Capsule Endoscopy in Crohn’s Disease—From a Relative Contraindication to Habitual Monitoring Tool
Abstract
:1. Introduction
2. CE—The Early Days
3. Current Data
4. Diagnostic Yield of VCE
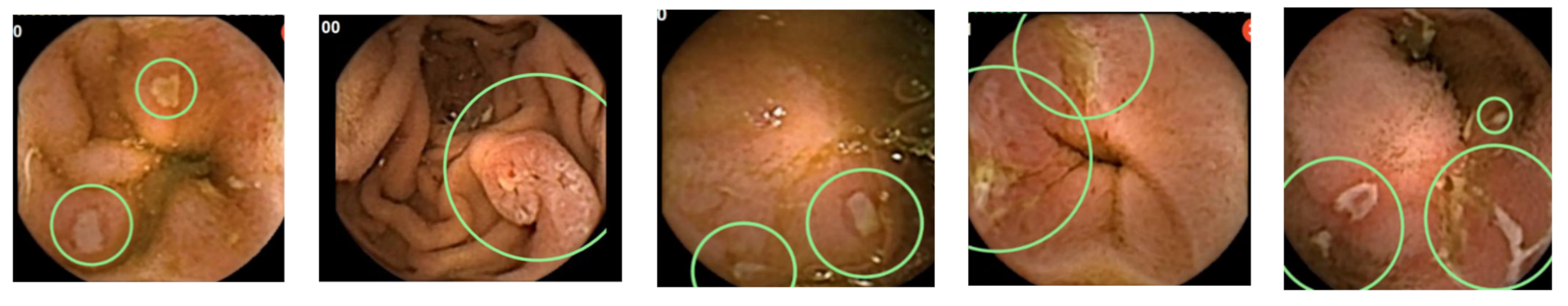
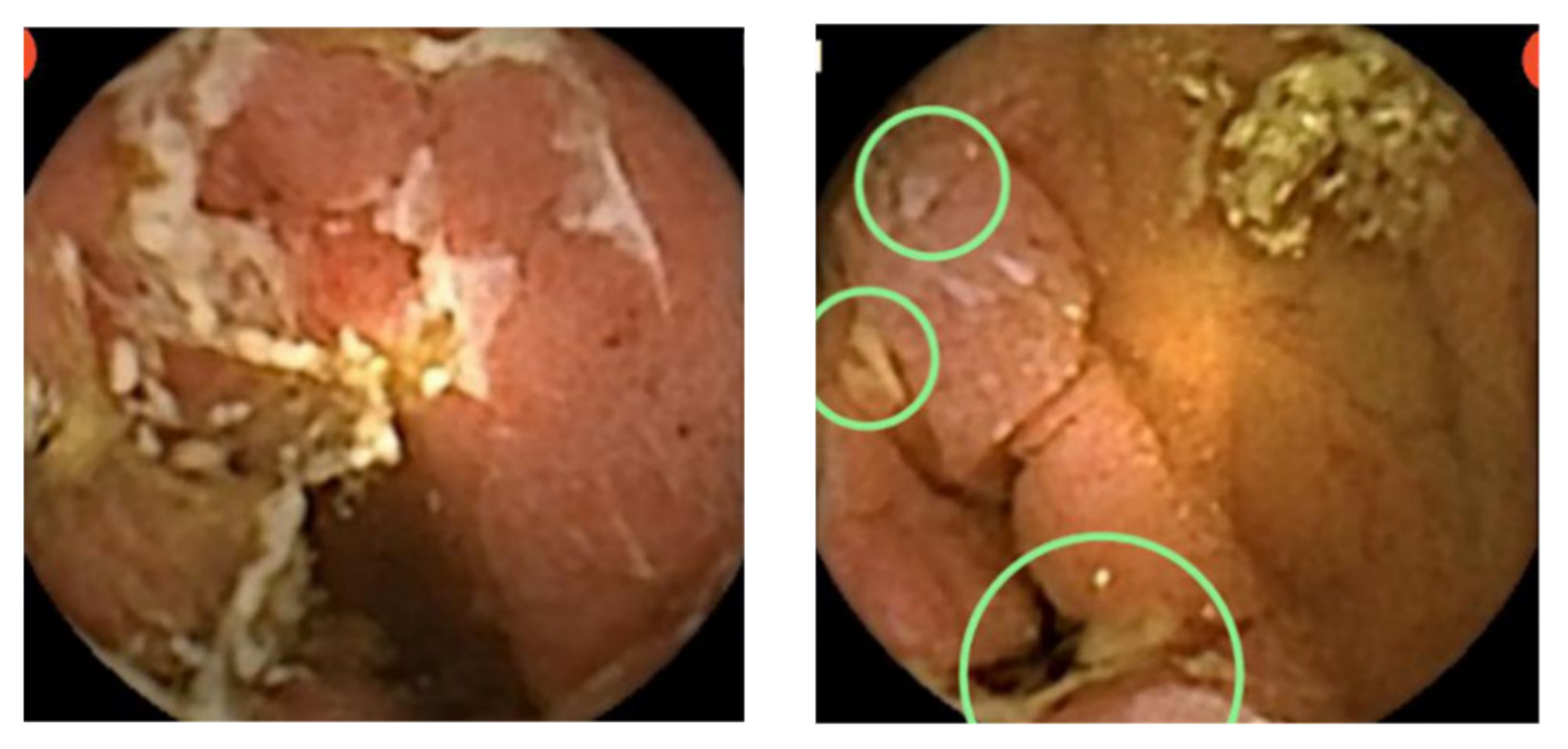
5. CD Extent and Disease Activity Evaluation
6. Adverse Events
7. Technical Advancements
8. Current International Recommendations
9. Appropriate Patients’ Selection
10. Disease Monitoring and Treatment
11. CE and COVID 19
12. CE and deep Learning Neural Network
13. Cost Effectiveness of CE in CD
14. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Burisch, J.; Pedersen, N.; Čuković-Čavka, S.; Brinar, M.; Kaimakliotis, I.; Duricova, D.; Shonová, O.; Vind, I.; Avnstrøm, S.; Thorsgaard, N.; et al. East-West gradient in the incidence of inflammatory bowel disease in Europe: The ECCO-EpiCom inception cohort. Gut 2014, 63, 588–597. [Google Scholar] [CrossRef]
- Dubcenco, E.; Jeejeebhoy, K.N.; Petroniene, R.; Tang, S.J.; Zalev, A.H.; Gardiner, G.W.; Baker, J.P. Capsule endoscopy findings in patients with established and suspected small-bowel Crohn’s disease: Correlation with radiologic, endoscopic, and histologic findings. Gastrointest. Endosc. 2005, 62, 538–544. [Google Scholar] [CrossRef] [PubMed]
- Cosnes, J.; Gower-Rousseau, C.; Seksik, P.; Cortot, A. Epidemiology and natural history of inflammatory bowel diseases. Gastroenterology 2011, 140, 1785–1794. [Google Scholar] [CrossRef] [PubMed]
- Torres, J.; Mehandru, S.; Colombel, J.F.; Peyrin-Biroulet, L. Crohn’s disease. Lancet 2017, 389, 1741–1755. [Google Scholar] [CrossRef]
- Love, J.R.; Irvine, E.J.; Fedorak, R.N. Quality of life in inflammatory bowel disease. J. Clin. Gastroenterol. 1992, 14, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Casellas, F.; Arenas, J.I.; Baudet, J.S.; Fábregas, S.; García, N.; Gelabert, J.; Medina, C.; Ochotorena, I.; Papo, M.; Rodrigo, L.; et al. Impairment of health-related quality of life in patients with inflammatory bowel disease: A Spanish multicenter study. Inflamm. Bowel. Dis. 2005, 11, 488–496. [Google Scholar] [CrossRef] [PubMed]
- Rubin, G.P.; Hungin, A.P.; Chinn, D.J.; Dwarakanath, D. Quality of life in patients with established inflammatory bowel disease: A UK general practice survey. Aliment. Pharm. Ther. 2004, 19, 529–535. [Google Scholar] [CrossRef]
- Casellas, F.; López-Vivancos, J.; Badia, X.; Vilaseca, J.; Malagelada, J.R. Influence of inflammatory bowel disease on different dimensions of quality of life. Eur. J. Gastroenterol. Hepatol. 2001, 13, 567–572. [Google Scholar] [CrossRef] [PubMed]
- Canavan, C.; Abrams, K.R.; Hawthorne, B.; Drossman, D.; Mayberry, J.F. Long-term prognosis in Crohn’s disease: Factors that affect quality of life. Aliment Pharm. Ther. 2006, 23, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Hjortswang, H.; Järnerot, G.; Curman, B.; Sandberg-Gertzén, H.; Tysk, C.; Blomberg, B.; Almer, S.; Ström, M. The influence of demographic and disease-related factors on health-related quality of life in patients with ulcerative colitis. Eur. J. Gastroenterol. Hepatol. 2003, 15, 1011–1020. [Google Scholar] [CrossRef]
- Latella, G.; Papi, C. Crucial steps in the natural history of inflammatory bowel disease. World J. Gastroenterol. 2012, 18, 3790–3799. [Google Scholar] [CrossRef]
