Analysis of a Routinely Used Commercial Anti-Chikungunya IgM ELISA Reveals Cross-Reactivities with Dengue in Brazil: A New Challenge for Differential Diagnosis?
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Clinical Samples
2.3. Anti-Chikungunya Virus IgM ELISA
2.4. Statistical Analysis
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Silva, N.M.D.; Teixeira, R.A.G.; Cardoso, C.G.; Junior, J.B.S.; Coelho, G.E.; Oliveira, E.S.F.D. Chikungunya surveillance in Brazil: Challenges in the context of Public Health. Epidemiol. Serv. Saúde 2018, 27. [Google Scholar] [CrossRef]
- de Souza, T.M.A.; Ribeiro, E.D.; Corrêa, V.C.E.; Damasco, P.V.; Santos, C.C.; de Bruycker-Nogueira, F.; Chouin-Carneiro, T.; Faria, N.; Nunes, P.C.G.; Heringer, M.; et al. Following in the Footsteps of the Chikungunya Virus in Brazil: The First Autochthonous Cases in Amapá in 2014 and Its Emergence in Rio de Janeiro during 2016. Viruses 2018, 10, 623. [Google Scholar] [CrossRef] [PubMed]
- Moreira, J.; Bressan, C.S.; Brasil, P.; Siqueira, A.M. Epidemiology of acute febrile illness in Latin America. Clin. Microbiol. Infect. 2018, 24, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Santos, F.B.D.; Filippis, A.M.B.D.; Araújo, E.S.M.D.; Lima, M.d.R.Q.; Nogueira, F.D.B.; Faria, N.R.D.C.; Simões, J.B.S.; Sampaio, S.A.; Nunes, P.C.G.; Silva, M.H.D.; et al. A Review on Dengue Diagnosis and Epidemiology by a Regional Reference Laboratory in 25 years, Rio de Janeiro, Brazil. Dengue Bull. 2013, 31, 61–76. [Google Scholar]
- Heringer, M.; Nogueira, R.M.; de Filippis, A.M.; Lima, M.R.; Faria, N.R.; Nunes, P.C.; Nogueira, F.B.; dos Santos, F.B. Impact of the emergence and re-emergence of different dengue viruses’ serotypes in Rio de Janeiro, Brazil, 2010 to 2012. Trans. R. Soc. Trop. Med. Hyg. 2015, 109, 268–274. [Google Scholar] [CrossRef]
- Azeredo, E.L.; Dos Santos, F.B.; Barbosa, L.S.; Souza, T.M.A.; Badolato-Corrêa, J.; Sánchez-Arcila, J.C.; Nunes, P.C.G.; de-Oliveira-Pinto, L.M.; de Filippis, A.M.; Dal Fabbro, M.; et al. Clinical and Laboratory Profile of Zika and Dengue Infected Patients: Lessons Learned From the Co-circulation of Dengue, Zika and Chikungunya in Brazil. PLoS Curr. 2018, 10. [Google Scholar] [CrossRef]
- Tauro, L.B.; Bandeira, A.C.; Ribeiro, G.S.; Reis, M.G.; Pizarro, C.P.; Araujo, K.A.; Castro, A.P.; Queiroz, J.M.; Simon, S.S.; Menezes, G.; et al. Potential use of saliva samples to diagnose Zika virus infection. J. Med. Virol. 2017, 89, 1–2. [Google Scholar] [CrossRef]
- Farias, L.; Beserra, F.; Fernandes, L.; Teixeira, A.A.R.; Ferragut, J.M.; Girão, E.S.; Pires Neto, R.D.J. Myocarditis Following Recent Chikungunya and Dengue Virus Coinfection: A Case Report. Arq. Bras. Cardiol. 2019, 113, 783–786. [Google Scholar] [CrossRef]
