Concordance between Response Assessment Using Prostate-Specific Membrane Antigen PET and Serum Prostate-Specific Antigen Levels after Systemic Treatment in Patients with Metastatic Castration Resistant Prostate Cancer: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Literature Search
2.2. Inclusion Criteria
2.3. Exclusion Criteria
2.4. Data Extraction
2.5. Quality Assessment
2.6. Data Synthesis and Analysis
3. Results
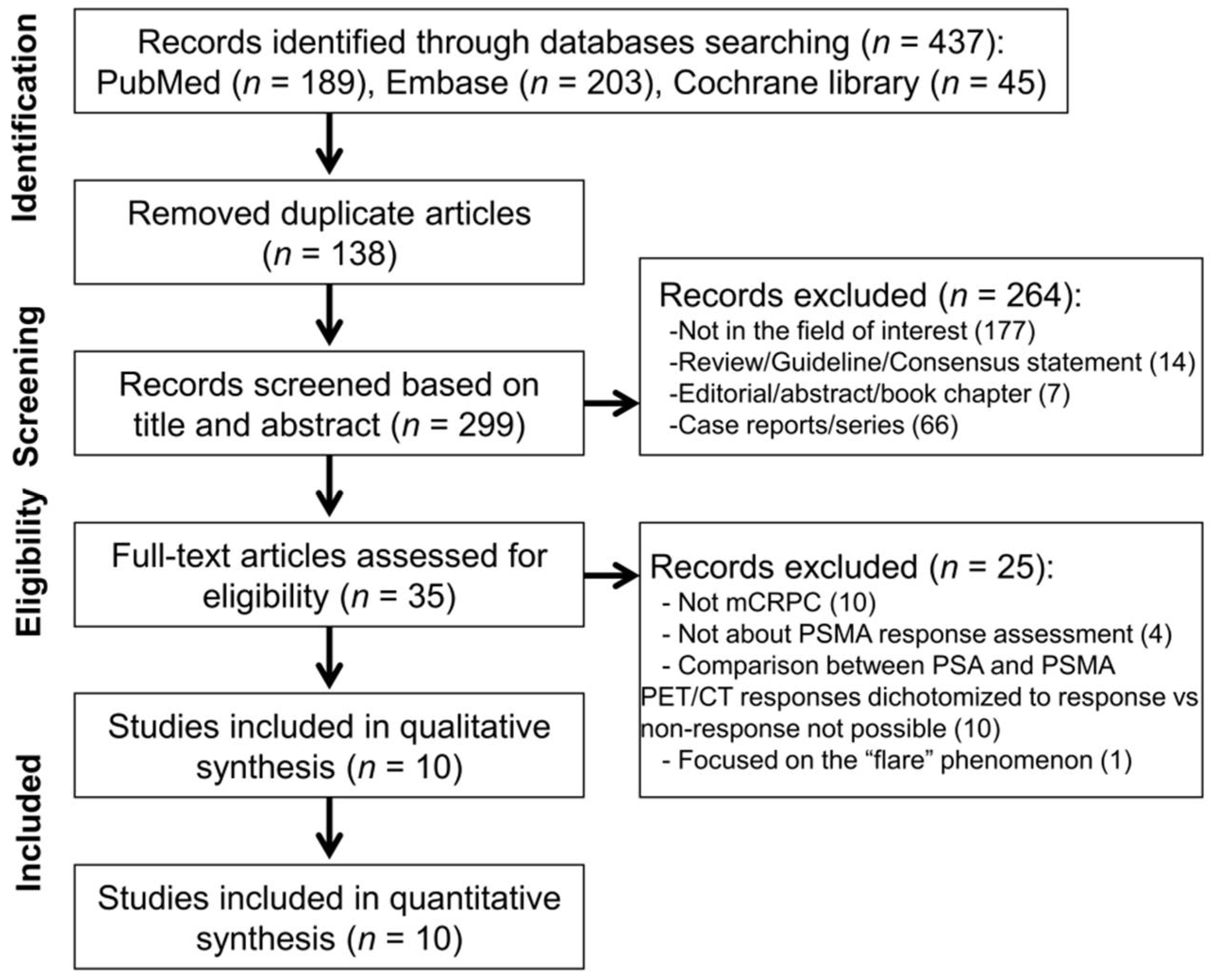
3.1. Literature Search
3.2. Characteristics of Included Studies
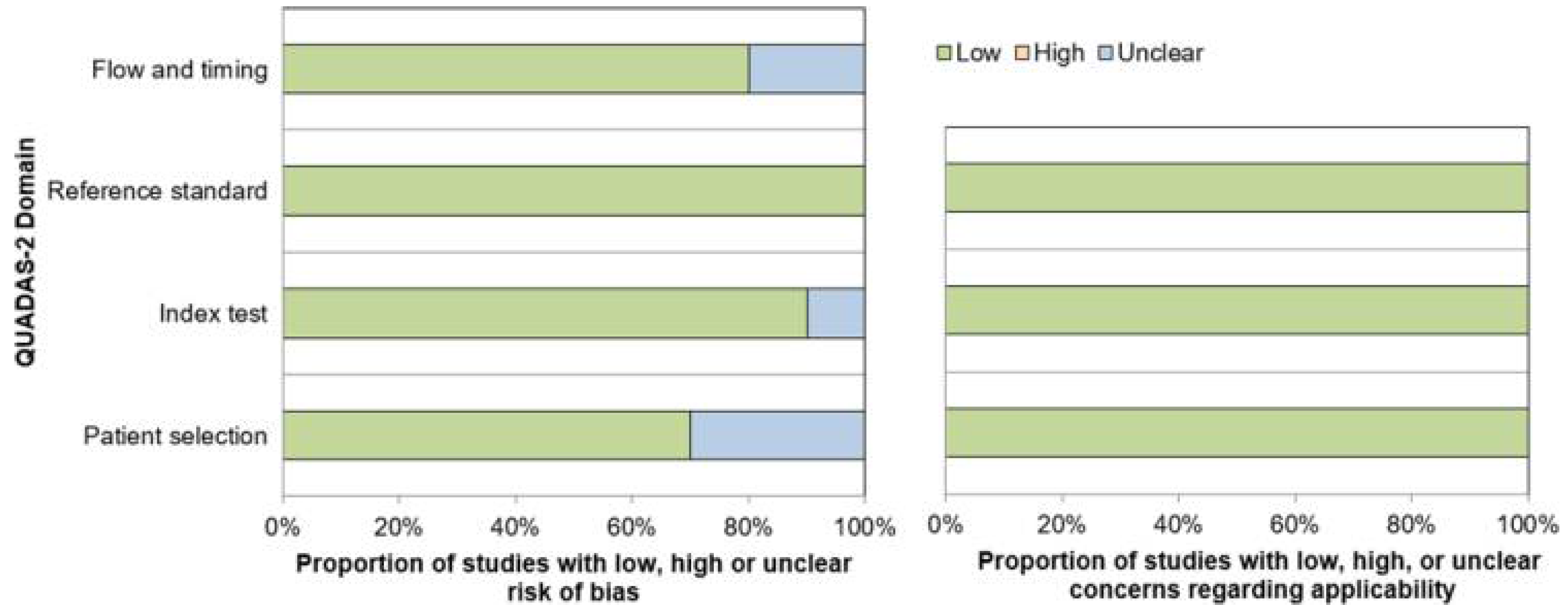
3.3. Quality Assessment
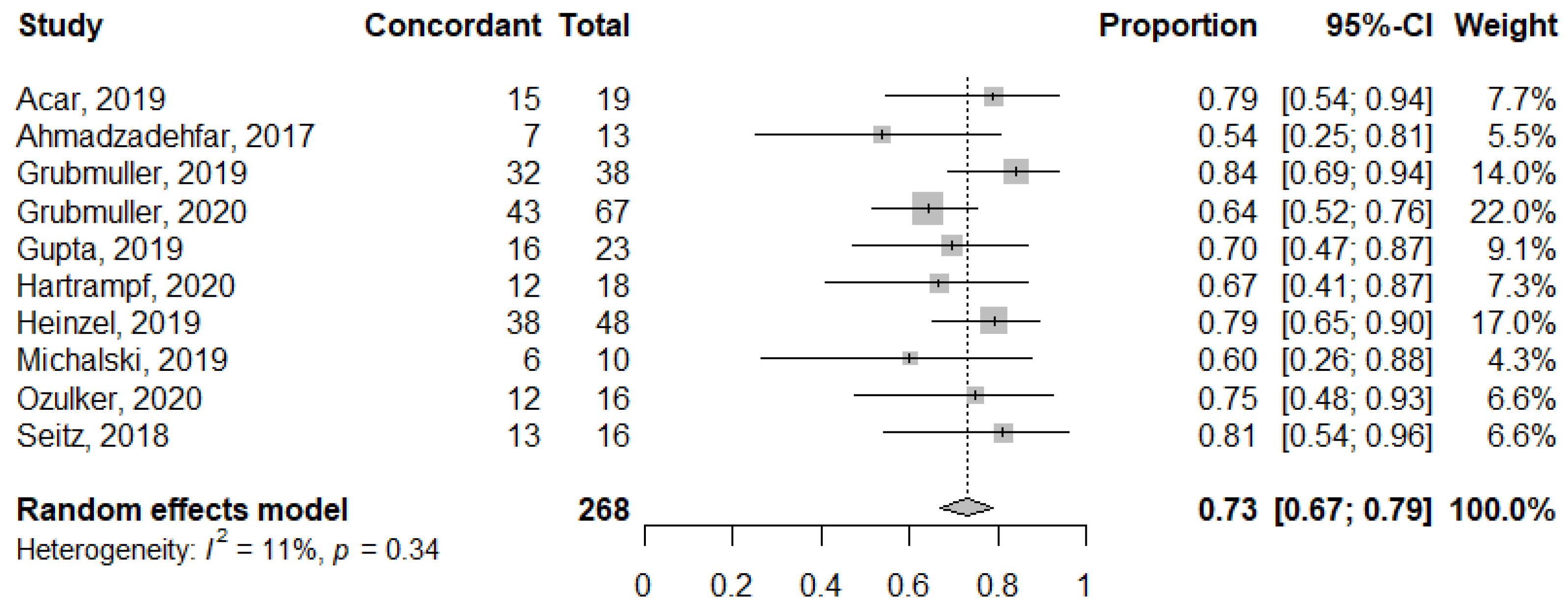
3.4. Concordance Rate between PSA and PSMA Response
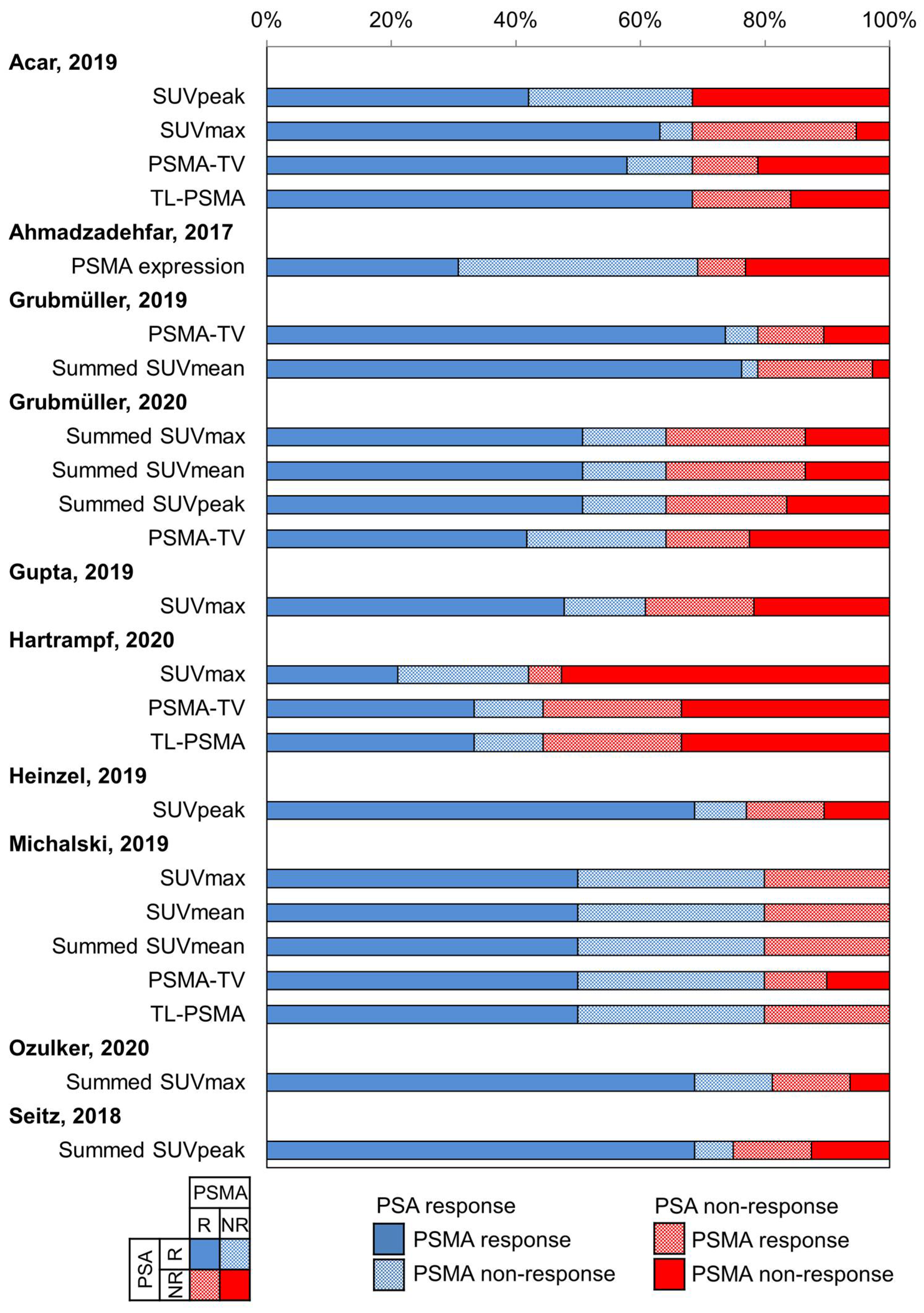
3.5. Directionality of Discordant Responses between PSA and PSMA
3.6. Correlation between PSMA Response and Clinical Outcomes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Ryan, C.J.; Smith, M.R.; de Bono, J.S.; Molina, A.; Logothetis, C.J.; de Souza, P.; Fizazi, K.; Mainwaring, P.; Piulats, J.M.; Ng, S.; et al. Abiraterone in metastatic prostate cancer without previous chemotherapy. N. Engl. J. Med. 2013, 368, 138–148. [Google Scholar] [CrossRef] [PubMed]
- Scher, H.I.; Fizazi, K.; Saad, F.; Taplin, M.E.; Sternberg, C.N.; Miller, K.; de Wit, R.; Mulders, P.; Chi, K.N.; Shore, N.D.; et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N. Engl. J. Med. 2012, 367, 1187–1197. [Google Scholar] [CrossRef] [PubMed]
- Parker, C.; Nilsson, S.; Heinrich, D.; Helle, S.I.; O’Sullivan, J.M.; Fosså, S.D.; Chodacki, A.; Wiechno, P.; Logue, J.; Seke, M.; et al. Alpha emitter radium-223 and survival in metastatic prostate cancer. N. Engl. J. Med. 2013, 369, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Scher, H.I.; Halabi, S.; Tannock, I.; Morris, M.; Sternberg, C.N.; Carducci, M.A.; Eisenberger, M.A.; Higano, C.; Bubley, G.J.; Dreicer, R.; et al. Design and end points of clinical trials for patients with progressive prostate cancer and castrate levels of testosterone: Recommendations of the Prostate Cancer Clinical Trials Working Group. J. Clin. Oncol. 2008, 26, 1148–1159. [Google Scholar] [CrossRef]
