COVID-19 Impact on Diagnostic Innovations: Emerging Trends and Implications
Abstract
1. Introduction
2. Materials and Methods
2.1. Identification
2.2. Inclusion and Date Extraction
2.3. Monitor
3. Results
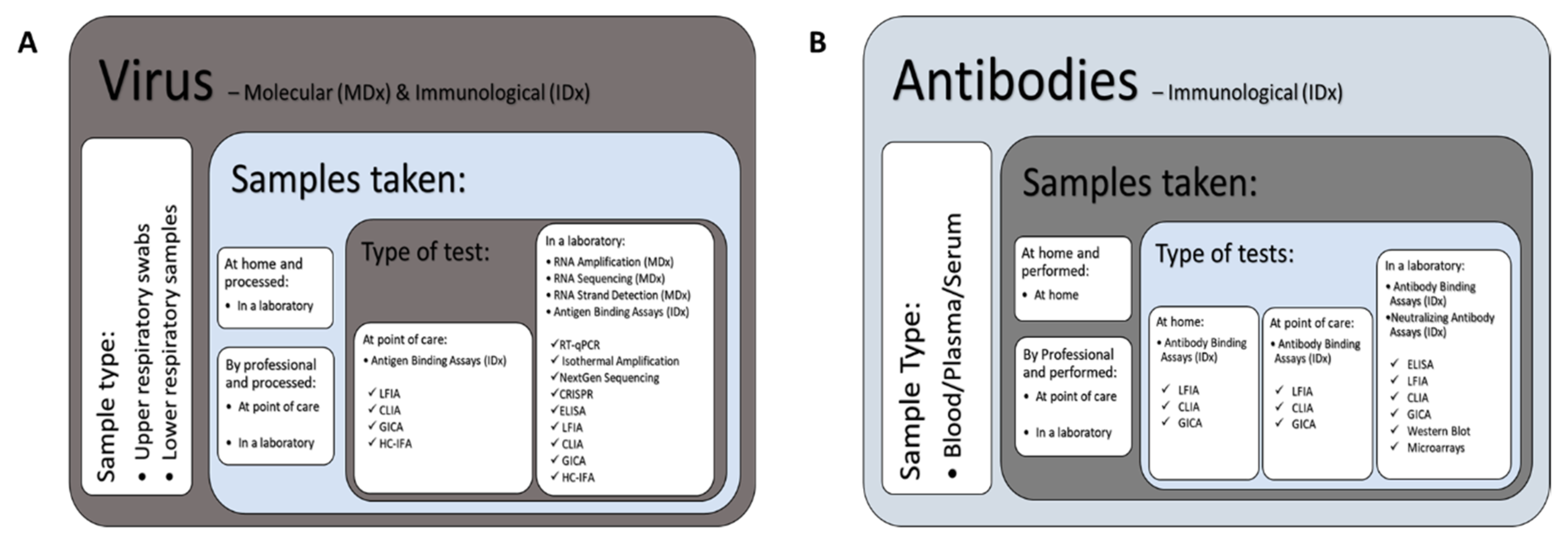
3.1. Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Diagnostic Technology Landscape
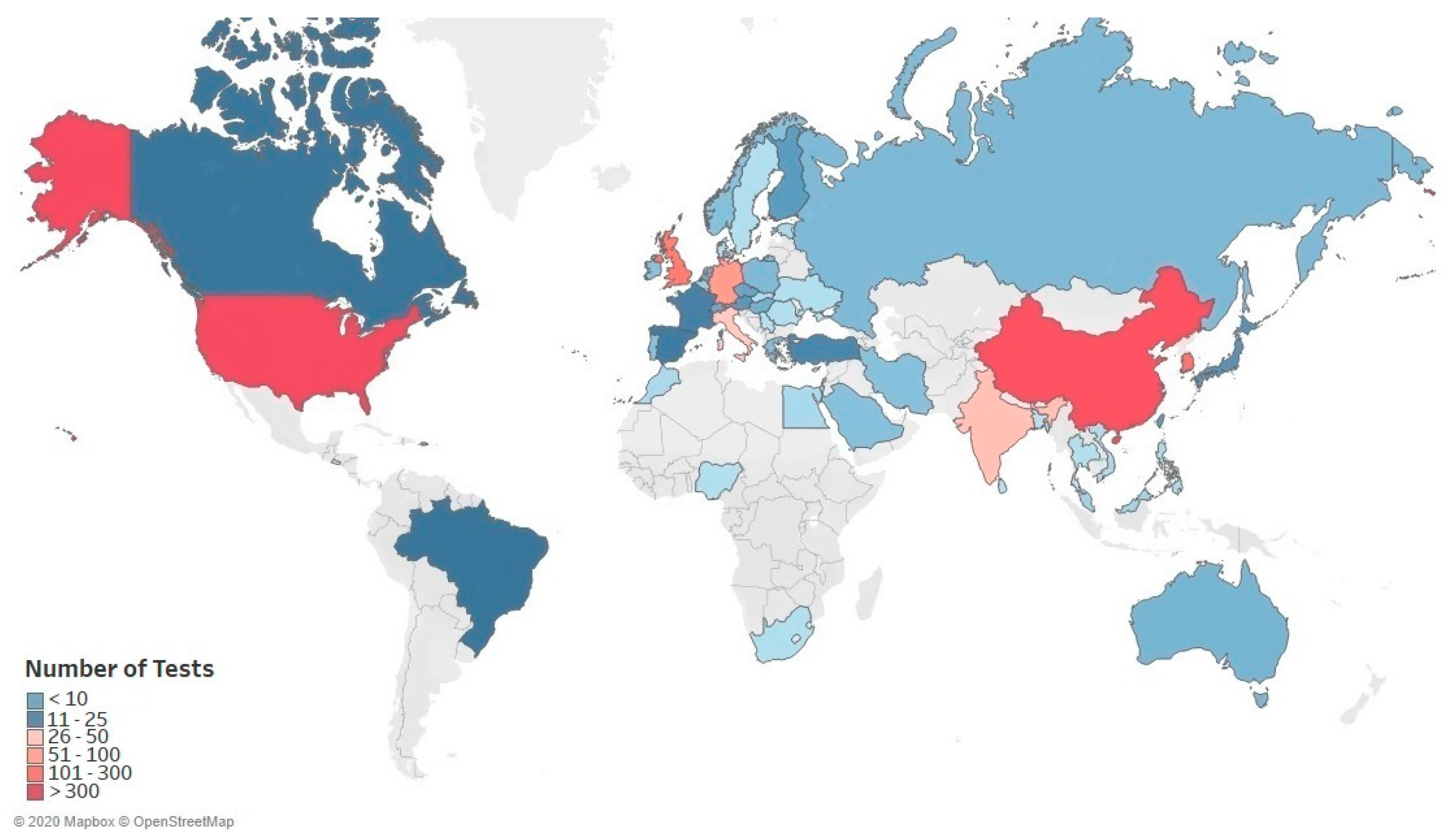
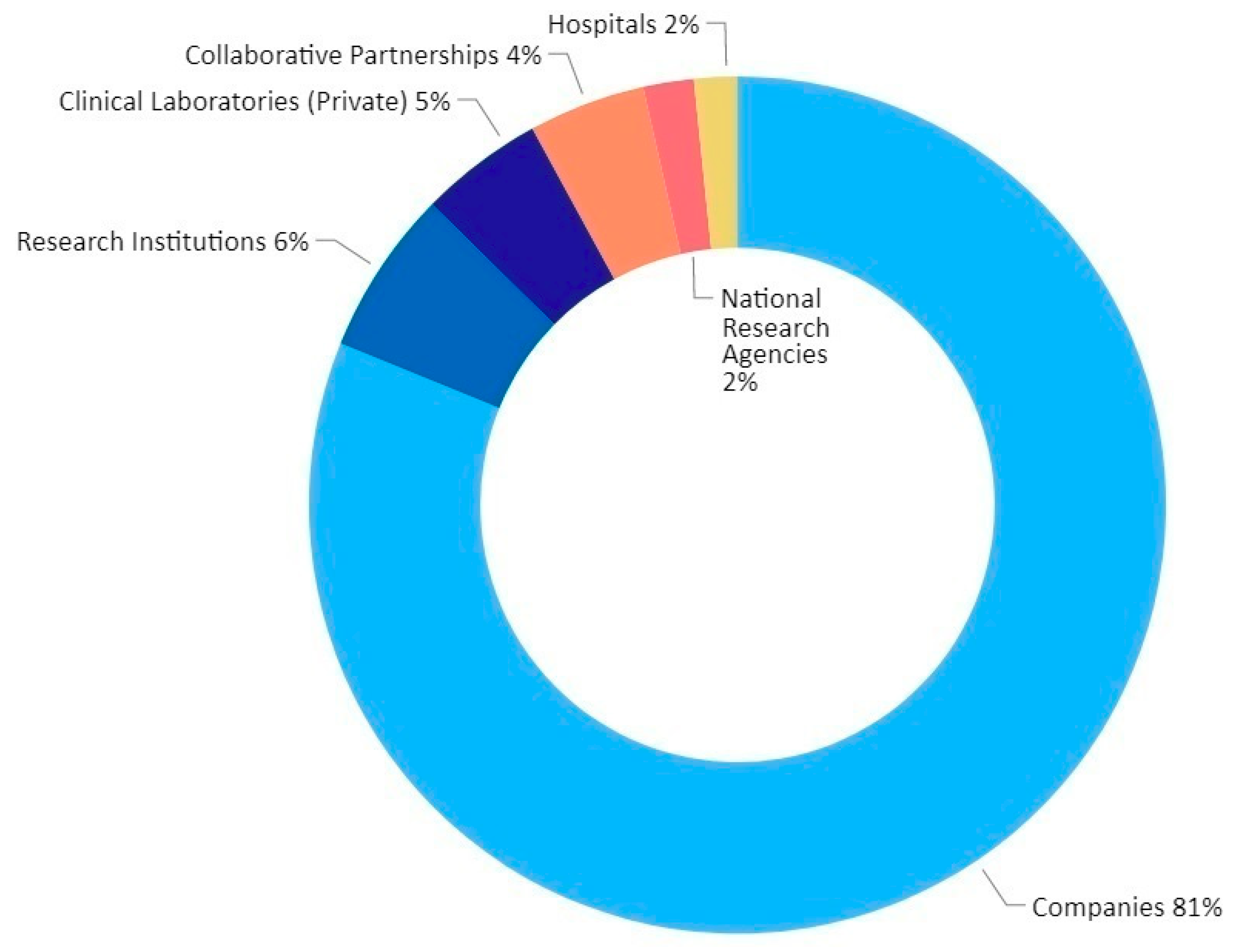
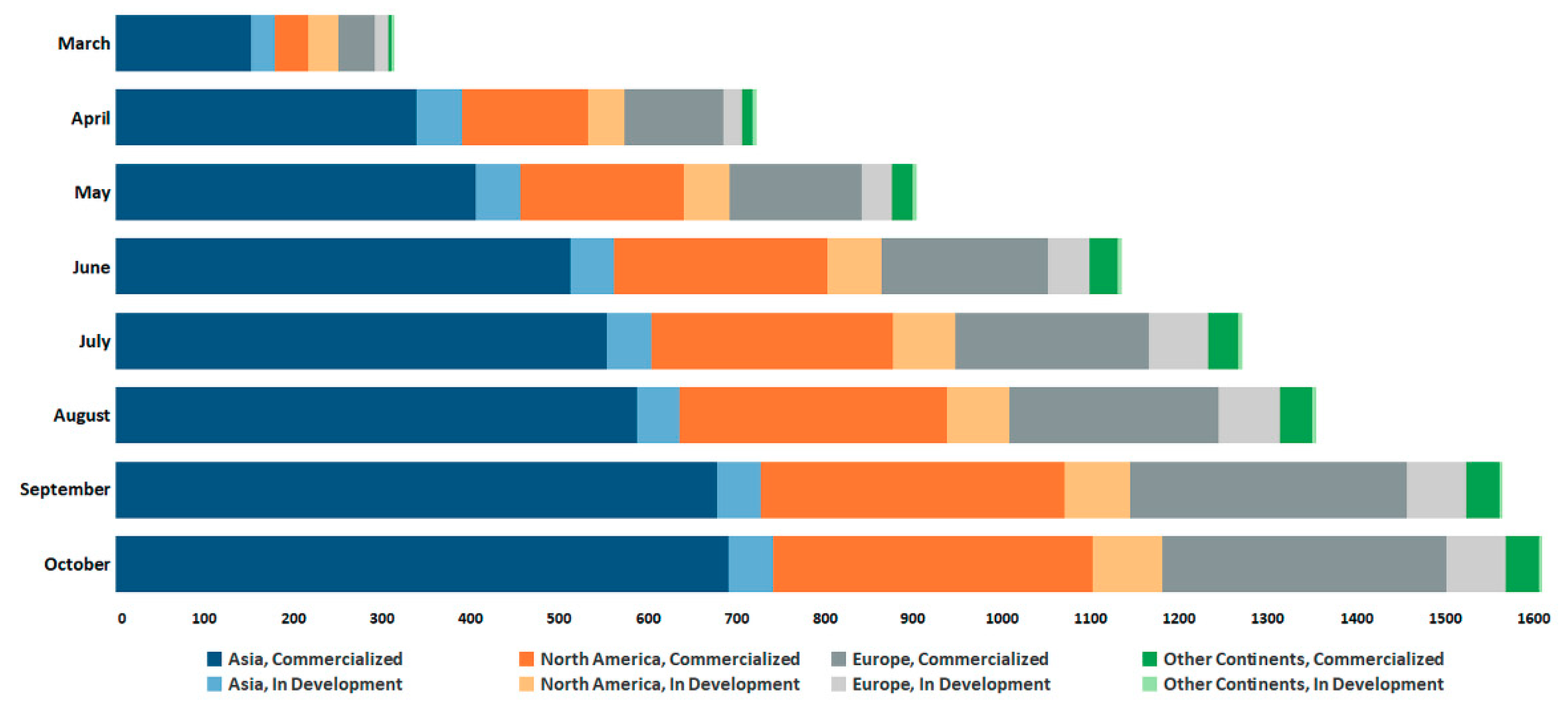
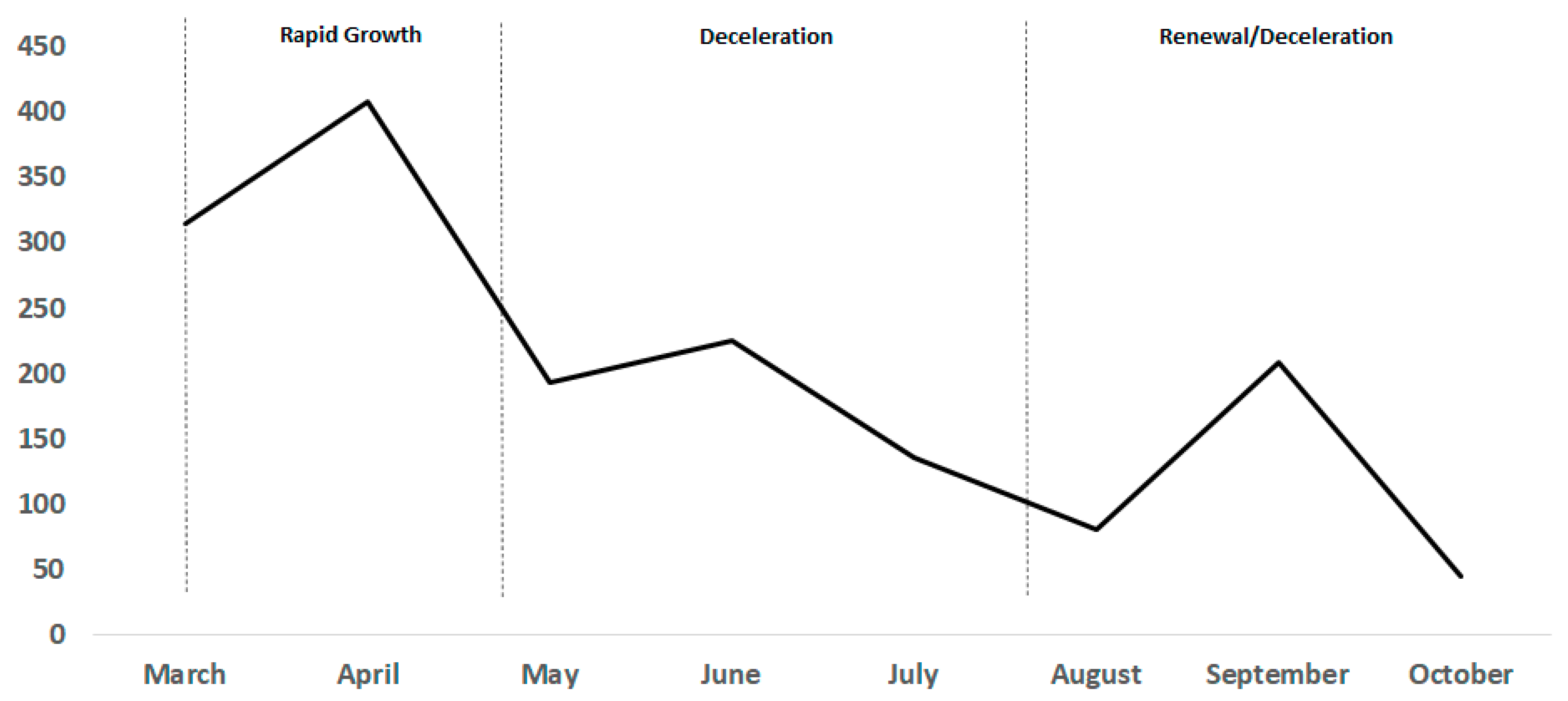
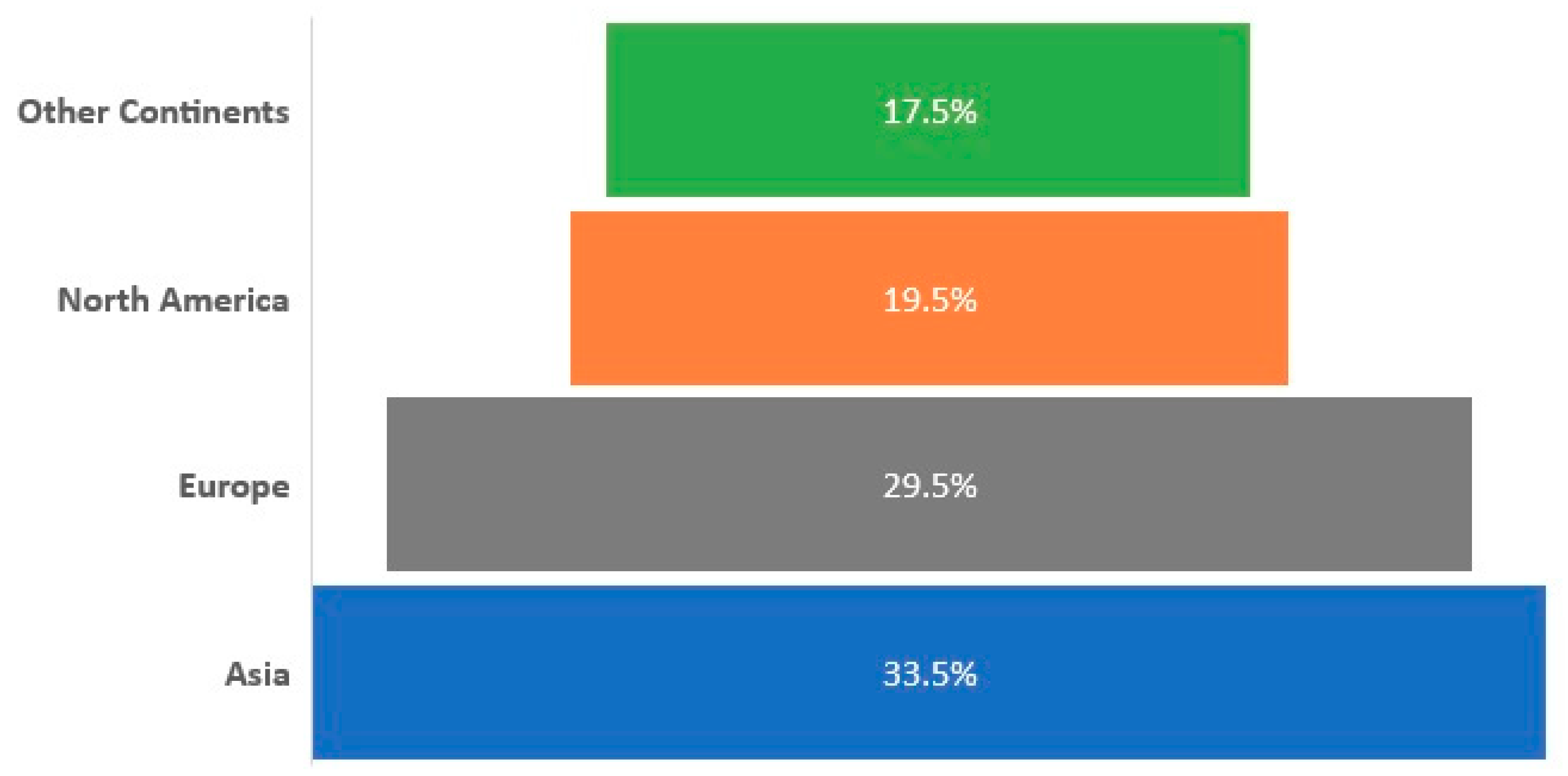
3.2. Global Trends in SARS-CoV-2 Diagnostic Market
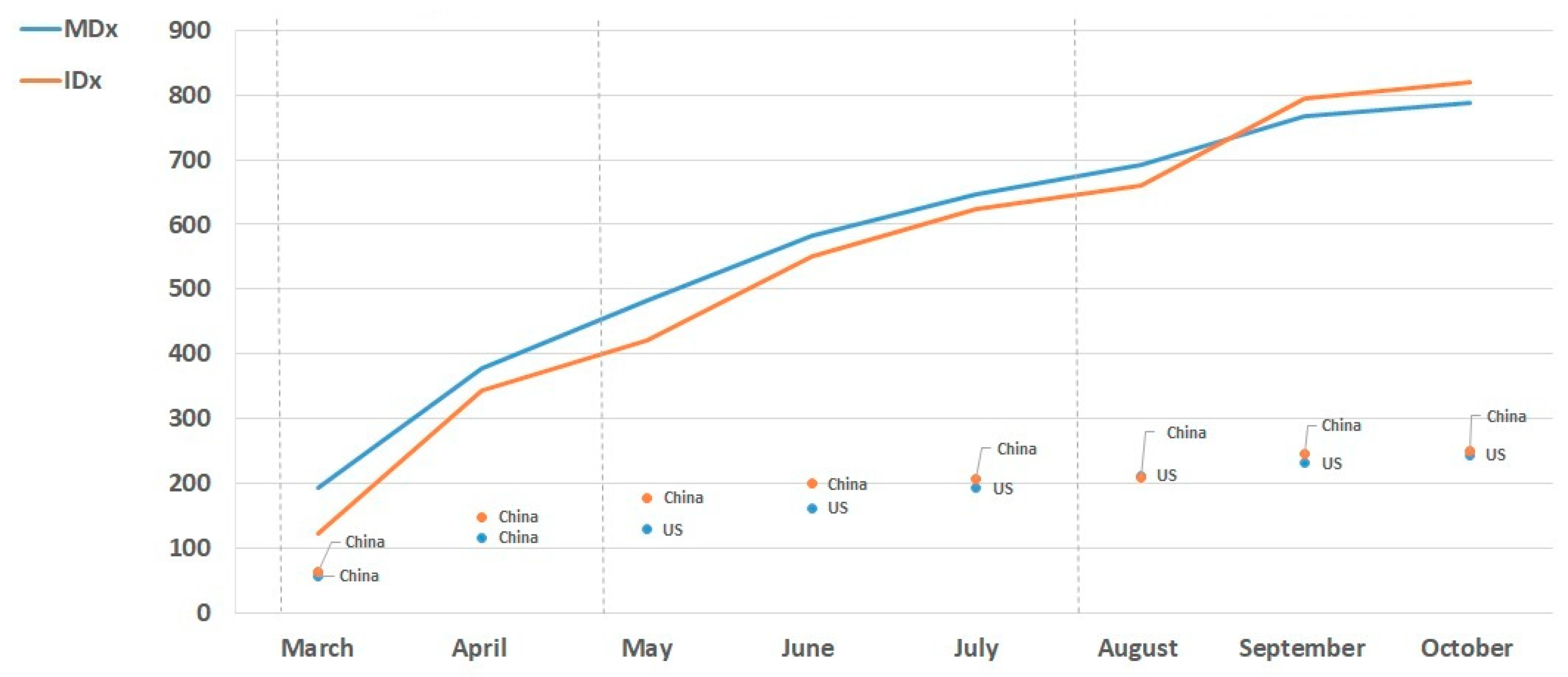
3.3. Global Trends in SARS-CoV-2 MDx and IDx Solutions
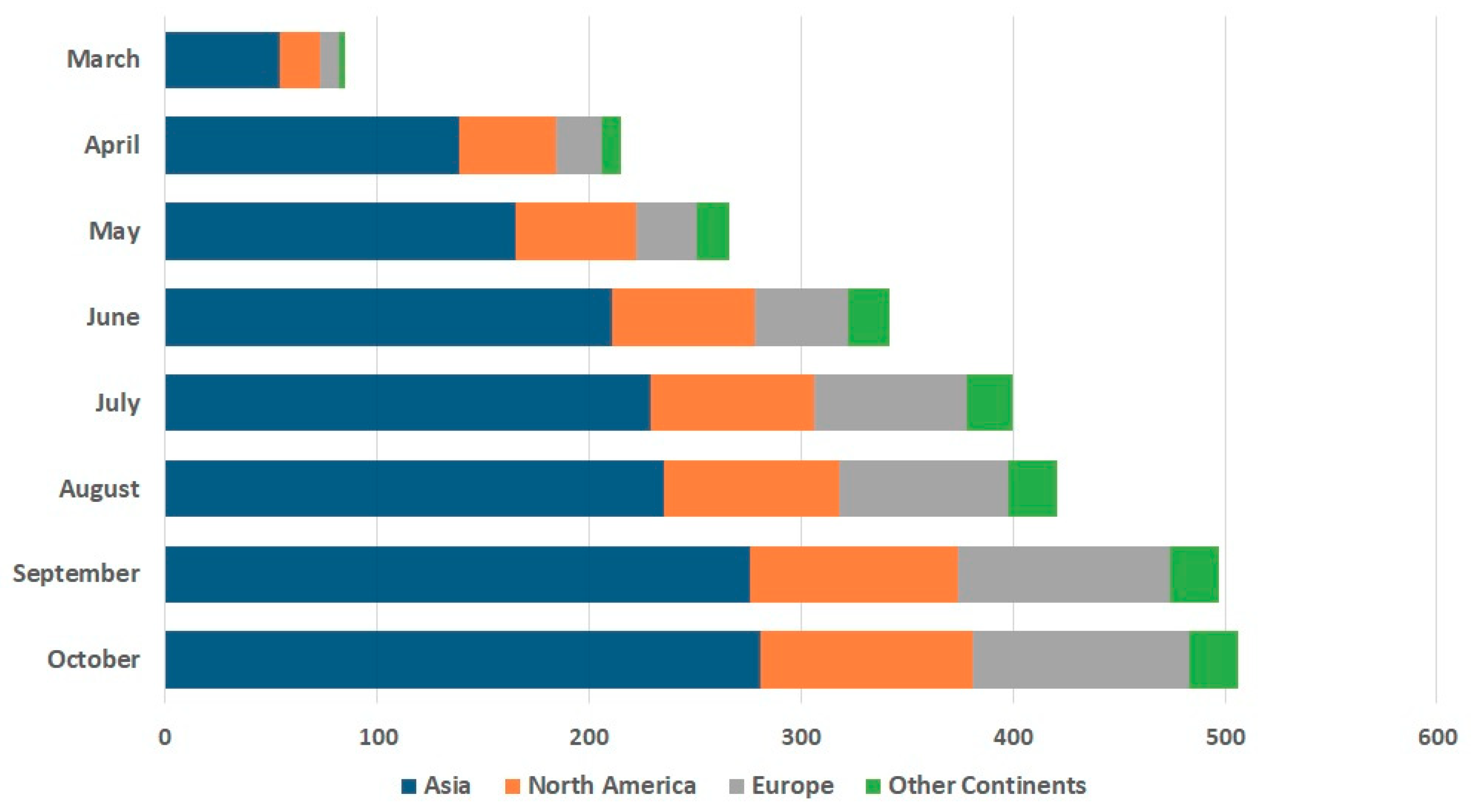
3.4. Global Trends in SARS-CoV-2 Rapid Diagnostic Solutions
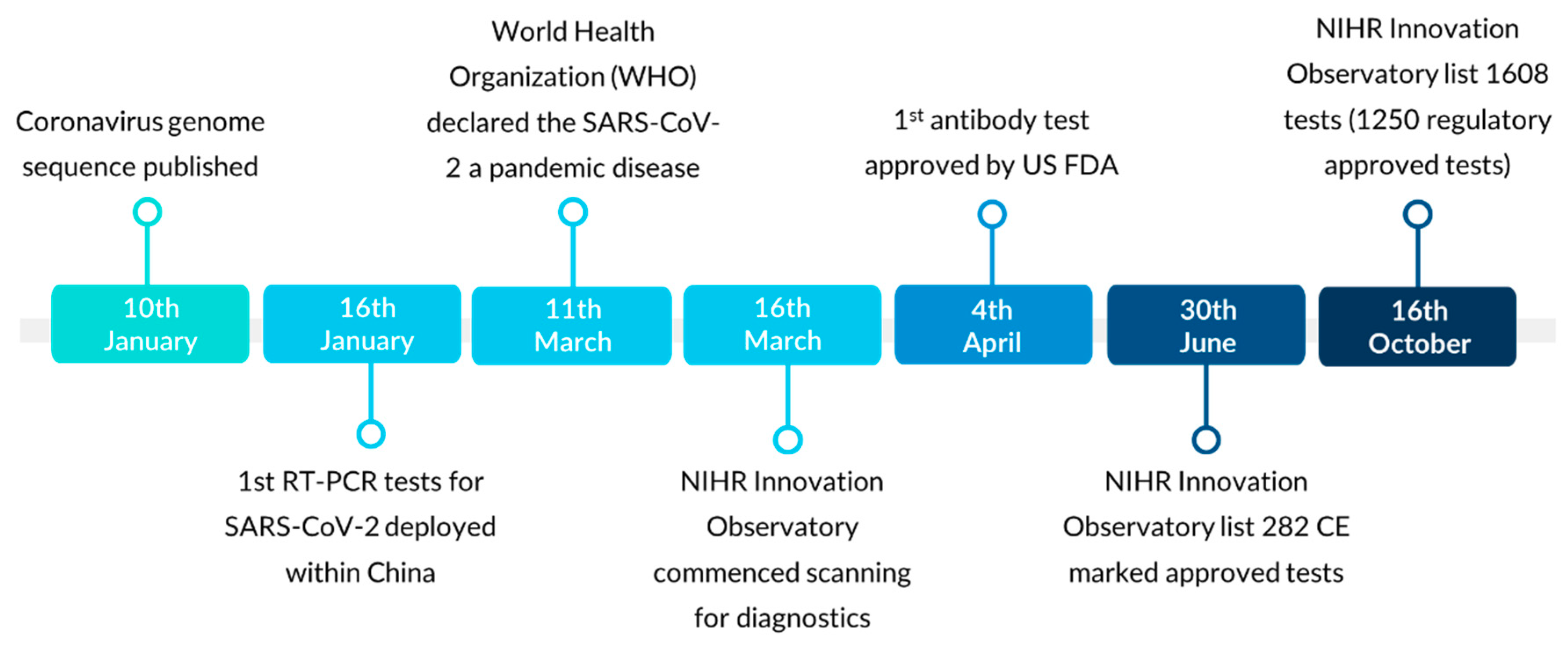
3.5. Evolving SARS-CoV-2 Diagnostics Regulatory Landscape
3.6. Direct-to-Consumer SARS-CoV-2 Service Tests
4. Discussion
4.1. Global Eruption of Diagnostic Technologies in the Wake of COVID-19
4.2. Impact of SARS-CoV-2 Technological Innovation on Regulatory Approval
