Role of Imaging in the Management of Patients with SARS-CoV-2 Lung Involvement Admitted to the Emergency Department: A Systematic Review
Abstract
1. Introduction
2. Search Strategy
Included Studies
3. Chest X-ray (CXR)
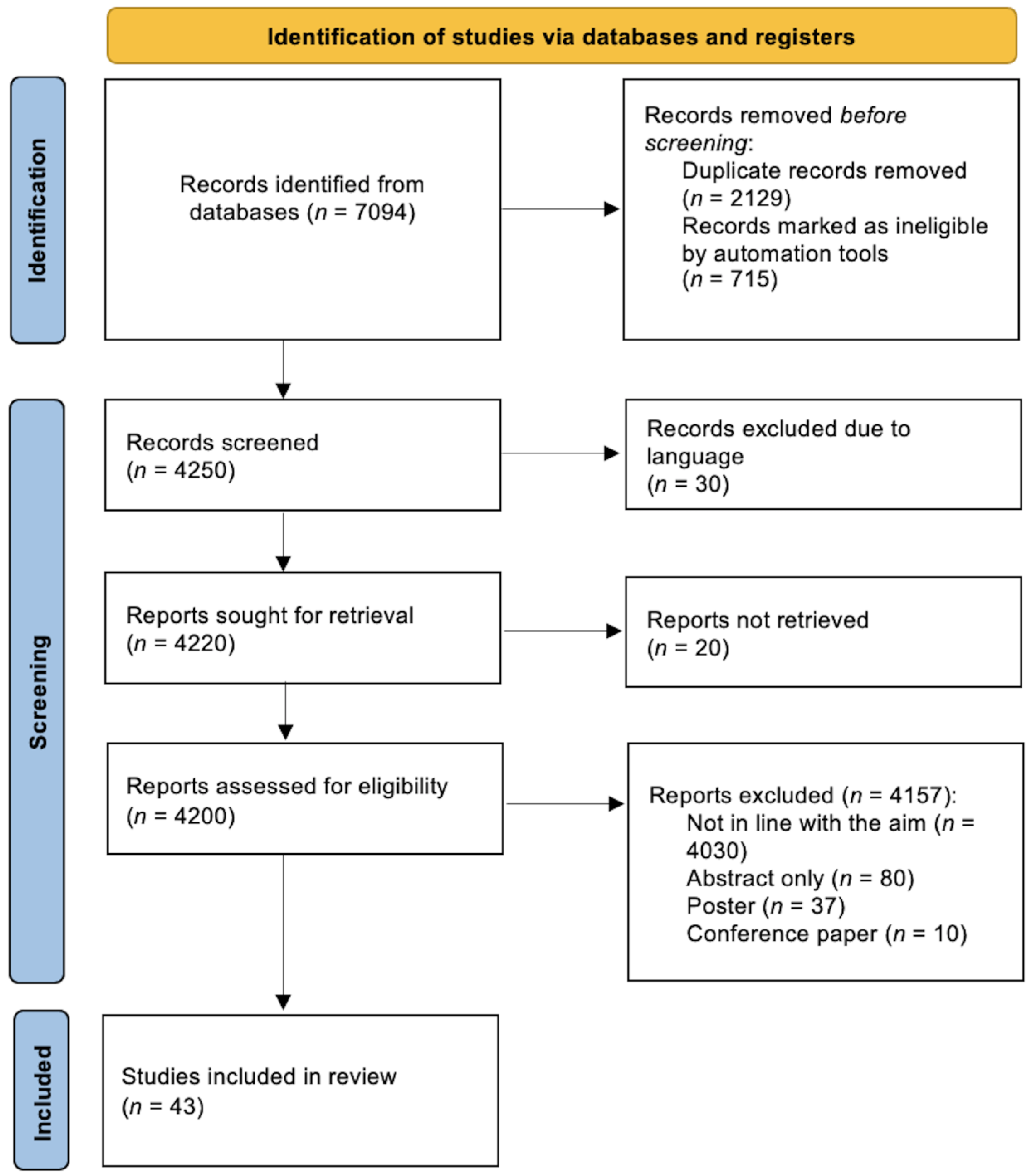
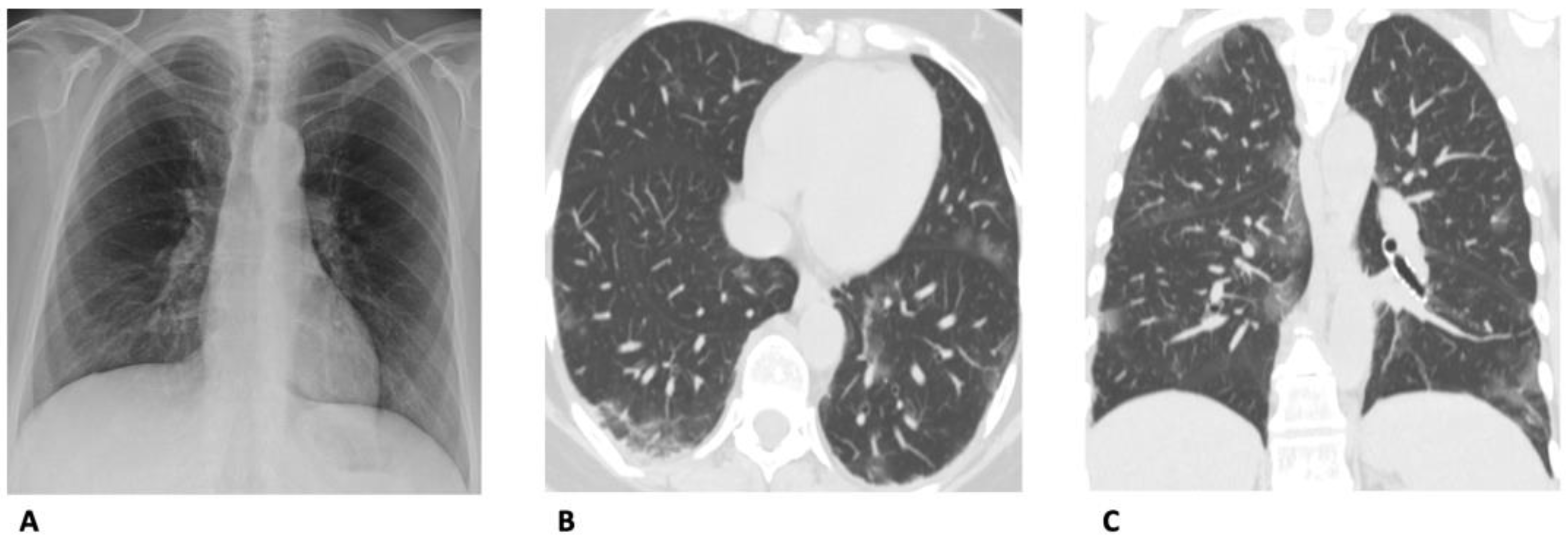