- Prat, C.M.; Flusin, O.; Panella, A.; Tenebray, B.; Lanciotti, R.; Leparc-Goffart, I. Evaluation of commercially available serologic diagnostic tests for chikungunya virus. Emerg. Infect. Dis. 2014, 20, 2129–2132. [Google Scholar] [CrossRef]
- Yap, G.; Pok, K.Y.; Lai, Y.L.; Hapuarachchi, H.C.; Chow, A.; Leo, Y.S.; Tan, L.K.; Ng, L.C. Evaluation of Chikungunya diagnostic assays: Differences in sensitivity of serology assays in two independent outbreaks. PLoS Negl. Trop. Dis. 2010, 4, e753. [Google Scholar] [CrossRef]
- Caglioti, C.; Lalle, E.; Castilletti, C.; Carletti, F.; Capobianchi, M.R.; Bordi, L. Chikungunya virus infection: An overview. New Microbiol. 2013, 36, 211–227. [Google Scholar]
- Grivard, P.; Le Roux, K.; Laurent, P.; Fianu, A.; Perrau, J.; Gigan, J.; Hoarau, G.; Grondin, N.; Staikowsky, F.; Favier, F.; et al. Molecular and serological diagnosis of Chikungunya virus infection. Pathol. Biol. 2007, 55, 490–494. [Google Scholar] [CrossRef] [PubMed]
- Chelluboina, S.; Robin, S.; Aswathyraj, S.; Arunkumar, G. Persistence of antibody response in chikungunya. Virusdisease 2019, 30, 469–473. [Google Scholar] [CrossRef] [PubMed]
- Nunes, P.C.G.; de Filippis, A.M.B.; Lima, M.; Faria, N.; de Bruycker-Nogueira, F.; Santos, J.B.; Heringer, M.; Chouin-Carneiro, T.; Couto-Lima, D.; de Santis Gonçalves, B.; et al. 30 years of dengue fatal cases in Brazil: A laboratorial-based investigation of 1047 cases. BMC Infect. Dis. 2018, 18, 346. [Google Scholar] [CrossRef] [PubMed]
- Panning, M.; Charrel, R.N.; Donoso Mantke, O.; Landt, O.; Niedrig, M.; Drosten, C. Coordinated implementation of chikungunya virus reverse transcription-PCR. Emerg. Infect Dis. 2009, 15, 469–471. [Google Scholar] [CrossRef]
- Joseph, A.Y.; Babu, V.S.; Dev, S.S.; Gopalakrishnapai, J.; Harish, M.; Rajesh, M.D.; Anisha, S.; Mohankumar, C. Rapid detection and characterization of Chikungunya virus by RT-PCR in febrile patients from Kerala, India. Indian J. Exp. Biol. 2008, 46, 573–578. [Google Scholar]
- Lanciotti, R.S.; Kosoy, O.L.; Laven, J.J.; Panella, A.J.; Velez, J.O.; Lambert, A.J.; Campbell, G.L. Chikungunya Virus in US Travelers Returning from India, 2006. Emerg. Infect. Dis. 2007, 13, 764–767. [Google Scholar]
- Carletti, F.; Bordi, L.; Chiappini, R.; Ippolito, G.; Sciarrone, M.R.; Capobianchi, M.R.; Di Caro, A.; Castilletti, C. Rapid detection and quantification of Chikungunya virus by a one-step reverse transcription polymerase chain reaction real-time assay. Am. J. Trop. Med. Hyg. 2007, 77, 521–524. [Google Scholar]
- Laurent, P.; Le Roux, K.; Grivard, P.; Bertil, G.; Naze, F.; Picard, M.; Staikowsky, F.; Barau, G.; Schuffenecker, I.; Michault, A. Development of a sensitive real-time reverse transcriptase PCR assay with an internal control to detect and quantify chikungunya virus. Clin. Chem. 2007, 53, 1408–1414. [Google Scholar] [CrossRef]
