Pancreatic Cystic Neoplasms: Diagnosis and Management
Abstract
1. Introduction
2. Classification/Definition
2.1. Mucinous Cystic Neoplasms
2.2. Intraductal Papillary Mucinous Neoplasms
2.3. Serous Cystic Neoplasm
2.4. Solid Pseudopapillary Neoplasm
2.5. Cystic Neuroendocrine Tumors
3. Diagnosis
3.1. Cyst Fluid Analysis and Next-Generation Sequencing
3.2. Endoscopic Ultrasound-Guided Through-the-Needle Microforceps Biopsy
3.3. Needle Confocal Laser Endomicroscopy
4. Surgical Resection
4.1. Cyst Size
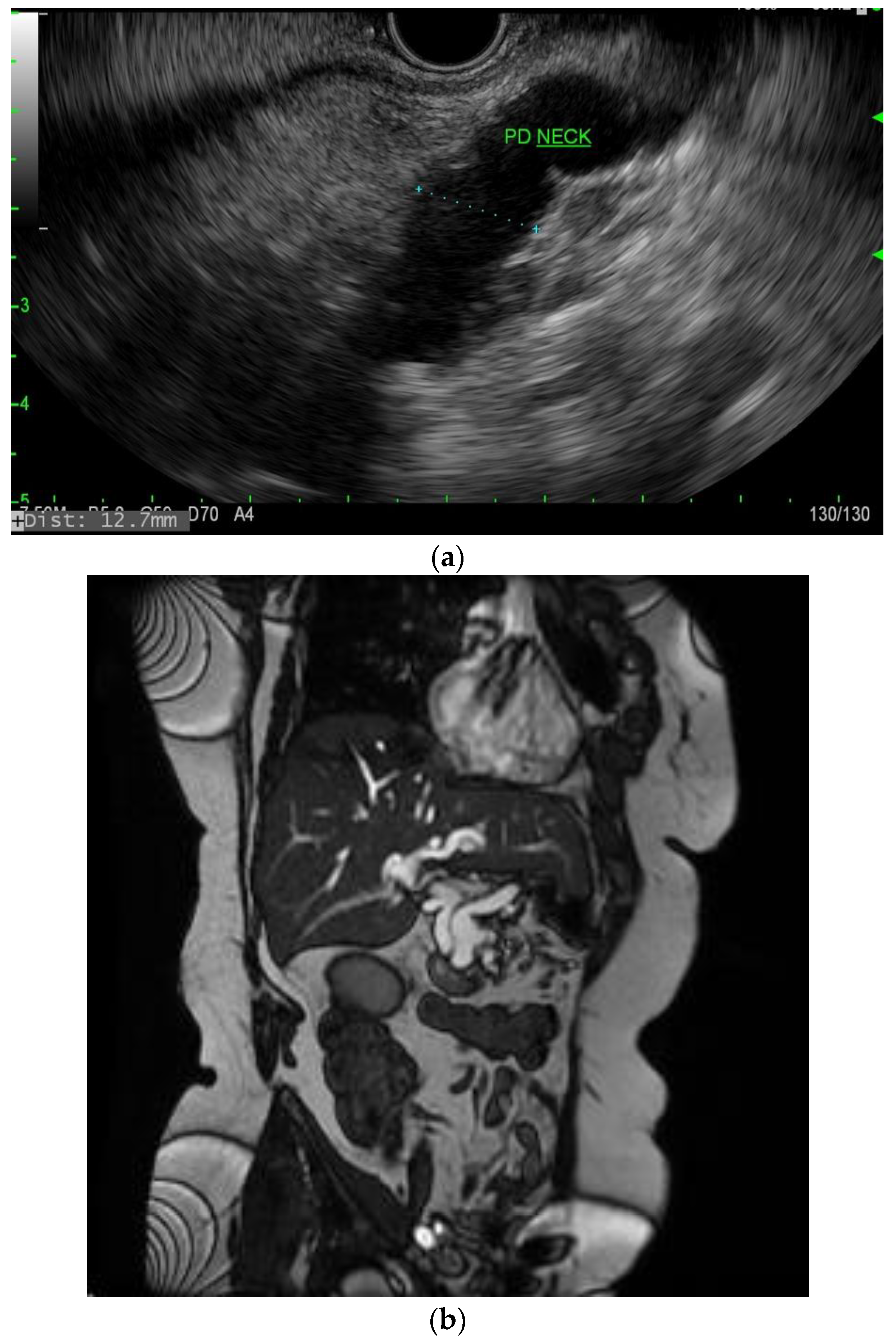
4.2. Pancreatic Ductal Dilatation
4.3. Symptoms
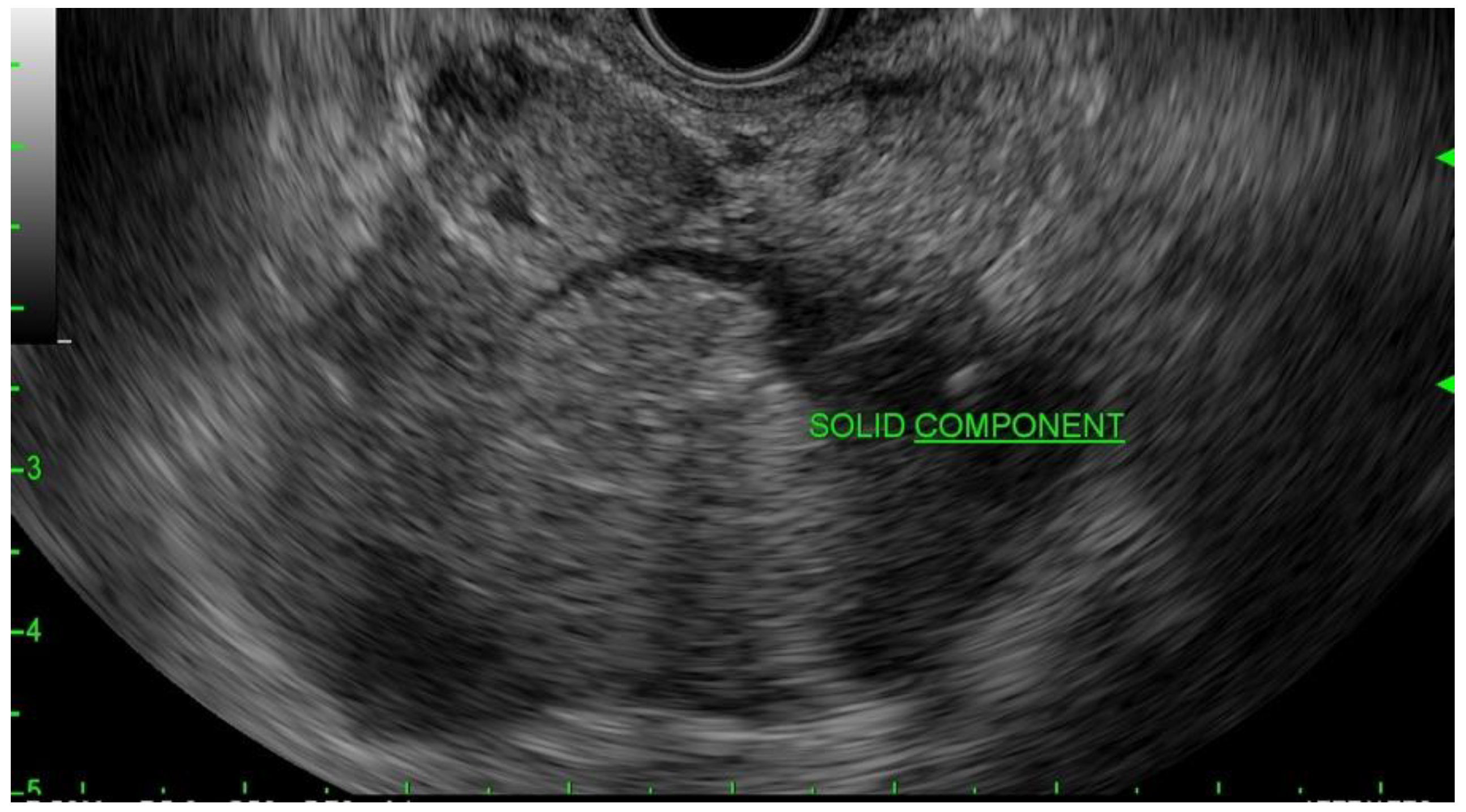
4.4. Enhancing Mural Nodule
| 2015 AGA [32] | MCN | - |
| IPMN | - | |
| 2017 IAP [30] | MCN | - |
| IPMN |
| |
| 2018 European [29] | MCN | - |
| IPMN |
| |
| 2018 ACG [10] | MCN | - |
| IPMN |
|
5. Surveillance
5.1. Active Surveillance of Non-Resected PCNs
5.2. Surveillance after Surgical Resection of PCNs
6. Future Directions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pelletier, A.L.; Hammel, P.; Rebours, V.; Couvelard, A.; Vullierme, M.-P.; Maire, F.; Hentic, O.; Aubert, A.; Sauvanet, A.; Lévy, P.; et al. Acute pancreatitis in patients operated on for intraductal papillary mucinous neoplasms of the pancreas: Frequency, severity, and clinicopathologic correlations. Pancreas 2010, 39, 658–661. [Google Scholar] [CrossRef] [PubMed]
- Ringold, D.A.; Shroff, P.; Sikka, S.K.; Ylagan, L.; Jonnalagadda, S.; Early, D.S.; Edmundowicz, S.A.; Azar, R. Pancreatitis is frequent among patients with side-branch intraductal papillary mucinous neoplasia diagnosed by EUS. Gastrointest. Endosc. 2009, 70, 488–494. [Google Scholar] [CrossRef] [PubMed]
- Canto, M.I.; Hruban, R.H.; Fishman, E.K.; Kamel, I.R.; Schulick, R.; Zhang, Z.; Topazian, M.; Takahashi, N.; Fletcher, J.; Petersen, G.; et al. Frequent Detection of Pancreatic Lesions in Asymptomatic High-Risk Individuals. Gastroenterology 2012, 142, 796–804. [Google Scholar] [CrossRef] [PubMed]
- Goh, B.K.P.; Tan, Y.-M.; Chung, Y.-F.A.; Chow, P.K.H.; Cheow, P.-C.; Wong, W.-K.; Ooi, L.L.P.J. A Review of Mucinous Cystic Neoplasms of the Pancreas Defined by Ovarian-type Stroma: Clinicopathological Features of 344 Patients. World J. Surg. 2006, 30, 2236–2245. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, M.; Suzuki, Y. Natural history of IPMN: Adenoma–carcinoma sequence in IPMN. In Pancreatic Cancer, Cystic Neoplasms and Endocrine Tumors: Diagnosis and Management; Wiley: Hoboken, NJ, USA, 2015; p. 225. [Google Scholar]
- Sahora, K.; Fernández-del Castillo, C.; Dong, F.; Marchegiani, G.; Thayer, S.; Ferrone, C.; Sahani, D.; Brugge, W.; Warshaw, A.; Lillemoe, K.; et al. Not all mixed-type intraductal papillary mucinous neoplasms behave like main-duct lesions: Implications of minimal involvement of the main pancreatic duct. Surgery 2014, 156, 611–621. [Google Scholar] [CrossRef]
- Sahora, K.; Mino-Kenudson, M.; Brugge, W.; Thayer, S.; Ferrone, C.; Sahani, D.; Pitman, M.; Warshaw, A.; Lillemoe, K.; Fernández-del Castillo, C. Branch duct intraductal papillary mucinous neoplasms: Does cyst size change the tip of the scale? A critical analysis of the revised international consensus guidelines in a large single-institutional series. Ann. Surg. 2013, 258, 466–475. [Google Scholar] [CrossRef]
