1. Introduction
Standard visual perception results from a single photon response, delimited in spectral sensitivity of 380 to 700 nm [
1]. Recently, a new concept of visual perception has been proposed, which involves the simultaneous absorption of two photons instead of one [
2,
3]. Pulsed infrared (IR) laser can trigger two photon absorption and produce color sensation corresponding to half of the wavelength of the stimulating beam (e.g., blue or green) [
2,
3,
4] despite that high-power IR emitters are typically seen as red or colorless in single-photon perception [
5]. The relationship between the brightness and the power of the stimulating beam is quadratic for the two-photon perceived stimuli. This attribute of two-photon vision may be advantageous, especially for the accuracy of visual threshold determination. A recent laboratory study demonstrated that the spread of the psychometric function for detecting two-photon stimuli was narrower (by a factor of 2) than for single-photon testing [
3]. The repeatability of two-photon visual threshold measurements was also improved compared to standard (visible-light) stimuli [
3]. Although both types of photoreceptors mediate two-photon vision [
2], studies of dark adaptation curves measured at 520 nm and 1040 nm have shown that the sensitivity difference between the two photoreceptors is substantially smaller than for a single-photon process [
3]. The pupil response is also significantly smaller for two-photon versus standard targets having the same color and brightness [
6]. Since the two-photon visibility threshold is a new parameter to assess retinal function, its clinical application has only recently been sought [
4,
7,
8].
In an initial study, we established the normative level of two-photon IR light sensitivity in adults aged 20 to 70 [
4]. We subsequently demonstrated that diabetic retinopathy patients have significantly decreased retinal sensitivity to IR light compared to their age-matched controls [
7]. A potential implementation of this new technology in the management of retinal diseases was proposed [
4,
7]. Additionally, in a recent analysis, the two-photon assessment has proven supportive in the detection of glaucomatous neuropathy [
8]. We found a significant correlation between optical coherence tomography (OCT) retinal biomarkers and IR light sensitivity, suggesting another application area.
Age-related macular degeneration (AMD) is a neurodegenerative disease affecting the macula, and it can result in severe loss of central vision [
9,
10]. Given the progressive, irreversible damage to photoreceptors and their subsequent atrophy, one may expect that the two-photon absorption is compromised in equal measure to the damage made by AMD to normal vision. This degenerative disease also affects other layers of the retina, e.g., retinal pigment epithelium (RPE), Bruch’s membrane, and choriocapillaris [
10]. A higher level of lipofuscin and decreased concentration of melanosomes have been reported as potential biomarkers of AMD [
11]. An increased number of confluent extracellular drusen, geographic atrophy (GA) of the RPE resulting in the loss of photoreceptors, and retinal pigment epithelial detachments are typical morphological features of nonexudative AMD. The exudative form is characterized by subretinal neovascularization due to abnormal growth of retinal blood vessels associated with the presence of subretinal fluid [
10].
Although such changes are often seen in standard fundoscopy, routinely performed by clinicians, the diagnosis and management of AMD patients are commonly supported by imaging techniques [
9]. Fundus photography has been particularly useful in disease-progression monitoring by providing a baseline image for subsequent appointments. The inclusion of spectral filters, e.g., autofluorescence imaging or fundus angiography, offers additional tools for improved detection of eye pathology [
9]. The introduction of OCT proved an important milestone in advancing AMD diagnosis as OCT increased the depth of examination by allowing a non-invasive assessment of the retinal anatomical structure. These advances in ophthalmology extended our understanding of the pathomechanism of various retinal disorders and expanded clinicians’ range of diagnostic tools. OCT proved valuable in diagnosing exudative AMD because with its high spatial resolution, even small subretinal fluid pockets or choroidal neovascularization can be detected [
9].
Microperimetry, which involves retinal sensitivity testing coupled with fundus imaging, has added a functional component to the morphological assessment [
12]. As opposed to standard (static) perimetry, microperimetry covers a narrow portion of the visual field, typically limited to the macula. Following a microperimetric examination, an exact retinal location of test points and measured sensitivity are superimposed over the patient’s fundus recording, which establishes a link between structure and function [
12]. Microperimetry has been applied successfully in studying the functional effects of AMD and the efficacy of treatment methods beyond standard visual acuity (VA) [
12,
13,
14,
15]. Fundus-driven perimetry has primarily been performed in a single-photon realm. However, the transition to two-photon retinal sensitivity may prove advantageous given its higher tolerance to eye turbidity and the nonlinearity of two-photon vision, which may yield more accurate testing [
3].
