Renal Biomarkers in Cardiovascular Patients with Acute Kidney Injury: A Case Report and Literature Review
Abstract
1. Case Presentation
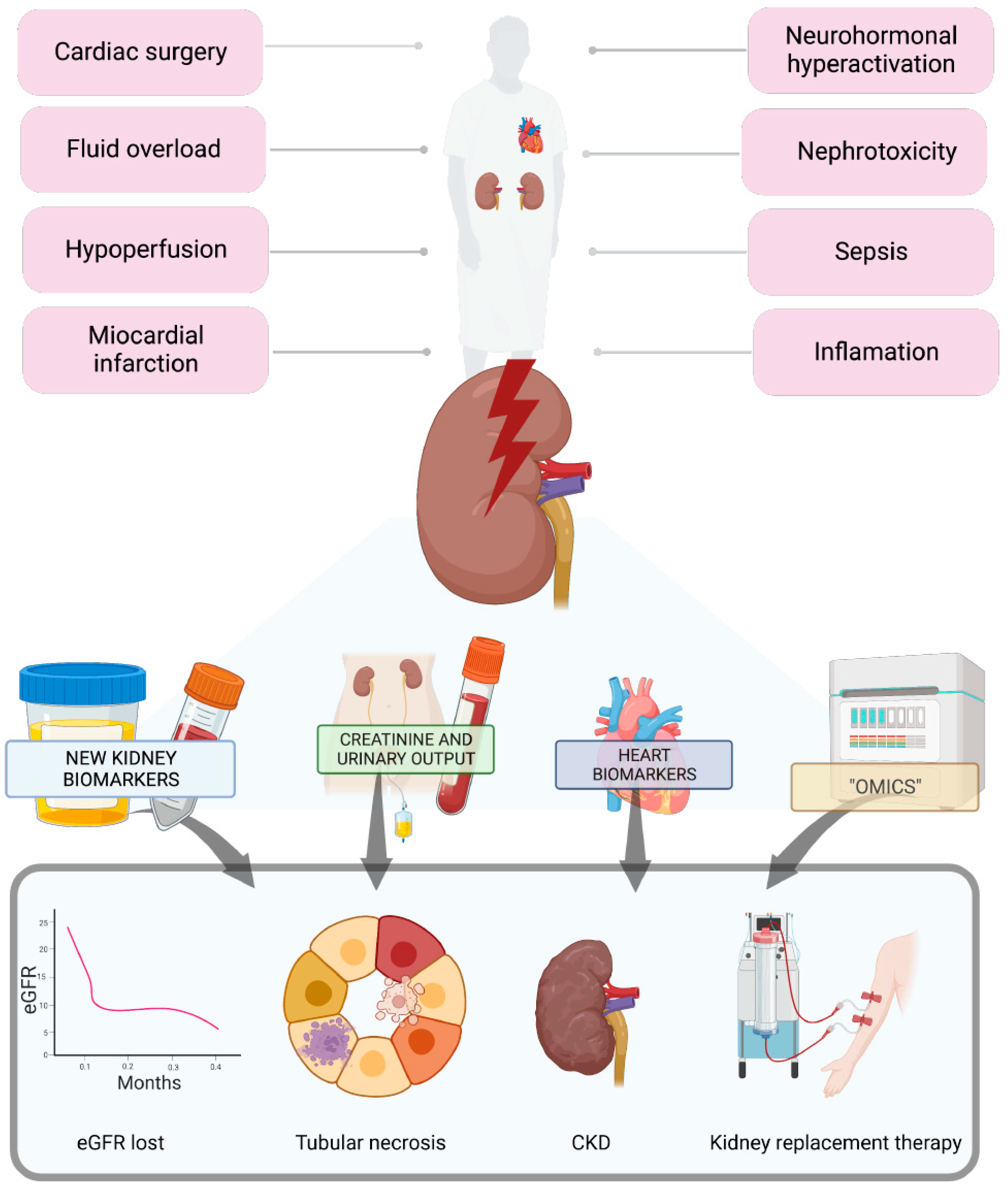
2. Introduction
3. Biomarkers for Diagnosing AKI during Cardiac Failure
4. Predicting AKI in Cardiac Surgery
5. Biomarkers for Assessing CRS Pathophysiology
6. Guiding Treatment with Biomarkers
7. Predicting Outcomes with Biomarkers
7.1. Biomarkers of Function
7.2. Biomarkers of Damage
8. Back to the Case
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chavez-Iniguez, J.S.; Sanchez-Villaseca, S.J.; Garcia-Macias, L.A. Cardiorenal syndrome: Classification, pathophysiology, diagnosis and management. Literature review. Arch. Cardiol. Mex. 2022, 92, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Hatamizadeh, P.; Fonarow, G.C.; Budoff, M.J.; Darabian, S.; Kovesdy, C.P.; Kalantar-Zadeh, K. Cardiorenal syndrome: Pathophysiology and potential targets for clinical management. Nat. Rev. Nephrol. 2013, 9, 99–111. [Google Scholar] [CrossRef] [PubMed]
- Vandenberghe, W.; Gevaert, S.; Kellum, J.A.; Bagshaw, S.M.; Peperstraete, H.; Herck, I.; Decruyenaere, J.; Hoste, E.A. Acute Kidney Injury in Cardiorenal Syndrome Type 1 Patients: A Systematic Review and Meta-Analysis. Cardiorenal Med. 2016, 6, 116–128. [Google Scholar] [CrossRef]
- Gigante, A.; Liberatori, M.; Gasperini, M.L.; Sardo, L.; Di Mario, F.; Dorelli, B.; Barbano, B.; Rosato, E.; Rossi Fanelli, F.; Amoroso, A. Prevalence and clinical features of patients with the cardiorenal syndrome admitted to an internal medicine ward. Cardiorenal Med. 2014, 4, 88–94. [Google Scholar] [CrossRef]
- Mavrakanas, T.A.; Khattak, A.; Singh, K.; Charytan, D.M. Epidemiology and Natural History of the Cardiorenal Syndromes in a Cohort with Echocardiography. Clin. J. Am. Soc. Nephrol. 2017, 12, 1624–1633. [Google Scholar] [CrossRef] [PubMed]
- Boorsma, E.M.; Ter Maaten, J.M.; Voors, A.A.; van Veldhuisen, D.J. Renal Compression in Heart Failure: The Renal Tamponade Hypothesis. JACC Heart Fail. 2022, 10, 175–183. [Google Scholar] [CrossRef]
- Kashani, K.; Rosner, M.H.; Ostermann, M. Creatinine: From physiology to clinical application. Eur. J. Intern. Med. 2020, 72, 9–14. [Google Scholar] [CrossRef]
- Claure-Del Granado, R.; Macedo, E.; Chavez-Iniguez, J.S. Biomarkers for Early Diagnosis of AKI: Could It Backfire? Kidney360 2022, 3, 1780–1784. [Google Scholar] [CrossRef]
- Ostermann, M.; Zarbock, A.; Goldstein, S.; Kashani, K.; Macedo, E.; Murugan, R.; Bell, M.; Forni, L.; Guzzi, L.; Joannidis, M.; et al. Recommendations on Acute Kidney Injury Biomarkers From the Acute Disease Quality Initiative Consensus Conference: A Consensus Statement. JAMA Netw. Open 2020, 3, e2019209. [Google Scholar] [CrossRef]