- Santhosh, S.R.; Parida, M.M.; Dash, P.K.; Pateriya, A.; Pattnaik, B.; Pradhan, H.K.; Tripathi, N.K.; Ambuj, S.; Gupta, N.; Saxena, P.; et al. Development and evaluation of SYBR Green I-based one-step real-time RT-PCR assay for detection and quantification of Chikungunya virus. J. Clin. Virol. 2007, 39, 188–193. [Google Scholar] [CrossRef]
- Pastorino, B.; Bessaud, M.; Grandadam, M.; Murri, S.; Tolou, H.J.; Peyrefitte, C.N. Development of a TaqMan RT-PCR assay without RNA extraction step for the detection and quantification of African Chikungunya viruses. J. Virol. Methods 2005, 124, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Hasebe, F.; Parquet, M.C.; Pandey, B.D.; Mathenge, E.G.; Morita, K.; Balasubramaniam, V.; Saat, Z.; Yusop, A.; Sinniah, M.; Natkunam, S.; et al. Combined detection and genotyping of Chikungunya virus by a specific reverse transcription-polymerase chain reaction. J. Med. Virol. 2002, 67, 370–374. [Google Scholar] [CrossRef] [PubMed]
- Pfeffer, M.; Linssen, B.; Parke, M.D.; Kinney, R.M. Specific detection of chikungunya virus using a RT-PCR/nested PCR combination. J. Vet. Med. B Infect Dis. Vet. Public Health 2002, 49, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Litzba, N.; Schuffenecker, I.; Zeller, H.; Drosten, C.; Emmerich, P.; Charrel, R.; Kreher, P.; Niedrig, M. Evaluation of the first commercial chikungunya virus indirect immunofluorescence test. J. Virol. Methods 2008, 149, 175–179. [Google Scholar] [CrossRef]
- Reddy, A.; Bosch, I.; Salcedo, N.; Herrera, B.B.; de Puig, H.; Narváez, C.F.; Caicedo-Borrero, D.M.; Lorenzana, I.; Parham, L.; García, K.; et al. Development and Validation of a Rapid Lateral Flow E1/E2-Antigen Test and ELISA in Patients Infected with Emerging Asian Strain of Chikungunya Virus in the Americas. Viruses 2020, 12, 971. [Google Scholar] [CrossRef]
- Prince, H.E.; Altrich, M.L.; Nowicki, M.J. Evaluation of Two Enzyme-Linked Immunosorbent Assay Kits for Chikungunya Virus IgM Using Samples from Deceased Organ and Tissue Donors. Clin. Vaccine Immunol. 2016, 23, 825–830. [Google Scholar] [CrossRef][Green Version]
- De Salazar, P.M.; Valadere, A.M.; Goodman, C.H.; Johnson, B.W. Evaluation of three commercially-available chikungunya virus immunoglobulin G immunoassays. Rev. Panam. Salud. Publica 2017, 41, e62. [Google Scholar] [CrossRef]
- Johnson, B.W.; Goodman, C.H.; Holloway, K.; de Salazar, P.M.; Valadere, A.M.; Drebot, M.A. Evaluation of Commercially Available Chikungunya Virus Immunoglobulin M Detection Assays. Am. J. Trop. Med. Hyg. 2016, 95, 182–192. [Google Scholar] [CrossRef]
- Mehta, R.; Soares, C.N.; Medialdea-Carrera, R.; Ellul, M.; da Silva, M.T.T.; Rosala-Hallas, A.; Jardim, M.R.; Burnside, G.; Pamplona, L.; Bhojak, M.; et al. The spectrum of neurological disease associated with Zika and chikungunya viruses in adults in Rio de Janeiro, Brazil: A case series. PLoS Negl. Trop. Dis. 2018, 12, e0006212. [Google Scholar] [CrossRef]