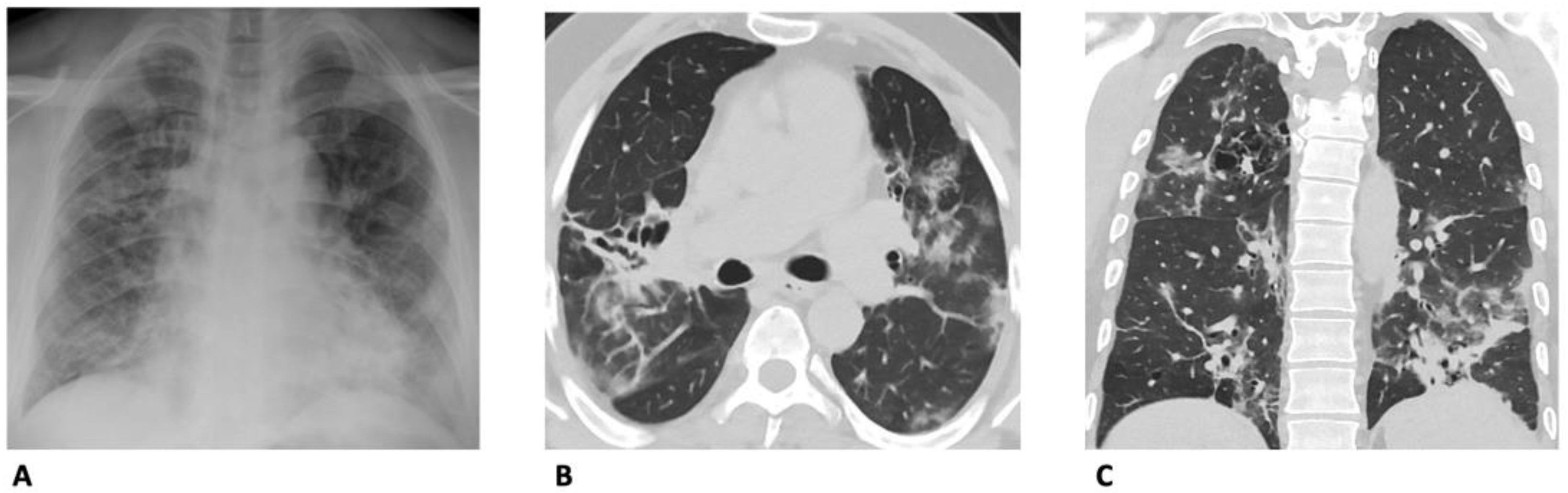
4. Computed Tomography (CT)
5. Management Strategies
6. Lung Ultrasounds (LUS)
7. Artificial Intelligence (AI)
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Available online: https://covid19.who.int/ (accessed on 15 May 2023).