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sohrabi, C.; Alsafi, Z.; O’Neill, N.; Khan, M.; Kerwan, A.; Al-Jabir, A.; Iosifidis, C.; Agha, R. World Health Organization declares global emergency: A review of the 2019 novel coronavirus (COVID-19). Int. J. Surg. 2020, 76, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Corman, V.M.; Landt, O; Kaiser, M.; Molenkamp, R.; Meijer, A.; Chu, D.K.W.; Bleicker, T.; Brünink, S.; Schneider, J.; Schmidt, M.L.; et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Euro Surveill 2020, 25, 2000045. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.P.; Papenburg, J.; Desjardins, M.; Kanjilal, S.; Quach, C.; Libman, M.; Dittrich, S.; Yansouni, C.P. Diagnostic Testing for Severe Acute Respiratory Syndrome–Related Coronavirus 2. Ann. Intern. Med. 2020, 172, 726–734. [Google Scholar] [CrossRef] [PubMed]
- Taipale, J.; Romer, P.; Linnarsson, S. Population-scale testing can suppress the spread of COVID-19. MedRxiv 2020. [Google Scholar] [CrossRef]
- Udugama, B.; Kadhiresan, P.; Kozlowski, H.; Malekjahani, A.; Osborne, M.; Li, V.; Chen, H.; Mubareka, S.; Gubbay, J.; Chan, W. Diagnosing COVID-19: The Disease and Tools for Detection. ACS Nano 2020, 14, 3822–3835. [Google Scholar] [CrossRef]
- Carter, L.; LGarner, L.; Smoot, J.; Li, Y.; Zhou, Q.; Saveson, C.; Sasso, J.; Gregg, A.; Soares, D.; Beskid, T.; et al. Assay Techniques and Test Development for COVID-19 Diagnosis. ACS Cent. Sci. 2020, 6, 591–605. [Google Scholar] [CrossRef]
- Sheridan, C. Fast, portable tests come online to curb coronavirus pandemic. Nat. Biotechnol. 2020, 38, 515–518. [Google Scholar] [CrossRef]
- Market Data Forecast. Global In-Vitro Diagnostics Market Size, Share, Trends, Growth Analysis Report—Segmented By Product, IVD Technology, Application and Region—Industry Forecast|2020 to 2025. Available online: https://www.marketdataforecast.com/market-reports/global-in-vitro-diagnostics-market (accessed on 28 October 2020).
- World Health Organization. Laboratory Testing for Coronavirus Disease (COVID-19) in Suspected Human Cases: Interim Guidance. Available online: https://apps.who.int/iris/handle/10665/331501 (accessed on 19 March 2020).
- Centers for Disease Control and Prevention (CDC). Interim Guidelines for Collecting, Handling, and Testing Clinical Specimens for COVID-19. Available online: https://www.cdc.gov/coronavirus/2019-ncov/lab/guidelines-clinical-specimens.html (accessed on 10 December 2020).
- Ejazi, S.A.; Ghosh, S.; Ali, N. Antibody detection assays for COVID-19 diagnosis: An early overview. Immunol. Cell Biol. 2021, 99, 21–33. [Google Scholar] [CrossRef]
- The Centre for Evidence-Based Medicine (CEBM). What Tests Could Potentially be Used for the Screening, Diagnosis and Monitoring of COVID-19 and What Are Their Advantages and Disadvantages? Available online: https://www.cebm.net/covid-19/what-tests-could-potentially-be-used-for-the-screening-diagnosis-and-monitoring-of-covid-19-and-what-are-their-advantages-and-disadvantages/ (accessed on 29 October 2020).
- Centers for Disease Control and Prevention (CDC). COVID-19 Testing Overview. Available online: https://www.cdc.gov/coronavirus/2019-ncov/symptoms-testing/testing.html (accessed on 29 October 2020).