- Miagostovich, M.P.; Nogueira, R.M.; dos Santos, F.B.; Schatzmayr, H.G.; Araújo, E.S.; Vorndam, V. Evaluation of an IgG enzyme-linked immunosorbent assay for dengue diagnosis. J. Clin. Virol. Off. Publ. Pan Am. Soc. Clin. Virol. 1999, 14, 183–189. [Google Scholar] [CrossRef]
- Lanciotti, R.S.; Calisher, C.H.; Gubler, D.J.; Chang, G.J.; Vorndam, A.V. Rapid detection and typing of dengue viruses from clinical samples by using reverse transcriptase-polymerase chain reaction. J. Clin. Microbiol. 1992, 30, 545–551. [Google Scholar] [CrossRef] [PubMed]
- Johnson, B.W.; Russell, B.J.; Lanciotti, R.S. Serotype-specific detection of dengue viruses in a fourplex real-time reverse transcriptase PCR assay. J. Clin. Microbiol. 2005, 43, 4977–4983. [Google Scholar] [CrossRef] [PubMed]
- Lanciotti, R.S.; Kosoy, O.L.; Laven, J.J.; Velez, J.O.; Lambert, A.J.; Johnson, A.J.; Stanfield, S.M.; Duffy, M.R. Genetic and Serologic Properties of Zika Virus Associated with an Epidemic, Yap State, Micronesia, 2007. Emerg. Infect Dis. 2008, 14. [Google Scholar] [CrossRef] [PubMed]
- Nunes, M.R.; Faria, N.R.; de Vasconcelos, J.M.; Golding, N.; Kraemer, M.U.; de Oliveira, L.F.; da Silva Azevedo, R.D.; da Silva, D.E.; da Silva, E.V.; da Silva, S.P.; et al. Emergence and potential for spread of Chikungunya virus in Brazil. BMC Med. 2015, 13, 102. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, R.M.R.; Miagostovich, M.P.; Lampe, E.; Schatzmayr, H.G. Isolation of dengue virus type 2 in Rio de Janeiro. Mem. Inst. Oswaldo. Cruz. 1990, 85, 253. [Google Scholar] [CrossRef][Green Version]
- Schatzmayr, H.G.; Nogueira, R.M.R.; Rosa, A.P.A.T. An outbreak of dengue virus at Rio de Janeiro—1986. Mem. Inst. Oswaldo. Cruz. 1986, 81, 245–246. [Google Scholar] [CrossRef]
- Soares, C.N.; Brasil, P.; Carrera, R.M.; Sequeira, P.; de Filippis, A.B.; Borges, V.A.; Theophilo, F.; Ellul, M.A.; Solomon, T. Fatal encephalitis associated with Zika virus infection in an adult. J Clin Virol. Off. Publ. Pan Am. Soc. Clin. Virol. 2016, 83, 63–65. [Google Scholar] [CrossRef]
- Zanluca, C.; Melo, V.C.; Mosimann, A.L.; Santos, G.I.; Santos, C.N.; Luz, K. First report of autochthonous transmission of Zika virus in Brazil. Mem. Inst. Oswaldo. Cruz. 2015, 110, 569–572. [Google Scholar] [CrossRef]
- Health-Surveillance-Secretariat. Epidemiological Bulletin—Monitoring of Dengue Cases, Chikungunya Fever and Acute Disease Caused by Zika Virus until the Epidemiological Week 52 of 2018; Brazilian-Ministry-of-Health: Brasília, Brazil, 2019; Volume 50, p. 14.
- Pialoux, G.; Gaüzère, B.-A.; Jauréguiberry, S.; Strobel, M. Chikungunya, an epidemic arbovirosis. Lancet Infect Dis. 2007, 7, 319–327. [Google Scholar] [CrossRef]
