Human Prion Disorders: Review of the Current Literature and a Twenty-Year Experience of the National Surveillance Center in the Czech Republic
Abstract
1. Background—Human Prion Diseases in Review
1.1. Sporadic Human Prion Diseases
1.1.1. Sporadic Creutzfeldt-Jakob Disease
1.1.2. Sporadic Fatal Insomnia
1.1.3. Variably Protease-Sensitive Prionopathy
1.2. Acquired CJD
1.2.1. Iatrogenic, Accidentally Transmitted CJD
1.2.2. Variant CJD
1.2.3. Kuru
1.3. Inherited Prion Diseases
1.3.1. Genetic CJD
1.3.2. Gerstmann–Sträussler–Scheinker Syndrome
1.3.3. Fatal Familial Insomnia
1.4. Differential Diagnosis of Human Prion Diseases
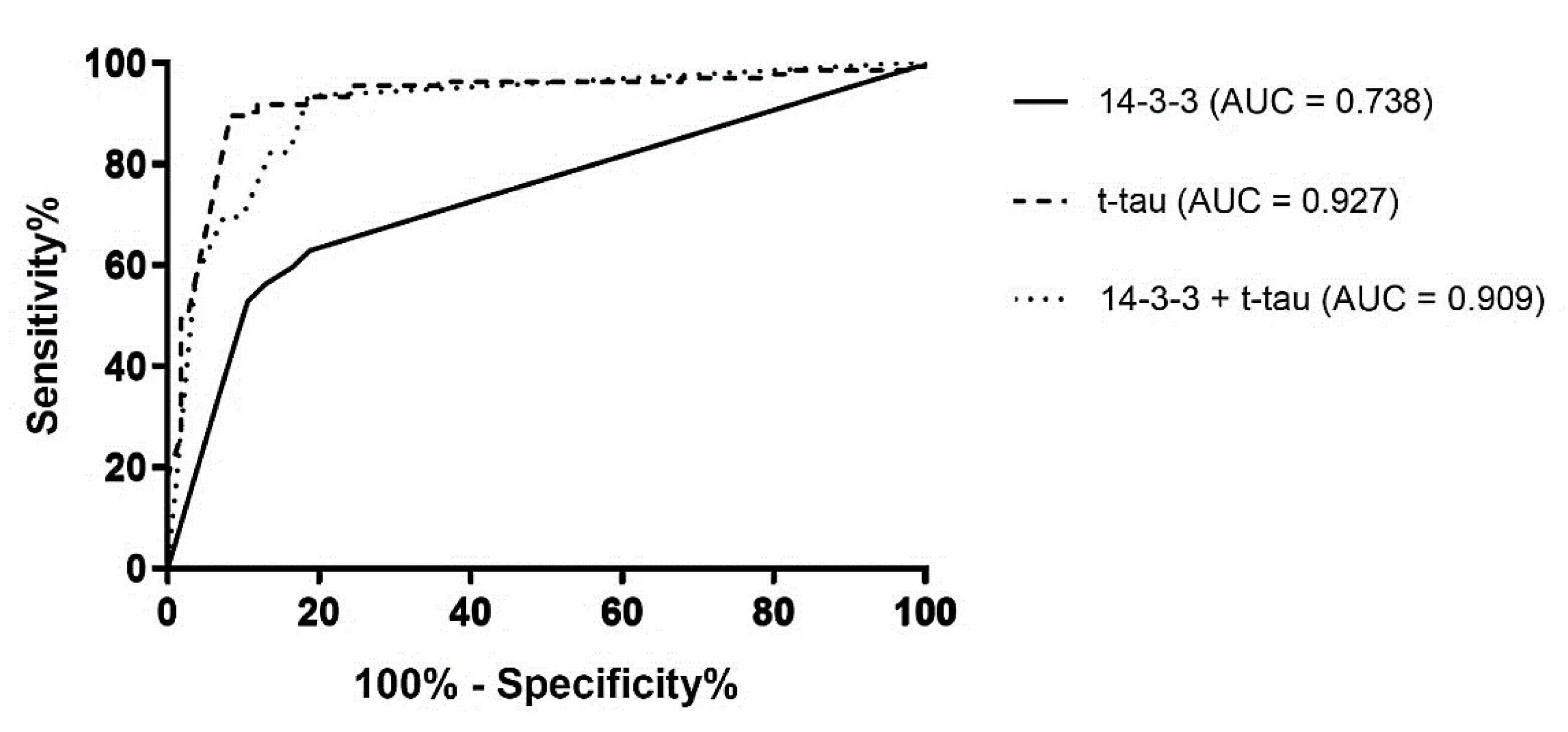
1.5. CSF Biomarkers in TSE
1.5.1. Protein 14-3-3
1.5.2. RT-QuIC
1.6. Definite Diagnosis of Human Prion Diseases
- MM1/MV1:
- Spongiform degeneration: formed by fine vacuoles predominantly affecting corticostriatal-thalamic and cerebellar areas, whereas hippocampal region is relatively spared.
- PrP deposits: prevailing synaptic location.
- MM2/MV2C:
- Spongiform degeneration: large confluent vacuoles.
- PrP deposits: predominant involvement of cortex and subiculum, minor involvement of brainstem with cerebellum.
- MV2K:
- PrP deposits: kuru-like plaques mainly in cerebellar granular layer, plaque-like deposits in other cortical regions.
- VV1:
- Spongiform degeneration: medium-sized vacuoles involving cortex, striatum, beside spared cerebellar region.
- PrP deposits: synaptic pattern.
- VV2:
- Spongiform degeneration: fine or medium-sized vacuoles; more severe involvement of subcortical grey matter by comparison with cerebral neocortex, and of hippocampus plus subiculum by comparison with occipital cortex.
- PrP deposits: cerebellar plaque-like structures and perioneuronal pattern in deep cortical layers and hippocampal region.
- Others: cerebellar atrophy.
- MM2T (sFI):
- Spongiform degeneration: absent in cerebellum.
- Others: moderate to severe selective atrophy of thalamus and olives.
- MVK+C:
- Spongiform degeneration: extensive vacuolarization.
- PrP deposits: same as MV2K (kuru-like plaques mainly in cerebellar granular layer, plaque-like deposits in other cortical regions), in addition with perivacuolar and coarse PrP deposits in the grey matter [113].
