Abstract
ADAM33 has been linked to airway structural changes in patients with asthma, leading to airway hyperresponsiveness, narrowing, and ultimately poor treatment responsiveness. This study aimed to evaluate the genetic association of ADAM33 SNPs with asthma, disease severity, and treatment responsiveness to ICS+LABA in the South Indian population. In this case–control study (486 controls and 503 cases), we performed genotyping using MassArray for six SNPs of ADAM33, namely rs2280091, rs2787094, rs3918396, rs67044, rs2853209, and rs3918392. We studied the association with asthma and treatment responsiveness to ICS+LABA, using genotype, allele frequency distribution, and haplotype analysis. A significant clinical finding of the study was that certain patients in the disease severity group (moderate and mild) showed poor or no improvement after a three-month follow-up of regular ICS+LABA therapy. Of the studied ADAM33 SNPs, rs2853209 showed an association with asthma. The further analysis of asthma patients according to disease severity suggested an association between moderate disease and the minor allele “T” for rs2853209. The homozygous minor allele of SNP rs2787094 was found to be associated with poorer lung function and the least lung-function improvement after three months of ICS+LABA therapy. The haplotype analysis of six SNPs showed a significant association between the rs2853209 and rs3918396 blocks and asthma. ADAM33 gene polymorphism has clinical relevance in terms of disease association and response to treatment. SNP rs2853209 seemed most relevant to asthma, and SNP rs2787094 could be a genetic marker for predicting response to ICS+LABA therapy in the study population.
1. Introduction
Asthma is a heterogeneous chronic inflammatory disease characterized by reversible airflow limitation [1]. It affects 300 million individuals globally, of which 37.9 million live in India [2,3]. In addition to the already well-established risk factors for asthma, including family, environmental, and individual factors such as smoking and medication adherence, genetics has a significant influence [4,5,6]. Recently, studies such as family and twin association studies [7,8], linkage studies [9], candidate gene studies, and genome-wide association studies [9,10] in asthma have proved strong genetic associations in asthma [8,11]. Structural changes in the airways, or “airway remodeling”, an umbrella term encompassing several modifications in the airways caused by chronic inflammation, is considered one of the significant complications of long-term asthma [12,13,14]. Studies have confirmed that structural changes in the airways impact asthma [15,16]. The first report on genes involved in asthma pathogenesis and airway hyperresponsiveness, other than those concerning non-immune and non-allergic pathways, addressed the positionally cloned asthma and bronchial hyperresponsiveness gene ADAM33 [17]. This codes for the ADAM33 protein, a disintegrin and metalloproteinase glycoprotein involved in intercellular and cell–matrix interactions. Soluble ADAM33 plays a crucial role in airway remodeling [18]. The variations of this gene have been studied in different populations independently and have been found to be associated with asthma [9]. Airway remodeling is involved in airway hyperresponsiveness; airway narrowing; and, finally, poor treatment responsiveness and fixed airflow obstruction. In routine clinical practice, it is difficult to confirm, but the non-reversibility of lung functions after adequate treatment has the potential to be a surrogate marker. Lung functions, especially FEV1, may be initially partly reversible and progress to irreversibility in some asthma patients [19].
Medical practitioners have observed that there are variations in response to asthma therapy. This implies that there are three categories of asthma patients in a particular class of asthma therapy (“responders”, “partial responders”, and “non-responders”), and considerations of genetics might aid in the choice of treatment for asthma [20]. In this study, we were keen to find the relationship between ADAM33 SNPs and asthma, its severity, and the treatment responsiveness after three months of regular inhaled corticosteroids (ICS) and long-acting beta-agonist (LABA) using dosages based on the GINA guidelines for the management of mild, moderate, and persistent asthma [21,22].
2. Materials and Methods
2.1. Study Population
The study recruited 1074 adult individuals (549 asthma cases and 525 controls, age- and gender-matched). Among them, 503 asthma cases and 486 normal controls were subjected to genotype analysis. The remaining 85 patients did not meet the inclusion criteria (unacceptable spirometry, non-atopic asthma, additional co-morbid conditions). The case and control recruitment period lasted from July 2012 to February 2017. The Institutional Human Ethical Committee, University of Mysore approved the study (IHEC-UOM No.79 Ph.D/2012-13). Written informed consent was obtained from all the asthma patients and participants of the study.
The study did not include individuals under 18 years and asthma patients with other respiratory diseases. Age- and gender-matched controls were selected from the general population in the same geographical area as the asthma cases. The study was conducted on consecutive cases of asthma visiting the department of Respiratory Medicine, JSS Medical College and Hospital, a tertiary-care University teaching hospital. The cases underwent spirometry, satisfying the American Thoracic Society (ATS) standards [23]. Asthma was confirmed according to GINA guidelines for the diagnosis and classification of asthma, i.e., a reversibility of more than 12% and 200 mL on post-bronchodilator spirometry, and asthma severity was classified as mild, moderate, and severe persistent asthma [22]. Spirometry was performed using an Easy-One portable spirometer (NDD Medizintechnik; Zurich, Switzerland). All asthma patients were treatment-naïve (ICS+LABA) at the time of recruitment into the study and used oral bronchodilators as needed before entry into the study. All asthma patients received ICS+LABA for three months, and pre-bronchodilator spirometry was repeated at the end of the three months of the study, when patients were stable and did not have any exacerbations. When there was a viral infection, the treatment was continued, and spirometry was performed at least 2 weeks after the resolution of the viral infection. All asthma patients underwent a skin prick test to confirm atopy; only atopic patients were included in the study. A skin prick test was conducted for common aeroallergens, including weeds, grasses, trees, fungi, house dust mites, and cockroaches. A skin prick test was considered positive when the wheal size was more than 3 mm larger than the saline control. Only patients who were atopic were included in the study. The controls had no asthma symptoms or known family history of asthma and had normal pre-bronchodilator spirometry. The controls underwent only pre-bronchodilator spirometry, as many did not consent to post-bronchodilator spirometry.