- Chang, S.; Malter, L.; Hudesman, D. Disease monitoring in inflammatory bowel disease. World J. Gastroenterol. 2015, 21, 11246–11259. [Google Scholar] [CrossRef] [PubMed]
- Sauter, B.; Beglinger, C.; Girardin, M.; Macpherson, A.; Michetti, P.; Schoepfer, A.; Seibold, F.; Vavricka, S.R.; Rogler, G. Monitoring disease activity and progression in Crohn’s disease. A Swiss perspective on the IBD ahead ‘optimised monitoring’ recommendations. Digestion 2014, 89, 299–309. [Google Scholar] [CrossRef] [Green Version]
- Maaser, C.; Sturm, A.; Vavricka, S.R.; Kucharzik, T.; Fiorino, G.; Annese, V.; Calabrese, E.; Baumgart, D.C.; Bettenworth, D.; Borralho Nunes, P.; et al. ECCO-ESGAR guideline for diagnostic assessment in IBD Part 1: Initial diagnosis, monitoring of known IBD, detection of complications. J. Crohns Colitis 2019, 13, 144–164. [Google Scholar] [CrossRef] [Green Version]
- Colombel, J.F.; Rutgeerts, P.J.; Sandborn, W.J.; Yang, M.; Camez, A.; Pollack, P.F.; Thakkar, R.B.; Robinson, A.M.; Chen, N.; Mulani, P.M.; et al. Adalimumab induces deep remission in patients with Crohn’s disease. Clin. Gastroenterol. Hepatol. 2014, 12, 414–422. [Google Scholar] [CrossRef]
- Orlando, A.; Guglielmi, F.W.; Cottone, M.; Orlando, E.; Romano, C.; Sinagra, E. Clinical implications of mucosal healing in the management of patients with inflammatory bowel disease. Dig. Liver Dis. 2013, 45, 986–991. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pennazio, M.; Spada, C.; Eliakim, R.; Keuchel, M.; May, A.; Mulder, C.J.; Rondonotti, E.; Adler, S.N.; Albert, J.; Baltes, P.; et al. Small-bowel capsule endoscopy and device-assisted enteroscopy for diagnosis and treatment of small-bowel disorders: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline. Endoscopy 2015, 47, 352–376. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ooi, C.J.; Makharia, G.K.; Hilmi, I.; Gibson, P.R.; Fock, K.M.; Ahuja, V.; Ling, K.L.; Lim, W.C.; Thia, K.T.; Wei, S.C.; et al. Asia Pacific consensus statements on Crohn’s disease. Part 1: Definition, diagnosis, and epidemiology: (Asia Pacific Crohn’s Disease Consensus—Part 1). J. Gastroenterol. Hepatol. 2016, 31, 45–55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reinink, A.R.; Lee, T.C.; Higgins, P.D. Endoscopic Mucosal healing predicts favorable clinical outcomes in inflammatory bowel disease: A meta-analysis. Inflamm. Bowel Dis. 2016, 22, 1859–1869. [Google Scholar] [CrossRef] [PubMed]
- Gomollón, F.; Dignass, A.; Annese, V.; Tilg, H.; Van Assche, G.; Lindsay, J.O.; Peyrin-Biroulet, L.; Cullen, G.J.; Daperno, M.; Kucharzik, T.; et al. 3rd European evidence-based consensus on the diagnosis and management of Crohn’s disease 2016: Part 1: Diagnosis and medical management. J. Crohns Colitis 2017, 11, 3–25. [Google Scholar] [CrossRef] [Green Version]
- Sandborn, W.J. The future of inflammatory bowel disease therapy: Where do we go from here? Dig. Dis. 2012, 30 (Suppl. S3), 140–144. [Google Scholar] [CrossRef]
- Kopylov, U.; Rosenfeld, G.; Bressler, B.; Seidman, E. Clinical utility of fecal biomarkers for the diagnosis and management of inflammatory bowel disease. Inflamm. Bowel Dis. 2014, 20, 742–756. [Google Scholar] [CrossRef] [PubMed]
