Transvaginal Ultrasound vs. Magnetic Resonance Imaging (MRI) Value in Endometriosis Diagnosis
Abstract
1. Introduction
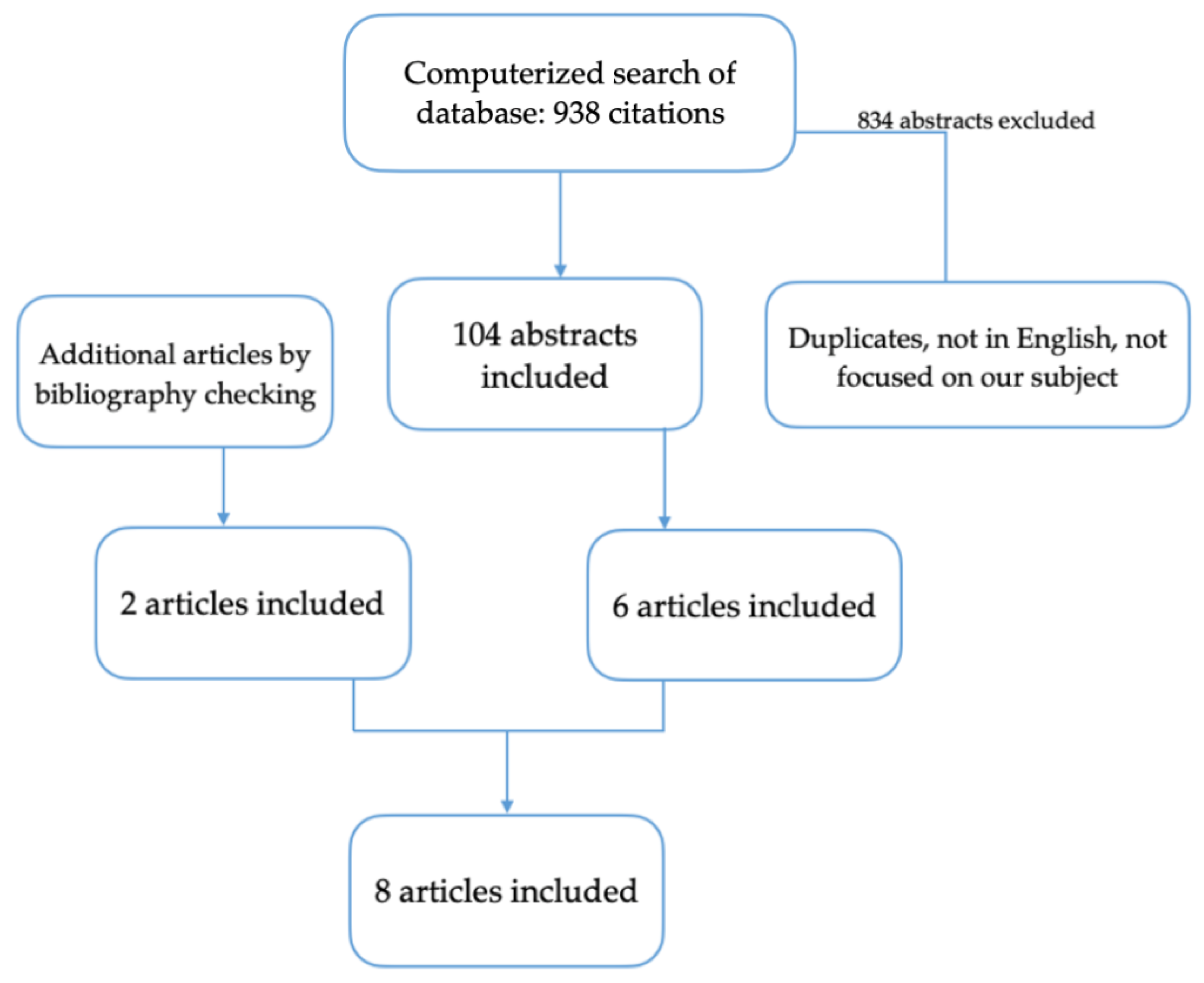
2. Materials and Methods
3. Results
3.1. Diagnosis Methods
3.1.1. Ultrasonography Overview
- Regular uterine and adnexal examinations (with sonographic evidence of adenomyosis or ovarian cysts).
- Transvaginal ultrasound “soft markers” evaluation (i.e., site-specific tenderness and adnexal mobility).
- POD status is evaluated by utilizing a real-time ultrasound “sliding sign”.
- Examination of the anterior and posterior compartments for DE nodules [38].