2.2. Genomic DNA Isolation
The venous blood samples were drawn from the asthma cases and control patients using the venipuncture method. The drawn blood samples were collected in EDTA BD-Vacutainer® PLUS blood collection tubes (Becton Dickinson Ltd., Franklin Lakes, NJ, USA). The Wizard genomic DNA isolation kit of the Promega Company was used to isolate genomic DNA according to the manufacturer’s protocol.
2.3. SNP Genotyping
SNP genotyping in the samples was carried out using the Sequenom-MassARRAY platform from Xcelris Genomics Company, Ahmedabad, India. The Sequenom-iPLEX® Gold SNP genotyping platform along with Spectro CHIP was used for the MassARRAY method, and the analysis was performed with MALDI-TOF MS.
2.4. Analysis of SNPs of the ADAM33 Gene
This study was performed on six SNPs of ADAM33 selected based on previous studies and our pilot study [17,24,25,26]. The SNPs and the primers used for SNP genotyping alongside the mass of unextended (E1) and extended (E2) primers in daltons are listed in Table S1.
2.5. Statistical Analysis
The clinical parameters of the study population are reported as mean ± SE. The association between variants of ADAM33 SNPs and asthma was examined using appropriate statistical tests. SPSS software V.19 (SPSS Inc. Chicago, IL, USA) was used to test the association between clinical variables and SNPs. Power analysis was carried out to calculate the power of the study using Quanto software.
The SNP haplotype and disease associations were tested using haploview software (V.4.0) (https://www.broadinstitute.org/haploview/haploview, accessed on 22 November 2017). The software’s case/control option was used to test the associations, and the individuals with >50% missing phenotype were excluded. The >1.0% linked haplotype option was considered to create haplotype blocks. The alluvial chart was generated with Microsoft Excel®, and the forest plot was generated using GraphPad Prism software (GraphPad Software Inc., San Diego, CA, USA).
3. Results
3.1. Demographic and Clinical Profiles of the Study Population
The demographic and clinical profiles of the study patients are summarized in Table 1. Of the asthma patients, 32.8% presented with severe persistent, 42.3% with moderate persistent, and 24.9% with mild persistent asthma (GINA guidelines). The pulmonary function test results for the cases and controls are summarized in Table 2. Asthma patients showed mean FVC, FEV1, FEV1/FVC%, and PEF (L/s) values lowers than those of the controls. However, lung function within the asthma patients improved after using a bronchodilator.

Table 1.
Demographic profile of the study patients.

Table 2.
Lung function profile of study patients.
3.2. Improvement in Lung Function in Various Severity Groups upon ICS and LABA Treatment
Asthma patients were treated for three months with inhaled corticosteroids and long-acting beta-agonist. The improvement in their lung function after treatment for three months with ICS+LABA was grouped based on the highest FEV1 achieved as normal (FEV1 > 80%); partially responsive (some improvement, FEV1 60–80%); or poorly responsive (some improvement, but FEV1 still < 60%). We observed that at baseline, 24.9% of the asthma patients had an FEV1 > 80, 42.3% between 60 and 80, and 32.8% < 60; after three months of treatment, we observed that 41.6% of the asthma patients had an FEV1 > 80, 37.2% between 60 and 80, and 21.2% < 60. The improvements in the asthma patients over the course of three months of treatment are depicted in Figure S1. The duration of asthma (asthma duration < 5 years versus > 5 years) did not seem to influence treatment responsiveness (chi-square 0.18; p-value 0.66). A small but important sub-group of patients had worse lung functions than at baseline. A detailed evaluation of the reasons was not performed. Possible reasons include non-adherence, exposure to environmental triggers, or post-viral bronchial hyperresponsiveness.
3.3. ADAM33 SNP Distribution in the Study Population
Table S2 summarizes the genotypic and allelic frequency of six SNPs in the asthma patients and controls. The Hardy–Weinberg equilibrium (HWE) was calculated for both cases and controls, showing no deviation in any SNPs except in the control group for rs2280091. The genotype and allele frequency distribution showed a significant association between the TT genotype and asthma for SNP rs2853209 (p-value 0.018; odds ratio [95% CI] 0.61 [0.40–0.92]). The minor allele ‘T’ of the same SNP rs2853209 showed a significant association with asthma (p-value of 0.035; odds ratio [95% CI] 0.80 [0.65–0.98]), with the minor allele seeming to have a protective effect against asthma (Table S2). The remaining SNPs showed no significant differences between the three asthma severity groups. The table also represents the sample size and odds ratio of the ADAM33 SNPs. The power analysis for six SNPs showed a relative risk ratio of 1.33 to 1.49 at a power of 80% with a significance of p < 0.05.
3.4. ADAM33 SNP Association with Disease Severity
We analyzed the genotype and allele frequency distribution of ADAM33 SNPs among the asthma patients classified as having severe persistent, moderate persistent, and mild persistent cases. We also calculated the odds ratios of the cases (based on severity) vs. the control. The genotype and allele frequency distribution showed an association for SNP rs2853209 between the TT genotype and moderate persistent asthma, with a significant p-value of 0.001, as well as the minor allele ‘T’, with a significant p-value of 0.002; the minor allele seemed to have a protective effect against moderate persistent asthma but not severe persistent asthma (Table S3).
3.5. ADAM33 SNP Association with Lung Function Improvement in Asthma Patients upon ICS and LABA Treatment
We next examined the ADAM33 SNPs and their association with lung function (FVC and FEV1). The SNP rs2787094 (CC) showed significantly lower baseline FVC and FEV1 values, with p-values of 0.043 and 0.038. The SNP rs3918396 (AA) also showed lower baseline FVC and FEV1 values; no significant difference was observed due to the lower number of patients (Table 3 and Table 4). We went on to examine whether the ADAM33 SNPs were associated with a healthy outcome, in terms of improved lung function and treatment responsiveness, in the asthma patients treated for three months with ICS+LABA. The results are summarized in Table 3 and Table 4. Most of the genotypes showed an improvement in FVC of around 5%, and the FEV1 improvement ranged from 4.4% to 10.2%. The SNP rs2787094 (CC genotype) showed a significantly lower improvement in both FVC and FEV1, with p-values of 0.048 and 0.049. The SNP rs2853209 (TT genotype) showed a significantly lower improvement in FEV1 (4.6%) as compared to the AA genotype, which showed a greater improvement in FEV1 (10.2%) after three months of ICS+LABA. The SNP rs3918396 (AA genotype) also showed a lower improvement in FVC, but this was not statistically significant due to the fewer patients with that polymorphism.

