Placental and Umbilical Cord Anomalies Diagnosed by Two- and Three-Dimensional Ultrasound
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Placental Anomalies
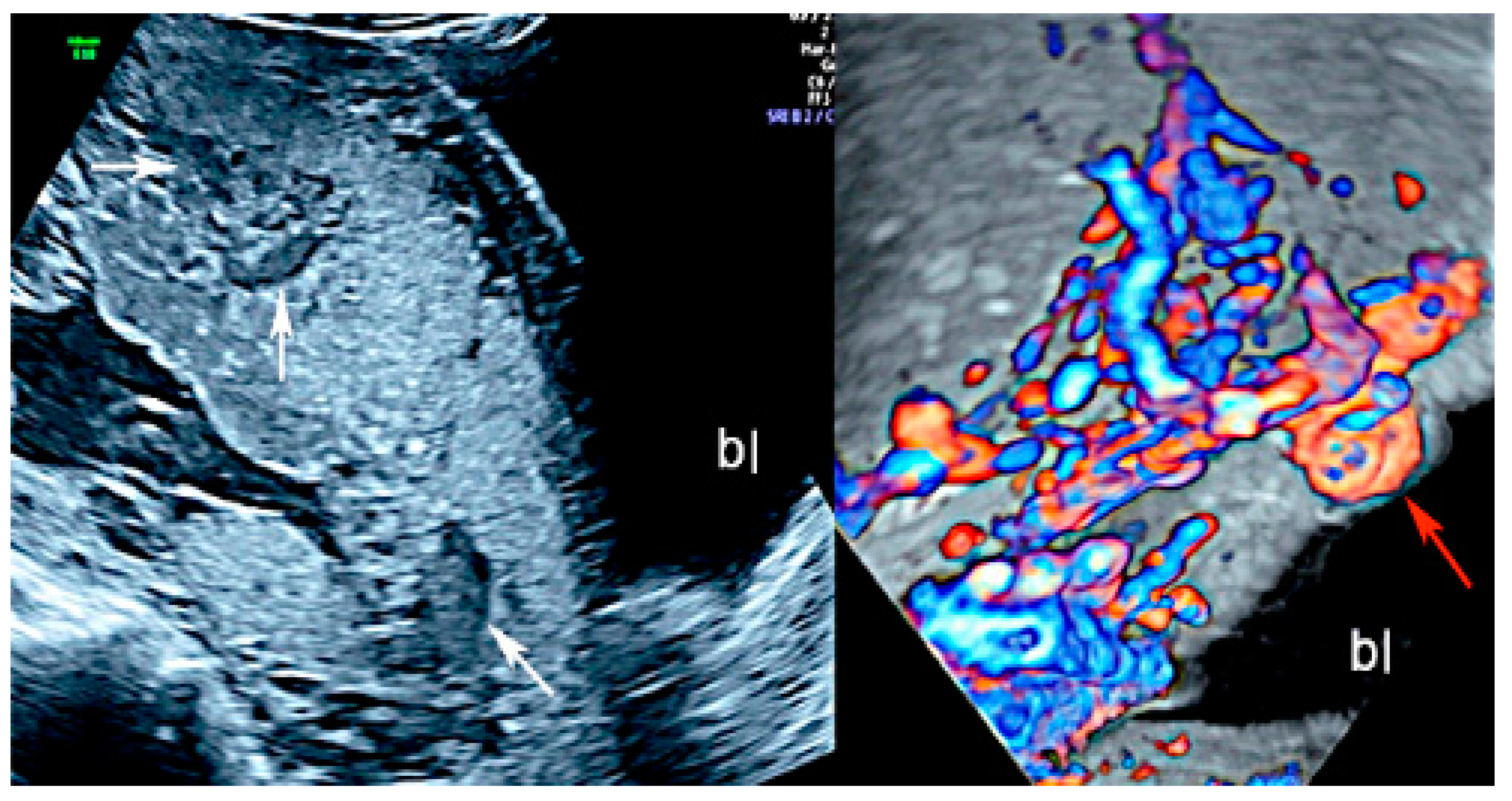
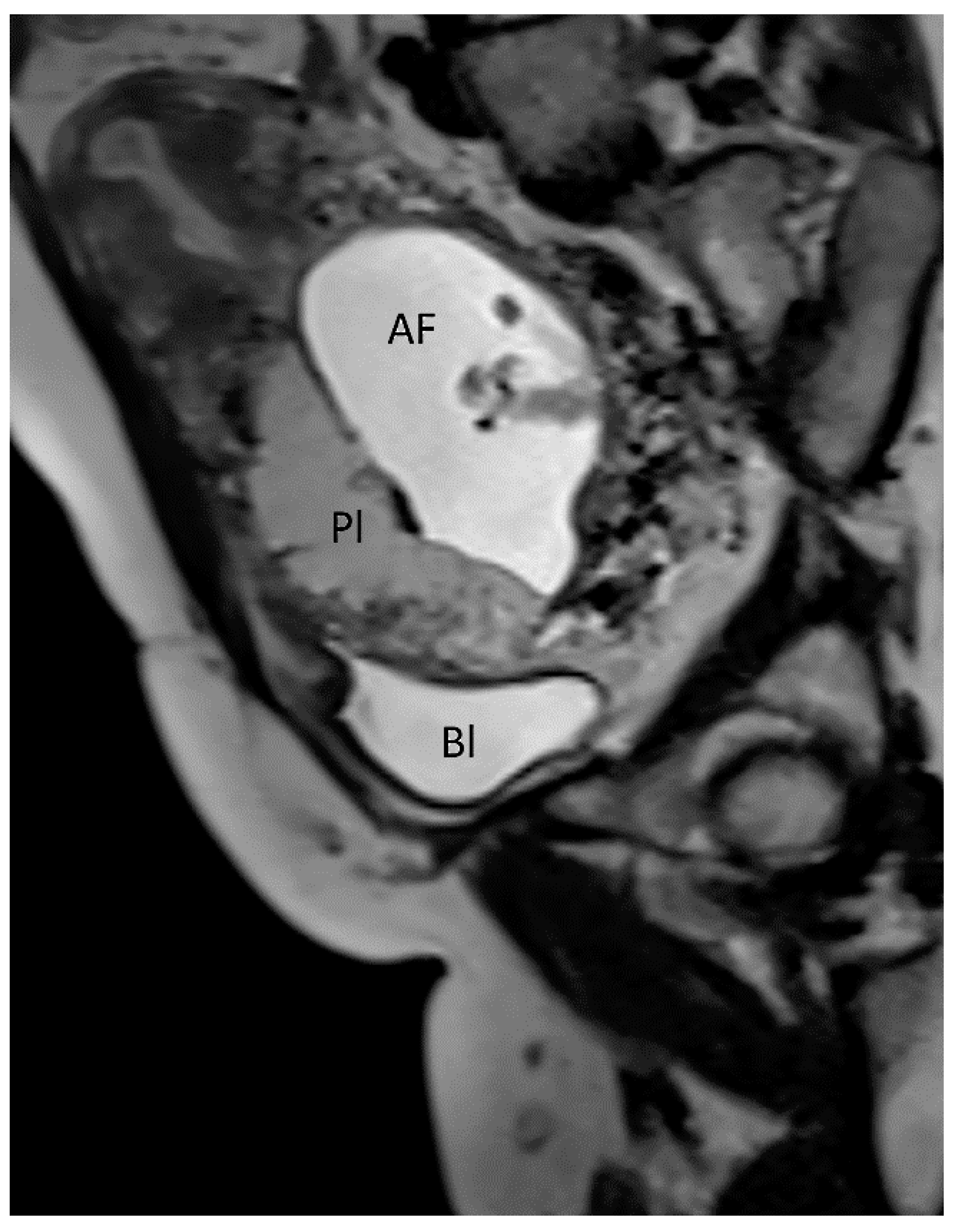
3.1.1. Placenta Accreta Spectrum
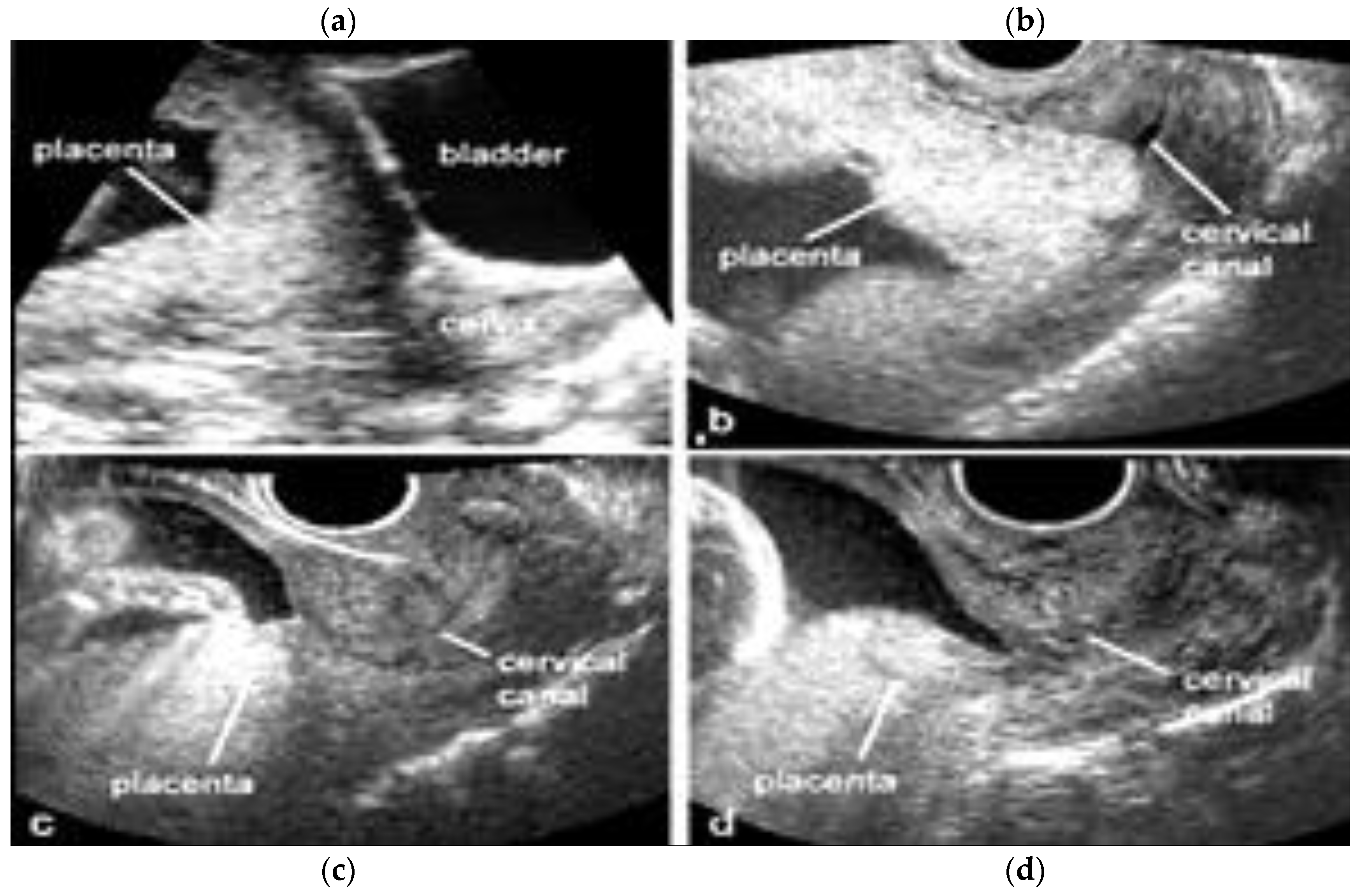
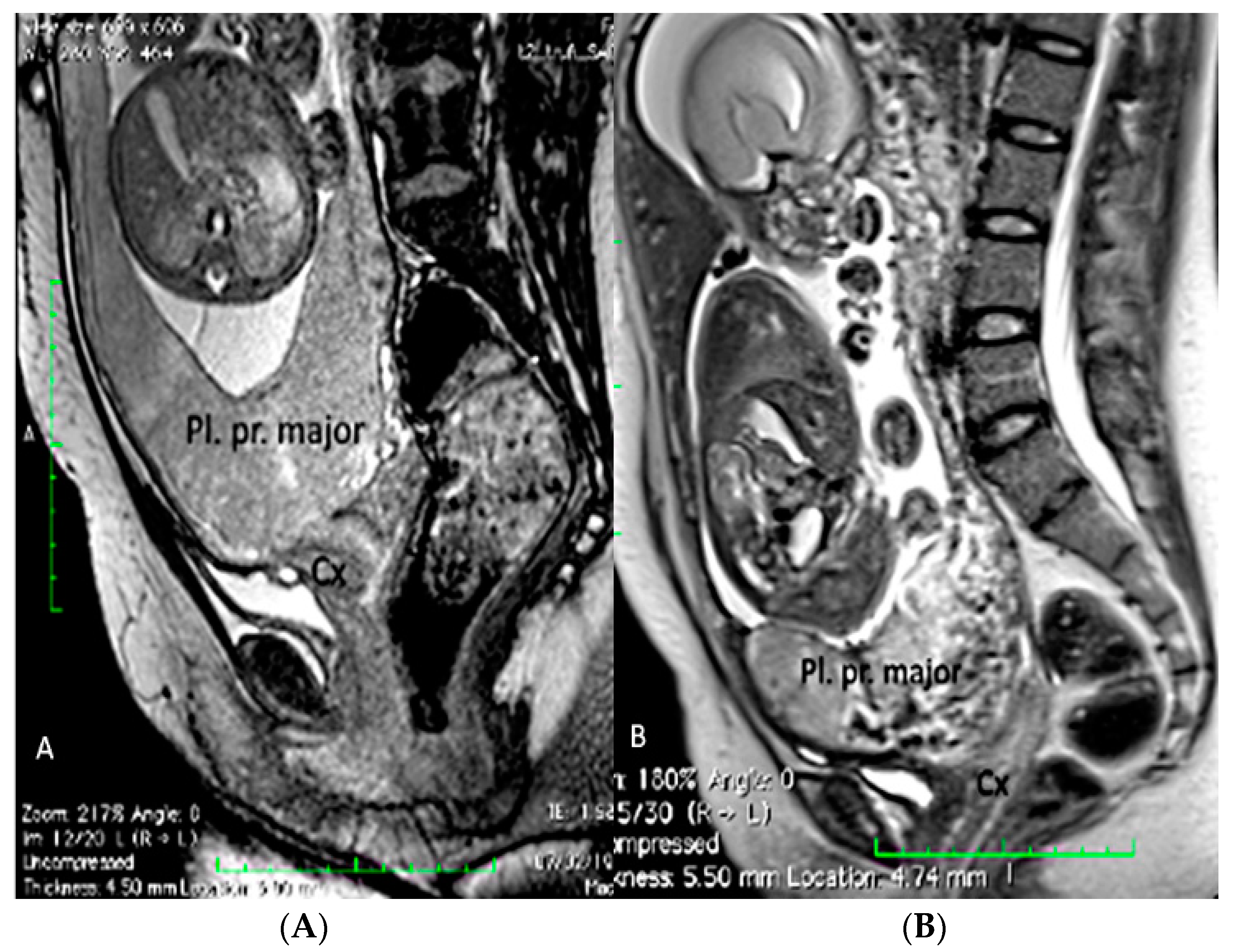
3.1.2. Placenta Praevia
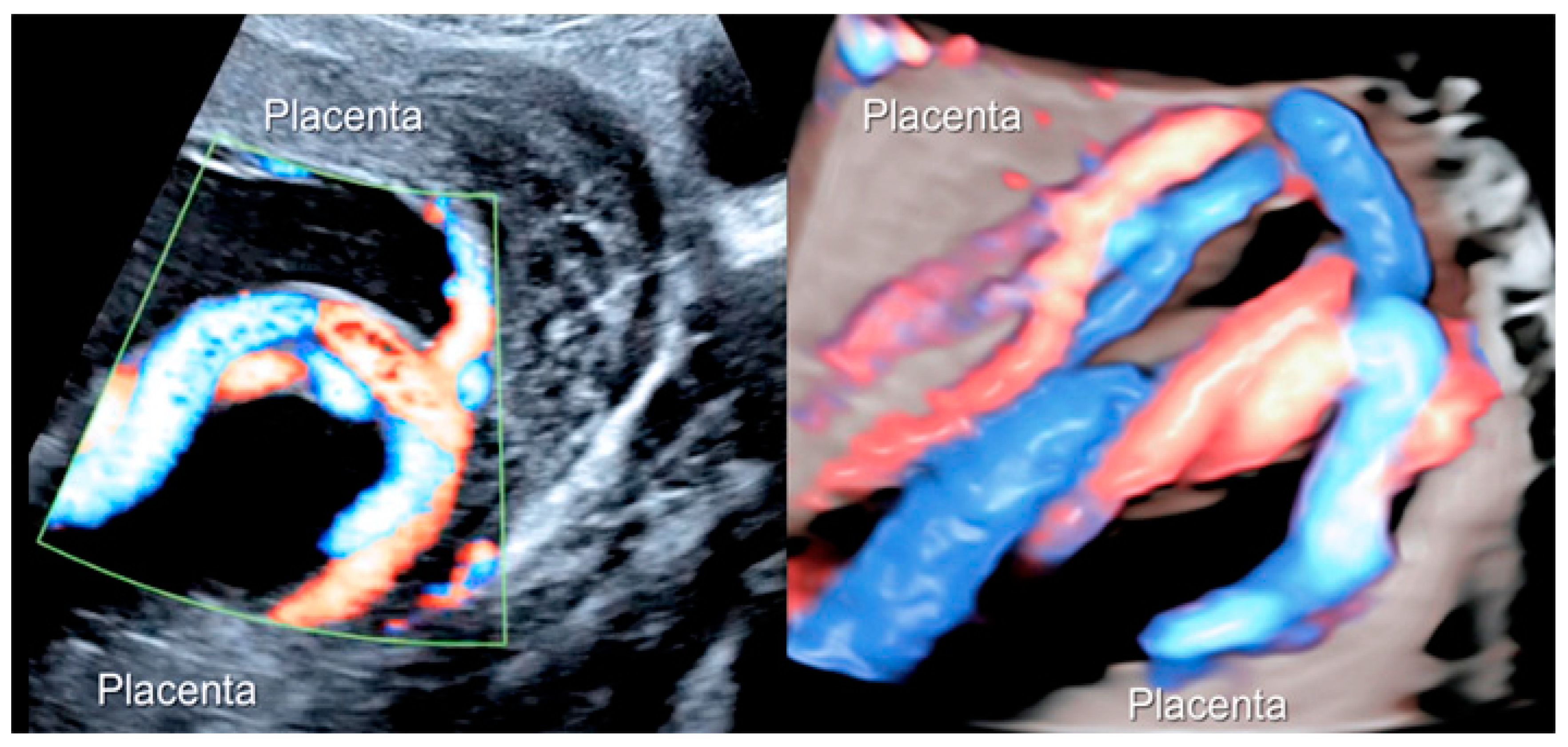
3.1.3. Bilobed Placenta and Succenturiate Lobe
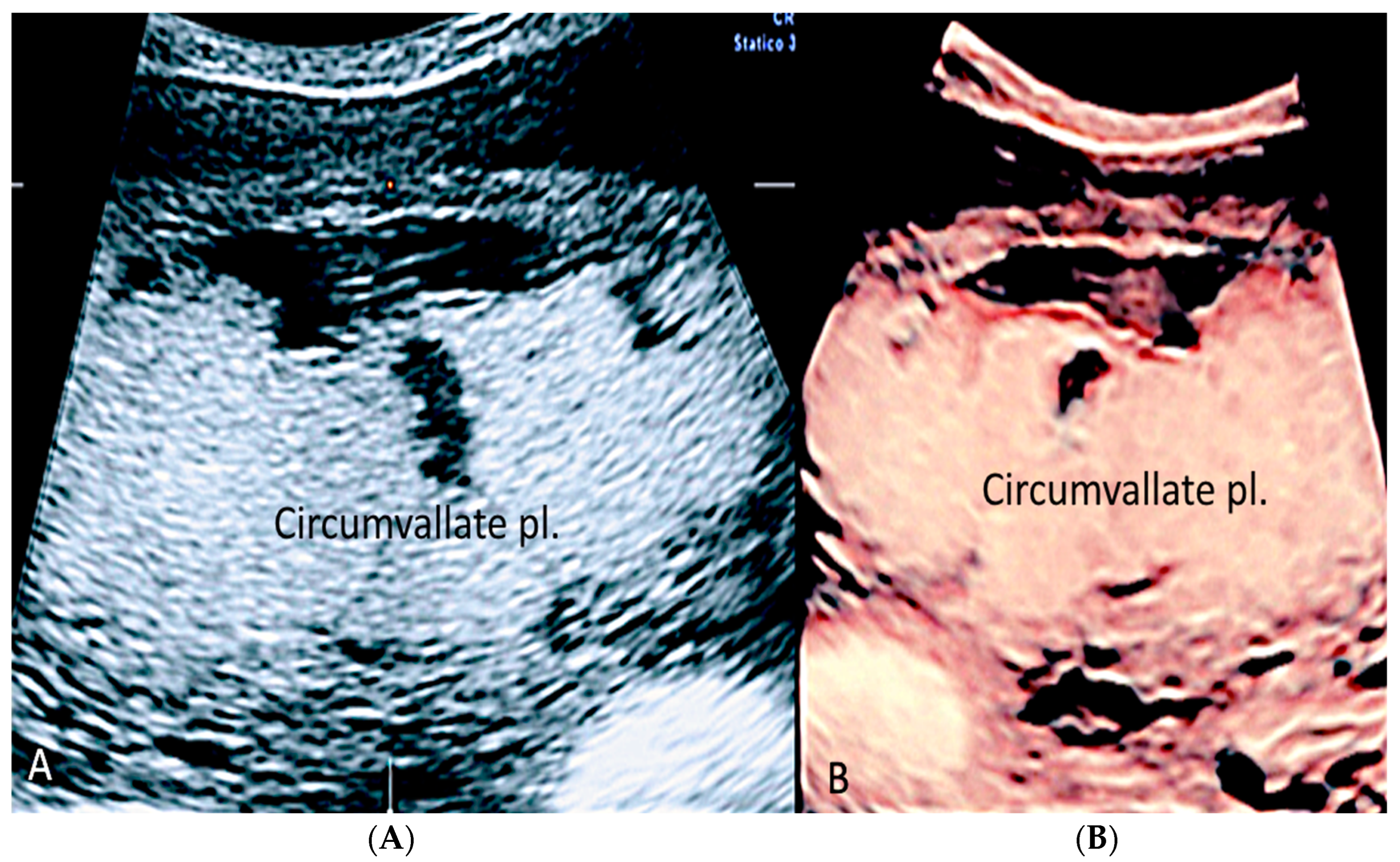
3.1.4. Circumvallate Placenta
3.1.5. Placental Mesenchymal Dysplasia
3.1.6. Chorioangioma
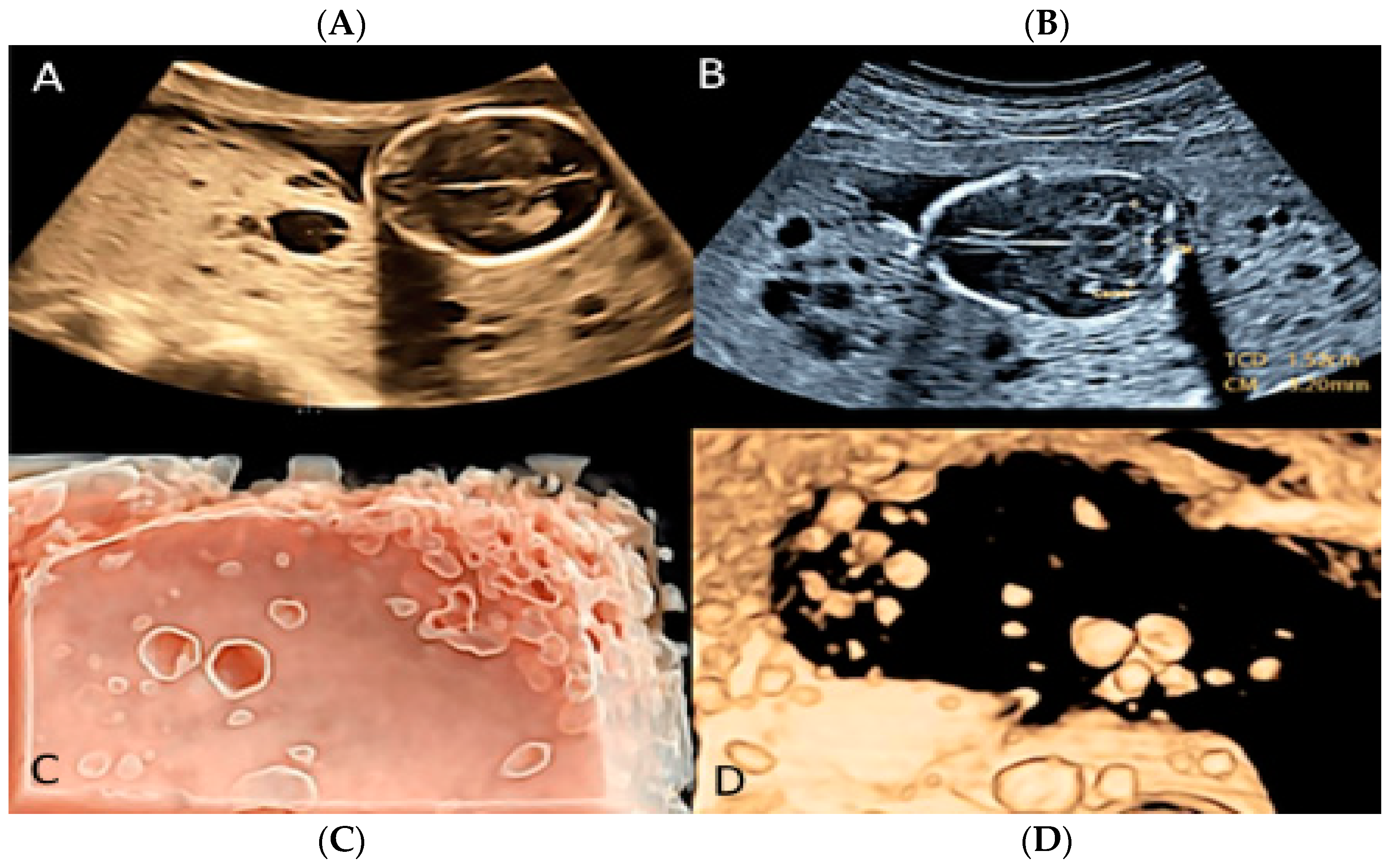
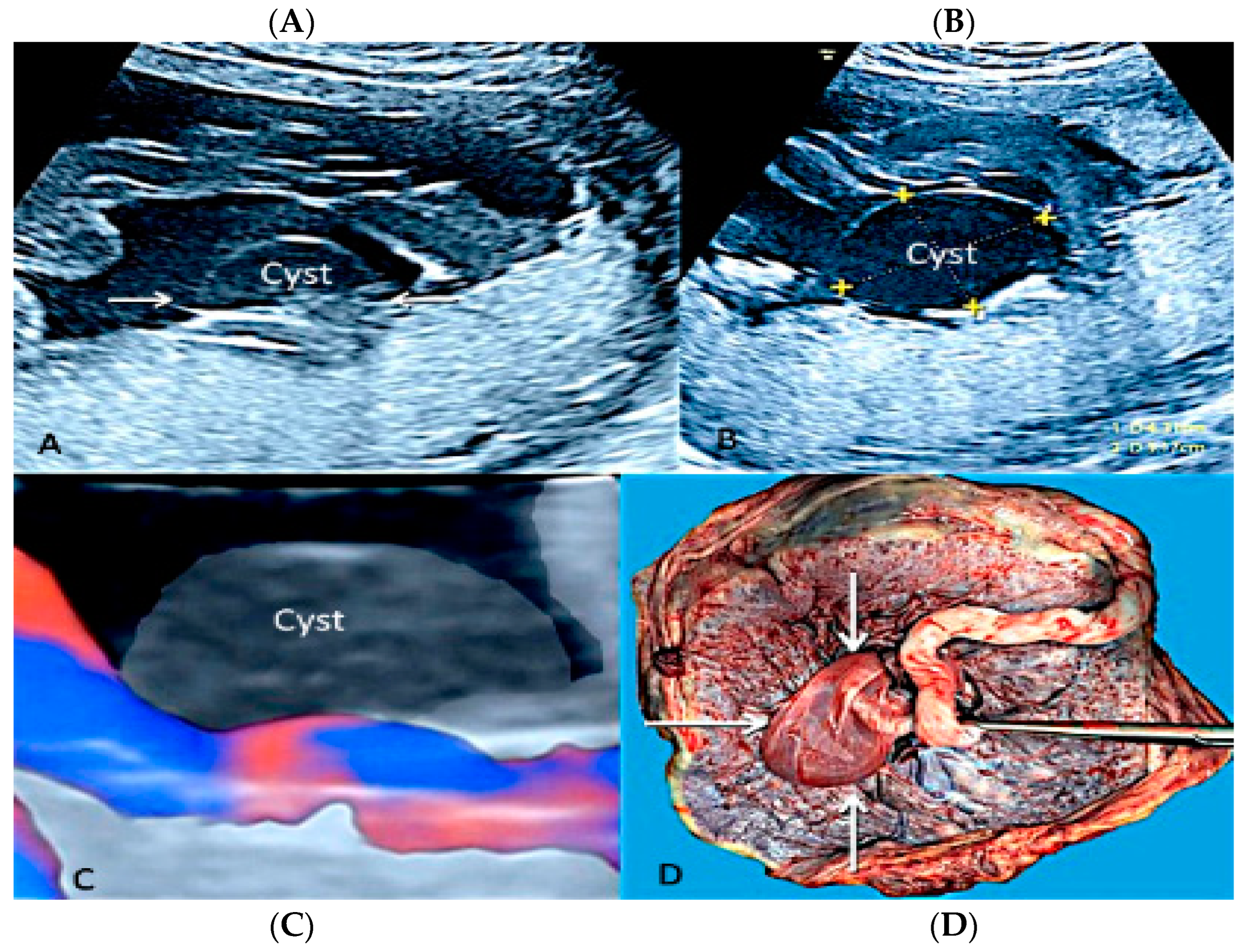
3.1.7. Membranous Placental Cysts
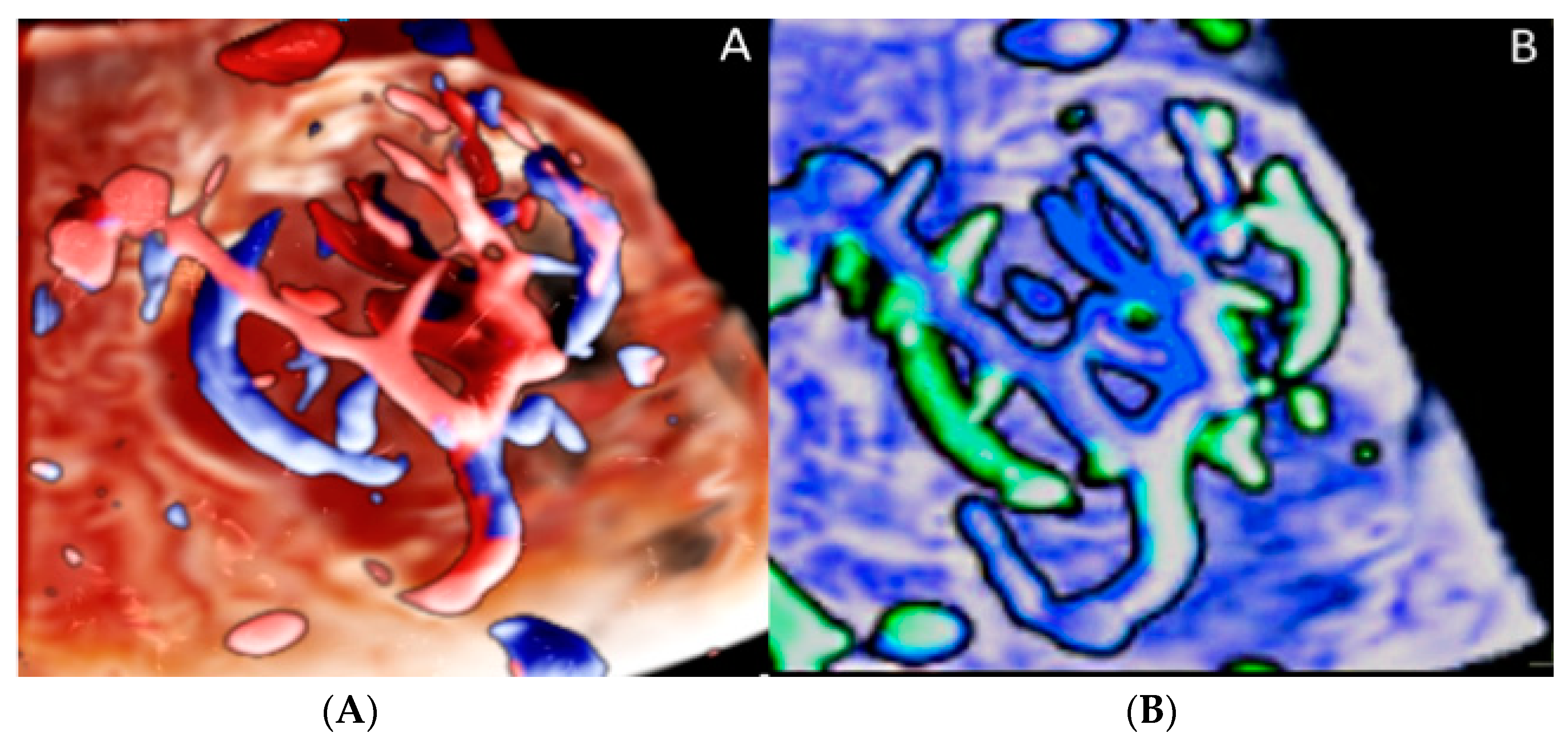
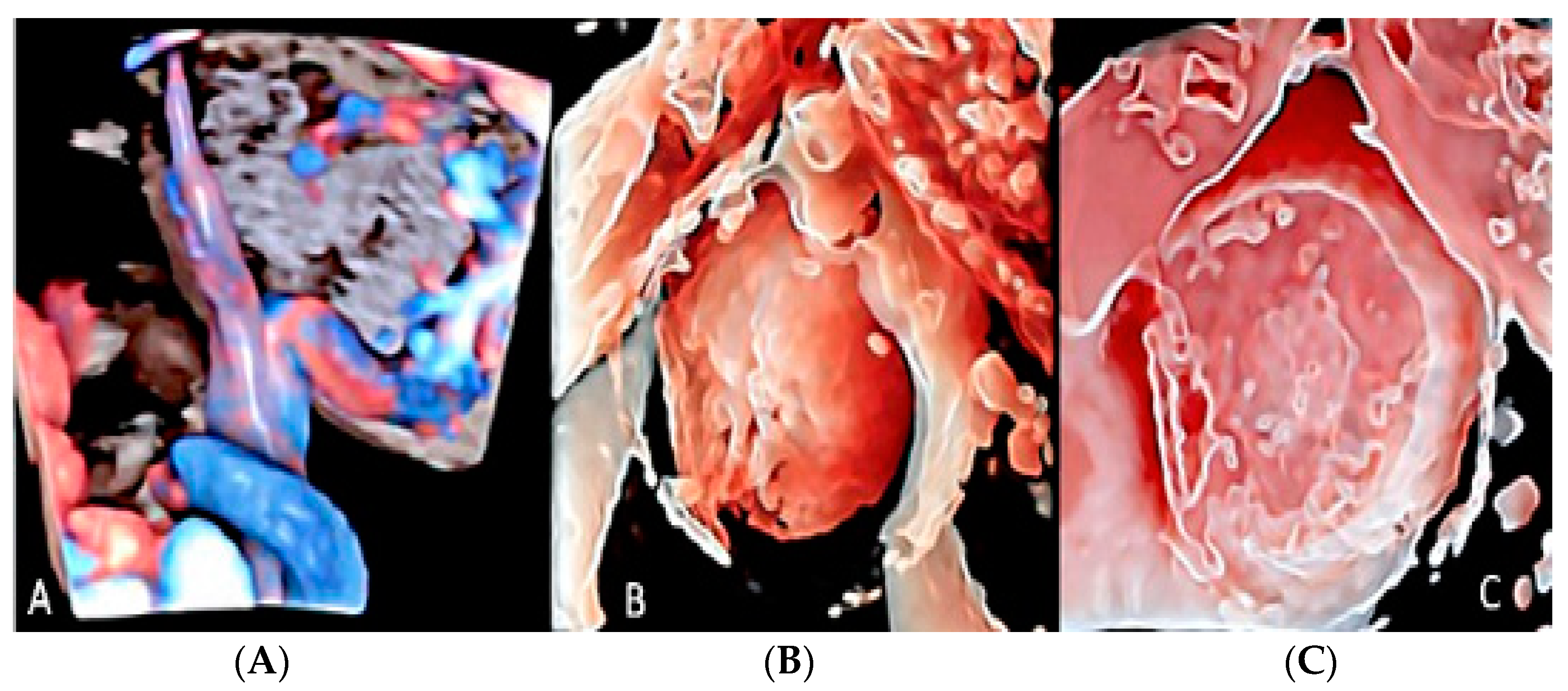
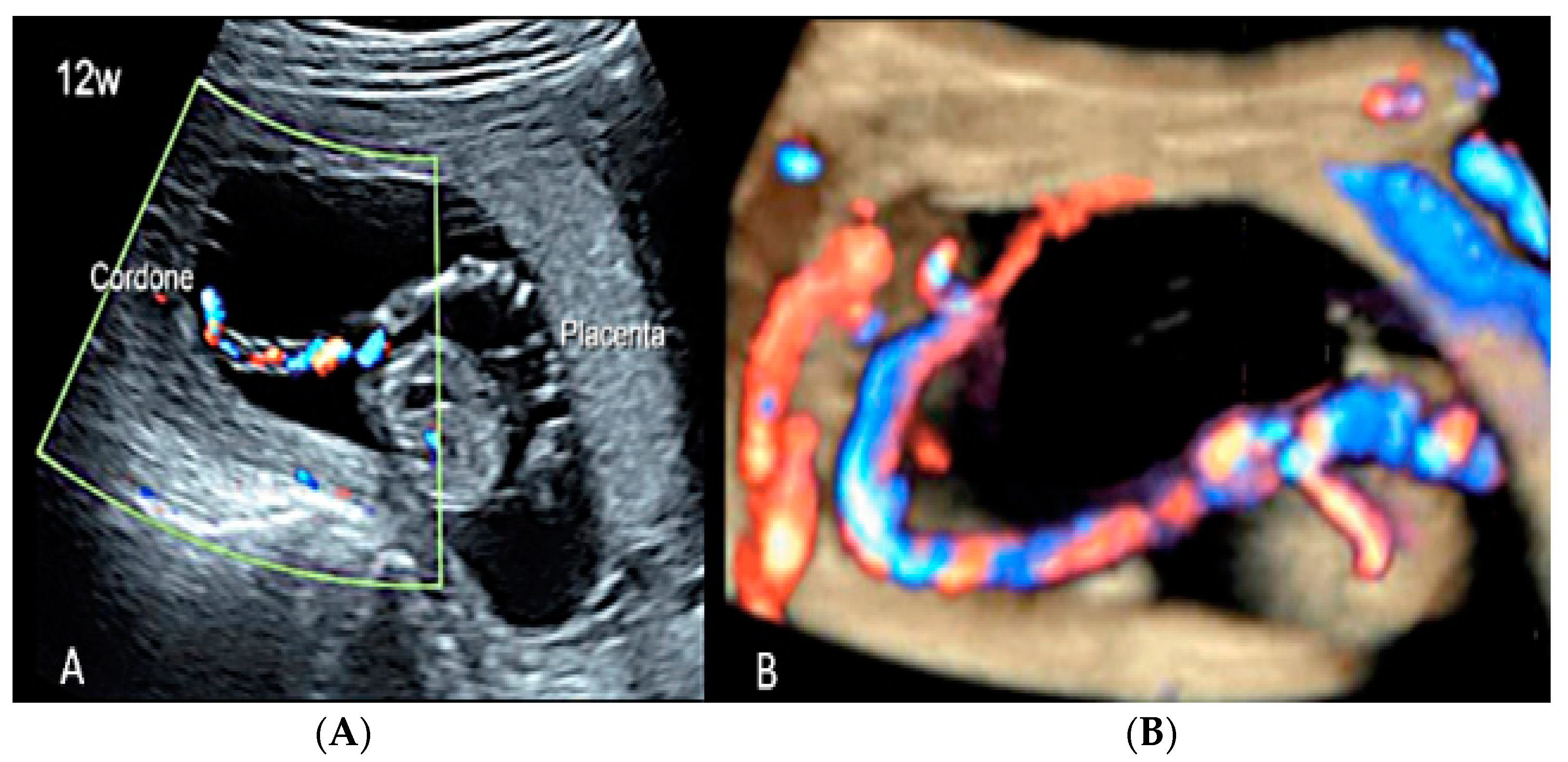
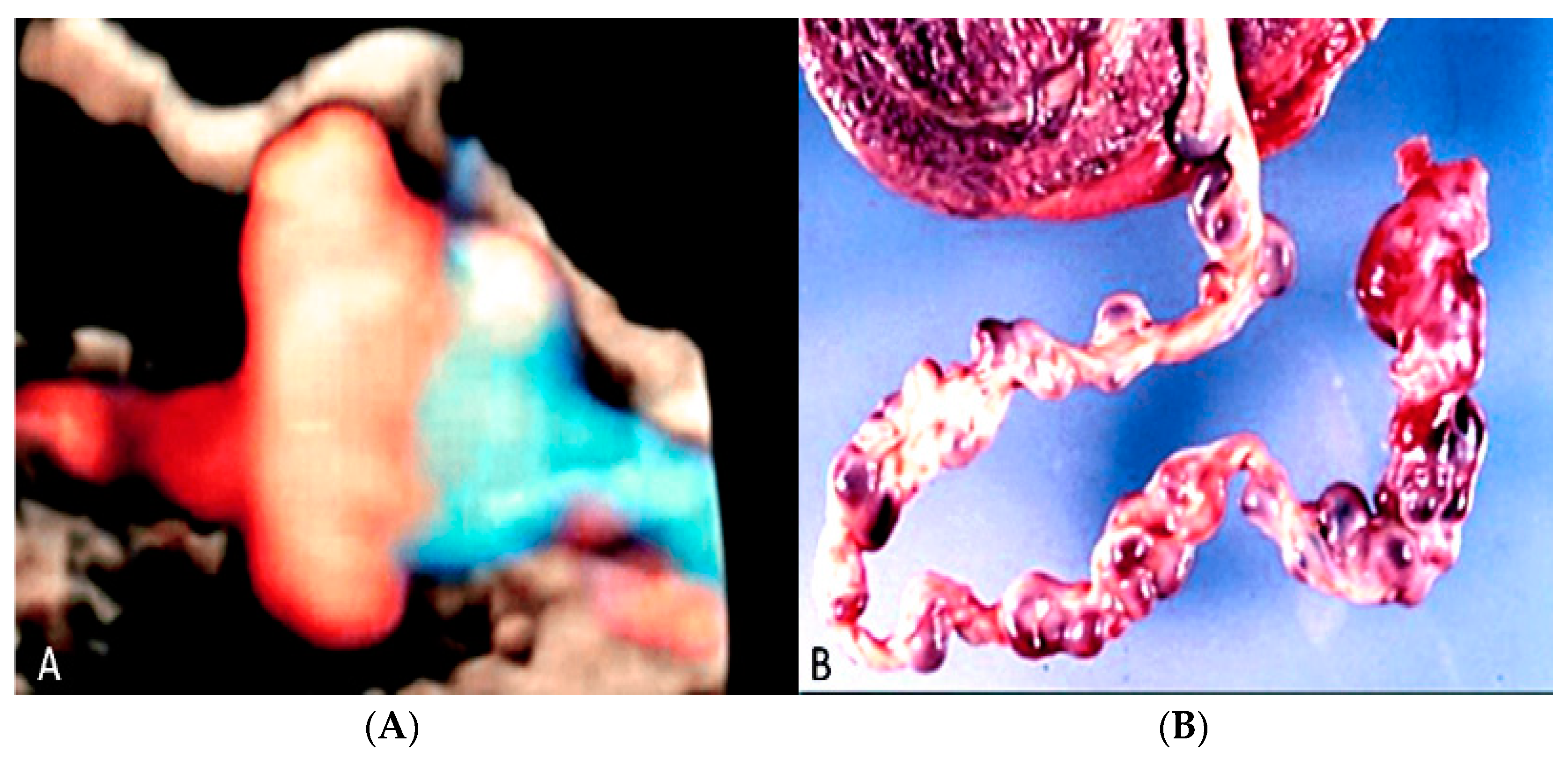
3.2. Umbilical Cord Anomalies
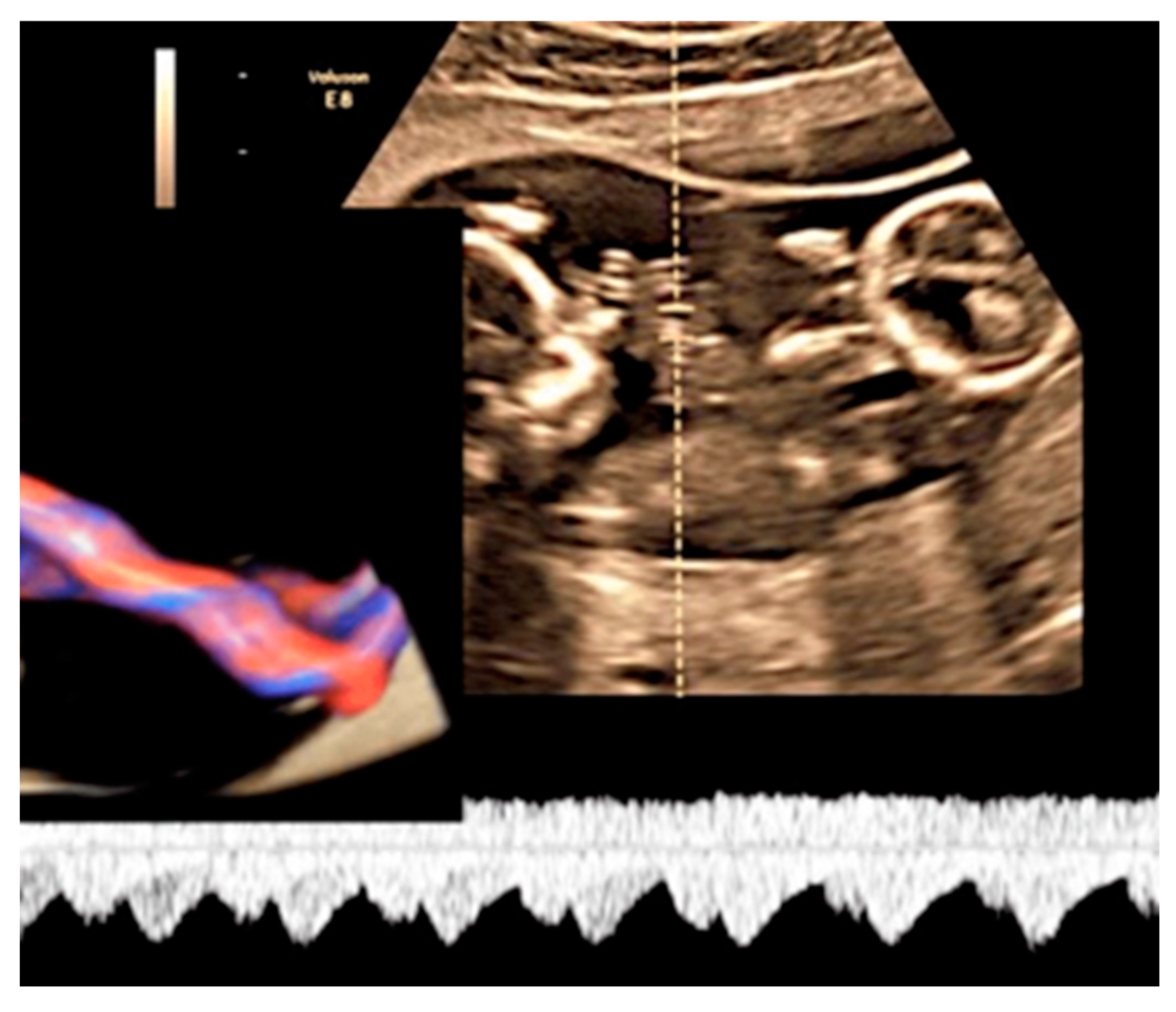
3.2.1. Nuchal Cords
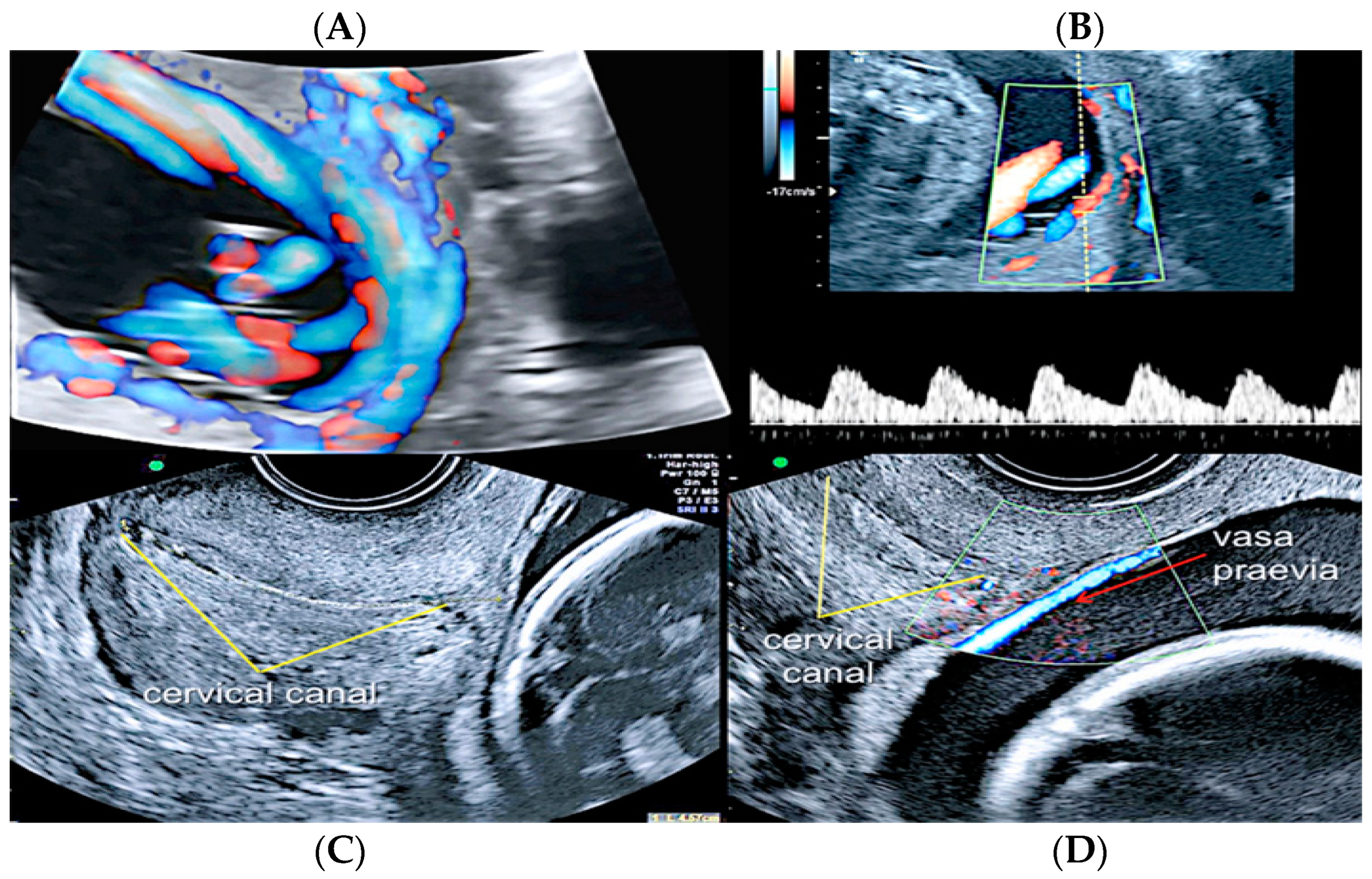
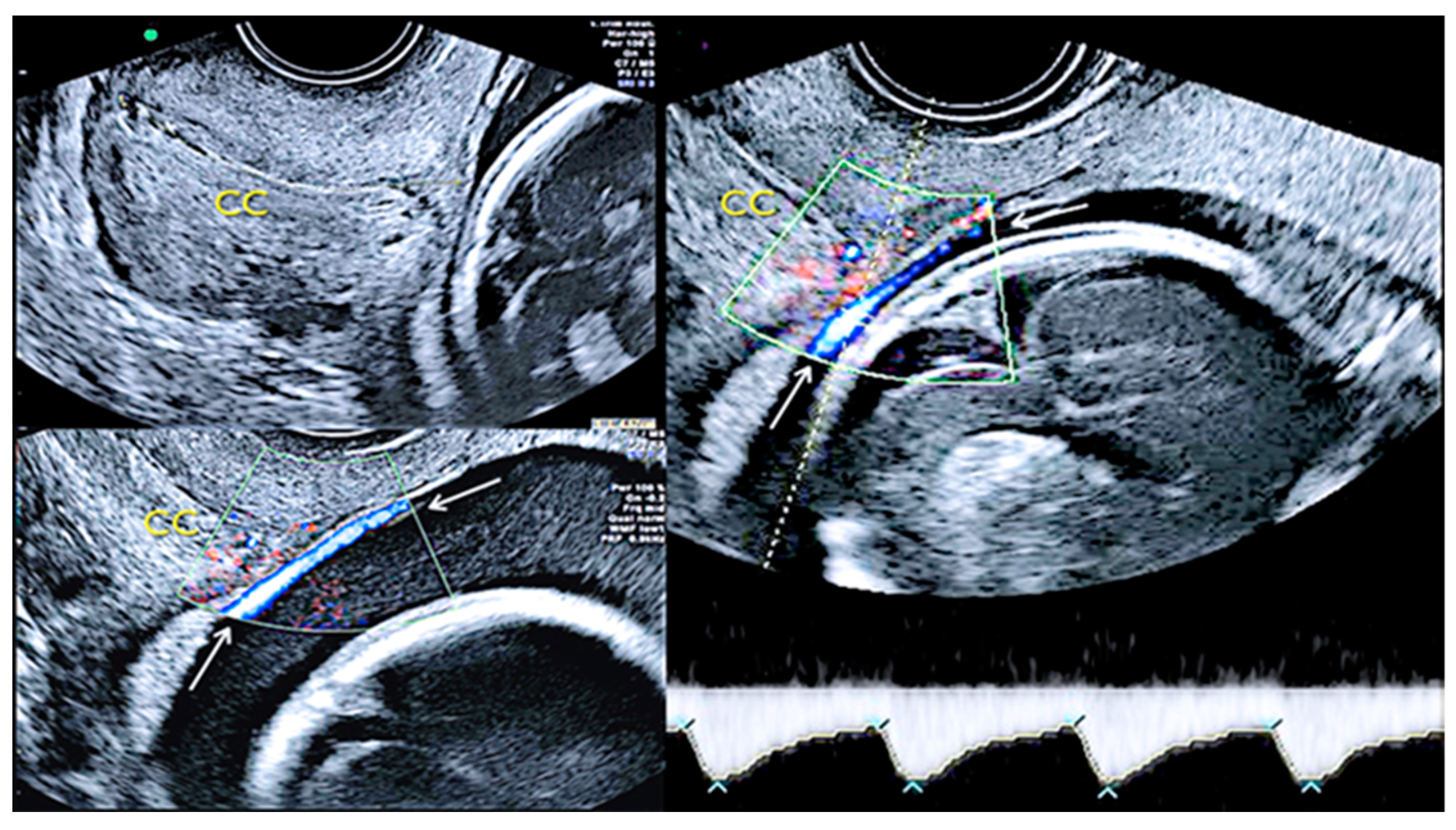
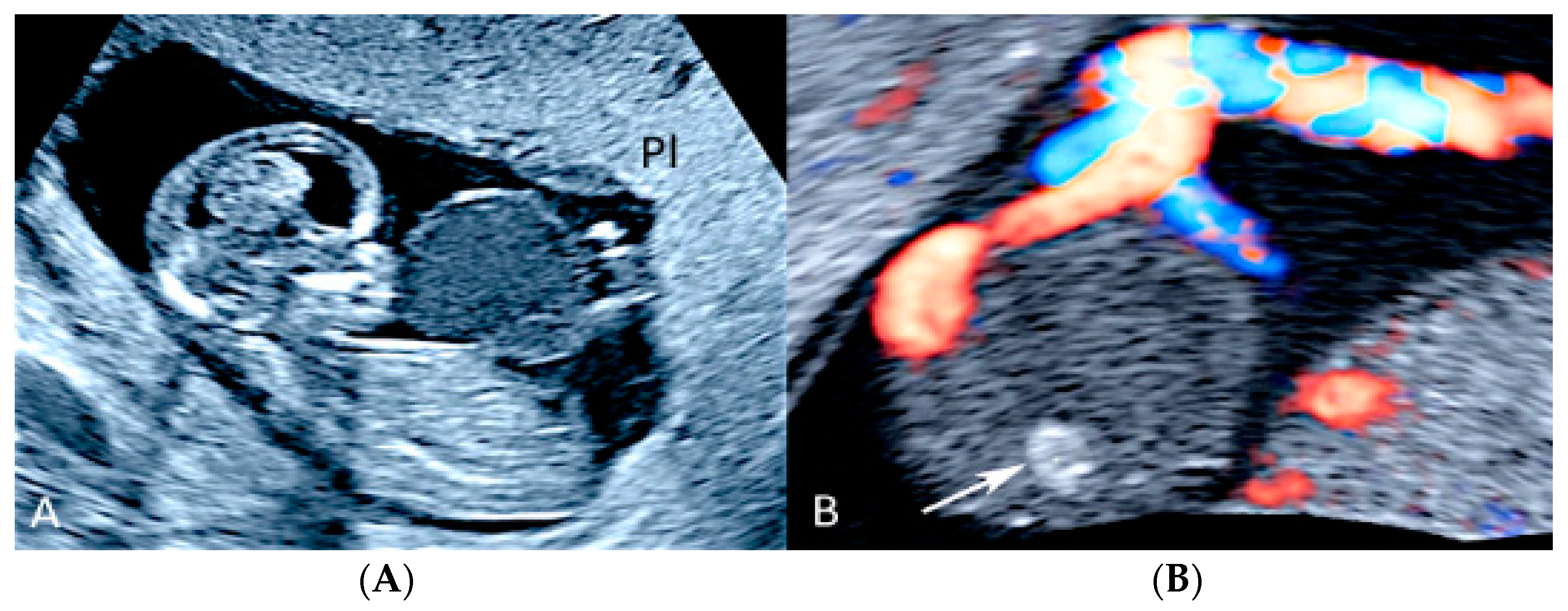
3.2.2. Vasa Praevia
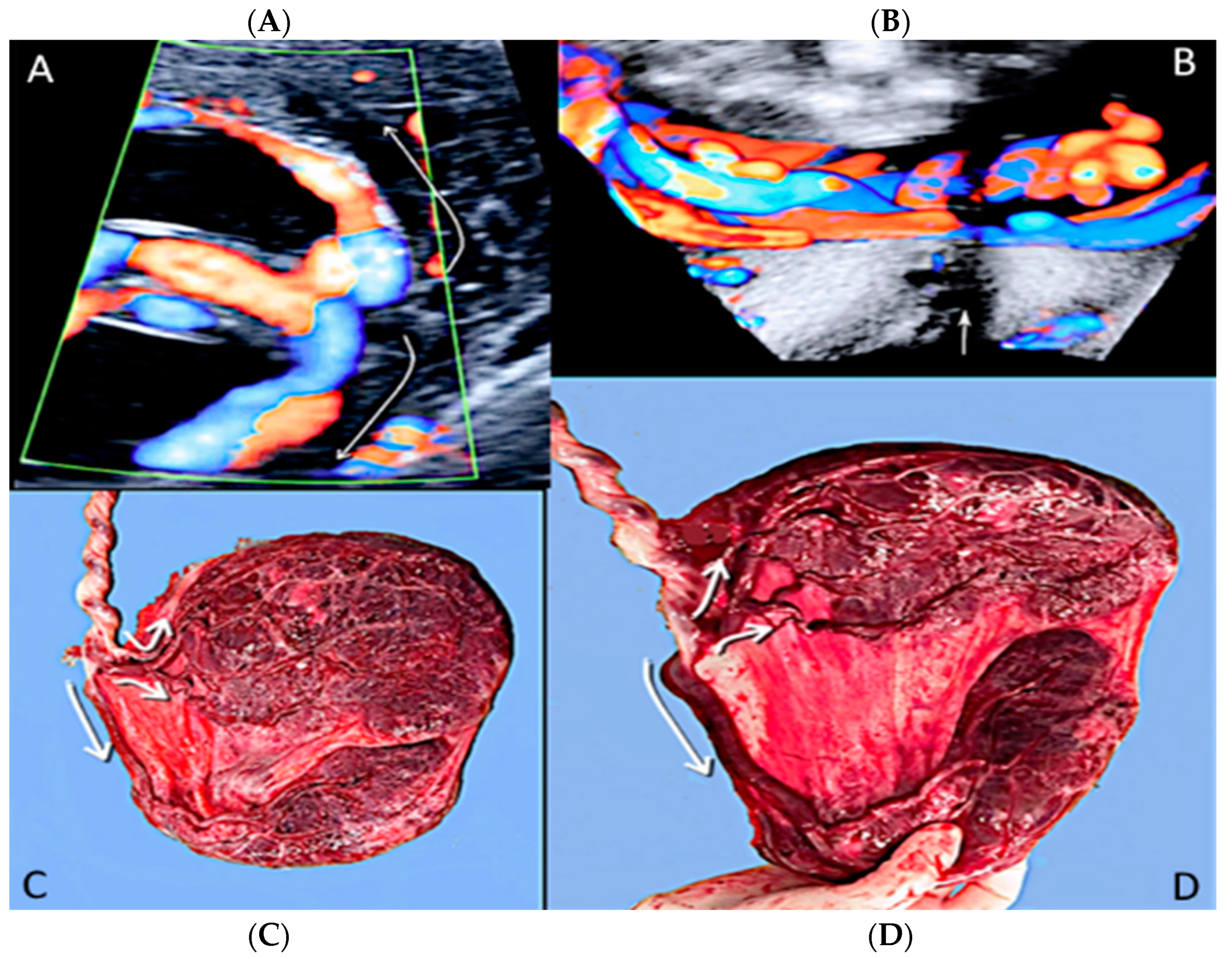
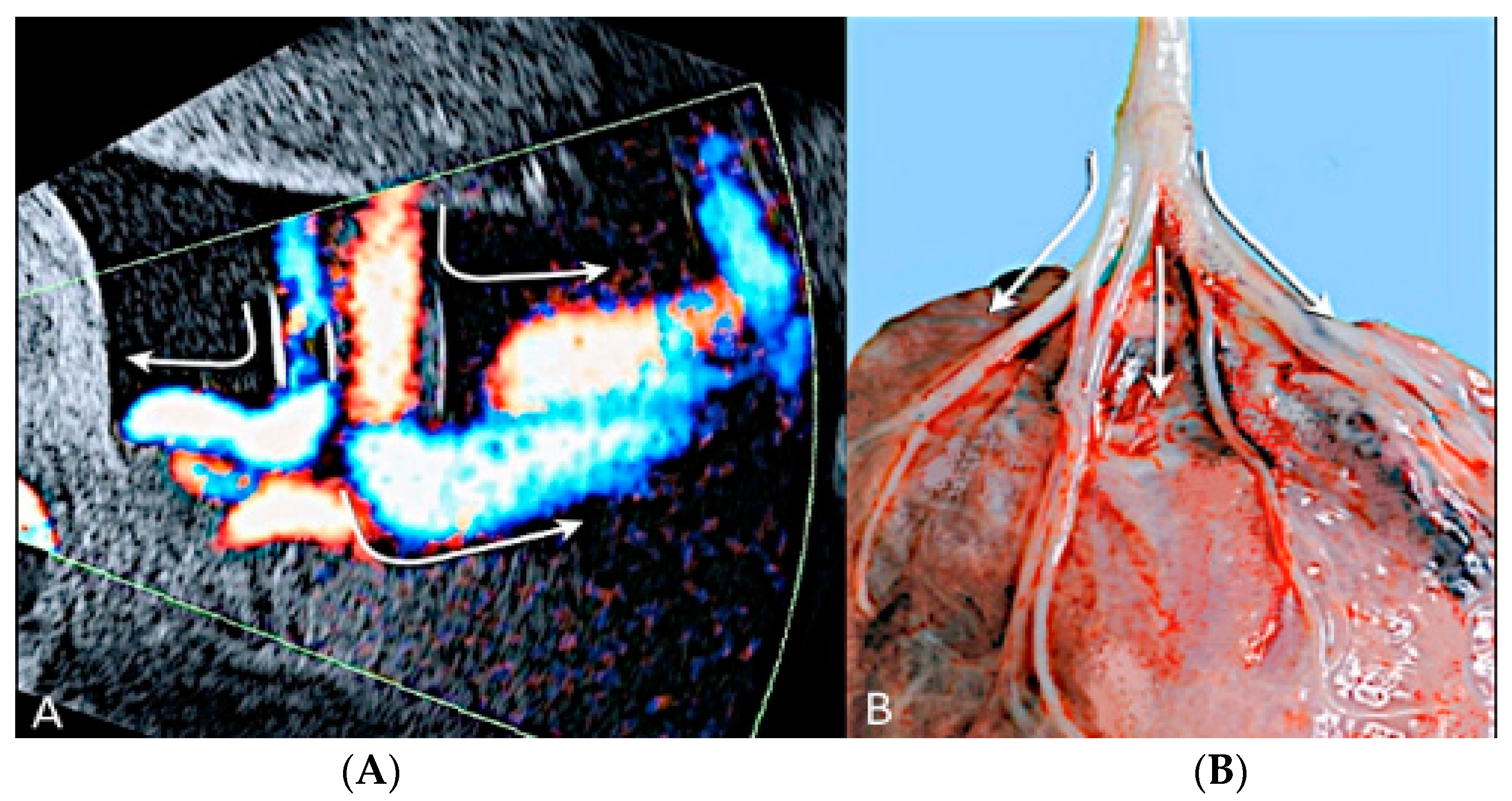
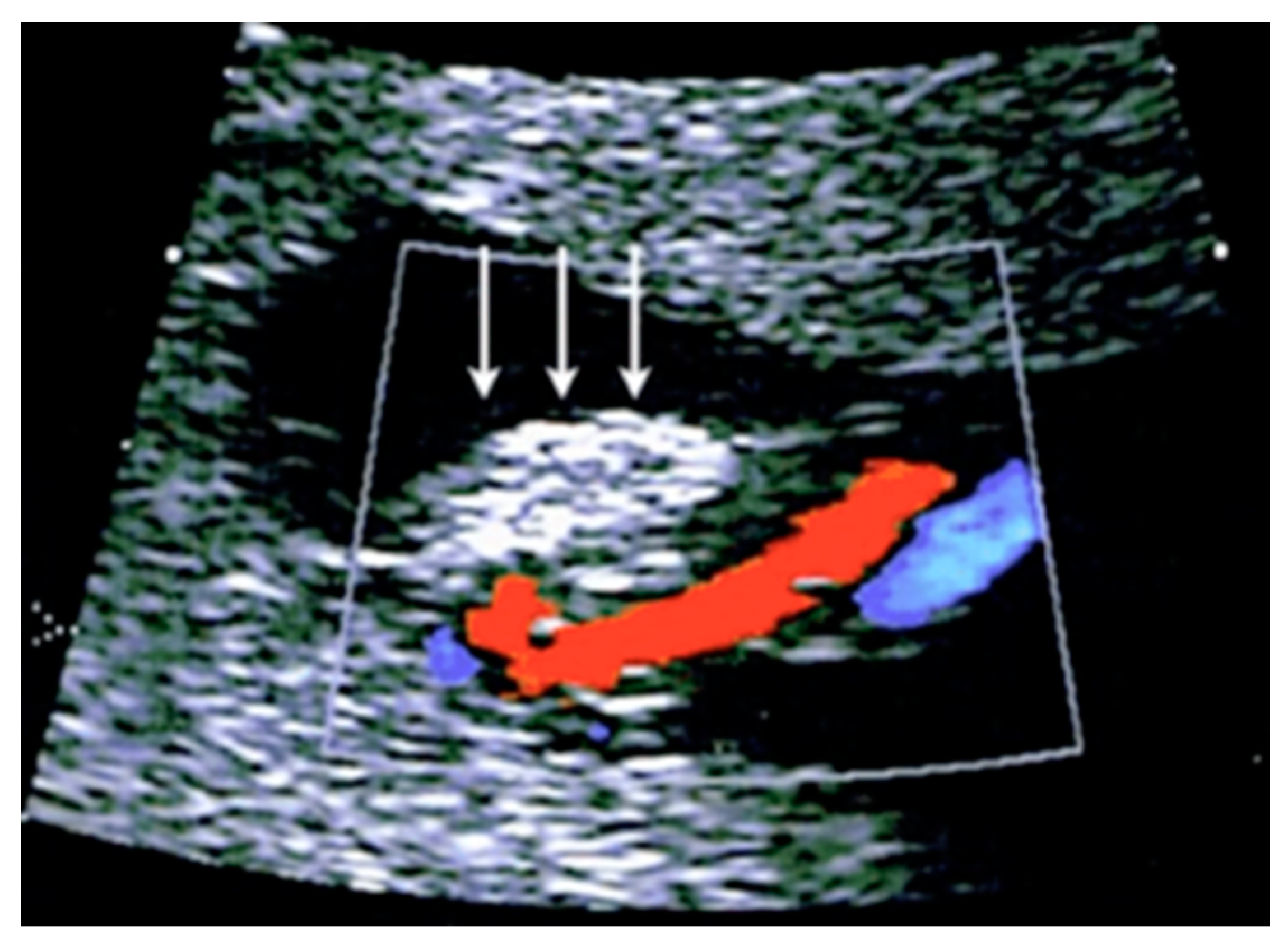
3.2.3. Velamentous Umbilical Cord Insertion
3.2.4. Marginal Umbilical Cord Insertion
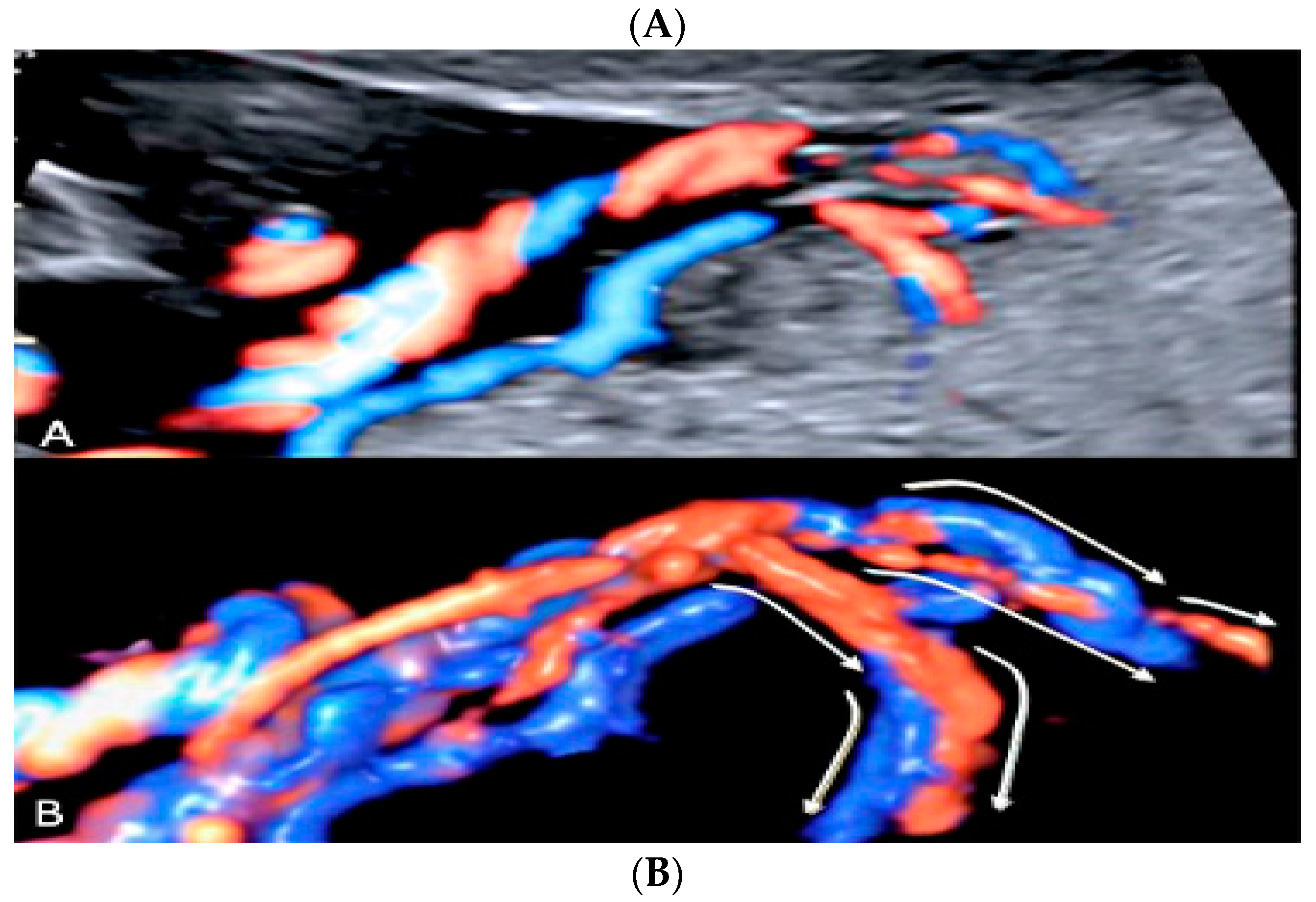
3.2.5. Furcate Umbilical Cord Insertion
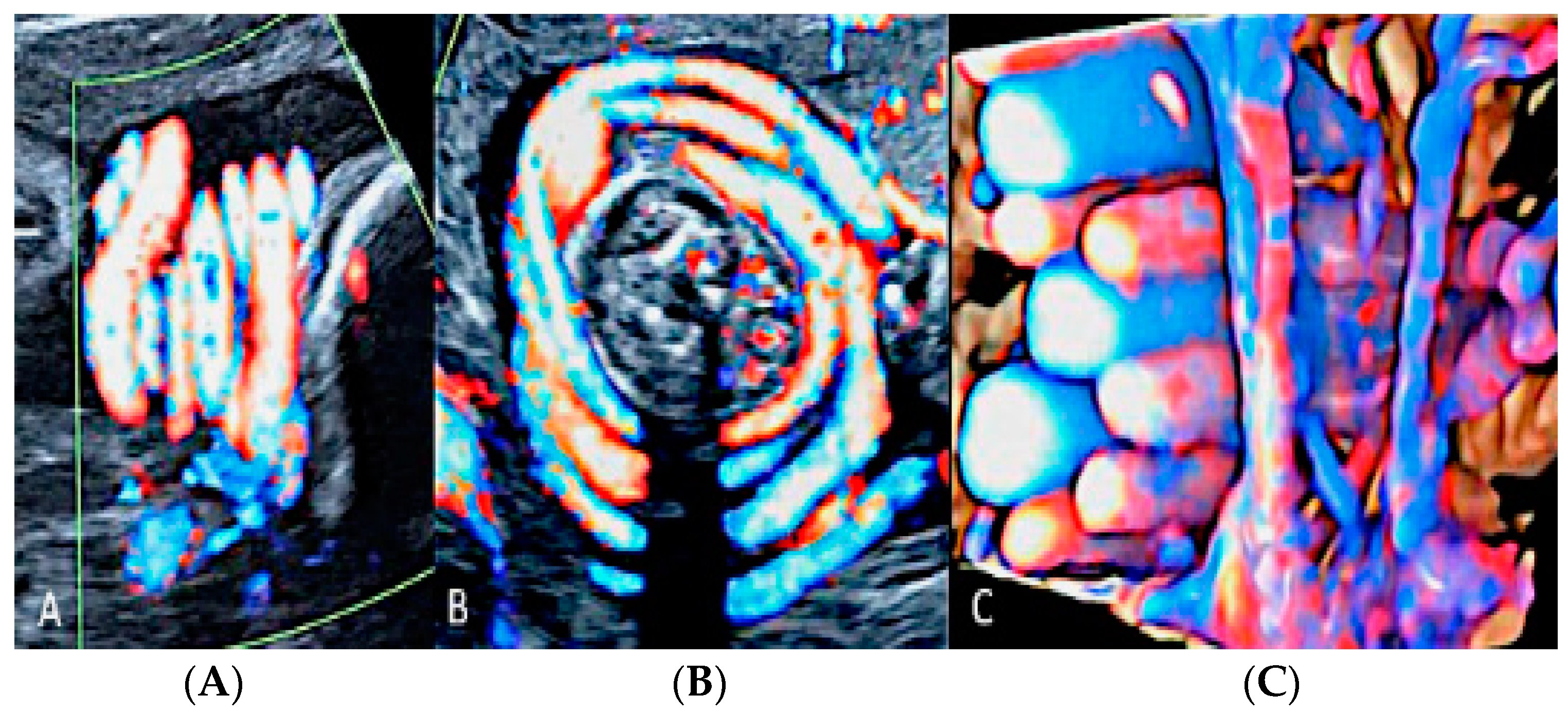
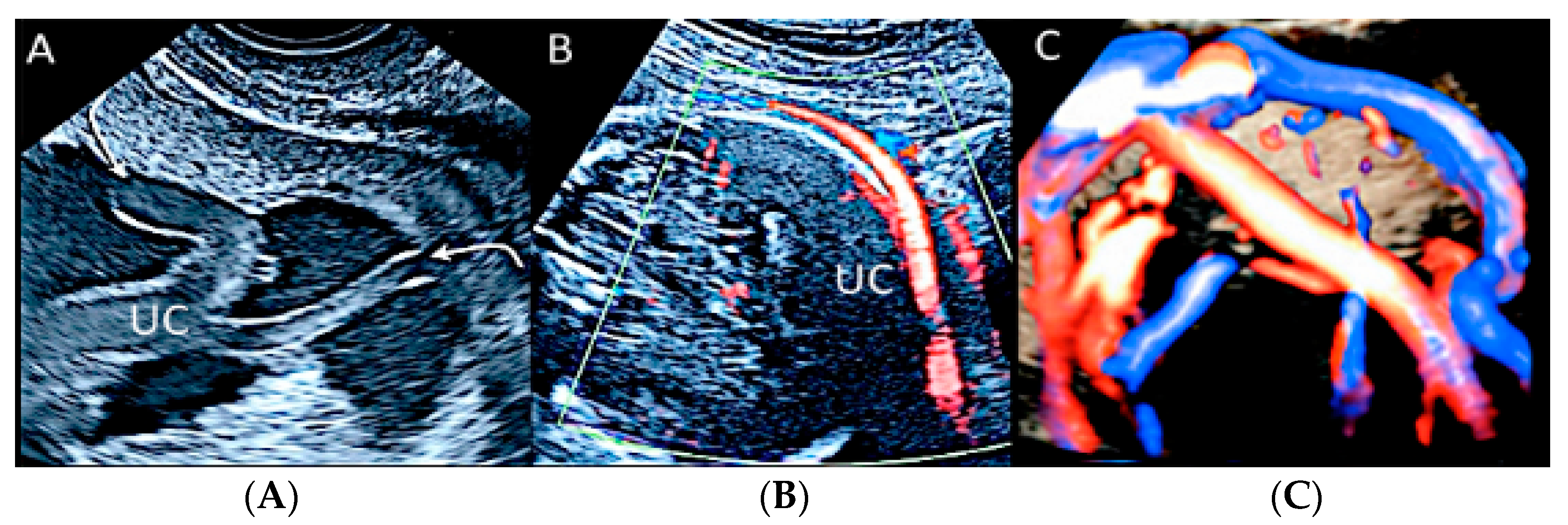
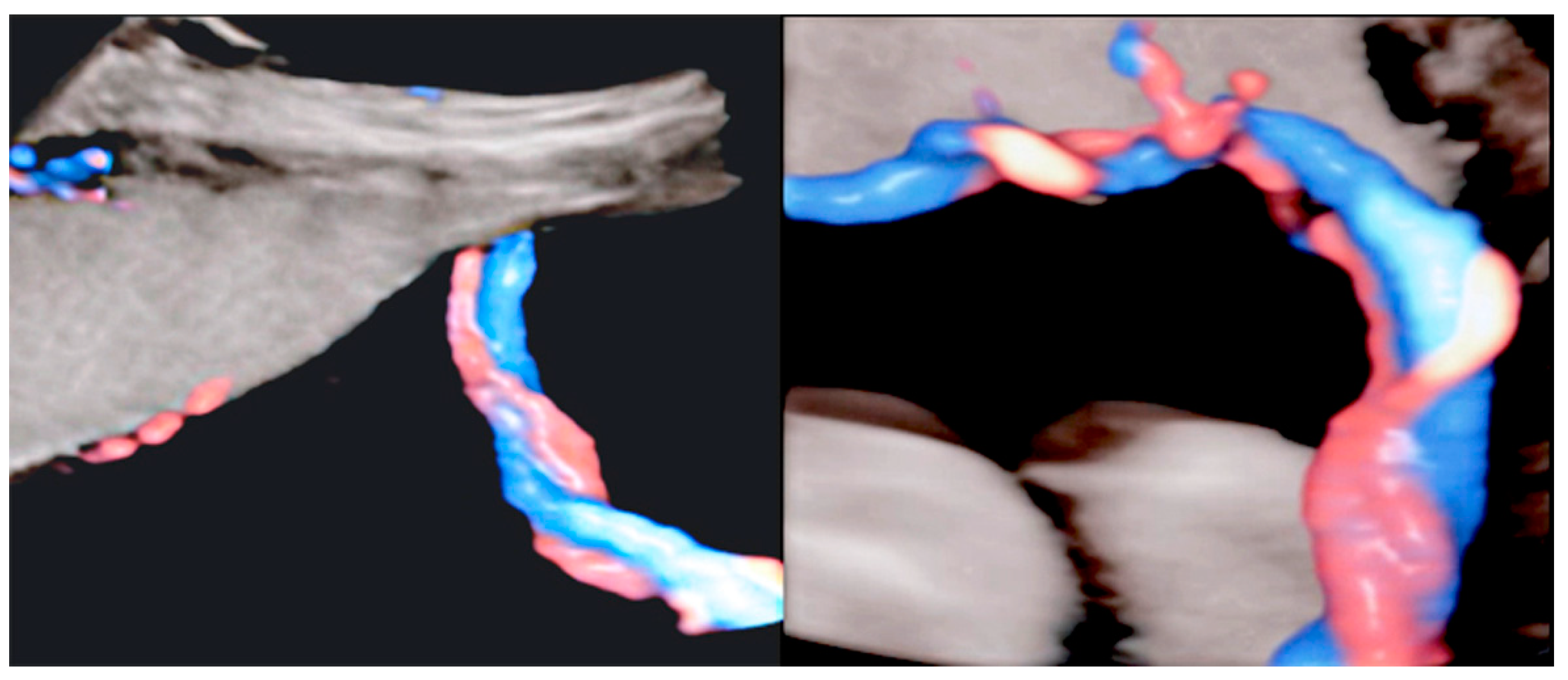
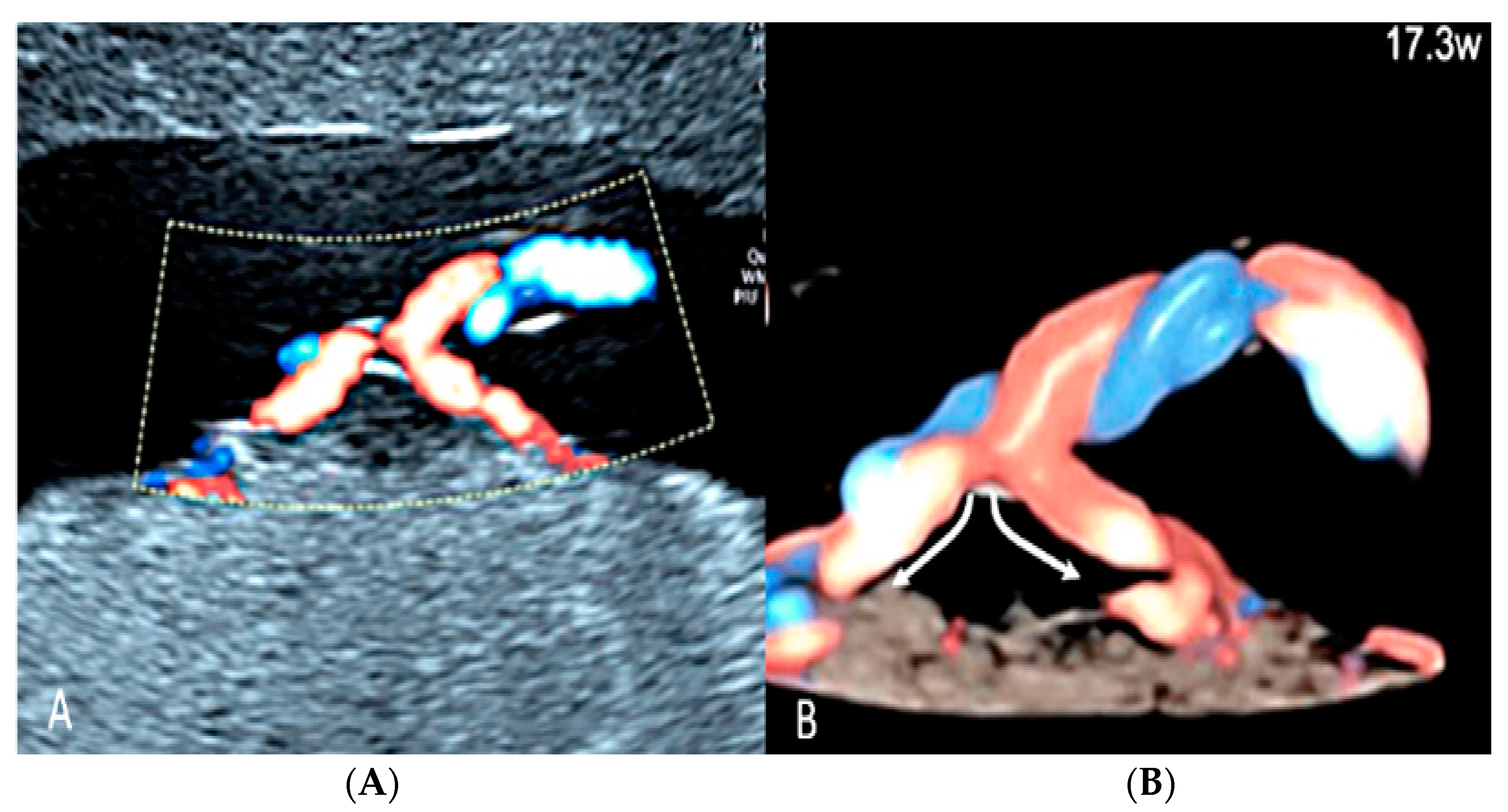
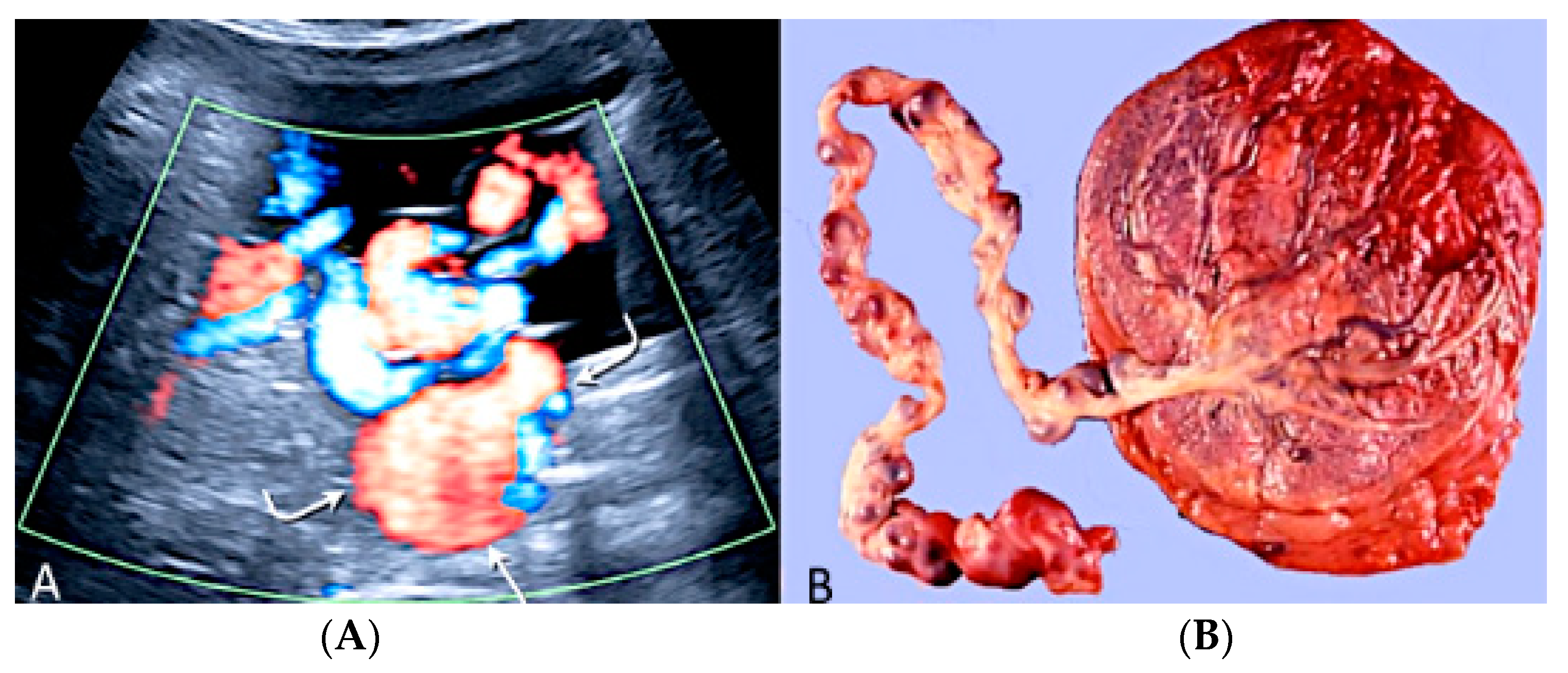
3.2.6. Umbilical Cord Entanglement
3.2.7. Umbilical Cord Hemangioma
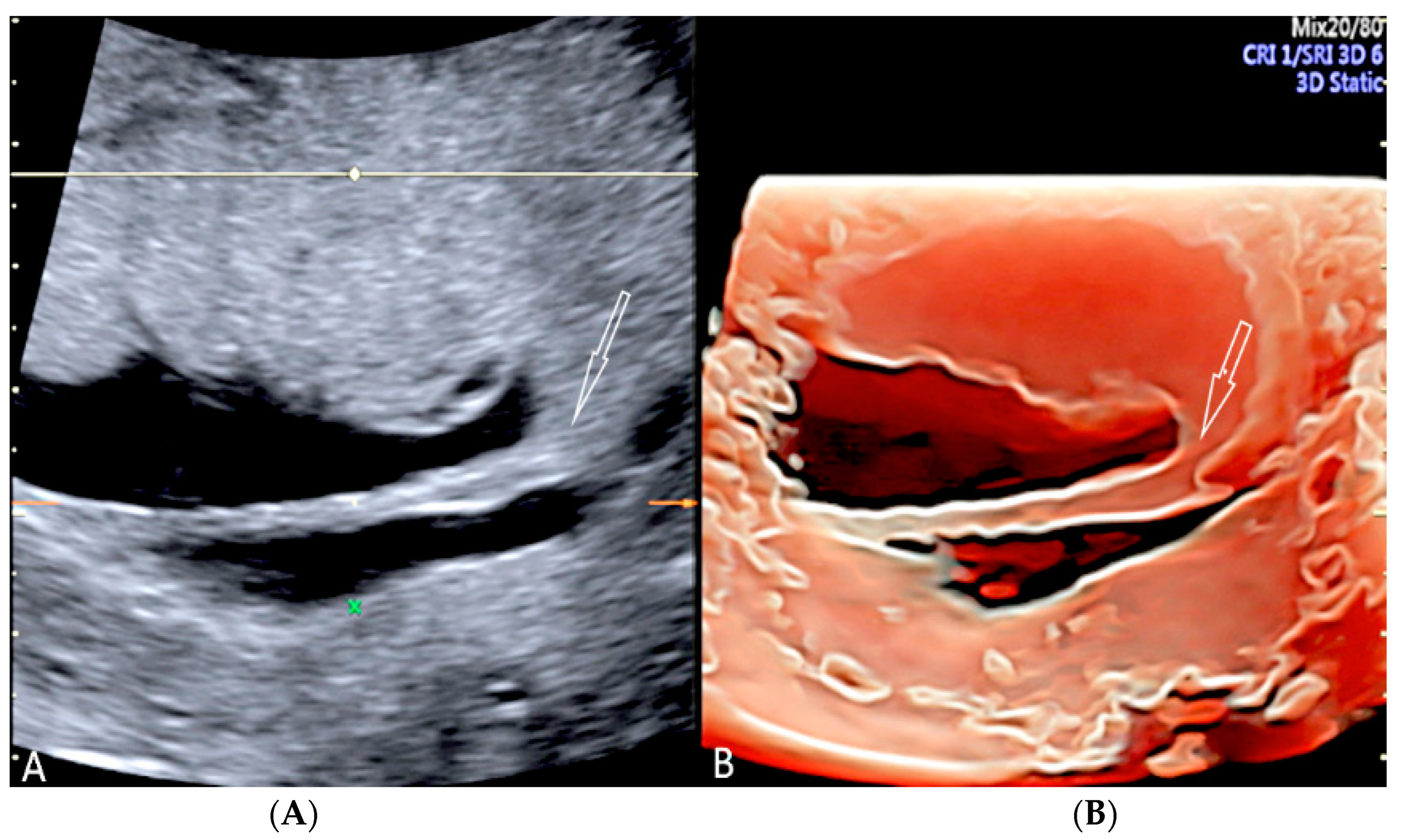
3.2.8. Umbilical Cord Hematoma
3.2.9. Umbilical Cord Cyst
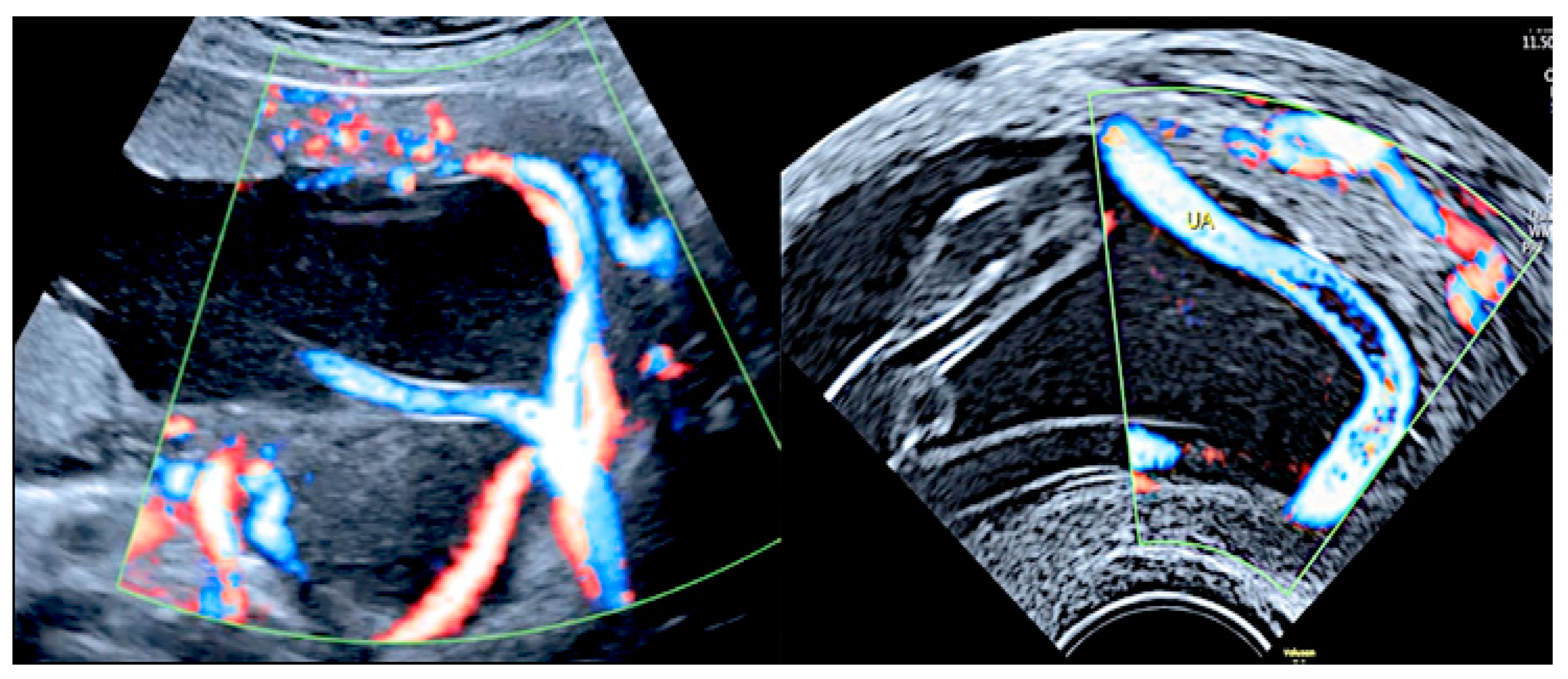
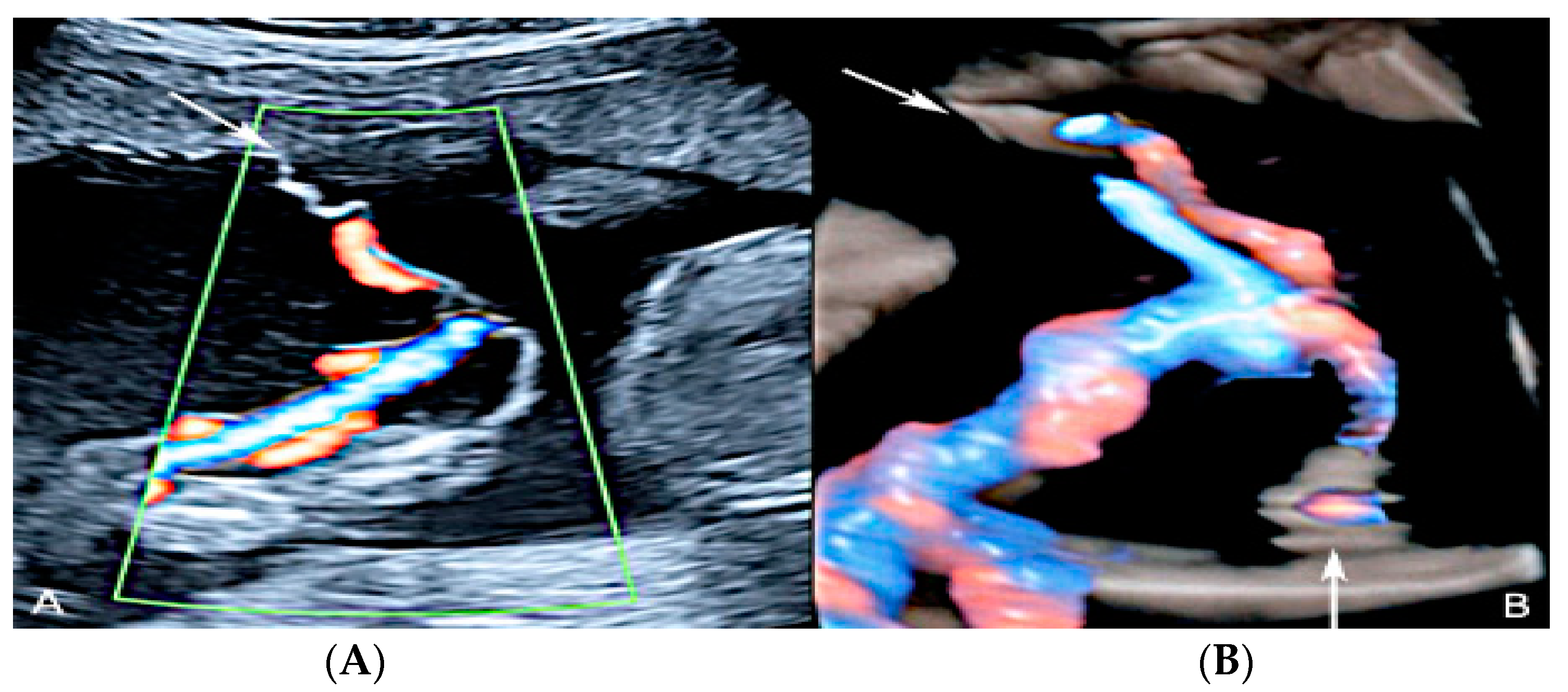
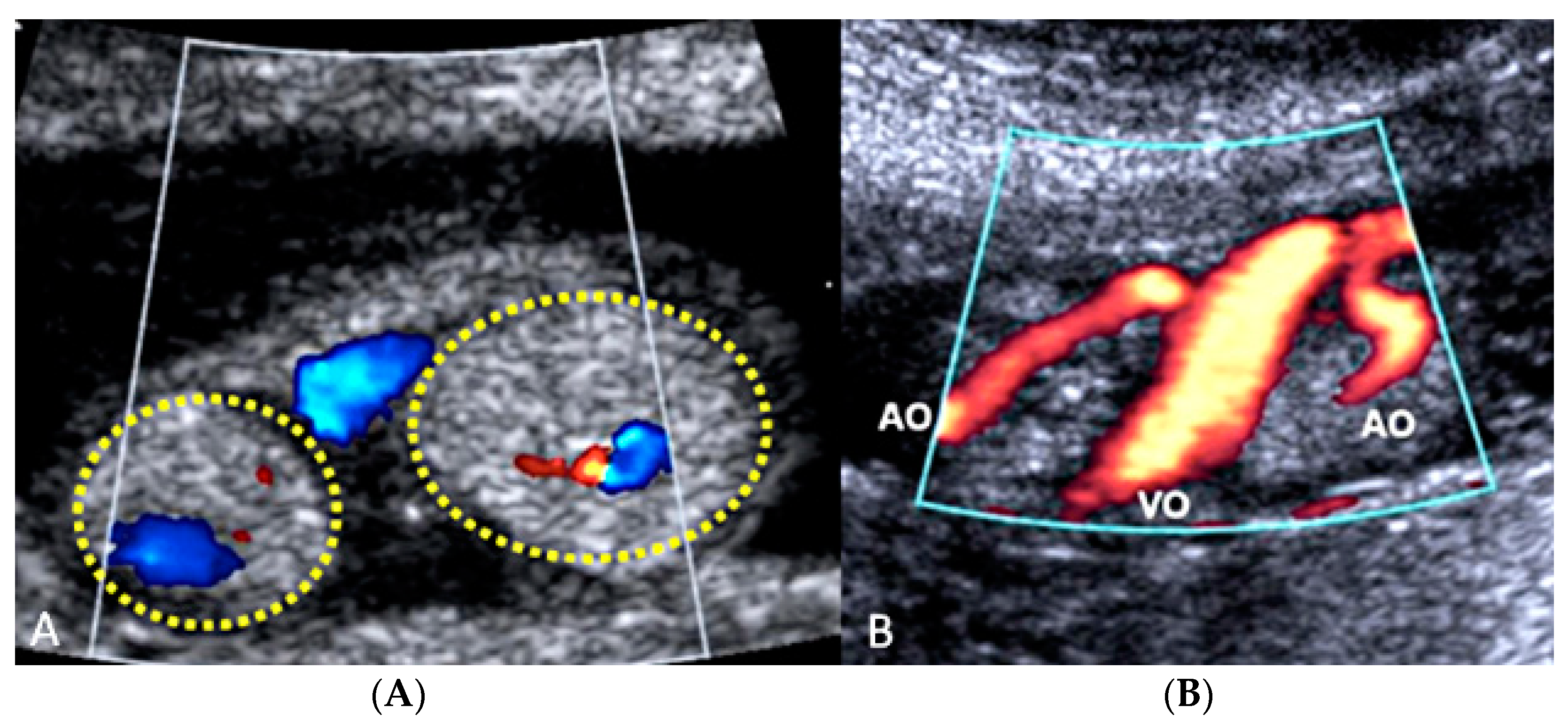
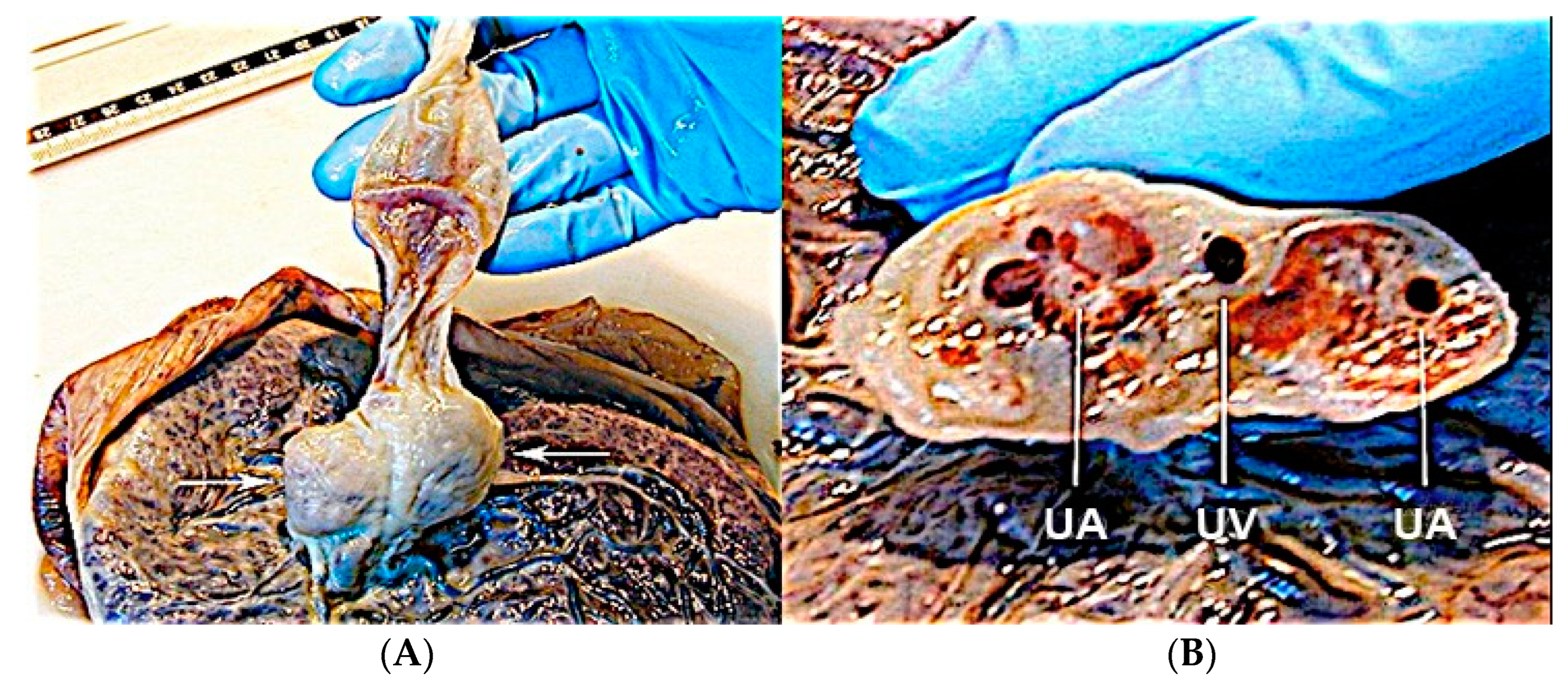
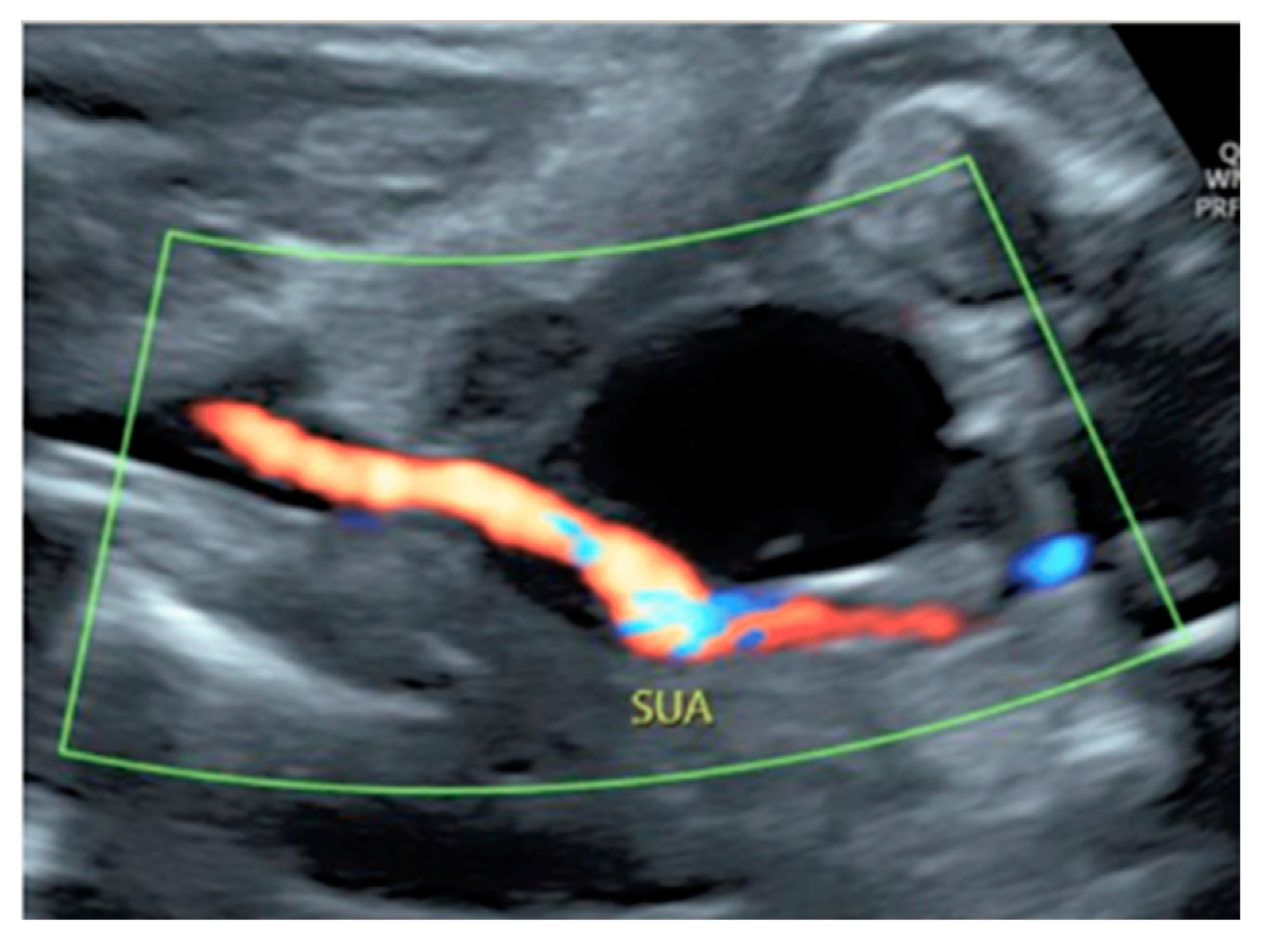
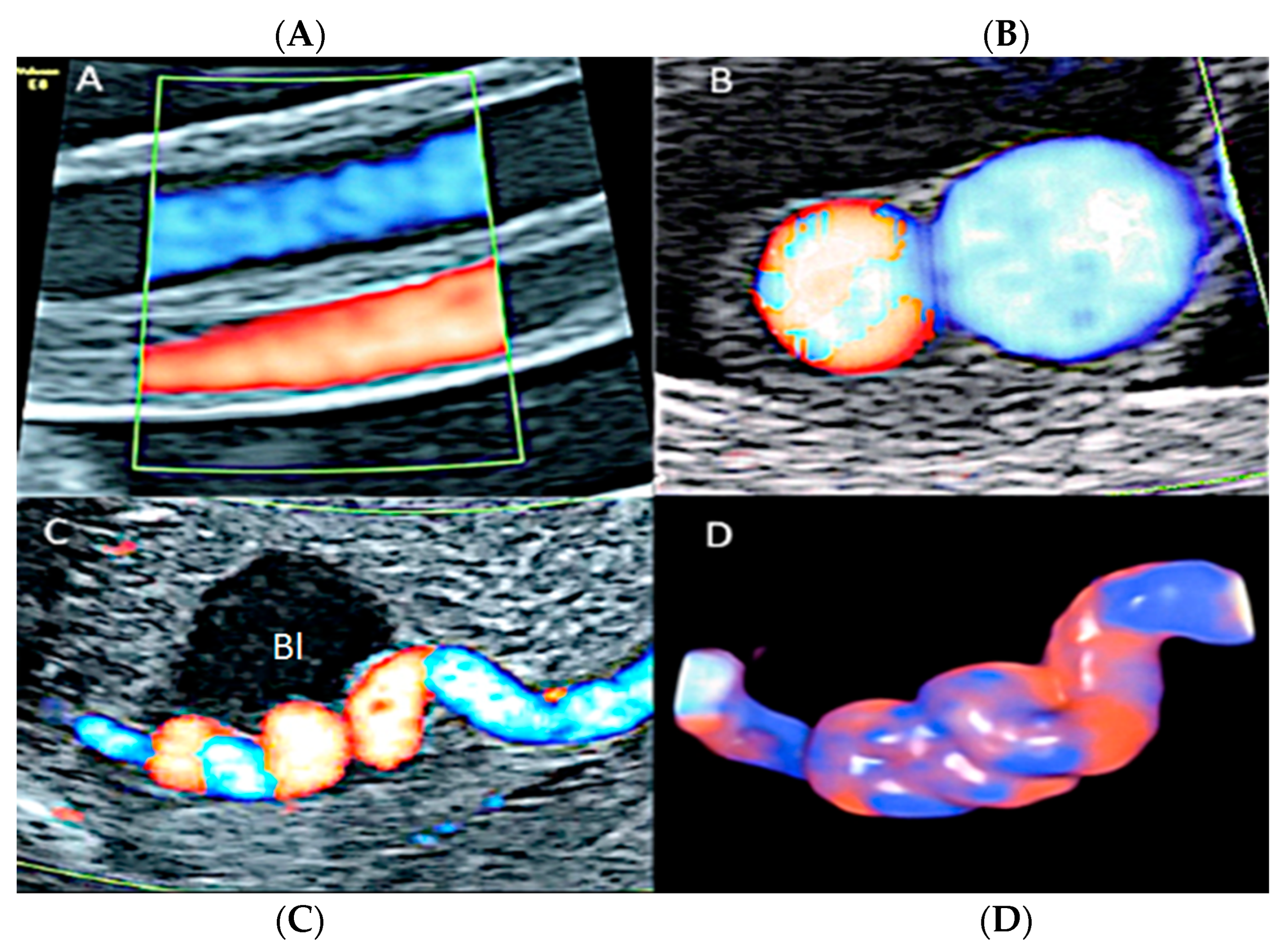
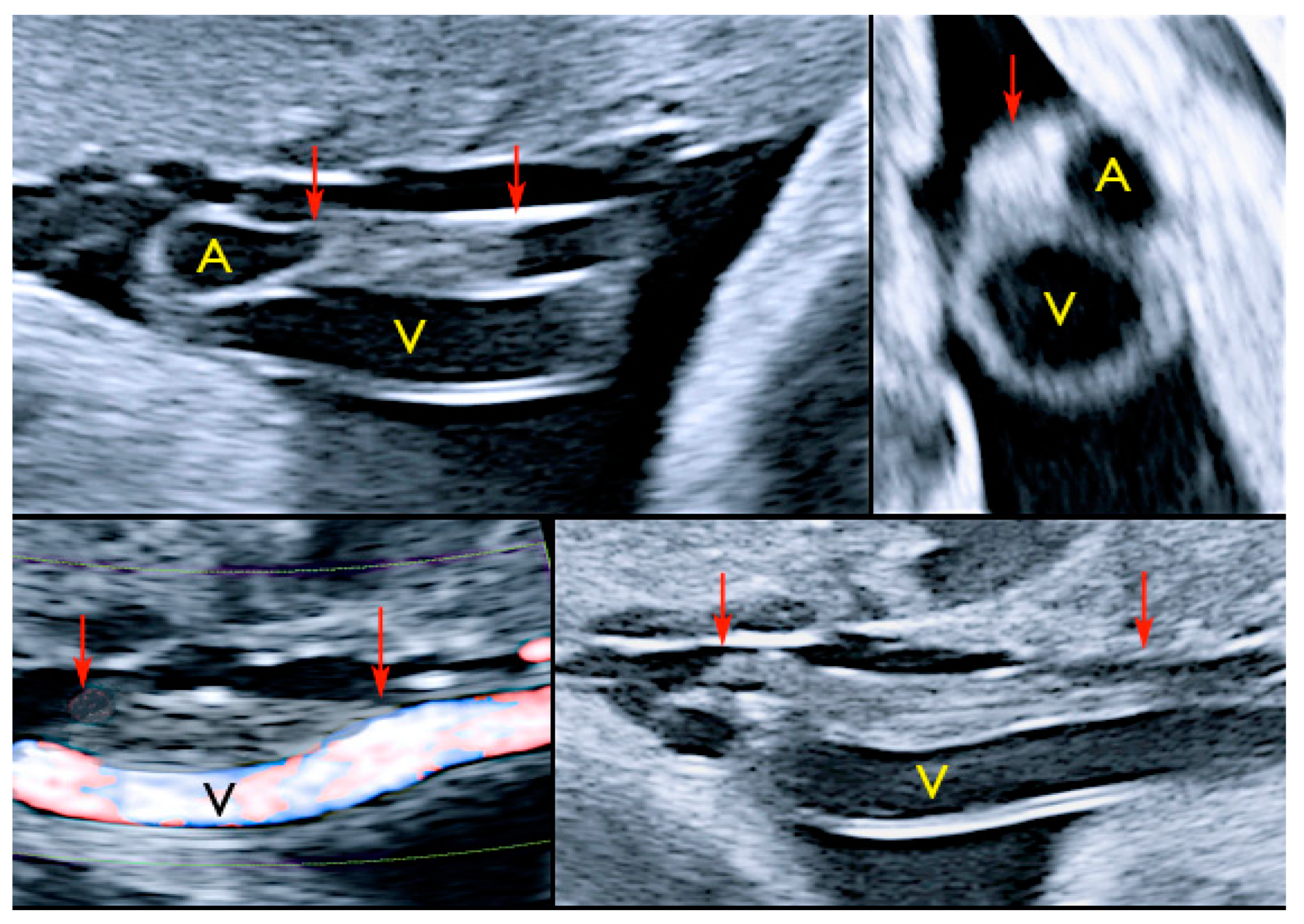
3.2.10. Single Umbilical Artery
3.2.11. Umbilical Cord Varix
3.2.12. Umbilical Vessel Thrombosis
3.2.13. Umbilical Cord Vessel Aneurysm
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rezende, G.C.; Araujo, E., Jr. Prenatal diagnosis of placenta and umbilical cord pathologies by three-dimensional ultrasound: Pictorial essay. Med. Ultrason. 2015, 14, 545–549. [Google Scholar]