3.1.2. Magnetic Resonance Imaging Overview
3.1.3. Comparison between the Diagnostic Accuracy of TVS and MRI
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Johnson, N.P.; Hummelshoj, L. World Endometriosis Society Montpellier Consortium. Consensus on current management of endometriosis. Hum Reprod. 2013, 28, 1552–1568. [Google Scholar] [CrossRef] [PubMed]
- Frincu, F.; Carp-Veliscu, A.; Petca, A.; Badiu, D.-C.; Bratila, E.; Cirstoiu, M.; Mehedintu, C. Maternal–Fetal Outcomes in Women with Endometriosis and Shared Pathogenic Mechanisms. Medicina 2021, 57, 1258. [Google Scholar] [CrossRef] [PubMed]
- Missmer, S.A.; Hankinson, S.E.; Spiegelman, D.; Barbieri, R.L.; Marshall, L.M.; Hunter, D.J. Incidence of laparoscopically confirmed endometriosis by demographic, anthropometric, and lifestyle factors. Am. J. Epidemiol. 2004, 160, 784–796. [Google Scholar] [CrossRef] [PubMed]
- Sampson, J.A. Peritoneal endometriosis due to menstrual dissemination of endometrial tissue into the peritoneal cavity. Am. J. Obstet. Gynecol. 1927, 14, 442–469. [Google Scholar] [CrossRef]
- Tammaa, A.; Fritzer, N.; Strunk, G.; Krell, A.; Salzer, H.; Hudelist, G. Learning curve for the detection of pouch of Douglas obliteration and deep infiltrating endometriosis of the rectum. Hum. Reprod. 2014, 29, 1199–1204. [Google Scholar] [CrossRef]
- Nisenblat, V.; Bossuyt, P.M.; Farquhar, C.; Johnson, N.; Hull, M.L. Imaging modalities for the non-invasive diagnosis of endometriosis. Cochrane Database Syst. Rev. 2016, 2, CD009591. [Google Scholar] [CrossRef]
- Koninckx, P.R.; Meuleman, C.; Demeyere, S.; Lesaffre, E.; Cornillie, F.J. Suggestive evidence that pelvic endometriosis is a progressive disease, whereas deeply infiltrating endometriosis is associated with pelvic pain. Fertil. Steril. 1991, 55, 759–765. [Google Scholar] [CrossRef]
- Saccardi, C.; Cosmi, E.; Borghero, A.; Tregnaghi, A.; Dessole, S.; Litta, P. Comparison between transvaginal sonography, saline contrast sonovaginography and magnetic resonance imaging in the diagnosis of posterior deep infiltrating endometriosis. Ultrasound Obstet. Gynecol. 2012, 40, 464–469. [Google Scholar] [CrossRef]
- Hudelist, G.; Ballard, K.; English, J.; Wright, J.; Banerjee, S.; Mastoroudes, H.; Thomas, A.; Singer, C.F.; Keckstein, J. Transvaginal sonography vs. clinical examination in the preoperative diagnosis of deep infiltrating endometriosis. Ultrasound Obstet. Gynecol. 2011, 37, 480–487. [Google Scholar] [CrossRef]
- Cazalis, C.G.; Koskas, M.; Martin, B.; Palazzo, L.; Madelenat, P.; Yazbeck, C. Imagerie préopératoire dans l’endométriose profonde: Échographie pelvienne, écho-endoscopie rectale et IRM. Gynécologie Obs. Fertil. 2012, 40, 634–641. [Google Scholar] [CrossRef]
- Alborzi, S.; Rasekhi, A.; Shomali, Z.; Madadi, G.; Alborzi, M.; Kazemi, M.; Nohandani, A.H. Diagnostic accuracy of magnetic resonance imaging, transvaginal, and transrectal ultrasonography in deep infiltrating endometriosis. Medicine 2018, 97, e9536. [Google Scholar] [CrossRef] [PubMed]
- Bazot, M.; Lafont, C.; Rouzier, R.; Roseau, G.; Thomassin-Naggara, I.; Daraï, E. Diagnostic accuracy of physical examination, transvaginal sonography, rectal endoscopic sonography, and magnetic resonance imaging to diagnose deep infiltrating endometriosis. Fertil. Steril. 2009, 92, 1825–1833. [Google Scholar] [CrossRef] [PubMed]