Table 3.
Predicted baseline FVC (%) and FVC (%) improvements after three months of inhaled corticosteroids (ICS) and long-acting beta-agonists (LABAs) with various SNPs of ADAM33 in asthma patients.

Table 4.
Predicted baseline FEV1 (%) and FEV1 (%) improvements after three months of inhaled corticosteroids (ICS) and long-acting beta-agonists (LABAs) with various SNPs of ADAM33 in asthma patients.
3.6. Haplotype Association of ADAM33 SNPs with Asthma
The association between haplotypes of ADAM33 SNPs and asthma was obtained through a six-marker haplotype analysis using the haploview tool (Table S4). Of the six markers analyzed, two markers, rs2853209 and rs3918396, which are adjacent to each other, showed a significant association with asthma: haplotype block AG, odds ratio 0.50 (0.46–0.55); haplotype block TG, odds ratio 0.38 (95% CI: 0.33–0.42); and haplotype block AA, odds ratio 0.12 (0.09–0.15). Information on the blocks, haplotypes, and case–control frequencies is presented in Figure 1 and Tables S4 and S5.
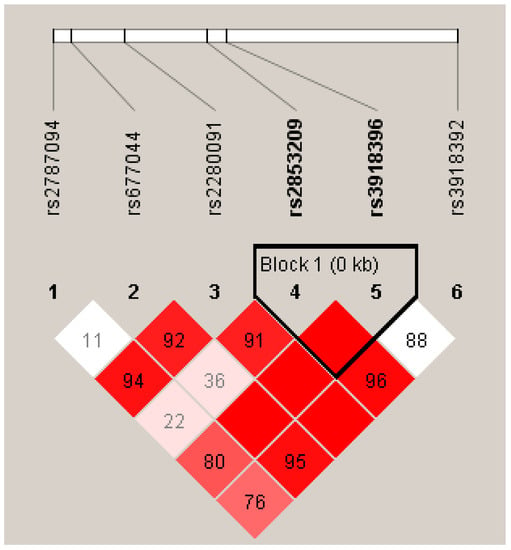
Figure 1.
The haplotype block of ADAM33 SNPs rs2853209 and rs3918396 was significantly associated with asthma. Color gradient description: The red blocks indicate strong linkage disequilibrium and white blocks indicate low linkage disequilibrium.
4. Discussion
ADAM33, a metalloproteinase enzyme, plays a significant role in airway structural abnormalities [27], being associated with changes such as epithelial damage, the hyperplasia of smooth muscle cells, fibroblast proliferation, and matrix depositions [28,29]. ADAM33 has been shown to be associated with these mechanisms [30]. Hence, the aim of this study was to identify the association between ADAM33 polymorphisms and asthma, disease severity, and treatment responsiveness to ICS+LABA. We observed that the ADAM33 SNP rs2853209 minor allele (T) was protective against asthma. According to the sub-group analysis, this effect was limited to moderate persistent asthma. The SNP rs2787094 (CC) was associated with significantly worse lung function (FVC and FEV1) at baseline as well as poor treatment responsiveness after three months of ICS+LABA. The SNP rs3918396 (AA) was also associated with lower baseline FVC and FEV1 values and poor treatment responsiveness, but this relationship was not statistically significant. The TT genotype of the SNP rs2853209 was associated with a significantly greater improvement in FVC and FEV1 after ICS+LABA. The GG genotype of the SNP rs3918392 demonstrated an even greater improvement in FEV1, but the association was not statistically significant. To the best of our knowledge, this is the first study to investigate the association between ADAM33 polymorphisms and treatment outcomes after three months of ICS+LABA.
Identifying the genetic variants predisposing an individual to acquire complex diseases is the primary purpose of contemporary human genetics. Case–control studies are the most widely accepted method for examining the association between any risk factor and a condition. However, the critical factor for the success of such case–control studies is achieving an acceptable sample size [31,32]. It has been stated that the frequency distribution of disease-associated alleles is a consequence of the power of the sample size rather than the distribution of disease alleles [33]. A power analysis was performed to estimate the association between SNPs and sample size [34]. All six studied SNPs (rs2280091, rs2787094, rs3918396, rs677044, rs2853209, and rs3918392) showed 80% power with an acceptable statistical significance level of 0.05 and were hence considered for further analysis.
Our study showed that most of the SNPs studied were not associated with asthma at any of the three severity levels (mild, moderate, and severe persistent asthma). One SNP (rs2787094) showed a nearly significant relationship between homozygous alleles and severe persistent asthma, with a p-value of 0.093. The same SNP was associated with mild persistent asthma in a North Indian population and a study on the Madeira island population [35,36]. An intronic SNP, rs2853209, showed a highly significant association between the minor homozygous genotype (TT) and asthma (p-value 0.001), which was limited to moderate persistent asthma (p-value 0.002) and not mild or severe persistent asthma. Though this SNP has been studied in other populations, it did not show any significant association with asthma in other populations [37].
Pharmacogenomics has the potential to help clinicians design personalized treatments for individual asthma patients, which may enhance the effectiveness of treatments. Different asthma patients with a similar disease severity treated with the same medication may respond differently. We observed that the percentage of non-responders or partial responders to treatment among the moderate and severe asthma cases was quite high. After excluding some non-genetic causes, such as adherence, and psychological and environmental factors, one probable reason for this variability is genetic diversity, including SNPs. In recent decades, the use of ICS+LABA has been the gold standard in asthma management, improving health status and quality of life, decreasing exacerbations, and improving lung function [38,39]. However, these therapies have variable outcomes and variable treatment responses, as observed in clinical studies. Few earlier studies have focused on therapy and the response to inhaled ICS+LABA in patients with asthma in relation to different polymorphisms. Only SNPs of the Arg16Gly and TBX21 genes have been studied to date [40,41]. No previous studies have been conducted on ADAM33 gene polymorphisms and response to treatment.