- Ptacek, I.; Sebire, N.J.; Man, J.A.; Brownbill, P.; Heazell, A.E.P. Systematic review of placental pathology reported in association with stillbirth. Placenta 2014, 5, 552–562. [Google Scholar]
- Bukowski, R.; Hansen, N.I.; Pinar, H.; Willinger, M.; Reddy, U.M.; Parker, C.B.; Silver, R.M.; Dudley, D.J.; Stoll, B.J.; Saade, G.R.; et al. Altered fetal growth, placental abnormalities, and stillbirth. PLoS ONE 2017, 12, e0182874. [Google Scholar] [CrossRef] [PubMed]
- Hayes, D.J.L.; Warland, J.; Parast, M.M.; Bendon, R.W.; Hasegawa, J.; Banks, J.; Clapham, L.; Heazell, A.E.P. Umbilical cord characteristics and their association with adverse pregnancy outcomes: A systematic review and meta-analysis. PLoS ONE 2020, 15, e0239630. [Google Scholar] [CrossRef]
- Hammad, I.A.; Blue, N.R.; Allshouse, A.A.; Silver, R.M.; Gibbins, K.J.; Page, J.M.; Goldenberg, R.L.; Reddy, U.M.; Saade, G.R.; Dudley, D.J.; et al. Umbilical Cord Abnormalities and Stillbirth. Obstet. Gynecol. 2020, 135, 644–652. [Google Scholar] [CrossRef]
- Silver, R.M.; Branch, D.W. Placenta Accreta Spectrum. N. Engl. J. Med. 2018, 378, 1529–1536. [Google Scholar] [CrossRef]
- Silver, R.M. Abnormal Placentation: Placenta Previa, Vasa Previa, and Placenta Accreta. Obstet. Gynecol. 2015, 126, 654–668. [Google Scholar] [CrossRef]
- Juniaux, E.; Ayres-de-Campos, D.; Langhoff-Roos, J.; Fox, K.A.; Collins, S.; Duncombe, G.; Klaritsch, P.; Chantraine, F.; Kingdom, J.; Grønbeck, L. FIGO classification for the clinical diagnosis of placenta accreta spectrum disorders. Int. J. Gynecol. Obstet. 2019, 46, 20–24. [Google Scholar]
- Society of Gynecologic Oncology; American College of Obstetricians and Gynecologists and the Society for Maternal–Fetal Medicine; Cahill, A.G.; Beigi, R.; Heine, P.; Silver, R.M.; Wax, J.R. Placenta Accreta Spectrum. Am. J. Obstet. Gynecol. 2018, 219, B2–B16. [Google Scholar] [CrossRef]
- Silver, R.M.; Barbour, K.D. Placenta Accreta Spectrum. Obstet. Gynecol. Clin. N. Am. 2015, 42, 381–402. [Google Scholar] [CrossRef]
- Fonseca, A.; de Campos, D.A. Maternal morbidity and mortality due to placenta accreta spectrum disorders. Best Pract. Res. Clin. Obstet. Gynaecol. 2021, 72, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Jauniaux, E.; Hussein, A.M.; Fox, K.A.; Collins, S.L. New evidence-based diagnostic and management strategies for placenta accreta spectrum disorders. Best Pract. Res. Clin. Obstet. Gynaecol. 2019, 61, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Berkley, E.M.; Abuhamad, A. Imaging of Placenta Accreta Spectrum. Clin. Obstet. Gynecol. 2018, 61, 755–765. [Google Scholar] [CrossRef] [PubMed]