- Choi, S.H.; Park, S.H.; Kim, K.W.; Lee, J.Y.; Lee, S.S. Progression of Unresected Intraductal Papillary Mucinous Neoplasms of the Pancreas to Cancer: A Systematic Review and Meta-analysis. Clin. Gastroenterol. Hepatol. 2017, 15, 1509–1520.e1504. [Google Scholar] [CrossRef]
- Zamboni, G.; Scarpa, A.; Bogina, G.; Iacono, C.; Bassi, C.; Talamini, G.; Sessa, F.; Capella, C.; Solcia, E.; Rickaert, F.; et al. Mucinous cystic tumors of the pancreas: Clinicopathological features, prognosis, and relationship to other mucinous cystic tumors. Am. J. Surg. Pathol. 1999, 23, 410–422. [Google Scholar] [CrossRef]
- Elta, G.H.; Enestvedt, B.K.; Sauer, B.G.; Lennon, A.M. ACG Clinical Guideline: Diagnosis and Management of Pancreatic Cysts. Am. J. Gastroenterol. 2018, 113, 464–479. [Google Scholar] [CrossRef]
- Reddy, R.P.; Smyrk, T.C.; Zapiach, M.; Levy, M.J.; Pearson, R.K.; Clain, J.E.; Farnell, M.B.; Sarr, M.G.; Chari, S.T. Pancreatic mucinous cystic neoplasm defined by ovarian stroma: Demographics, clinical features, and prevalence of cancer. Clin. Gastroenterol. Hepatol. 2004, 2, 1026–1031. [Google Scholar] [CrossRef]
- Park, J.W.; Jang, J.Y.; Kang, M.J.; Kwon, W.; Chang, Y.R.; Kim, S.W. Mucinous cystic neoplasm of the pancreas: Is surgical resection recommended for all surgically fit patients? Pancreatology 2014, 14, 131–136. [Google Scholar] [CrossRef]
- Baker, M.L.; Seeley, E.S.; Pai, R.; Suriawinata, A.A.; Mino-Kenudson, M.; Zamboni, G.; Klöppel, G.; Longnecker, D.S. Invasive mucinous cystic neoplasms of the pancreas. Exp. Mol. Pathol. 2012, 93, 345–349. [Google Scholar] [CrossRef]
- Crippa, S.; Salvia, R.; Warshaw, A.L.; Domínguez, I.; Bassi, C.; Falconi, M.; Thayer, S.P.; Zamboni, G.; Lauwers, G.Y.; Mino-Kenudson, M. Mucinous cystic neoplasm of the pancreas is not an aggressive entity: Lessons from 163 resected patients. Ann. Surg. 2008, 247, 571. [Google Scholar] [CrossRef]
- van Huijgevoort, N.C.M.; Del Chiaro, M.; Wolfgang, C.L.; van Hooft, J.E.; Besselink, M.G. Diagnosis and management of pancreatic cystic neoplasms: Current evidence and guidelines. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 676–689. [Google Scholar] [CrossRef]
- Capurso, G.; Boccia, S.; Salvia, R.; Del Chiaro, M.; Frulloni, L.; Arcidiacono, P.G.; Zerbi, A.; Manta, R.; Fabbri, C.; Ventrucci, M.; et al. Risk factors for intraductal papillary mucinous neoplasm (IPMN) of the pancreas: A multicentre case-control study. Am. J. Gastroenterol. 2013, 108, 1003–1009. [Google Scholar] [CrossRef]
- Stark, A.; Donahue, T.R.; Reber, H.A.; Hines, O.J. Pancreatic Cyst Disease: A Review. JAMA 2016, 315, 1882–1893. [Google Scholar] [CrossRef]
- Jais, B.; Rebours, V.; Malleo, G.; Salvia, R.; Fontana, M.; Maggino, L.; Bassi, C.; Manfredi, R.; Moran, R.; Lennon, A.M.; et al. Serous cystic neoplasm of the pancreas: A multinational study of 2622 patients under the auspices of the International Association of Pancreatology and European Pancreatic Club (European Study Group on Cystic Tumors of the Pancreas). Gut 2016, 65, 305–312. [Google Scholar] [CrossRef]
- Law, J.K.; Ahmed, A.; Singh, V.K.; Akshintala, V.S.; Olson, M.T.; Raman, S.P.; Ali, S.Z.; Fishman, E.K.; Kamel, I.; Canto, M.I.; et al. A systematic review of solid-pseudopapillary neoplasms: Are these rare lesions? Pancreas 2014, 43, 331–337. [Google Scholar] [CrossRef]