- Saba, L.; Guerriero, S.; Sulis, R.; Pilloni, M.; Ajossa, S.; Melis, G.; Mallarini, G. Learning curve in the detection of ovarian and deep endometriosis by using Magnetic Resonance: Comparison with surgical results. Eur. J. Radiol. 2011, 79, 237–244. [Google Scholar] [CrossRef]
- Krüger, K.; Behrendt, K.; Niedobitek-Kreuter, G.; Koltermann, K.; Ebert, A.D. Location-dependent value of pelvic MRI in the preoperative diagnosis of endometriosis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2013, 169, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Indrielle-Kelly, T.; Frühauf, F.; Fanta, M.; Burgetova, A.; Lavu, D.; Dundr, P.; Cibula, D.; Fischerova, D. Diagnostic Accuracy of Ultrasound and MRI in the Mapping of Deep Pelvic Endometriosis Using the International Deep Endometriosis Analysis (IDEA) Consensus. BioMed Res. Int. 2020, 2020, 3583989. [Google Scholar] [CrossRef]
- Zondervan, K.T.; Becker, C.M.; Missmer, S.A. Endometriosis. N. Engl. J. Med. 2020, 382, 1244–1256. [Google Scholar] [CrossRef] [PubMed]
- Lopes, L.; Hindman, N.; Huang, K. Accuracy of Magnetic Resonance Imaging (MRI) in the Diagnosis of Endometriosis-Evaluation of an Institutional Protocol. J. Minim. Invasive Gynecol. 2015, 22, S52–S54. [Google Scholar] [CrossRef]
- Abrao, M.S.; Gonçalves, M.O.; Dias JAJr Podgaec, S.; Chamie, L.P.; Blasbalg, R. Comparison between clinical examination, transvaginal sonography and magnetic resonance imaging for the diagnosis of deep endometriosis. Hum. Reprod. 2007, 22, 3092–3097. [Google Scholar] [CrossRef]
- Agarwal, S.K.; Chapron, C.; Giudice, L.C.; Laufer, M.R.; Leyland, N.; Missmer, S.A.; Singh, S.S.; Taylor, H.S. Clinical diagnosis of endometriosis: A call to action. Am. J. Obstet. Gynecol. 2019, 220, 354.e1–354.e12. [Google Scholar] [CrossRef]
- Ballard, K.D.; Seaman, H.E.; De Vries, C.S.; Wright, J.T. Can symptomatology help in the diagnosis of endometriosis? Findings from a national case–control study–part 1. BJOG 2008, 115, 1382–1391. [Google Scholar] [CrossRef]
- Denny, E.; Mann, C.H. Endometriosis and the primary care consultation. Eur. J. Obstet. Gynecol. Reprod. Biol. 2008, 139, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, S.; Bergqvist, A.; Chapron, C.; D’Hooghe, T.; Dunselman, G.; Greb, R.; Hummelshoj, L.; Prentice, A.; Saridogan, E. ESHRE guideline for the diagnosis and treatment of endometriosis. Hum. Reprod. 2005, 20, 2698–2704. [Google Scholar] [CrossRef]
- Prusova, K.; Churcher, L.; Tyler, A.; Lokugamage, A.U. Royal College of Obstetricians and Gynaecologists guidelines: How evidence-based are they? J. Obstet. Gynaecol. 2014, 34, 706–711. [Google Scholar] [CrossRef] [PubMed]
- Soliman, A.M.; Coyne, K.S.; Zaiser, E.; Castelli-Haley, J.; Fuldeore, M.J. The burden of endometriosis symptoms on health-related quality of life in women in the United States: A cross-sectional study. J. Psychosom. Obstet. Gynaecol. 2017, 38, 238–248. [Google Scholar] [CrossRef] [PubMed]