- Jauniaux, E.; Collins, S.; Burton, G.J. Placenta Accreta spectrum: Pathophysiology and evidence-based anatomy for prenatal ultrasound imaging. Am. J. Obstet. Gynecol. 2018, 218, 75–87. [Google Scholar] [CrossRef] [PubMed]
- Shainker, S.A.; Coleman, B.; Timor, I.E.; Bhide, A.; Bromley, B.; Cahill, A.G.; Gandhi, M.; Hecht, J.L.; Johnson, K.M.; Levine, D.; et al. Special Report of the Society for Maternal-Fetal Medicine Placenta Accreta Spectrum Ultrasound Marker Task Force: Consensus on definition of markers and approach to the ultrasound examination in pregnancies at risk for placenta accreta spectrum. Am. J. Obstet. Gynecol. 2021, 224, B2–B14. [Google Scholar] [CrossRef]
- Shih, J.C.; Kang, J.; Tsai, S.J.; Lee, J.K.; Liu, K.L.; Huang, K.Y. The “rail sign”: An ultrasound finding in placenta accreta spectrum indicating deep villous invasion and adverse outcomes. Am. J. Obstet. Gynecol. 2012, 225, e1–e17. [Google Scholar] [CrossRef]
- Januaux, E.; Kingdom, J.; Silver, R.M. A comparison of recent guidelines in diagnosis and management of placenta accreta spectrum disorders. Best Pract. Res. Clin. Obstet. Gynaecol. 2021, 72, 102–116. [Google Scholar] [CrossRef]
- Finazzo, F.; D’Antonio, F.; Masseli, G.; Folnani, F.; Palacios-Jaraquemada, J.; Minneci, G.; Gambarini, S.; Timor-Tritsch, I.; Prefumo, F.; Buca, D.; et al. Interobserver agreement in MRI assessment of severity of placenta accreta spectrum disorders. Ultrasound Obstet. Gynecol. 2020, 55, 467–473. [Google Scholar] [CrossRef]
- Arthuis, C.; Millischer, A.E.; Bussieres, L.; Mahallati, H.; Henry, C.; Ville, Y.; Salomon, L.J.; Grévent, D. MRI based morpohological examination of the placenta. Placenta 2021, 115, 20–26. [Google Scholar] [CrossRef]
- Kapoor, H.; Hanaoka, M.; Dawkins, A.; Khurana, A. Review of MRI imaging for placenta accreta spectrum: Pathophysiologic insights, imaging signs and recent developments. Placenta 2021, 104, 31–39. [Google Scholar]
- Jauniaux, E.R.M.; Alfirevic, Z.; Bhide, A.G.; Belfort, M.A.; Burton, G.J.; Collins, S.L.; Dornan, S.; Jurkovic, D.; Kayem, G.; Kingdom, J.; et al. Placenta Praevia and Placenta Accreta: Diagnosis and Management. Green-top Guideline No. 27a. BJOG 2019, 126, e1–e48. [Google Scholar] [CrossRef] [PubMed]
- Jain, V.; Bos, H.; Bujold, E. Diagnosis and management of placenta previa. J. Obstet. Gynaecol. Can. 2020, 42, 906–917. [Google Scholar] [CrossRef] [PubMed]
