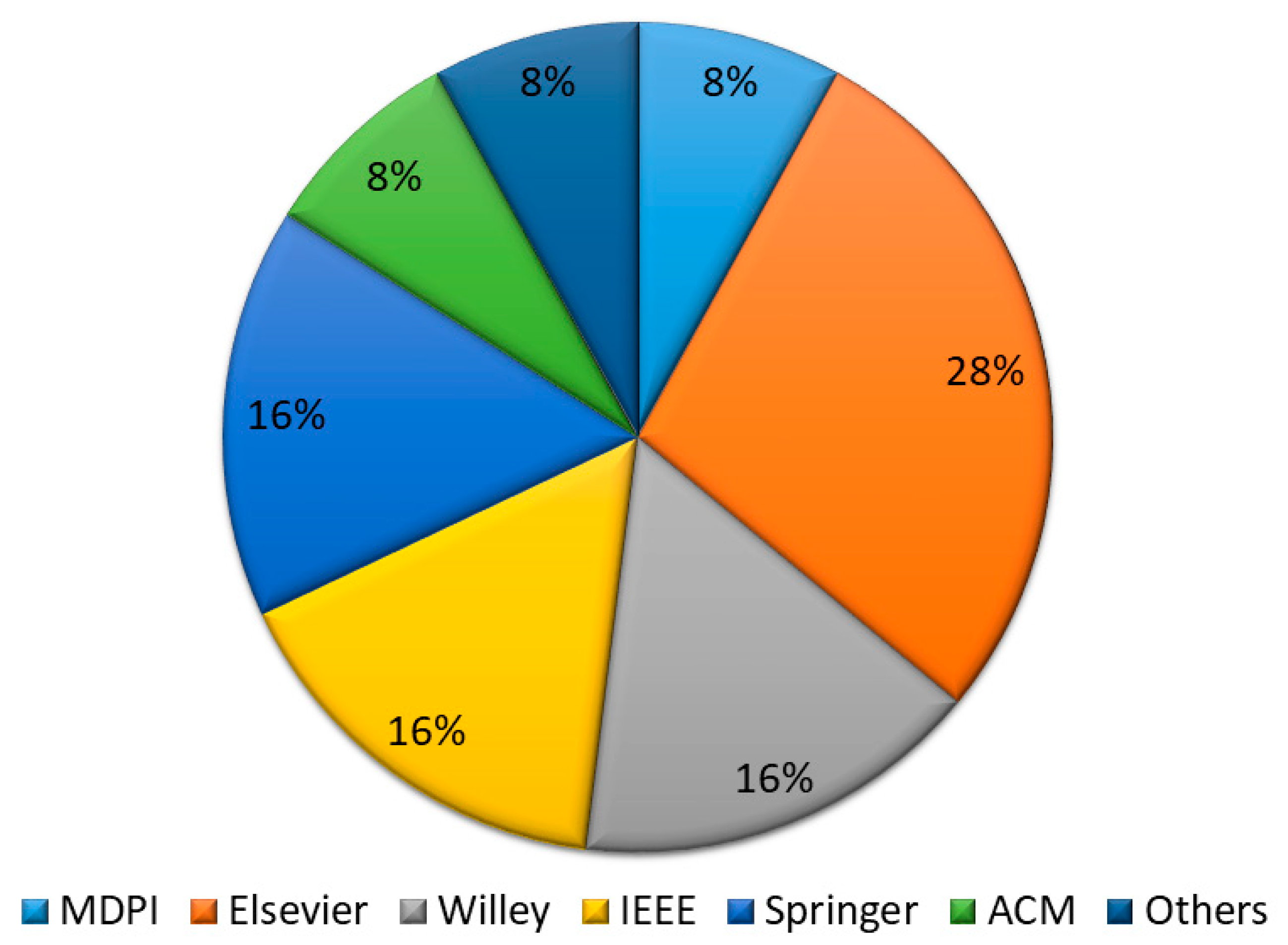
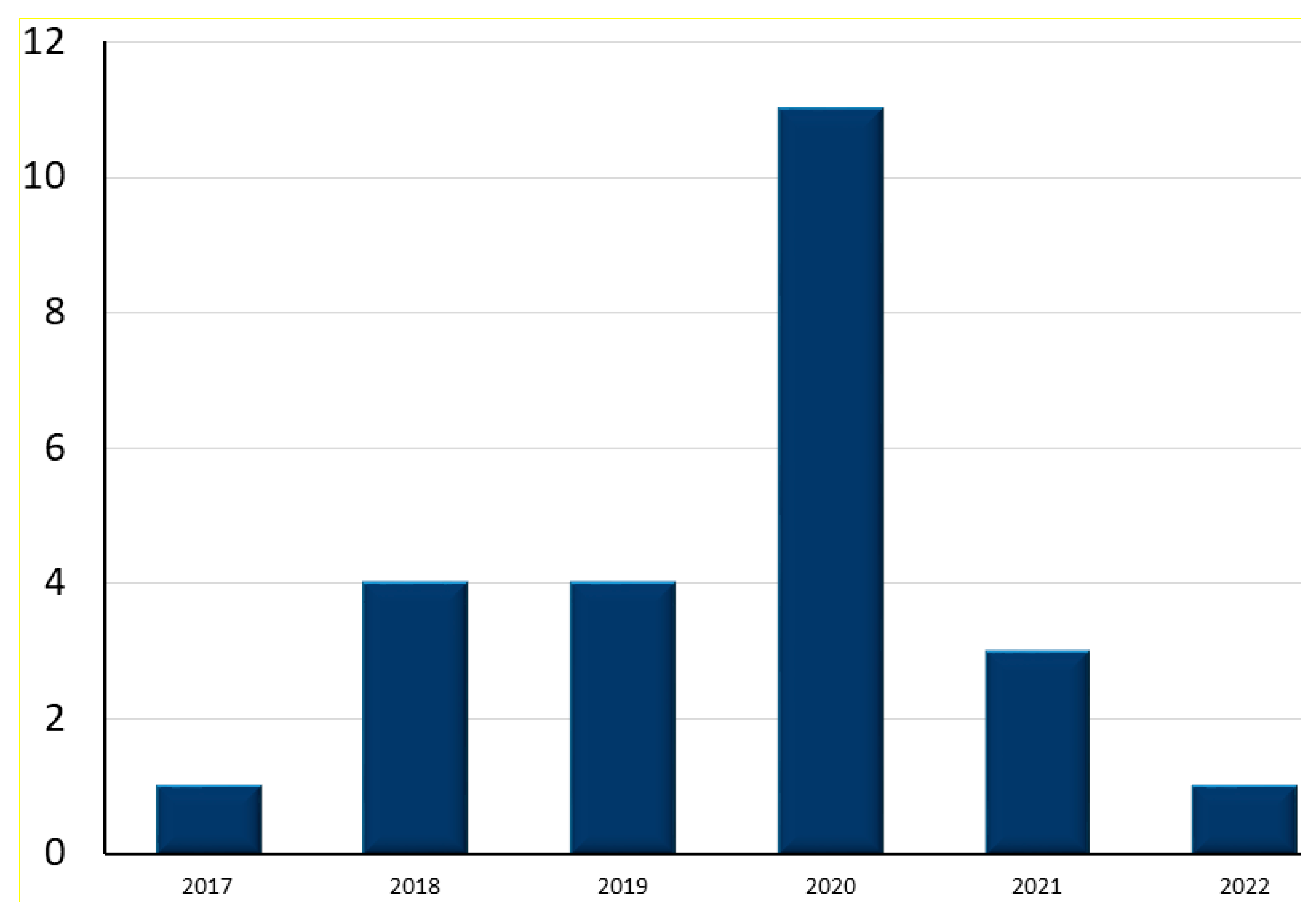
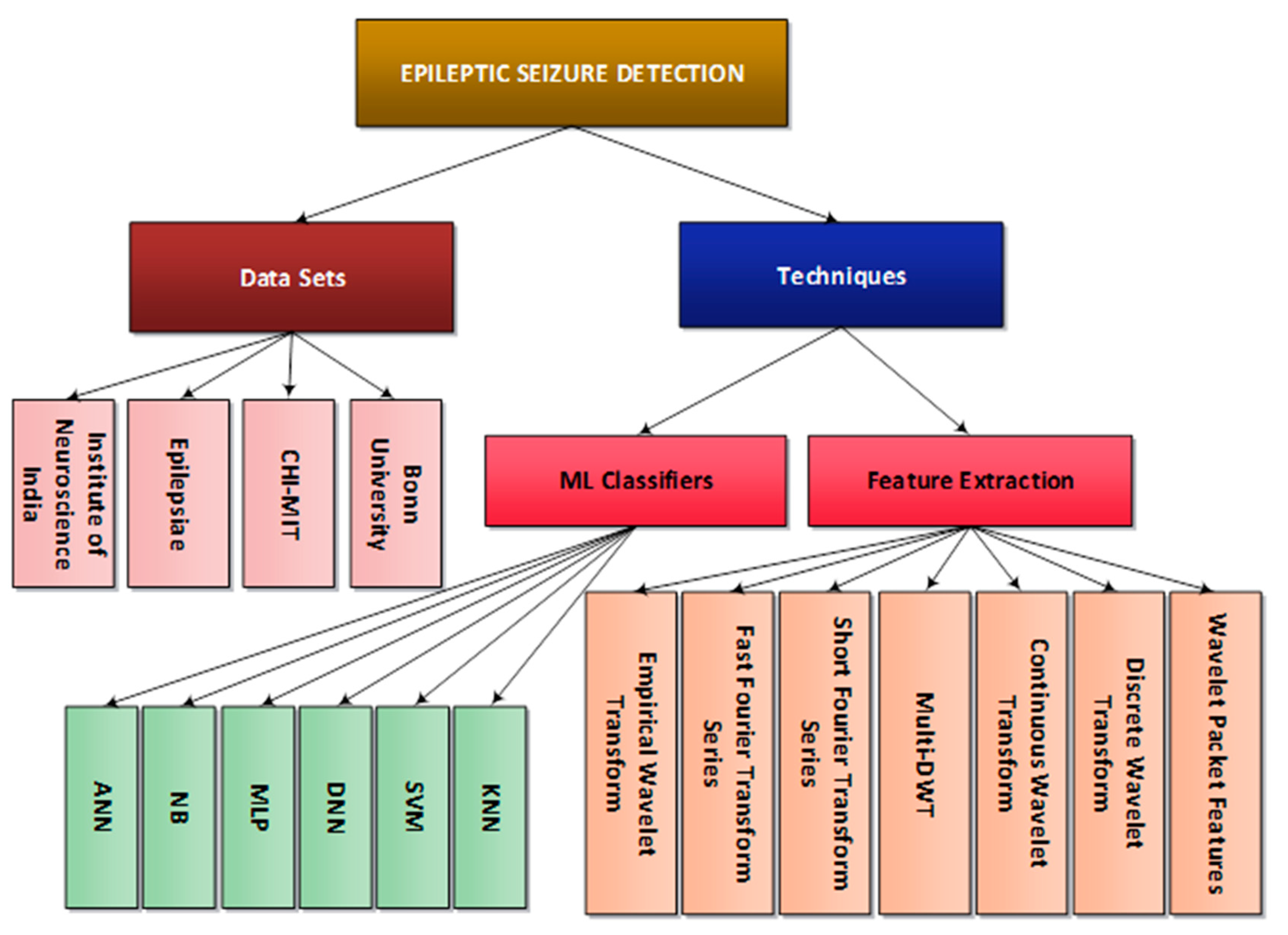
| Research Questions | Motivation | |
|---|---|---|
| RQ1 | What machine learning classifiers are used in the majority of the research for the diagnostics of epileptic seizure? | For the understanding of the model requirements for the prediction of epileptic seizure |
| RQ2 | What kind of feature-extracting methods are being used, and what kind of features are being extracted from the EEG signal? | To understand the performance of the classification process based on the features extracted |
| RQ3 | What are the gaps and challenges in the detection of epileptic seizures? | This question aims to identify strengths and limitations in pattern recognition techniques and the performance of classifiers based on feature extraction on various datasets. |
| RQ4 | What datasets are used for epileptic seizure detection? | To check the biases of the dataset |
| Terms (Keywords) | Synonyms/Alternate Keywords |
|---|---|
| +Machine Learning | Classification Techniques, ML classifiers, Classification |
| +Epileptic Seizure | Seizure detection, Epilepsy detection, Convulsions, Epileptic seizure detection |
| +Feature Extraction | - |
| Data Repository | Relevant Search Strings |
|---|---|
| Science Direct | ((Machine learning or supervised learning or deeplearning) and (“epileptic seizure” or “convulsions” or “epilepsy”) and (feature extraction or feature selection methods) and (diagnosis or detection or classification)) |
| Springer link | ((machine learning or supervised learning or deeplearning) and (“epileptic seizure” or “convulsions” or “epilepsy”) and (feature extraction or feature selection methods) and (diagnosis or detection or classification)[fields] [all fields]) |
| Ieee Xplore | (((((((((((“all metadata”:”machine learning”) or “all metadata”:supervised learning) or “all metadata”:deep learning) and “all metadata”:epileptic seizure) or “all metadata”:convulsions) or “all metadata”:epilepsy) and “all metadata”:feature extraction) or “all metadata”:feature selection method) and “all metadata”:diagnosis) or “all metadata”:detection) or “all metadata”:classification) |
