Comparative Study of the Impact of Human Leukocyte Antigens on Renal Transplant Survival in Andalusia and the United States
Abstract
1. Introduction
2. Methods and Materials
2.1. Methodology
- Bibliographic search of survival analysis studies that consider blood group and HLA incompatibilities as factors that have an impact on renal transplant survival.
- Pre-processing of the available databases by performing cleaning and quality improvement process, including standardisation of date-type variables, identification of null values, elimination of unimportant variables, renaming of some variables in order to better describe their content, standardisation of values using the scaling technique and conversion of categorical variables.
- Analysis of renal graft survival using the Kaplan–Meier estimator considering the different HLA incompatibilities, both in isolation and together as well as in combination with the blood group.
- Obtaining the coefficients of the Cox model for each of the identified factors with greater significance identified in the previous step.
- Graphical representation of the survival probabilities obtained and discussion of the results.
2.2. Data Characteristics
3. Results
Results of the Data Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lewis, A.; Koukoura, A.; Tsianos, G.-I.; Gargavanis, A.A.; Nielsen, A.A.; Vassiliadis, E. Organ Donation in the US and Europe: The Supply vs. Demand Imbalance. Transplant. Rev. 2021, 35, 100585. [Google Scholar] [CrossRef] [PubMed]
- Browne, T.; McPherson, L.; Retzloff, S.; Darius, A.; Wilk, A.S.; Cruz, A.; Wright, S.; Pastan, S.O.; Gander, J.C.; Berlin, A.A.; et al. Improving Access to Kidney Transplantation: Perspectives from Dialysis and Transplant Staff in the Southeastern United States. Kidney Med. 2021, 3, 799–807.e1. [Google Scholar] [CrossRef]
- Wang, J.H.; Hart, A. Global Perspective on Kidney Transplantation: United States. Kidney360 2021, 2, 1836–1839. [Google Scholar] [CrossRef] [PubMed]