- Garofalo, A.; Pilloni, E.; Alemanno, M.G.; Garofalo, G.; Sciarrone, A.; Todros, T.; Viora, E. Ultrasound accuracy in prenatal diagnosis of abnormal placentation of posterior placenta previa. Eur. J. Obstet. Gynecol. Reprod. Biol. 2019, 242, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Fratelli, N.; Prefumo, F.; Maggi, C.; Cavalli, C.; Sciarrone, A.; Garofalo, A.; Viora, E.; Vergani, P.; Ornaghi, S.; Betti, M.; et al. Third-trimester ultrasound for antenatal diagnosis of placenta accreta spectrum in women with placenta previa: Results from the ADoPAD study. Ultrasound Obstet. Gynecol. 2022, 60, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Romeo, V.; Verde, F.; Sarno, L.; Migliorini, S.; Petretta, M.; Mainenti, P.P.; D’Armiento, M.; Guida, M.; Brunetti, A.; Maurea, S. Prediction of placenta accreta spectrum in patients with placenta previa using clinical risk factors, ultrasound and magnetic resonance imaging findings. Radiol. Med. 2021, 9, 1216–1225. [Google Scholar] [CrossRef]
- Guo, P.; Wu, Y.; Yuan, X.; Wan, Z. Clinical diagnostic value and analysis of MRI combined with ultrasound in prenatal pernicious placenta previa with placenta accreta. Ann. Palliat. Med. 2021, 10, 6753–6759. [Google Scholar] [CrossRef]
- Drăgușin, R.C.; Șorop-Florea, M.; Pătru, C.L.; Zorilă, L.; Marinaș, C.; Șorop, B.V.; Căpitănescu, R.; Iliescu, D.G. Abnormalities of the Placenta. In Congenital Anomalies—From the Embryo to the Neonate; Tudorache, S., Ed.; Intech Open Limited: London, UK, 2018. [Google Scholar]
- Strauss, F.; Benirschke, K.; Driscoll, S.G. Unusual Shapes of the Placenta. Placenta Accreta. In Placenta; Strauss, F., Benirschke, K., Driscoll, S.G., Eds.; Springer: Berlin/Heidelberg, Germany, 1967; pp. 106–134. [Google Scholar]
- Holzman, J.; Zalud, I.; Bartholomew, M.L. Ultrasound of the placenta. Donald. Sch. J. Ultrasound Obs. Gynecol. 2007, 1, 47–60. [Google Scholar]
- Chihara, H.; Otsubo, Y.; Ohta, Y.; Araki, T. Prenatal diagnosis of succenturiate lobe by ultrasonography and color Doppler imaging. Arch. Gynceol. Obstet. 2000, 263, 137–138. [Google Scholar]
- Kikuchi, A.; Uemura, R.; Serikawa, T.; Takakuwa, K.; Tanaka, K. Clinical significances of magnetic resonance imaging in prenatal diagnosis of vasa previa in a woman with bilobed placentas. J. Obstet. Gynaecol. Res. 2011, 31, 75–78. [Google Scholar] [CrossRef]
- Taniguchi, H.; Aoki, S.; Sakamaki, K.; Kurasawa, K.; Okuda, M.; Takahashi, T.; Hirahara, F. Circumvallate placenta: Associated clinical manifestations and complications—A retrospective study. Obstet. Gynecol. Int. 2014, 2014, 986230. [Google Scholar] [CrossRef]