- Koh, Y.X.; Chok, A.Y.; Zheng, H.L.; Tan, C.S.; Goh, B.K. A systematic review and meta-analysis of the clinicopathologic characteristics of cystic versus solid pancreatic neuroendocrine neoplasms. Surgery 2014, 156, 83–96.e82. [Google Scholar] [CrossRef]
- Kazanjian, K.K.; Reber, H.A.; Hines, O.J. Resection of Pancreatic Neuroendocrine Tumors: Results of 70 Cases. Arch. Surg. 2006, 141, 765–770. [Google Scholar] [CrossRef]
- Waters, J.A.; Schmidt, C.M.; Pinchot, J.W.; White, P.B.; Cummings, O.W.; Pitt, H.A.; Sandrasegaran, K.; Akisik, F.; Howard, T.J.; Nakeeb, A.; et al. CT vs. MRCP: Optimal classification of IPMN type and extent. J. Gastrointest. Surg. 2008, 12, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Pilleul, F.; Rochette, A.; Partensky, C.; Scoazec, J.Y.; Bernard, P.; Valette, P.J. Preoperative evaluation of intraductal papillary mucinous tumors performed by pancreatic magnetic resonance imaging and correlated with surgical and histopathologic findings. J. Magn. Reson. Imaging. 2005, 21, 237–244. [Google Scholar] [CrossRef] [PubMed]
- de Jong, K.; van Hooft, J.E.; Nio, C.Y.; Gouma, D.J.; Dijkgraaf, M.G.; Bruno, M.J.; Fockens, P. Accuracy of preoperative workup in a prospective series of surgically resected cystic pancreatic lesions. Scand. J. Gastroenterol. 2012, 47, 1056–1063. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Eun, H.W.; Park, H.-J.; Hong, S.S.; Kim, Y.J. Diagnostic performance of MRI and EUS in the differentiation of benign from malignant pancreatic cyst and cyst communication with the main duct. Eur. J. Radiol. 2012, 81, 2927–2935. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, N.A.; Kochman, M.L.; Lewis, J.D.; Ginsberg, G.G. Can EUS alone differentiate between malignant and benign cystic lesions of the pancreas? Am. J. Gastroenterol. 2001, 96, 3295–3300. [Google Scholar] [CrossRef]
- Cho, C.S.; Russ, A.J.; Loeffler, A.G.; Rettammel, R.J.; Oudheusden, G.; Winslow, E.R.; Weber, S.M. Preoperative classification of pancreatic cystic neoplasms: The clinical significance of diagnostic inaccuracy. Ann. Surg. Oncol. 2013, 20, 3112–3119. [Google Scholar] [CrossRef]
- Keane, M.G.; Afghani, E. A Review of the Diagnosis and Management of Premalignant Pancreatic Cystic Lesions. J. Clin. Med. 2021, 10, 1284. [Google Scholar] [CrossRef]
- European evidence-based guidelines on pancreatic cystic neoplasms. Gut 2018, 67, 789–804. [CrossRef]
- Tanaka, M.; Fernández-del Castillo, C.; Kamisawa, T.; Jang, J.Y.; Levy, P.; Ohtsuka, T.; Salvia, R.; Shimizu, Y.; Tada, M.; Wolfgang, C.L. Revisions of international consensus Fukuoka guidelines for the management of IPMN of the pancreas. Pancreatology 2017, 17, 738–753. [Google Scholar] [CrossRef]
- Muthusamy, V.R.; Chandrasekhara, V.; Acosta, R.D.; Bruining, D.H.; Chathadi, K.V.; Eloubeidi, M.A.; Faulx, A.L.; Fonkalsrud, L.; Gurudu, S.R.; Khashab, M.A.; et al. The role of endoscopy in the diagnosis and treatment of cystic pancreatic neoplasms. Gastrointest. Endosc. 2016, 84, 1–9. [Google Scholar] [CrossRef]
- Vege, S.S.; Ziring, B.; Jain, R.; Moayyedi, P. American gastroenterological association institute guideline on the diagnosis and management of asymptomatic neoplastic pancreatic cysts. Gastroenterology 2015, 148, 819–822. [Google Scholar] [CrossRef]