In this study, we measured retinal sensitivity in AMD patients with standard visible-light microperimetry and a two-photon approach and compared their performance against a control group to evaluate the application of two-photon technology in the assessment of AMD.
3. Results
The study population comprised 45 subjects: 23 in the AMD group and 22 controls. Of the 23 AMD eyes, five were classified as grade I, seven as grade II, and 11 as grade III. The median (IQR) age of the AMD subjects was 77.3 (72.2; 79.9) years and 71.6 (68.0; 77.9) years for the controls, which did not yield a statistically significant difference (p = 0.07). BCVA was compromised in the disease group compared to non-AMD patients, which was 0.15 (0.05; 0.24) logMAR vs. −0.02 (−0.06; 0.02) logMAR. Median spherical equivalent was 0.00 D in both groups. Similarly, the level of straylight was also comparable, as we found 1.17 (1.06; 1.45) log(s) in AMD and 1.18 (1.01; 1.29) log(s) in normal eyes.
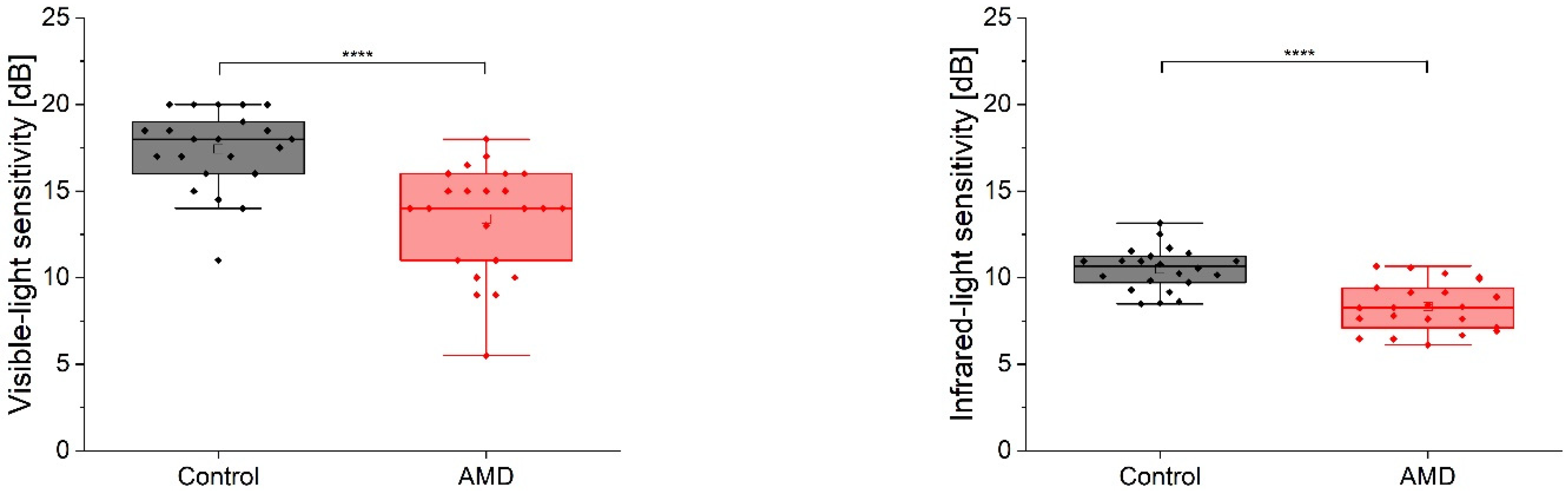
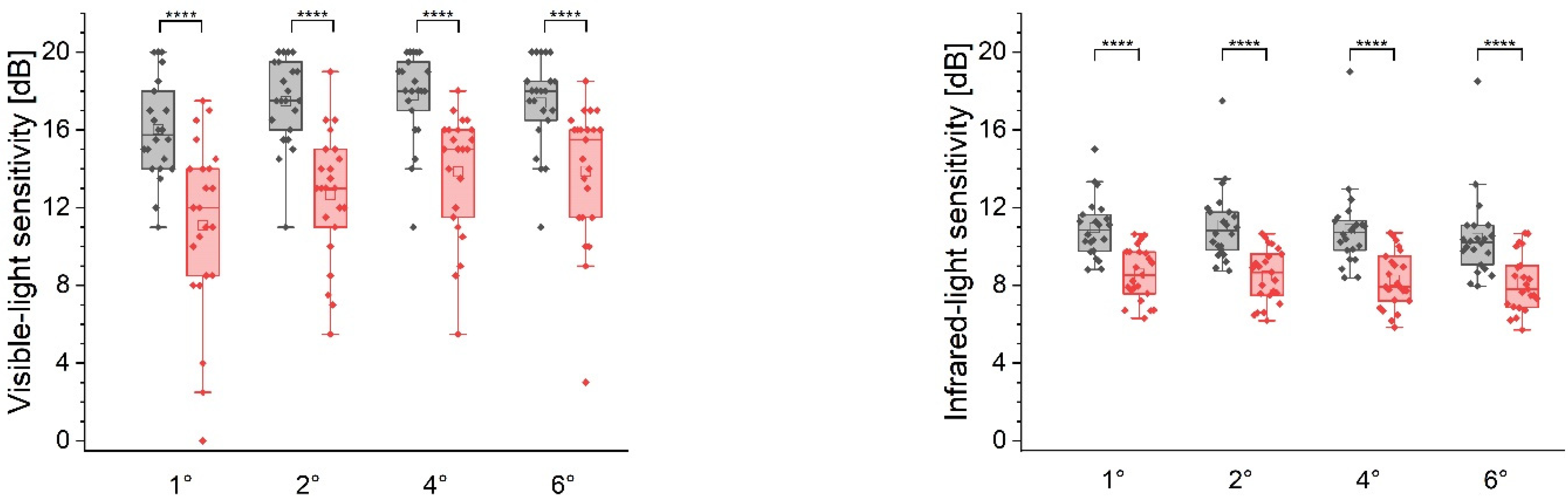
The median sensitivity to IR light was 8.3 (7.2, 9.3) dB in the AMD cases and 10.7 (9.7, 11.2) dB in the normal population. The MP1 assessment showed 14.0 (11.0; 15.5) dB for disease and 18.0 (16.3; 18.9) dB for control subjects. Both comparisons (
Figure 4) yielded statistically significant differences (
p < 0.001).
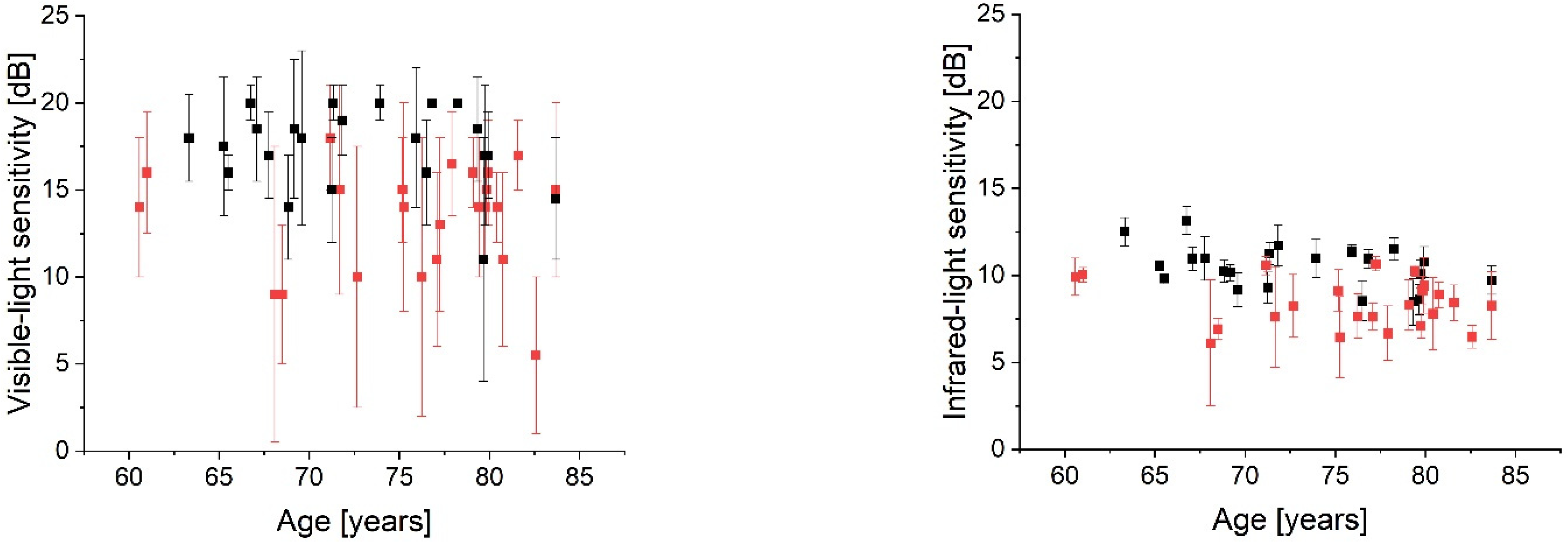
Figure 5 shows the age distribution of the (pooled) retinal sensitivity. The correlation coefficient (ρ) found in the control group was −0.35 for IR- and −0.11 for visible-light; however, it did not reach significance level (
p > 0.05) for both approaches. An increased intrasubject variability (manifested by a higher IQR) can be observed in the MP1 results of both cohorts (
Figure 5).