1.6.1. Western Blot
Western Blot in Corneal Pre-Transplantation Testing
1.6.2. Immunohistochemistry
1.6.3. Genetic Testing
2. Human Prion Diseases in the Czech Republic 2001–2020—Results from a Nation-Wide Survey
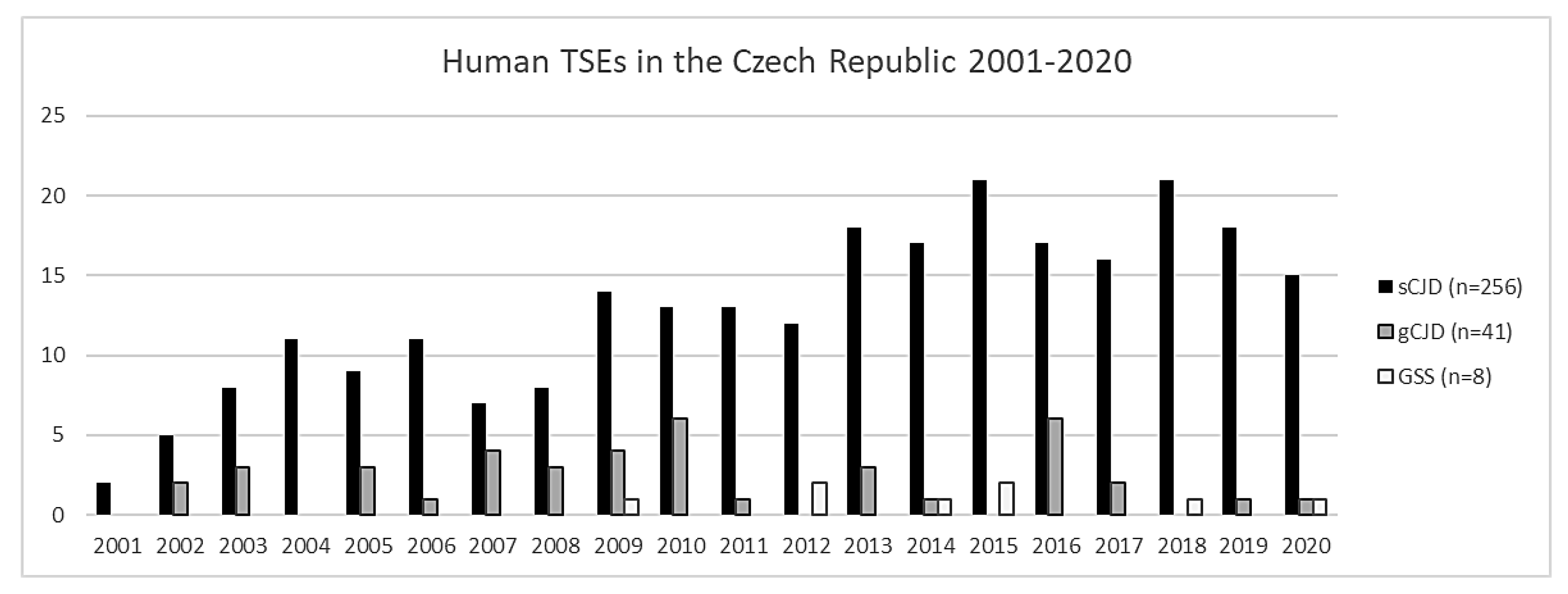
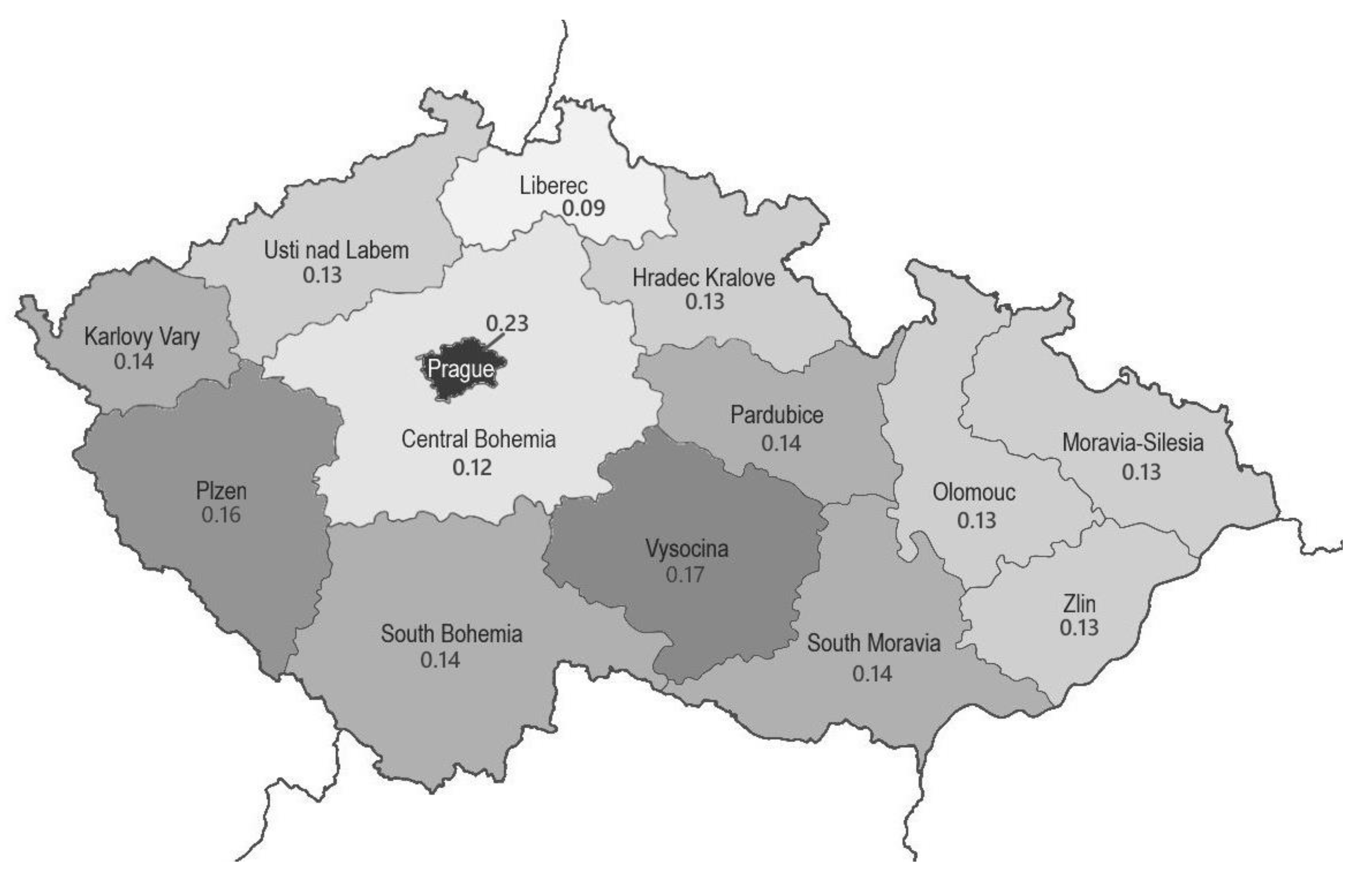
2.1. Numbers of Detected Cases
2.2. CJD in Comorbidity
2.3. Brain Biopsy
3. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Imran, M.; Mahmood, S. An overview of human prion diseases. Virol. J. 2011, 8, 559. [Google Scholar] [CrossRef]
- Asher, D.M.; Gregori, L. Human transmissible spongiform encephalopathies: Historic view. Handb. Clin. Neurol. 2018, 153, 1–17. [Google Scholar] [PubMed]
- Kuwahara, C.; Takeuchi, A.M.; Nishimura, T.; Haraguchi, K.; Kubosaki, A.; Matsumoto, Y.; Saeki, K.; Matsumoto, Y.; Yokoyama, T.; Itohara, S.; et al. Prions prevent neuronal cell-line death. Nature 1999, 400, 225–226. [Google Scholar] [CrossRef]
- Wulf, M.A.; Senatore, A.; Aguzzi, A. The biological function of the cellular prion protein: An update. BMC Biol. 2017, 15, 34. [Google Scholar] [CrossRef] [PubMed]
- Chiesa, R.; Harris, D.A. Fishing for prion protein function. PLoS Biol. 2009, 7, e75. [Google Scholar] [CrossRef] [PubMed]
- Steele, A.D.; Lindquist, S.; Aguzzi, A. The prion protein knockout mouse: A phenotype under challenge. Prion 2007, 1, 83–93. [Google Scholar] [CrossRef]
- Linden, R.; Martins, V.R.; Prado, M.A.; Cammarota, M.; Izquierdo, I.; Brentani, R.R. Physiology of the prion protein. Physiol. Rev. 2008, 88, 673–728. [Google Scholar] [CrossRef]
- McLennan, N.F.; Brennan, P.M.; McNeill, A.; Davies, I.; Fotheringham, A.; Rennison, K.A.; Ritchie, D.; Brannan, F.; Head, M.W.; Ironside, J.W.; et al. Prion protein accumulation and neuro-protection in hypoxic brain damage. Am. J. Pathol. 2004, 165, 227–235. [Google Scholar] [CrossRef]
- Spudich, A.; Frigg, R.; Kilic, E.; Kilic, U.; Oesch, B.; Raeber, A.; Bassetti, C.L.; Hermann, D.M. Aggravation of ischemic brain injury by prion protein deficiency: Role of ERK-1/-2 and STAT-1. Neurobiol. Dis. 2005, 20, 442–449. [Google Scholar] [CrossRef]
- Prusiner, S.B. Novel proteinaceous infectious particles cause scrapie. Science 1982, 216, 136–144. [Google Scholar] [CrossRef]
- Prusiner, S.B. Prions. Proc. Natl. Acad. Sci. USA 1998, 95, 13363–13383. [Google Scholar] [CrossRef] [PubMed]
- Wolschner, C.; Giese, A.; Kretzschmar, H.A.; Huber, R.; Moroder, L.; Budisa, N. Design of anti- and pro-aggregation variants to assess the effects of methionine oxidation in human prion protein. Proc. Natl. Acad. Sci. USA 2009, 106, 7756–7761. [Google Scholar] [CrossRef] [PubMed]
- Nelson, R.; Eisenberg, D. Recent atomic models of amyloid fibril structure. Curr. Opin. Struct. Biol. 2006, 16, 260–265. [Google Scholar] [CrossRef] [PubMed]