Besides playing an important role in health, ADAM33 also plays an important role in disease. ADAM33 has been associated with various asthma characteristics, such as bronchial hyperreactivity [42,43], disease progression [44], airway remodeling [28,45], worse lung function [46], and accelerated lung function decline [45]. Consequently, it is possible that the response to therapy is genetically complex [47]. After three months of ICS+LABA treatment, most of the asthma patients with different polymorphisms showed an improvement in FEV1 of 4.4% to 10.2%. One of the SNPs, the TT genotype of rs2853209, was associated with significantly higher treatment responsiveness in patients with asthma.
In contrast, the GG genotype of rs3918392 was associated with an even higher improvement in FEV1, but this was not statistically significant due to the smaller number of patients. In the case of the SNP rs2787094, a significantly lower (p-value 0.0001) improvement in both FVC and FEV1 was observed. The rs3918396 SNP (AA genotype) also showed a relatively lower improvement in FVC and FEV1, but this was not statistically significant due to the fewer patients with that polymorphism. The association between ADAM33 SNPs and spirometric parameters indicated the involvement of genetic factors in determining therapeutic response. rs2853209 is an intronic SNP, and rs2787094 is a 3′-UTR variant, wherein the 3′-UTR position affects the mRNA translation regulation [48]. Our study found an association between failure to improve FEV1 after a follow-up of three months with ICS+LABA and the SNP rs2787094 minor genotype variant “CC”, which is a probable factor influencing poor treatment responsiveness. This study could not further delineate the exact reasons for the poor treatment responsiveness and ADAM33 polymorphisms. Possibilities include structural changes in the airway that limited the improvement in FEV1 with ICS+LABA. Another possibility is that these patients had small lungs, since ADAM33 is also important for embryonic lung development, and no further improvement is possible. The Indian population has worse lung function than the Caucasian population, and the possible reasons for this are varied, including antenatal factors, nutrition during childhood, the prevalence of indoor and outdoor air pollution in India, and genetic polymorphisms such as ADAM33 [49]. Another possibility could be the influence of polymorphisms of the ADAM33 gene that may lead to the varied expression of ADAM33, which may affect smooth muscle function and impact the response to asthma treatment. There is a need for future studies on both longitudinal lung development in children and treatment responsiveness with various alternatives including biologics and ADAM33 polymorphisms in different ethnic populations across the world.
Every individual’s genotype includes multiple closely linked SNPs, referred to as haplotypes. Each individual’s genome consists of two haplotype alleles, each from one parent [50]. Haplotypes encompass the polymorphisms, variations, and markers on the same chromosome or gene that are inherited together, with a significantly lower or no chance of recombination, since they are located close to each other. Haplotypes are analyzed for two main reasons. Primarily, haplotype alleles may exist in a closer linkage disequilibrium with causal variants compared to single measured SNPs. The other reason is that the haplotypes can be causal variants of significance by themselves [51]. Many computational algorithms are available to measure haplotype frequency and predict haplotype phases from unrelated individuals’ genotype data [52,53,54]. Common haplotype frequencies can be evaluated based on the known marker phenotypes in unrelated individuals of a population [55]. The association between haplotype and asthma in our study was analyzed using haploview software, similarly to other studies [56,57,58]. A study from North India showed a haplotype association between ADAM33 and asthma, but the SNPs they studied did not include the SNPs rs2853209 and rs3918396 [59] evaluated in this study. In the present study, the rs2853209 and rs3918396 SNPs were associated with asthma. We observed a T-int value of 178.95, with a T-int value greater than 100 denoting that the genomic variants have a tendency to be inherited together. Among the studied SNPs, only these two SNPs were found to be associated significantly with asthma via haplotype analysis. The combination of rs2853209 and rs3918396 has not been observed to be significantly associated with asthma in previous studies. There is a need for the further evaluation of these SNPs and haplotypes in multiple populations and ethnicities to confirm their relevance in the global general population.
The review of previous studies on the six SNP polymorphisms of ADAM33 evaluated in this study is presented in Figure 2. Many of the studies did not observe a significant difference between various ADAM33 polymorphisms and asthma. Few studies found a strong association between different polymorphisms and asthma. The study of Su et al. [60] (China) observed that patients with the rs2280091 (T1) genotype AG had 3.19 greater odds and those with the genotype GG 8.28 greater odds of suffering from asthma. Awasthi et al. [61] (India) observed a weaker association, with the rs2280091 (T1) genotype AG having 1.6 greater odds and the genotype GG 2.8 greater odds of suffering asthma. For the ADAM33 SNP rs2787094 (V4), Su et al. [60] (China) observed that patients with the CG and GG genotypes had 2.82 and 10.28 greater odds of suffering asthma. Khadim et al. [62] (Iraq) observed a similarly strong association between the genotypes CG and GG and asthma, presenting 4.5 and 10.6 greater odds, respectively. Sinha et al. [63] (India) observed a weaker association between the CG and GG genotypes and asthma, with 1.40 and 2.10 greater odds, respectively. Xue et al. [64] (China) and Tripathi et al., [59] (India) observed 1.91 and 3.3 higher odds for the GG genotype, but the CG genotype was not significantly associated with asthma. Awasthi et al. [61] (India) observed that patients with the ADAM33 SNP rs3918396 (S1) genotypes AG and GG had a significant association with asthma, presenting 2.1 and 3.6 greater odds, respectively. For the ADAM33 SNP rs677044 (V3), Xue et al. [64] (China) observed that patients with the CT and CC genotypes had 1.51 and 2.77 greater odds of suffering from asthma. For the ADAM33 SNP rs3918392 (F1), Bukvic et al. [65] (Croatia) observed that patients with the GG and GA genotypes had 2.03 greater odds of suffering asthma (Figure 2).
