The Role of Fecal Calprotectin in Patients with Systemic Sclerosis and Small Intestinal Bacterial Overgrowth (SIBO)
Abstract
1. Introduction
2. Patients and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ayling, R.M.; Kok, K. Fecal Calprotectin. Adv. Clin. Chem 2018, 87, 161–190. [Google Scholar] [CrossRef] [PubMed]
- Moore, B.W. A soluble protein characteristic of the nervous system. Biochem. Biophys. Res. Commun 1965, 19, 739–744. [Google Scholar] [CrossRef]
- Røseth, A.G.; Fagerhol, M.K.; Aadland, E.; Schjønsby, H. Assessment of the neutrophil dominating protein calprotectin in feces a methodological study. Scand. J. Gastroenterol 1992, 27, 793–798. [Google Scholar] [CrossRef] [PubMed]
- Besold, A.N.; Culbertson, E.M.; Nam, L.; Hobbs, R.P.; Boyko, A.; Maxwell, C.N.; Chazin, W.J.; Marques, A.R.; Culotta, V.C. Antimicrobial action of calprotectin that does not involve metal withholding. Metallomics 2018, 10, 1728–1742. [Google Scholar] [CrossRef] [PubMed]
- Kehl-Fie, T.E.; Chitayat, S.; Hood, M.; Damo, S.; Restrepo, N.; Garcia, C.; Munro, K.A.; Chazin, W.J.; Skaar, E.P. Nutrient metal sequestration by calprotectin inhibits bacterial superoxide defense, enhancing neutrophil killing of Staphylococcus aureus. Cell Host Microbe 2011, 10, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Nakashige, T.G.; Zygiel, E.M.; Drennan, C.L.; Nolan, E.M. Nickel sequestration by the host-defence protein human calprotectin. J. Am. Chem. Soc. 2017, 139, 8828–8836. [Google Scholar] [CrossRef] [PubMed]
- D’Angelo, F.; Felley, C.; Frossard, J.L. Calprotectin in Daily Practice: Where Do We Stand in 2017? Digestion 2017, 95, 293–301. [Google Scholar] [CrossRef]
- Vogl, T.; Tenbrock, K.; Ludwig, S.; Leukert, N.; Ehrhardt, C.; van Zoelen, M.A.D.; Nacken, W.; Foell, D.; van der Poll, T.; Sorg, C. Mrp8 and Mrp14 are endogenous activators of Toll-like receptor 4, promoting lethal, endotoxin-induced shock. Nat. Med. 2007, 13, 1042–1049. [Google Scholar] [CrossRef]
- D’Haens, G.; Ferrante, M.; Vermeire, S.; Baert, F.; Noman, M.; Moortgat, L.; Geens, P.; Iwens, D.; Aerden, I.; Van Assche, G.; et al. Fecal calprotectin is a surrogate marker for endoscopic lesions in inflammatory bowel disease. Inflamm Bowel Dis. 2012, 18, 2218–2224. [Google Scholar] [CrossRef]
- Sipponen, T.; Kolho, K.L. Fecal calprotectin in diagnosis and clinical assessment of inflammatory bowel disease. Scand J. Gastroenterol. 2015, 50, 74–80. [Google Scholar] [CrossRef]
- Tibble, J.; Teahon, K.; Thjodleifsson, B.; Roseth, A.; Sigthorsson, G.; Bridger, S.; Foster, R.; Sherwood, R.; Fagerhol, M.; Bjarnason, I. A simple method for assessing intestinal inflammation in Crohn’s disease. Gut 2000, 47, 506–513. [Google Scholar] [CrossRef] [PubMed]
- Carroccio, A.; Iacono, G.; Cottone, M.; Di Prima, L.; Cartabellotta, F.; Cavataio, F.; Scalici, C.; Montalto, G.; Di Fede, G.; Rini, G.B.; et al. Diagnostic accuracy of fecal calprotectin assay in distinguishing organic causes of chronic diarrhea from irritable bowel syndrome: A prospective study in adults and children. Clin. Chem. 2003, 49, 861–867. [Google Scholar] [PubMed]