- Pastore, A.; Zagari, A. A structural overview of the vertebrate prion proteins. Prion 2007, 1, 185–197. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sikorska, B.; Knight, R.; Ironside, J.W.; Liberski, P.P. Creutzfeldt-Jakob disease. Adv. Exp. Med. Biol. 2012, 724, 76–90. [Google Scholar]
- Vacca, V.M., Jr. CJD: Understanding Creutzfeldt-Jakob disease. Nursing 2016, 46, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Gençer, A.G.; Pelin, Z.; Küçükali, C.İ.; Topçuoğlu, O.B.; Yilmaz, N. Creutzfeldt-Jakob disease. Psychogeriatrics 2011, 11, 119–124. [Google Scholar] [CrossRef]
- Creutzfeld, H.G. Über eine eigenartige herdförmige Erkrankung des Zentralnervensystems. Z. Ges. Neurol. Psychiatr. 1920, 57, 1–18. [Google Scholar] [CrossRef]
- Jakob, A. Über eine eigenartige Erkrankung des Zentral-nervensystems mit bemerkenswertem anatomischem Befunde (spastische pseudosklerotische Encephalomyelopathie mit disseminierten Degenerationsherden). Dtsch. Z. Nervenheilk 1921, 70, 132–146. [Google Scholar]
- Jakob, A. Über eigenartige Erkrankrungen des Zentralnervensystems mit bemerkenswerten anatomischen Befunden (Spastische Pseudosklerose—Enzephalomyelopathie mit disseminierten Degenerationsherden). Z. Ges. Neurol. Psychiatr. 1921, 64, 147–228. [Google Scholar] [CrossRef]
- Jakob, A. Über eine der multiplen Sklerose klinisch nahestehende Erkrankung des Zentralnervensystems (spastische Pseudoskerlose) mit bemerkenswertem anatomischem Befunde. Mitteilung eines vierten Falles. Med. Klin. 1921, 17, 372–376. [Google Scholar]
- Liberski, P.P. Kuru: A journey back in time from papua new Guinea to the neanderthals’ extinction. Pathogens 2013, 2, 472–505. [Google Scholar] [CrossRef]
- Will, R.G.; Ironside, J.W.; Zeidler, M.; Cousens, S.N.; Estibeiro, K.; Alperovitch, A.; Poser, S.; Pocchiari, M.; Hofman, A.; Smith, P.G. A new variant of Creutzfeldt- Jakob disease in the UK. Lancet 1996, 347, 921. [Google Scholar] [CrossRef]
- Hill, A.F.; Desbruslais, M.; Joiner, S.; Sidle, K.C.; Gowland, I.; Collinge, J.; Doey, L.J.; Lantos, P. The same prion strain causes vCJD and BSE. Nature 1997, 389, 448–526. [Google Scholar] [CrossRef]
- The national CJD research and surveillance unit. The University of Edinburgh. Available online: https://www.cjd.ed.ac.uk/surveillance (accessed on 9 September 2021).
- Brandel, J.P.; Vlaicu, M.B.; Culeux, A.; Belondrade, M.; Bougard, D.; Grznarova, K.; Denouel, A.; Plu, I.; Bouaziz-Amar, E.; Seilhean, D.; et al. Variant Creutzfeldt-Jakob Disease Diagnosed 7.5 Years after Occupational Exposure. N. Engl. J. Med. 2020, 383, 83–85. [Google Scholar] [CrossRef]
- France Issues Moratorium on Prion Research after Fatal Brain Disease Strikes Two Lab Workers. Science. Available online: https://www.science.org/news/2021/07/france-issues-moratorium-prion-research-after-fatal-brain-disease-strikes-two-lab (accessed on 9 September 2021).