- Horiuchi, Y.U.; Wettersten, N.; Veldhuisen, D.J.V.; Mueller, C.; Filippatos, G.; Nowak, R.; Hogan, C.; Kontos, M.C.; Cannon, C.M.; Mueller, G.A.; et al. Potential Utility of Cardiorenal Biomarkers for Prediction and Prognostication of Worsening Renal Function in Acute Heart Failure. J. Card. Fail. 2021, 27, 533–541. [Google Scholar] [CrossRef]
- Jang, S.Y.; Yang, D.H.; Kim, H.J.; Park, B.E.; Park, Y.J.; Kim, H.N.; Kim, N.K.; Bae, M.H.; Lee, J.H.; Park, H.S.; et al. Prognostic Value of Cystatin C-Derived Estimated Glomerular Filtration Rate in Patients with Acute Heart Failure. Cardiorenal Med. 2020, 10, 232–242. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.S.; Shahid, I.; Anker, S.D.; Fonarow, G.C.; Fudim, M.; Hall, M.E.; Hernandez, A.; Morris, A.A.; Shafi, T.; Weir, M.R.; et al. Albuminuria and Heart Failure: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2023, 81, 270–282. [Google Scholar] [CrossRef] [PubMed]
- Nishimoto, M.; Murashima, M.; Kokubu, M.; Matsui, M.; Eriguchi, M.; Samejima, K.I.; Akai, Y.; Tsuruya, K. Pre-operative proteinuria and post-operative acute kidney injury in noncardiac surgery: The NARA-Acute Kidney Injury cohort study. Nephrol. Dial. Transplant. 2020, 35, 2111–2116. [Google Scholar] [CrossRef] [PubMed]
- Salah, K.; Kok, W.E.; Eurlings, L.W.; Bettencourt, P.; Pimenta, J.M.; Metra, M.; Verdiani, V.; Tijssen, J.G.; Pinto, Y.M. Competing Risk of Cardiac Status and Renal Function During Hospitalization for Acute Decompensated Heart Failure. JACC Heart Fail. 2015, 3, 751–761. [Google Scholar] [CrossRef] [PubMed]
- Ng, L.L.; Squire, I.B.; Jones, D.J.L.; Cao, T.H.; Chan, D.C.S.; Sandhu, J.K.; Quinn, P.A.; Davies, J.E.; Struck, J.; Hartmann, O.; et al. Proenkephalin, Renal Dysfunction, and Prognosis in Patients With Acute Heart Failure: A GREAT Network Study. J. Am. Coll. Cardiol. 2017, 69, 56–69. [Google Scholar] [CrossRef]
- Breidthardt, T.; Socrates, T.; Drexler, B.; Noveanu, M.; Heinisch, C.; Arenja, N.; Klima, T.; Zusli, C.; Reichlin, T.; Potocki, M.; et al. Plasma neutrophil gelatinase-associated lipocalin for the prediction of acute kidney injury in acute heart failure. Crit. Care 2012, 16, R2. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Yang, X.; Lei, Y.; Zha, Y.; Liu, H.; Ma, C.; Tian, J.; Chen, P.; Yang, T.; Hou, F.F. Urinary Biomarkers at the Time of AKI Diagnosis as Predictors of Progression of AKI among Patients with Acute Cardiorenal Syndrome. Clin. J. Am. Soc. Nephrol. 2016, 11, 1536–1544. [Google Scholar] [CrossRef] [PubMed]
- Aronson, D.; Darawsha, W.; Promyslovsky, M.; Kaplan, M.; Abassi, Z.; Makhoul, B.F.; Goldberg, A.; Azzam, Z.S. Hyponatraemia predicts the acute (type 1) cardio-renal syndrome. Eur. J. Heart Fail. 2014, 16, 49–55. [Google Scholar] [CrossRef]