- Costa, F.; Mumolo, M.G.; Ceccarelli, L.; Bellini, M.; Romano, M.R.; Sterpi, C.; Ricchiuti, A.; Marchi, S.; Bottai, M. Calprotectin is a stronger predictive marker of relapse in ulcerative colitis than in Crohn’s disease. Gut 2005, 54, 364–368. [Google Scholar] [CrossRef]
- Dolwani, S.; Metzner, M.; Wassell, J.J.; Yong, A.; Hawthorne, A.B. Diagnostic accuracy of faecal calprotectin estimation in prediction of abnormal small bowel radiology. Aliment. Pharm. Ther. 2004, 20, 615–621. [Google Scholar] [CrossRef] [PubMed]
- D’Inca, R.; Dal Pont, E.; Di Leo, V.; Ferronato, A.; Fries, W.; Vettorato, M.G.; Martines, D.; Sturniolo, G.C. Calprotectin and lactoferrin in the assessment of intestinal inflammation and organic disease. Int J. Colorectal Dis. 2007, 22, 429–437. [Google Scholar] [CrossRef]
- Schoepfer, A.M.; Trummler, M.; Seeholzer, P.; Seibold-Schmid, B.; Seibold, F. Discriminating IBD from IBS: Comparison of the test performance of fecal markers, blood leukocytes, CRP, and IBD antibodies. Inflamm Bowel Dis. 2008, 14, 32–39. [Google Scholar] [CrossRef]
- Schroder, O.; Naumann, M.; Shastri, Y.; Povse, N.; Stein, J. Prospective evaluation of faecal neutrophil-derived proteins in identifying intestinal inflammation: Combination of parameters does not improve diagnostic accuracy of calprotectin. Aliment. Pharm. Ther. 2007, 26, 1035–1042. [Google Scholar] [CrossRef]
- Manceau, H.; Chicha-Cattoir, V.; Puy, H.; Peoc’h, K. Fecal calprotectin in inflammatory bowel diseases: Update and perspectives. Clin. Chem Lab. Med. 2017, 55, 474–483. [Google Scholar] [CrossRef]
- Davidson, F.; Lock, R.J. Paediatric reference ranges for faecal calprotectin: A UK study. Ann. Clin. Biochem. 2017, 54, 214–218. [Google Scholar] [CrossRef]
- Denton, C.P.; Khanna, D. Systemic sclerosis. Lancet 2017, 390, 1685–1699. [Google Scholar] [CrossRef]
- Bures, J.; Cyrany, J.; Kohoutova, D.; Förstl, M.; Rejchrt, S.; Kvetina, J.; Vorisek, V.; Kopacova, M. Small intestinal bacterial overgrowth syndrome. World J. Gastroenterol. 2010, 16, 2978–2990. [Google Scholar] [CrossRef] [PubMed]
- Sakkas, L.I.; Simopoulou, T.; Daoussis, D.; Liossis, S.N.; Potamianos, S. Intestinal Involvement in Systemic Sclerosis: A Clinical Review. Dig. Dis Sci. 2018, 63, 834–844. [Google Scholar] [CrossRef] [PubMed]
- Jaeger, V.K.; Wirz, E.G.; Allanore, Y.; Rossbach, P.; Riemekasten, G.; Hachulla, E.; Distler, O.; Airo, P.; Carreira, P.E.; Gurman, A.B.; et al. Incidences and Risk Factors of Organ Manifestations in the Early Course of Systemic Sclerosis: A Longitudinal EUSTAR Study. PLoS ONE 2016, 11, e0163894. [Google Scholar] [CrossRef] [PubMed]
- Miazga, A.; Osiński, M.; Cichy, W.; Żaba, R. Current views on the etiopathogenesis, clinical manifestation, diagnostics, treatment and correlation with other nosological entities of SIBO. Adv. Med. Sci. 2015, 60, 118–124. [Google Scholar] [CrossRef]
- Quigley, E.M.M. The Spectrum of Small Intestinal Bacterial Overgrowth (SIBO). Curr Gastroenterol Rep. 2019, 21, 3. [Google Scholar] [CrossRef]