- Suzuki, S. Clinical significance of pregnancies with circumvallate placenta. J. Obstet. Gynaecol. Res. 2008, 34, 51–54. [Google Scholar] [PubMed]
- Celik, O.Y.; Obut, M.; Keles, A.; Calik, M.G.; Dagdeviren, G.; Yucel, A.; Sahin, D. Outcomes of pregnancies diagnosed with circumvallate placenta, and use of uterine artery pulsatility index and maternal serum alpha-fetoprotein for prediction of adverse outcomes. Eur. J. Obstet. Gynecol. Reprod. Biol. 2021, 262, 57–61. [Google Scholar] [CrossRef] [PubMed]
- Arlicot, C.; Herve, P.; Simon, E.; Perrotin, F. Three-dimensional surface rendering of the chorionic placental plate: The “Tire” Sign for the diagnosis of a circumvallate placenta. J. Ultrasound Med. 2012, 31, 337–341. [Google Scholar] [CrossRef]
- Suzuki, S. Antenatal screening for circumvallate placenta. J. Med. Ultrason. 2008, 35, 71–73. [Google Scholar] [CrossRef] [PubMed]
- Quintana, R.L.; Baez, C.A.F.; Moraga, F.A.L.; Moraga, E.L.; Zepeda, J.H.G.; Leyva, F.; Manjarrez, R.E.A. Placental mesenchymal dysplasia, a rare pathology. Eur. J. Obstet. Gynecol. Reprod. Biol. 2019, 240, 122–138. [Google Scholar]
- Pawoo, N.; Heller, D.S. Placental Mesenchymal Dysplasia. Arch. Pathol. Lab. Med. 2014, 138, 1247–1249. [Google Scholar] [CrossRef]
- Psarris, A.; Sindos, M.; Kourtis, P.; Pampanos, A.; Antsaklis, P.; Theodora, M.; Chondrogianni, M.E.; Morphopoulos, G.; Loutradis, D.; Daskalakis, G. Placental Mesenchymal Dysplasia: Ultrasound Characteristics and Diagnostic Pitfalls. Ultrasound Int. Open 2020, 6, E2–E3. [Google Scholar] [CrossRef]
- Fan, M.; Skupski, D.W. Placental chorioangioma: Literature review. J. Perinat. Med. 2014, 42, 273–279. [Google Scholar] [CrossRef]
- Buca, D.; Iacovella, C.; Khalil, A.; Rizzo, G.; Sirotkina, M.; Makatsariya, A.; Liberati, M.; Silvi, C.; Acharya, G.; D’Antonio, F. Perinatal outcome of pregnancies complicated by placental chorioangioma: Systematic review and meta-analysis. Ultrasound Obstet. Gynecol. 2020, 55, 441–449. [Google Scholar] [CrossRef]
- D’Antonio, F.; Bhide, A. Ultrasound in placental disorders. Best Pract. Res. Clin. Obstet. Gynaecol. 2014, 28, 429–442. [Google Scholar] [CrossRef]
- Brown, D.L.; DiSlavo, D.N.; Frates, M.C.; Davidson, K.M.; Genest, D.R. Placental surface cysts detected on sonography: Histiologic and clinical correlation. J. Ultrasound Med. 2002, 1, 641–646. [Google Scholar]