- Smyrlaki, I.; Ekman, M.; Lentini, A.; Rufino de Sousa, N.; Papanicolaou, N.; Vondracek, M.; Aarum, J.; Safari, H.; Muradrasoli, S.; Rothfuchs, A.G.; et al. Massive and rapid COVID-19 testing is feasible by extraction-free SARS-CoV-2 RT-PCR. Nat. Commun. 2020, 11, 4812. [Google Scholar] [CrossRef]
- Garg, J.; Singh, V.; Pandey, P.; Verma, A.; Sen, M.; Das, A.; Agarwal, J. Evaluation of sample pooling for diagnosis of COVID-19 by real time-PCR: A resource-saving combat strategy. J. Med. Virol. 2020. [Google Scholar] [CrossRef]
- Esbin, M.N.; Whitney, O.N.; Chong, S.; Maurer, A.; Darzacq, X.; Tjian, R. Overcoming the bottleneck to widespread testing: A rapid review of nucleic acid testing approaches for COVID-19 detection. RNA 2020, 26, 771–783. [Google Scholar] [CrossRef] [PubMed]
- Beetz, C.; Skrahina, V.; Förster, T.M.; Gaber, H.; Paul, J.J.; Curado, F.; Rolfs, A.; Bauer, P.; Schäfer, S.; Weckesser, V.; et al. Rapid Large-Scale COVID-19 Testing during Shortages. Diagnostics 2020, 10, 464. [Google Scholar] [CrossRef] [PubMed]
- Fomsgaard, A.S.; Rosenstierne, M.W. An alternative workflow for molecular detection of SARS-CoV-2—escape from the NA extraction kit-shortage, Copenhagen, Denmark, March 2020. Eurosurveillance 2020, 25, 2000398. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.; Cui, J.; Huang, L.; Du, B.; Chen, L.; Xue, G.; Li, S.; Zhang, W.; Zhao, L.; Sun, Y.; et al. Rapid and visual detection of 2019 novel coronavirus (SARS-CoV-2) by a reverse transcription loop-mediated isothermal amplification assay. Clin. Microbiol. Infect. 2020, 26, 773–779. [Google Scholar] [CrossRef]
- Zhao, J.; Yuan, Q.; Wang, H.; Liu, W.; Lioa, X.; Su, Y.; Wang, X.; Yuan, J.; Li, T.; Li, J.; et al. Antibody Responses to SARS-CoV-2 in Patients With Novel Coronavirus Disease 2019. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- De Marinis, Y.; Sunnerhagen, T.; Bompada, P.; Bläckberg, A.; Yang, R.; Svensson, J.; Ekström, O.; Eriksson, K.-F.; Hansson, O.; Groop, L.; et al. Serology assessment of antibody response to SARS-CoV-2 in patients with COVID-19 by rapid IgM/IgG antibody test. Infect. Ecol. Epidemiol. 2020, 10, 1821513. [Google Scholar] [CrossRef]
- European Centre for Disease Prevention and Control (ECDC). Options for the Use of Rapid Antigen Tests for COVID-19 in the EU/EEA and the UK. Available online: https://www.ecdc.europa.eu/sites/default/files/documents/Options-use-of-rapid-antigen-tests-for-COVID-19.pdf (accessed on 20 January 2021).
- World Health Organisation (WHO). Global Partnership to Make Available 120 Million Affordable, Quality COVID-19 Rapid Tests for Low- and Middle-Income Countries. Available online: https://www.who.int/news/item/28-09-2020-global-partnership-to-make-available-120-million-affordable-quality-covid-19-rapid-tests-for-low--and-middle-income-countries (accessed on 20 January 2021).
- UK Department of Health and Social Care. More Rapid COVID-19 Tests to be Rolled out across England. Available online: https://www.gov.uk/government/news/more-rapid-covid-19-tests-to-be-rolled-out-across-england (accessed on 20 January 2021).
- Tromberg, B.J.; Schwetz, T.A.; Pérez-Stable, E.J.; Hodes, R.J.; Woychik, R.P.; Bright, R.A.; Fleurence, R.L.; Collins, F.S. Rapid Scaling Up of Covid-19 Diagnostic Testing in the United States—The NIH RADx Initiative. N. Engl. J. Med. 2020, 383, 1071–1077. [Google Scholar] [CrossRef]
- Ganguli, A.; Mostafa, A.; Berger, J.; Aydin, M.Y.; Sun, F.; Ramirez, S.A.S.d.; Valera, E.; Cunningham, B.T.; King, W.P.; Bashir, R. Rapid isothermal amplification and portable detection system for SARS-CoV-2. Proc. Natl. Acad. Sci. USA 2020, 117, 22727. [Google Scholar] [CrossRef]
- Nguyen, T.; Duong Bang, D.; Wolff, A. 2019 Novel Coronavirus Disease (COVID-19): Paving the Road for Rapid Detection and Point-of-Care Diagnostics. Micromachines (Basel) 2020, 11, 306. [Google Scholar] [CrossRef]
- Ahmad, S.; Ali, N.; Kausar, M.; Misbah, H.; Wahid, A. Road toward rapid-molecular point of care test to detect novel SARS-coronavirus 2019 (COVID-19): Review from updated literature. Allergol. Et Immunopathol. 2020, 48, 518–520. [Google Scholar] [CrossRef] [PubMed]
- Diao, B.; Wen, K.; Chen, J.; Liu, Y.; Han, C.; Yuan, Z.; Chen, J.; Pan, Y.; Chen, L.; Dan, Y.; et al. Diagnosis of Acute Respiratory Syndrome Coronavirus 2 Infection by Detection of Nucleocapsid Protein. MedRxiv 2020. [Google Scholar] [CrossRef]
- Che, X.; Qiu, L.; Pan, Y.; Wen, K.; Hao, W.; Zhang, L.; Wang, Y.; Liao, Z.; Hua, X.; Cheng, V.; et al. Sensitive and Specific Monoclonal Antibody-Based Capture Enzyme Immunoassay for Detection of Nucleocapsid Antigen in Sera from Patients with Severe Acute Respiratory Syndrome. J. Clin. Microbiol. 2004, 42, 2629–2635. [Google Scholar] [CrossRef] [PubMed][Green Version]
- World Health Organisation (WHO). Antigen-Detection in the Diagnosis of SARS-CoV-2 Infection Using Rapid Immunoassays. 2020. Available online: https://www.who.int/publications/i/item/antigen-detection-in-the-diagnosis-of-sars-cov-2infection-using-rapid-immunoassays (accessed on 10 December 2020).
- Traugott, M.; Aberle, S.; Aberle, J.; Griebler, H.; Karolyi, M.; Pawelka, E.; Puchhammer-Stöckl, E.; Zoufaly, A.; Weseslindtner, L. Performance of Severe Acute Respiratory Syndrome Coronavirus 2 Antibody Assays in Different Stages of Infection: Comparison of Commercial Enzyme-Linked Immunosorbent Assays and Rapid Tests. J. Infect. Dis. 2020, 222, 362–366. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Yi, Y.; Luo, X.; Xiong, N.; Liu, Y.; Li, S.; Sun, R.; Wang, Y.; Chen, W. Development and clinical application of a rapid IgM-IgG combined antibody test for SARS-CoV-2 infection diagnosis. J. Med. Virol. 2020, 92. [Google Scholar] [CrossRef] [PubMed]
- Maloney, B.E.; Perera, K.D.; Saunders, D.R.D.; Shadipeni, N.; Fleming, S.D. Interactions of viruses and the humoral innate immune response. Clin. Immunol. 2020, 212, 108351. [Google Scholar] [CrossRef]
- Lau, E.H.Y.; Tsang, O.T.Y.; Hui, D.S.C.; Kwan, M.Y.W.; Chan, W.-h.; Chiu, S.S.; Ko, R.L.W.; Chan, K.H.; Cheng, S.M.S.; Perera, R.A.P.M.; et al. Neutralizing antibody titres in SARS-CoV-2 infections. Nat. Commun. 2021, 12, 63. [Google Scholar] [CrossRef]
- Favresse, J.; Elsen, M.; Eucher, C.; Laffineur, K.; Van Eeckhoudt, S.; Nicolas, J.-B.; Gillot, C.; Dogné, J.-M.; Douxfils, J. Long-term kinetics of anti-SARS-CoV-2 antibodies in a cohort of 197 hospitalized and non-hospitalized COVID-19 patients. Clin. Chem. Lab. Med. (Cclm) 2020, 000010151520201736. [Google Scholar] [CrossRef]
- Lu, R.; Zhao, X.; Li, J.; Niu, P.; Yang, B.; Wu, H.; Wang, W.; Song, H.; Huang, B.; Zhu, N.; et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: Implications for virus origins and receptor binding. Lancet 2020, 395, 565–574. [Google Scholar] [CrossRef]
- GenBank. Severe Acute Respiratory Syndrome Coronavirus 2 Isolate Wuhan-Hu-1, Complete Genome. Available online: https://www.ncbi.nlm.nih.gov/nuccore/MN908947 (accessed on 9 November 2020).
- Drosten, C.; Günther, S.; Preiser, W.; van der Werf, S.; Brodt, H.-R.; Becker, S.; Rabenau, H.; Panning, M.; Kolesnikova, L.; Fouchier, R.A.M.; et al. Identification of a Novel Coronavirus in Patients with Severe Acute Respiratory Syndrome. N. Engl. J. Med. 2003, 348, 1967–1976. [Google Scholar] [CrossRef]
- Bedford, J.; Enria, D.; Giesecke, J.; Heymann, D.L.; Ihekweazu, C.; Kobinger, G.; Lane, H.C.; Memish, Z.; Oh, M.-D.; Sall, A.A.; et al. COVID-19: Towards controlling of a pandemic. Lancet 2020, 395, 1015–1018. [Google Scholar] [CrossRef]
- FINDDX. SARS-COV-2 Diagnostic Pipeline. Available online: https://www.finddx.org/covid-19/pipeline/ (accessed on 14 December 2020).
- Medicines and Healthcare products Regulatory Agency (MHRA). For Industry and Manufacturers: COVID-19 Tests and Testing Kits. 2020. Available online: https://www.gov.uk/government/publications/how-tests-and-testing-kits-for-coronavirus-covid-19-work/for-industry-and-manufactures-covid-19-tests-and-testing-kits (accessed on 10 December 2020).