- Gondos, A.; Döhler, B.; Brenner, H.; Opelz, G. Kidney Graft Survival in Europe and the United States: Strikingly Different Long-Term Outcomes. Transplantation 2013, 95, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Pullen, L.C. The Economy of Transplant: Saving Money and Saving Lives. Am. J. Transplant. 2021, 21, 1353–1354. [Google Scholar] [CrossRef]
- Kelly, D.M.; Anders, H.-J.; Bello, A.K.; Choukroun, G.; Coppo, R.; Dreyer, G.; Eckardt, K.-U.; Johnson, D.W.; Jha, V.; Harris, D.C.H.; et al. International Society of Nephrology Global Kidney Health Atlas: Structures, Organization, and Services for the Management of Kidney Failure in Western Europe. Kidney Int. Suppl. 2021, 11, e106–e118. [Google Scholar] [CrossRef]
- Assfalg, V.; Selig, K.; Tolksdorf, J.; van Meel, M.; de Vries, E.; Ramsoebhag, A.-M.; Rahmel, A.; Renders, L.; Novotny, A.; Matevossian, E.; et al. Repeated Kidney Re-Transplantation—The Eurotransplant Experience: A Retrospective Multicenter Outcome Analysis. Transpl. Int. 2020, 33, 617–631. [Google Scholar]
- Sistema Español de Donación y Trasplante, Consumo y Bienestar social (Gobierno de España) Plan Estratégico En Donación y Trasplante de Órganos 2018–2022. 2018. Available online: http://www.ont.es/infesp/Paginas/plan-estrategico-2018-2022.aspx (accessed on 22 January 2023).
- Tambur, A.R.; Kosmoliaptsis, V.; Claas, F.H.J.; Mannon, R.B.; Nickerson, P.; Naesens, M. Significance of HLA-DQ in Kidney Transplantation: Time to Reevaluate Human Leukocyte Antigen-Matching Priorities to Improve Transplant Outcomes? An Expert Review and Recommendations. Kidney Int. 2021, 100, 1012–1022. [Google Scholar] [CrossRef]
- Becker, L.E.; Süsal, C.; Morath, C. Kidney Transplantation across HLA and ABO Antibody Barriers. Curr. Opin. Organ. Transpl. 2013, 18, 445–454. [Google Scholar] [CrossRef]