- Scher, H.I.; Morris, M.J.; Stadler, W.M.; Higano, C.; Basch, E.; Fizazi, K.; Antonarakis, E.S.; Beer, T.M.; Carducci, M.A.; Chi, K.N.; et al. Trial Design and Objectives for Castration-Resistant Prostate Cancer: Updated Recommendations from the Prostate Cancer Clinical Trials Working Group 3. J. Clin. Oncol. 2016, 34, 1402–1418. [Google Scholar] [CrossRef] [PubMed]
- Fanti, S.; Hadaschik, B.; Herrmann, K. Proposal for Systemic-Therapy Response-Assessment Criteria at the Time of PSMA PET/CT Imaging: The PSMA PET Progression Criteria. J. Nucl. Med. 2020, 61, 678–682. [Google Scholar] [CrossRef]
- Fanti, S.; Goffin, K.; Hadaschik, B.A.; Herrmann, K.; Maurer, T.; MacLennan, S.; Oprea-Lager, D.E.; Oyen, W.J.; Rouvière, O.; Mottet, N.; et al. Consensus statements on PSMA PET/CT response assessment criteria in prostate cancer. Eur. J. Nucl. Med. Mol. Imaging 2020. [Google Scholar] [CrossRef] [PubMed]
- Scher, H.I.; Jia, X.; de Bono, J.S.; Fleisher, M.; Pienta, K.J.; Raghavan, D.; Heller, G. Circulating tumour cells as prognostic markers in progressive, castration-resistant prostate cancer: A reanalysis of IMMC38 trial data. Lancet Oncol. 2009, 10, 233–239. [Google Scholar] [CrossRef]
- Calais, J.; Ceci, F.; Eiber, M.; Hope, T.A.; Hofman, M.S.; Rischpler, C.; Bach-Gansmo, T.; Nanni, C.; Savir-Baruch, B.; Elashoff, D.; et al. (18)F-fluciclovine PET-CT and (68)Ga-PSMA-11 PET-CT in patients with early biochemical recurrence after prostatectomy: A prospective, single-centre, single-arm, comparative imaging trial. Lancet Oncol. 2019, 20, 1286–1294. [Google Scholar] [CrossRef]
- Hofman, M.S.; Lawrentschuk, N.; Francis, R.J.; Tang, C.; Vela, I.; Thomas, P.; Rutherford, N.; Martin, J.M.; Frydenberg, M.; Shakher, R.; et al. Prostate-specific membrane antigen PET-CT in patients with high-risk prostate cancer before curative-intent surgery or radiotherapy (proPSMA): A prospective, randomised, multicentre study. Lancet 2020, 395, 1208–1216. [Google Scholar] [CrossRef]
- Han, S.; Woo, S.; Kim, Y.J.; Suh, C.H. Impact of (68)Ga-PSMA PET on the Management of Patients with Prostate Cancer: A Systematic Review and Meta-analysis. Eur. Urol. 2018, 74, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Fendler, W.P.; Weber, M.; Iravani, A.; Hofman, M.S.; Calais, J.; Czernin, J.; Ilhan, H.; Saad, F.; Small, E.J.; Smith, M.R.; et al. Prostate-Specific Membrane Antigen Ligand Positron Emission Tomography in Men with Nonmetastatic Castration-Resistant Prostate Cancer. Clin. Cancer Res. 2019, 25, 7448–7454. [Google Scholar] [CrossRef] [PubMed]
