Using Machine Learning to Predict Abnormal Carotid Intima-Media Thickness in Type 2 Diabetes
Abstract
1. Introduction
2. Materials and Methods
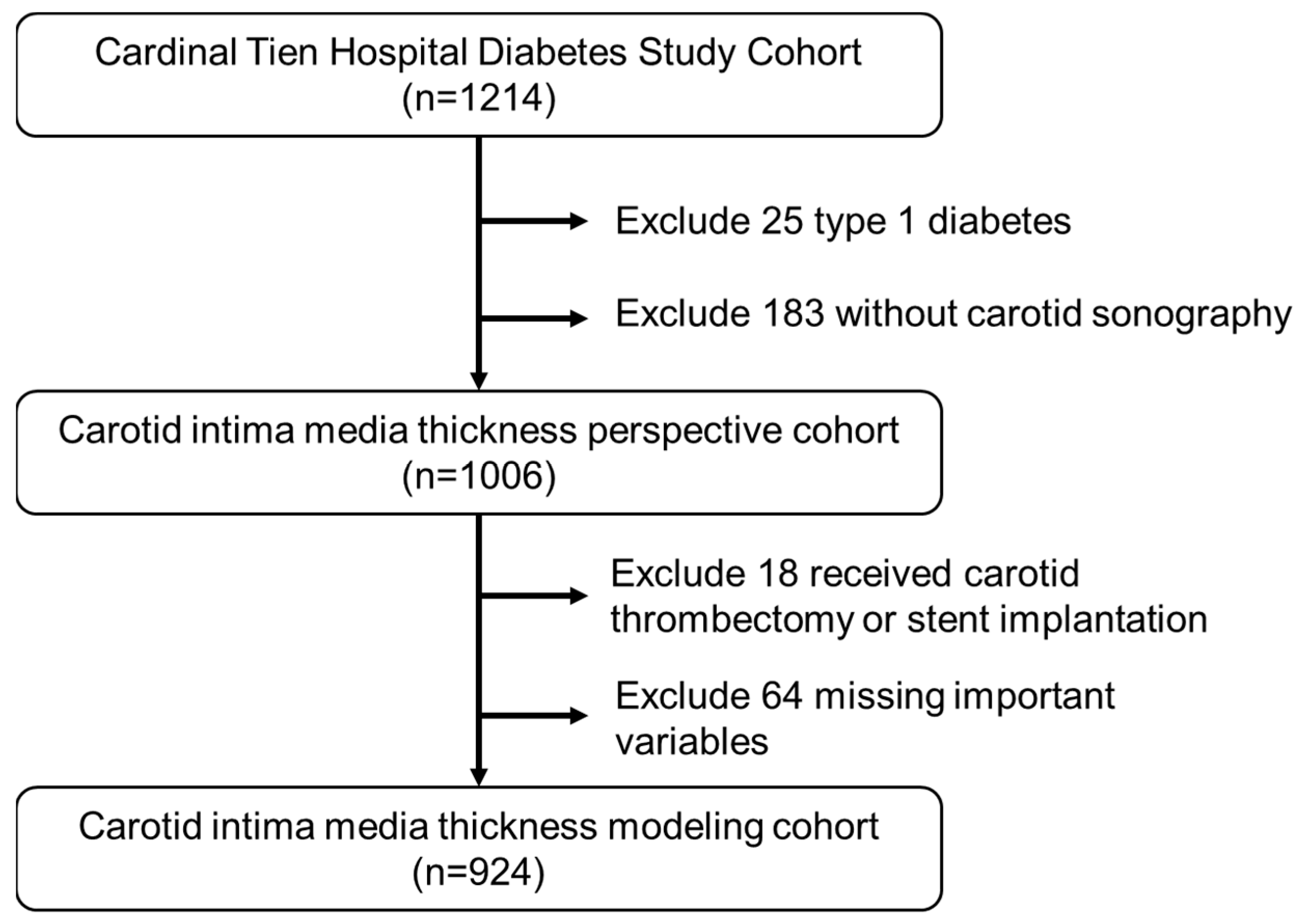
2.1. Participants and Study Design
2.2. Measurements of Anthropometry and Biochemistry
2.3. Quantification of Carotid MIT and Plaque
2.4. Description of the Study Data Set
2.5. Proposed Scheme
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khan, M.A.B.; Hashim, M.J.; King, J.K.; Govender, R.D.; Mustafa, H.; Al Kaabi, J. Epidemiology of Type 2 Diabetes—Global Burden of Disease and Forecasted Trends. J. Epidemiol. Glob. Health 2020, 10, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Saeedi, P.; Karuranga, S.; Pinkepank, M.; Ogurtsova, K.; Duncan, B.B.; Stein, C.; Basit, A.; Chan, J.C.; Mbanya, J.C.; et al. IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res. Clin. Pract. 2022, 183, 109119. [Google Scholar] [CrossRef] [PubMed]
- Faselis, C.; Katsimardou, A.; Imprialos, K.; Deligkaris, P.; Kallistratos, M.; Dimitriadis, K. Microvascular Complications of Type 2 Diabetes Mellitus. Curr. Vasc. Pharmacol. 2020, 18, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Ley, S.H.; Hu, F.B. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat. Rev. Endocrinol. 2018, 14, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Katakami, N.; Matsuoka, T.A.; Shimomura, I. Clinical utility of carotid ultrasonography: Application for the management of patients with diabetes. J. Diabetes Investig. 2019, 10, 883–898. [Google Scholar] [CrossRef]
- Johansson, A.; Acosta, S. Diet and Lifestyle as Risk Factors for Carotid Artery Disease: A Prospective Cohort Study. Cerebrovasc. Dis. 2020, 49, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Kaul, S.; Alladi, S.; Mridula, K.R.; Bandaru, V.S.; Umamashesh, M.; Anjanikumar, D.; Lalitha, P.; Chandrasekhar, R. Prevalence and risk factors of asymptomatic carotid artery stenosis in Indian population: An 8-year follow-up study. Neurol. India 2017, 65, 279–285. [Google Scholar] [CrossRef]