- Chavez-Iniguez, J.S.; Ibarra-Estrada, M.; Sanchez-Villaseca, S.; Romero-Gonzalez, G.; Font-Yanez, J.J.; De la Torre-Quiroga, A.; de Quevedo, A.A.; Romero-Munoz, A.; Maggiani-Aguilera, P.; Chavez-Alonso, G.; et al. The Effect in Renal Function and Vascular Decongestion in Type 1 Cardiorenal Syndrome Treated with Two Strategies of Diuretics, a Pilot Randomized Trial. BMC Nephrol. 2022, 23, 3. [Google Scholar] [CrossRef]
- Chavez-Iniguez, J.S.; Ivey-Miranda, J.B.; De la Vega-Mendez, F.M.; Borges-Vela, J.A. How to interpret serum creatinine increases during decongestion. Front. Cardiovasc. Med. 2022, 9, 1098553. [Google Scholar] [CrossRef]
- Ho, J.; Reslerova, M.; Gali, B.; Nickerson, P.W.; Rush, D.N.; Sood, M.M.; Bueti, J.; Komenda, P.; Pascoe, E.; Arora, R.C.; et al. Serum creatinine measurement immediately after cardiac surgery and prediction of acute kidney injury. Am. J. Kidney Dis. 2012, 59, 196–201. [Google Scholar] [CrossRef] [PubMed]
- Coca, S.G.; Garg, A.X.; Thiessen-Philbrook, H.; Koyner, J.L.; Patel, U.D.; Krumholz, H.M.; Shlipak, M.G.; Parikh, C.R.; Consortium, T.-A. Urinary biomarkers of AKI and mortality 3 years after cardiac surgery. J. Am. Soc. Nephrol. 2014, 25, 1063–1071. [Google Scholar] [CrossRef]
- Priyanka, P.; Zarbock, A.; Izawa, J.; Gleason, T.G.; Renfurm, R.W.; Kellum, J.A. The impact of acute kidney injury by serum creatinine or urine output criteria on major adverse kidney events in cardiac surgery patients. J. Thorac. Cardiovasc. Surg. 2021, 162, 143–151.E7. [Google Scholar] [CrossRef]
- Molnar, A.O.; Parikh, C.R.; Sint, K.; Coca, S.G.; Koyner, J.; Patel, U.D.; Butrymowicz, I.; Shlipak, M.; Garg, A.X. Association of postoperative proteinuria with AKI after cardiac surgery among patients at high risk. Clin. J. Am. Soc. Nephrol. 2012, 7, 1749–1760. [Google Scholar] [CrossRef] [PubMed]
- Kaufeld, T.; Foerster, K.A.; Schilling, T.; Kielstein, J.T.; Kaufeld, J.; Shrestha, M.; Haller, H.G.; Haverich, A.; Schmidt, B.M.W. Preoperative serum uric acid predicts incident acute kidney injury following cardiac surgery. BMC Nephrol. 2018, 19, 161. [Google Scholar] [CrossRef] [PubMed]
- Husain-Syed, F.; Ferrari, F.; Sharma, A.; Danesi, T.H.; Bezerra, P.; Lopez-Giacoman, S.; Samoni, S.; de Cal, M.; Corradi, V.; Virzi, G.M.; et al. Preoperative Renal Functional Reserve Predicts Risk of Acute Kidney Injury After Cardiac Operation. Ann. Thorac. Surg. 2018, 105, 1094–1101. [Google Scholar] [CrossRef]
- Digvijay, K.; Neri, M.; Fan, W.; Ricci, Z.; Ronco, C. International Survey on the Management of Acute Kidney Injury and Continuous Renal Replacement Therapies: Year 2018. Blood Purif. 2019, 47, 113–119. [Google Scholar] [CrossRef]