- Budka, H.; Aguzzi, A.; Brown, P.; Brucher, J.M.; Bugiani, O.; Gullotta, F.; Haltia, N.; Hauw, J.J.; Ironside, J.W.; Jellinger, K.; et al. Neuropathological Diagnostic Criteria for Creutzfeldt-Jakob Disease (CJD) and Other Human Spongiform Encephalopathies (Prion Diseases). Brain Pathol. 1995, 5, 459–466. [Google Scholar] [CrossRef]
- Bell, J.E.; Ironside, J.W. Neuropathology of spongiform encephalopathies in humans. Br. Med. Bull. 1993, 49, 738–777. [Google Scholar] [CrossRef]
- Collinge, J. Variant Creutzfeldt-Jakob disease. Lancet 1999, 354, 317–323. [Google Scholar] [CrossRef]
- Hill, A.F.; Joiner, S.; Wadsworth, J.D.F.; Sidle, K.C.L.; Bell, J.E.; Budka, H.; Ironside, J.W.; Collinge, J. Molecular classification of sporadic Creutzfeldt–Jakob disease. Brain 2003, 126, 1333–1346. [Google Scholar] [CrossRef]
- Parchi, P.; Giese, A.; Capellari, S.; Brown, P.; Schulz-Schaeffer, W.; Windl, O.; Zerr, I.; Budka, H.; Kopp, N.; Piccardo, P.; et al. Classification of sporadic Creutzfeldt-Jakob disease based on molecular and phenotypic analysis of 300 subjects. Ann. Neurol. 1999, 46, 224–233. [Google Scholar] [CrossRef]
- Brown, P.; Cathala, F.; Raubertas, R.F.; Gajdusek, D.C.; Castaigne, P. The epidemiology of Creutzfeldt–Jakob disease: Conclusion of a 15-year investigation in France and review of the world literature. Neurology 1987, 37, 895–904. [Google Scholar] [CrossRef] [PubMed]
- Uttley, L.; Carroll, C.; Wong, R.; Hilton, D.A.; Stevenson, M. Creutzfeldt-Jakob disease: A systematic review of global incidence, prevalence, infectivity, and incubation. Lancet Infect Dis. 2020, 20, e2–e10. [Google Scholar] [CrossRef]
- Manix, M.; Kalakoti, P.; Henry, M.; Thakur, J.; Menger, R.; Guthikonda, B.; Nanda, A. Creutzfeldt-Jakob disease: Updated diagnostic criteria, treatment algorithm, and the utility of brain biopsy. Neurosurg. Focus 2015, 39, E2. [Google Scholar] [CrossRef]
- Watson, N.; Brandel, J.P.; Green, A.; Hermann, P.; Ladogana, A.; Lindsay, T.; Mackenzie, J.; Pocchiari, M.; Smith, C.; Zerr, I.; et al. The importance of ongoing international surveillance for Creutzfeldt–Jakob disease. Nat. Rev. Neurol. 2021, 17, 362–379. [Google Scholar] [CrossRef]
- Rudge, P.; Hyare, H.; Green, A.; Collinge, J.; Mead, S. Imaging and CSF analyses effectively distinguish CJD from its mimics. J. Neurol. Neurosurg. Psychiatry 2018, 89, 461–466. [Google Scholar] [CrossRef]
- Bizzi, A.; Pascuzzo, R.; Blevins, J.; Grisoli, M.; Lodi, R.; Moscatelli, M.E.M.; Castelli, G.; Cohen, M.L.; Schonberger, L.B.; Foutz, A.; et al. Evaluation of a New Criterion for Detecting Prion Disease with Diffusion Magnetic Resonance Imaging. JAMA Neurol. 2020, 77, 1141–1149. [Google Scholar] [CrossRef]
- Wieser, H.G.; Schindler, K.; Zumsteg, D. EEG in Creutzfeldt-Jakob disease. Clin Neurophysiol. 2006, 117, 935–951. [Google Scholar] [CrossRef]
- Steinhoff, B.J.; Räcker, S.; Herrendorf, G.; Poser, S.; Grosche, S.; Zerr, I.; Kretzschmar, H.; Weber, T. Accuracy and reliability of periodic sharp wave complexes in Creutzfeldt-Jakob disease. Arch. Neurol. 1996, 53, 162–166. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention: CDC’s Diagnostic Criteria for Creutzfeldt-Jakob Disease (CJD). 2018. Available online: https://www.cdc.gov/prions/cjd/diagnostic-criteria.html (accessed on 6 January 2021).
- Mader, E.C., Jr.; El-Abassi, R.; Villemarette-Pittman, N.R.; Santana-Gould, L.; Olejniczak, P.W.; England, J.D. Sporadic Creutzfeldt-Jakob disease with focal findings: Caveats to current diagnostic criteria. Neurol. Int. 2013, 5, e1. [Google Scholar] [CrossRef]
- Heinemann, U.; Krasnianski, A.; Meissner, B.; Kallenberg, K.; Kretzschmar, H.A.; Schulz-Schaeffer, W. Brain biopsy in patients with suspected Creutzfeldt-Jakob disease. J. Neurosurg. 2008, 109, 735–741. [Google Scholar] [CrossRef] [PubMed]
- Cracco, L.; Appleby, B.S.; Gambetti, P. Chapter 15: Fatal familial insomnia and sporadic fatal insomnia. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2018; Volume 153, pp. 175–190. ISSN 0072-9752. ISBN 9780444639455. [Google Scholar] [CrossRef]
- Lugaresi, E.; Medori, R.; Montagna, P.; Baruzzi, A.; Cortelli, P.; Lugaresi, A.; Tinuper, P.; Zucconi, M.; Gambetti, P. Fatal familial insomnia and dysautonomia with selective degeneration of thalamic nuclei. N. Engl. J Med. 1986, 315, 997–1003. [Google Scholar] [CrossRef] [PubMed]
- Montagna, P.; Gambetti, P.; Cortelli, P.; Lugaresi, E. Familial and sporadic fatal insomnia. Lancet Neurol. 2003, 2, 167–176. [Google Scholar] [CrossRef]
- Mehta, L.R.; Huddleston, B.J.; Skalabrin, E.J.; Burns, J.B.; Zou, W.Q.; Gambetti, P.; Chin, S.S. Sporadic fatal insomnia masquerading as a paraneoplastic cerebellar syndrome. Arch Neurol. 2008, 65, 971–973. [Google Scholar] [CrossRef][Green Version]
- Moda, F.; Suardi, S.; Di Fede, G.; Indaco, A.; Limido, L.; Vimercati, C.; Ruggerone, M.; Campagnani, I.; Langeveld, J.; Terruzzi, A.; et al. MM2-thalamic Creutzfeldt-Jakob disease: Neuropathological, biochemical and transmission studies identify a distinctive prion strain. Brain Pathol. 2012, 22, 662–669. [Google Scholar] [CrossRef]
- Piao, Y.S.; Kakita, A.; Watanabe, H.; Kitamoto, T.; Takahashi, H. Sporadic fatal insomnia with spongiform degeneration in the thalamus and widespread PrPSc deposits in the brain. Neuropathology 2005, 25, 144–149. [Google Scholar] [CrossRef]