- Bielen, D.; Tomassetti, C.; Van Schoubroeck, D.; Vanbeckevoort, D.; De Wever, L.; Van den Bosch, T.; D’Hooghe, T.; Bourne, T.; D’Hoore, A.; Wolthuis, A.; et al. IDEAL study: Magnetic resonance imaging for suspected deep endometriosis assessment prior to laparoscopy is as reliable as radiological imaging as a complement to transvaginal ultrasonography. Ultrasound Obstet. Gynecol. 2020, 56, 255–266. [Google Scholar] [CrossRef]
- Capozzi, V.A.; Merisio, C.; Rolla, M.; Pugliese, M.; Morganelli, G.; Cianciolo, A.; Gambino, G.; Armano, G.; Sozzi, G.; Ricco, M.; et al. Confounding factors of transvaginal ultrasound accuracy in endometrial cancer. J. Obstet. Gynaecol. 2021, 41, 779–784. [Google Scholar] [CrossRef]
- Bazot, M.; Thomassin, I.; Hourani, R.; Cortez, A.; Darai, E. Diagnostic accuracy of transvaginal sonography for deep pelvic endometriosis. Ultrasound Obstet. Gynecol. 2004, 24, 180–185. [Google Scholar] [CrossRef]
- Kupfer, M.C.; Schwimer, S.R.; Lebovic, J. Transvaginal sonographic appearance of endometriomata: Spectrum of findings. J. Ultrasound Med. 1992, 11, 129–133. [Google Scholar] [CrossRef]
- Hoyle, A.T.; Puckett, Y. Endometrioma. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Ghezzi, F.; Raio, L.; Cromi, A.; Duwe, D.G.; Beretta, P.; Buttarelli, M.; Mueller, M.D. “Kissing ovaries”: A sonographic sign of moderate to severe endometriosis. Fertil. Steril. 2005, 83, 143–147. [Google Scholar] [CrossRef]
- Carneiro, M.M.; Filogônio, I.D.; Costa, L.M.; de Ávila, I.; Ferreira, M.C. Clinical prediction of deeply infiltrating endometriosis before surgery: Is it feasible? A review of the literature. Biomed. Res. Int. 2013, 2013, 564153. [Google Scholar] [CrossRef][Green Version]
- Hudelist, G.; Oberwinkler, K.H.; Singer, C.F.; Tuttlies, F.; Rauter, G.; Ritter, O.; Keckstein, J. Combination of transvaginal sonography and clinical examination for preoperative diagnosis of pelvic endometriosis. Hum. Reprod. 2009, 24, 1018–1024. [Google Scholar] [CrossRef] [PubMed]
- Lorusso, F.; Scioscia, M.; Rubini, D.; Stabile Ianora, A.A.; Scardigno, D.; Leuci, C.; De Ceglie, M.; Sardaro, A.; Lucarelli, N.; Scardapane, A. Magnetic resonance imaging for deep infiltrating endometriosis: Current concepts, imaging technique and key findings. Insights Imaging 2021, 12, 105. [Google Scholar] [CrossRef] [PubMed]
- Guerriero, S.; Ajossa, S.; Gerada, M.; D’Aquila, M.; Piras, B.; Melis, G.B. “Tenderness-guided” transvaginal ultrasonography: A new method for the detection of deep endometriosis in patients with chronic pelvic pain. Fertil. Steril. 2007, 88, 1293–1297. [Google Scholar] [CrossRef] [PubMed]
- Holland, T.K.; Cutner, A.; Saridogan, E.; Mavrelos, D.; Pateman, K.; Jurkovic, D. Ultrasound mapping of pelvic endometriosis: Does the location and number of lesions affect the diagnostic accuracy? A multicentre diagnostic accuracy study. BMC Womens Health 2013, 13, 43. [Google Scholar] [CrossRef]
- Guerriero, S.; Ajossa, S.; Gerada, M.; Virgilio, B.; Angioni, S.; Melis, G.B. Diagnostic value of transvaginal ‘tenderness-guided’ ultrasonography for the prediction of location of deep endometriosis. Hum. Reprod. 2008, 23, 2452–2457. [Google Scholar] [CrossRef]
- Lee, S.-Y.; Koo, Y.-J.; Lee, D.-H. Classification of endometriosis. Yeungnam Univ. J. Med. 2021, 38, 10–18. [Google Scholar] [CrossRef]