- Hu, Y.; Liu, H.; Du, L.; Wan, J.; Li, X. Serum Cystatin C Predicts AKI and the Prognosis of Patients in Coronary Care Unit: A Prospective, Observational Study. Kidney Blood Press. Res. 2017, 42, 961–973. [Google Scholar] [CrossRef] [PubMed]
- Ho, J.; Tangri, N.; Komenda, P.; Kaushal, A.; Sood, M.; Brar, R.; Gill, K.; Walker, S.; MacDonald, K.; Hiebert, B.M.; et al. Urinary, Plasma, and Serum Biomarkers’ Utility for Predicting Acute Kidney Injury Associated With Cardiac Surgery in Adults: A Meta-analysis. Am. J. Kidney Dis. 2015, 66, 993–1005. [Google Scholar] [CrossRef]
- Schley, G.; Koberle, C.; Manuilova, E.; Rutz, S.; Forster, C.; Weyand, M.; Formentini, I.; Kientsch-Engel, R.; Eckardt, K.U.; Willam, C. Comparison of Plasma and Urine Biomarker Performance in Acute Kidney Injury. PLoS ONE 2015, 10, e0145042. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.Z.L.; Martin, A.; Cochrane, A.D.; Smith, J.A.; Thrift, A.G.; Harrop, G.K.; Ngo, J.P.; Evans, R.G. Urinary hypoxia: An intraoperative marker of risk of cardiac surgery-associated acute kidney injury. Nephrol. Dial. Transplant. 2018, 33, 2191–2201. [Google Scholar] [CrossRef] [PubMed]
- Guzzi, L.M.; Bergler, T.; Binnall, B.; Engelman, D.T.; Forni, L.; Germain, M.J.; Gluck, E.; Gocze, I.; Joannidis, M.; Koyner, J.L.; et al. Clinical use of [TIMP-2]*[IGFBP7] biomarker testing to assess risk of acute kidney injury in critical care: Guidance from an expert panel. Crit. Care 2019, 23, 225. [Google Scholar] [CrossRef] [PubMed]
- Heung, M.; Ortega, L.M.; Chawla, L.S.; Wunderink, R.G.; Self, W.H.; Koyner, J.L.; Shi, J.; Kellum, J.A.; Sapphire; Topaz, I. Common chronic conditions do not affect performance of cell cycle arrest biomarkers for risk stratification of acute kidney injury. Nephrol. Dial. Transplant. 2016, 31, 1633–1640. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zou, Z.; Jin, J.; Teng, J.; Xu, J.; Shen, B.; Jiang, W.; Zhuang, Y.; Liu, L.; Luo, Z.; et al. Urinary TIMP-2 and IGFBP7 for the prediction of acute kidney injury following cardiac surgery. BMC Nephrol. 2017, 18, 177. [Google Scholar] [CrossRef]
- Heringlake, M.; Charitos, E.I.; Erber, K.; Berggreen, A.E.; Heinze, H.; Paarmann, H. Preoperative plasma growth-differentiation factor-15 for prediction of acute kidney injury in patients undergoing cardiac surgery. Crit. Care 2016, 20, 317. [Google Scholar] [CrossRef]
- Yang, X.; Chen, C.; Teng, S.; Fu, X.; Zha, Y.; Liu, H.; Wang, L.; Tian, J.; Zhang, X.; Liu, Y.; et al. Urinary Matrix Metalloproteinase-7 Predicts Severe AKI and Poor Outcomes after Cardiac Surgery. J. Am. Soc. Nephrol. 2017, 28, 3373–3382. [Google Scholar] [CrossRef]