- Oh, S.H.; Lee, J.K.; Lee, K.T.; Lee, K.H.; Woo, Y.S.; Noh, D.H. The Combination of Cyst Fluid Carcinoembryonic Antigen, Cytology and Viscosity Increases the Diagnostic Accuracy of Mucinous Pancreatic Cysts. Gut Liver 2017, 11, 283–289. [Google Scholar] [CrossRef]
- Bick, B.L.; Enders, F.T.; Levy, M.J.; Zhang, L.; Henry, M.R.; Abu Dayyeh, B.K.; Chari, S.T.; Clain, J.E.; Farnell, M.B.; Gleeson, F.C.; et al. The string sign for diagnosis of mucinous pancreatic cysts. Endoscopy 2015, 47, 626–631. [Google Scholar] [CrossRef]
- Brugge, W.R.; Lewandrowski, K.; Lee-Lewandrowski, E.; Centeno, B.A.; Szydlo, T.; Regan, S.; del Castillo, C.F.; Warshaw, A.L. Diagnosis of pancreatic cystic neoplasms: A report of the cooperative pancreatic cyst study. Gastroenterology 2004, 126, 1330–1336. [Google Scholar] [CrossRef]
- Thornton, G.D.; McPhail, M.J.; Nayagam, S.; Hewitt, M.J.; Vlavianos, P.; Monahan, K.J. Endoscopic ultrasound guided fine needle aspiration for the diagnosis of pancreatic cystic neoplasms: A meta-analysis. Pancreatology 2013, 13, 48–57. [Google Scholar] [CrossRef]
- Dumonceau, J.M.; Deprez, P.H.; Jenssen, C.; Iglesias-Garcia, J.; Larghi, A.; Vanbiervliet, G.; Aithal, G.P.; Arcidiacono, P.G.; Bastos, P.; Carrara, S.; et al. Indications, results, and clinical impact of endoscopic ultrasound (EUS)-guided sampling in gastroenterology: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline—Updated January 2017. Endoscopy 2017, 49, 695–714. [Google Scholar] [CrossRef]
- Smith, Z.L.; Satyavada, S.; Simons-Linares, R.; Mok, S.R.S.; Martinez Moreno, B.; Aparicio, J.R.; Chahal, P. Intracystic Glucose and Carcinoembryonic Antigen in Differentiating Histologically Confirmed Pancreatic Mucinous Neoplastic Cysts. Am. J. Gastroenterol. 2022, 117, 478–485. [Google Scholar] [CrossRef]
- Zikos, T.; Pham, K.; Bowen, R.; Chen, A.M.; Banerjee, S.; Friedland, S.; Dua, M.M.; Norton, J.A.; Poultsides, G.A.; Visser, B.C. Cyst fluid glucose is rapidly feasible and accurate in diagnosing mucinous pancreatic cysts. Am. J. Gastroenterol. 2015, 110, 909–914. [Google Scholar] [CrossRef]
- Park, W.G.; Wu, M.; Bowen, R.; Zheng, M.; Fitch, W.L.; Pai, R.K.; Wodziak, D.; Visser, B.C.; Poultsides, G.A.; Norton, J.A. Metabolomic-derived novel cyst fluid biomarkers for pancreatic cysts: Glucose and kynurenine. Gastrointest. Endosc. 2013, 78, 295–302.e292. [Google Scholar] [CrossRef]
- Cameron, M.E.; Yakovenko, A.; Trevino, J.G. Glucose and lactate transport in pancreatic cancer: Glycolytic metabolism revisited. J. Oncol. 2018, 2018, 6214838. [Google Scholar] [CrossRef]
- McCarty, T.R.; Garg, R.; Rustagi, T. Pancreatic cyst fluid glucose in differentiating mucinous from nonmucinous pancreatic cysts: A systematic review and meta-analysis. Gastrointest. Endosc. 2021, 94, 698–712.e696. [Google Scholar] [CrossRef] [PubMed]
- Simons-Linares, C.R.; Yadav, D.; Lopez, R.; Bhatt, A.; Jang, S.; El-Khider, F.; Sanaka, M.; Stevens, T.; Vargo, J.; Chahal, P. The utility of intracystic glucose levels in differentiating mucinous from non-mucinous pancreatic cysts. Pancreatology 2020, 20, 1386–1392. [Google Scholar] [CrossRef] [PubMed]