- Ochani, R.; Asad, A.; Yasmin, F.; Shaikh, S.; Khalid, H.; Batra, S.; Sohail, M.R.; Mahmood, S.F.; Ochani, R.; Arshad, M.H.; et al. COVID-19 Pandemic: From Origins to Outcomes. A Comprehensive Review of Viral Pathogenesis, Clinical Manifestations, Diagnostic Evaluation, and Management. Infez. Med. 2021, 29, 20–36. [Google Scholar]
- Butler-Laporte, G.; Lawandi, A.; Schiller, I.; Yao, M.; Dendukuri, N.; McDonald, E.G.; Lee, T.C. Comparison of Saliva and Nasopharyngeal Swab Nucleic Acid Amplification Testing for Detection of SARS-CoV-2: A Systematic Review and Meta-Analysis. JAMA Intern. Med. 2021, 181, 353–360. [Google Scholar] [CrossRef]
- Long, B.; Carius, B.M.; Chavez, S.; Liang, S.Y.; Brady, W.J.; Koyfman, A.; Gottlieb, M. Clinical Update on COVID-19 for the Emergency Clinician: Presentation and Evaluation. Am. J. Emerg. Med. 2022, 54, 46–57. [Google Scholar] [CrossRef]
- Available online: https://www.cdc.gov/coronavirus/2019-ncov/symptoms-testing/symptoms.html (accessed on 20 February 2023).
- Shi, H.; Han, X.; Jiang, N.; Cao, Y.; Alwalid, O.; Gu, J.; Fan, Y.; Zheng, C. Radiological Findings from 81 Patients with COVID-19 Pneumonia in Wuhan, China: A Descriptive Study. Lancet Infect. Dis. 2020, 20, 425–434. [Google Scholar] [CrossRef]
- Bernheim, A.; Mei, X.; Huang, M.; Yang, Y.; Fayad, Z.A.; Zhang, N.; Diao, K.; Lin, B.; Zhu, X.; Li, K.; et al. Chest CT Findings in Coronavirus Disease-19 (COVID-19): Relationship to Duration of Infection. Radiology 2020, 295, 200463. [Google Scholar] [CrossRef]
- Landete, P.; Loaiza, C.A.Q.; Aldave-Orzaiz, B.; Muñiz, S.H.; Maldonado, A.; Zamora, E.; Cerna, A.C.S.; Cerro, E.D.; Alonso, R.C.; Couñago, F. Clinical Features and Radiological Manifestations of COVID-19 Disease. WJR 2020, 12, 247–260. [Google Scholar] [CrossRef]
- Pan, F.; Ye, T.; Sun, P.; Gui, S.; Liang, B.; Li, L.; Zheng, D.; Wang, J.; Hesketh, R.L.; Yang, L.; et al. Time Course of Lung Changes at Chest CT during Recovery from Coronavirus Disease 2019 (COVID-19). Radiology 2020, 295, 715–721. [Google Scholar] [CrossRef]
- Rubin, G.D.; Ryerson, C.J.; Haramati, L.B.; Sverzellati, N.; Kanne, J.P.; Raoof, S.; Schluger, N.W.; Volpi, A.; Yim, J.-J.; Martin, I.B.K.; et al. The Role of Chest Imaging in Patient Management during the COVID-19 Pandemic: A Multinational Consensus Statement from the Fleischner Society. Radiology 2020, 296, 172–180. [Google Scholar] [CrossRef]
- Li, D.; Wang, D.; Dong, J.; Wang, N.; Huang, H.; Xu, H.; Xia, C. False-Negative Results of Real-Time Reverse-Transcriptase Polymerase Chain Reaction for Severe Acute Respiratory Syndrome Coronavirus 2: Role of Deep-Learning-Based CT Diagnosis and Insights from Two Cases. Korean J. Radiol. 2020, 21, 505–508. [Google Scholar] [CrossRef]
- Kepka, S.; Zarca, K.; Viglino, D.; Marjanovic, N.; Taheri, O.; Peyrony, O.; Desmettre, T.; Wilme, V.; Marx, T.; Muller, J.; et al. Imaging Strategies Used in Emergency Departments for the Diagnostic Workup of COVID-19 Patients during the First Wave of the Pandemic: A Cost-Effectiveness Analysis. Clin. Microbiol. Infect. 2022, 28, 1651.e1–1651.e8. [Google Scholar] [CrossRef]
- Ippolito, D.; Pecorelli, A.; Maino, C.; Capodaglio, C.; Mariani, I.; Giandola, T.; Gandola, D.; Bianco, I.; Ragusi, M.; Franzesi, C.T.; et al. Diagnostic Impact of Bedside Chest X-ray Features of 2019 Novel Coronavirus in the Routine Admission at the Emergency Department: Case Series from Lombardy Region. Eur. J. Radiol. 2020, 129, 109092. [Google Scholar] [CrossRef]
- Smith, D.L.; Grenier, J.-P.; Batte, C.; Spieler, B. A Characteristic Chest Radiographic Pattern in the Setting of the COVID-19 Pandemic. Radiol. Cardiothorac. Imaging 2020, 2, e200280. [Google Scholar] [CrossRef] [PubMed]
- Jacobi, A.; Chung, M.; Bernheim, A.; Eber, C. Portable Chest X-ray in Coronavirus Disease-19 (COVID-19): A Pictorial Review. Clin. Imaging 2020, 64, 35–42. [Google Scholar] [CrossRef]
- ACR Recommendations for the Use of Chest Radiography and Computed Tomography (CT) for Suspected COVID-19 Infection. Available online: https://www.acr.org/Advocacy-and-Economics/ACR-Position-Statements/Recommendations-for-Chest-Radiography-and-CT-for-Suspected-COVID19-Infection (accessed on 26 February 2023).