- Guerriero, S.; Condous, G.; Van den Bosch, T.; Valentin, L.; Leone, F.P.G.; Van Schoubroeck, D.; Exacoustos, C.; Installé, A.J.F.; Martins, W.P.; Abrao, M.S.; et al. Systematic approach to sonographic evaluation of the pelvis in women with suspected endometriosis, including terms, definitions and measurements: A consensus opinion from the International Deep Endometriosis Analysis (IDEA) group: IDEA consensus opinion. Ultrasound Obstet. Gynecol. 2016, 48, 318–322. [Google Scholar] [CrossRef]
- Menakaya, U.; Infante, F.; Lu, C.; Phua, C.; Model, A.; Messyne, F.; Brainwood, M.; Reid, S.; Condous, G. Interpreting the real-time dynamic ‘sliding sign’ and predicting pouch of Douglas obliteration: An interobserver, intraobserver, diagnostic-accuracy and learning-curve study. Ultrasound Obstet Gynecol. 2016, 48, 113–120. [Google Scholar] [CrossRef]
- Fraser, M.A.; Agarwal, S.; Chen, I.; Singh, S.S. Routine vs. expert-guided transvaginal ultrasound in the diagnosis of endometriosis: A retrospective review. Abdom. Imaging 2015, 40, 587–594. [Google Scholar] [CrossRef]
- Hudelist, G.; English, J.; Thomas, A.E.; Tinelli, A.; Singer, C.F.; Keckstein, J. Diagnostic accuracy of transvaginal ultrasound for non-invasive diagnosis of bowel endometriosis: Systematic review and meta-analysis. Ultrasound Obstet. Gynecol. 2011, 37, 257–263. [Google Scholar] [CrossRef]
- Coutinho, A., Jr.; Bittencourt, L.K.; Pires, C.E.; Junqueira, F.; Lima, C.M.; Coutinho, E.; Domingues, M.A.; Domingues, R.C.; Marchiori, E. MR imaging in deep pelvic endometriosis: A pictorial essay. Radiographics 2011, 31, 549–567. [Google Scholar] [CrossRef] [PubMed]
- Bazot, M.; Darai, E.; Hourani, R.; Thomassin, I.; Cortez, A.; Uzan, S.; Buy, J.N. Deep pelvic endometriosis: MR imaging for diagnosis and prediction of extension of disease. Radiology 2004, 232, 379–389. [Google Scholar] [CrossRef] [PubMed]
- Cheewadhanaraks, S.; Peeyananjarassri, K.; Dhanaworavibul, K.; Liabsuetrakul, T. Valoarea predictivă pozitivă a diagnosticului clinic al endometriozei. J. Med. Conf. Univ. Thai. 2004, 87, 740–744. [Google Scholar]
- Bruyere, C.; Maniou, I.; Habre, C.; Kalovidouri, A.; Pluchino, N.; Montet, X.; Botsikas, D. Pelvic MRI for Endometriosis: A Diagnostic Challenge for the Inexperienced Radiologist. How Much Experience Is Enough? Acad. Radiol. 2021, 28, 345–353. [Google Scholar] [CrossRef] [PubMed]
- De Venecia, C.; Ascher, S.M. Pelvic Endometriosis: Spectrum of Magnetic Resonance Imaging Findings. Semin. Ultrasound CT MRI 2015, 36, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.; Daniels, I.R.; Richardson, C.; Revell, P.; Peppercorn, D.; Bourne, M. Techniques and trouble-shooting in high spatial resolution thin slice MRI for rectal cancer. Br. J. Radiol. 2005, 78, 245–251. [Google Scholar] [CrossRef]
- Bazot, M.; Bharwani, N.; Huchon, C.; Kinkel, K.; Cunha, T.M.; Guerra, A.; Manganaro, L.; Buñesch, L.; Kido, A.; Togashi, K.; et al. European society of urogenital radiology [ESUR] guidelines: MR imaging of pelvic endometriosis. Eur. Radiol. 2017, 27, 2765–2775. [Google Scholar] [CrossRef]