- Parchi, P.; Capellari, S.; Chin, S.; Schwarz, H.B.; Schecter, N.P.; Butts, J.D.; Hudkins, P.; Burns, D.K.; Powers, J.M.; Gambetti, P. A subtype of sporadic prion disease mimicking fatal familial insomnia. Neurology 1999, 52, 1757–1763. [Google Scholar] [CrossRef]
- Mastrianni, J.A.; Nixon, R.; Layzer, R.; Telling, G.C.; Han, D.; DeArmond, S.J. Prion protein conformation in a patient with sporadic fatal insomnia. N. Engl. J. Med. 1999, 340, 1630–1638. [Google Scholar] [CrossRef]
- Gambetti, P.; Dong, Z.; Yuan, J.; Xiao, X.; Zheng, M.; Alshekhlee, A.; Castellani, R.; Cohen, M.; Barria, M.A.; Gonzalez-Romero, D.; et al. A novel human disease with abnormal prion protein sensitive to protease. Ann. Neurol. 2008, 63, 697–708. [Google Scholar] [CrossRef]
- Notari, S.; Appleby, B.S.; Gambetti, P. Chapter 10: Variably protease-sensitive prionopathy. In Handbook of Clinical Neurology; Pocchiari, M., Manson, J., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; Volume 153, pp. 175–190. ISSN 0072-9752. ISBN 9780444639455. [Google Scholar] [CrossRef]
- Zou, W.Q.; Puoti, G.; Xiao, X.; Yuan, J.; Qing, L.; Cali, I.; Shimoji, M.; Langeveld, J.P.; Castellani, R.; Notari, S.; et al. Variably protease-sensitive prionopathy: A new sporadic disease of the prion protein. Ann. Neurol. 2010, 68, 162–172. [Google Scholar] [CrossRef] [PubMed]
- Aizpurua, M.; Selvackadunco, S.; Yull, H.; Kipps, C.M.; Ironside, J.W.; Bodi, I. Variably protease-sensitive prionopathy mimicking frontotemporal dementia. Neuropathology 2019, 39, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Will, R.G. Acquired prion disease: Iatrogenic CJD, variant CJD, kuru. Br. Med. Bulletin. 2003, 66, 255–265. [Google Scholar] [CrossRef] [PubMed]
- Moffatt, S.L.; Pollock, G. Creutzfeldt-Jakob disease: Perceptions and realities of risk. Clin Exp. Ophthalmol. 2006, 34, 635–636. [Google Scholar] [CrossRef] [PubMed]
- Bonda, D.J.; Manjila, S.; Mehndiratta, P.; Khan, F.; Miller, B.R.; Onwuzulike, K.; Puoti, G.; Cohen, M.L.; Schonberger, L.B.; Cali, I. Human prion diseases: Surgical lessons learned from iatrogenic prion transmission. Neurosurg. Focus. 2016, 41, E10. [Google Scholar] [CrossRef] [PubMed]
- Brown, P.; Brandel, J.P.; Sato, T.; Nakamura, Y.; MacKenzie, J.; Will, R.G. Iatrogenic Creutzfeldt-Jakob disease, final assessment. Emerg. Infect Dis. 2012, 18, 901–907. [Google Scholar] [CrossRef] [PubMed]
- Will, R.G.; Matthews, W.B. Evidence for case-to-case transmission of Creutzfeldt-Jakob disease. J. Neurol. Neurosurg. Psychiatry 1982, 45, 235–238. [Google Scholar] [CrossRef]
- Hilton, D.A. Pathogenesis and prevalence of variant Creutzfeldt-Jakob disease. J. Pathol. 2006, 208, 134–141. [Google Scholar] [CrossRef]
- Sikorska, B.; Liberski, P.P.; Sobów, T.; Budka, H.; Ironside, J.W. Ultrastructural study of florid plaques in variant Creutzfeldt-Jakob disease: A comparison with amyloid plaques in kuru, sporadic Creutzfeldt-Jakob disease and Gerstmann-Sträussler-Scheinker disease. Neuropathol. Appl. Neurobiol. 2009, 35, 46–59. [Google Scholar] [CrossRef]
- Zeidler, M.; Sellar, R.J.; Collie, D.A.; Knight, R.; Stewart, G.; Macleod, M.A.; Ironside, J.W.; Cousens, S.; Colchester, A.C.; Hadley, D.M.; et al. The pulvinar sign on magnetic resonance imaging in variant Creutzfeldt-Jakob disease. Lancet 2000, 355, 1412–1418. [Google Scholar] [CrossRef]
- Hill, A.F.; Butterworth, R.J.; Joiner, S.; Jackson, G.; Rossor, M.N.; Thomas, D.J.; Frosh, A.; Tolley, N.; Bell, J.E.; Spencer, M.; et al. Investigation of variant Creutzfeldt-Jakob disease and other human prion diseases with tonsil biopsy samples. Lancet 1999, 353, 183–189. [Google Scholar] [CrossRef]
- Mok, T.; Jaunmuktane, Z.; Joiner, S.; Campbell, T.; Morgan, C.; Wakerley, B.; Golestani, F.; Rudge, P.; Mead, S.; Jäger, H.R.; et al. Variant Creutzfeldt-Jakob Disease in a Patient with Heterozygosity at PRNP Codon 129. N. Engl. J. Med. 2017, 376, 292–294. [Google Scholar] [CrossRef]
- Collinge, J. Molecular neurology of prion disease. J. Neurol. Neurosurg. Psychiatry 2005, 76, 906–919. [Google Scholar] [CrossRef]
- Head, M.W.; Bunn, T.J.R.; Bishop, M.T.; McLoughlin, V.; Lowrie, S.; McKimmie, C.S.; Williams, M.C.; McCardle, L.; Mackenzie, J.; Knight, R.; et al. Prion protein heterogeneity in sporadic but not variant Creutzfeldt-Jakob disease: United Kingdom cases 1991–2002. Ann. Neurol. 2004, 55, 851–859. [Google Scholar] [CrossRef]
- Llewelyn, C.A.; Hewitt, P.E.; Knight, R.S.; Amar, K.; Cousens, S.; Mackenzie, J. Possible transmission of variant Creutzfeldt-Jakob disease by blood transfusion. Lancet 2004, 363, 417–421. [Google Scholar] [CrossRef]
- Gajdusek, D.C.; Zigas, V. Degenerative disease of the central nervous system in New Guinea. The endemic occurrence of “kuru” in the native population. N. Engl. J. Med. 1957, 257, 974–978. [Google Scholar] [CrossRef] [PubMed]
- Alpers, M.P. The epidemiology of kuru: Monitoring the epidemic from its peak to the end. Philos. Trans. R. Soc. B 2008, 363, 3707–3713. [Google Scholar] [CrossRef] [PubMed]
- Beasley, A. The promised medicine: Fore reflections on the scientific investigation of kuru. Oceania 2006, 76, 186–202. [Google Scholar] [CrossRef]