- Khalid, A.; Zahid, M.; Finkelstein, S.D.; LeBlanc, J.K.; Kaushik, N.; Ahmad, N.; Brugge, W.R.; Edmundowicz, S.A.; Hawes, R.H.; McGrath, K.M. Pancreatic cyst fluid DNA analysis in evaluating pancreatic cysts: A report of the PANDA study. Gastrointest. Endosc. 2009, 69, 1095–1102. [Google Scholar] [CrossRef] [PubMed]
- Nikiforova, M.N.; Khalid, A.; Fasanella, K.E.; McGrath, K.M.; Brand, R.E.; Chennat, J.S.; Slivka, A.; Zeh, H.J.; Zureikat, A.H.; Krasinskas, A.M.; et al. Integration of KRAS testing in the diagnosis of pancreatic cystic lesions: A clinical experience of 618 pancreatic cysts. Mod. Pathol. 2013, 26, 1478–1487. [Google Scholar] [CrossRef] [PubMed]
- Singhi, A.D.; McGrath, K.; Brand, R.E.; Khalid, A.; Zeh, H.J.; Chennat, J.S.; Fasanella, K.E.; Papachristou, G.I.; Slivka, A.; Bartlett, D.L.; et al. Preoperative next-generation sequencing of pancreatic cyst fluid is highly accurate in cyst classification and detection of advanced neoplasia. Gut 2018, 67, 2131–2141. [Google Scholar] [CrossRef]
- McCarty, T.; Rustagi, T. Endoscopic ultrasound-guided through-the-needle microforceps biopsy improves diagnostic yield for pancreatic cystic lesions: A systematic review and meta-analysis. Endosc. Int. Open 2020, 8, E1280–E1290. [Google Scholar]
- Cho, S.H.; Song, T.J.; Seo, D.-W.; Oh, D.; Park, D.H.; Lee, S.S.; Lee, S.K.; Kim, M.-H. Efficacy and safety of EUS-guided through-the-needle microforceps biopsy sampling in categorizing the type of pancreatic cystic lesions. Gastrointest. Endosc. 2022, 95, 299–309. [Google Scholar] [CrossRef]
- Tacelli, M.; Celsa, C.; Magro, B.; Barchiesi, M.; Barresi, L.; Capurso, G.; Arcidiacono, P.G.; Cammà, C.; Crinò, S.F. Diagnostic performance of endoscopic ultrasound through-the-needle microforceps biopsy of pancreatic cystic lesions: Systematic review with meta-analysis. Dig. Endosc. 2020, 32, 1018–1030. [Google Scholar] [CrossRef]
- Krishna, S.G.; Swanson, B.; Hart, P.A.; El-Dika, S.; Walker, J.P.; McCarthy, S.T.; Malli, A.; Shah, Z.K.; Conwell, D.L. Validation of diagnostic characteristics of needle based confocal laser endomicroscopy in differentiation of pancreatic cystic lesions. Endosc. Int. Open 2016, 4, E1124–E1135. [Google Scholar] [CrossRef]
- Li, F.; Malli, A.; Cruz-Monserrate, Z.; Conwell, D.L.; Krishna, S.G. Confocal endomicroscopy and cyst fluid molecular analysis: Comprehensive evaluation of pancreatic cysts. World J. Gastrointest. Endosc. 2018, 10, 1–9. [Google Scholar] [CrossRef]
- Napoleon, B.; Lemaistre, A.I.; Pujol, B.; Caillol, F.; Lucidarme, D.; Bourdariat, R.; Morellon-Mialhe, B.; Fumex, F.; Lefort, C.; Lepilliez, V.; et al. In vivo characterization of pancreatic cystic lesions by needle-based confocal laser endomicroscopy (nCLE): Proposition of a comprehensive nCLE classification confirmed by an external retrospective evaluation. Surg. Endosc. 2016, 30, 2603–2612. [Google Scholar] [CrossRef]
- Nakai, Y.; Iwashita, T.; Park, D.H.; Samarasena, J.B.; Lee, J.G.; Chang, K.J. Diagnosis of pancreatic cysts: EUS-guided, through-the-needle confocal laser-induced endomicroscopy and cystoscopy trial: DETECT study. Gastrointest. Endosc. 2015, 81, 1204–1214. [Google Scholar] [CrossRef]
- Keane, M.G.; Wehnert, N.; Perez-Machado, M.; Fusai, G.K.; Thorburn, D.; Oppong, K.W.; Carroll, N.; Metz, A.J.; Pereira, S.P. A prospective trial of CONfocal endomicroscopy in CYSTic lesions of the pancreas: CONCYST-01. Endosc. Int. Open 2019, 7, E1117–E1122. [Google Scholar] [CrossRef]