- Burri, E.; Beglinger, C.; Lehmann, F.S. Monitoring of therapy for inflammatory bowel disease. Digestion 2012, 86 (Suppl. S1), 1–5. [Google Scholar] [CrossRef] [PubMed]
- Laharie, D.; Mesli, S.; El Hajbi, F.; Chabrun, E.; Chanteloup, E.; Capdepont, M.; Razaire, S.; de Lédinghen, V.; Zerbib, F. Prediction of Crohn’s disease relapse with faecal calprotectin in infliximab responders: A prospective study. Aliment. Pharm. Ther. 2011, 34, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Koulaouzidis, A.; Douglas, S.; Rogers, M.A.; Arnott, I.D.; Plevris, J.N. Fecal calprotectin: A selection tool for small bowel capsule endoscopy in suspected IBD with prior negative bi-directional endoscopy. Scand. J. Gastroenterol. 2011, 46, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Lahat, A.; Fidder, H.H. Bowel imaging in IBD patients: Review of the literature and current recommendations. J. Gastroint. Dig. Syst. 2014, 4. [Google Scholar] [CrossRef]
- Kopylov, U.; Yablecovitch, D.; Lahat, A.; Neuman, S.; Levhar, N.; Greener, T.; Klang, E.; Rozendorn, N.; Amitai, M.M.; Ben-Horin, S.; et al. Detection of small bowel mucosal healing and deep remission in patients with known small bowel Crohn’s disease using biomarkers, capsule endoscopy, and imaging. Am. J. Gastroenterol. 2015, 110, 1316–1323. [Google Scholar] [CrossRef]
- Leighton, J.A.; Helper, D.J.; Gralnek, I.M.; Dotan, I.; Fernandez-Urien, I.; Lahat, A.; Malik, P.; Mullin, G.E.; Rosa, B. Comparing diagnostic yield of a novel pan-enteric video capsule endoscope with ileocolonoscopy in patients with active Crohn’s disease: A feasibility study. Gastrointest. Endosc. 2017, 85, 196–205.e191. [Google Scholar] [CrossRef]
- Davies, G.R.; Benson, M.J.; Gertner, D.J.; Van Someren, R.M.; Rampton, D.S.; Swain, C.P. Diagnostic and therapeutic push type enteroscopy in clinical use. Gut 1995, 37, 346–352. [Google Scholar] [CrossRef] [Green Version]
- Murphy, K.P.; McLaughlin, P.D.; O’Connor, O.J.; Maher, M.M. Imaging the small bowel. Curr. Opin. Gastroenterol. 2014, 30, 134–140. [Google Scholar] [CrossRef]
- Rafael Development Corporation Ltd. Given Imaging Company History. Available online: https://rdc.co.il/#success (accessed on 18 July 2021). Archived from the original on 2 August 2014.
- Redondo-Cerezo, E.; Sánchez-Capilla, A.D.; De La Torre-Rubio, P.; De Teresa, J. Wireless capsule endoscopy: Perspectives beyond gastrointestinal bleeding. World J. Gastroenterol. 2014, 20, 15664–15673. [Google Scholar] [CrossRef]
- Swain, P. Wireless capsule endoscopy and Crohn’s disease. Gut 2005, 54, 323–326. [Google Scholar] [CrossRef] [Green Version]
- Lewis, B.S.; Swain, P. Capsule endoscopy in the evaluation of patients with suspected small intestinal bleeding: Results of a pilot study. Gastrointest. Endosc. 2002, 56, 349–353. [Google Scholar] [CrossRef]
- Ell, C.; Remke, S.; May, A.; Helou, L.; Henrich, R.; Mayer, G. The first prospective controlled trial comparing wireless capsule endoscopy with push enteroscopy in chronic gastrointestinal bleeding. Endoscopy 2002, 34, 685–689. [Google Scholar] [CrossRef]
- Mylonaki, M.; Fritscher-Ravens, A.; Swain, P. Wireless capsule endoscopy: A comparison with push enteroscopy in patients with gastroscopy and colonoscopy negative gastrointestinal bleeding. Gut 2003, 52, 1122–1126. [Google Scholar] [CrossRef] [PubMed]