- Zanchetti, A.; Crepaldi, G.; Bond, M.G.; Gallus, G.V.; Veglia, F.; Ventura, A.; Mancia, G.; Baggio, G.; Sampieri, L.; Rubba, P.; et al. Systolic and pulse blood pressures (but not diastolic blood pressure and serum cholesterol) are associated with alterations in carotid intima-media thickness in the moderately hypercholesterolaemic hypertensive patients of the Plaque Hypertension Lipid Lowering Italian Study. PHYLLIS study group. J. Hypertens. 2001, 19, 79–88. [Google Scholar]
- Mitchell, T. Machine Learning; McGraw Hill: New York, NY, USA, 1997. [Google Scholar]
- Marateb, H.R.; Mansourian, M.; Faghihimani, E.; Amini, M.; Farina, D. A hybrid intelligent system for diagnosing microalbuminuria in type 2 diabetes patients without having to measure urinary albumin. Comput. Biol. Med. 2014, 45, 34–42. [Google Scholar] [CrossRef]
- Nusinovici, S.; Tham, Y.C.; Yan, M.Y.C.; Ting, D.S.W.; Li, J.; Sabanayagam, C.; Wong, T.Y.; Cheng, C.Y. Logistic regression was as good as machine learning for predicting major chronic diseases. J. Clin. Epidemiol. 2020, 122, 56–69. [Google Scholar] [CrossRef]
- Ye, Y.; Xiong, Y.; Zhou, Q.; Wu, J.; Li, X.; Xiao, X. Comparison of Machine Learning Methods and Conventional Logistic Regressions for Predicting Gestational Diabetes Using Routine Clinical Data: A Retrospective Cohort Study. J. Diabetes Res. 2020, 2020, 4168340. [Google Scholar] [CrossRef] [PubMed]
- Miller, D.D.; Brown, E.W. Artificial Intelligence in Medical Practice: The Question to the Answer? Am. J. Med. 2018, 131, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Du, C.; Shao, W.; Ma, G. Diagnostic Role of Carotid Intima-Media Thickness for Coronary Artery Disease: A Meta-Analysis. Biomed Res. Int. 2020, 2020, 9879463. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.C.; Cheng, Y.C.; Jhou, M.J.; Chen, M.; Lu, C.J. Important Risk Factors in Patients with Nonvalvular Atrial Fibrillation Taking Dabigatran Using Integrated Machine Learning Scheme-A Post Hoc Analysis. J. Pers. Med. 2022, 12, 756. [Google Scholar] [CrossRef]
- Breiman, L.; Friedman, J.H.; Olshen, R.A.; Stone, C.J. Classification and Regression Trees; Champman & Hall/CRC: New York, NY, USA, 1984. [Google Scholar]