- Phillips, R.; Shi, W.Y.; Deek, M.; Radwan, N.; Lim, S.J.; Antonarakis, E.S.; Rowe, S.P.; Ross, A.E.; Gorin, M.A.; Deville, C.; et al. Outcomes of Observation vs. Stereotactic Ablative Radiation for Oligometastatic Prostate Cancer: The ORIOLE Phase 2 Randomized Clinical Trial. JAMA Oncol. 2020, 6, 650–659. [Google Scholar] [CrossRef]
- Acar, E.; Özdoğan, Ö.; Aksu, A.; Derebek, E.; Bekiş, R.; Çapa Kaya, G. The use of molecular volumetric parameters for the evaluation of Lu-177 PSMA I&T therapy response and survival. Ann. Nucl. Med. 2019, 33, 681–688. [Google Scholar] [CrossRef] [PubMed]
- Ahmadzadehfar, H.; Azgomi, K.; Hauser, S.; Wei, X.; Yordanova, A.; Gaertner, F.C.; Kürpig, S.; Strunk, H.; Essler, M. (68)Ga-PSMA-11 PET as a Gatekeeper for the Treatment of Metastatic Prostate Cancer with (223)Ra: Proof of Concept. J. Nucl. Med. 2017, 58, 438–444. [Google Scholar] [CrossRef]
- Grubmüller, B.; Senn, D.; Kramer, G.; Baltzer, P.; D’Andrea, D.; Grubmüller, K.H.; Mitterhauser, M.; Eidherr, H.; Haug, A.R.; Wadsak, W.; et al. Response assessment using (68)Ga-PSMA ligand PET in patients undergoing (177)Lu-PSMA radioligand therapy for metastatic castration-resistant prostate cancer. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1063–1072. [Google Scholar] [CrossRef]
- Grubmüller, B.; Rasul, S.; Baltzer, P.; Fajkovic, H.; D’Andrea, D.; Berndl, F.; Maj-Hes, A.; Grubmüller, K.H.; Mitterhauser, M.; Wadsak, W.; et al. Response assessment using [(68) Ga]Ga-PSMA ligand PET in patients undergoing systemic therapy for metastatic castration-resistant prostate cancer. Prostate 2020, 80, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.; Choudhury, P.S.; Rawal, S.; Goel, H.C.; Rao, S.A. Evaluation of response in patients of metastatic castration resistant prostate cancer undergoing systemic radiotherapy with lutetium177-prostate-specific membrane antigen: A comparison between response evaluation criteria in solid tumors, positron-emission tomography response criteria in solid tumors, European organization for research and treatment of cancer, and