- Hasegawa, J. Ultrasound Assessment of the Umbilical Cord. Donald Sch. J. Ultrasound Obs. Gynecol. 2014, 84, 382–390. [Google Scholar]
- Strong, T.H.; Jarles, D.L.; Vega, J.S.; Feldman, D.B. The umbilical coiling index. Am. J. Obstet. Gynecol. 1994, 170, 29–32. [Google Scholar] [PubMed]
- Mittal, A.; Nanda, S.; Sen, J. Antenatal umbilical coiling index as a predictor of perinatal outcome. Arch. Gynecol. Obstet. 2015, 291, 763–768. [Google Scholar]
- Moshiri, M.; Zaidi, S.F.; Robinson, T.J.; Bhargava, P.; Siebert, J.R.; Dubinsky, T.J.; Katz, D.S. Comprehensive Imaging Review of Abnormalities of the Umbilical Cord. Radiographics 2014, 34, 179–196. [Google Scholar] [CrossRef] [PubMed]
- Catanzarite, V.; Cousins, L.; Daneshmand, S.; Schwendemann, W.; Casele, H.; Adamczak, J.; Tevy, T.; Patel, A. Prenatally Diagnosed Vasa Previa: A Single-Institution Series of 96 Cases. Obstet. Gynecol. 2016, 128, 1153–1161. [Google Scholar] [CrossRef]
- Pavalagantharajah, S.; Villani, L.; D’Souza, R. Prevalence of obstetric risk factors in pregnancies with vasa previa. Am. J. Obstet. Gynecol. 2020, 2, 100117. [Google Scholar]
- Erfani, H.; Haeri, S.; Shainker, S.A.; Saad, A.F.; Ruano, R.; Dunn, T.N.; Rezaei, A.; Aalipour, S.; Nassr, A.A.; Shamshirsaz, A.A.; et al. Vasa Previa: A multicenter retrospective cohort study. Am. J. Obstet. Gynecol. 2019, 221, e1–e644. [Google Scholar]
- Zhang, W.; Geris, S.; Al-Emara, N.; Ramadan, G.; Sotiriadis, A.; Akolekar, R. Perinatal outcome of pregnancies with prenatal diagnosis of vasa previa: Systematic review and meta-analysis. Ultrasound Obstet. Gynecol. 2021, 57, 710–719. [Google Scholar] [CrossRef]
- Westcott, J.M.; Simpson, M.S.; Chasen, S.; Vieira, L.; Stone, J.; Doulaveris, G.; Dar, P.; Bernstein, P.S.; Atallah, F.; Dolin, C.D.; et al. Prenatally diagnosed vasa previa: Association with adverse obstetrical and neonatal outcomes. Am. J. Obstet. Gynecol. 2020, 2, 100206. [Google Scholar] [CrossRef]
- Klahr, R.; Fox, N.S.; Zafman, K.; Hill, M.B.; Connolly, C.T.; Rebarber, A. Frequency of spontaneous resolution of vasa previa with advancing gestational age. Am. J. Obstet. Gynecol. 2019, 221, e1–e646. [Google Scholar] [CrossRef] [PubMed]
- Yeaton-Massey, A.; Girsen, A.I.; Mayo, J.A.; Blumenfeld, Y.J.; El-Sayed, Y.Y.; Stevenson, D.K.; Shaw, G.M. Vasa previa and extreme prematurity: A population-based study. J. Perinatol. 2019, 39, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Ruiter, L.; Kok, N.; Limpens, J.; Derks, J.B.; de Graaf, I.M.; Mol, B.W.J.; Pajkrt, E. Incidence of and risk indicators for vasa praevia: A systematic review. BJOG 2016, 123, 1278–1287. [Google Scholar] [CrossRef] [PubMed]