- Mamode, N.; Bestard, O.; Claas, F.; Furian, L.; Griffin, S.; Legendre, C.; Pengel, L.; Naesens, M. European Guideline for the Management of Kidney Transplant Patients with HLA Antibodies: By the European Society for Organ Transplantation Working Group. Transpl. Int. 2022, 35, 10511. [Google Scholar] [CrossRef]
- Krishnan, N.; Abimbola, A.; Machan, N.; Daga, S.; Gopalakrishnan, K.; Lam, F.; Tan, L.; Kashi, H.; Imray, C.; Zehnder, D.; et al. HLA Antibody Incompatible Renal Transplantation: Long-Term Outcomes Similar to Deceased Donor Transplantation. Transplant. Direct 2021, 7, e732. [Google Scholar] [CrossRef] [PubMed]
- Talaminos-Barroso, A.; Reina-Tosina, J.; Roa-Romero, L.M.; Calvillo-Arbizu, J.; Pérez-Valdivia, M.A.; Medina-López, R.; Castro-de-la-Nuez, P. Análisis Del Impacto Combinado Del Grupo Sanguíneo y Los Antígenos Leucocitarios Humanos En La Supervivencia Del Trasplante Renal En Andalucía. In Proceedings of the XXXIX Congreso Anual de la Sociedad Española de Ingeniería Biomédica (CASEIB 2021), Ciudad Real, Spain, 21–23 November 2021. [Google Scholar]
- Oellerich, M.; Shipkova, M.; Asendorf, T.; Walson, P.D.; Schauerte, V.; Mettenmeyer, N.; Kabakchiev, M.; Hasche, G.; Gröne, H.-J.; Friede, T.; et al. Absolute Quantification of Donor-Derived Cell-Free DNA as a Marker of Rejection and Graft Injury in Kidney Transplantation: Results from a Prospective Observational Study. Am. J. Transplant. 2019, 19, 3087–3099. [Google Scholar] [CrossRef] [PubMed]
- Erlich, H.A.; Opelz, G.; Hansen, J. HLA DNA Typing and Transplantation. Immunity 2001, 14, 347–356. [Google Scholar] [CrossRef]
- dela Cruz, T.; Dames, C.; Pagaduan, L.; Cho, Y.; Kong, D.; Rajalingam, R. Concurrent Use of Two Independent Methods Prevents Erroneous HLA Typing of Deceased Organ Donors—An Important Strategy for Patient Safety and Accurate Virtual Crossmatching for Broader Sharing. Hum. Immunol. 2022, 83, 458–466. [Google Scholar] [CrossRef] [PubMed]