- Kalokairinou, L.; Zettler, P.J.; Nagappan, A.; Kyweluk, M.A.; Wexler, A. The promise of direct-to-consumer COVID-19 testing: Ethical and regulatory issues. J. Law Biosci. 2020. [Google Scholar] [CrossRef]
- Medicines and Healthcare Products Regulatory Agency (MHRA). Action Taken to Halt Sales of Fingerprick Coronavirus (COVID-19) Antibody Testing Kits. Available online: https://www.gov.uk/government/news/action-taken-to-halt-sales-of-fingerprick-coronavirus-covid-19-antibody-testing-kits (accessed on 10 December 2020).
- Which? Coronavirus: Can You Get a Home Test Kit for COVID-19? Available online: https://www.which.co.uk/news/2020/11/coronavirus-can-you-get-a-home-test-kit/ (accessed on 10 December 2020).
- Medicines and Healthcare Products Regulatory Agency (MHRA). For Patients, the Public and Professional Users: A Guide to COVID-19 Tests and Testing Kits. 2020. Available online: https://www.gov.uk/government/publications/how-tests-and-testing-kits-for-coronavirus-covid-19-work/for-patients-the-public-and-professional-users-a-guide-to-covid-19-tests-and-testing-kits (accessed on 10 December 2020).
- National Institiute for Health Research Innovation Observatory. COVID-19 Diagnostics. Available online: http://www.io.nihr.ac.uk/report/covid-19-diagnostics/ (accessed on 16 October 2020).
- US Food and Drug Administration. Emergency Use Authorization. Available online: https://www.fda.gov/emergency-preparedness-and-response/mcm-legal-regulatory-and-policy-framework/emergency-use-authorization#covidinvitrodev (accessed on 16 November 2020).
- Canada Health. Testing Devices for COVID-19: Overview. Available online: https://www.canada.ca/en/health-canada/services/drugs-health-products/covid19-industry/medical-devices/testing.html#a4 (accessed on 10 December 2020).
- Australia Therapeutic Goods Administration. COVID-19 Test Kits Included in the ARTG for Legal Supply in Australia. Available online: https://www.tga.gov.au/covid-19-test-kits-included-artg-legal-supply-australia (accessed on 14 December 2020).
- South African Health Products Regulatory Authority. COVID-19 Information. Available online: http://www.sahpra.org.za/be-prepared-for-covid-19/ (accessed on 16 October 2020).
- European Commission. COVID-19 In Vitro Diagnostic Devices and Test Methods Database. Available online: https://covid-19-diagnostics.jrc.ec.europa.eu/devices#form_content (accessed on 14 December 2020).
- Pharmaceuticals and Medical Devices Agency. Activities Related to Measures against New Coronavirus Infections at PMDA. Available online: https://www.pmda.go.jp/about-pmda/news-release/0012.html (accessed on 14 December 2020).
- Ministry of Food and Drug Safety. Diagnostics Devices, MFDS-regulated Mask Information. Available online: https://www.mfds.go.kr/eng/brd/m_65/list.do?page=1&srchFr=&srchTo=&srchWord=&srchTp=&itm_seq_1=0&itm_seq_2=0&multi_itm_seq=0&company_cd=&company_nm= (accessed on 14 December 2020).
- Health Sciences Authority. HSA Expedites Approval of COVID-19 Diagnostic Tests in Singapore via Provisional Authorisation. Available online: https://www.hsa.gov.sg/announcements/regulatory-updates/hsa-expedites-approval-of-covid-19-diagnostic-tests-in-singapore-via-provisional-authorisation (accessed on 10 December 2020).
- Food and Drug Administration Philippines. Category: General Announcements. Available online: https://www.fda.gov.ph/category/general-announcements/ (accessed on 14 December 2020).
- ANVISA. Queries. Available online: https://consultas.anvisa.gov.br/#/saude/q/?nomeTecnico=coronav%C3%ADrus (accessed on 14 December 2020).
- World Health Organisation (WHO). Emergency Use Listing. Available online: https://www.who.int/teams/regulation-prequalification/eul (accessed on 14 December 2020).
- Lee, D.; Lee, J. Testing on the move: South Korea’s rapid response to the COVID-19 pandemic. Transp. Res. Interdiscip. Perspect. 2020, 5, 100111. [Google Scholar] [CrossRef]
- Kim, J.Y.; Choe, P.G.; Oh, Y.; Oh, K.J.; Kim, J.; Park, S.J.; Park, J.H.; Na, H.K.; Oh, M.-D. The First Case of 2019 Novel Coronavirus Pneumonia Imported into Korea from Wuhan, China: Implication for Infection Prevention and Control Measures. J. Korean Med. Sci. 2020, 35. [Google Scholar] [CrossRef] [PubMed]
- Report on the Epidemiological Features of Coronavirus Disease 2019 (COVID-19) Outbreak in the Republic of Korea from 19 January to 2 March 2020. J. Korean Med. Sci. 2020, 35. [CrossRef]
- Normile, D. Coronavirus Cases Have Dropped Sharply in South Korea. What’s the Secret to Its Success? Available online: https://www.sciencemag.org/news/2020/03/coronavirus-cases-have-dropped-sharply-south-korea-whats-secret-its-success (accessed on 16 November 2020).
- National Medical Products Administration (NMPA). Cabinet Ramps up COVID-19 Testing. Available online: http://subsites.chinadaily.com.cn/nmpa/2020-08/04/c_526417.htm (accessed on 16 November 2020).
- National Medical Products Administration (NMPA). Premier Urges Development of More Efficient Nucleic Acid Testing Products. Available online: http://subsites.chinadaily.com.cn/nmpa/2020-08/03/c_525962.htm (accessed on 16 November 2020).
- National Medical Products Administration (NMPA). Beijing ups Its Nucleic Acid Testing Capability. Available online: http://subsites.chinadaily.com.cn/nmpa/2020-06/29/c_504741.htm (accessed on 16 November 2020).
- Oh, J.; Lee, J.-K.; Schwarz, D.; Ratcliffe, H.L.; Markuns, J.F.; Hirschhorn, L.R. National Response to COVID-19 in the Republic of Korea and Lessons Learned for Other Countries. Health Syst. Reform 2020, 6, e1753464. [Google Scholar] [CrossRef] [PubMed]
- National Medical Products Administration (NMPA). Mass Testing a Go-to Response. Available online: http://subsites.chinadaily.com.cn/nmpa/2020-07/31/c_525625.htm (accessed on 16 November 2020).
- National Medical Products Administration (NMPA). NMPA Gives Emergency Approvals to COVID-19 Test Kits. Available online: http://subsites.chinadaily.com.cn/nmpa/2020-03/27/c_465663.htm (accessed on 16 November 2020).
- National Medical Products Administration (NMPA). China’s Daily Nucleic Acid Testing Capacity Tops 3m: Official. Available online: http://subsites.chinadaily.com.cn/nmpa/2020-06/28/c_504442.htm (accessed on 16 November 2020).
- World Health Organisation (WHO). Report of the WHO-China Joint Mission on Coronavirus Disease 2019 (COVID-19). Available online: https://www.who.int/docs/default-source/coronaviruse/who-china-joint-mission-on-covid-19-final-report.pdf (accessed on 16 November 2020).