- Liberski, P.P.; Gajos, A.; Sikorska, B.; Lindenbaum, S. Kuru, the First Human Prion Disease. Viruses 2019, 11, 232. [Google Scholar] [CrossRef] [PubMed]
- Le Bouvier, G. Slow, Latent, and Temperate Virus Infections. Yale J. Biol. Med. 1967, 39, 83–84. [Google Scholar]
- Beck, E.; Daniel, P.M. Prion diseases from a neuropathologist’s perspective. In Prion Diseases of Humans and Animals; Prusiner, S.B., Collinge, J., Powell, J., Anderton, B., Eds.; Ellis Horwood: New York, NY, USA; London, UK; Toronto, ON, Canada; Sydney, Australia; Singapore, 1993; pp. 63–65. [Google Scholar]
- Ladogana, A.; Kovacs, G.G. Chapter 13: Genetic Creutzfeldt-Jakob disease. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2018; Volume 153, pp. 219–242. ISSN 0072-9752. ISBN 9780444639455. [Google Scholar] [CrossRef]
- Gao, L.P.; Shi, Q.; Xiao, K.; Wang, J.; Zhou, W.; Chen, C.; Dong, X.P. The genetic Creutzfeldt-Jakob disease with E200K mutation: Analysis of clinical, genetic and laboratory features of 30 Chinese patients. Sci. Rep. 2019, 9, 1836. [Google Scholar] [CrossRef] [PubMed]
- Kovács, G.G.; Puopolo, M.; Ladogana, A.; Pocchiari, M.; Budka, H.; van Duijn, C.; Collins, S.J.; Boyd, A.; Giulivi, A.; Coulthart, M.; et al. EUROCJD. Genetic prion disease: The EUROCJD experience. Hum. Genet. 2005, 118, 166–174. [Google Scholar] [CrossRef]
- Rohan, Z.; Rusina, R.; Marešová, M.; Matěj, R. Lidská prionová onemocnění v České republice. Epidemiol. Mikrobiol. Imunol. 2015, 64, 115–120. [Google Scholar] [PubMed]
- Nozaki, I.; Hamaguchi, T.; Sanjo, N.; Noguchi-Shinohara, M.; Sakai, K.; Nakamura, Y.; Sato, T.; Kitamoto, T.; Mizusawa, H.; Moriwaka, F.; et al. Prospective 10-year surveillance of human prion diseases in Japan. Brain 2010, 133, 3043–3057. [Google Scholar] [CrossRef] [PubMed]
- Chapman, J.; Ben-Israel, J.; Goldhammer, Y.; Korczyn, A.D. The risk of developing Creutzfeldt-Jakob disease in subjects with the PRNP gene codon 200 point mutation. Neurology 1994, 44, 1683–1686. [Google Scholar] [CrossRef] [PubMed]
- Liberski, P.P. Gerstmann-Sträussler-Scheinker disease. Adv. Exp. Med. Biol. 2012, 724, 128–137. [Google Scholar]
- Gambetti, P.; Kong, Q.; Zou, W.; Parchi, P.; Chen, S.G. Sporadic and familial CJD: Classification and characterisation. Br. Med. Bull. 2003, 66, 213–239. [Google Scholar] [CrossRef] [PubMed]
- Ghetti, B.; Tagliavini, F.; Takao, M.; Bugiani, O.; Piccardo, P. Hereditary prion protein amyloidoses. Clin. Lab. Med. 2003, 23, 65–85. [Google Scholar] [CrossRef]
- Galatioto, S.; Ruggeri, D.; Gullotta, F. Gerstmann-Sträussler-Scheinker syndrome in a Sicilian patient. Neuropathological aspects. Pathologica 1995, 87, 659–665. [Google Scholar]
- Keller, J.; Eliasova, I.; Parobkova, E.; Smetakova, M.; Musova, Z.; Rusina, R. Clinical Variability in P102L Gerstmann-Sträussler-Scheinker Syndrome. Ann. Neurol. 2019, 86, 643–652. [Google Scholar] [CrossRef]
- Jansen, C.; Voet, W.; Head, M.W.; Parchi, P.; Yull, H.; Verrips, A.; Wesseling, P.; Meulstee, J.; Baas, F.; van Gool, W.A.; et al. A novel seven-octapeptide repeat insertion in the prion protein gene (PRNP) in a Dutch pedigree with Gerstmann-Sträussler-Scheinker disease phenotype: Comparison with similar cases from the literature. Acta Neuropathol. 2011, 121, 59–68. [Google Scholar] [CrossRef]
- Medori, R.; Tritschler, H.J.; LeBlanc, A.; Villare, F.; Manetto, V.; Chen, H.Y.; Xue, R.; Leal, S.; Montagna, P.; Cortelli, P.; et al. Fatal familial insomnia, a prion disease with a mutation at codon 178 of the prion protein gene. N. Engl. J. Med. 1992, 326, 444–449. [Google Scholar] [CrossRef]
- Llorens, F.; Zarranz, J.J.; Fischer, A.; Zerr, I.; Ferrer, I. Fatal Familial Insomnia: Clinical Aspects and Molecular Alterations. Curr Neurol. Neurosci. Rep. 2017, 17, 30. [Google Scholar] [CrossRef]
- Geschwind, M.D.; Shu, H.; Haman, A.; Sejvar, J.J.; Miller, B.L. Rapidly progressive dementia. Ann. Neurol. 2008, 64, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Chitravas, N.; Jung, R.S.; Kofskey, D.M.; Blevins, J.E.; Gambetti, P.; Leigh, R.J.; Cohen, M.L. Treatable neurological disorders misdiagnosed as Creutzfeldt–Jakob disease. Ann. Neurol. 2011, 70, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Poser, S.; Mollenhauer, B.; Kraubeta, A.; Zerr, I.; Steinhoff, B.J.; Schroeter, A.; Finkenstaedt, M.; Schulz-Schaeffer, W.J.; Kretzschmar, H.A.; Felgenhauer, K. How to improve the clinical diagnosis of Creutzfeldt-Jakob disease. Brain 1999, 122, 2345–2351. [Google Scholar] [CrossRef] [PubMed]
- Saiz, A.; Graus, F.; Dalmau, J.; Pifarré, A.; Marin, C.; Tolosa, E. Detection of 14-3-3 brain protein in the cerebrospinal fluid of patients with paraneoplastic neurological disorders. Ann. Neurol. 1999, 46, 774–777. [Google Scholar] [CrossRef]
- Fermo, O.P.; Izbudak, I.; Sutter, R.; Venkatesan, A.; Kaplan, P.W.; Probasco, J.C. Autoimmune encephalitis mimicking Creutzfeldt–Jakob disease. Neurol. Clin. Pract. 2014, 4, 493–495. [Google Scholar] [CrossRef]
- World Health Organization. WHO Manual for Surveillance of Human Transmissible Spongiform Encephalopathies, Including Variant Creutzfeldt-Jakob Disease. World Health Organization. 2003. Available online: https://apps.who.int/iris/handle/10665/42656 (accessed on 3 February 2021).