- Pennazio, M.; Santucci, R.; Rondonotti, E.; Abbiati, C.; Beccari, G.; Rossini, F.P.; De Franchis, R. Outcome of patients with obscure gastrointestinal bleeding after capsule endoscopy: Report of 100 consecutive cases. Gastroenterology 2004, 126, 643–653. [Google Scholar] [CrossRef] [PubMed]
- Saurin, J.C.; Delvaux, M.; Gaudin, J.L.; Fassler, I.; Villarejo, J.; Vahedi, K.; Bitoun, A.; Canard, J.M.; Souquet, J.C.; Ponchon, T.; et al. Diagnostic value of endoscopic capsule in patients with obscure digestive bleeding: Blinded comparison with video push-enteroscopy. Endoscopy 2003, 35, 576–584. [Google Scholar] [CrossRef] [PubMed]
- Costamagna, G.; Shah, S.K.; Riccioni, M.E.; Foschia, F.; Mutignani, M.; Perri, V.; Vecchioli, A.; Brizi, M.G.; Picciocchi, A.; Marano, P. A prospective trial comparing small bowel radiographs and video capsule endoscopy for suspected small bowel disease. Gastroenterology 2002, 123, 999–1005. [Google Scholar] [CrossRef]
- Voderholzer, W.A.; Ortner, M.; Rogalla, P.; Beinhölzl, J.; Lochs, H. Diagnostic yield of wireless capsule enteroscopy in comparison with computed tomography enteroclysis. Endoscopy 2003, 35, 1009–1014. [Google Scholar] [CrossRef] [PubMed]
- Hilmi, I.; Kobayashi, T. Capsule endoscopy in inflammatory bowel disease: When and how. Intest. Res. 2020, 18, 265–274. [Google Scholar] [CrossRef]
- Annese, V.; Daperno, M.; Rutter, M.D.; Amiot, A.; Bossuyt, P.; East, J.; Ferrante, M.; Götz, M.; Katsanos, K.H.; Kießlich, R.; et al. European evidence based consensus for endoscopy in inflammatory bowel disease. J. Crohns Colitis 2013, 7, 982–1018. [Google Scholar] [CrossRef] [Green Version]
- Rondonotti, E.; Spada, C.; Adler, S.; May, A.; Despott, E.J.; Koulaouzidis, A.; Panter, S.; Domagk, D.; Fernandez-Urien, I.; Rahmi, G.; et al. Small-bowel capsule endoscopy and device-assisted enteroscopy for diagnosis and treatment of small-bowel disorders: European Society of Gastrointestinal Endoscopy (ESGE) technical review. Endoscopy 2018, 50, 423–446. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cleynen, I.; González, J.R.; Figueroa, C.; Franke, A.; McGovern, D.; Bortlík, M.; Crusius, B.J.; Vecchi, M.; Artieda, M.; Szczypiorska, M.; et al. Genetic factors conferring an increased susceptibility to develop Crohn’s disease also influence disease phenotype: Results from the IBDchip European Project. Gut 2013, 62, 1556–1565. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, M.; Lim, S.; Choi, M.G.; Shim, K.N.; Lee, S.H. Effectiveness of capsule endoscopy compared with other diagnostic modalities in patients with small bowel Crohn’s Disease: A meta-analysis. Gut Liver 2017, 11, 62–72. [Google Scholar] [CrossRef] [Green Version]
- Triester, S.L.; Leighton, J.A.; Leontiadis, G.I.; Gurudu, S.R.; Fleischer, D.E.; Hara, A.K.; Heigh, R.I.; Shiff, A.D.; Sharma, V.K. A meta-analysis of the yield of capsule endoscopy compared to other diagnostic modalities in patients with non-stricturing small bowel Crohn’s disease. Am. J. Gastroenterol. 2006, 101, 954–964. [Google Scholar] [CrossRef]
- Dionisio, P.M.; Gurudu, S.R.; Leighton, J.A.; Leontiadis, G.I.; Fleischer, D.E.; Hara, A.K.; Heigh, R.I.; Shiff, A.D.; Sharma, V.K. Capsule endoscopy has a significantly higher diagnostic yield in patients with suspected and established small-bowel Crohn’s disease: A meta-analysis. Am. J. Gastroenterol. 2010, 105, 1240–1248. [Google Scholar] [CrossRef]