- Liu, T.Y.; Rai, A.; Ditkofsky, N.; Deva, D.P.; Dowdell, T.R.; Ackery, A.D.; Mathur, S. Cost Benefit Analysis of Portable Chest Radiography through Glass: Initial Experience at a Tertiary Care Centre during COVID-19 Pandemic. J. Med. Imaging Radiat. Sci. 2021, 52, 186–190. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, S.H.R.; Shahi, P.K.; Sharma, S.; Dhar, R. Utility of Chest Radiography on Admission for Initial Triaging of COVID-19 in Symptomatic Patients. ERJ Open Res. 2020, 6, 00357–02020. [Google Scholar] [CrossRef] [PubMed]
- Nishtar, T.; Ullah, N.; Ahmad, F.S.; Rahim, S. Radiographic Patterns on Chest X-ray as a Supporting Imaging Tool in Triaging of Suspected Corona Virus Disease (COVID) Patients. Pak. J. Med. Sci. 2022, 38, 1639–1643. [Google Scholar] [CrossRef]
- Velissaris, D.; Lagadinou, M.; Paraskevas, T.; Oikonomou, E.; Karamouzos, V.; Sampsonas, F.; Marangos, M.; Karanikolas, M. Evaluation of Admission Chest X-ray Findings in Patients with Respiratory Infection during the COVID-19 Pandemic. Cureus 2021, 13, e18114. [Google Scholar] [CrossRef]
- Kohli, A.; Hande, P.C.; Chugh, S. Role of Chest Radiography in the Management of COVID-19 Pneumonia: An Overview and Correlation with Pathophysiologic Changes. Indian J. Radiol. Imaging 2021, 31, S70–S79. [Google Scholar] [CrossRef]
- Kotok, D.; Robles, J.R.; Girard, C.E.; Shettigar, S.K.; Lavina, A.P.; Gillenwater, S.R.; Kim, A.I.; Hadeh, A. Chest Radiograph Severity and Its Association with Outcomes in Subjects with COVID-19 Presenting to the Emergency Department. Respir. Care 2022, 67, 871–878. [Google Scholar] [CrossRef]
- Moroni, C.; Cozzi, D.; Albanesi, M.; Cavigli, E.; Bindi, A.; Luvarà, S.; Busoni, S.; Mazzoni, L.N.; Grifoni, S.; Nazerian, P.; et al. Chest X-ray in the Emergency Department during COVID-19 Pandemic Descending Phase in Italy: Correlation with Patients’ Outcome. Radiol. Med. 2021, 126, 661–668. [Google Scholar] [CrossRef]
- Giraudo, C.; Cavaliere, A.; Fichera, G.; Weber, M.; Motta, R.; Pelloso, M.; Tosato, F.; Lupi, A.; Calabrese, F.; Carretta, G.; et al. Validation of a Composed COVID-19 Chest Radiography Score: The CARE Project. ERJ Open Res. 2020, 6, 00359–02020. [Google Scholar] [CrossRef] [PubMed]
- Rawashdeh, M.A.; Saade, C. Radiation Dose Reduction Considerations and Imaging Patterns of Ground Glass Opacities in Coronavirus: Risk of over Exposure in Computed Tomography. Radiol. Med. 2021, 126, 380–387. [Google Scholar] [CrossRef]
- Zhou, Y.; Zheng, Y.; Wen, Y.; Dai, X.; Liu, W.; Gong, Q.; Huang, C.; Lv, F.; Wu, J. Radiation Dose Levels in Chest Computed Tomography Scans of Coronavirus Disease 2019 Pneumonia: A Survey of 2119 Patients in Chongqing, Southwest China. Medicine 2021, 100, e26692. [Google Scholar] [CrossRef]
- Agostini, A.; Floridi, C.; Borgheresi, A.; Badaloni, M.; Pirani, P.E.; Terilli, F.; Ottaviani, L.; Giovagnoni, A. Proposal of a Low-Dose, Long-Pitch, Dual-Source Chest CT Protocol on Third-Generation Dual-Source CT Using a Tin Filter for Spectral Shaping at 100 KVp for CoronaVirus Disease 2019 (COVID-19) Patients: A Feasibility Study. Radiol. Med. 2020, 125, 365–373. [Google Scholar] [CrossRef]
- Dangis, A.; Gieraerts, C.; De Bruecker, Y.; Janssen, L.; Valgaeren, H.; Obbels, D.; Gillis, M.; Van Ranst, M.; Frans, J.; Demeyere, A.; et al. Accuracy and Reproducibility of Low-Dose Submillisievert Chest CT for the Diagnosis of COVID-19. Radiol. Cardiothorac. Imaging 2020, 2, e200196. [Google Scholar] [CrossRef]