- Patel, N.; Upadhyay, S. Study of Various Decision Tree Pruning Methods with their Empirical Comparison in WEKA. Int. J. Comp. Appl. 2012, 60, 20–25. [Google Scholar] [CrossRef]
- Tierney, N.J.; Harden, F.A.; Harden, M.J.; Mengersen, K.L. Using decision trees to understand structure in missing data. BMJ Open 2015, 5, e007450. [Google Scholar] [CrossRef]
- Breiman, L. Random forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Calle, M.; Urrea, V. Letter to the editor: Stability of random forest importance measures. Brief. Bioinform. 2011, 12, 86–89. [Google Scholar] [CrossRef]
- Chen, T.; Guestrin, C. XGBoost: A Scalable Tree Boosting System. In Proceedings of the KDD ‘16: Proceedings of the 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining, San Francisco, CA, USA, 13–17 August 2016; pp. 785–794. [Google Scholar]
- Torlay, L.; Perrone-Bertolotti, M.; Thomas, E.; Baciu, M. Machine learning–XGBoost analysis of language networks to classify patients with epilepsy. Brain Inform. 2017, 4, 159–169. [Google Scholar] [CrossRef]
- Basa, A.L.; Garber, A.J. Cardiovascular disease and diabetes: Modifying risk factors other than glucose control. Ochsner J. 2001, 3, 132–137. [Google Scholar]
- Kabłak-Ziembicka, A.; Przewłocki, T. Clinical Significance of Carotid Intima-Media Complex and Carotid Plaque Assessment by Ultrasound for the Prediction of Adverse Cardiovascular Events in Primary and Secondary Care Patients. J. Clin. Med. 2021, 10, 4628. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.D.; Chang, C.H.; Tai, T.Y.; Chen, J.F.; Chuang, L.M. Incidence and prevalence rates of diabetes mellitus in Taiwan: Analysis of the 2000–2009 Nationwide Health Insurance database. J. Formos. Med. Assoc. 2012, 111, 599–604. [Google Scholar] [CrossRef]
- Tseng, C.J.; Lu, C.J.; Chang, C.C.; Chen, G.D.; Cheewakriangkrai, C. Integration of data mining classification techniques and ensemble learning to identify risk factors and diagnose ovarian cancer recurrence. Artif. Intell. Med. 2017, 78, 47–54. [Google Scholar] [CrossRef] [PubMed]