- Quigley, E.M. Small intestinal bacterial overgrowth: What it is and what it is not. Curr. Opin. Gastroenterol. 2014, 30, 141–146. [Google Scholar] [CrossRef]
- Kaye, S.A.; Lim, S.G.; Taylor, M.; Patel, S.; Gillespie, S.; Black, C.M. Small bowel bacterial overgrowth in systemic sclerosis: Detection using direct and indirect methods and treatment outcome. Br. J. Rheumatol. 1995, 34, 265–269. [Google Scholar] [CrossRef]
- Marie, I.; Leroi, A.M.; Menard, J.F.; Levesque, H.; Quillard, M.; Ducrotte, P. Fecal calprotectin in systemic sclerosis and review of the literature. Autoimmun. Rev. 2015, 14, 547–554. [Google Scholar] [CrossRef]
- Andréasson, K.; Saxne, T.; Scheja, A.; Bartosik, I.; Mandl, T.; Hesselstrand, R. Faecal levels of calprotectin in systemic sclerosis are stable over time and are higher compared to primary Sjögren’s syndrome and rheumatoid arthritis. Arthritis Res. 2014, 16, R46. [Google Scholar] [CrossRef]
- Andréasson, K.; Scheja, A.; Saxne, T.; Ohlsson, B.; Hesselstrand, R. Faecal calprotectin: A biomarker of gastrointestinal disease in systemic sclerosis. J. Intern. Med. 2011, 270, 50–57. [Google Scholar] [CrossRef]
- van den Hoogen, F.; Khanna, D.; Fransen, J.; Johnson, S.R.; Baron, M.; Tyndall, A.; Matucci-Cerinic, M.; Naden, R.; Riemekasten, G.; Carreira, P.; et al. 2013 classification criteria for systemic sclerosis: An American College of Rheumatology/European League against Rheumatism collaborative initiative. Arthritis Rheum. 2013, 65, 2737–2747. [Google Scholar] [CrossRef] [PubMed]
- Khanna, D.; Nagaraja, V.; Gladue, H.; Chey, W.; Pimentel, M.; Frech, T. Measuring Response in the Gastrointestinal Tract in systemic sclerosis. Curr. Opin. Rheumatol. 2013, 25, 700–706. [Google Scholar] [CrossRef] [PubMed]
- Gyger, G.; Baron, M. Systemic sclerosis: Gastrointestinal disease and its management. Rheum. Dis. Clin. N. Am. 2015, 41, 459–473. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Xu, D.; Li, M.T.; Yao, Y.; Jin, M.; Zeng, X.F.; Qian, J. Gastrointestinal manifestations on impaired quality of life in systemic sclerosis. J. Dig. Dis. 2019, 20, 256–261. [Google Scholar] [CrossRef]
- Emmanuel, A. Current management of the gastrointestinal complications of systemic sclerosis. Nat. Rev. Gastroenterol Hepatol. 2016, 13, 461–472. [Google Scholar] [CrossRef]
- Marie, I.; Ducrotté, P.; Denis, P.; Hellot, M.F.; Levesque, H. Outcome of small-bowel motor impairment in systemic sclerosis—A prospective manometric 5-yr follow-up. Rheumatol. Oxf. 2007, 46, 150–153. [Google Scholar] [CrossRef]
- McFarlane, I.M.; Bhamra, M.S.; Kreps, A.; Iqbal, S.; Al-Ani, F.; Aponte, C.S.; Grant, C.; Singh, S.; Awwal, K.; Koci, K.; et al. Gastrointestinal Manifestations of Systemic Sclerosis. Rheumatol. Sunnyvale 2018, 8, 235. [Google Scholar] [CrossRef]
- Akesson, A.; Wollheim, F.A. Organ manifestations in 100 patients with progressive systemic sclerosis: A comparison between the CREST syndrome and diffuse scleroderma. Br. J. Rheumatol. 1989, 28, 81–86. [Google Scholar] [CrossRef]
- Polkowska-Pruszyńska, B.; Gerkowicz, A.; Szczepanik-Kułak, P.; Krasowska, D. Small intestinal bacterial overgrowth in systemic sclerosis: A review of the literature. Arch. Derm. Res. 2019, 311, 1–8. [Google Scholar] [CrossRef]