- Kwee, R.M.; Adams, H.J.A.; Kwee, T.C. Pulmonary Embolism in Patients with COVID-19 and Value of D-Dimer Assessment: A Meta-Analysis. Eur. Radiol. 2021, 31, 8168–8186. [Google Scholar] [CrossRef]
- Kwee, T.C.; Kwee, R.M. Chest CT in COVID-19: What the Radiologist Needs to Know. RadioGraphics 2020, 40, 1848–1865. [Google Scholar] [CrossRef]
- Teichgräber, U.; Malouhi, A.; Ingwersen, M.; Neumann, R.; Reljic, M.; Deinhardt-Emmer, S.; Löffler, B.; Behringer, W.; Lewejohann, J.-C.; Stallmach, A.; et al. Ruling out COVID-19 by Chest CT at Emergency Admission When Prevalence Is Low: The Prospective, Observational SCOUT Study. Respir. Res. 2021, 22, 13. [Google Scholar] [CrossRef]
- Aslan, S.; Bekçi, T.; Çakır, İ.M.; Ekiz, M.; Yavuz, İ.; Şahin, A.M. Diagnostic Performance of Low-Dose Chest CT to Detect COVID-19: A Turkish Population Study. Diagn. Interv. Radiol. 2021, 27, 181–187. [Google Scholar] [CrossRef]
- Palmisano, A.; Scotti, G.M.; Ippolito, D.; Morelli, M.J.; Vignale, D.; Gandola, D.; Sironi, S.; De Cobelli, F.; Ferrante, L.; Spessot, M.; et al. Chest CT in the Emergency Department for Suspected COVID-19 Pneumonia. Radiol. Med. 2021, 126, 498–502. [Google Scholar] [CrossRef]
- Prokop, M.; Van Everdingen, W.; Van Rees Vellinga, T.; Quarles Van Ufford, H.; Stöger, L.; Beenen, L.; Geurts, B.; Gietema, H.; Krdzalic, J.; Schaefer-Prokop, C.; et al. CO-RADS: A Categorical CT Assessment Scheme for Patients Suspected of Having COVID-19—Definition and Evaluation. Radiology 2020, 296, E97–E104. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Chen, Y.; Runa, A.; Liu, J. Diagnostic Performance of CO-RADS for COVID-19: A Systematic Review and Meta-Analysis. Eur. Radiol. 2022, 32, 4414–4426. [Google Scholar] [CrossRef]
- Özel, M.; Aslan, A.; Araç, S. Use of the COVID-19 Reporting and Data System (CO-RADS) Classification and Chest Computed Tomography Involvement Score (CT-IS) in COVID-19 Pneumonia. Radiol. Med. 2021, 126, 679–687. [Google Scholar] [CrossRef] [PubMed]
- Poor, H.D. Pulmonary Thrombosis and Thromboembolism in COVID-19. Chest 2021, 160, 1471–1480. [Google Scholar] [CrossRef]
- Schulz, K.; Mao, L.; Kanne, J. Computed Tomography Pulmonary Angiography Utilization in the Emergency Department during the COVID-19 Pandemic. J. Thorac. Imaging 2022, 37, 225–230. [Google Scholar] [CrossRef]
- Li, S.; Liu, S.; Wang, B.; Li, Q.; Zhang, H.; Zeng, L.; Ge, H.; Ma, Q.; Shen, N. Predictive Value of Chest CT Scoring in COVID-19 Patients in Wuhan, China: A Retrospective Cohort Study. Respir. Med. 2021, 176, 106271. [Google Scholar] [CrossRef]
- Kang, J.; Kang, J.; Seo, W.J.; Park, S.H.; Kang, H.K.; Park, H.K.; Song, J.E.; Kwak, Y.G.; Chang, J.; Kim, S.; et al. Quantitative Computed Tomography Parameters in Coronavirus Disease 2019 Patients and Prediction of Respiratory Outcomes Using a Decision Tree. Front. Med. 2022, 9, 914098. [Google Scholar] [CrossRef]
- Liu, J.; Liu, S. The Management of Coronavirus Disease 2019 (COVID-19). J. Med. Virol. 2020, 92, 1484–1490. [Google Scholar] [CrossRef]
- Stramare, R.; Carretta, G.; Capizzi, A.; Boemo, D.G.; Contessa, C.; Motta, R.; De Conti, G.; Causin, F.; Giraudo, C.; Donato, D. Radiological Management of COVID-19: Structure Your Diagnostic Path to Guarantee a Safe Path. Radiol. Med. 2020, 125, 691–694. [Google Scholar] [CrossRef]