MDA criteria assessed by gallium 68-prostate-specific membrane antigen positron-emission tomography-computed tomography. Urol. Ann. 2019, 11, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Hartrampf, P.E.; Heinrich, M.; Seitz, A.K.; Brumberg, J.; Sokolakis, I.; Kalogirou, C.; Schirbel, A.; Kübler, H.; Buck, A.K.; Lapa, C.; et al. Metabolic Tumour Volume from PSMA PET/CT Scans of Prostate Cancer Patients during Chemotherapy-Do Different Software Solutions Deliver Comparable Results? J. Clin. Med. 2020, 9, 1390. [Google Scholar] [CrossRef] [PubMed]
- Heinzel, A.; Boghos, D.; Mottaghy, F.M.; Gaertner, F.; Essler, M.; von Mallek, D.; Ahmadzadehfar, H. (68)Ga-PSMA PET/CT for monitoring response to (177)Lu-PSMA-617 radioligand therapy in patients with metastatic castration-resistant prostate cancer. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1054–1062. [Google Scholar] [CrossRef]
- Michalski, K.; Mix, M.; Meyer, P.T.; Ruf, J. Determination of whole-body tumour burden on [68Ga]PSMA-11 PET/CT for response assessment of [177Lu]PSMA-617 radioligand therapy: A retrospective analysis of serum PSA level and imaging derived parameters before and after two cycles of therapy. Nuklearmedizin 2019, 58, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Özülker, T.; Özülker, F. Assessment of the role of Ga-68 PSMA I&T PET/CT in response evaluation to docetaxel therapy in castration resistant prostate cancer patients. Rev. Esp. Med. Nucl. Imagen Mol. 2020, 39, 292–298. [Google Scholar] [CrossRef]
- Seitz, A.K.; Rauscher, I.; Haller, B.; Krönke, M.; Luther, S.; Heck, M.M.; Horn, T.; Gschwend, J.E.; Schwaiger, M.; Eiber, M.; et al. Preliminary results on response assessment using (68)Ga-HBED-CC-PSMA PET/CT in patients with metastatic prostate cancer undergoing docetaxel chemotherapy. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 602–612. [Google Scholar] [CrossRef] [PubMed]
- McInnes, M.D.F.; Moher, D.; Thombs, B.D.; McGrath, T.A.; Bossuyt, P.M.; Clifford, T.; Cohen, J.F.; Deeks, J.J.; Gatsonis, C.; Hooft, L.; et al. Preferred Reporting Items for a Systematic Review and Meta-analysis of Diagnostic Test Accuracy Studies: The PRISMA-DTA Statement. JAMA 2018, 319, 388–396. [Google Scholar] [CrossRef]