- Ronco, C.; Cicoira, M.; McCullough, P.A. Cardiorenal syndrome type 1: Pathophysiological crosstalk leading to combined heart and kidney dysfunction in the setting of acutely decompensated heart failure. J. Am. Coll. Cardiol. 2012, 60, 1031–1042. [Google Scholar] [CrossRef]
- Anand, I.S.; Bishu, K.; Rector, T.S.; Ishani, A.; Kuskowski, M.A.; Cohn, J.N. Proteinuria, chronic kidney disease, and the effect of an angiotensin receptor blocker in addition to an angiotensin-converting enzyme inhibitor in patients with moderate to severe heart failure. Circulation 2009, 120, 1577–1584. [Google Scholar] [CrossRef]
- Jackson, C.E.; Solomon, S.D.; Gerstein, H.C.; Zetterstrand, S.; Olofsson, B.; Michelson, E.L.; Granger, C.B.; Swedberg, K.; Pfeffer, M.A.; Yusuf, S.; et al. Albuminuria in chronic heart failure: Prevalence and prognostic importance. Lancet 2009, 374, 543–550. [Google Scholar] [CrossRef]
- Masson, S.; Latini, R.; Milani, V.; Moretti, L.; Rossi, M.G.; Carbonieri, E.; Frisinghelli, A.; Minneci, C.; Valisi, M.; Maggioni, A.P.; et al. Prevalence and prognostic value of elevated urinary albumin excretion in patients with chronic heart failure: Data from the GISSI-Heart Failure trial. Circ. Heart Fail. 2010, 3, 65–72. [Google Scholar] [CrossRef]
- Sokolski, M.; Zymlinski, R.; Biegus, J.; Siwolowski, P.; Nawrocka-Millward, S.; Todd, J.; Yerramilli, M.R.; Estis, J.; Jankowska, E.A.; Banasiak, W.; et al. Urinary levels of novel kidney biomarkers and risk of true worsening renal function and mortality in patients with acute heart failure. Eur. J. Heart Fail. 2017, 19, 760–767. [Google Scholar] [CrossRef] [PubMed]
- Manzano-Fernandez, S.; Boronat-Garcia, M.; Albaladejo-Oton, M.D.; Pastor, P.; Garrido, I.P.; Pastor-Perez, F.J.; Martinez-Hernandez, P.; Valdes, M.; Pascual-Figal, D.A. Complementary prognostic value of cystatin C, N-terminal pro-B-type natriuretic Peptide and cardiac troponin T in patients with acute heart failure. Am. J. Cardiol. 2009, 103, 1753–1759. [Google Scholar] [CrossRef] [PubMed]
- Carrasco-Sanchez, F.J.; Galisteo-Almeda, L.; Paez-Rubio, I.; Martinez-Marcos, F.J.; Camacho-Vazquez, C.; Ruiz-Frutos, C.; Pujol-De La Llave, E. Prognostic value of cystatin C on admission in heart failure with preserved ejection fraction. J. Card. Fail. 2011, 17, 31–38. [Google Scholar] [CrossRef] [PubMed]
- van Deursen, V.M.; Damman, K.; Voors, A.A.; van der Wal, M.H.; Jaarsma, T.; van Veldhuisen, D.J.; Hillege, H.L. Prognostic value of plasma neutrophil gelatinase-associated lipocalin for mortality in patients with heart failure. Circ. Heart Fail. 2014, 7, 35–42. [Google Scholar] [CrossRef]