- Long, C.; Xu, H.; Shen, Q.; Zhang, X.; Fan, B.; Wang, C.; Zeng, B.; Li, Z.; Li, X.; Li, H. Diagnosis of the Coronavirus Disease (COVID-19): RRT-PCR or CT? Eur. J. Radiol. 2020, 126, 108961. [Google Scholar] [CrossRef]
- Wong, H.Y.F.; Lam, H.Y.S.; Fong, A.H.-T.; Leung, S.T.; Chin, T.W.-Y.; Lo, C.S.Y.; Lui, M.M.-S.; Lee, J.C.Y.; Chiu, K.W.-H.; Chung, T.W.-H.; et al. Frequency and Distribution of Chest Radiographic Findings in Patients Positive for COVID-19. Radiology 2020, 296, E72–E78. [Google Scholar] [CrossRef] [PubMed]
- Ai, T.; Yang, Z.; Hou, H.; Zhan, C.; Chen, C.; Lv, W.; Tao, Q.; Sun, Z.; Xia, L. Correlation of Chest CT and RT-PCR Testing for Coronavirus Disease 2019 (COVID-19) in China: A Report of 1014 Cases. Radiology 2020, 296, E32–E40. [Google Scholar] [CrossRef] [PubMed]
- Bellini, D.; Panvini, N.; Rengo, M.; Vicini, S.; Lichtner, M.; Tieghi, T.; Ippoliti, D.; Giulio, F.; Orlando, E.; Iozzino, M.; et al. Diagnostic Accuracy and Interobserver Variability of CO-RADS in Patients with Suspected Coronavirus Disease-2019: A Multireader Validation Study. Eur. Radiol. 2021, 31, 1932–1940. [Google Scholar] [CrossRef]
- Ghosh, S.; Deshwal, H.; Saeedan, M.B.; Khanna, V.K.; Raoof, S.; Mehta, A.C. Imaging Algorithm for COVID-19: A Practical Approach. Clin. Imaging 2021, 72, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Ng, M.-Y.; Lee, E.Y.P.; Yang, J.; Yang, F.; Li, X.; Wang, H.; Lui, M.M.-S.; Lo, C.S.-Y.; Leung, B.; Khong, P.-L.; et al. Imaging Profile of the COVID-19 Infection: Radiologic Findings and Literature Review. Radiol. Cardiothorac. Imaging 2020, 2, e200034. [Google Scholar] [CrossRef]
- Shumilov, E.; Hosseini, A.S.A.; Petzold, G.; Treiber, H.; Lotz, J.; Ellenrieder, V.; Kunsch, S.; Neesse, A. Comparison of Chest Ultrasound and Standard X-ray Imaging in COVID-19 Patients. Ultrasound Int. Open 2020, 6, E36–E40. [Google Scholar] [CrossRef]
- Cappa, G.; Secco, G.; Nganso, A.; Ruzga, R.; Perlini, S. The Role of Lung Ultrasound in Low-Resource Settings during the Coronavirus (SARS-CoV-2) Pandemic. J. Ultrasound Med. 2022, 41, 543–545. [Google Scholar] [CrossRef]
- Di Serafino, M.; Notaro, M.; Rea, G.; Iacobellis, F.; Delli Paoli, V.; Acampora, C.; Ianniello, S.; Brunese, L.; Romano, L.; Vallone, G. The Lung Ultrasound: Facts or Artifacts? In the Era of COVID-19 Outbreak. Radiol. Med. 2020, 125, 738–753. [Google Scholar] [CrossRef]
- Abrams, E.R.; Rose, G.; Fields, J.M.; Esener, D. Point-of-Care Ultrasound in the Evaluation of COVID-19. J. Emerg. Med. 2020, 59, 403–408. [Google Scholar] [CrossRef]
- Toma, T.P.; Trigiani, M.; Zanforlin, A.; Inchingolo, R.; Zanobetti, M.; Sammicheli, L.; Conte, E.G.; Buggio, G.; Villari, L.; Corbetta, L.; et al. Competence in Thoracic Ultrasound. Panminerva Med. 2019, 61, 344–366. [Google Scholar] [CrossRef]
- Gil-Rodríguez, J.; de Rojas, J.P.; Aranda-Laserna, P.; Benavente-Fernández, A.; Martos-Ruiz, M.; Peregrina-Rivas, J.-A.; Guirao-Arrabal, E. Ultrasound Findings of Lung Ultrasonography in COVID-19: A Systematic Review. Eur. J. Radiol. 2022, 148, 110156. [Google Scholar] [CrossRef] [PubMed]
- Vetrugno, L.; Bove, T.; Orso, D.; Barbariol, F.; Bassi, F.; Boero, E.; Ferrari, G.; Kong, R. B Lines in COVID-19: “Unspecificity” Is Not “Meaningless”. Echocardiography 2020, 37, 1140–1141. [Google Scholar] [CrossRef] [PubMed]
- Boero, E.; Schreiber, A.; Rovida, S.; Vetrugno, L.; Blaivas, M. The Role of Lung Ultrasonography in COVID-19 Disease Management. J. Am. Coll. Emerg. Physicians Open 2020, 1, 1357–1363. [Google Scholar] [CrossRef]