- Fiaschetti, V.; Crusco, S.; Meschini, A.; Cama, V.; Di Vito, L.; Marziali, M.; Piccione, E.; Calabria, F.; Simonetti, G. Deeply infiltrating endometriosis: Evaluation of retro-cervical space on MRI after vaginal opacification. Eur. J. Radiol. 2012, 81, 3638–3645. [Google Scholar] [CrossRef]
- Scardapane, A.; Lorusso, F.; Francavilla, M.; Bettocchi, S.; Fascilla, F.D.; Angelelli, G.; Scioscia, M. Magnetic resonance colonography may predict the need for bowel resection in colorectal endometriosis. Biomed. Res. Int. 2017, 2017, 5981217. [Google Scholar] [CrossRef]
- Craig, E.V.; Shannon, L.M.; Andreotti, R.F. The Complementary Role of Ultrasound and Magnetic Resonance Imaging in the Evaluation of Endometriosis: A Review. Ultrasound Q. 2020, 36, 123–132. [Google Scholar] [CrossRef]
- Dias, J.L.; Veloso Gomes, F.; Lucas, R.; Cunha, T.M. The shading sign: Is it exclusive of endometriomas? Abdom. Imaging 2015, 40, 2566–2572. [Google Scholar] [CrossRef] [PubMed]
- Lupean, R.A.; Ștefan, P.A.; Csutak, C.; Lebovici, A.; Măluțan, A.M.; Buiga, R.; Melincovici, C.S.; Mihu, C.M. Differentiation of Endometriomas from Ovarian Hemorrhagic Cysts at Magnetic Resonance: The Role of Texture Analysis. Medicina 2020, 56, 487. [Google Scholar] [CrossRef] [PubMed]
- Bazot, M.; Gasner, A.; Ballester, M.; Daraï, E. Value of thin-section oblique axial T2-weighted magnetic resonance images to assess uterosacral ligament endometriosis. Hum. Reprod. 2011, 26, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Foti, P.V.; Farina, R.; Palmucci, S.; Vizzini, I.A.A.; Libertini, N.; Coronella, M.; Spadola, S.; Caltabiano, R.; Iraci, M.; Basile, A.; et al. Endometriosis: Clinical features, MR imaging findings and pathologic correlation. Insights Imaging 2018, 9, 149–172. [Google Scholar] [CrossRef]
- Yoon, J.H.; Choi, D.; Jang, K.T.; Kim, C.K.; Kim, H.; Lee, S.J.; Chun, H.K.; Lee, W.Y.; Yun, S.H. Deep rectosigmoid endometriosis: “mushroom cap” sign on T2-weighted MR imaging. Abdom. Imaging 2010, 35, 726–731. [Google Scholar] [CrossRef]
- Loubeyre, P.; Petignat, P.; Jacob, S.; Egger, J.F.; Dubuisson, J.B.; Wenger, J.M. Anatomic distribution of posterior deeply infiltrating endometriosis on MRI after vaginal and rectal gel opacification. Am. J. Roentgenol. 2009, 192, 1625–1631. [Google Scholar] [CrossRef]
- Tian, Z.; Zhang, Y.C.; Sun, X.H.; Wang, Y.; Zhao, Y.; Chang, X.H.; Zhu, H.L. Accuracy of transvaginal ultrasound and magnetic resonance imaging for diagnosis of deep endometriosis in bladder and ureter: A meta-analysis. J. Obstet. Gynaecol. 2022, 1–10. [Google Scholar] [CrossRef]
- Zhang, X.; He, T.; Shen, W. Comparison of physical examination, ultrasound techniques and magnetic resonance imaging for the diagnosis of deep infiltrating endometriosis: A systematic review and meta-analysis of diagnostic accuracy studies. Exp. Ther. Med. 2020, 20, 3208–3220. [Google Scholar] [CrossRef]
- Kaponis, A.; Taniguchi, F.; Azuma, Y.; Deura, I.; Vitsas, C.; Decavalas, G.O.; Harada, T. Current treatment of endometrioma. Obstet. Gynecol. Surv. 2015, 70, 183–195. [Google Scholar] [CrossRef]
- Rousset, P.; Bischoff, E.; Charlot, M.; Grangeon, F.; Dubernard, G.; Paparel, P.; Lega, J.C.; Golfier, F. Bladder endometriosis: Preoperative MRI analysis with assessment of extension to ureteral orifices. Diagn. Interv. Imaging 2021, 102, 255–263. [Google Scholar] [CrossRef]