| MDPI | (“machine learning”[all fields] or “supervised learning” [all fields] or “deep learning”[all fields]) and (“epileptic seizures”[all fields] or “convulsions”[all fields] or “epilepsy”[all fields] or “feature extraction”[all fields] or “feature selection methods” [all fields]) and (“diagnosis”[all fields] OR “detection”[all fields] or “classification”[all fields]) |
| WILEY | ((machine learning or supervised learning or deep learning) and (“epileptic seizure” or “convulsions” or “epilepsy”) and (feature extraction or feature selection methods) and (diagnosis or detection or classification)) |
| ACM Digital Library | ((((“machine” or “supervised learning”) and learning) or deep learning or (“machine” and “learning”) and ((“epileptic seizure” or “convulsions”) and (“feature “ or “feature extraction”) and selection) or ((“feature extraction method”) and classification) or (“detection of” and (“classification” or “diagnosis”)))) |
| Ref: | Year | Problem Tackled | Classifiers | Technique | Findings | Datasets |
|---|---|---|---|---|---|---|
| [1] | 2020 | Differentiating normal EEG signals with Epileptic seizure signals in ictal and inter-ictal stage | Linear and Non-linear ML techniques NB, KNN, MLP, SVM | CAD-based diagnoses using DWT, Wavelet Decomposition, Feature computation, and classification, Arithmetic Coding | Overall 100% accuracy | Bonn University |
| [4] | 2021 | Identifying pre-ictal and ictal state of EEG signals | Fuzzy classifier | Pattern adaptive wavelet transform | 96% accuracy | CHB-MIT |
| [5] | 2019 | Misdiagnose in manual methods, so the procedure is automated | SVM KNN Deep Neural Networks | Feature Scaling Loss Function | SVM = 94% accuracy KNN = 74% accuracy | Bonn University |
| [9] | 2022 | Detecting seizure and non-seizure events | SVM RF | Tunable Q-wavelet transform | RF-sensitivity = 91.5% RF-accuracy = 93% SVM-sensitivity = 9 = 89.2% SVM-accuracy = 90.4% | CHB-MIT |