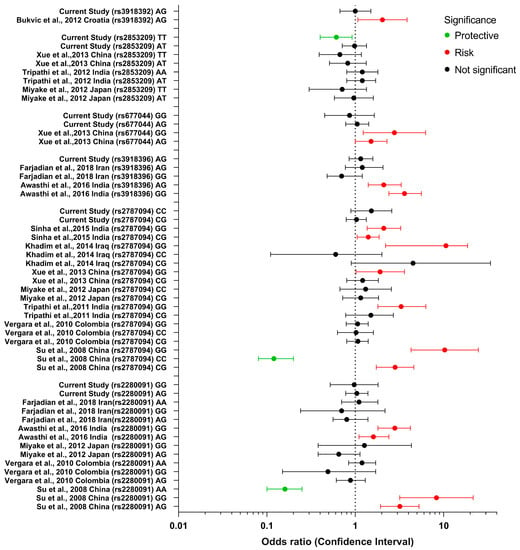
Figure 2.
Forest plot of ADAM33 SNPs genotypes rs2280091, rs2787094, rs3918396, rs677044, rs2853209, rs3918392 and their association with asthma according to various studies conducted globally [56,59,60,61,62,63,64,65,66,67,68].
Strength and Limitations
To our knowledge, this is the first study examining the relationship between ADAM33 polymorphisms and treatment responsiveness. Our study contained a large number of patients, including both cases and controls. We studied not only asthma but the severity of asthma and treatment responsiveness. The main limitation of this study was that it followed a single-center study design. We did not examine the effects of other ADAM33 polymorphisms that may be relevant for asthma. We were not able to perform post-bronchodilator spirometry on controls. Neither could we perform in vitro functional experiments to assess the potential role of the studied polymorphisms and their influence on treatment responsiveness in asthma patients.
5. Conclusions
This study indicated that the ADAM33 SNP rs2853209 is associated with asthma and treatment responsiveness, with the minor allele conferring improved treatment responsiveness to steroids. Contrastingly, the minor allele of the SNP rs2787094 had poor treatment responsiveness. According to the haplotype analysis, rs2853209 and rs3918396, which are adjacent to each other, showed a significant association with asthma.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/diagnostics13030405/s1, Table S1: Unextended primer (UEP) sequences used for massARRAY analysis along with the extended (E1/E2) alleles and mass in daltons used for the genotyping of ADAM33 variants in controls and asthma patients, Table S2: Hardy–Weinberg equilibrium (HWE) test, genotype, and minor allele frequency (MAF) of ADAM33 in healthy controls (C) and asthma patients (P), Table S3: Genotype and allele frequency distribution of ADAM33 SNPs among controls (C) and asthma patients with severe (Se), moderate (Mo), and mild (Mi) asthma, Table S4: Haplotype analysis of six SNPs of ADAM33, Table S5: Details of significant associations between haplotype SNPs of ADAM33 and controls and asthma patients in the study population, Figure S1: Alluvial chart depicting the changes in FEV1 of asthma patients after three months of treatment. FEV1: forced expiratory volume in one minute.
Author Contributions
S.V., N.B.R., S.C., A.A., and P.A.M. conceptualized the research idea and created the research design. S.V., N.B.R., N.J., A.P., M.K.U., J.B.S., and A.D.H. performed the data acquisition and analysis. S.V., N.B.R., N.J., M.K.U., S.C., A.A., and P.A.M. interpreted the data and verified the underlying data. S.V., N.B.R., N.J., A.P., M.K.U., J.B.S., and A.D.H. drafted the manuscript’s intellectual content. S.C., A.A., and P.A.M. critically revised the manuscript’s intellectual content. S.V., N.B.R., A.P., M.K.U., S.C., A.A., and P.A.M. managed the submission process. All authors had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. All authors have read and agreed to the published version of the manuscript.
Funding
This research received funding from the Indian Council of Medical Research (RFC No: NCD-Ad-hoc/3/2011-12).
Institutional Review Board Statement
The Institutional Human Ethical Committee, University of Mysore approved the study (IHEC-UOM No.79 Ph.D/2012-13).
Informed Consent Statement
Informed consent was obtained from all participants.
Data Availability Statement
All data generated or analyzed during this study are included in this published article and are available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Hargreave, F.E.; Nair, P. The Definition and Diagnosis of Asthma. Clin. Exp. Allergy 2009, 39, 1652–1658. [Google Scholar] [CrossRef]
- Krishna, M.T.; Mahesh, P.A.; Vedanthan, P.K.; Mehta, V.; Moitra, S.; Christopher, D.J. The Burden of Allergic Diseases in the Indian Subcontinent: Barriers and Challenges. Lancet Glob. Health 2020, 8, e478–e479. [Google Scholar] [CrossRef]
- Parthasarathi, A.; Padukudru, S.; Rajgopal, N.; Holla, A.D.; Krishna, M.T.; Mahesh, P.A. Allergic Disease Prevalence in School Children in Bengaluru, India: A Cross-sectional Survey. Clin. Exp. Allergy 2021, 51, 955–958. [Google Scholar] [CrossRef] [PubMed]
- Haughney, J.; Price, D.; Kaplan, A.; Chrystyn, H.; Horne, R.; May, N.; Moffat, M.; Versnel, J.; Shanahan, E.R.; Hillyer, E.V.; et al. Achieving Asthma Control in Practice: Understanding the Reasons for Poor Control. Respir. Med. 2008, 102, 1681–1693. [Google Scholar] [CrossRef] [PubMed]
- Martel, M.-J.; Rey, E.; Malo, J.-L.; Perreault, S.; Beauchesne, M.-F.; Forget, A.; Blais, L. Determinants of the Incidence of Childhood Asthma: A Two-Stage Case-Control Study. Am. J. Epidemiol. 2009, 169, 195–205. [Google Scholar] [CrossRef]