- Emmett, L.; Yin, C.; Crumbaker, M.; Hruby, G.; Kneebone, A.; Epstein, R.; Nguyen, Q.; Hickey, A.; Ihsheish, N.; O’Neill, G.; et al. Rapid Modulation of PSMA Expression by Androgen Deprivation: Serial (68)Ga-PSMA-11 PET in Men with Hormone-Sensitive and Castrate-Resistant Prostate Cancer Commencing Androgen Blockade. J. Nucl. Med. 2019, 60, 950–954. [Google Scholar] [CrossRef] [PubMed]
- Whiting, P.F.; Rutjes, A.W.; Westwood, M.E.; Mallett, S.; Deeks, J.J.; Reitsma, J.B.; Leeflang, M.M.; Sterne, J.A.; Bossuyt, P.M. QUADAS-2: A revised tool for the quality assessment of diagnostic accuracy studies. Ann. Intern. Med. 2011, 155, 529–536. [Google Scholar] [CrossRef]
- Barendregt, J.J.; Doi, S.A.; Lee, Y.Y.; Norman, R.E.; Vos, T. Meta-analysis of prevalence. J. Epidemiol. Community Health 2013, 67, 974–978. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef]
- Brito, A.E.T.; Mourato, F.A.; de Oliveira, R.P.M.; Leal, A.L.G.; Filho, P.J.A.; de Filho, J.L.L. Evaluation of whole-body tumor burden with (68)Ga-PSMA PET/CT in the biochemical recurrence of prostate cancer. Ann. Nucl. Med. 2019, 33, 344–350. [Google Scholar] [CrossRef]
- Vargas, H.A.; Wassberg, C.; Fox, J.J.; Wibmer, A.; Goldman, D.A.; Kuk, D.; Gonen, M.; Larson, S.M.; Morris, M.J.; Scher, H.I.; et al. Bone metastases in castration-resistant prostate cancer: Associations between morphologic CT patterns, glycolytic activity, and androgen receptor expression on PET and overall survival. Radiology 2014, 271, 220–229. [Google Scholar] [CrossRef]
- Han, S.; Kim, H.; Kim, Y.J.; Suh, C.H.; Woo, S. Prognostic value of volume-based metabolic parameters of (18)F-FDG PET/CT in ovarian cancer: A systematic review and meta-analysis. Ann. Nucl. Med. 2018, 32, 669–677. [Google Scholar] [CrossRef]
- Im, H.J.; Pak, K.; Cheon, G.J.; Kang, K.W.; Kim, S.J.; Kim, I.J.; Chung, J.K.; Kim, E.E.; Lee, D.S. Prognostic value of volumetric parameters of (18)F-FDG PET in non-small-cell lung cancer: A meta-analysis. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Hillier, S.M.; Kern, A.M.; Maresca, K.P.; Marquis, J.C.; Eckelman, W.C.; Joyal, J.L.; Babich, J.W. 123I-MIP-1072, a small-molecule inhibitor of prostate-specific membrane antigen, is effective at monitoring tumor response to taxane therapy. J. Nucl. Med. 2011, 52, 1087–1093. [Google Scholar] [CrossRef] [PubMed]