- Sinkey, R.G.; Odibo, A.O. Vasa previa screening strategies: Decision and cost-effectiveness analysis. Ultrasound Obstet. Gynecol. 2018, 52, 522–529. [Google Scholar] [CrossRef]
- Tsakiridis, I.; Mamopoulos, A.; Athanasiadis, A.; Dagklis, T. Diagnosis and management of vasa previa: A comparison of 4 national guidelines. Obstet. Gynecol. Surv. 2019, 74, 436–442. [Google Scholar] [CrossRef]
- Fishel Bartal, M.; Sibai, B.M.; Ilan, H.; Katz, S.; Eisen, I.S.; Kassif, E.; Yinon, Y.; Mazaki-Tovi, S.; Bartal, M.F. Prenatal Diagnosis of Vasa Previa: Outpatient versus Inpatient Management. Am. J. Perinatol. 2019, 36, 422–427. [Google Scholar]
- Rodriguez, D.; Eliner, Y. Performance of ultrasound for the visualization of the placental cord insertion. Curr. Opin. Obstet. Gynecol. 2019, 31, 403–409. [Google Scholar] [CrossRef]
- Kelley, B.P.; Klochko, C.L.; Atkinson, S.; Hillman, D.; Craig, B.M.; Sandberg, S.A.; Gaba, A.R.; Halabi, S.S. Sonographic Diagnosis of Velamentous and Marginal Placental Cord Insertion. Ultrasound Q. 2020, 36, 247–254. [Google Scholar] [CrossRef]
- Aurioles-Garibay, A.; Hernandez-Andrade, E.; Romero, R.; Garcia, M.; Qureshi, F.; Jacques, S.M.; Ahn, H.; Yeo, L.; Chaiworapongsa, T.; Hassan, S.S. Presence of an Umbilical Artery Notch in Monochorionic/Monoamniotic Twins. Fetal Diagn. Ther. 2014, 36, 305–311. [Google Scholar] [CrossRef]
- Papadopoulos, V.G.; Kourea, H.P.; Adonakis, G.L.; Decavalas, G.O. A case of umbilical cord hemangioma: Doppler studies and review of the literature. Eur. J. Obstet. Gynecol. Reprod. Biol. 2009, 144, 8–14. [Google Scholar] [CrossRef]
- Lok, W.Y.; Law, K.M.; Ho, C.L.; Leung, T.Y. Prenatal diagnosis of umbilical cord hemangioma. Ultrasound Obstet. Gynecol. 2022, 59, 392–393. [Google Scholar] [PubMed]
- Clemont-Hama, Y.; Thibouw, K.; Devisme, L.; Franquet-Ansart, H.; Stichelbout, M.; Subtil, D. Risk factors for spontaneous hematoma of the umbilical cord: A case-control study. Placenta 2020, 99, 152–156. [Google Scholar] [CrossRef] [PubMed]
- Zangen, R.; Boldes, R.; Yaffe, H.; Schwed, P.; Weiner, Z. Umbilical cord cysts in the second and third trimesters: Significance and prenatal approach. Ultrasound Obstet. Gynecol. 2010, 36, 296–301. [Google Scholar] [PubMed]
- Weissman, A.; Drugan, A. Sonographic findings of the umbilical cord: Implications for the risk of fetal chromosomal anomalies. Ultrasound Obstet. Gynecol. 2001, 17, 536–541. [Google Scholar]