- Piliego, C.; Strumia, A.; Stone, M.B.; Pascarella, G. The Ultrasound-Guided Triage: A New Tool for Prehospital Management of COVID-19 Pandemic. Anesth. Analg. 2020, 131, e93–e94. [Google Scholar] [CrossRef]
- Narinx, N.; Smismans, A.; Symons, R.; Frans, J.; Demeyere, A.; Gillis, M. Feasibility of Using Point-of-Care Lung Ultrasound for Early Triage of COVID-19 Patients in the Emergency Room. Emerg. Radiol. 2020, 27, 663–670. [Google Scholar] [CrossRef] [PubMed]
- Guzmán-García, M.B.; Mohedano-Moriano, A.; González-González, J.; Morales-Cano, J.M.; Campo-Linares, R.; Lozano-Suárez, C.; Estrada-Álvarez, T.P.; Romero-Fernández, M.M.; Aguilar-Galán, E.V.; Criado-Álvarez, J.J. Lung Ultrasound as a Triage Method in Primary Care for Patients with Suspected SARS-CoV-2 Pneumonia. J. Clin. Med. 2022, 11, 6420. [Google Scholar] [CrossRef]
- Volpicelli, G.; Gargani, L.; Perlini, S.; Spinelli, S.; Barbieri, G.; Lanotte, A.; Casasola, G.G.; Nogué-Bou, R.; Lamorte, A.; Agricola, E.; et al. Lung Ultrasound for the Early Diagnosis of COVID-19 Pneumonia: An International Multicenter Study. Intensive Care Med. 2021, 47, 444–454. [Google Scholar] [CrossRef]
- Pivetta, E.; Goffi, A.; Tizzani, M.; Locatelli, S.M.; Porrino, G.; Losano, I.; Leone, D.; Calzolari, G.; Vesan, M.; Steri, F.; et al. Lung Ultrasonography for the Diagnosis of SARS-CoV-2 Pneumonia in the Emergency Department. Ann. Emerg. Med. 2021, 77, 385–394. [Google Scholar] [CrossRef]
- Sorlini, C.; Femia, M.; Nattino, G.; Bellone, P.; Gesu, E.; Francione, P.; Paternò, M.; Grillo, P.; Ruffino, A.; Bertolini, G.; et al. The Role of Lung Ultrasound as a Frontline Diagnostic Tool in the Era of COVID-19 Outbreak. Intern. Emerg. Med. 2021, 16, 749–756. [Google Scholar] [CrossRef]
- Bonadia, N.; Carnicelli, A.; Piano, A.; Buonsenso, D.; Gilardi, E.; Kadhim, C.; Torelli, E.; Petrucci, M.; Di Maurizio, L.; Biasucci, D.G.; et al. Lung Ultrasound Findings Are Associated with Mortality and Need for Intensive Care Admission in COVID-19 Patients Evaluated in the Emergency Department. Ultrasound Med. Biol. 2020, 46, 2927–2937. [Google Scholar] [CrossRef]
- Si, X.; Yi, H.; Liu, D.; Wang, R.; Liu, Y.; Wu, J.; Guan, X. Lung Ultrasound: A Valuable Tool for Assessing COVID-19 Patients with Different Severity. Chin. Med. J. 2022, 135, 1114–1116. [Google Scholar] [CrossRef] [PubMed]
- Gottlieb, M.; Jay, A.; Mogni, B. What Is the Diagnostic Accuracy of Chest Radiography, Ultrasound, and Computed Tomography for COVID-19? Ann. Emerg. Med. 2022, 79, 64–65. [Google Scholar] [CrossRef] [PubMed]
- Colombi, D.; Petrini, M.; Maffi, G.; Villani, G.D.; Bodini, F.C.; Morelli, N.; Milanese, G.; Silva, M.; Sverzellati, N.; Michieletti, E. Comparison of Admission Chest Computed Tomography and Lung Ultrasound Performance for Diagnosis of COVID-19 Pneumonia in Populations with Different Disease Prevalence. Eur. J. Radiol. 2020, 133, 109344. [Google Scholar] [CrossRef] [PubMed]
- Pare, J.R.; Camelo, I.; Mayo, K.C.; Leo, M.M.; Dugas, J.N.; Nelson, K.P.; Baker, W.E.; Shareef, F.; Mitchell, P.M.; Schechter-Perkins, E.M. Point-of-Care Lung Ultrasound Is More Sensitive than Chest Radiograph for Evaluation of COVID-19. West. J. Emerg. Med. 2020, 21, 771–778. [Google Scholar] [CrossRef]
- Mateos González, M.; García de Casasola Sánchez, G.; Muñoz, F.J.T.; Proud, K.; Lourdo, D.; Sander, J.-V.; Jaimes, G.E.O.; Mader, M.; Canora Lebrato, J.; Restrepo, M.I.; et al. Comparison of Lung Ultrasound versus Chest X-ray for Detection of Pulmonary Infiltrates in COVID-19. Diagnostics 2021, 11, 373. [Google Scholar] [CrossRef]