- Falconi, M.; Eriksson, B.; Kaltsas, G.; Bartsch, D.K.; Capdevila, J.; Caplin, M.; Kos-Kudla, B.; Kwekkeboom, D.; Rindi, G.; Klöppel, G.; et al. ENETS Consensus Guidelines Update for the Management of Patients with Functional Pancreatic Neuroendocrine Tumors and Non-Functional Pancreatic Neuroendocrine Tumors. Neuroendocrinology 2016, 103, 153–171. [Google Scholar] [CrossRef]
- Clancy, T.E. Surgery for Pancreatic Cancer. Hematol. Oncol. Clin. N. Am. 2015, 29, 701–716. [Google Scholar] [CrossRef]
- Xiao, W.; Zhu, J.; Peng, L.; Hong, L.; Sun, G.; Li, Y. The role of central pancreatectomy in pancreatic surgery: A systematic review and meta-analysis. HPB 2018, 20, 896–904. [Google Scholar] [CrossRef]
- Nagai, K.; Doi, R.; Kida, A.; Kami, K.; Kawaguchi, Y.; Ito, T.; Sakurai, T.; Uemoto, S. Intraductal Papillary Mucinous Neoplasms of the Pancreas: Clinicopathologic Characteristics and Long-Term Follow-Up After Resection. World J. Surg. 2008, 32, 271–278. [Google Scholar] [CrossRef]
- Anand, N.; Sampath, K.; Wu, B.U. Cyst features and risk of malignancy in intraductal papillary mucinous neoplasms of the pancreas: A meta-analysis. Clin. Gastroenterol. Hepatol. 2013, 11, 913–921, quiz e959–e960. [Google Scholar] [CrossRef]
- Scheiman, J.M.; Hwang, J.H.; Moayyedi, P. American gastroenterological association technical review on the diagnosis and management of asymptomatic neoplastic pancreatic cysts. Gastroenterology 2015, 148, 824–848.e822. [Google Scholar] [CrossRef]
- Höhn, P.; Soydemir, M.A.; Luu, A.M.; Janot-Matuschek, M.; Tannapfel, A.; Uhl, W.; Belyaev, O. It’s not all about the size-characteristics and risk factors for malignancy of mucinous cystic neoplasms of the pancreas. Ann. Transl. Med. 2020, 8, 1572. [Google Scholar] [CrossRef]
- Postlewait, L.M.; Ethun, C.G.; McInnis, M.R.; Merchant, N.; Parikh, A.; Idrees, K.; Isom, C.A.; Hawkins, W.; Fields, R.C.; Strand, M.; et al. Association of Preoperative Risk Factors With Malignancy in Pancreatic Mucinous Cystic Neoplasms: A Multicenter Study. JAMA Surg. 2017, 152, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Kwon, W.; Han, Y.; Byun, Y.; Kang, J.S.; Choi, Y.J.; Kim, H.; Jang, J.Y. Predictive Features of Malignancy in Branch Duct Type Intraductal Papillary Mucinous Neoplasm of the Pancreas: A Meta-Analysis. Cancers 2020, 12, 2618. [Google Scholar] [CrossRef] [PubMed]
- Del Chiaro, M.; Beckman, R.; Ateeb, Z.; Orsini, N.; Rezaee, N.; Manos, L.; Valente, R.; Yuan, C.; Ding, D.; Margonis, G.A.; et al. Main Duct Dilatation Is the Best Predictor of High-grade Dysplasia or Invasion in Intraductal Papillary Mucinous Neoplasms of the Pancreas. Ann. Surg. 2020, 272, 1118–1124. [Google Scholar] [CrossRef] [PubMed]
- Marchegiani, G.; Andrianello, S.; Borin, A.; Dal Borgo, C.; Perri, G.; Pollini, T.; Romanò, G.; D’Onofrio, M.; Gabbrielli, A.; Scarpa, A.; et al. Systematic review, meta-analysis, and a high-volume center experience supporting the new role of mural nodules proposed by the updated 2017 international guidelines on IPMN of the pancreas. Surgery 2018, 163, 1272–1279. [Google Scholar] [CrossRef] [PubMed]