- Lubusky, M.; Dhaifalah, I.; Prochazka, M.; Hyjanek, J.; Mickova, I.; Vomackova, K.; Santavy, J. Single umbilical artery and its siding in the second trimester of pregnancy: Relation to chromosomal defects. Prenat. Diagn. 2007, 27, 327–331. [Google Scholar]
- Di Pasquo, E.; Kuleva, M.; O’Gorman, N.; Ville, Y.; Salomon, L.J. Fetal intra-abdominal umbilical vein varix: Retrospective cohort study and systematic review and meta-analysis. Ultrasound Obstet. Gynecol. 2018, 51, 580–585. [Google Scholar]
- Sherer, D.M.; Al-Haddad, S.; Cheng, R.; Dalloul, M. Current perspectives of prenatal sonography of umbilical cord morphology. Int. J. Womens Health 2021, 13, 939–971. [Google Scholar]
- Zaidi, A.F.; Moshiri, M.; Osman, S.; Robinson, T.J.; Siebert, J.; Bhargava, P.; Katz, D.S. Comprehensive imaging review of abnormalities of the placenta. Ultrasound Q. 2016, 32, 25–42. [Google Scholar]
- Abramowicz, J.S.; Sheiner, E. Ultrasound of the placenta: A systematic approach. Part 1: Imaging. Placenta. 2008, 29, 225–240. [Google Scholar]
- Hasegawa, J. Ultrasound screening of umbilical cord abnormalities and delivery management. Placenta 2018, 62, 66–78. [Google Scholar]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santana, E.F.M.; Castello, R.G.; Rizzo, G.; Grisolia, G.; Araujo Júnior, E.; Werner, H.; Lituania, M.; Tonni, G. Placental and Umbilical Cord Anomalies Diagnosed by Two- and Three-Dimensional Ultrasound. Diagnostics 2022, 12, 2810. https://doi.org/10.3390/diagnostics12112810
Santana EFM, Castello RG, Rizzo G, Grisolia G, Araujo Júnior E, Werner H, Lituania M, Tonni G. Placental and Umbilical Cord Anomalies Diagnosed by Two- and Three-Dimensional Ultrasound. Diagnostics. 2022; 12(11):2810. https://doi.org/10.3390/diagnostics12112810
Chicago/Turabian StyleSantana, Eduardo Félix Martins, Renata Gomes Castello, Giuseppe Rizzo, Gianpaolo Grisolia, Edward Araujo Júnior, Heron Werner, Mario Lituania, and Gabriele Tonni. 2022. "Placental and Umbilical Cord Anomalies Diagnosed by Two- and Three-Dimensional Ultrasound" Diagnostics 12, no. 11: 2810. https://doi.org/10.3390/diagnostics12112810
APA StyleSantana, E. F. M., Castello, R. G., Rizzo, G., Grisolia, G., Araujo Júnior, E., Werner, H., Lituania, M., & Tonni, G. (2022). Placental and Umbilical Cord Anomalies Diagnosed by Two- and Three-Dimensional Ultrasound. Diagnostics, 12(11), 2810. https://doi.org/10.3390/diagnostics12112810