- van den Munckhof, I.C.; Jones, H.; Hopman, M.T.; de Graaf, J.; Nyakayiru, J.; van Dijk, B.; Eijsvogels, T.M.; Thijssen, D.H. Relation between age and carotid artery intima-medial thickness: A systematic review. Clin. Cardiol. 2018, 41, 698–704. [Google Scholar] [CrossRef]
- Boulos, N.M.; Gardin, J.M.; Malik, S.; Postley, J.; Wong, N.D. Carotid Plaque Characterization, Stenosis, and Intima-Media Thickness According to Age and Gender in a Large Registry Cohort. Am. J. Cardiol. 2016, 117, 1185–1191. [Google Scholar] [CrossRef]
- Paramsothy, P.; Knopp, R.H.; Bertoni, A.G.; Blumenthal, R.S.; Wasserman, B.A.; Tsai, M.Y.; Rue, T.; Wong, N.D.; Heckbert, S.R. Association of combinations of lipid parameters with carotid intima-media thickness and coronary artery calcium in the MESA (Multi-Ethnic Study of Atherosclerosis). J. Am. Coll. Cardiol. 2010, 56, 1034–1041. [Google Scholar] [CrossRef]
- Abd El-Hafez, H.; Elrakhawy, M.M.; El-Baiomy, A.A.; El-Eshmawy, M.M. Carotid Intima Media Thickness Is Independently Associated with Male Gender, Middle Age, and IGF-1 in Metabolically Healthy Obese Individuals. ISRN Obes. 2014, 2014, 545804. [Google Scholar] [CrossRef] [PubMed]
- Ogbera, A.O. Prevalence and gender distribution of the metabolic syndrome. Diabetol. Metab. Syndr. 2010, 2, 1. [Google Scholar] [CrossRef]
- Yang, Y.M.; Shin, B.C.; Son, C.; Ha, I.H. An analysis of the associations between gender and metabolic syndrome components in Korean adults: A national cross-sectional study. BMC Endocr. Disord. 2019, 19, 67. [Google Scholar] [CrossRef]
- Kastarinen, H.; Ukkola, O.; Kesäniemi, Y.A. Glomerular filtration rate is related to carotid intima-media thickness in middle-aged adults. Nephrol. Dial. Transplant. 2009, 24, 2767–2772. [Google Scholar] [CrossRef]
- Kawamoto, R.; Ohtsuka, N.; Kusunoki, T.; Yorimitsu, N. An association between the estimated glomerular filtration rate and carotid atherosclerosis. Intern. Med. 2008, 47, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Gentile, M.; Panico, S.; Mattiello, A.; de Michele, M.; Iannuzzi, A.; Jossa, F.; Marotta, G.; Rubba, P. Plasma creatinine levels, estimated glomerular filtration rate and carotid intima media thickness in middle-aged women: A population based cohort study. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 677–680. [Google Scholar] [CrossRef] [PubMed]