- González-Suárez, B.; Rodriguez, S.; Ricart, E.; Ordás, I.; Rimola, J.; Díaz-González, Á.; Romero, C.; de Miguel, C.R.; Jáuregui, A.; Araujo, I.K.; et al. Comparison of capsule endoscopy and magnetic resonance enterography for the assessment of small bowel lesions in Crohn’s disease. Inflamm. Bowel Dis. 2018, 24, 775–780. [Google Scholar] [CrossRef]
- Liu, B.; Ramalho, M.; AlObaidy, M.; Busireddy, K.K.; Altun, E.; Kalubowila, J.; Semelka, R.C. Gastrointestinal imaging-practical magnetic resonance imaging approach. World J. Radiol. 2014, 6, 544–566. [Google Scholar] [CrossRef]
- Moy, M.P.; Sauk, J.; Gee, M.S. The role of MR Enterography in assessing Crohn’s disease activity and treatment response. Gastroenterol. Res. Pract. 2016, 2016, 8168695. [Google Scholar] [CrossRef]
- Yung, D.E.; Har-Noy, O.; Tham, Y.S.; Ben-Horin, S.; Eliakim, R.; Koulaouzidis, A.; Kopylov, U. Capsule endoscopy, magnetic resonance enterography, and small bowel ultrasound for evaluation of postoperative recurrence in Crohn’s Disease: Systematic review and meta-analysis. Inflamm. Bowel Dis. 2017, 24, 93–100. [Google Scholar] [CrossRef] [Green Version]
- Kopylov, U.; Nemeth, A.; Koulaouzidis, A.; Makins, R.; Wild, G.; Afif, W.; Bitton, A.; Johansson, G.W.; Bessissow, T.; Eliakim, R.; et al. Small bowel capsule endoscopy in the management of established Crohn’s disease: Clinical impact, safety, and correlation with inflammatory biomarkers. Inflamm. Bowel Dis. 2015, 21, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Dussault, C.; Gower-Rousseau, C.; Salleron, J.; Vernier-Massouille, G.; Branche, J.; Colombel, J.F.; Maunoury, V. Small bowel capsule endoscopy for management of Crohn’s disease: A retrospective tertiary care centre experience. Dig. Liver Dis. 2013, 45, 558–561. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Jeon, S.R.; Choi, S.M.; Kim, H.G.; Lee, T.H.; Cho, J.H.; Jung, Y.; Kim, W.J.; Ko, B.M.; Kim, J.O.; et al. Practice patterns and clinical significance of use of capsule endoscopy in suspected and established Crohn’s disease. Intest. Res. 2017, 15, 467–474. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cave, D.; Legnani, P.; de Franchis, R.; Lewis, B.S. ICCE consensus for capsule retention. Endoscopy 2005, 37, 1065–1067. [Google Scholar] [CrossRef]
- Pasha, S.F.; Pennazio, M.; Rondonotti, E.; Wolf, D.; Buras, M.R.; Albert, J.G.; Cohen, S.A.; Cotter, J.; D’Haens, G.; Eliakim, R.; et al. Capsule retention in Crohn’s disease: A meta-analysis. Inflamm. Bowel Dis. 2020, 26, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Rozendorn, N.; Klang, E.; Lahat, A.; Yablecovitch, D.; Kopylov, U.; Eliakim, A.; Ben-Horin, S.; Amitai, M.M. Prediction of patency capsule retention in known Crohn’s disease patients by using magnetic resonance imaging. Gastrointest. Endosc. 2016, 83, 182–187. [Google Scholar] [CrossRef]
- Wang, Y.C.; Pan, J.; Liu, Y.W.; Sun, F.Y.; Qian, Y.Y.; Jiang, X.; Zou, W.B.; Xia, J.; Jiang, B.; Ru, N.; et al. Adverse events of video capsule endoscopy over the past two decades: A systematic review and proportion meta-analysis. BMC Gastroenterol. 2020, 20, 364. [Google Scholar] [CrossRef]