| [18] | 2017 | The automated framework created for the automated detection of epilepsy | ANN model RPROP+, RPROP-, SAG, SLR | Feature Selection Back Propagation | SLR = 99%, SAG = 97% in balanced classes and SLR = 87%, SAG = 89% in balanced classes | Preprocessed Epileptic seizure recognition on UCI repository. Balanced and Imbalanced classes. |
| [21] | 2019 | Seizure prediction and detection | Random Forest | DWT with 5-level decomposition | High classification sensitivity was achieved by this method, reaching 99.95% in comparison with other studies | Bonn University. Freiburg Hospital. |
| [22] | 2018 | Automated onset prediction | SVM MLP KNN RF | Multiscale principle analyzing for de-noising. EMD DWT Wavelet packet decomposition Inter vs. inter-ictal | With the Freiburg dataset and CHB-MIT dataset, high accuracy has been achieved, indicating both well-known datasets worked well with the used technique | Freiburg Hospital. CHB-MIT. |
| [23] | 2019 | A hybrid model for epileptic seizure prediction | SVM | PSO-based SVM GA-based SVM DWT with db4 | SVM= 97.87% accuracy GA-SVM = 98.75% PSO-SVM = 99.38% | Publically available dataset |
| [24] | 2020 | Automated seizure detection process with a comparison of the proposed method with existing methods | SVM | Successive Decomposition Index SDI. Wavelet energy | The sensitivity achieved is 97.53% with F-measure with 97.22% | Ramaiah College Hospital. CHB-MIT. Temple Unit |
| [25] | 2021 | Detecting epileptic seizures and defining the right features | SVM ANN | DWT 5 level and statistical calculations. Statistical features were extracted | 2-class: ANN = 100% accuracy, SVM = 100% accuracy 3-class: SVM = 98.7% accuracy ANN = 98.7% accuracy | Bonn University |
| [26] | 2018 | Monitoring of brain activity during an epileptic seizure and normal state | SVM KNN Decision trees | Time-frequency domain characteristics Non-linear wavelet-based entropy | SVM = 98% accuracy KNN = 94% accuracy | Bonn University |