- Hirano, T. Abnormal lipoprotein metabolism in diabetic nephropathy. Clin. Exp. Nephrol. 2014, 18, 206–209. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Ma, X.; Ren, X.L.; Xiao, H.; Yan, L.; Li, Z.; Wang, S. The Role of Blood Pressure in Carotid Plaque Incidence: Interactions With Body Mass Index, Age, and Sex-Based on a 7-Years Cohort Study. Front. Physiol. 2021, 12, 690094. [Google Scholar] [CrossRef] [PubMed]
- Lou, Y.; Li, B.; Su, L.; Mu, Z.; Sun, M.; Gu, H.; Ni, J.; Wu, Y.; Tu, J.; Wang, J.; et al. Association between body mass index and presence of carotid plaque among low-income adults aged 45 years and older: A population-based cross-sectional study in rural China. Oncotarget 2017, 8, 81261–81272. [Google Scholar] [CrossRef]
- Stevens, J.; Tyroler, H.A.; Cai, J.; Paton, C.C.; Folsom, A.R.; Tell, G.S.; Schrener, P.J.; Chambless, L.E. Body weight change and carotid artery wall thickness. The Atherosclerosis Risk in Communities (ARIC) Study. Am. J. Epidemiol. 1998, 147, 563–573. [Google Scholar] [CrossRef]
- Rajala, U.; Päivänsalo, M.; Laakso, M.; Pelkonen, O.; Suramo, I.; Keinänen-Kiukaanniemi, S. Associations of blood pressure with carotid intima-media thickness in elderly Finns with diabetes mellitus or impaired glucose tolerance. J. Hum. Hypertens. 2003, 17, 705–711. [Google Scholar] [CrossRef]
- Saif, A.; Karawya, S.; Abdelhamid, A. Blood pressure is a risk factor for progression of diabetic retinopathy in normotensive patients with type 2 diabetes: Correlation with carotid intima-media thickness. Endocr. Regul. 2014, 48, 189–194. [Google Scholar] [CrossRef]
- Itoh, H.; Kaneko, H.; Kiriyama, H.; Yoshida, Y.; Nakanishi, K.; Mizuno, Y.; Daimon, M.; Morita, H.; Yatomi, Y.; Komuro, I. Relation between the Updated Blood Pressure Classification according to the American College of Cardiology/American Heart Association Guidelines and Carotid Intima-Media Thickness. Am. J. Cardiol. 2019, 124, 396–401. [Google Scholar] [CrossRef]