- Gralnek, I.M.; Defranchis, R.; Seidman, E.; Leighton, J.A.; Legnani, P.; Lewis, B.S. Development of a capsule endoscopy scoring index for small bowel mucosal inflammatory change. Aliment Pharm. Ther. 2008, 27, 146–154. [Google Scholar] [CrossRef]
- Niv, Y.; Ilani, S.; Levi, Z.; Hershkowitz, M.; Niv, E.; Fireman, Z.; O’Donnel, S.; O’Morain, C.; Eliakim, R.; Scapa, E.; et al. Validation of the capsule endoscopy Crohn’s disease activity index (CECDAI or Niv score): A multicenter prospective study. Endoscopy 2012, 44, 21–26. [Google Scholar] [CrossRef]
- Yablecovitch, D.; Lahat, A.; Neuman, S.; Levhar, N.; Avidan, B.; Ben-Horin, S.; Eliakim, R.; Kopylov, U. The Lewis score or the capsule endoscopy Crohn’s disease activity index: Which one is better for the assessment of small bowel inflammation in established Crohn’s disease? Ther. Adv. Gastroenterol. 2018, 11, 1756283X17747780. [Google Scholar] [CrossRef] [Green Version]
- Eliakim, R.; Yablecovitch, D.; Lahat, A.; Ungar, B.; Shachar, E.; Carter, D.; Selinger, L.; Neuman, S.; Ben-Horin, S.; Kopylov, U. A novel PillCam Crohn’s capsule score (Eliakim score) for quantification of mucosal inflammation in Crohn’s disease. United Eur. Gastroenterol. J. 2020, 8, 544–551. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goran, L.; Negreanu, A.M.; Stemate, A.; Negreanu, L. Capsule endoscopy: Current status and role in Crohn’s disease. World J. Gastrointest. Endosc. 2018, 10, 184–192. [Google Scholar] [CrossRef]
- Park, S.K.; Ye, B.D.; Kim, K.O.; Park, C.H.; Lee, W.S.; Jang, B.I.; Jeen, Y.T.; Choi, M.G.; Kim, H.J.; Group, K.G.I.S. Guidelines for video capsule endoscopy: Emphasis on Crohn’s disease. Clin. Endosc. 2015, 48, 128–135. [Google Scholar] [CrossRef]
- Enns, R.A.; Hookey, L.; Armstrong, D.; Bernstein, C.N.; Heitman, S.J.; Teshima, C.; Leontiadis, G.I.; Tse, F.; Sadowski, D. Clinical practice guidelines for the use of video capsule endoscopy. Gastroenterology 2017, 152, 497–514. [Google Scholar] [CrossRef] [Green Version]
- Eliakim, R.; Spada, C.; Lapidus, A.; Eyal, I.; Pecere, S.; Fernández-Urién, I.; Lahat, A.; Costamagna, G.; Schwartz, A.; Ron, Y.; et al. Evaluation of a new pan-enteric video capsule endoscopy system in patients with suspected or established inflammatory bowel disease—Feasibility study. Endosc. Int. Open 2018, 6, E1235–E1246. [Google Scholar] [CrossRef] [Green Version]
- Yamada, K.; Nakamura, M.; Yamamura, T.; Maeda, K.; Sawada, T.; Mizutani, Y.; Ishikawa, E.; Ishikawa, T.; Kakushima, N.; Furukawa, K.; et al. Diagnostic yield of colon capsule endoscopy for Crohn’s disease lesions in the whole gastrointestinal tract. BMC Gastroenterol. 2021, 21, 75. [Google Scholar] [CrossRef]
- Levine, A.; Koletzko, S.; Turner, D.; Escher, J.C.; Cucchiara, S.; de Ridder, L.; Kolho, K.L.; Veres, G.; Russell, R.K.; Paerregaard, A.; et al. ESPGHAN revised porto criteria for the diagnosis of inflammatory bowel disease in children and adolescents. J. Pediatr. Gastroenterol. Nutr. 2014, 58, 795–806. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jensen, M.D.; Nathan, T.; Rafaelsen, S.R.; Kjeldsen, J. Diagnostic accuracy of capsule endoscopy for small bowel Crohn’s disease is superior to that of MR enterography or CT enterography. Clin. Gastroenterol. Hepatol. 2011, 9, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Efthymiou, A.; Viazis, N.; Mantzaris, G.; Papadimitriou, N.; Tzourmakliotis, D.; Raptis, S.; Karamanolis, D.G. Does clinical response correlate with mucosal healing in patients with Crohn’s disease of the small bowel? A prospective, case-series study using wireless capsule endoscopy. Inflamm. Bowel Dis. 2008, 14, 1542–1547. [Google Scholar] [CrossRef]