- Gauthier, A.C.; Baehring, J.M. Hashimoto’s encephalopathy mimicking Creutzfeldt–Jakob disease. J. Clin. Neurosci. 2017, 35, 72–73. [Google Scholar] [CrossRef]
- Mead, S.; Rudge, P. CJD mimics and chameleons. Pract. Neurol. 2017, 17, 113–121. [Google Scholar] [CrossRef]
- Lanata, S.; Gambetti, P.; Geschwind, M. Wernicke-Korsakoff Syndrome Mimicking Sporadic Jakob-Creutzfeldt Disease. Neurology. 2014, 82, P5.235. [Google Scholar]
- Rusina, R.; Krajcovicova, L.; Srpova, B.; Matěj, R. Metastatic intracranial spread of adenocarcinoma mimicking Creutzfeldt-Jakob disease in two cases. Rev. Neurol. 2021. In press. [Google Scholar] [CrossRef]
- Toledo, J.B.; Brettschneider, J.; Grossman, M.; Arnold, S.E.; Hu, W.T.; Xie, S.X.; Lee, V.M.; Shaw, L.M.; Trojanowski, J.Q. CSF biomarkers cutoffs: The importance of coincident neuropathological diseases. Acta Neuropathol. 2012, 124, 23–35. [Google Scholar] [CrossRef]
- Foote, M.; Zhou, Y. 14-3-3 proteins in neurological disorders. Int. J. Biochem. Mol. Biol. 2012, 3, 152–164. [Google Scholar] [PubMed]
- Satoh, K.; Tobiume, M.; Matsui, Y.; Mutsukura, K.; Nishida, N.; Shiga, Y.; Eguhchi, K.; Shirabe, S.; Sata, T. Establishment of a standard 14-3-3 protein assay of cerebrospinal fluid as a diagnostic tool for Creutzfeldt-Jakob disease. Lab. Investig. 2010, 90, 1637–1644. [Google Scholar] [CrossRef]
- Cuadrado-Corrales, N.; Jiménez-Huete, A.; Albo, C.; Hortigüela, R.; Vega, L.; Cerrato, L.; Sierra-Moros, M.; Rábano, A.; de Pedro-Cuesta, J.; Calero, M. Impact of the clinical context on the 14-3-3 test for the diagnosis of sporadic CJD. BMC Neurol. 2006, 6, 25. [Google Scholar] [CrossRef] [PubMed]
- Skillbäck, T.; Rosén, C.; Asztely, F.; Mattsson, N.; Blennow, K.; Zetterberg, H. Diagnostic performance of cerebrospinal fluid total tau and phosphorylated tau in Creutzfeldt-Jakob disease: Results from the Swedish Mortality Registry. JAMA Neurol. 2014, 71, 476–483. [Google Scholar] [CrossRef] [PubMed]
- Rohan, Z.; Smetakova, M.; Kukal, J.; Rusina, R.; Matěj, R. Proteinase-activated receptor 2 and disease biomarkers in cerebrospinal fluid in cases with autopsy-confirmed prion diseases and other neurodegenerative diseases. BMC Neurol. 2015, 31, 50. [Google Scholar] [CrossRef]
- Bruzova, M.; Rusina, R.; Stejskalova, Z.; Matěj, R. Autopsy-diagnosed neurodegenerative dementia cases support the use of cerebrospinal fluid protein biomarkers in the diagnostic work-up. Sci. Rep. 2021. In press. [Google Scholar] [CrossRef]
- Green, A.J.E. RT-QuIC: A new test for sporadic CJD. Pract. Neurol. 2019, 19, 49–55. [Google Scholar] [CrossRef]
- Orrú, C.D.; Groveman, B.R.; Hughson, A.G.; Zanusso, G.; Coulthart, M.B.; Caughey, B. Rapid and sensitive RT-QuIC detection of human Creutzfeldt-Jakob disease using cerebrospinal fluid. mBio 2015, 6, e02451-14. [Google Scholar] [CrossRef]
- Wilham, J.M.; Orrú, C.D.; Bessen, R.A.; Atarashi, R.; Sano, K.; Race, B.; Meade-White, K.D.; Taubner, L.M.; Timmes, A.; Caughey, B. Rapid end-point quantitation of prion seeding activity with sensitivity comparable to bioassays. PLoS Pathog. 2021, 6, e1001217. [Google Scholar] [CrossRef]
- Bongianni, M.; Orrù, C.; Groveman, B.R.; Sacchetto, L.; Fiorini, M.; Tonoli, G.; Triva, G.; Capaldi, S.; Testi, S.; Ferrari, S.; et al. Diagnosis of Human Prion Disease Using Real-Time Quaking-Induced Conversion Testing of Olfactory Mucosa and Cerebrospinal Fluid Samples. JAMA Neurol. 2017, 74, 155–162. [Google Scholar] [CrossRef]
- Foutz, A.; Appleby, B.S.; Hamlin, C.; Liu, X.; Yang, S.; Cohen, Y.; Chen, W.; Blevins, J.; Fausett, C.; Wang, H.; et al. Diagnostic and prognostic value of human prion detection in cerebrospinal fluid. Ann Neurol. 2017, 81, 79–92. [Google Scholar] [CrossRef]
- Franceschini, A.; Baiardi, S.; Hughson, A.G.; McKenzie, N.; Moda, F.; Rossi, M.; Capellari, S.; Green, A.; Giaccone, G.; Caughey, B.; et al. High diagnostic value of second generation CSF RT-QuIC across the wide spectrum of CJD prions. Sci. Rep. 2017, 7, 10655. [Google Scholar] [CrossRef]
- Parchi, P.; Strammiello, R.; Notari, S.; Giese, A.; Langeveld, J.P.; Ladogana, A.; Zerr, I.; Roncaroli, F.; Cras, P.; Ghetti, B.; et al. Incidence and spectrum of sporadic Creutzfeldt-Jakob disease variants with mixed phenotype and co-occurrence of PrPSc types: An updated classification. Acta Neuropathol. 2009, 118, 659–671. [Google Scholar] [CrossRef] [PubMed]
- Parchi, P.; de Boni, L.; Saverioni, D.; Cohen, M.L.; Ferrer, I.; Gambetti, P.; Gelpi, E.; Giaccone, G.; Hauw, J.J.; Höftberger, R.; et al. Consensus classification of human prion disease histotypes allows reliable identification of molecular subtypes: An inter-rater study among surveillance centres in Europe and USA. Acta Neuropathol. 2012, 124, 517–529. [Google Scholar] [CrossRef] [PubMed]
- Armitage, W.; Tullo, A.; Ironside, J. Risk of Creutzfeldt–Jakob disease transmission by ocular surgery and tissue transplantation. Eye 2009, 23, 1926–1930. [Google Scholar] [CrossRef]
- Mehta, J.S.; Franks, W.A. The sclera, the prion, and the ophthalmologist. Br. J. Ophthalmol. 2002, 86, 587–592. [Google Scholar] [CrossRef] [PubMed]
- Sbírka Zákonů: Předpis 97/2019 Sb. Available online: https://www.psp.cz/sqw/sbirka.sqw?cz=97&r=2019 (accessed on 15 January 2021).