| [27] | 2020 | Brain activity at different regions for timely and accurate detection of epileptic seizure | SVM KNN | Feature engineering (FT) Wavelet transform The sequential forward floating selection | SVM = 99%, 100%, and 100% in time, frequency, and time-frequency, respectively, KNN = 99.5%, 99%, 99.5%, respectively | Bonn University |
| [28] | 2020 | Classifying normal brain signals with an epileptic seizure while increasing accuracy and reducing the computational cost | ANN KNN NB SVM | 54-DWT wavelets Derived features minimization by using Genetic Algorithm to select relevant features | ANN achieved higher accuracy, reaching 97.82% in comparison with the rest. | 14 classification combinations using Bonn University dataset |
| [29] | 2020 | Effective real-time epilepsy diagnosis | Feedforward multi-layer neural network, MLP, ANN | Field programmable gate array solution (FPGA) | 95% accuracy | TUH-EEG corpus database |
| [30] | 2020 | Predicting a seizure in a pre-ictal stage in terms of specificity and sensitivity | Deep learning techniques SVM CNN | FT EMD WT Feature extraction and handcrafted feature extraction methods. | Sensitivity = 92.7% Specificity = 90.8% | CHB-MIT |
| [31] | 2018 | Development of automated seizure detection | Random forest | Synthesizing generalized Stockwell transform (GST), singular value decomposition (SVD) based feature extraction. Changing n values for 4 cases to see if it affects the accuracies | Highest classification accuracies are 99.12%, 99.16%, 98.65%, 98.62% for four cases | Bonn University |
| [32] | 2020 | Checking performance in terms of accuracy to find relevant patterns related to different mental activities using feature extraction. Extracting features based on spectrogram. | K-means SVM Multilayer perceptron | STFT is used, and window parameters are set to obtain good results. K-means to extract features. Descriptors: Spectral peaks Frequency Time | The comparison was made with other works, and it was noted that the SVM kernel gave a better performance. | Bonn University |