- Parthasarathi, A.; Srinivas, S.; Biligere Siddaiah, J.; Anand Mahesh, P. Local Adverse Drug Reactions in Ambulatory Asthma Patients TreatedWith Inhaled Corticosteroids: An Experience from a South Indian TeachingHospital. Curr. Respir. Med. Rev. 2022, 18, 217–227. [Google Scholar] [CrossRef]
- Vercelli, D. Discovering Susceptibility Genes for Asthma and Allergy. Nat. Rev. Immunol. 2008, 8, 169–182. [Google Scholar] [CrossRef]
- Bijanzadeh, M.; Mahesh, P.A.; Ramachandra, N.B. An Understanding of the Genetic Basis of Asthma. Indian J. Med. Res. 2011, 134, 149–161. [Google Scholar]
- Ober, C. Genome-Wide Search for Asthma Susceptibility Loci in a Founder Population. The Collaborative Study on the Genetics of Asthma. Hum. Mol. Genet. 1998, 7, 1393–1398. [Google Scholar] [CrossRef]
- Teerlink, C.C.; Camp, N.J.; Bansal, A.; Crapo, R.; Hughes, D.; Kort, E.; Rowe, K.; Cannon-Albright, L.A. Significant Evidence for Linkage to Chromosome 5q13 in a Genome-Wide Scan for Asthma in an Extended Pedigree Resource. Eur. J. Hum. Genet. 2009, 17, 636–643. [Google Scholar] [CrossRef]
- Meng, J.-F.; Rosenwasser, L.J. Unraveling the Genetic Basis of Asthma and Allergic Diseases. Allergy Asthma Immunol. Res. 2010, 2, 215–227. [Google Scholar] [CrossRef]
- Bergeron, C.; Al-Ramli, W.; Hamid, Q. Remodeling in Asthma. Proc. Am. Thorac. Soc. 2009, 6, 301–305. [Google Scholar] [CrossRef]
- Davies, D.E. The Role of the Epithelium in Airway Remodeling in Asthma. Proc. Am. Thorac. Soc. 2009, 6, 678–682. [Google Scholar] [CrossRef] [PubMed]
- Hough, K.P.; Curtiss, M.L.; Blain, T.J.; Liu, R.-M.; Trevor, J.; Deshane, J.S.; Thannickal, V.J. Airway Remodeling in Asthma. Front. Med. 2020, 7, 191. [Google Scholar] [CrossRef] [PubMed]
- Chakir, J.; Shannon, J.; Molet, S.; Fukakusa, M.; Elias, J.; Laviolette, M.; Boulet, L.-P.; Hamid, Q. Airway Remodeling-Associated Mediators in Moderate to Severe Asthma: Effect of Steroids on TGF-β, IL-11, IL-17, and Type I and Type III Collagen Expression. J. Allergy Clin. Immunol. 2003, 111, 1293–1298. [Google Scholar] [CrossRef]
- Grainge, C.L.; Lau, L.C.K.; Ward, J.A.; Dulay, V.; Lahiff, G.; Wilson, S.; Holgate, S.; Davies, D.E.; Howarth, P.H. Effect of Bronchoconstriction on Airway Remodeling in Asthma. N. Engl. J. Med. 2011, 364, 2006–2015. [Google Scholar] [CrossRef] [PubMed]
- Van Eerdewegh, P.; Little, R.D.; Dupuis, J.; Del Mastro, R.G.; Falls, K.; Simon, J.; Torrey, D.; Pandit, S.; McKenny, J.; Braunschweiger, K.; et al. Association of the ADAM33 Gene with Asthma and Bronchial Hyperresponsiveness. Nature 2002, 418, 426–430. [Google Scholar] [CrossRef]
- Davies, E.R.; Kelly, J.F.C.; Howarth, P.H.; Wilson, D.I.; Holgate, S.T.; Davies, D.E.; Whitsett, J.A.; Haitchi, H.M. Soluble ADAM33 Initiates Airway Remodeling to Promote Susceptibility for Allergic Asthma in Early Life. JCI Insight 2016, 1. [Google Scholar] [CrossRef]
- Durrani, S.R.; Viswanathan, R.K.; Busse, W.W. What Effect Does Asthma Treatment Have on Airway Remodeling? Current Perspectives. J. Allergy Clin. Immunol. 2011, 128, 439–448. [Google Scholar] [CrossRef]
- Oppenheimer, J. Role of Genetic Polymorphisms in Therapeutic Response to Anti-Asthma Therapy. Allergy Asthma Clin. Immunol. 2007, 3, 50. [Google Scholar] [CrossRef][Green Version]
- Berger, W. New Approaches to Managing Asthma: A US Perspective. Ther. Clin. Risk Manag. 2008, 4, 363–379. [Google Scholar] [CrossRef]
- Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention. 2012. p. 225. Available online: Www.Ginasthma.Org (accessed on 20 September 2022).
- Miller, M.R. Standardisation of Spirometry. Eur. Respir. J. 2005, 26, 319–338. [Google Scholar] [CrossRef] [PubMed]
- Vishweswaraiah, S.; Ramachandra, N.; Jayaraj, B.; Holla, A.; Chakraborty, S.; Agrawal, A.; Mahesh, P. Haplotype Analysis of ADAM33 Polymorphisms in Asthma: A Pilot Study. Indian J. Med. Res. 2019, 150, 272. [Google Scholar] [CrossRef] [PubMed]
- Holgate, S.T. Is Big Beautiful? The Continuing Story of ADAM33 and Asthma. Thorax 2005, 60, 263–264. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bijanzadeh, M.; Ramachandra, N.B.; Mahesh, P.A.; Savitha Mysore, R.; Kumar, P.; Manjunath, B.S.; Jayaraj, B.S. Association of IL-4 and ADAM33 Gene Polymorphisms with Asthma in an Indian Population. Lung 2010, 188, 415–422. [Google Scholar] [CrossRef]
- Holgate, S.T.; Davies, D.E.; Rorke, S.; Cakebread, J.; Murphy, G.; Powell, R.M.; Holloway, J.W. ADAM 33 and Its Association with Airway Remodeling and Hyperresponsiveness in Asthma. Clin. Rev. Allergy Immunol. 2004, 27, 023–034. [Google Scholar] [CrossRef] [PubMed]