- Ari-Veikko, A. Successful Government Responses to the Pandemic: Contextualizing National and Urban Responses to the COVID-19 Outbreak in East and West. Int. J. E-Plan. Res. (IJEPR) 2021, 10, 1–17. [Google Scholar] [CrossRef]
- Ravi, N.; Cortade, D.L.; Ng, E.; Wang, S.X. Diagnostics for SARS-CoV-2 detection: A comprehensive review of the FDA-EUA COVID-19 testing landscape. Biosens. Bioelectron. 2020, 165, 112454. [Google Scholar] [CrossRef]
- Afzal, A. Molecular diagnostic technologies for COVID-19: Limitations and challenges. J. Adv. Res. 2020, 26, 149–159. [Google Scholar] [CrossRef]
- Wise, J. Covid-19: Which rapid tests is the UK pinning its hopes on? BMJ 2020, 371, m3868. [Google Scholar] [CrossRef]
- Vashist, S.K. In Vitro Diagnostic Assays for COVID-19: Recent Advances and Emerging Trends. Diagnostics 2020, 10, 202. [Google Scholar] [CrossRef]
- Bialek, S.; Bowen, V.; Chow, N.; Curns, A.; Gierke, R.; Hall, A.; Hughes, M.; Pilishvili, T.; Ritchey, M.; Roguski, K.; et al. Geographic differences in COVID-19 cases, deaths, and incidence—United States, 12 February–7 April 2020. Morbidity and Mortality Weekly Report 2020, 68, 465–471. [Google Scholar] [CrossRef]
- Alanezi, F.; Aljahdali, A.; Alyousef, S.M.; Alrashed, H.; Mushcab, H.; AlThani, B.; Alghamedy, F.; Alotaibi, H.; Saadah, A.; Alanzi, T. A Comparative Study on the Strategies Adopted by the United Kingdom, India, China, Italy, and Saudi Arabia to Contain the Spread of the COVID-19 Pandemic. J. Healthc. Lead. 2020, 12, 117–131. [Google Scholar] [CrossRef] [PubMed]
- Winter, A.K.; Hegde, S.T. The important role of serology for COVID-19 control. Lancet Infect. Dis. 2020, 20, 758–759. [Google Scholar] [CrossRef]
- Long, Q.-X.; Deng, H.-J.; Chen, J.; Hu, J.-L.; Liu, B.-Z.; Liao, P.; Lin, Y.; Yu, L.-H.; Mo, Z.; Xu, Y.-Y.; et al. Antibody responses to SARS-CoV-2 in COVID-19 patients: The perspective application of serological tests in clinical practice. medRxiv 2020. [Google Scholar] [CrossRef]
- Lee, C.Y.-P.; Lin, R.T.P.; Renia, L.; Ng, L.F.P. Serological Approaches for COVID-19: Epidemiologic Perspective on Surveillance and Control. Front. Immunol. 2020, 11. [Google Scholar] [CrossRef]
- Chan, P.K.S.; Ng, K.-C.; Chan, R.C.W.; Lam, R.K.Y.; Chow, V.C.Y.; Hui, M.; Wu, A.; Lee, N.; Yap, F.H.Y.; Cheng, F.W.T.; et al. Immunofluorescence Assay for Serologic Diagnosis of SARS. Emerg. Infect. Dis. J. 2004, 10, 530. [Google Scholar] [CrossRef]
- Okba, N.M.A.; Müller, M.A.; Li, W.; Wang, C.; GeurtsvanKessel, C.H.; Corman, V.M.; Lamers, M.M.; Sikkema, R.S.; de Bruin, E.; Chandler, F.D.; et al. SARS-CoV-2 specific antibody responses in COVID-19 patients. medRxiv 2020. [Google Scholar] [CrossRef]
- Global Data. United States In Vitro Diagnostics (IVD) Market Outlook to 2025—Clinical Chemistry, Genetic Testing, Hematology, Histology and Cytology, Immuno Chemistry, Infectious Diagnostics and Microbiology Culture. 2017. Available online: https://medical.globaldata.com/Analysis/TableOfContents/United-States-In-Vitro-Diagnostics-Market-Outlook-to-2025---Cardiac-Disease--Clinical-Chemistry--Hematological-Disorders-and-Others (accessed on 7 July 2020).
- Global Data. China In Vitro Diagnostics (IVD) Market Outlook to 2025—Clinical Chemistry, Genetic Testing, Hematology, Histology and Cytology, Immuno Chemistry, Infectious Diagnostics and Microbiology Culture. 2017. Available online: https://medical.globaldata.com/Analysis/TableOfContents/China-In-Vitro-Diagnostics-Market-Outlook-to-2025---Cardiac-Disease--Clinical-Chemistry--Hematological-Disorders-and-Others (accessed on 7 July 2020).
- Science. Coronavirus Antigen Tests: Quick and Cheap, but Too Often Wrong? Available online: https://www.sciencemag.org/news/2020/05/coronavirus-antigen-tests-quick-and-cheap-too-often-wrong (accessed on 14 December 2020).
- GlobalData Healthcare. Fast, Accurate, and Cost-Effective POC Covid-19 Tests Are Set to Revolutionise the Testing Market. Available online: https://www.medicaldevice-network.com/comment/poc-covid-19-tests/ (accessed on 20 January 2021).
- European Centre for Disease Prevention and Control (ECDC). An Overview of the Rapid Test Situation for COVID-19 Diagnosis in the EU/EEA. Available online: https://www.ecdc.europa.eu/sites/default/files/documents/Overview-rapid-test-situation-for-COVID-19-diagnosis-EU-EEA.pdf (accessed on 07 December 2020).
- Kilic, T.; Weissleder, R.; Lee, H. Molecular and Immunological Diagnostic Tests of COVID-19: Current Status and Challenges. iScience 2020, 23, 101406. [Google Scholar] [CrossRef]
- Porte, L.; Legarraga, P.; Vollrath, V.; Aguilera, X.; Munita, J.M.; Araos, R.; Pizarro, G.; Vial, P.; Iruretagoyena, M.; Dittrich, S.; et al. Evaluation of a novel antigen-based rapid detection test for the diagnosis of SARS-CoV-2 in respiratory samples. Int. J. Infect. Dis. 2020, 99, 328–333. [Google Scholar] [CrossRef]
- Gremmels, H.; Winkel, B.M.F.; Schuurman, R.; Rosingh, A.; Rigter, N.A.M.; Rodriguez, O.; Ubijaan, J.; Wensing, A.M.J.; Bonten, M.J.M.; Hofstra, L.M. Real-life validation of the Panbio™ COVID-19 antigen rapid test (Abbott) in community-dwelling subjects with symptoms of potential SARS-CoV-2 infection. EClinicalMedicine 2020, 100677. [Google Scholar] [CrossRef]
- Lambert-Niclot, S.; Cuffel, A.; Le Pape, S.; Vauloup-Fellous, C.; Morand-Joubert, L.; Roque-Afonso, A.-M.; Le Goff, J.; Delaugerre, C. Evaluation of a Rapid Diagnostic Assay for Detection of SARS-CoV-2 Antigen in Nasopharyngeal Swabs. J. Clin. Microbiol. 2020, 58, e00977-00920. [Google Scholar] [CrossRef] [PubMed]
- Weitzel, T.; Legarraga, P.; Iruretagoyena, M.; Pizarro, G.; Vollrath, V.; Araos, R.; Munita, J.M.; Porte, L. Head-to-head comparison of four antigen-based rapid detection tests for the diagnosis of SARS-CoV-2 in respiratory samples. bioRxiv 2020. [Google Scholar] [CrossRef]
- Kyosei, Y.; Namba, M.; Yamura, S.; Takeuchi, R.; Aoki, N.; Nakaishi, K.; Watabe, S.; Ito, E. Proposal of De Novo Antigen Test for COVID-19: Ultrasensitive Detection of Spike Proteins of SARS-CoV-2. Diagnostics 2020, 10, 594. [Google Scholar] [CrossRef]
- Ying, L.; Yue-ping, L.; Bo, D.; Feifei, R.; Yue, W.; Jinya, D.; Qianchuan, H. Diagnostic Indexes of a Rapid IgG/IgM Combined Antibody Test for SARS-CoV-2. medRxiv 2020. [Google Scholar] [CrossRef]
- Andrey, D.O.; Cohen, P.; Meyer, B.; Torriani, G.; Yerly, S.; Mazza, L.; Calame, A.; Arm-Vernez, I.; Guessous, I.; Stringhini, S.; et al. Head-to-Head Accuracy Comparison of Three Commercial COVID-19 IgM/IgG Serology Rapid Tests. J. Clin. Med. 2020, 9, 2369. [Google Scholar] [CrossRef]
- David, N.; Mash, R. Community-based screening and testing for Coronavirus in Cape Town, South Africa: Short report. Afr. J. Prim Health Care Fam. Med. 2020, 12. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.-W.; Schmitz, J.E.; Persing, D.H.; Stratton, C.W. Laboratory Diagnosis of COVID-19: Current Issues and Challenges. J. Clin. Microbiol. 2020, 58, e00512–e00520. [Google Scholar] [CrossRef]
- World Health Organisation (WHO). Laboratory Testing Strategy Recommendations for COVID-19. 2020. Available online: https://www.who.int/publications/i/item/laboratory-testing-for-2019-novel-coronavirus-in-suspected-human-cases-20200117 (accessed on 28 October 2020).
- Ricks, S.; Kendall, E.A.; Dowdy, D.W.; Sacks, J.A.; Schumacher, S.G.; Arinaminpathy, N. Quantifying the potential value of antigen-detection rapid diagnostic tests for COVID-19: A modelling analysis. medRxiv 2020. [Google Scholar] [CrossRef]
- Pan American Health Organization. Rapid Antigen Tests Arriving in Countries to Assist in COVID-19 Response in the Americas. Available online: https://www.paho.org/en/news/10-11-2020-rapid-antigen-tests-arriving-countries-assist-covid-19-response-americas (accessed on 10 December 2020).
- Scohy, A.; Anantharajah, A.; Bodéus, M.; Kabamba-Mukadi, B.; Verroken, A.; Rodriguez-Villalobos, H. Low Performance of Rapid Antigen Detection Test as Frontline Testing for COVID-19 Diagnosis. J. Clin. Virol. 2020, 129, 104455. [Google Scholar] [CrossRef]
- National Medical Products Administration (NMPA). Regulatory Requirements and Standards for Coronavirus Reagent Test Kits and Protective Equipment in China. Available online: http://english.nmpa.gov.cn/2020-03/30/c_467202.htm (accessed on 10 December 2020).
- Bchetnia, M.; Girard, C.; Duchaine, C.; Laprise, C. The outbreak of the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): A review of the current global status. J. Infect. Public Health 2020, 13, 1601–1610. [Google Scholar] [CrossRef]
- National Medical Products Administration (NMPA). Expanded COVID-19 Testing to Cover Key Groups. Available online: http://english.nmpa.gov.cn/2020-06/09/c_499083.htm (accessed on 10 December 2020).