- Foster, B.J.; Dahhou, M.; Zhang, X.; Platt, R.W.; Hanley, J.A. Relative Importance of HLA Mismatch and Donor Age to Graft Survival in Young Kidney Transplant Recipients. Transplantation 2013, 96, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Opelz, G.; Döhler, B.; Middleton, D.; Süsal, C. A Collaborative Transplant Study Report HLA Matching in Pediatric Kidney Transplantation: HLA Poorly Matched Living Donor Transplants Versus HLA Well-Matched Deceased Donor Transplants. Transplantation 2017, 101, 2789–2792. [Google Scholar] [CrossRef] [PubMed]
- Calvillo-Arbizu, J.; Pérez-Valdivia, M.A.; Gentil-Govantes, M.A.; Castro-de-la-Nuez, P.; Mazuecos-Blanca, A.; Rodríguez-Benot, A.; Gracia-Guindo, M.C.; Borrego-Utiel, F.; Cabello-Díaz, M.; Bedoya-Pérez, R.; et al. Does the Kidney Donor Profile Index (KDPI) Predict Graft and Patient Survival in a Spanish Population? Nefrología 2018, 38, 587–595. [Google Scholar] [CrossRef]
- Williams, R.C.; Opelz, G.; McGarvey, C.J.; Weil, E.J.; Chakkera, H.A. The Risk of Transplant Failure with HLA Mismatch in First Adult Kidney Allografts from Deceased Donors. Transplantation 2016, 100, 1094–1102. [Google Scholar] [CrossRef]
- Kadatz, M.; Lan, J.H.; Brar, S.; Vaishnav, S.; Chang, D.T.; Gill, J.; Gill, J.S. Transplantation of Patients with Long Dialysis Vintage in the Current Deceased Donor Kidney Allocation System (KAS). Am. J. Kidney Dis. 2022, 80, 319–329.e1. [Google Scholar] [CrossRef]
- Hariharan, S.; Israni, A.K.; Danovitch, G. Long-Term Survival after Kidney Transplantation. N. Engl. J. Med. 2022, 386, 497–500. [Google Scholar] [CrossRef]
- Poggio, E.D.; Augustine, J.J.; Arrigain, S.; Brennan, D.C.; Schold, J.D. Long-Term Kidney Transplant Graft Survival—Making Progress When Most Needed. Am. J. Transplant. 2021, 21, 2824–2832. [Google Scholar] [CrossRef] [PubMed]
- Seifert, M.E.; Agarwal, G.; Bernard, M.; Kasik, E.; Raza, S.S.; Fatima, H.; Gaston, R.S.; Hauptfeld-Dolejsek, V.; Julian, B.A.; Kew, C.E.; et al. Impact of Subclinical Borderline Inflammation on Kidney Transplant Outcomes. Transpl. Direct 2021, 7, e663. [Google Scholar] [CrossRef]