- Honorio, N.A.; Camara, D.C.; Calvet, G.A.; Brasil, P. Chikungunya: An arbovirus infection in the process of establishment and expansion in Brazil. Cad. Saúde Pública 2015, 31, 906–908. [Google Scholar] [CrossRef]
- Dash, M.; Mohanty, I.; Padhi, S. Laboratory diagnosis of chikungunya virus: Do we really need it? Indian J. Med. Sci. 2011, 65, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Reddy, V.; Ravi, V.; Desai, A.; Parida, M.; Powers, A.M.; Johnson, B.W. Utility of IgM ELISA, TaqMan real-time PCR, reverse transcription PCR, and RT-LAMP assay for the diagnosis of Chikungunya fever. J. Med. Virol. 2012, 84, 1771–1778. [Google Scholar] [CrossRef]
- Carrillo-Hernández, M.Y.; Ruiz-Saenz, J.; Villamizar, L.J.; Gómez-Rangel, S.Y.; Martínez-Gutierrez, M. Co-circulation and simultaneous co-infection of dengue, chikungunya, and zika viruses in patients with febrile syndrome at the Colombian-Venezuelan border. BMC Infect. Dis. 2018, 18, 61. [Google Scholar] [CrossRef] [PubMed]
- de Souza Costa, M.C.; Siqueira Maia, L.M.; Costa de Souza, V.; Gonzaga, A.M.; Correa de Azevedo, V.; Ramos Martins, L.; Chavez Pavoni, J.H.; Gomes Naveca, F.; Dezengrini Slhessarenko, R. Arbovirus investigation in patients from Mato Grosso during Zika and Chikungunya virus introdution in Brazil, 2015–2016. Acta Trop. 2019, 190, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Mercado-Reyes, M.; Acosta-Reyes, J.; Navarro-Lechuga, E.; Corchuelo, S.; Rico, A.; Parra, E.; Tolosa, N.; Pardo, L.; González, M.; Martìn-Rodriguez-Hernández, J.; et al. Dengue, chikungunya and zika virus coinfection: Results of the national surveillance during the zika epidemic in Colombia. Epidemiol. Infect. 2019, 147, e77. [Google Scholar] [CrossRef] [PubMed]
- Castellanos, J.E.; Jaimes, N.; Coronel-Ruiz, C.; Rojas, J.P.; Mejía, L.F.; Villarreal, V.H.; Maya, L.E.; Claros, L.M.; Orjuela, C.; Calvo, E.; et al. Dengue-chikungunya coinfection outbreak in children from Cali, Colombia, in 2018–2019. Int. J. Infect. Dis. 2020, 102, 97–102. [Google Scholar] [CrossRef]
- Sánchez-Arcila, J.C.; Badolato-Correa, J.; de Souza, T.M.A.; Paiva, I.A.; Barbosa, L.S.; Nunes, P.C.G.; Lima, M.; Dos Santos, F.B.; Damasco, P.V.; da Cunha, R.V.; et al. Clinical, Virol.ogical, and Immunological Profiles of DENV, ZIKV, and/or CHIKV-Infected Brazilian Patients. Intervirology 2020, 63, 33–45. [Google Scholar] [CrossRef]
- Silva, M.M.O.; Tauro, L.B.; Kikuti, M.; Anjos, R.O.; Santos, V.C.; Gonçalves, T.S.F.; Paploski, I.A.D.; Moreira, P.S.S.; Nascimento, L.C.J.; Campos, G.S.; et al. Concomitant Transmission of Dengue, Chikungunya, and Zika Viruses in Brazil: Clinical and Epidemiological Findings From Surveillance for Acute Febrile Illness. Clin. Infect. Dis. 2019, 69, 1353–1359. [Google Scholar] [CrossRef]