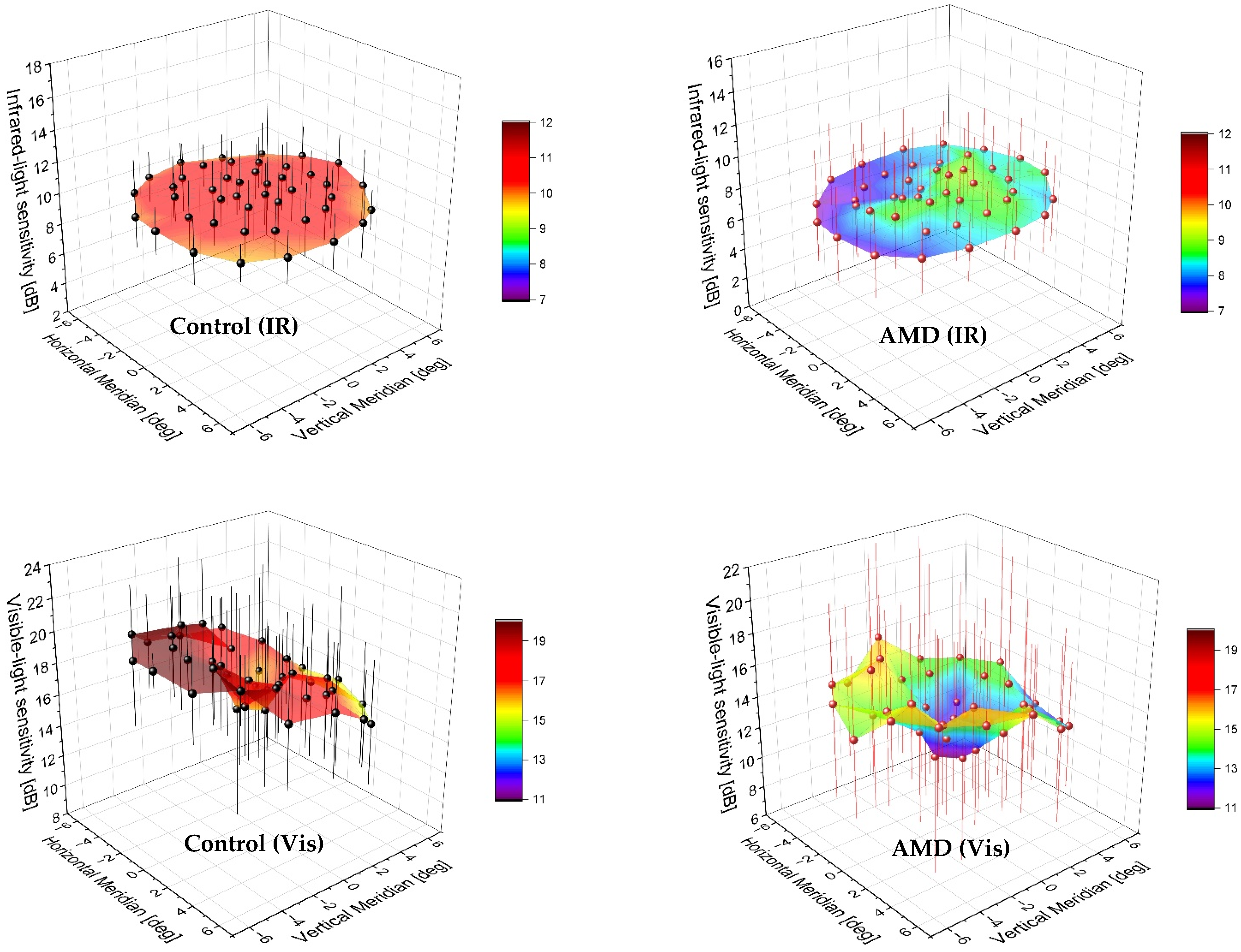
The distribution of sensitivity in each retinal locus is visualized in
Figure 6. Both methods confirmed the debilitating effect of AMD on retinal sensitivity with a decreased distribution profile. The two-photon map of the controls produced a nearly uniform data spread with a slight increase in the center. Note that sensitivity was not tested at the fixation point.