- Vanholder, R.; Stel, V.S.; Jager, K.J.; Lameire, N.; Loud, F.; Oberbauer, R.; de Jong, R.W.; Zoccali, C. How to Increase Kidney Transplant Activity throughout Europe—An Advocacy Review by the European Kidney Health Alliance. Nephrol. Dial. Transplant. 2019, 34, 1254–1261. [Google Scholar] [CrossRef] [PubMed]
- Saez-Giménez, B.; Berastegui, C.; Barrecheguren, M.; Revilla-López, E.; Arcos, I.L.; Alonso, R.; Aguilar, M.; Mora, V.M.; Otero, I.; Reig, J.P.; et al. COVID-19 in Lung Transplant Recipients: A Multicenter Study. Am. J. Transplant. 2021, 21, 1816–1824. [Google Scholar] [CrossRef] [PubMed]
- Krampe, N.; Nebra Puertas, A.C.; Povar Echeverría, M.; Elmer, J.; Povar Marco, J. Comparing Demographics of Organ Donor Referrals from the Intensive Care Unit and Outside Units. Transpl. Int. 2021, 34, 2146–2153. [Google Scholar] [CrossRef] [PubMed]
- Sureshkumar, K.K.; Chopra, B. Induction Type and Outcomes in HLA-DR Mismatch Kidney Transplantation. Transplant. Proc. 2019, 51, 1796–1800. [Google Scholar] [CrossRef]
- Al-Otaibi, T.; Gheith, O.; Mosaad, A.; Nampoory, M.R.N.; Halim, M.; Said, T.; Nair, P. Human Leukocyte Antigen-DR Mismatched Pediatric Renal Transplant: Patient and Graft Outcome with Different Kidney Donor Sources. Exp. Clin. Transpl. 2015, 13 (Suppl. S1), 117–123. [Google Scholar]
- Opelz, G.; Döhler, B. Effect of Human Leukocyte Antigen Compatibility on Kidney Graft Survival: Comparative Analysis of Two Decades. Transplantation 2007, 84, 137–143. [Google Scholar] [CrossRef]
- Su, X.; Zenios, S.A.; Chakkera, H.; Milford, E.L.; Chertow, G.M. Diminishing Significance of HLA Matching in Kidney Transplantation. Am. J. Transpl. 2004, 4, 1501–1508. [Google Scholar] [CrossRef]
- Firoz, A.; Kashem, M.; Zhao, H.; Geier, S.; Toyoda, Y. Human Leukocyte Antigen Mismatch on Lung Transplantation Outcomes. Eur. J. Cardio-Thorac. Surg. 2022, 62, ezac132. [Google Scholar] [CrossRef]