- Jha, P.; Sakala, M.; Chamie, L.P.; Feldman, M.; Hindman, N.; Huang, C.; Kilcoyne, A.; Laifer-Narin, S.; Nicola, R.; Poder, L.; et al. Endometriosis MRI lexicon: Consensus statement from the society of abdominal radiology endometriosis disease-focused panel. Abdom. Radiol. 2020, 45, 1552–1568. [Google Scholar] [CrossRef] [PubMed]
- Dunselman, G.A.; Vermeulen, N.; Becker, C.; Calhaz-Jorge, C.; D’Hooghe, T.; De Bie, B.; Heikinheimo, O.; Horne, A.W.; Kiesel, L.; Nap, A.; et al. European Society of Human Reproduction and Embryology. ESHRE guideline: Management of women with endometriosis. Hum. Reprod. 2014, 29, 400–412. [Google Scholar] [CrossRef] [PubMed]
- Wild, M.; Pandhi, S.; Rendle, J.; Swift, I.; Ofuasia, E. MRI for the diagnosis and staging of deeply infiltrating endometriosis: A national survey of BSGE accredited endometriosis centres and review of the literature. Br. J. Radiol. 2020, 93, 20200690. [Google Scholar] [CrossRef] [PubMed]
- Hudelist, G.; Fritzer, N.; Thomas, A.; Niehues, C.; Oppelt, P.; Haas, D.; Tammaa, A.; Salzer, H. Diagnostic delay for endometriosis in Austria and Germany: Causes and possible consequences. Hum. Reprod. 2012, 27, 3412–3416. [Google Scholar] [CrossRef]
- Mehedintu, C.; Frincu, F.; Brinduse, L.A.; Carp-Veliscu, A.; Bratila, E.; Hennetier, C.; Roman, H. Postoperative Assessment of the Quality of Life in Patients with Colorectal Endometriosis. J. Clin. Med. 2021, 10, 5211. [Google Scholar] [CrossRef]
- Deguara, C.S.; Pepas, L.; Davis, C. Does minimally invasive surgery for endometriosis improve pelvic symptoms and quality of life? Curr. Opin. Obstet. Gynecol. 2012, 24, 241–244. [Google Scholar] [CrossRef]
- La Verde, M.; Riemma, G.; Tropea, A.; Biondi, A.; Cianci, S. Ultra-minimally invasive surgery in gynecological patients: A review of the literature. Updates Surg. 2022, 74, 843–855. [Google Scholar] [CrossRef]
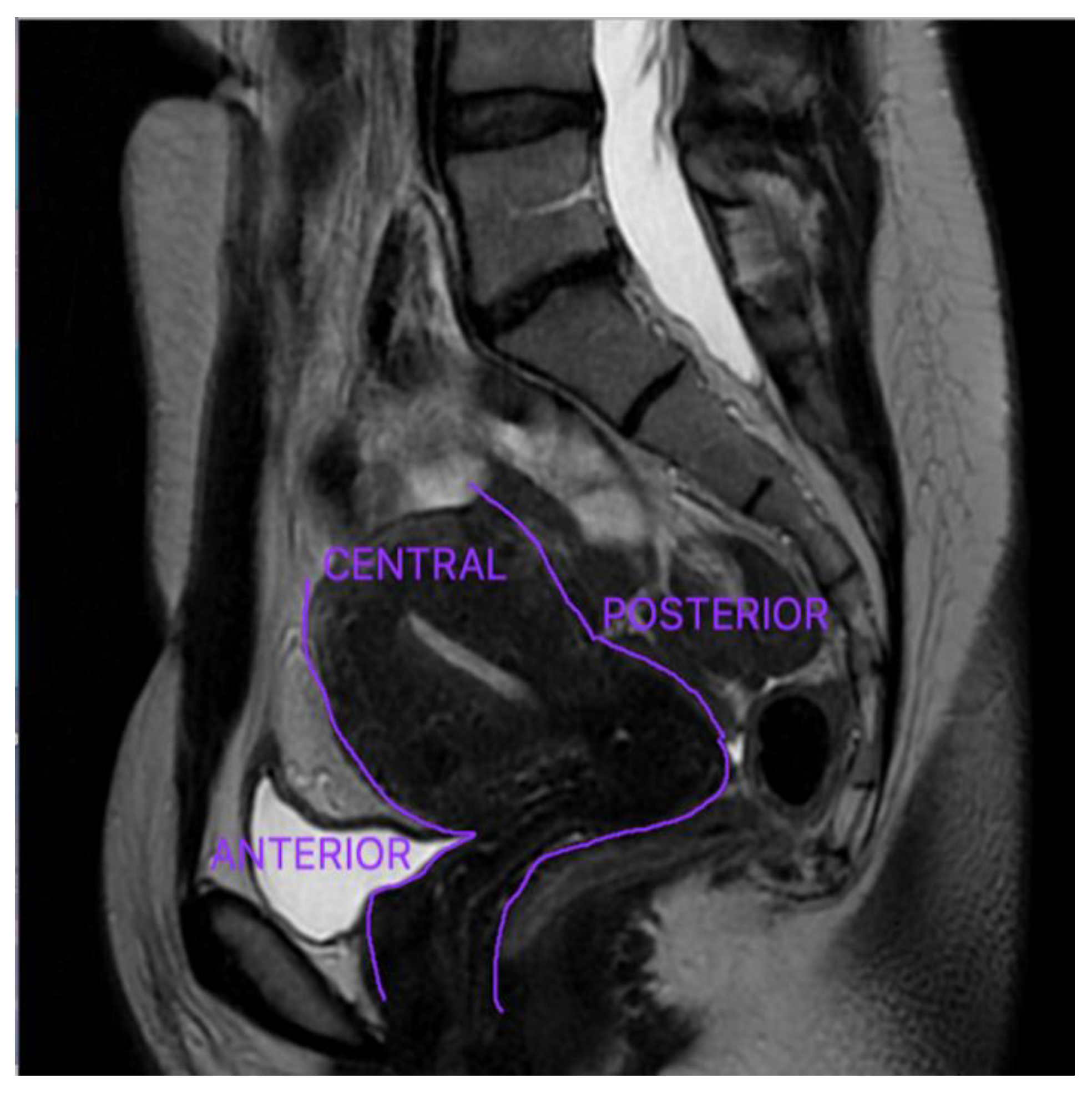
| Reference | Number of Patients | Locations of DE | Imaging Techniques | Mean Age | Year of Publication |
|---|---|---|---|---|---|
| Hudelist et al. [9] | 126 | Ovaries, uterosacral ligaments, rectovaginal space, bladder, rectosigmoid | TVS | 32.2 | 2011 |
| Cazalis et al. [10] | 25 | Ovaries, uterosacral ligaments, rectovaginal space, bladder, rectosigmoid | TVS + MRI | 35.4 | 2012 |
| Alborzi et al. [11] | 317 | Ovaries, uterosacral ligaments, rectovaginal space, bladder, rectosigmoid | TVS + MRI | 31 | 2018 |
| Bazot et al. [12] | 92 | Uterosacral ligaments, rectovaginal space, rectosigmoid | TVS + MRI | 31.8 | 2009 |
| Idrielle et al. [15] | 49 | Uterosacral ligaments, rectovaginal space, bladder, rectosigmoid | TVS + MRI | - | 2019 |
| Kruger et al. [14] | 152 | Uterosacral ligaments, ovaries, bladder, rectosigmoid | MRI | 33.5 | 2013 |
| Saccardi et al. [8] | 102 | Uterosacral ligaments, rectovaginal space | TVS + MRI | 32.3 | 2012 |
| Saba et al. [13] | 30 | Ovaries, uterosacral ligaments, rectosigmoid | MRI | 34 | 2011 |
| Ovaries | TVS | MRI | ||
|---|---|---|---|---|
| Sensitivity | Specificity | Sensitivity | Specificity | |
| Hudelist et al. [9] | 96% | 96% | - | - |