- Liu, J.; Lin, Q.; Guo, D.; Yang, Y.; Zhang, X.; Tu, J.; Ning, X.; Song, Y.; Wang, J. Association Between Pulse Pressure and Carotid Intima-Media Thickness Among Low-Income Adults Aged 45 Years and Older: A Population-Based Cross-Sectional Study in Rural China. Front. Cardiovasc. Med. 2020, 7, 547365. [Google Scholar] [CrossRef]
- Yokoyama, H.; Katakami, N.; Yamasaki, Y. Recent advances of intervention to inhibit progression of carotid intima-media thickness in patients with type 2 diabetes mellitus. Stroke 2006, 37, 2420–2427. [Google Scholar] [CrossRef] [PubMed]
- Wagenknecht, L.E.; D’Agostino, R., Jr.; Savage, P.J.; O’Leary, D.H.; Saad, M.F.; Haffner, S.M. Duration of diabetes and carotid wall thickness. The Insulin Resistance Atherosclerosis Study (IRAS). Stroke 1997, 28, 999–1005. [Google Scholar] [CrossRef] [PubMed]
- Tönnies, T.; Stahl-Pehe, A.; Baechle, C.; Castillo, K.; Kuss, O.; Yossa, R.; Lindner, L.M.; Holl, R.W.; Rosenbauer, J. Risk of Microvascular Complications and Macrovascular Risk Factors in Early-Onset Type 1 Diabetes after at Least 10 Years Duration: An Analysis of Three Population-Based Cross-Sectional Surveys in Germany between 2009 and 2016. Int. J. Endocrinol. 2018, 2018, 7806980. [Google Scholar] [CrossRef] [PubMed]
- Bjerg, L.; Gudbjörnsdottir, S.; Franzén, S.; Carstensen, B.; Witte, D.R.; Jørgensen, M.E.; Svensson, A.M. Duration of diabetes-related complications and mortality in type 1 diabetes: A national cohort study. Int. J. Epidemiol. 2021, 50, 1250–1259. [Google Scholar] [CrossRef]
- Bebu, I.; Braffett, B.H.; Schade, D.; Sivitz, W.; Malone, J.I.; Pop-Busui, R.; Lorenzi, G.M.; Lee, P.; Trapani, V.R.; Wallia, A.; et al. An Observational Study of the Equivalence of Age and Duration of Diabetes to Glycemic Control Relative to the Risk of Complications in the Combined Cohorts of the DCCT/EDIC Study. Diabetes Care 2020, 43, 2478–2484. [Google Scholar] [CrossRef]
- Zoungas, S.; Woodward, M.; Li, Q.; Cooper, M.E.; Hamet, P.; Harrap, S.; Heller, S.; Marre, M.; Patel, A.; Poulter, N.; et al. Impact of age, age at diagnosis and duration of diabetes on the risk of macrovascular and microvascular complications and death in type 2 diabetes. Diabetologia 2014, 57, 2465–2474. [Google Scholar] [CrossRef]