| [33] | 2020 | Combining four different approaches to decompose non-linear and non-stationary signals into a finite number of oscillations (IMFs) | SVM KNN NB Logistic Regression | EMD and its DWT derivatives and use them to generate EEG into oscillations called IMF | EEMD provided better accuracies than EMD analysis. EEMD provided a robust feature extraction and results | Kahib-Celebi School of Medicine |
| [34] | 2019 | Pre-ictal stage prediction multi-class classification | Random forest | Wavelet packet features Wavelet packet decomposition | 84% accuracy | CHB-MIT |
| [35] | 2021 | Detection of epileptic seizure | ANN, SVM, NN, CNN | Wavelet transform Singular value decomposition entropy Petrosian fractal dimension Higuchi fractal dimension | ANN outperformed other classifiers | Bonn University |
| [36] | 2020 | Reducing seizure frequency or prevention of epileptic seizure by early prediction | Random forest Decision trees | DWT Coefficient of variance with all sub-bands | 99.81% accuracy | CHB-MIT |
| [37] | 2018 | Pre-clinical seizure state | Random forest | Cross-frequency coupling Multistate classifier | Sensitivity = 87.9% Specificity = 82.4% Area under ROC = 93.4% | Toronto Western Hospital Epilepsy Monitoring Unit |
| [38] | 2020 | Computationally efficient automated seizure detection | SVM | Successive decomposition Index | SDI = higher detection rate of epileptic seizure in terms of sensitivity, db1 = 97.53%, db2 = 97.28%, db3 = 95.80%, false-detection rate, db1 = 0.4/h, db2 = 0.57/h, db3 = 0.49/h median detection delay, db1 = 1.5 s, db2 = 1.7 s, db3 = 1.5 s f-measure db1 = 97.22%, db2 = 96.29%, db3 = 94.70% | CHB-MIT Ramaiah Medical College Temple University Hospital |
| [39] | 2021 | Presented an approach for the classification of EEG signals based on fuzzy classifier | FDT, Fuzzy random forest (FRF) | fuzzy classifier | 99.3% | Bonn University |
| Ref: | Approaches | Feature Selection Methods | Datasets | Performance Metrics | Limitations | Accuracy |
|---|---|---|---|---|---|---|
| [40] | Random forest classifiers | L1-penalized robust regression | BONN, CHB-MIT | Class Acc | - | 100 |
| [22] | ANN, random forest, SVM, KNN | Power, mean, kurtosis, absolute mean std dev, skewness | CHB-MIT | Sen, spec, Acc | Time complexity | 100 |