- Abhishek, K.S.; Chakravarti, A. Simultaneous detection of IgM antibodies against dengue and chikungunya: Coinfection or cross-reactivity? J. Fam. Med. Prim. Care 2019, 8, 2420–2423. [Google Scholar] [CrossRef]
- Londhey, V.; Agrawal, S.; Vaidya, N.; Kini, S.; Shastri, J.S.; Sunil, S. Dengue and Chikungunya Virus Co-infections: The Inside Story. J. Assoc. Physicians India 2016, 64, 36–40. [Google Scholar]
- Pessoa, R.; Patriota, J.V.; Lourdes de Souza, M.; Felix, A.C.; Mamede, N.; Sanabani, S.S. Investigation Into an Outbreak of Dengue-like Illness in Pernambuco, Brazil, Revealed a Cocirculation of Zika, Chikungunya, and Dengue Virus Type 1. Medicine 2016, 95, e3201. [Google Scholar] [CrossRef] [PubMed]
- Cabral-Castro, M.J.; Cavalcanti, M.G.; Peralta, R.H.S.; Peralta, J.M. Molecular and serological techniques to detect co-circulation of DENV, ZIKV and CHIKV in suspected dengue-like syndrome patients. J. Clin. Virol. Off. Publ. Pan Am. Soc. Clin. Virol. 2016, 82, 108–111. [Google Scholar] [CrossRef] [PubMed]
- Vongpunsawad, S.; Intharasongkroh, D.; Thongmee, T.; Poovorawan, Y. Seroprevalence of antibodies to dengue and chikungunya viruses in Thailand. PLoS ONE 2017, 12, e0180560. [Google Scholar] [CrossRef] [PubMed]
- Kaur, M.; Singh, K.; Sidhu, S.K.; Devi, P.; Kaur, M.; Soneja, S.; Singh, N. Coinfection of chikungunya and dengue viruses: A serological study from North Western region of Punjab, India. J. Lab. Physicians 2018, 10, 443–447. [Google Scholar] [CrossRef]
- Chaves, T.; Pellini, A.; Mascheretti, M.; Jahnel, M.; Ribeiro, A.; Rodrigues, S. Travelers as Sentinels for Chikungunya Fever, Brazil. Emerg. Infect. Dis. 2012, 18, 530–531. [Google Scholar] [CrossRef]
- Albuquerque, I.G.C.; Marandino, R.; Mendonça, A.P.; Nogueira, R.M.R.; Vasconcelos, P.F.C.; Guerra, L.R.; Brandão, B.C.; Mendonça, A.P.P.; Aguiar, G.R.; Bacco, P.A.M. Chikungunya virus infection: Report of the first case diagnosed in Rio de Janeiro, Brazil. Rev. Soc. Bras. Med. 2012, 45, 128–129. [Google Scholar] [CrossRef]
- Felix, A.C.; Souza, N.C.S.; Figueiredo, W.M.; Costa, A.A.; Inenami, M.; da Silva, R.M.G.; Levi, J.E.; Pannuti, C.S.; Romano, C.M. Cross reactivity of commercial anti-dengue immunoassays in patients with acute Zika virus infection. J. Med. Virol. 2017, 89, 1477–1479. [Google Scholar] [CrossRef]
- Zaidi, M.B.; Cedillo-Barron, L.; Gonzalez, Y.A.M.E.; Garcia-Cordero, J.; Campos, F.D.; Namorado-Tonix, K.; Perez, F. Serological tests reveal significant cross-reactive human antibody responses to Zika and Dengue viruses in the Mexican population. Acta Trop. 2020, 201, 105201. [Google Scholar] [CrossRef]
- Kikuti, M.B.; Tauro, L.; Moreira, P.S.S.; Nascimento, L.C.J.; Portilho, M.M.; Soares, G.C.; Weaver, S.C.; Reisa, M.G.; Kitrona, U.; Ribeiro, G.S. Evaluation of two commercially available chikungunya virus IgM enzyme-linked immunoassays (ELISA) in a setting of concomitant transmission of chikungunya, dengue and Zika viruses. Int. J. Infect. Dis. 2020, 91, 38–43. [Google Scholar] [CrossRef]