- Sadasivam, K.; Nagarajan, P.; Durai, I.; Sundari, M.; Ayyavoo, S.; Ramamoorthy, T. Carotid Artery Intima-Media Thickness in Young Adults with Family History of Coronary Artery Disease. J. Clin. Diagn. Res. 2015, 9, CC01–CC04. [Google Scholar] [CrossRef]
- Polak, J.F.; Pencina, M.J.; Pencina, K.M.; O’Donnell, C.J.; Wolf, P.A.; D’Agostino, R.B., Sr. Carotid-wall intima-media thickness and cardiovascular events. N. Engl. J. Med. 2011, 365, 213–222. [Google Scholar] [CrossRef]

| Men | Women | |
|---|---|---|
| n | 495 | 429 |
| Age (y) *** | 63.8 ± 10.9 | 66.5 ± 10.0 |
| Smoking (n (%)) | 191 (38.5%) | 15 (3.5%) |
| Alcoholic drinking (n (%)) | 53 (10.7%) | 1 (0.2%) |
| BMI (Kg/m2) | 26.3 ± 3.6 | 26.6 ± 4.4 |
| Duration of diabetes (y) | 15.3 ± 7.5 | 15.6 ± 7.8 |
| HgbA1c (%) | 7.5 ± 1.5 | 7.5 ± 1.3 |
| TG (mg/dL) | 140.0 ± 120.1 | 133.0 ± 72.6 |
| HDL-C (mg/dL) *** | 41.7 ± 11.1 | 47.4 ± 10.7 |
| LDL-C (mg/dL) | 96.6 ± 26.6 | 97.5 ± 28.3 |
| ALT (U/dL) *** | 31.5 ± 22.1 | 25.6 ± 16.5 |
| Creatinine (mg/dL) * | 1.05 ± 0.48 | 0.76 ± 0.55 |
| SBP (mmHg) | 130.0 ± 13.3 | 130.9 ± 14.6 |
| DBP (mmHg) * | 76.5 ± 8.2 | 74.5 ± 15.1 |
| Microalbuminuria (mg/g) | 180.4 ± 689.2 | 114.0 ± 606.5 |
| Normal | Abnormal | |
|---|---|---|
| n | 710 | 214 |
| Age (y) *** | 63.4 ± 10.3 | 70.6 ± 9.5 |
| Smoking (n (%)) | 150 (21.1%) | 56 (26.2%) |
| Alcoholic drinking (n (%)) | 47 (6.6%) | 7 (3.3%) |
| BMI (Kg/m2) ** | 26.6 ± 4.0 | 25.8 ± 3.6 |
| Duration of diabetes (y) | 14.8 ± 7.1 | 17.4 ± 9.0 |
| HgbA1c (%) | 7.5 ± 1.4 | 7.5 ± 1.4 |
| TG (mg/dL) | 138.5 ± 108.1 | 131.1 ± 74.9 |
| HDL-C (mg/dL) | 44.6 ± 10.9 | 43.4 ± 12.3 |
| LDL-C (mg/dL) | 97.2 ± 27.9 | 96.2 ± 25.8 |
| ALT (U/dL) | 25.2 ± 19.7 | 27.4 ± 20.7 |
| Creatinine (mg/dL) *** | 0.87 ± 0.51 | 1.05 ± 0.59 |
| SBP (mmHg) | 130.0 ± 14.0 | 132.0 ± 13.4 |
| DBP (mmHg) * | 76.2 ± 12.8 | 73.7 ± 8.2 |
| Microalbuminuria (mg/g) | 129.8 ± 613.5 | 216.9 ± 767.7 |
| Accuracy | Sensitivity | Specificity | AUC | |
|---|---|---|---|---|
| Logit | 0.669 ± 0.081 | 0.682 ± 0.116 | 0.665 ± 0.134 | 0.692 ± 0.030 |
| CART | 0.523 ± 0.082 | 0.488 ± 0.040 | 0.532 ± 0.112 | 0.511 ± 0.036 |