- Zachary, A.A.; Leffell, M.S. HLA Mismatching Strategies for Solid Organ Transplantation—A Balancing Act. Front. Immunol. 2016, 7, 575. [Google Scholar] [CrossRef]
- Casey, M.J.; Wen, X.; Rehman, S.; Santos, A.H.; Andreoni, K.A. Rethinking the Advantage of Zero-HLA Mismatches in Unrelated Living Donor Kidney Transplantation: Implications on Kidney Paired Donation. Transpl. Int. 2015, 28, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Williams, R.C.; Opelz, G.; Weil, E.J.; McGarvey, C.J.; Chakkera, H.A. The Risk of Transplant Failure With HLA Mismatch in First Adult Kidney Allografts 2: Living Donors, Summary, Guide. Transpl. Direct 2017, 3, e152. [Google Scholar] [CrossRef] [PubMed]
- Morales, J.M.; Marcén, R.; Andrés, A.; Molina, M.G.; Castillo, D.D.; Cabello, M.; Capdevila, L.; Campistol, J.M.; Oppenheimer, F.; Serón, D.; et al. Renal Transplantation in the Modern Immunosuppressive Era in Spain: Four-Year Results from a Multicenter Database Focus on Post-Transplant Cardiovascular Disease. Kidney Int. Suppl. 2008, 74, S94–S99. [Google Scholar] [CrossRef] [PubMed]
- Lazda, V.A. The Impact of HLA Frequency Differences in Races on the Access to Optimally HLA-Matched Cadaver Renal Transplants. The Medical Advisory Committee. Transplantation 1992, 53, 352–357. [Google Scholar] [CrossRef] [PubMed]
- Bekbolsynov, D.; Mierzejewska, B.; Khuder, S.; Ekwenna, O.; Rees, M.; Green, R.C.; Stepkowski, S.M. Improving Access to HLA-Matched Kidney Transplants for African American Patients. Front. Immunol. 2022, 13, 832488. [Google Scholar] [CrossRef] [PubMed]
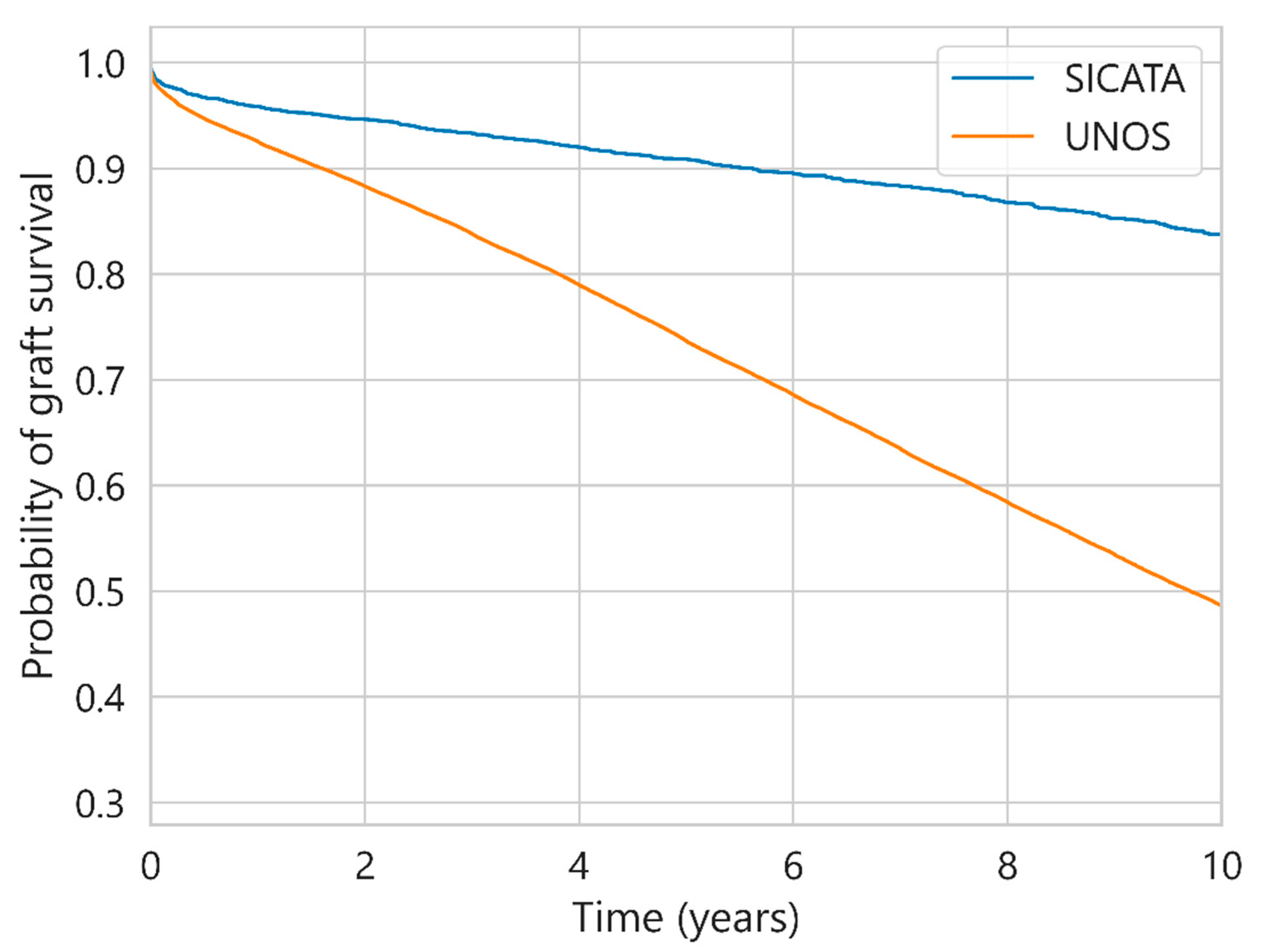

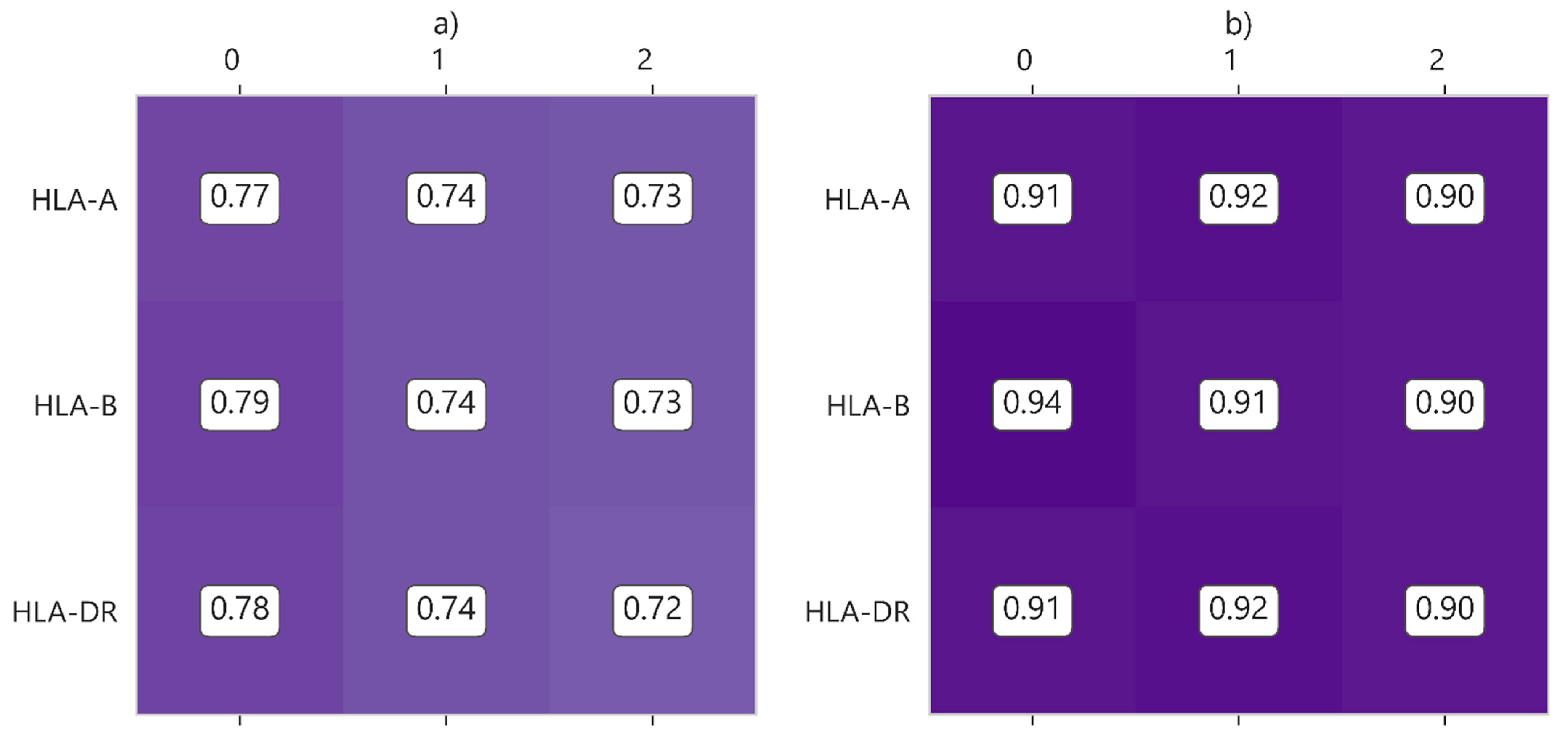
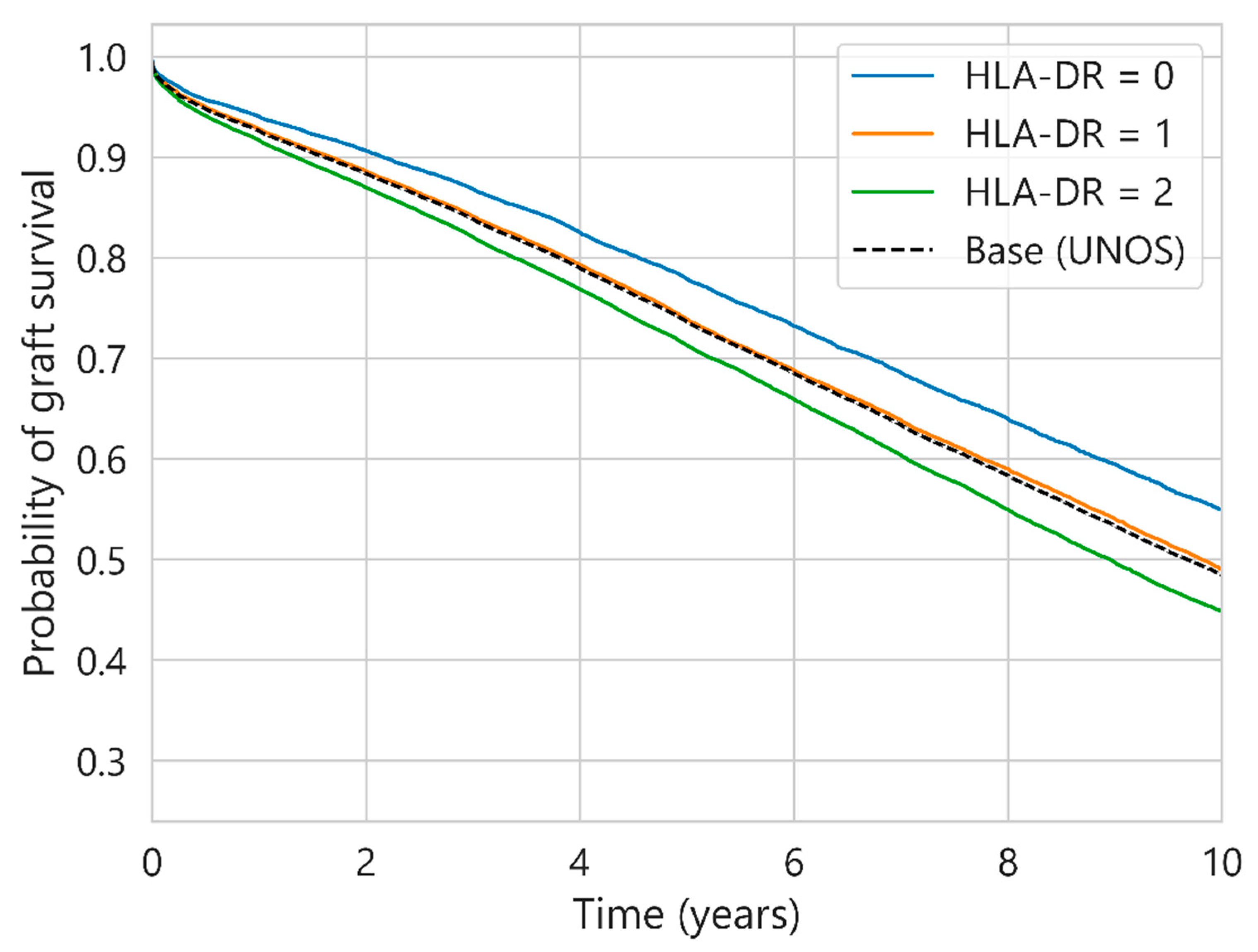
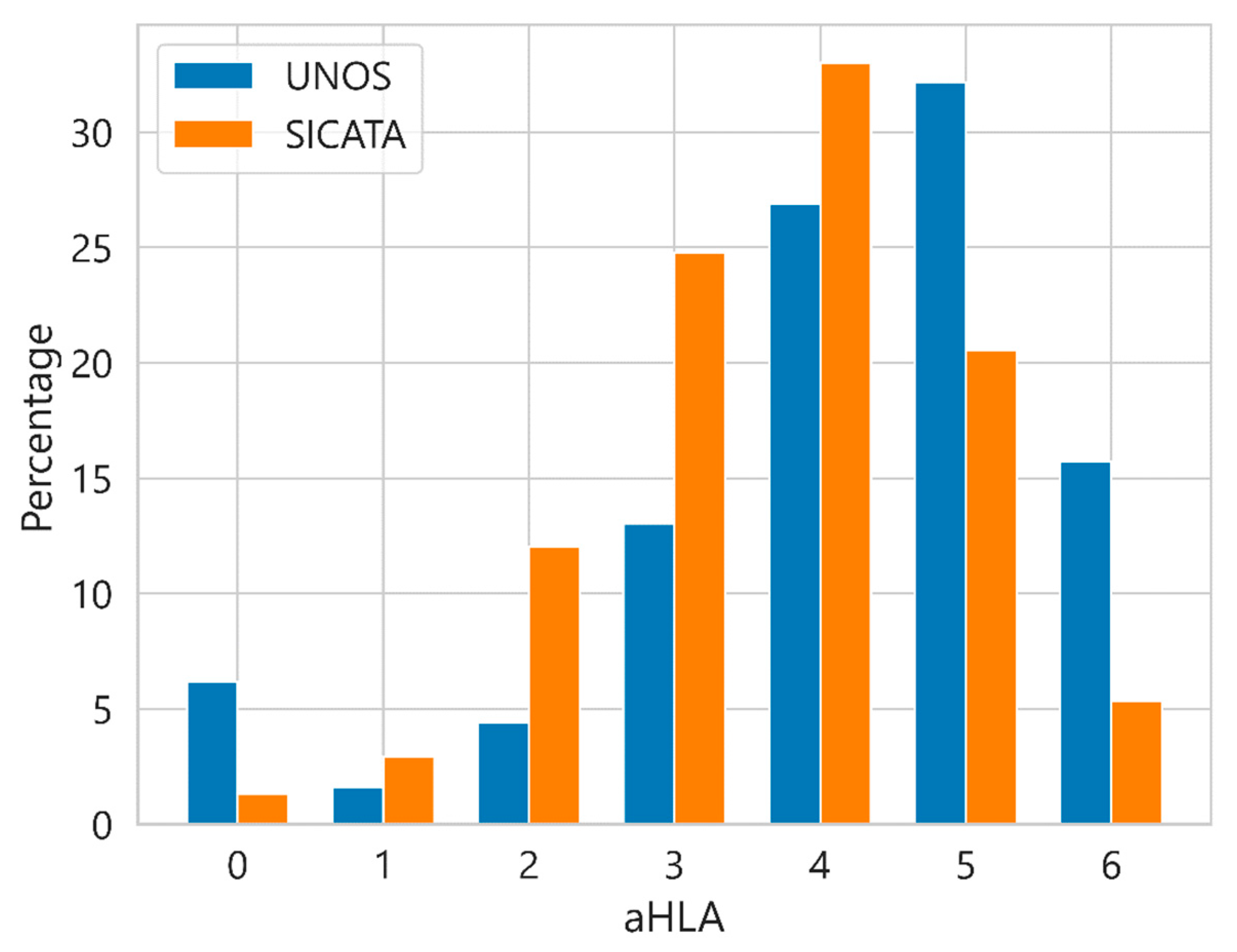
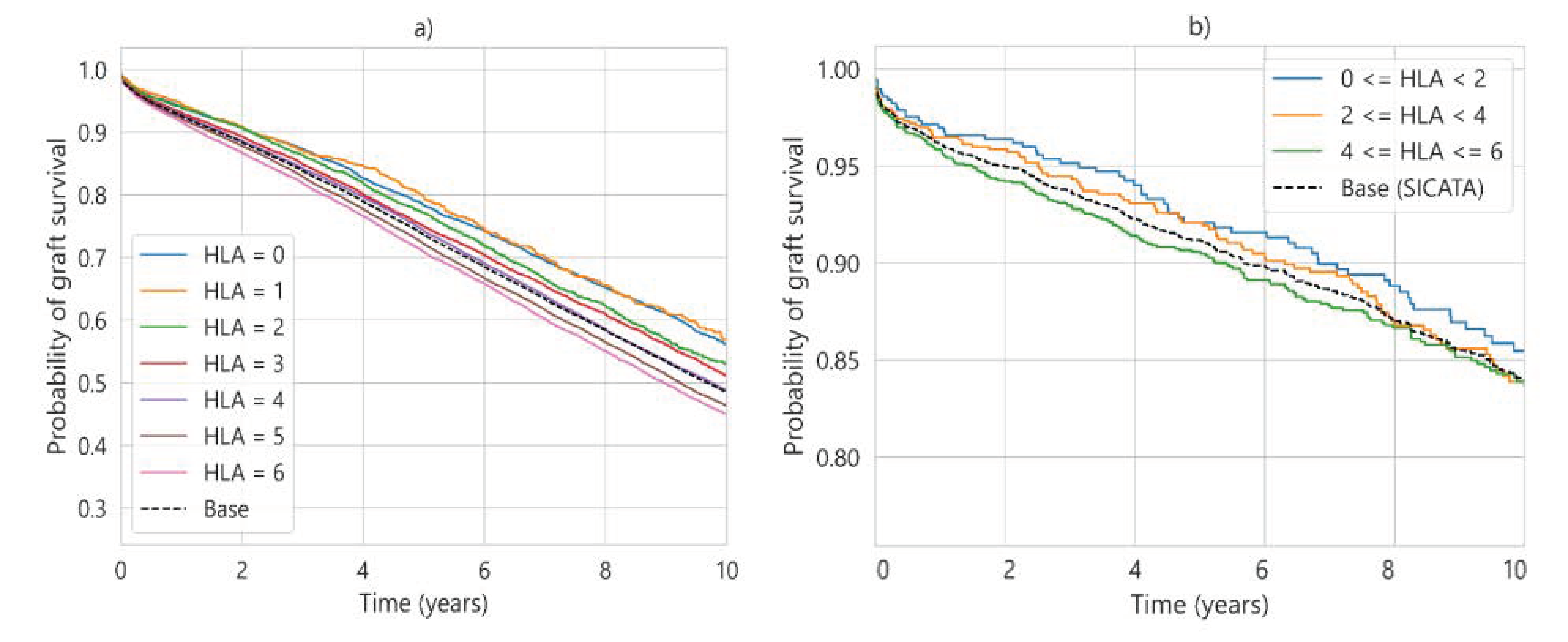
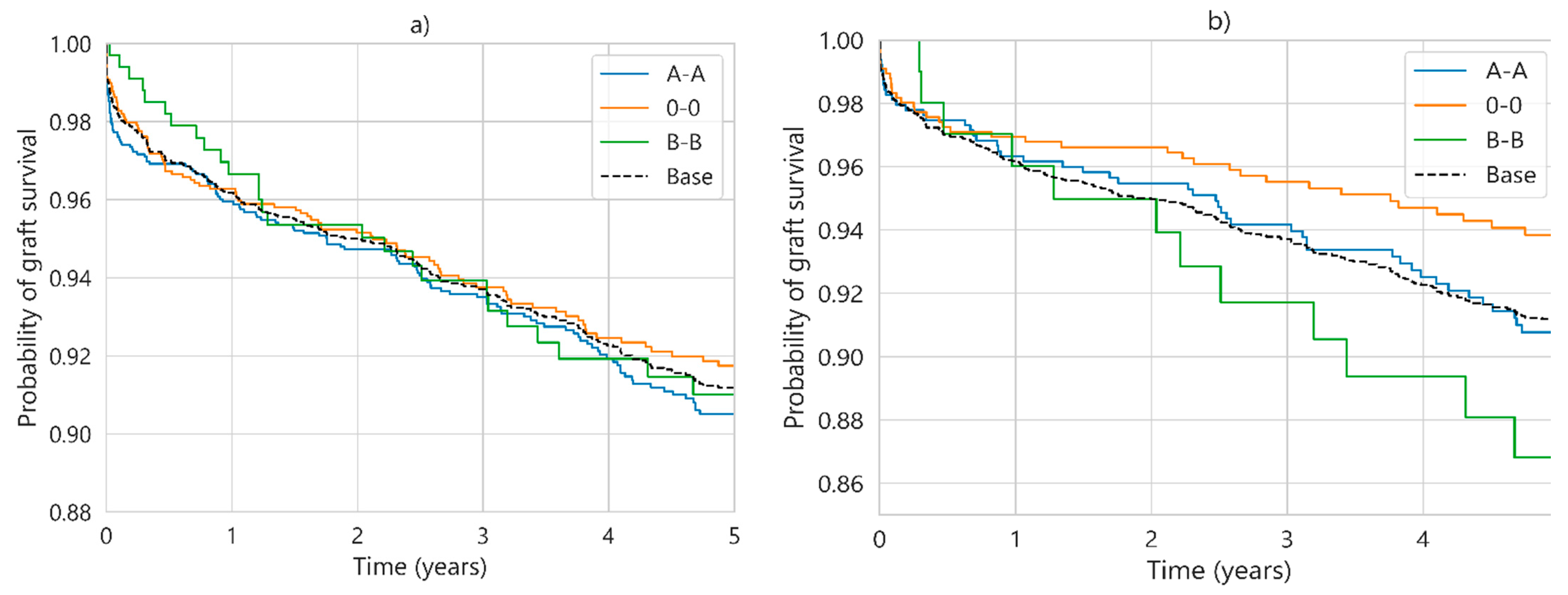
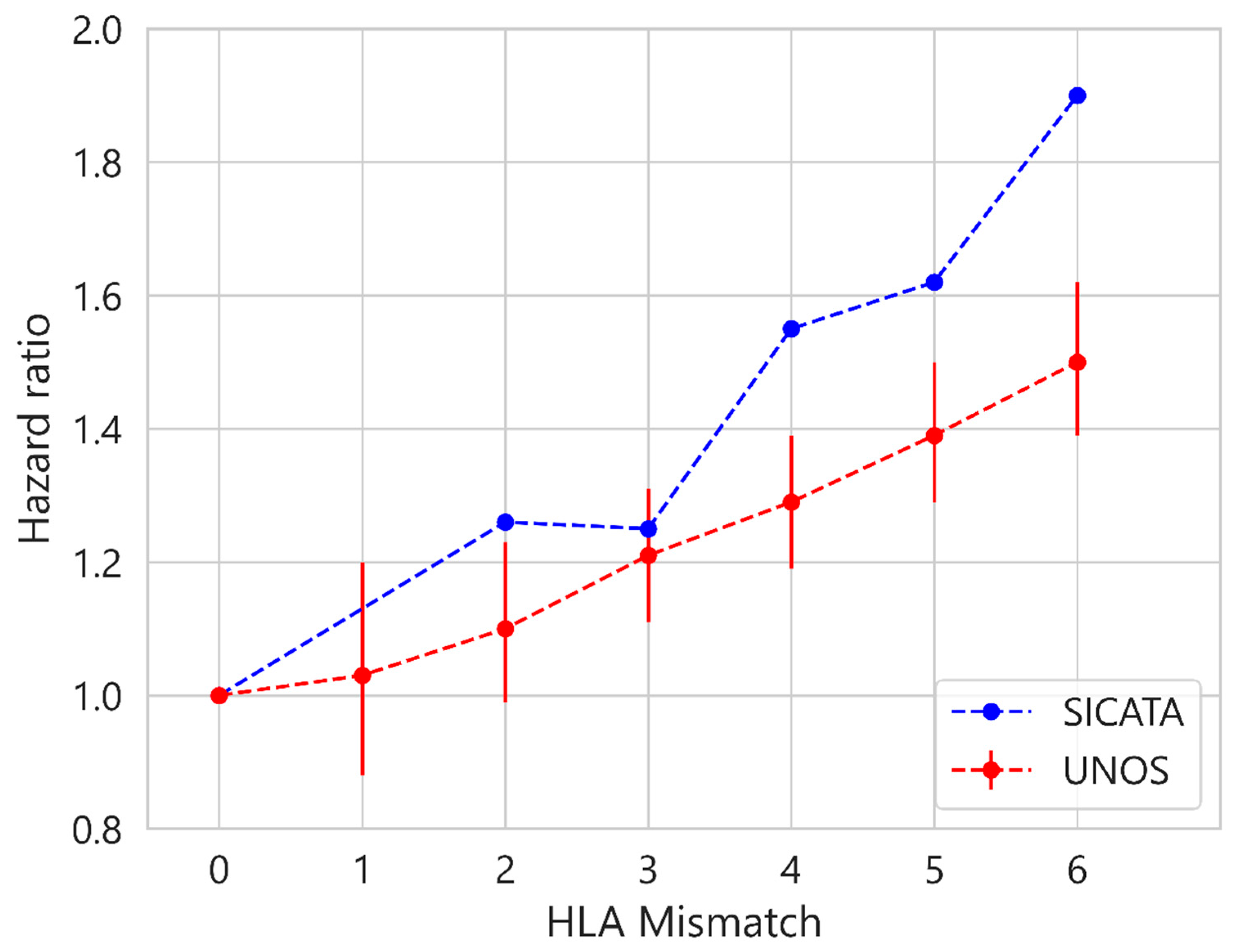
| Characteristic | UNOS (112,877 Cases) | SICATA (3478 Cases) |