The presence of AMD upsets this uniform distribution, showing a sensitivity peak shift and a valley starting near 1° and extending temporally to 6°. The MP1 measurements of the normal and AMD eyes yielded a depression in the center of the testing area. A tilt of the surface map indicated nonuniformity of retinal sensitivity results, which was less pronounced in AMD. Although the IQR recorded in the AMD cases was comparable between the two methods, the visible-light microperimetry appeared to exhibit higher intersubject variability in the control group.
The stratification of measurement points based on their radial distance from the fovea is summarized in
Figure 7. The control subjects had the highest IR light sensitivity along the 1° and 2° radii (r) and demonstrated a gradual decrease with a 0.8 dB difference occurring between 1° and 6°. By contrast, the AMD patients showed lower values at r = 1° compared to r = 2° by 0.3 dB. Beyond this point, the sensitivity reduction was reminiscent of our observations in the controls. The radial distribution of the MP1 results exhibits a different pattern. Normal eyes’ sensitivity was the lowest at r = 1° with a gradual increase up to 4° by 2.3 dB and a minimal (0.6 dB) decrease at 6°. A similar trend was maintained in the AMD population. However, the sensitivity values were significantly lower and demonstrated a higher IQR.
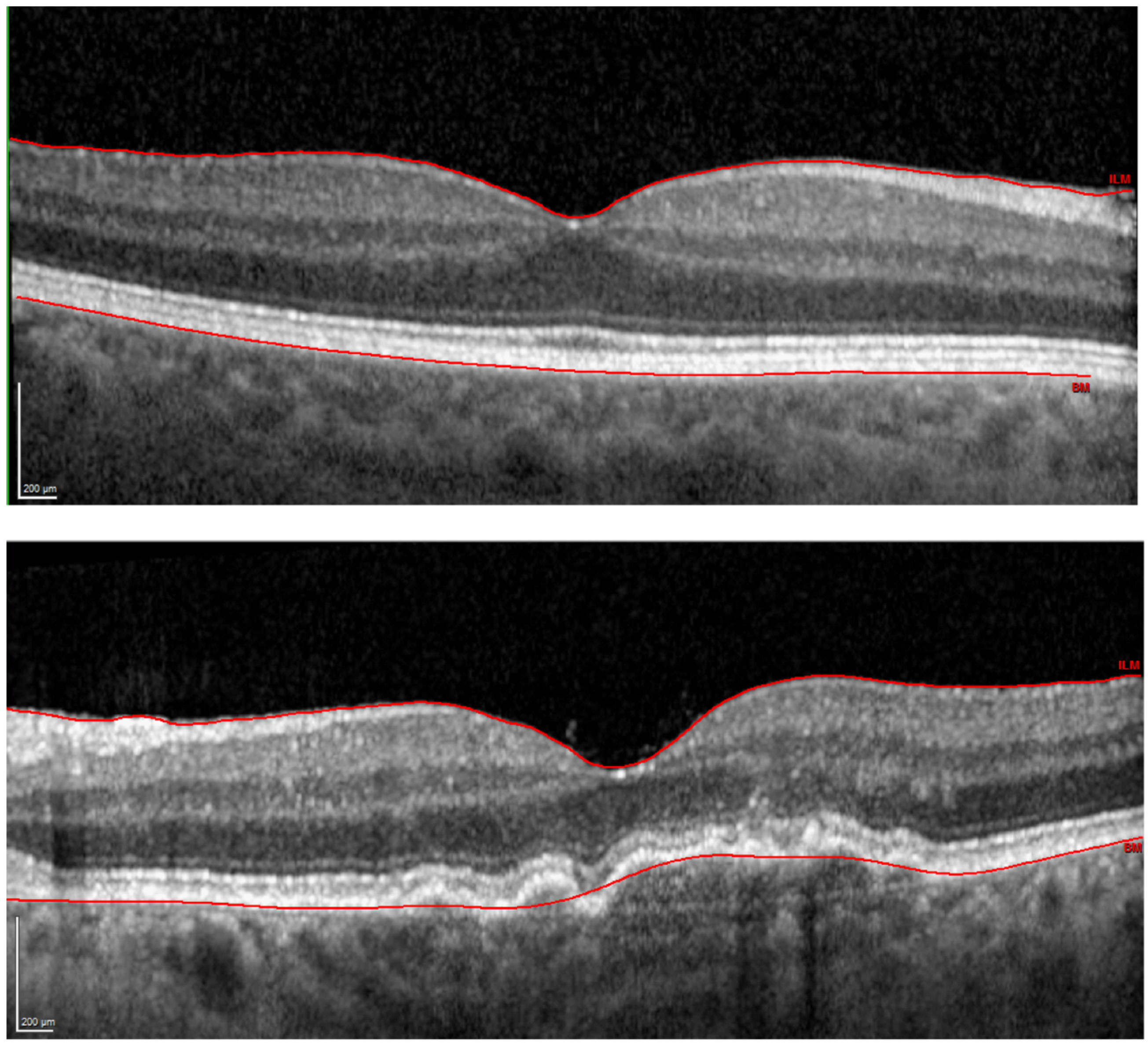
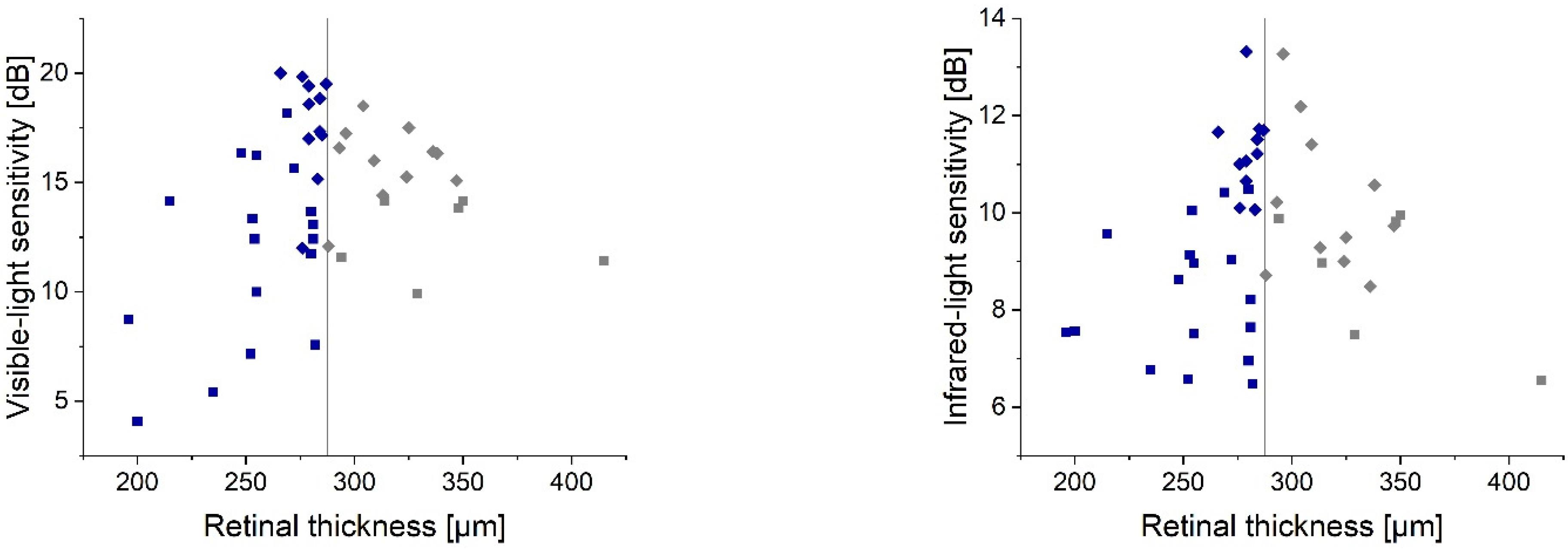
The AMD eyes’ central subfield thickness was 272.0 (252.5; 288) µm, which was lower than that found in the controls: 287.5 (280; 312.0) µm. The median value of the control group provided a reference for the macular thickness vs. sensitivity plot (
Figure 8) to create two subgroups for statistical analysis. Retinal thinning showed a significant correlation with the loss of retinal sensitivity detected with visible (ρ = 0.43,
p = 0.02) and IR light (ρ = 0.48,
p = 0.01). However, the increase of the central subfield thickness demonstrated a lower correlation (ρ
vis = 0.26 and ρ
IR = 0.31), which did not reach the significance level.