| RF | 0.703 ± 0.071 | 0.622 ± 0.122 | 0.724 ± 0.132 | 0.692 ± 0.036 |
| XGBoost | 0.716 ± 0.048 | 0.616 ± 0.123 | 0.742 ± 0.097 | 0.688 ± 0.030 |
| NB | 0.683 ± 0.059 | 0.664 ± 0.094 | 0.688 ± 0.101 | 0.692 ± 0.029 |
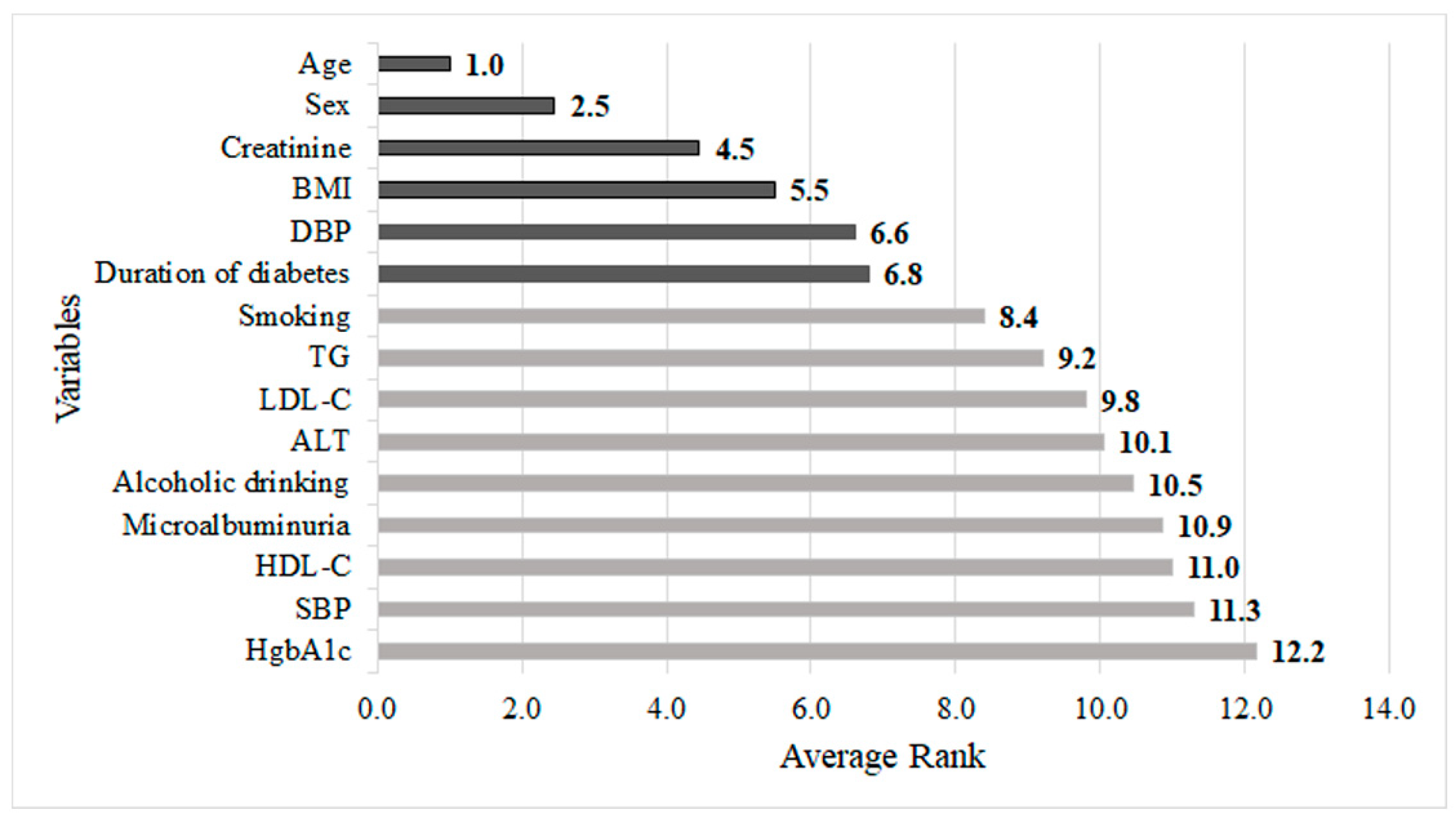
| Logit | CART | RF | XGBoost | NB | ||
|---|---|---|---|---|---|---|
| Sex | 2.0 | 2.0 | 2.4 | 1.0 | 2.5 | |
| Age | 1.0 | 6.0 | 1.0 | 2.0 | 1.0 | |
| BMI | 7.8 | 9.0 | 7.1 | 3.0 | 3.3 | |
| Duration of diabetes | 12.3 | 5.0 | 7.0 | 4.0 | 7.1 | |
| Smoking | 7.4 | 3.0 | 5.4 | 5.0 | 11.4 | |
| Alcoholic drinking | 10.2 | 1.0 | 9.8 | 6.0 | 10.5 | |
| HgbA1c | 10.2 | 7.0 | 11.4 | 7.0 | 13.2 | |
| TG | 8.1 | 4.0 | 11.7 | 8.0 | 7.4 | |
| HDL-C | 11.9 | 8.0 | 11.0 | 9.0 | 11.1 | |
| LDL-C | 13.1 | 15.0 | 10.2 | 10.0 | 9.2 | |
| ALT | 5.4 | 15.0 | 8.1 | 11.4 | 11.5 | |
| Creatinine | 10.4 | 15.0 | 3.5 | 12.3 | 5.4 | |
| Microalbuminuria | 11.4 | 15.0 | 12.1 | 13.2 | 10.3 | |
| SBP | 5.7 | 15.0 | 9.6 | 14.4 | 12.4 | |
| DBP | 3.1 | 15.0 | 9.7 | 15 | 3.7 | |
| Rank value | 1.0~1.4 | 1.5~2.4 | 2.5~3.4 | 3.5~4.4 | 4.5~5.4 | 5.5~ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, C.-Z.; Huang, L.-Y.; Chen, F.-Y.; Kuo, C.-H.; Yeih, D.-F. Using Machine Learning to Predict Abnormal Carotid Intima-Media Thickness in Type 2 Diabetes. Diagnostics 2023, 13, 1834. https://doi.org/10.3390/diagnostics13111834
Wu C-Z, Huang L-Y, Chen F-Y, Kuo C-H, Yeih D-F. Using Machine Learning to Predict Abnormal Carotid Intima-Media Thickness in Type 2 Diabetes. Diagnostics. 2023; 13(11):1834. https://doi.org/10.3390/diagnostics13111834
Chicago/Turabian StyleWu, Chung-Ze, Li-Ying Huang, Fang-Yu Chen, Chun-Heng Kuo, and Dong-Feng Yeih. 2023. "Using Machine Learning to Predict Abnormal Carotid Intima-Media Thickness in Type 2 Diabetes" Diagnostics 13, no. 11: 1834. https://doi.org/10.3390/diagnostics13111834
APA StyleWu, C.-Z., Huang, L.-Y., Chen, F.-Y., Kuo, C.-H., & Yeih, D.-F. (2023). Using Machine Learning to Predict Abnormal Carotid Intima-Media Thickness in Type 2 Diabetes. Diagnostics, 13(11), 1834. https://doi.org/10.3390/diagnostics13111834