|---|---|---|
| Age of donor | 38.93 (±15.42) | 53.32 (±15.02) |
| Gender of donor (% men) | 68,903 (61.04%) | 2143 (61.62%) |
| BMI of donor (kg/m2) | 27.77 (±6.82) | 27.4 (±15.02) |
| Age of recipient | 52.81 (±13.14) | 52.74 (±12.89) |
| Gender of recipient (% men) | 68,289 (60.5%) | 2205 (63.4%) |
| BMI of recipient (kg/m2) | 28.11 (±5.4) | 27.18 (±7.87) |
| Blood group A–A | 20,461 (18.13%) | 1624 (46.69%) |
| Blood group B–B | 18,570 (16.45%) | 335 (9.63%) |
| Blood group O–O | 71,386 (63.24%) | 1386 (39.85%) |
| Blood group AB–AB | 2460 (2.18%) | 133 (3.82%) |
| HLA-A: class 0 | 14,966 (13.26%) | 568 (16.33%) |
| HLA-A: class 1 | 42,965 (38.06%) | 1760 (50.6%) |
| HLA-A: class 2 | 54,946 (48.68%) | 1150 (33.06%) |
| HLA-B: class 0 | 10,216 (9.05%) | 247 (7.1%) |
| HLA-B: class 1 | 27,492 (26.36%) | 1140 (40.54%) |
| HLA-B: class 2 | 75,169 (66.59%) | 1821 (52.36%) |
| HLA-DR: class 0 | 19,658 (17.42%) | 647 (18.6%) |
| HLA-DR: class 1 | 51,656 (45.76%) | 1965 (56.5%) |
| HLA-DR: class 2 | 41,563 (36.82%) | 866 (24.9%) |
| aHLA | Cases | Probability of Survival | ||
|---|---|---|---|---|
| 1 Year | 3 Years | 5 Years | ||
| 1 | 1789 | 0.95 | 0.88 | 0.81 |
| 2 | 4927 | 0.94 | 0.87 | 0.78 |
| 3 | 14,627 | 0.94 | 0.85 | 0.75 |
| 4 | 16,236 | 0.93 | 0.85 | 0.74 |
| 5 | 36,085 | 0.93 | 0.84 | 0.73 |
| 6 | 17,644 | 0.92 | 0.83 | 0.72 |
| aHLA | Cases | Probability of Survival | ||
|---|---|---|---|---|
| 1 Year | 3 Years | 5 Years | ||
| 1 | 102 | 0.99 | 0.98 | 0.98 |
| 2 | 419 | 0.94 | 0.93 | 0.91 |
| 3 | 862 | 0.96 | 0.95 | 0.92 |
| 4 | 617 | 0.97 | 0.91 | 0.89 |
| 5 | 715 | 0.95 | 0.92 | 0.89 |
| 6 | 186 | 0.94 | 0.92 | 0.9 |
| Factor | SICATA | UNOS | ||
|---|---|---|---|---|
| Hazard Ratio | p | Hazard Ratio | p | |
| Age of donor | 1.04 (1.02–1.05) | <0.005 | 1.01 (1.01–1.01) | <0.005 |
| Gender of donor | 0.96 (0.69–1.34) | 0.81 | 1.11 (1.07–1.14) | <0.005 |
| BMI of donor | 1.01 (0.97–1.04) | 0.69 | 1 (1–1) | 0.75 |