| [41] | Forest CERN | 9-statistical features | BONN, CHB-MIT | Class Acc | - | 100 |
| [42] | Random forest | IMF | Kaggle | Sen, spec, Acc | Sen, spec not mentioned | 98.4 |
| [43] | Random forest, SVM | Frequency, 10-time | UCI | ROC-AUC | - | 98 |
| [44] | Random forest, boosting, decision forest | Nine statistical features | Bern Barcelona | Pre, Rec, Fmeasure | High time complexity | 96.67 |
| [45] | Random forest | Time, frequency | EPILEPSY | Sensitivity | Spec not mentioned | 93.8 |
| Ref: | Approaches | Feature Selection Methods | Datasets | Performance Metrics | Limitations | Accuracy |
|---|---|---|---|---|---|---|
| [46] | LS-SVM | DWT, FFT | Class Acc | BONN | High time | 100 |
| [47] | SVM | Energy | BONN, Barcelona | Class Acc | - | 99.5 |
| [48] | SVM | DWT | BONN | Class Acc | - | 99.38 |
| [49] | SVM | Time-frequency | CHB-MIT | Sen, spec | High time complexity | 99.32 |
| [50] | SVM | Time-frequency | CHB- MIT | Sensitivity (sen) | - | 96 |
| [51] | SVM | DWT | CHB-MIT | Avg | - | 94.8 |
| [14] | SVM | Permutation entropy | CHB-MIT | Pre, Rec, Fmeasure | Low prec and accuracy | 93.55 |
| [52] | SVM | DWT | BONN | Confusion Matrix | Low sen, pres | 86.83 |
| Ref: | Approaches | Feature Selection Methods | Datasets | Performance Metrics | Limitations | Accuracy |
|---|---|---|---|---|---|---|
| [53] | KNN and GHE | - | BONN | Class Acc | - | 100 |
| [54] | Naive Bayes, KNN | Energy | EPILEPSY | Class Acc | - | 98.75 |
| [55] | KNN | Time-frequency | BernBarcelona | Sen, pre, NPR, ROC | NFR not mentioned | 97.6 |
| [56] | KNN | 15-features | BONN | Acc, sen, spec | - | 98 |
| [57] | KNN | Genetic programming | BONN | Class Acc | Low accuracy | 93.50 |
| [58] | QDA, DT, KNN | Time-frequency | BONN | Sen, spec | Low sen, pres | 85 |
| Ref: | Approaches | Feature Selection Methods | Datasets | Performance Metrics | Limitations | Accuracy |
|---|---|---|---|---|---|---|
| [50] | ANN | Time-frequency features | BONN | Pre, Rec, Fmeasure | - | 100 |
| [14] | ANN, random forest, SVM, KNN | Power, mean, kurtosis, absolute mean std dev, skewness | CHB-MIT | Sen, spec, Acc | Time complexity | 100 |