- Kedda, M.-A.; Duffy, D.L.; Bradley, B.; O’Hehir, R.E.; Thompson, P.J. ADAM33 Haplotypes Are Associated with Asthma in a Large Australian Population. Eur. J. Hum. Genet. 2006, 14, 1027–1036. [Google Scholar] [CrossRef] [PubMed]
- Vishweswaraiah, S.; Veerappa, A.M.; Mahesh, P.A.; Jayaraju, B.S.; Krishnarao, C.S.; Ramachandra, N.B. Molecular Interaction Network and Pathway Studies of ADAM33 Potentially Relevant to Asthma. Ann. Allergy Asthma. Immunol. 2014, 113, 418–424.e1. [Google Scholar] [CrossRef]
- Mahesh, P.A. Unravelling the Role of ADAM 33 in Asthma. Indian J. Med. Res. 2013, 137, 447–450. [Google Scholar] [PubMed]
- Gauderman, W.J. Sample Size Requirements for Matched Case-Control Studies of Gene-Environment Interaction. Stat. Med. 2002, 21, 35–50. [Google Scholar] [CrossRef]
- Lloyd-Jones, G. Design and Control Issues in Qualitative Case Study Research. Int. J. Qual. Methods 2003, 2, 33–42. [Google Scholar] [CrossRef]
- Iles, M.M. What Can Genome-Wide Association Studies Tell Us about the Genetics of Common Disease? PLoS Genet. 2008, 4, e33. [Google Scholar] [CrossRef] [PubMed]
- Gauderman, W.J.; Murcray, C.; Gilliland, F.; Conti, D.V. Testing Association between Disease and Multiple SNPs in a Candidate Gene. Genet. Epidemiol. 2007, 31, 383–395. [Google Scholar] [CrossRef] [PubMed]
- Awasthi, S.; Tripathi, P.; Ganesh, S.; Husain, N. Association of ADAM33 Gene Polymorphisms with Asthma in Indian Children. J. Hum. Genet. 2011, 56, 188–195. [Google Scholar] [CrossRef]
- Berenguer, A.; Fernandes, A.; Oliveira, S.; Rodrigues, M.; Ornelas, P.; Romeira, D.; Serrão, T.; Rosa, A.; Câmara, R. Genetic Polymorphisms and Asthma: Findings from a Case–Control Study in the Madeira Island Population. Biol. Res. 2014, 47, 40. [Google Scholar] [CrossRef] [PubMed]
- Thongngarm, T.; Jameekornrak, A.; Limwongse, C.; Sangasapaviliya, A.; Jirapongsananuruk, O.; Assawamakin, A.; Chaiyaratana, N.; Luangwedchakarn, V.; Thongnoppakhun, W. Association between ADAM33 Polymorphisms and Asthma in a Thai Population. Asian Pac. J. Allergy Immunol. 2008, 26, 205–211. [Google Scholar]
- Van Den Berge, M.; Ten Hacken, N.; Kerstjens, H.; Postma, D. Management of Asthma with ICS and LABAs: Different Treatment Strategies. Clin. Med. Ther. 2009, 1, CMT.S2283. [Google Scholar] [CrossRef]
- Tamm, M.; Richards, D.H.; Beghé, B.; Fabbri, L. Inhaled Corticosteroid and Long-Acting Β2-Agonist Pharmacological Profiles: Effective Asthma Therapy in Practice. Respir. Med. 2012, 106, S9–S19. [Google Scholar] [CrossRef] [PubMed]
- Lopert, A.; Rijavec, M.; Žavbi, M.; Korošec, P.; Fležar, M. Asthma Treatment Outcome in Adults Is Associated with Rs9910408 in TBX21 Gene. Sci. Rep. 2013, 3, 2915. [Google Scholar] [CrossRef]
- Qiu, Y.; Zhang, D.; Qin, Y.; Yin, K.-S. Effect of Β2 -Adrenergic Receptor Gene Arg16Gly Polymorphisms on Response to Long-Acting Β2-Agonist in Chinese Han Asthmatic Patients. Multidiscip. Respir. Med. 2014, 9, 22. [Google Scholar] [CrossRef]
- Holgate, S.T. ADAM 33: Just Another Asthma Gene or a Breakthrough in Understanding the Origins of Bronchial Hyperresponsiveness? Thorax 2003, 58, 466–469. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lee, J.H.; Park, H.-S.; Park, S.W.; Jang, A.S.; Uh, S.T.; Rhim, T.; Park, C.-S.; Hong, S.-J.; Holgate, S.T.; Holloway, J.W.; et al. ADAM33 Polymorphism: Association with Bronchial Hyper-Responsiveness in Korean Asthmatics. Clin. Htmlent Glyphamp Asciiamp Exp. Allergy 2004, 34, 860–865. [Google Scholar] [CrossRef] [PubMed]
- Blakey, J.D.; Sayers, I.; Ring, S.M.; Strachan, D.P.; Hall, I.P. Positionally Cloned Asthma Susceptibility Gene Polymorphisms and Disease Risk in the British 1958 Birth Cohort. Thorax 2009, 64, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Jongepier, H.; Boezen, H.M.; Dijkstra, A.; Howard, T.D.; Vonk, J.M.; Koppelman, G.H.; Zheng, S.L.; Meyers, D.A.; Bleecker, E.R.; Postma, D.S. Polymorphisms of the ADAM33 Gene Are Associated with Accelerated Lung Function Decline in Asthma. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2004, 34, 757–760. [Google Scholar] [CrossRef]
- Lee, J.-Y.; Park, S.-W.; Chang, H.K.; Kim, H.Y.; Rhim, T.; Lee, J.-H.; Jang, A.-S.; Koh, E.-S.; Park, C.-S. A Disintegrin and Metalloproteinase 33 Protein in Patients with Asthma: Relevance to Airflow Limitation. Am. J. Respir. Crit. Care Med. 2006, 173, 729–735. [Google Scholar] [CrossRef]
- Drazen, J.M.; Silverman, E.K.; Lee, T.H. Heterogeneity of Therapeutic Responses in Asthma. Br. Med. Bull. 2000, 56, 1054–1070. [Google Scholar] [CrossRef]
- Szostak, E.; Gebauer, F. Translational Control by 3’-UTR-Binding Proteins. Brief. Funct. Genom. 2013, 12, 58–65. [Google Scholar] [CrossRef]
- Abdullah, N.; Borhanuddin, B.; Shah, S.A.; Hassan, T.; Jamal, R. Global Lung Initiative 2012 Spirometry Reference Values in a Large Asian Cohort of Malay, Chinese and Indian Ancestry. Respirology 2018, 23, 1173–1179. [Google Scholar] [CrossRef]