- Verbrugge, F.H.; Dupont, M.; Shao, Z.; Shrestha, K.; Singh, D.; Finucan, M.; Mullens, W.; Tang, W.H. Novel urinary biomarkers in detecting acute kidney injury, persistent renal impairment, and all-cause mortality following decongestive therapy in acute decompensated heart failure. J. Card. Fail. 2013, 19, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Chung, E.Y.M.; Trinh, K.; Li, J.; Hahn, S.H.; Endre, Z.H.; Rogers, N.M.; Alexander, S.I. Biomarkers in Cardiorenal Syndrome and Potential Insights Into Novel Therapeutics. Front. Cardiovasc. Med. 2022, 9, 868658. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, T.; Jackson, K.; Rao, V.S.; Tang, W.H.W.; Brisco-Bacik, M.A.; Chen, H.H.; Felker, G.M.; Hernandez, A.F.; O’Connor, C.M.; Sabbisetti, V.S.; et al. Worsening Renal Function in Patients With Acute Heart Failure Undergoing Aggressive Diuresis Is Not Associated With Tubular Injury. Circulation 2018, 137, 2016–2028. [Google Scholar] [CrossRef]
- Mullens, W.; Damman, K.; Harjola, V.P.; Mebazaa, A.; Brunner-La Rocca, H.P.; Martens, P.; Testani, J.M.; Tang, W.H.W.; Orso, F.; Rossignol, P.; et al. The use of diuretics in heart failure with congestion—A position statement from the Heart Failure Association of the European Society of Cardiology. Eur. J. Heart Fail. 2019, 21, 137–155. [Google Scholar] [CrossRef]
- Zeillemaker, A.M.; Verbrugh, H.A.; Hoynck van Papendrecht, A.A.; Leguit, P. CA 125 secretion by peritoneal mesothelial cells. J. Clin. Pathol. 1994, 47, 263–265. [Google Scholar] [CrossRef]
- Nunez, J.; Minana, G.; Nunez, E.; Chorro, F.J.; Bodi, V.; Sanchis, J. Clinical utility of antigen carbohydrate 125 in heart failure. Heart Fail. Rev. 2014, 19, 575–584. [Google Scholar] [CrossRef]
- Nunez, J.; Bayes-Genis, A.; Revuelta-Lopez, E.; Ter Maaten, J.M.; Minana, G.; Barallat, J.; Cserkoova, A.; Bodi, V.; Fernandez-Cisnal, A.; Nunez, E.; et al. Clinical Role of CA125 in Worsening Heart Failure: A BIOSTAT-CHF Study Subanalysis. JACC Heart Fail. 2020, 8, 386–397. [Google Scholar] [CrossRef] [PubMed]
- Nunez, J.; Llacer, P.; Bertomeu-Gonzalez, V.; Bosch, M.J.; Merlos, P.; Garcia-Blas, S.; Montagud, V.; Bodi, V.; Bertomeu-Martinez, V.; Pedrosa, V.; et al. Carbohydrate Antigen-125-Guided Therapy in Acute Heart Failure: CHANCE-HF: A Randomized Study. JACC Heart Fail. 2016, 4, 833–843. [Google Scholar] [CrossRef] [PubMed]
- Nunez, J.; Llacer, P.; Garcia-Blas, S.; Bonanad, C.; Ventura, S.; Nunez, J.M.; Sanchez, R.; Facila, L.; de la Espriella, R.; Vaquer, J.M.; et al. CA125-Guided Diuretic Treatment Versus Usual Care in Patients With Acute Heart Failure and Renal Dysfunction. Am. J. Med. 2020, 133, 370–380 e374. [Google Scholar] [CrossRef] [PubMed]