4. Discussion
We demonstrated a significant reduction in two-photon retinal sensitivity in eyes with AMD. Although we could observe a similar outcome in standard single-photon microperimetry, this method appears to be more affected by inter- and intrasubject variability.
Standard visible-light microperimetry is a popular screening option for visual-function evaluation in AMD patients beyond VA. Dinc et al. performed an analogous comparison between normal and AMD patients [
13]. They recruited 30 patients diagnosed with intermediate AMD and a VA of 0.20 logMAR or better. The MP1 microperimeter used in their study had a Goldmann III size stimulus and a 1.27 cd/m
2 background, which are standard settings also applied in our investigation, but they used a larger grid consisting of 76 retinal loci extending to 20° [
13], whereas we assessed the retinal function at 44 points across 12°. They reported a mean sensitivity (±standard deviation) of 12.7 ± 2.8 dB in AMD patients and 18.0 ± 0.6 dB in age-matched controls. Although we found a similar median level in our controls, the AMD subjects were reported by Dinc and co-workers to have lower retinal sensitivity by 1.3 dB, despite being nearly 10 years younger. Their finding may indicate that any age effect may be suppressed by AMD severity, which appears to be the primary factor affecting retinal sensitivity in those patients. Still, the values reported by Dinc et al. fall within a broad IQR outlining our population, which again indicates a substantial variability in visual function between AMD patients, primarily determined by their unique course of the disease [
13]. One may assume that these factors also affect two-photon retinal sensitivity.
Late-stage AMD, characterized by GA and choroidal neovascularization, has the most substantial compromising impact on retinal sensitivity. Takahashi et al. enrolled 25 patients with nonexudative AMD and GA for a comprehensive morphological and visual function examination [
15]. Microperimetry was performed with the MP1 device over 57 points unevenly distributed within 10° from the center. They applied color fundus photography, SLO imaging with fundus autofluorescence, and spectral-domain OCT in order to assess the retinal structure [
15]. They reported a worse BCVA (0.40 logMAR) than that of the current study and found a reduced retinal sensitivity of 8.42 ± 4.24 dB. This value decreased to 3.28 ± 2.70 dB at confined areas of photoreceptor damage and further to 1.84 ± 2.68 dB in regions with identified retinal pigment epithelium loss. Therefore, they established a clear link between retinal morphology and visual function in GA patients [
15]. Munk et al. assessed patients with neovascular AMD in a longitudinal study of the effect of monthly intravitreal anti-vascular endothelial growth factor (VEGF) therapy on visual function [
14]. MP1 microperimetry was performed at 14 follow-up visits scheduled within 12 months of a monthly administration of an anti-VEGF agent. A baseline VA of their 61 patients who completed that study was 0.61 ± 032 logMAR, which improved to 0.46 ± 0.36 logMAR at 12 months. Retinal sensitivity was measured within an identical angular range of 12° as applied in our investigation, but with a different (non-radial) shape of their 33-point grid pattern [
14]. The mean value of 7.3 ± 4.5 dB that had been recorded before the initial injection was decreased two-fold compared to what we found in our population. A gradual improvement of retinal sensitivity was observed in the course of that study with 10.8 ± 4.5 dB at the last visit. However, between the fourth and 12th months, mean retinal sensitivity varied between 10.6 dB and 11.6 dB, which might have resulted from a higher variability of MP1. Whether the two-photon IR light stimulation, which demonstrated a narrower spread of sensitivity values, proves more advantageous in longitudinal studies requires further research.
Higher repeatability of the two-photon approach compared to a standard one-photon stimulation was demonstrated by Rumiński et al. [
3] They studied a psychometric function with an optical setup featuring visible- (522.5 nm) and IR light (1045 nm) paths for single- and two-photon stimulation. Although one (IR) light source was used for both methods, visible light was a product of second-harmonic generation by a nonlinear crystal added to the visible path. Thus, only the wavelength of stimuli differed between the two conditions. Both, however, were perceived as green. Rumiński et al. reported that the psychometric function derived from IR stimuli had a two-fold (on a log scale) steeper slope than that obtained with visible light, which indicates a lower spread of values [
3]. Indeed, they found that 99.7% of recorded changes fell within ±1.1 dB for two-photon testing while single-photo perception yielded ±2.2 dB. It was suggested that a higher accuracy of IR light testing results from a nonlinear behavior of the two-photon absorption process [
3]. Our investigation also noted a lower spread of retinal sensitivity values in two-photon testing compared to the single-photon approach, which for the AMD and control patient was 2.1-fold and 1.7-fold, respectively. Since it was close to a two-fold difference (expected from the square ratio), this study may also support the nonlinearity of the two-photon process [
3]. However, our assessment was performed on two different devices, which despite the attempts to mirror the MP1 conditions in the two-photon realm, still may not account for all existing confounders [
19].