- Ben Jemaa, A.; Bouraoui, Y.; Oueslati, R. Insight into the heterogeneity of prostate cancer through PSA-PSMA prostate clones: Mechanisms and consequences. Histol. Histopathol. 2014, 29, 1263–1280. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, F.G.; Queiroz, M.A.; Ferraro, D.A.; Nunes, R.F.; Dreyer, P.R.; Zaniboni, E.C.; Costa, L.B.; Bastos, D.A.; Marin, J.F.G.; Buchpiguel, C.A. Prostate-specific Membrane Antigen PET: Therapy Response Assessment in Metastatic Prostate Cancer. Radiographics 2020, 40, 1412–1430. [Google Scholar] [CrossRef] [PubMed]
- Parimi, V.; Goyal, R.; Poropatich, K.; Yang, X.J. Neuroendocrine differentiation of prostate cancer: A review. Am. J. Clin. Exp. Urol. 2014, 2, 273–285. [Google Scholar] [PubMed]
- Costa, L.B.; Queiroz, M.A.; Barbosa, F.G.; Nunes, R.F.; Marin, J.F.G.; Dzik, C.; Buchpiguel, C.A. Pseudoprogression on PSMA PET imaging of a mCRPC patient under anti-PD1 treatment. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1576–1577. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, R.; Wei, X.; Kim, W.; Small, E.J.; Ryan, C.J.; Carroll, P.; Cooperberg, M.; Evans, M.J.; Hope, T. Heterogeneous Flare in Prostate-specific Membrane Antigen Positron Emission Tomography Tracer Uptake with Initiation of Androgen Pathway Blockade in Metastatic Prostate Cancer. Eur. Urol. Oncol. 2018, 1, 78–82. [Google Scholar] [CrossRef]
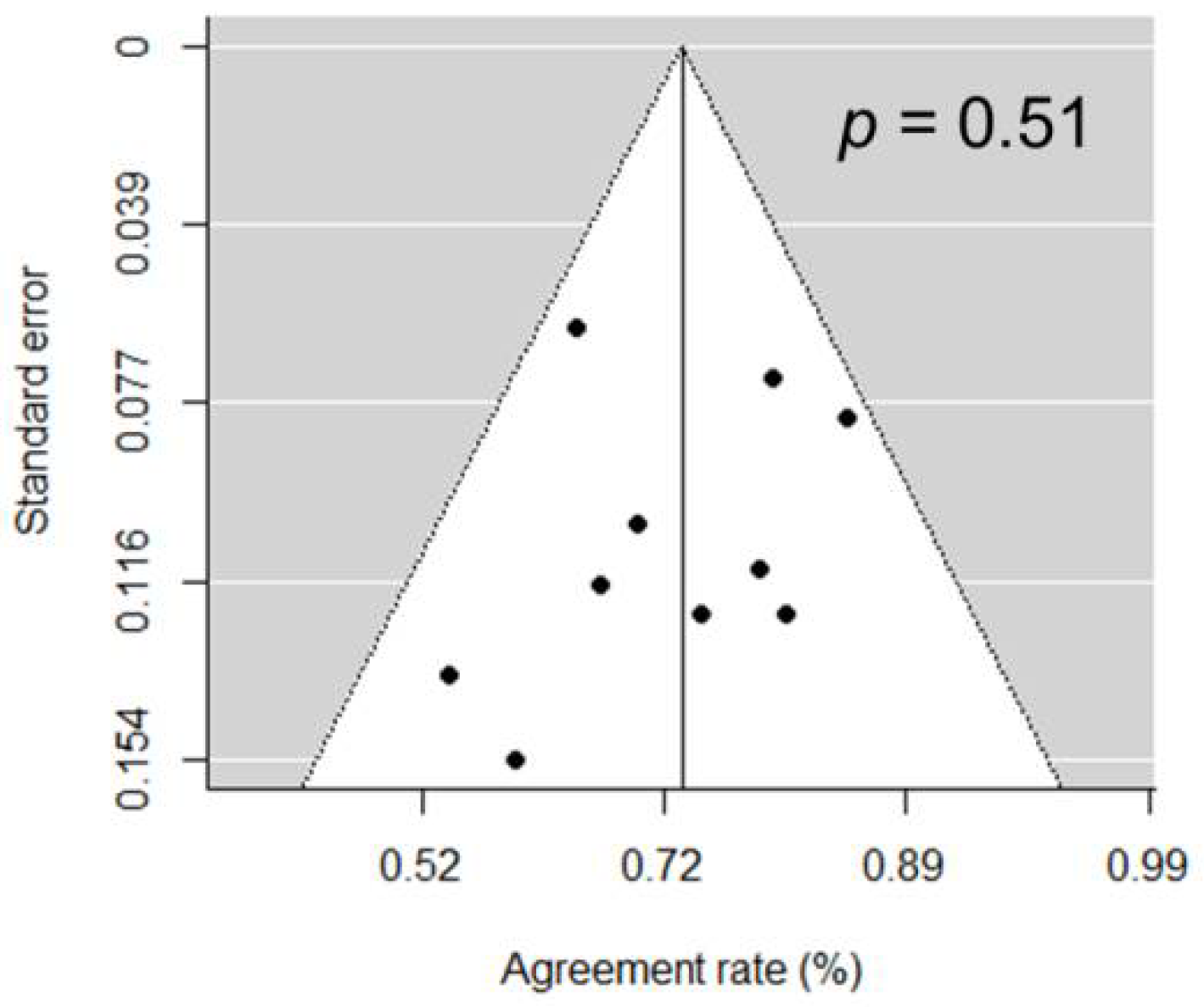
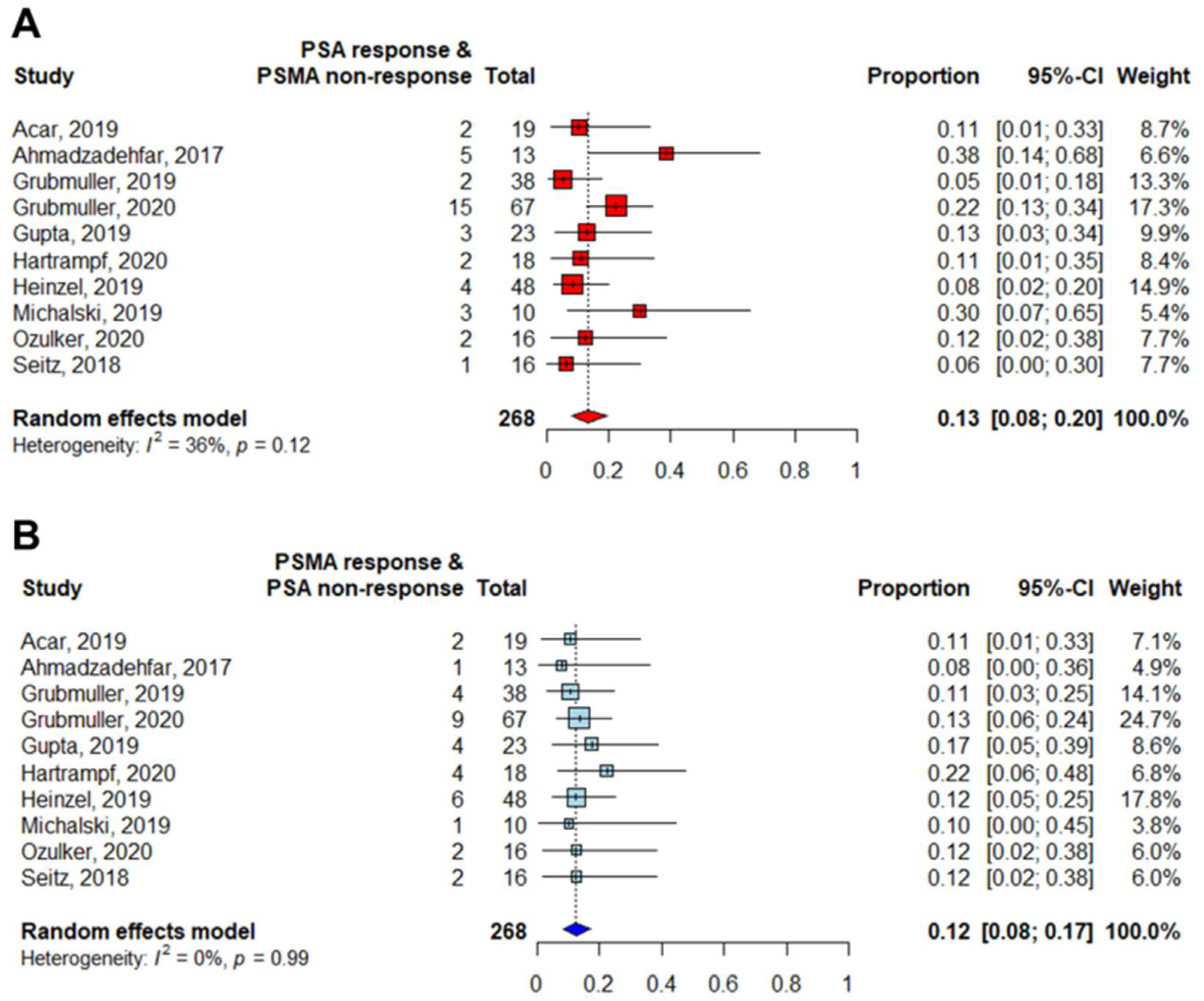
| Author | Journal | Year | Institution | Country | Period | Design | Treatment | n | Pretreatment PSA (Median, ng/mL) | Initial Gleason Score (Median, Range) |
|---|---|---|---|---|---|---|---|---|---|---|
| Acar [15] | Ann Nucl Med | 2019 | Ataturk Training and Research Hospital | Turkey | 2015–2018 | R | 177Lu-PSMA I&T | 19 | NR | 9 (6–10) |
| Ahmadzadehfar [16] | J Nucl Med | 2017 | University Hospital Bonn | Germany | 2014–2016 | R | 223Ra | 13 | NR | 8 (6–10) |
| Grubmüller [17] | Eur J Nucl Med Mol Imaging | 2019 | Medical University of Vienna | Austria | 2015–2018 | R | 177Lu-PSMA 617 | 38 | 60.8 (IQR 15.4–264.2) | NR |
| Grubmüller [18] | The prostate | 2020 | Medical University of Vienna | Austria | 2015–2019 | R | 223Ra/cabazitaxel/docetaxel abiraterone/enzalutamide | 43 * | 11.3 (IQR 3.3–30.1) | NR |