- Daniels, L.B.; Maisel, A.S. Natriuretic peptides. J. Am. Coll. Cardiol. 2007, 50, 2357–2368. [Google Scholar] [CrossRef] [PubMed]
- Kazanegra, R.; Cheng, V.; Garcia, A.; Krishnaswamy, P.; Gardetto, N.; Clopton, P.; Maisel, A. A rapid test for B-type natriuretic peptide correlates with falling wedge pressures in patients treated for decompensated heart failure: A pilot study. J. Card. Fail. 2001, 7, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Maisel, A. The coming of age of natriuretic peptides: The emperor does have clothes! J. Am. Coll. Cardiol. 2006, 47, 61–64. [Google Scholar] [CrossRef]
- Fu, S.; Zhao, S.; Ye, P.; Luo, L. Biomarkers in Cardiorenal Syndromes. Biomed Res. Int. 2018, 2018, 9617363. [Google Scholar] [CrossRef]
- van Kimmenade, R.R.; Januzzi, J.L., Jr.; Ellinor, P.T.; Sharma, U.C.; Bakker, J.A.; Low, A.F.; Martinez, A.; Crijns, H.J.; MacRae, C.A.; Menheere, P.P.; et al. Utility of amino-terminal pro-brain natriuretic peptide, galectin-3, and apelin for the evaluation of patients with acute heart failure. J. Am. Coll. Cardiol. 2006, 48, 1217–1224. [Google Scholar] [CrossRef]
| Cardiorenal Subtype | Description | Examples/Etiology |
|---|---|---|
| CRS Type 1 (acute CRS) | Rapid worsening of cardiac function leading to acute kidney injury | Acute MI with cardiogenic shock, ADSHF, acute valvular insufficiency |
| CRS Type 2 (chronic CRS) | Chronic abnormalities in cardiac function leading to chronic kidney disease | Chronic inflammation, long-term RAAS and SNS activation, chronic hypoperfusion |
| CRS Type 3 (acute renocardiac syndrome) | Acute worsening of renal function leading to cardiac dysfunction (HF, arrhythmia, and so forth) | Uremia causing impaired contractility, hyperkalemia causing arrhythmias, volume overload causing pulmonary edema |
| CRS Type 4 (chronic renocardiac syndrome) | Chronic worsening of renal function leading to worsening cardiac function | CKD leading to LVH, coronary disease and calcification, diastolic dysfunction, and so forth |
| CRS Type 5 | Acute or chronic systemic disease leading to both cardiac and renal dysfunction | Diabetes mellitus, amyloidosis, sepsis, vasculitis |
| Function of Biomarker | Predictive Value (AUC-ROC) | Prognostic Value (Increase Times Risk of Outcome) | References | |
|---|---|---|---|---|
| Biomarkers of function | ||||
| Albuminuria | Marker of glomerular injury | Unclear | Type 2 CRS: all-cause/CV death or HF hospitalization (1.4–1.8 times) | [38,39,40] |
| Plasma Cystatin-C | Produced by all nucleated cells, marker of eGFR | Type 1 CRS: AKI (0.68), all-cause death or hospitalization (0.73) | Type 1 and 2 CRS: all-cause death (2–3 times) | [41,42,43] |