- Hall, B.; Holleran, G.; Chin, J.L.; Smith, S.; Ryan, B.; Mahmud, N.; McNamara, D. A prospective 52 week mucosal healing assessment of small bowel Crohn’s disease as detected by capsule endoscopy. J. Crohns Colitis 2014, 8, 1601–1609. [Google Scholar] [CrossRef] [Green Version]
- Lahat, A.; Kopylov, U.; Amitai, M.M.; Neuman, S.; Levhar, N.; Yablecovitch, D.; Avidan, B.; Yanai, H.; Dotan, I.; Chowers, Y.; et al. Magnetic resonance enterography or video capsule endoscopy—What do Crohn’s disease patients prefer? Patient Prefer. Adherence 2016, 10, 1043–1050. [Google Scholar] [CrossRef] [Green Version]
- Mir, A.; Nguyen, V.Q.; Soliman, Y.; Sorrentino, D. Wireless capsule endoscopy for diagnosis and management of post-operative recurrence of Crohn’s disease. Life 2021, 11, 602. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Director-General’s Opening Remarks at the Media Briefing on COVID-19—11 March 2020. 2020. Available online: https://www.who.int/director-general/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020 (accessed on 1 September 2021).
- Yeo, C.; Kaushal, S.; Yeo, D. Enteric involvement of coronaviruses: Is faecal-oral transmission of SARS-CoV-2 possible? Lancet Gastroenterol. Hepatol. 2020, 5, 335–337. [Google Scholar] [CrossRef] [Green Version]
- Johnston, E.R.; Habib-Bein, N.; Dueker, J.M.; Quiroz, B.; Corsaro, E.; Ambrogio, M.; Kingsley, M.; Papachristou, G.I.; Kreiss, C.; Khalid, A. Risk of bacterial exposure to the endoscopist’s face during endoscopy. Gastrointest. Endosc. 2019, 89, 818–824. [Google Scholar] [CrossRef] [PubMed]
- Repici, A.; Maselli, R.; Colombo, M.; Gabbiadini, R.; Spadaccini, M.; Anderloni, A.; Carrara, S.; Fugazza, A.; Di Leo, M.; Galtieri, P.A.; et al. Coronavirus (COVID-19) outbreak: What the department of endoscopy should know. Gastrointest. Endosc. 2020, 92, 192–197. [Google Scholar] [CrossRef] [Green Version]
- Chiu, P.W.Y.; Ng, S.C.; Inoue, H.; Reddy, D.N.; Ling Hu, E.; Cho, J.Y.; Ho, L.K.; Hewett, D.G.; Chiu, H.M.; Rerknimitr, R.; et al. Practice of endoscopy during COVID-19 pandemic: Position statements of the Asian Pacific Society for Digestive Endoscopy (APSDE-COVID statements). Gut 2020, 69, 991–996. [Google Scholar] [CrossRef] [PubMed]
- Lui, R.N.; Wong, S.H.; Sánchez-Luna, S.A.; Pellino, G.; Bollipo, S.; Wong, M.Y.; Chiu, P.W.Y.; Sung, J.J.Y. Overview of guidance for endoscopy during the coronavirus disease 2019 pandemic. J. Gastroenterol. Hepatol. 2020, 35, 749–759. [Google Scholar] [CrossRef] [PubMed]
- Perisetti, A.; Gajendran, M.; Boregowda, U.; Bansal, P.; Goyal, H. COVID-19 and gastrointestinal endoscopies: Current insights and emergent strategies. Dig. Endosc. 2020, 32, 715–722. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lahat, A.; Benjamin, A. Changes in policy and endoscopic procedures during the 2019 coronavirus disease outbreak: A single center experience. Clin. Endosc. 2021, 54, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Gajendran, M.; Perisetti, A.; Aziz, M.; Raghavapuram, S.; Bansal, P.; Tharian, B.; Goyal, H. Inflammatory bowel disease amid the COVID-19 pandemic: Impact, management strategies, and lessons learned. Ann. Gastroenterol. 2020, 33, 591–602. [Google Scholar] [CrossRef]