- Umaru, F.A. Scaling up testing for COVID-19 in Africa: Responding to the pandemic in ways that strengthen health systems. Afr. J. Lab. Med. 2020, 9. [Google Scholar] [CrossRef]
- Kobia, F.; Gitaka, J. COVID-19: Are Africa’s diagnostic challenges blunting response effectiveness? Aas Open Res. 2020, 3. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation (WHO). Accelerating regulation in response to COVID-19. Bull. World Health Organ. 2020, 98, 514–515. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, J.; Kühne, V.; Lunguya, O.; Affolabi, D.; Hardy, L.; Vandenberg, O. Implementing COVID-19 (SARS-CoV-2) Rapid Diagnostic Tests in Sub-Saharan Africa: A Review. Front. Med. 2020, 7. [Google Scholar] [CrossRef] [PubMed]
- Idris Nasir, A.; Anthony Uchenna, E.; Azeez Oyebanji, A.; Peter Elisha, G.; Hafeez Aderinsayo, A.; Yakubu, I.; Amos, D. Roles and challenges of coordinated public health laboratory response against COVID-19 pandemic in Africa. J. Infect. Dev. Ctries. 2020, 14. [Google Scholar] [CrossRef]
- Kaye, J. The regulation of direct-to-consumer genetic tests. Hum. Mol. Genet. 2008, 17, R180–R183. [Google Scholar] [CrossRef]
- Charbonneau, J.; Nicol, D.; Chalmers, D.; Kato, K.; Yamamoto, N.; Walshe, J.; Critchley, C. Public reactions to direct-to-consumer genetic health tests: A comparison across the US, UK, Japan and Australia. Eur. J. Hum. Genet. 2020, 28, 339–348. [Google Scholar] [CrossRef]
- 23andMe. Available online: https://www.23andme.com/ (accessed on 10 December 2020).
- AncestryDNA. Available online: https://www.ancestry.co.uk/dna/ (accessed on 10 December 2020).
- MyoGenes. Personalising Your Health. Available online: https://www.myogenes.com/ (accessed on 10 December 2020).
- MyHeritage. Available online: https://www.myheritage.com/ (accessed on 10 December 2020).
- CloudPharmacy. Coronavirus (COVID-19) IgG/IgM Antibody Test. Available online: https://www.cloudpharmacy.co.uk/medications/coronavirus-antibody-igg-test/ (accessed on 10 December 2020).
- Better2Know. Covid-19 Swab Test. Available online: https://www.better2know.co.uk/shop/products/home-testing-kits/covid-19-test (accessed on 10 December 2020).
- LloydsPharmacy. Coronavirus (COVID-19) Swab Test Kit. Available online: https://onlinedoctor.lloydspharmacy.com/uk/coronavirus-covid-19-testing/covid-19-swab-test-kit (accessed on 10 December 2020).
- EUR-LEX. Commission Implementing Decision (EU) 2020/439 of 24 March 2020 on the Harmonised Standards for In Vitro Diagnostic Medical Devices Drafted in Support of Directive 98/79/EC of the European Parliament and of the Council. 2020. Available online: http://data.europa.eu/eli/dec_impl/2020/439/oj (accessed on 10 December 2020).
- Financial Times. UK Regulator Halts Antibody Home Tests for Coronavirus. Available online: https://www.ft.com/content/aed8d0c4-2e60-46c4-88c9-846f11104f9e (accessed on 10 December 2020).
- Biosciences, S. Sherlock™ CRISPR SARS-CoV-2. Available online: https://sherlock.bio/crispr-sars-cov-2/ (accessed on 14 December 2020).
- MammothBiosciences. A CRISPR-Based Detection Platform for SARS-CoV-2. Available online: https://mammoth.bio/covid/ (accessed on 14 December 2020).
- Luminostics. The Clip COVID Rapid Antigen Test. Available online: https://luminostics.com/ (accessed on 14 December 2020).
- Nicoya. Nicoya Receives over $290K in Funding to Develop a Portable COVID-19 Diagnostic Test. Available online: https://nicoyalife.com/blog/nicoya-covid-19-diagnostic-test/?utm_campaign=COVID-19%20-%20Atlas&utm_source=Website&utm_medium=Website%20-%20Top%20Banner (accessed on 14 December 2020).
- Quer, G.; Radin, J.M.; Gadaleta, M.; Baca-Motes, K.; Ariniello, L.; Ramos, E.; Kheterpal, V.; Topol, E.J.; Steinhubl, S.R. Wearable sensor data and self-reported symptoms for COVID-19 detection. Nat. Med. 2020. [Google Scholar] [CrossRef]
- Ford, J.; Goldstein, T.; Trahan, S.; Neuwirth, A.; Tatoris, K.; Decker, S. A 3D-printed nasopharyngeal swab for COVID-19 diagnostic testing. 3D Print. Med. 2020, 6, 21. [Google Scholar] [CrossRef]
- Callahan, C.J.; Lee, R.; Zulauf, K.E.; Tamburello, L.; Smith, K.P.; Previtera, J.; Cheng, A.; Green, A.; Abdul Azim, A.; Yano, A.; et al. Open Development and Clinical Validation of Multiple 3D-Printed Nasopharyngeal Collection Swabs: Rapid Resolution of a Critical COVID-19 Testing Bottleneck. J. Clin. Microbiol. 2020, 58, e00876-00820. [Google Scholar] [CrossRef]
- US Food and Drug Administration. 3D Printing in FDA’s Rapid Response to COVID-19. Available online: https://www.fda.gov/emergency-preparedness-and-response/coronavirus-disease-2019-covid-19/3d-printing-fdas-rapid-response-covid-19 (accessed on 14 December 2020).
- Walker, H.J.; Burrell, M.M. Could breath analysis by MS could be a solution to rapid, non-invasive testing for COVID-19? Bioanalysis 2020, 12, 1213–1217. [Google Scholar] [CrossRef] [PubMed]
- Chris Brooker. Ohio State Researchers Testing Breathalyzer to Detect COVID-19. Available online: https://news.osu.edu/ohio-state-researchers-testing-breathalyzer-to-detect-covid-19/ (accessed on 14 December 2020).
- IONICON. COVID-19 Detection in Breath. Available online: https://www.ionicon.com/blog/2020/covid-19-detection-in-breath (accessed on 14 December 2020).
- Price, C.P.; Christenson, R.H. Evaluating New Diagnostic Technologies: Perspectives in the UK and US. Clin. Chem. 2008, 54, 1421–1423. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bossuyt, P.M. Testing COVID-19 tests faces methodological challenges. J. Clin. Epidemiol. 2020, 126, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Bossuyt, P.M.M.; Reitsma, J.B.; Linnet, K.; Moons, K.G.M. Beyond Diagnostic Accuracy: The Clinical Utility of Diagnostic Tests. Clin. Chem. 2012, 58, 1636–1643. [Google Scholar] [CrossRef] [PubMed]
- Brownstein, N.C.; Chen, Y.A. Predictive values, uncertainty, and interpretation of serology tests for the novel coronavirus. medRxiv 2020. [Google Scholar] [CrossRef]
- Walley, T. Evaluating laboratory diagnostic tests. BMJ 2008, 336, 569. [Google Scholar] [CrossRef]
- Applied Clinical Trials. Big Changes for EU Medical and In Vitro Diagnostic Device Regulations. Available online: https://www.appliedclinicaltrialsonline.com/view/big-changes-eu-medical-and-vitro-diagnostic-device-regulations (accessed on 14 December 2020).
- PHG Foundation. What is the IVDR? Available online: https://www.phgfoundation.org/briefing/what-is-the-ivdr (accessed on 14 December 2020).
- African Union. Available online: https://au.int/en (accessed on 18 January 2021).
- African Union. Africa CDC. Available online: https://africacdc.org/ (accessed on 18 January 2021).
- African Union. Partnership to Accelerate COVID-19 Testing (PACT) in Africa—Resources. Available online: https://africacdc.org/download/partnership-to-accelerate-covid-19-testing-pact-in-africa/ (accessed on 18 January 2021).
- Centers for Disease Control and Prevention (CDC). Africa CDC Launches Continent-Wide Response. Available online: https://www.cdc.gov/globalhealth/healthprotection/fieldupdates/fall-2020/africa-cdc-covid.html (accessed on 18 January 2021).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oyewole, A.O.; Barrass, L.; Robertson, E.G.; Woltmann, J.; O’Keefe, H.; Sarpal, H.; Dangova, K.; Richmond, C.; Craig, D. COVID-19 Impact on Diagnostic Innovations: Emerging Trends and Implications. Diagnostics 2021, 11, 182. https://doi.org/10.3390/diagnostics11020182
Oyewole AO, Barrass L, Robertson EG, Woltmann J, O’Keefe H, Sarpal H, Dangova K, Richmond C, Craig D. COVID-19 Impact on Diagnostic Innovations: Emerging Trends and Implications. Diagnostics. 2021; 11(2):182. https://doi.org/10.3390/diagnostics11020182
Chicago/Turabian StyleOyewole, Anne O., Lucy Barrass, Emily G. Robertson, James Woltmann, Hannah O’Keefe, Harsimran Sarpal, Kim Dangova, Catherine Richmond, and Dawn Craig. 2021. "COVID-19 Impact on Diagnostic Innovations: Emerging Trends and Implications" Diagnostics 11, no. 2: 182. https://doi.org/10.3390/diagnostics11020182
APA StyleOyewole, A. O., Barrass, L., Robertson, E. G., Woltmann, J., O’Keefe, H., Sarpal, H., Dangova, K., Richmond, C., & Craig, D. (2021). COVID-19 Impact on Diagnostic Innovations: Emerging Trends and Implications. Diagnostics, 11(2), 182. https://doi.org/10.3390/diagnostics11020182