| Plasma proenkephalin A | Involved in opiod receptor-mediated negative inotropic effects; inversely related to eGFR | Type 1 CRS: AKI (0.69) | Type 1 CRS: all-cause death or HF hospitalization (1.3 times) | [15] |
| Biomarkers of kidney damage | ||||
| Plasma and/or urinary NGAL | Secreted by neutrophils and epithelial cells in response to inflammation. Mediates cardiac fibrosis by aldosterone | Type 1 CRS: AKI (0.775–0.996) | Type 1 CRS: all-cause death (1.3–2 times); AKI (5 times) | [17,41,44] |
| Urinary KIM-1 | Facilitates phagocytosis of apoptotic renal tubular cells | Type 1 CRS: AKI (0.83–0.88) | Type 1 CRS: all-cause death (2 times)Type 2 CRS: all-cause death or HF hospitalizations (1.1–1.5 times) | [22,41,45] |
| Urinary IL-18 | Marker of injury from NLRP3-inflammasome on cardiac myocytes and renal tubular cells | Type 1 CRS: AKI (0.61–0.75); AKI to CKD (0.674) | Type 1 CRS: AKI (3.6 times); all-cause death (1.2 times) | [17,22,45] |
| Urinary L-FABP | Binds fatty acid oxidation products | Type 1 CRS: AKI (0.86) when combined with NAG | Unclear | [29] |
| Urinary NAG | Renal proximal tubule brush border marker | Unclear | Type 2 CRS: all-cause death (1.3–1.4 times); HF hospitalizations (1.2 times) | [46] |
| Urinary angiotensinogen | Marker of intrarenal RAAS activation | Type 1 CRS: AKI (0.78); all-cause death (0.86) | Unclear | [17] |
| Cell-cycle arrest biomarkers and other biomarkers | ||||
| Urinary (TIMP2) × (IGFBP7) | Involved in G1 cell-cycle arrest during early phase of cell injury | Type 1 CRS: AKI (0.75–0.84) | Unclear | [32,46] |
| Plasma sydecan-1 | Marker of glycocalyx injury | Type 1 CRS: AKI (0.741); severe AKI (0.812); all-cause death (0.788) | Type 1 CRS: all-cause death (1.3 times) | [46] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Claure-Del Granado, R.; Chávez-Íñiguez, J.S. Renal Biomarkers in Cardiovascular Patients with Acute Kidney Injury: A Case Report and Literature Review. Diagnostics 2023, 13, 1922. https://doi.org/10.3390/diagnostics13111922
Claure-Del Granado R, Chávez-Íñiguez JS. Renal Biomarkers in Cardiovascular Patients with Acute Kidney Injury: A Case Report and Literature Review. Diagnostics. 2023; 13(11):1922. https://doi.org/10.3390/diagnostics13111922
Chicago/Turabian StyleClaure-Del Granado, Rolando, and Jonathan S. Chávez-Íñiguez. 2023. "Renal Biomarkers in Cardiovascular Patients with Acute Kidney Injury: A Case Report and Literature Review" Diagnostics 13, no. 11: 1922. https://doi.org/10.3390/diagnostics13111922
APA StyleClaure-Del Granado, R., & Chávez-Íñiguez, J. S. (2023). Renal Biomarkers in Cardiovascular Patients with Acute Kidney Injury: A Case Report and Literature Review. Diagnostics, 13(11), 1922. https://doi.org/10.3390/diagnostics13111922