One potential explanation for increased IQR obtained with the MP1 device in the normal population is the nonuniformity of the device background. Given that our testing spanned 6° from the fixation point, one may assume that a healthy eye has an equal radial-density distribution of photoreceptors and thus retinal sensitivity. It was confirmed in the IR light testing (
Figure 6). The MP1 approach demonstrated, however, a titled surface of the sensitivity map, which may suggest background intensity fluctuations over the test area. Another discrepancy between the two methods was observed in the central area. The visualization of the IR light result shows the so-called “hill of vision” with a higher sensitivity at 1° and a gradual decrease with eccentricity indicating cone-mediated vision. Although the two-photon absorption can also activate rods, cones achieve greater efficiency in this process [
3,
6]. Therefore, this method might be better suited for sensitivity tests performed in photopic conditions. Despite identical background illuminance, the single-photon sensitivity map showed lower values in the center than at higher angles. This might be caused by a higher contribution of rods whose density increases with eccentricity to reach a peak at approximately 20° [
20]. Alternatively, radiation from the fixation circle confounded measurements at 1° despite being set at 0.5°. We used the first-generation microperimeter in our study on account of its availability at the Heidelberg Eye Clinic. We have yet to address how the latest MP3 device compares to the two-photon excitation device.
Two-photon sensitivity is a new parameter that has only recently been introduced with the first clinical outcomes reported in 2020 [
4], where the age-dependency of IR light sensitivity was studied by our group in a normal population. As a proof-of-concept, five AMD subjects were also included and measured following the same protocol of 30-min dark adaptation [
4]. The median scotopic sensitivity was 17.9 (17.0; 19.1) dB in young, healthy participants and 9.6 (9.4; 11.2) dB in AMD patients, yielding a difference of 8.3 dB. In the current evaluation, IR light sensitivity was reduced only by 2.4 dB in the diseased eyes. The reason for a smaller effect might be an insignificant age difference observed here, while in the earlier study, the median value of the healthy population was 44.1 (31.6; 53.1) years. Another explanation is faster degradation and loss of rods than cones in AMD [
21,
22], which may result in the compromised visual function of dark-adapted eyes. Therefore, performing dark-adapted microperimetry with two-photon stimuli may prove a more sensitive measure of disease progression and its functional effect. This conjecture, however, should be validated in a clinical study.
The assessment of retinal morphology in AMD patients has improved our knowledge about the pathomechanism of this disease [
9,
10]. A routine application of OCT in AMD patient management has led to the introduction of various retinal biomarkers, which can predict the disease’s progression and a patient’s functional outcome [
23]. Central retinal thickness is one parameter that is readily accessible to clinicians as it is automatically generated in each OCT macular report. Although OCT devices may differ in anatomical landmarks used for thickness measurements, a clear correlation has been observed between retinal thickness changes and eye sensitivity [
24,
25]. Alexander et al. assessed 14 neovascular AMD patients during anti-VEGF therapy [
24]. They studied the association between the OCT-derived central retinal thickness and microperimetry results, showing a coefficient of determination (R
2) of 0.69 for the nonlinear fit of their data clusters. Alexander et al. also demonstrated that an excessive increase or decrease in retinal thickness results in degradation of the visual function [
24], which is consistent with the findings of our analysis. A potential explanation is that retinal thinning may indicate atrophy, while active neovascularization may increase retinal thickness [
24]. Both conditions yield a significant reduction of retinal sensitivity. However, predicting visual function by measuring retinal thickness would appear to have limited application given the broad range of median sensitivity points along a vertical line demarcating the reference level. The Spearman’s rank correlation coefficient also indicates a worse prediction value in cases with increased retinal thickness, perhaps because only a few study cases presented subretinal fluid. However, in the report by Sabour-Pickett et al., a significant correlation was noted with a Pearson correlation coefficient of −0.59 for their entire cohort of patients treated with anti-VEFG intravitreal injections due to retinal fluid build-up or cyst formation [
25]. Thus, in a more uniformly phenotyped population, the predicting value of the retinal thickness parameter may increase. Interestingly, despite differences in the mediation of two- and single-photon vision, both instruments appear to have a comparable predictive value.