- Wang, M.; Xia, C.; Huang, L.; Xu, S.; Qin, C.; Liu, J.; Cao, Y.; Yu, P.; Zhu, T.; Zhu, H.; et al. Deep Learning-Based Triage and Analysis of Lesion Burden for COVID-19: A Retrospective Study with External Validation. Lancet Digit. Health 2020, 2, e506–e515. [Google Scholar] [CrossRef]
- Mei, X.; Lee, H.-C.; Diao, K.-Y.; Huang, M.; Lin, B.; Liu, C.; Xie, Z.; Ma, Y.; Robson, P.M.; Chung, M.; et al. Artificial Intelligence-Enabled Rapid Diagnosis of Patients with COVID-19. Nat. Med. 2020, 26, 1224–1228. [Google Scholar] [CrossRef]
- Kataoka, Y.; Kimura, Y.; Ikenoue, T.; Matsuoka, Y.; Matsumoto, J.; Kumasawa, J.; Tochitatni, K.; Funakoshi, H.; Hosoda, T.; Kugimiya, A.; et al. Integrated Model for COVID-19 Diagnosis Based on Computed Tomography Artificial Intelligence, and Clinical Features: A Multicenter Cohort Study. Ann. Transl. Med. 2022, 10, 130. [Google Scholar] [CrossRef]
- Wang, B.; Jin, S.; Yan, Q.; Xu, H.; Luo, C.; Wei, L.; Zhao, W.; Hou, X.; Ma, W.; Xu, Z.; et al. AI-Assisted CT Imaging Analysis for COVID-19 Screening: Building and Deploying a Medical AI System. Appl. Soft Comput. 2021, 98, 106897. [Google Scholar] [CrossRef]
- Carlile, M.; Hurt, B.; Hsiao, A.; Hogarth, M.; Longhurst, C.A.; Dameff, C. Deployment of Artificial Intelligence for Radiographic Diagnosis of COVID-19 Pneumonia in the Emergency Department. J. Am. Coll. Emerg. Physicians Open 2020, 1, 1459–1464. [Google Scholar] [CrossRef]
- Panwar, H.; Gupta, P.K.; Siddiqui, M.K.; Morales-Menendez, R.; Singh, V. Application of Deep Learning for Fast Detection of COVID-19 in X-rays Using NCOVnet. Chaos Solitons Fractals 2020, 138, 109944. [Google Scholar] [CrossRef] [PubMed]
- Drozdov, I.; Szubert, B.; Reda, E.; Makary, P.; Forbes, D.; Chang, S.L.; Ezhil, A.; Puttagunta, S.; Hall, M.; Carlin, C.; et al. Development and Prospective Validation of COVID-19 Chest X-ray Screening Model for Patients Attending Emergency Departments. Sci. Rep. 2021, 11, 20384. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maino, C.; Franco, P.N.; Talei Franzesi, C.; Giandola, T.; Ragusi, M.; Corso, R.; Ippolito, D. Role of Imaging in the Management of Patients with SARS-CoV-2 Lung Involvement Admitted to the Emergency Department: A Systematic Review. Diagnostics 2023, 13, 1856. https://doi.org/10.3390/diagnostics13111856
Maino C, Franco PN, Talei Franzesi C, Giandola T, Ragusi M, Corso R, Ippolito D. Role of Imaging in the Management of Patients with SARS-CoV-2 Lung Involvement Admitted to the Emergency Department: A Systematic Review. Diagnostics. 2023; 13(11):1856. https://doi.org/10.3390/diagnostics13111856
Chicago/Turabian StyleMaino, Cesare, Paolo Niccolò Franco, Cammillo Talei Franzesi, Teresa Giandola, Maria Ragusi, Rocco Corso, and Davide Ippolito. 2023. "Role of Imaging in the Management of Patients with SARS-CoV-2 Lung Involvement Admitted to the Emergency Department: A Systematic Review" Diagnostics 13, no. 11: 1856. https://doi.org/10.3390/diagnostics13111856
APA StyleMaino, C., Franco, P. N., Talei Franzesi, C., Giandola, T., Ragusi, M., Corso, R., & Ippolito, D. (2023). Role of Imaging in the Management of Patients with SARS-CoV-2 Lung Involvement Admitted to the Emergency Department: A Systematic Review. Diagnostics, 13(11), 1856. https://doi.org/10.3390/diagnostics13111856