| Gupta [19] | Urol Ann | 2019 | Rajiv Gandhi Cancer Insttute and Research Centre | India | NR | R | 177Lu-PSMA 617 | 23 | 66 (5.4–550) | 7 (6–9) |
| Hartrampf [20] | J Clin Med | 2020 | University Hospital Würzburg | Germany | 2014–2018 | R | docetaxel/cabazitaxel | 21 | 15.0 (0–800) | 8 (6–10) |
| Heinzel [21] | Eur J Nucl Med Mol Imaging | 2019 | university hospitals of Bonn and Aachen | Germany | 2014–2018 | R | 177Lu-PSMA 617 | 48 | NR | NR |
| Michalski [22] | Nuklearmedizin | 2019 | University of Freiburg | Germany | 2015–2018 | R | 177Lu PSMA-617 | 10 | 812.1 ± 972.4 | 8 (7–9) |
| Ozulker [23] | Rev Esp Med Nucl Imagen Mol | 2020 | Okmeydanı Training and Research Hospital | Turkey | 2017–2018 | R | docetaxel | 16 | 61.37 ± 102.2 | 8.50 ± 0.97 |
| Seitz [24] | Eur J Nucl Med Mol Imaging | 2018 | Technical University of Munich | Germany | 2013–2016 | R | docetaxel | 16 | 28 (2–3176) | ≤6: 1 7: 6 ≥8: 7 |
| Author | Tracer | Injected Dose (MBq) | Uptake Time (min) | Machine | PET Timing | PET Response Criteria |
|---|---|---|---|---|---|---|
| Acar [15] | 68Ga-PSMA-I&T | 115 * | 60 | PET/CT | after last cycle | SUVpeak SUVmax PSMA-TV TL-PSMA |
| Ahmadzadehfar [16] | 68Ga-PSMA-11 | 2/kg | 77 † | PET/CT | after last cycle | PSMA expression (not explicit) |
| Grubmüller [17] | 68Ga-PSMA-11 | 2/kg | 60 | PET/CT or PET/MRI | after 3 cycle | PSMA-TV Summed SUVmean |
| Grubmüller [18] | 68Ga-PSMA-11 | 2/kg | 60 | PET/CT or MRI | 223Ra or systemic Tx.: after 6 cycle abiraterone or enzalutamide: at 3 mo. | Summed SUVmax Summed SUVmean Summed SUVpeak PSMA-TV |
| Gupta [19] | 68Ga-PSMA-11 | 2/kg | 60 | PET/CT | after radionuclide therapy | SUVmax |
| Hartrampf [20] | 68Ga-PSMA-I&T | 119–125 * | 71–82 † | PET/CT | after completion or termination of chemotherapy | SUVmax PSMA-TV TL-PSMA |
| Heinzel [21] | 68Ga-PSMA-11 | 2/kg | 60 | PET/CT | after 3 or 4 cycle | SUVpeak |
| Michalski [22] | 68Ga-PSMA-11 | 206 * | 60 | PET/CT | after 2 cycle | SUVmax SUVmean Summed SUVmean PSMA-TV TL-PSMA |
| Ozulker [23] | 68Ga-PSMA-I&T | 2/kg | 60 | PET/CT | after ≥3 cycles | Summed SUVmax ‡ |
| Seitz [24] | 68Ga-PSMA-11 | 154 * | 57 * | PET/CT | after 3 cycle | Summed SUVpeak |