| Cazalis et al. [10] | 88.2% | 71% | 87.5% | 71% |
| Alborzi et al. [11] | 70.86% | 92.7% | 63.5% | 93.9% |
| Saba et al. [13] | - | - | 92.6% | 91.3% |
| Kruger et al. [14] | - | - | 86.3% | 73.6% |
| USL | TVS | MRI | ||
|---|---|---|---|---|
| Sensitivity | Specificity | Sensitivity | Specificity | |
| Hudelist et al. [9] | 63% | 98% | - | - |
| Cazalis et al. [10] | 63% | 82.6% | 69% | 82.6% |
| Alborzi et al. [11] | 70.8% | 92.7% | 63.5% | 93.9% |
| Bazot et al. [12] | 78.3% | 66.7% | 84.4% | 88.9% |
| Idrielle et al. [15] | 74% | 67% | 94% | 60% |
| Kruger et al. [14] | - | - | 77% | 68% |
| Saccardi et al. [8] | 55.6% | 95.6% | 95.6% | 75% |
| Saba et al. [13] | - | - | 80% | 84.6% |
| Rectovaginal Space | TVS | MRI | ||
|---|---|---|---|---|
| Sensitivity | Specificity | Sensitivity | Specificity | |
| Hudelist et al. [9] | 64% | 99% | - | - |
| Cazalis et al. [10] | 63.2% | 100% | 47.4% | 100% |
| Alborzi et al. [11] | 86.3% | 94.8% | 95.2% | 71.1% |
| Bazot et al. [12] | 9% | 98.7% | 54.5% | 98.7% |
| Idrielle et al. [15] | 67% | 100% | 83% | 93% |
| Saccardi et al. [8] | 63.9% | 88.9% | 83.3% | 77.8% |
| Bladder | TVS | MRI | ||
|---|---|---|---|---|
| Sensitivity | Specificity | Sensitivity | Specificity | |
| Hudelist et al. [9] | 50% | 98% | - | - |
| Cazalis et al. [10] | 16.7% | 100% | 33.3% | 89.5% |
| Alborzi et al. [11] | 100% | 99.6% | 100% | 99.6% |
| Idrielle et al. [15] | 89% | 100% | 100% | 95% |
| Kruger et al. [14] | - | - | 81% | 94% |
| Rectosigmoid | TVS | MRI | ||
|---|---|---|---|---|
| Sensitivity | Specificity | Sensitivity | Specificity | |
| Hudelist et al. [9] | 90% | 99% | - | - |
| Cazalis et al. [10] | 73.7% | 66.7% | 89.5% | 50% |
| Alborzi et al. [11] | 88.4% | 98.8% | 76.9% | 96.6% |
| Bazot et al. [12] | 93.6% | 100% | 87.3% | 93.1% |
| Idrielle et al. [15] | 94% | 84% | 94% | 84% |
| Kruger et al. [14] | - | - | 80% | 77% |
| Saba et al. [13] | - | - | 73.9% | 83.9% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baușic, A.; Coroleucă, C.; Coroleucă, C.; Comandașu, D.; Matasariu, R.; Manu, A.; Frîncu, F.; Mehedințu, C.; Brătilă, E. Transvaginal Ultrasound vs. Magnetic Resonance Imaging (MRI) Value in Endometriosis Diagnosis. Diagnostics 2022, 12, 1767. https://doi.org/10.3390/diagnostics12071767
Baușic A, Coroleucă C, Coroleucă C, Comandașu D, Matasariu R, Manu A, Frîncu F, Mehedințu C, Brătilă E. Transvaginal Ultrasound vs. Magnetic Resonance Imaging (MRI) Value in Endometriosis Diagnosis. Diagnostics. 2022; 12(7):1767. https://doi.org/10.3390/diagnostics12071767
Chicago/Turabian StyleBaușic, Alexandra, Ciprian Coroleucă, Cătălin Coroleucă, Diana Comandașu, Roxana Matasariu, Andrei Manu, Francesca Frîncu, Claudia Mehedințu, and Elvira Brătilă. 2022. "Transvaginal Ultrasound vs. Magnetic Resonance Imaging (MRI) Value in Endometriosis Diagnosis" Diagnostics 12, no. 7: 1767. https://doi.org/10.3390/diagnostics12071767
APA StyleBaușic, A., Coroleucă, C., Coroleucă, C., Comandașu, D., Matasariu, R., Manu, A., Frîncu, F., Mehedințu, C., & Brătilă, E. (2022). Transvaginal Ultrasound vs. Magnetic Resonance Imaging (MRI) Value in Endometriosis Diagnosis. Diagnostics, 12(7), 1767. https://doi.org/10.3390/diagnostics12071767