- Balding, D.J. A Tutorial on Statistical Methods for Population Association Studies. Nat. Rev. Genet. 2006, 7, 781–791. [Google Scholar] [CrossRef]
- Stram, D.O.; Seshan, V.E. Multi-SNP Haplotype Analysis Methods for Association Analysis. In Statistical Human Genetics; Elston, R.C., Satagopan, J.M., Sun, S., Eds.; Methods in Molecular Biology; Humana Press: Totowa, NJ, USA, 2012; Volume 850, pp. 423–452. ISBN 978-1-61779-554-1. [Google Scholar]
- Tishkoff, S.A.; Pakstis, A.J.; Ruano, G.; Kidd, K.K. The Accuracy of Statistical Methods for Estimation of Haplotype Frequencies: An Example from the CD4 Locus. Am. J. Hum. Genet. 2000, 67, 518–522. [Google Scholar] [CrossRef]
- Xu, C.-F.; Lewis, K.; Cantone, K.L.; Khan, P.; Donnelly, C.; White, N.; Crocker, N.; Boyd, P.R.; Zaykin, D.V.; Purvis, I.J. Effectiveness of Computational Methods in Haplotype Prediction. Hum. Genet. 2002, 110, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Halldórsson, B.V.; Bafna, V.; Edwards, N.; Lippert, R.; Yooseph, S.; Istrail, S. Combinatorial Problems Arising in SNP and Haplotype Analysis. In Discrete Mathematics and Theoretical Computer Science; Calude, C.S., Dinneen, M.J., Vajnovszki, V., Eds.; Lecture Notes in Computer Science; Springer Berlin Heidelberg: Berlin/Heidelberg, Germany, 2003; Volume 273, pp. 26–47. ISBN 978-3-540-40505-4. [Google Scholar]
- McKeigue, P.M. Efficiency of Estimation of Haplotype Frequencies: Use of Marker Phenotypes of Unrelated Individuals versus Counting of Phase-Known Gametes. Am. J. Hum. Genet. 2000, 67, 1626–1627. [Google Scholar] [CrossRef] [PubMed]
- Vergara, C.I.; Acevedo, N.; Jiménez, S.; Martínez, B.; Mercado, D.; Gusmão, L.; Barnes, K.C.; Caraballo, L. A Six-SNP Haplotype of ADAM33 Is Associated with Asthma in a Population of Cartagena, Colombia. Int. Arch. Allergy Immunol. 2010, 152, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Bush, W.S.; Moore, J.H. Chapter 11: Genome-Wide Association Studies. PLoS Comput. Biol. 2012, 8, e1002822. [Google Scholar] [CrossRef]
- Singh, K.; Singh, V.K.; Agrawal, N.K.; Gupta, S.K.; Singh, K. Association of Toll-Like Receptor 4 Polymorphisms with Diabetic Foot Ulcers and Application of Artificial Neural Network in DFU Risk Assessment in Type 2 Diabetes Patients. BioMed Res. Int. 2013, 2013, 1–9. [Google Scholar] [CrossRef]
- Tripathi, P.; Awasthi, S.; Prasad, R.; Husain, N.; Ganesh, S. Association of ADAM33 Gene Polymorphisms with Adult-Onset Asthma and Its Severity in an Indian Adult Population. J. Genet. 2011, 90, 265–273. [Google Scholar] [CrossRef]
- Su, D.; Zhang, X.; Sui, H.; Lü, F.; Jin, L.; Zhang, J. Association of ADAM33 Gene Polymorphisms with Adult Allergic Asthma and Rhinitis in a Chinese Han Population. BMC Med. Genet. 2008, 9, 82. [Google Scholar] [CrossRef]
- Awasthi, S.; Tripathi, P.; Prasad, R.; Ganesh, S. ASSOCIATION OF A DISINTEGRIN AND METALLOPROTEASE 33 GENE POLYMORPHISMS AND THEIR HAPLOTYPES WITH ASTHMA IN THE NORTH-INDIAN POPULATION. Indian J. Med. Sci. 2016, 68, 54. [Google Scholar] [CrossRef]
- Manal, M.K.; Al-damerchi, A.; Kazaal, M.A. Evaluation of A Disintegrin and Metalloprotein33 Gene Polymorphism in Bronchial Asthma. AL-Qadisiya Med. J. 2014, 11, 9. [Google Scholar]
- Sinha, S.; Singh, J.; Kumar Jindal, S. Significant Association of ADAM33 V4C>G Polymorphisms with Asthma in a North Indian Population. Biores. Commun. 2015, 1, 62–68. Available online: https://Www.Bioresearchcommunications.Com/Index.Php/Brc/Article/View/149 (accessed on 15 October 2022).
- Xue, W.; Han, W.; Zhou, Z.-S. ADAM33 Polymorphisms Are Associated with Asthma and a Distinctive Palm Dermatoglyphic Pattern. Mol. Med. Rep. 2013, 8, 1795–1800. [Google Scholar] [CrossRef] [PubMed]
- Bukvic, B.K.; Blekic, M.; Simpson, A.; Marinho, S.; Curtin, J.A.; Hankinson, J.; Aberle, N.; Custovic, A. Asthma Severity, Polymorphisms in 20p13 and Their Interaction with Tobacco Smoke Exposure. Pediatr. Allergy Immunol. 2013, 24, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Miyake, Y.; Tanaka, K.; Arakawa, M. ADAM33 Polymorphisms, Smoking and Asthma in Japanese Women: The Kyushu Okinawa Maternal and Child Health Study. Int. J. Tuberc. Lung Dis. 2012, 16, 974–979. [Google Scholar] [CrossRef] [PubMed]
- Farjadian, S.; Moghtaderi, M.; Hoseini-Pouya, B.-A.; Ebrahimpour, A.; Nasiri, M. ADAM33 Gene Polymorphisms in Southwestern Iranian Patients with Asthma. Iran. J. Basic Med. Sci. 2018, 21, 813–817. [Google Scholar] [CrossRef]
- Tripathi, P.; Awasthi, S.; Prasad, R.; Ganesh, S. Haplotypic Association of ADAM33 (T+1, S+1 and V − 3) Gene Variants in Genetic Susceptibility to Asthma in Indian Population. Ann. Hum. Biol. 2012, 39, 479–483. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).