- Jirsova, K.; Krabcova, I.; Novakova, J.; Hnathova, I.; Koukolik, F.; Kubesova, B.; Netukova, M.; Matěj, R. The assessment of pathogenic prions in the brains of eye tissue donors Cesk Slov Neurol: 2-years experience in the Czech Republic. Cornea 2010, 29, 996–999. [Google Scholar] [CrossRef] [PubMed]
- Mitrová, E.; Belay, G. Creutzfeldt-Jakob disease with E200K mutation in Slovakia: Characterization and development. Acta Virol. 2002, 46, 31–39. [Google Scholar] [PubMed]
- Mitrová, E.; Cernák, A.; Slivarichová, D.; Koščová, S.; Bernovská, V.; Cernák, M. Experience with preventive genetic testing of corneal donors in slovakia. Cornea 2011, 30, 987–990. [Google Scholar] [CrossRef]
- Sikora, J.; Srbová, A.; Koukolík, F.; Matěj, R. Retrospective sequence analysis of the human PRNP gene from the formaldehyde-fixed paraffin-embedded tissues: Report of two cases of Creutzfeldt-Jakob disease. Folia Microbiol. 2006, 51, 619–625. [Google Scholar] [CrossRef]
- Rusina, R.; Fiala, J.; Holada, K.; Matějčková, M.; Nováková, J.; Ampapa, R.; Koukolík, F.; Matěj, R. Gerstmann-Sträussler-Scheinker syndrome with the P102L pathogenic mutation presenting as familial Creutzfeldt-Jakob disease: A case report and review of the literature. Neurocase 2013, 19, 41–53. [Google Scholar] [CrossRef]
- Matěj, R.; Kovacs, G.G.; Johanidesová, S.; Keller, J.; Matějčková, M.; Nováková, J.; Sigut, V.; Keller, O.; Rusina, R. Genetic Creutzfeldt-Jakob disease with R208H mutation presenting as progressive supranuclear palsy. Mov. Disord. 2012, 27, 476–479. [Google Scholar] [CrossRef] [PubMed]
- Rohan, Z.; Parobkova, E.; Johanidesova, S.; Koukolik, F.; Matěj, R.; Rusina, R. Human Prion Diseases in the Czech Republic—10 Years of Experience with the Diagnosis. Cesk. Slov. Neurol. Neurochir. 2013, 76, 300–306. [Google Scholar]
- Klug, G.M.; Wand, H.; Simpson, M.; Boyd, A.; Law, M.; Masters, C.L.; Matěj, R.; Howley, R.; Farrell, M.; Breithaupt, M.; et al. Intensity of human prion disease surveillance predicts observed disease incidence. J. Neurol. Neurosurg. Psychiatry 2013, 84, 1372–1377. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, G.G.; Rahimi, J.; Ströbel, T.; Lutz, M.I.; Regelsberger, G.; Streichenberger, N.; Perret-Liaudet, A.; Höftberger, R.; Liberski, P.P.; Budka, H.; et al. Tau pathology in Creutzfeldt-Jakob disease revisited. Brain Pathol. 2017, 27, 332–344. [Google Scholar] [CrossRef] [PubMed]
- Parobkova, E.; van der Zee, J.; Dillen, L.; Van Broeckhoven, C.; Rusina, R.; Matěj, R. Sporadic Creutzfeldt-Jakob Disease and Other Proteinopathies in Comorbidity. Front. Neurol. 2020, 11, 596108. [Google Scholar] [CrossRef] [PubMed]
| Possible Creutzfeldt–Jakob disease: Rapidly progressive dementia with at least two of the following symptoms:
|
Probable Creutzfeldt–Jakob disease: Fulfilled criteria for possible CJD with:
|
| Definite Creutzfeldt–Jakob disease: Progressive neurological syndrome and either neuropathological, immunocytochemical or biochemical confirmation. |
Probable iatrogenic Creutzfeldt–Jakob disease diagnosis:
|
Definite iatrogenic Creutzfeldt–Jakob disease diagnosis:
|
Suspected Variant CJD:
|
| Definite Variant CJD: Neuropathologic examination of brain tissue is required to confirm a diagnosis of Variant CJD. The following confirmatory features should be present.
|
Probable genetic Creutzfeldt–Jakob disease diagnosis:
|
Definite genetic Creutzfeldt–Jakob disease diagnosis:
|
| Neurodegenerative disorders (usually comorbid) | Alzheimer’s disease | 88 |
| Frontotemporal dementia | 66 | |
| Dementia with Lewy bodies | 27 | |
| Progressive supranuclear palsy | 8 | |
| Multiple system atrophy | 4 | |
| Corticobasal degeneration | 2 | |
| Parkinson disease | 1 | |
| Neuroinfection and autoimmune diseases | Encephalitis | 20 |
| Malignant multiple sclerosis (Marburg variant) | 2 | |
| Ischemic and anoxic conditions | Subcortical vascular dementia | 7 |
| Post-anoxic encephalopathy | 7 | |
| Tumors | Primary CNS lymphoma | 5 |
| Gliomatosis cerebri | 1 | |
| Meningeal carcinomatosis | 1 | |
| Metastatic carcinoma | 1 | |
| Metabolic encephalopathy | Wernicke-Korsakoff | 5 |
| Others | Subdural hematoma | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jankovska, N.; Rusina, R.; Bruzova, M.; Parobkova, E.; Olejar, T.; Matej, R. Human Prion Disorders: Review of the Current Literature and a Twenty-Year Experience of the National Surveillance Center in the Czech Republic. Diagnostics 2021, 11, 1821. https://doi.org/10.3390/diagnostics11101821
Jankovska N, Rusina R, Bruzova M, Parobkova E, Olejar T, Matej R. Human Prion Disorders: Review of the Current Literature and a Twenty-Year Experience of the National Surveillance Center in the Czech Republic. Diagnostics. 2021; 11(10):1821. https://doi.org/10.3390/diagnostics11101821
Chicago/Turabian StyleJankovska, Nikol, Robert Rusina, Magdalena Bruzova, Eva Parobkova, Tomas Olejar, and Radoslav Matej. 2021. "Human Prion Disorders: Review of the Current Literature and a Twenty-Year Experience of the National Surveillance Center in the Czech Republic" Diagnostics 11, no. 10: 1821. https://doi.org/10.3390/diagnostics11101821
APA StyleJankovska, N., Rusina, R., Bruzova, M., Parobkova, E., Olejar, T., & Matej, R. (2021). Human Prion Disorders: Review of the Current Literature and a Twenty-Year Experience of the National Surveillance Center in the Czech Republic. Diagnostics, 11(10), 1821. https://doi.org/10.3390/diagnostics11101821