- Lahat, A.; Shatz, Z. Telemedicine in clinical gastroenterology practice: What do patients prefer? Ther. Adv. Gastroenterol. 2021, 14, 1–9. [Google Scholar] [CrossRef]
- Klang, E. Deep learning and medical imaging. J. Thorac. Dis. 2018, 10, 1325–1328. [Google Scholar] [CrossRef] [PubMed]
- Soffer, S.; Ben-Cohen, A.; Shimon, O.; Amitai, M.M.; Greenspan, H.; Klang, E. Convolutional neural networks for radiologic images: A radiologist’s guide. Radiology 2019, 290, 590–606. [Google Scholar] [CrossRef]
- Barash, Y.; Klang, E. Automated quantitative assessment of oncological disease progression using deep learning. Ann. Transl. Med. 2019, 7, S379. [Google Scholar] [CrossRef] [PubMed]
- Soffer, S.; Klang, E.; Shimon, O.; Nachmias, N.; Eliakim, R.; Ben-Horin, S.; Kopylov, U.; Barash, Y. Deep learning for wireless capsule endoscopy: A systematic review and meta-analysis. Gastrointest. Endosc. 2020, 92, 831–839.e838. [Google Scholar] [CrossRef]
- Klang, E.; Barash, Y.; Margalit, R.Y.; Soffer, S.; Shimon, O.; Albshesh, A.; Ben-Horin, S.; Amitai, M.M.; Eliakim, R.; Kopylov, U. Deep learning algorithms for automated detection of Crohn’s disease ulcers by video capsule endoscopy. Gastrointest. Endosc. 2020, 91, 606–613.e602. [Google Scholar] [CrossRef] [PubMed]
- Klang, E.; Grinman, A.; Soffer, S.; Margalit Yehuda, R.; Barzilay, O.; Amitai, M.M.; Konen, E.; Ben-Horin, S.; Eliakim, R.; Barash, Y.; et al. Automated detection of Crohn’s disease intestinal strictures on capsule endoscopy images using deep neural networks. J. Crohns Colitis 2021, 15, 749–756. [Google Scholar] [CrossRef]
- Saunders, R.; Torrejon Torres, R.; Konsinski, L. Evaluating the clinical and economic consequences of using video capsule endoscopy to monitor Crohn’s disease. Clin. Exp. Gastroenterol. 2019, 12, 375–384. [Google Scholar] [CrossRef] [Green Version]
- Lobo, A.; Torrejon Torres, R.; McAlindon, M.; Panter, S.; Leonard, C.; van Lent, N.; Saunders, R. Economic analysis of the adoption of capsule endoscopy within the British NHS. Int. J. Qual. Health Care 2020, 32, 332–341. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lahat, A.; Veisman, I. Capsule Endoscopy in Crohn’s Disease—From a Relative Contraindication to Habitual Monitoring Tool. Diagnostics 2021, 11, 1737. https://doi.org/10.3390/diagnostics11101737
Lahat A, Veisman I. Capsule Endoscopy in Crohn’s Disease—From a Relative Contraindication to Habitual Monitoring Tool. Diagnostics. 2021; 11(10):1737. https://doi.org/10.3390/diagnostics11101737
Chicago/Turabian StyleLahat, Adi, and Ido Veisman. 2021. "Capsule Endoscopy in Crohn’s Disease—From a Relative Contraindication to Habitual Monitoring Tool" Diagnostics 11, no. 10: 1737. https://doi.org/10.3390/diagnostics11101737
APA StyleLahat, A., & Veisman, I. (2021). Capsule Endoscopy in Crohn’s Disease—From a Relative Contraindication to Habitual Monitoring Tool. Diagnostics, 11(10), 1737. https://doi.org/10.3390/diagnostics11101737