| [39] | QDA, DT, KNN | Time-frequency | BONN | Sen, spec | Low sen, pres | 85 |
| [12] | ANN | Line length feature | CHB-MIT | CHB-MIT | Low accuracy | 52 |
| Sr. No. | Classifiers | Used in |
|---|---|---|
| 1 | SVM | [1,10,13,14,15,16,33,34,41,42,46,47,49] |
| 2 | RF | [8,10,16,34,45,48,50,57] |
| 3 | ANN | [9,33,42,43,46,49] |
| 4 | KNN | [1,10,12,32,34,35,41,42,46,47] |
| Ref: | ML Approaches | Feature Extraction Methods |
|---|---|---|
| [22,40,41,42,43,44,45] | Random Forest (RF) | L1-penalized robust regression, Power, mean, kurtosis, absolute mean std dev, skewness, 9-statistical features, DWT, entropy, IMF, Frequency, 10-time, Std, dev, energy, energy, STFT, mean, Nine statistical features, Time, frequency |
| [14,47,48,49,50,51,52] | Support Vector Machine (SVM) | DWT, FFT, Energy, Time-frequency, Permutation entropy |
| [53,58] | K- Nearest Neighbour (KNN) | Energy, 15-features, Time-frequency, Genetic programming, Time-frequency |
| [22] | Artificial Neural Network (ANN) | Time-frequency features, Power, mean, kurtosis, absolute mean std dev, skewness, Time-frequency, Line length feature |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Farooq, M.S.; Zulfiqar, A.; Riaz, S. Epileptic Seizure Detection Using Machine Learning: Taxonomy, Opportunities, and Challenges. Diagnostics 2023, 13, 1058. https://doi.org/10.3390/diagnostics13061058
Farooq MS, Zulfiqar A, Riaz S. Epileptic Seizure Detection Using Machine Learning: Taxonomy, Opportunities, and Challenges. Diagnostics. 2023; 13(6):1058. https://doi.org/10.3390/diagnostics13061058
Chicago/Turabian StyleFarooq, Muhammad Shoaib, Aimen Zulfiqar, and Shamyla Riaz. 2023. "Epileptic Seizure Detection Using Machine Learning: Taxonomy, Opportunities, and Challenges" Diagnostics 13, no. 6: 1058. https://doi.org/10.3390/diagnostics13061058
APA StyleFarooq, M. S., Zulfiqar, A., & Riaz, S. (2023). Epileptic Seizure Detection Using Machine Learning: Taxonomy, Opportunities, and Challenges. Diagnostics, 13(6), 1058. https://doi.org/10.3390/diagnostics13061058