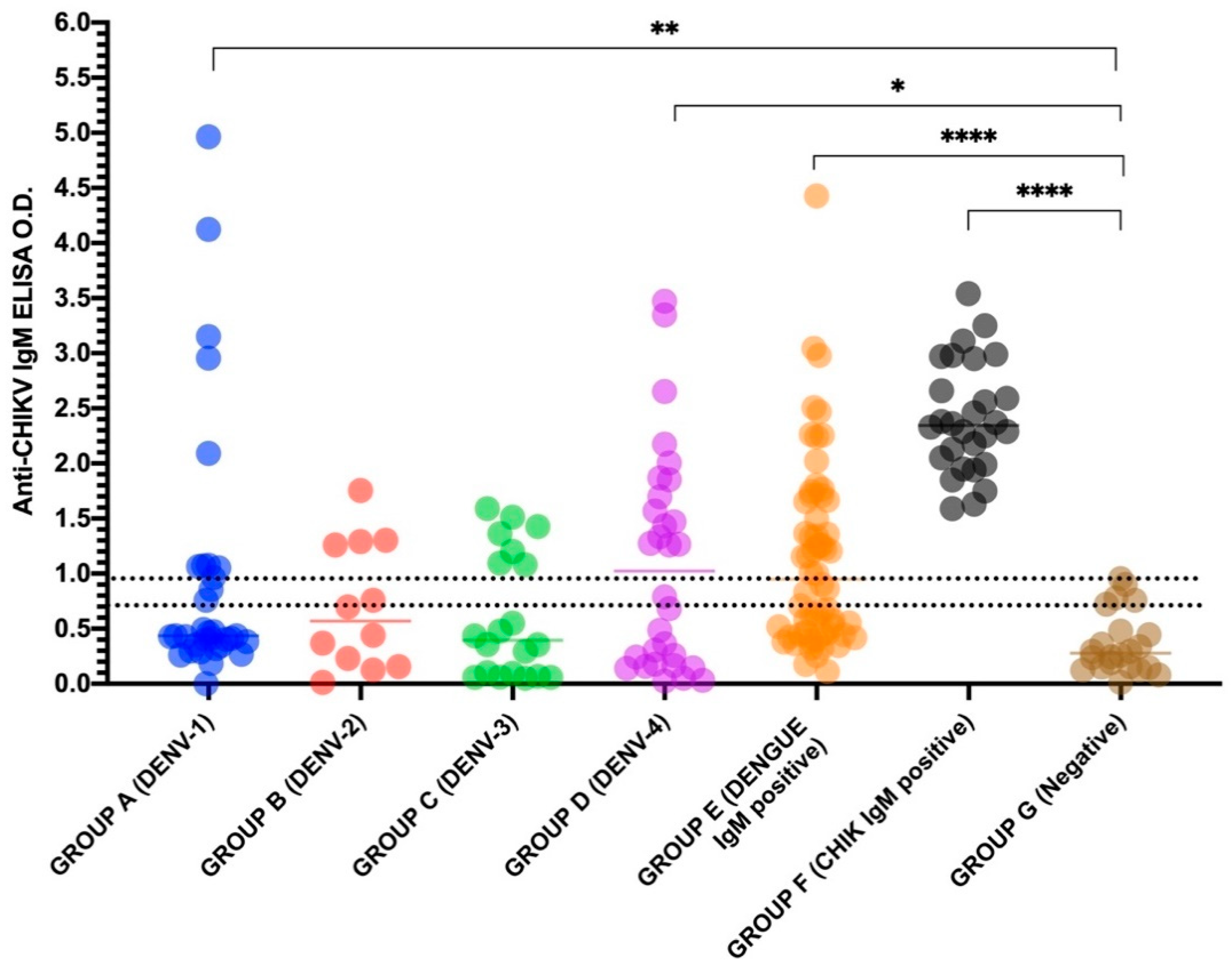
| Groups | Sample Year | Anti-Chikungunya Virus IgM ELISA (Euroimmun) Positive/Tested (%) |
|---|---|---|
| A (DENV-1 cases, n = 33) | 1998–2001 | 6/33 (18.2) |
| B (DENV-2 cases, n = 12) | 1999–2010 | 4/12 (33.3) |
| C (DENV-3 cases, n = 20) | 2001–2002 | 5/20 (25.0) |
| D (DENV-4 cases, n = 30) | 2012–2013 | 15/30 (50.0) |
| E (Dengue IgM positive cases, n = 60) | 1998–2003 | 28/60 (46.7) |
| TOTAL OF GROUP A–E (n = 155) | 58/155 (37.4) | |
| F (Chikungunya cases, n = 28) | 2018 | 28/28 (100) |
| TOTAL OF GROUP F (n = 28) | 28/28 (100) | |
| G (Healthy individuals, n= 22) | 2007–2018 | 0/22 |
| H (Individuals negative for dengue and chikungunya; n = 52) | 1998–2010 | 7/52 (13.5) |
| I (Yellow fever positive cases, n = 09) | 1997–1998 | 0/09 |
| J (Individuals vaccinated for yellow fever, n = 07) | 1999–2018 | 0/07 |
| K (Measles cases, n = 12) | 2004–2005 | 0/12 |
| L (Rubella cases, n = 12) | 2005 | 0/12 |
| M (Zika cases, n = 16) | 2015 | 0/16 |
| N (Leptospirosis cases, n= 17) | 2009 | 0/17 |
| O (Hepatitis C cases, n = 10) | 2009 | 0/10 |
| TOTAL OF GROUP I–O (n = 83) | 0/83 | |
| TOTAL | 1997–2018 | 93/340 (27.3) |
| Performance * of the Anti-Chikungunya Virus IgM ELISA (Euroimmun) | % |
|---|---|
| Sensitivity | 100 |
| Specificity | 25.3 |
| Efficiency | 43.5 |
| Positive Predictive Value | 30.1 |
| Negative Predictive Value | 100 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lima, M.d.R.Q.; de Lima, R.C.; de Azeredo, E.L.; dos Santos, F.B. Analysis of a Routinely Used Commercial Anti-Chikungunya IgM ELISA Reveals Cross-Reactivities with Dengue in Brazil: A New Challenge for Differential Diagnosis? Diagnostics 2021, 11, 819. https://doi.org/10.3390/diagnostics11050819
Lima MdRQ, de Lima RC, de Azeredo EL, dos Santos FB. Analysis of a Routinely Used Commercial Anti-Chikungunya IgM ELISA Reveals Cross-Reactivities with Dengue in Brazil: A New Challenge for Differential Diagnosis? Diagnostics. 2021; 11(5):819. https://doi.org/10.3390/diagnostics11050819
Chicago/Turabian StyleLima, Monique da Rocha Queiroz, Raquel Curtinhas de Lima, Elzinandes Leal de Azeredo, and Flavia Barreto dos Santos. 2021. "Analysis of a Routinely Used Commercial Anti-Chikungunya IgM ELISA Reveals Cross-Reactivities with Dengue in Brazil: A New Challenge for Differential Diagnosis?" Diagnostics 11, no. 5: 819. https://doi.org/10.3390/diagnostics11050819
APA StyleLima, M. d. R. Q., de Lima, R. C., de Azeredo, E. L., & dos Santos, F. B. (2021). Analysis of a Routinely Used Commercial Anti-Chikungunya IgM ELISA Reveals Cross-Reactivities with Dengue in Brazil: A New Challenge for Differential Diagnosis? Diagnostics, 11(5), 819. https://doi.org/10.3390/diagnostics11050819