| Age of recipient | 0.98 (0.96–0.99) | <0.005 | 1.01 (1.01–1.01) | <0.005 |
| Gender of recipient | 1.47 (1.06–2.02) | 0.02 | 0.88 (0.85–0.91) | <0.005 |
| BMI del receptor | 1 (0.99–1.02) | 0.76 | 1.01 (1.01–1.01) | <0.005 |
| HLA-A | 1.1 (0.87–1.39) | 0.44 | 1.02 (1–1.05) | 0.09 |
| HLA-B | 1.26 (0.93–1.56) | 0.1 | 1.02 (0.99–1.05) | 0.2 |
| HLA-DR | 1.2 (0.93–1.54) | 0.16 | 1.12 (1.1–1.15) | <0.005 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Talaminos Barroso, A.; Reina Tosina, J.; Roa, L.M.; Calvillo Arbizu, J.; Pérez Valdivia, M.A.; Medina, R.; Rocha Castilla, J.L.; Castro-de-la-Nuez, P. Comparative Study of the Impact of Human Leukocyte Antigens on Renal Transplant Survival in Andalusia and the United States. Diagnostics 2023, 13, 608. https://doi.org/10.3390/diagnostics13040608
Talaminos Barroso A, Reina Tosina J, Roa LM, Calvillo Arbizu J, Pérez Valdivia MA, Medina R, Rocha Castilla JL, Castro-de-la-Nuez P. Comparative Study of the Impact of Human Leukocyte Antigens on Renal Transplant Survival in Andalusia and the United States. Diagnostics. 2023; 13(4):608. https://doi.org/10.3390/diagnostics13040608
Chicago/Turabian StyleTalaminos Barroso, Alejandro, Javier Reina Tosina, Laura M. Roa, Jorge Calvillo Arbizu, Miguel Angel Pérez Valdivia, Rafael Medina, Jose Luis Rocha Castilla, and Pablo Castro-de-la-Nuez. 2023. "Comparative Study of the Impact of Human Leukocyte Antigens on Renal Transplant Survival in Andalusia and the United States" Diagnostics 13, no. 4: 608. https://doi.org/10.3390/diagnostics13040608
APA StyleTalaminos Barroso, A., Reina Tosina, J., Roa, L. M., Calvillo Arbizu, J., Pérez Valdivia, M. A., Medina, R., Rocha Castilla, J. L., & Castro-de-la-Nuez, P. (2023). Comparative Study of the Impact of Human Leukocyte Antigens on Renal Transplant Survival in Andalusia and the United States. Diagnostics, 13(4), 608. https://doi.org/10.3390/diagnostics13040608