| Variables | Subgroups | Studies (n) | Pooled Proportion | 95% CI | I2 | p * |
|---|---|---|---|---|---|---|
| Therapeutic agents | 177Lu-PSMA | 5 | 0.78 | 0.71–0.85 | 0% | 0.69 |
| Chemotherapy | 3 | 0.74 | 0.61–0.86 | 0% | ||
| PET criteria | PSMA-TV | 5 | 0.72 | 0.62–0.82 | 36% | 0.33 |
| SUVmax | 4 | 0.68 | 0.56–0.79 | 0% | ||
| Summed SUVmean | 3 | 0.68 | 0.54–0.81 | 50% | ||
| TL-PSMA | 3 | 0.70 | 0.50–0.87 | 45% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, S.; Woo, S.; Kim, Y.-i.; Lee, J.-L.; Wibmer, A.G.; Schoder, H.; Ryu, J.-S.; Vargas, H.A. Concordance between Response Assessment Using Prostate-Specific Membrane Antigen PET and Serum Prostate-Specific Antigen Levels after Systemic Treatment in Patients with Metastatic Castration Resistant Prostate Cancer: A Systematic Review and Meta-Analysis. Diagnostics 2021, 11, 663. https://doi.org/10.3390/diagnostics11040663
Han S, Woo S, Kim Y-i, Lee J-L, Wibmer AG, Schoder H, Ryu J-S, Vargas HA. Concordance between Response Assessment Using Prostate-Specific Membrane Antigen PET and Serum Prostate-Specific Antigen Levels after Systemic Treatment in Patients with Metastatic Castration Resistant Prostate Cancer: A Systematic Review and Meta-Analysis. Diagnostics. 2021; 11(4):663. https://doi.org/10.3390/diagnostics11040663
Chicago/Turabian StyleHan, Sangwon, Sungmin Woo, Yong-il Kim, Jae-Lyun Lee, Andreas G. Wibmer, Heiko Schoder, Jin-Sook Ryu, and Hebert Alberto Vargas. 2021. "Concordance between Response Assessment Using Prostate-Specific Membrane Antigen PET and Serum Prostate-Specific Antigen Levels after Systemic Treatment in Patients with Metastatic Castration Resistant Prostate Cancer: A Systematic Review and Meta-Analysis" Diagnostics 11, no. 4: 663. https://doi.org/10.3390/diagnostics11040663
APA StyleHan, S., Woo, S., Kim, Y.-i., Lee, J.-L., Wibmer, A. G., Schoder, H., Ryu, J.-S., & Vargas, H. A. (2021). Concordance between Response Assessment Using Prostate-Specific Membrane Antigen PET and Serum Prostate-Specific Antigen Levels after Systemic Treatment in Patients with Metastatic Castration Resistant Prostate Cancer: A Systematic Review and Meta-Analysis. Diagnostics, 11(4), 663. https://doi.org/10.3390/diagnostics11040663