- Griffin, J.F.; Page, A.J.; Samaha, G.J.; Christopher, A.; Bhaijee, F.; Pezhouh, M.K.; Peters, N.A.; Hruban, R.H.; He, J.; Makary, M.A.; et al. Patients with a resected pancreatic mucinous cystic neoplasm have a better prognosis than patients with an intraductal papillary mucinous neoplasm: A large single institution series. Pancreatology 2017, 17, 490–496. [Google Scholar] [CrossRef]
- Nilsson, L.N.; Keane, M.G.; Shamali, A.; Millastre Bocos, J.; Marijinissen van Zanten, M.; Antila, A.; Verdejo Gil, C.; Del Chiaro, M.; Laukkarinen, J. Nature and management of pancreatic mucinous cystic neoplasm (MCN): A systematic review of the literature. Pancreatology 2016, 16, 1028–1036. [Google Scholar] [CrossRef]
- Cuillerier, E.; Cellier, C.; Palazzo, L.; Devière, J.; Wind, P.; Rickaert, F.; Cugnenc, P.H.; Cremer, M.; Barbier, J.P. Outcome after surgical resection of intraductal papillary and mucinous tumors of the pancreas. Am. J. Gastroenterol. 2000, 95, 441–445. [Google Scholar] [CrossRef]
- He, J.; Cameron, J.L.; Ahuja, N.; Makary, M.A.; Hirose, K.; Choti, M.A.; Schulick, R.D.; Hruban, R.H.; Pawlik, T.M.; Wolfgang, C.L. Is it necessary to follow patients after resection of a benign pancreatic intraductal papillary mucinous neoplasm? J. Am. Coll. Surg. 2013, 216, 657–665, discussion 665–657. [Google Scholar] [CrossRef]
- Pflüger, M.J.; Griffin, J.F.; Hackeng, W.M.; Kawamoto, S.; Yu, J.; Chianchiano, P.; Shin, E.; Lionheart, G.; Tsai, H.L.; Wang, H.; et al. The Impact of Clinical and Pathological Features on Intraductal Papillary Mucinous Neoplasm Recurrence After Surgical Resection: Long-Term Follow-Up Analysis. Ann. Surg. 2022, 275, 1165–1174. [Google Scholar] [CrossRef]
- Attila, T.; Adsay, V.; Faigel, D.O. The efficacy and safety of endoscopic ultrasound-guided ablation of pancreatic cysts with alcohol and paclitaxel: A systematic review. Eur. J. Gastroenterol. Hepatol. 2019, 31, 1–9. [Google Scholar] [CrossRef]
- Othman, M.; Patel, K.; Krishna, S.G.; Mendoza-Ladd, A.; Verco, S.; Abidi, W.; Verco, J.; Wendt, A.; diZerega, G. Early phase trial of intracystic injection of large surface area microparticle paclitaxel for treatment of mucinous pancreatic cysts. Endosc. Int. Open 2022, 10, E1517–E1525. [Google Scholar] [CrossRef]
- Gadenz, S.D.; Basso, J.; de Oliviera, P.R.B.P.; Sperling, S.; Zuanazzi, M.V.D.; Oliveira, G.G.; da Silva, I.M.; Motta, R.M.; Gehres, L.G.; de Brito Mallmann, É. Telehealth to support referral management in a universal health system: A before-and-after study. BMC Health. Serv. Res. 2021, 21, 1012. [Google Scholar] [CrossRef]
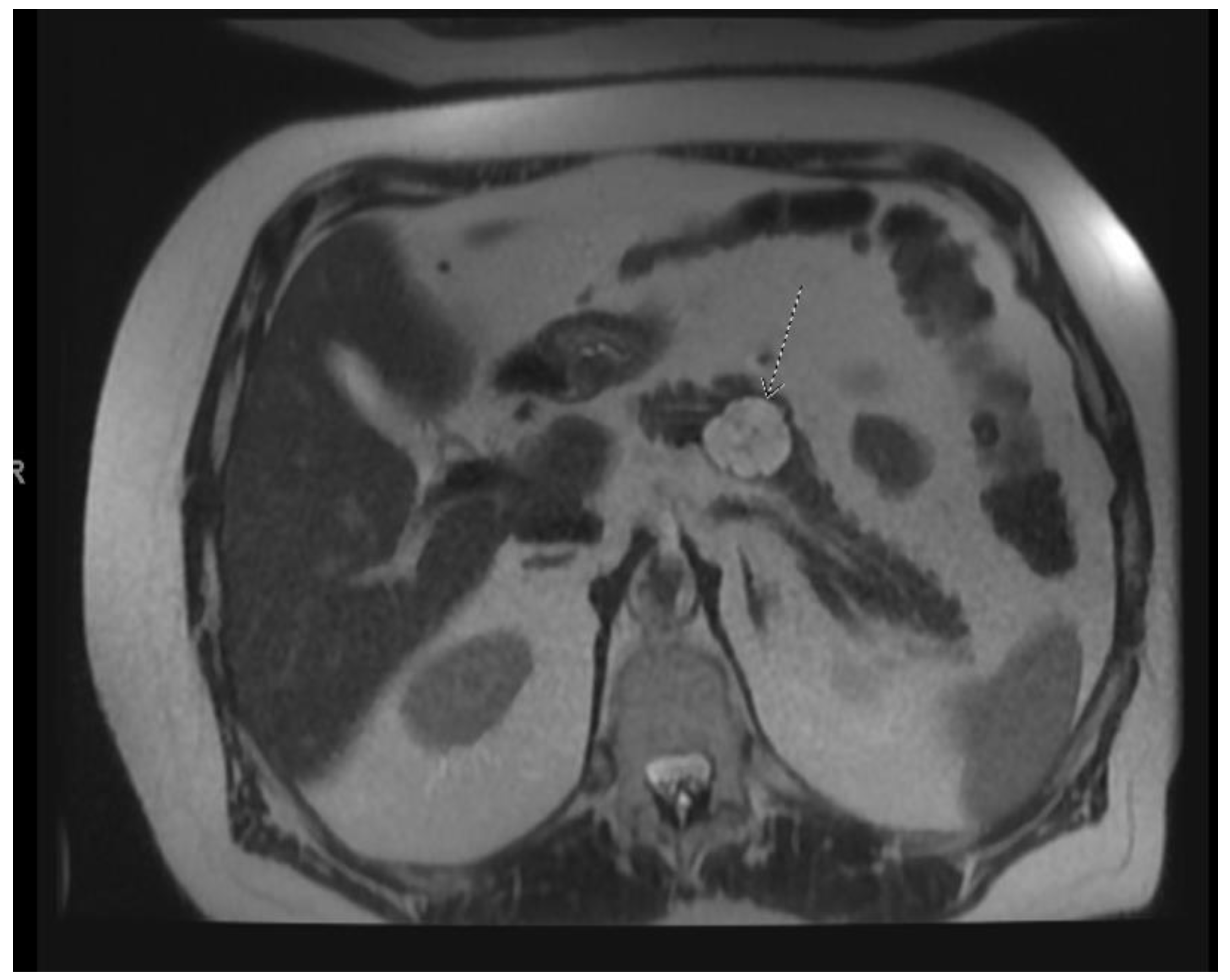
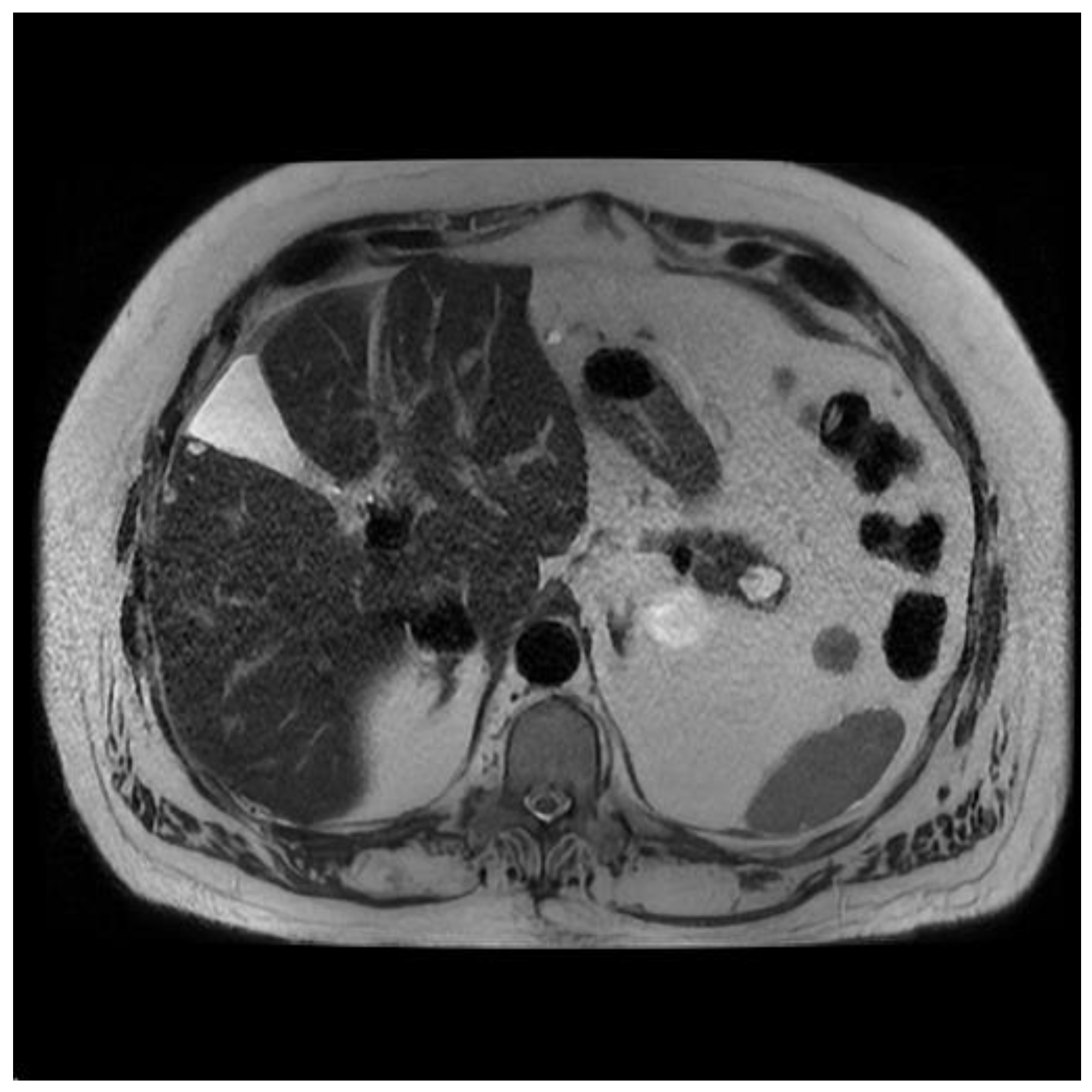
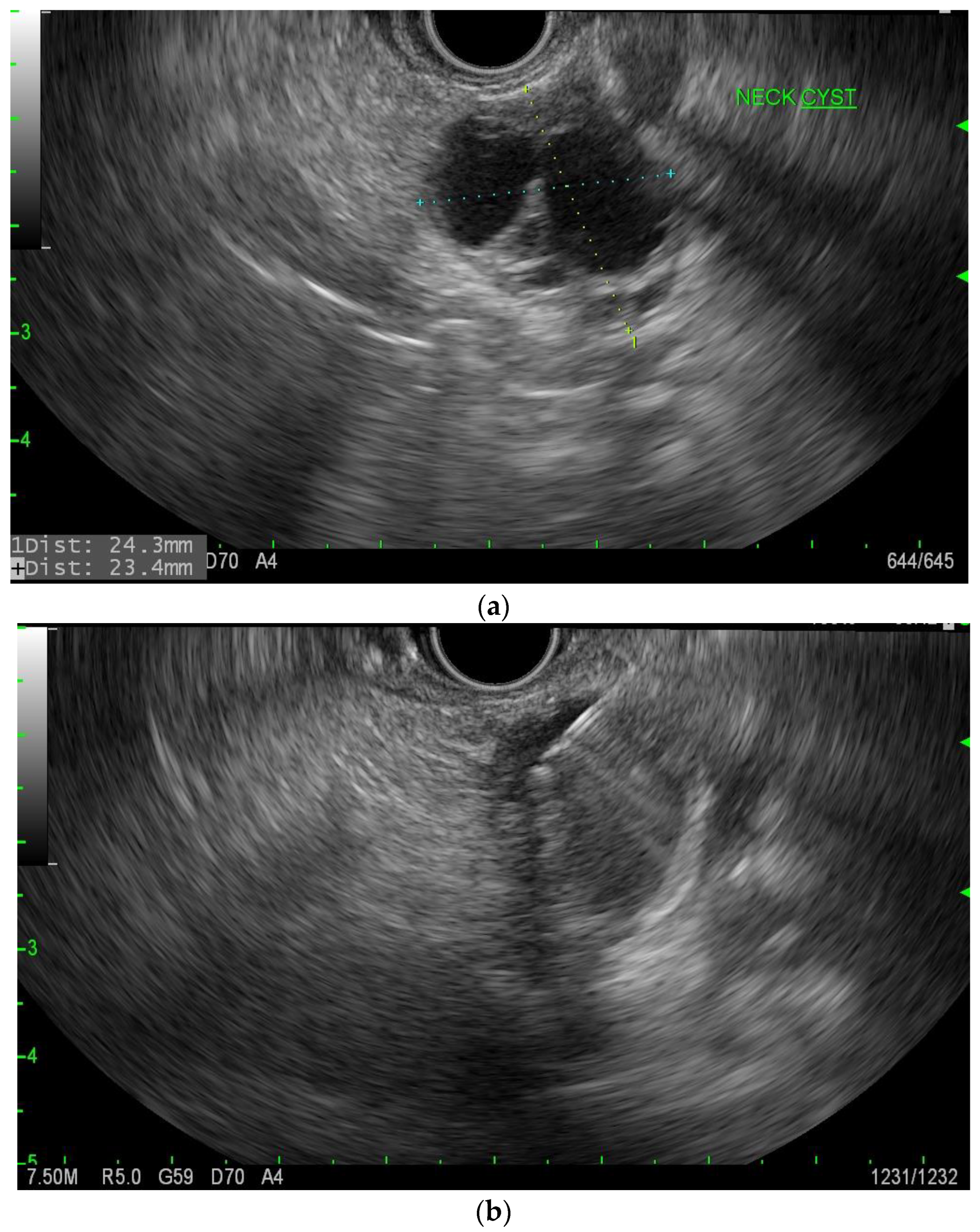
| Society | American College of Gastroenterology (2018) [10] | European Guideline (2018) [29] | International Association of Pancreatology (2017) [30] | American Society of Gastrointestinal Endoscopy (2016) [31] | American Gastrointestinal Association (2015) [32] |
| Cyst Diameter | >3 cm diameter | N/A | >3 cm diameter | >3 cm diameter | >3 cm diameter |
| Pancreatic Duct Dilatation | Yes | N/A | Yes, also an abrupt change in the caliber of the pancreatic duct with distal pancreatic atrophy | Yes | Yes |
| Nodule | Yes | N/A | Enhancing mural nodule <5 mm | Presence of epithelial nodule or suspicious mass lesion | Yes |
| Rate of Growth | Cyst growth rate greater than 3 mm per year | N/A | Cyst growth rate greater than or equal to 5 mm over 2 years | N/A | |
| Biomarker | Elevated Ca 19-9 | N/A | Elevated Ca 19-9 | N/A | |
| Symptoms | Jaundice or Acute Pancreatitis | N/A | N/A | N/A | N/A |
| Additional Recommendations | If not clearly an IPMN or MCN based on cross-sectional imaging, EUS-FNA is recommended. | EUS-(FNA) should only be performed when the results are expected to change clinical management or PCN has either clinical or radiological features of concern identified during the initial investigation or surveillance. | Thickened/enhancing cyst walls, Lymphadenopathy | EUS-FNA is optional for cysts <3 cm without other indications for performing EUS-FNA. |
| Guideline | Cyst Type | Size | Pancreatic Duct Dilatation | Symptoms | Enhancing Mural Nodule | Cytology Positive for Malignancy |
|---|---|---|---|---|---|---|
| 2015 AGA [32] | MCN | All | All | All | All | All |
| IPMN | - | >5 mm and solid component | - | - | Yes | |
| 2017 IAP [30] | MCN | All | All | All | All | All |
| IPMN | ≥5 mm | ≥10 mm | Jaundice | Yes | Yes | |
| 2018 European [29] | MCN | ≥40 mm | - | Jaundice, acute pancreatitis, new-onset diabetes mellitus | Yes | - |
| IPMN | - | ≥10 mm | Jaundice | Yes ≥5 mm | Yes | |
| cNET | >20 mm | - | - | - | Yes | |
| 2018 ACG [10] | MCN | - | - | - | - | - |
| IPMN | - | - | - | - | - |
| Guideline | Cyst Type | Cyst Size | Surveillance Interval | Imaging Modality | When to Lengthen Interval or Stop If Stable |
|---|---|---|---|---|---|
| 2015 AGA [32] | IPMN | <30 mm | Yearly for 1 year, then every 2 years | MRI with MRCP | Stop after 5 years |
| 2017 IAP [30] | IPMN | <10 mm | 6 months and then every 2 years | CT or MRI with MRCP | Lengthen interval after 3 years |
| 10–20 mm | Every 6 months for one year and then yearly for every 2 years | CT or MRI with MRCP | Lengthen interval after 3 years | ||
| 20–30 mm | 3–6 months, then yearly | EUS alternating MRI with EUS | Lifelong surveillance | ||
| 2018 European [29] | MCN and IPMN | <40 mm | Every 6 months for 1 year, then yearly | Ca-19-9, EUS and/or MRI | Lifelong surveillance |
| cNET | <20 mm | No guidance on interval or screening modality | |||
| 2018 ACG [10] | MCN and IPMN | <10 mm | Every 2 years | MRI | Lifelong surveillance |
| 10–20 mm | Yearly for 3 years, then every 2 years | MRI | Lifelong surveillance | ||
| 20–30 mm | Every 6–12 months for 3 years, then yearly | MRI or EUS then MRI | Lifelong surveillance | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schubach, A.; Kothari, S.; Kothari, T. Pancreatic Cystic Neoplasms: Diagnosis and Management. Diagnostics 2023, 13, 207. https://doi.org/10.3390/diagnostics13020207
Schubach A, Kothari S, Kothari T. Pancreatic Cystic Neoplasms: Diagnosis and Management. Diagnostics. 2023; 13(2):207. https://doi.org/10.3390/diagnostics13020207
Chicago/Turabian StyleSchubach, Abigail, Shivangi Kothari, and Truptesh Kothari. 2023. "Pancreatic Cystic Neoplasms: Diagnosis and Management" Diagnostics 13, no. 2: 207. https://doi.org/10.3390/diagnostics13020207
APA StyleSchubach, A., Kothari, S., & Kothari, T. (2023). Pancreatic Cystic Neoplasms: Diagnosis and Management. Diagnostics, 13(2), 207. https://doi.org/10.3390/diagnostics13020207