- Erdogan, A.; Rao, S.S.; Gulley, D.; Jacobs, C.; Lee, Y.Y.; Badger, C. Small intestinal bacterial overgrowth: Duodenal aspiration vs. glucose breath test. Neurogastroenterol Motil. 2015, 27, 481–489. [Google Scholar] [CrossRef]
- McMahan, Z.H. Gastrointestinal involvement in systemic sclerosis: An update. Curr. Opin. Rheumatol. 2019, 31, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, C.; Coss, A.E.; Attaluri, A.; Valestin, J.; Rao, S.S.C. Dysmotility and ppi use are independent risk factors for small intestinal bacterial and/or fungal overgrowth. Aliment. Pharm. Ther. 2013, 37, 1103–1111. [Google Scholar] [CrossRef] [PubMed]
- Walecka, I. Systemic sclerosis and the gastrointestinal tract. Prz. Gastroenterol. 2017, 12, 163–168. [Google Scholar] [CrossRef] [PubMed]
| Variable | Study Group | Absence of SIBO 1 | Presence of SIBO 1 | p | Fecal Calprotectin Levels [ug/g] Me | p | ||
|---|---|---|---|---|---|---|---|---|
| Gender Female Male | 39 (97.5%) 1 (2.5%) | 20 (95.2%) 1 (4.8%) | 19 (100%) - | 0.9596 | 118 20 | - | ||
| Disease subtype dcSCC 2 IcSCC 3 | 6 (15.4%) 33 (84.6%) | 2 (9.52%) 19 (90.48%) | 4 (22.2%) 14 (77.8%) | 0.5153 | 221.00 74 | 0.0943 | ||
| Antibodies ACA 4 Scl-70 5 Not specified ACA 4/Scl-70 5 | 22 (59.5%) 12 (32.4%) 1 (2.7%) 2 (5.4%) | 12 (60%) 7 (35%) - 1 (5%) | 10 (58.8%) 5 (29.4%) 1 (5.9%) 1 (5.9%) | 0.7338 | 72 206 227 717 | 0.0729 | ||
| Gastrointestinal comorbidities No Yes | 24 (60%) 16 (40%) | 12 (57.1%) 9 (42.9%) | 12 (63.2%) 7 (36.8%) | 0.9485 | 96.5 119 | 0.2227 | ||
| Gastrointestinal comorbidities GERD 6 Esophagal hiatal hernia Others (i.a. mechanical bowel obstruction) | 5 (31.3%) 7 (43.7%) 4 (25%) | 3 (33.3%) 4 (44.4%) 2 (22.2%) | 2 (28.6%) 3 (42.9%) 2 (28.6%) | 0.9539 | 162 66.5 380 | 0.3878 | ||
| SIBO 1 presence No Yes | 21 (52.5%) 19 (47.5%) | 21 (100%) - | - 19 (100%) | - | 24 206 | 0.0010 | ||
| SIBO 1 eradication No Yes | 4 (26.7%) 11 (73.3%) | - | 4 (26.7%) 11 (73.3%) | - | 7 345 156 | 8 311 137 | 7 0.1059 | 8 0.1198 |
| 8Characteristics of the control group | ||||||||
| Variable | Control group | Absence of SIBO | Presence of SIBO | p | Fecal calprotectin levels [ug/g] Me | P | ||
| Gender Female Male | 36 (92.3%) 3 (7.7%) | 31 (91.2%) 3 (8.8%) | 5 (100%) - | 0.8357 | 20 33 | 0.1617 | ||
| Variable | Study Group | |||||
|---|---|---|---|---|---|---|
| SIBO Absence | SIBO Presence | |||||
| n | rho | p | n | rho | p | |
| Age (years) | 19 | 0.505 | 0.0275 | 17 | −0.006 | 0.9813 |
| Weight (kilograms) | 19 | −0.143 | 0.5592 | 17 | 0.215 | 0.4076 |
| BMI 1 | 19 | −0.150 | 0.5387 | 17 | 0.140 | 0.5928 |
| Disease duration (years) | 19 | 0.093 | 0.7038 | 17 | 0.336 | 0.1873 |
| TP 2 (g/dL) | 6 | 0.395 | 0.4387 | 4 | −0.400 | 0.6000 |
| Albumins (% TP) | 6 | −0.273 | 0.6004 | 4 | 0.200 | 0.8000 |
| ESR 3 (mm/h) | 16 | 0.208 | 0.4405 | 15 | −0.075 | 0.7905 |
| AST 4 (IU/L) | 19 | −0.404 | 0.0865 | 17 | 0.125 | 0.6326 |
| ALT 5 (U/L) | 19 | −0.567 | 0.0114 | 16 | 0.153 | 0.5717 |
| CRP 6 (mg/L) | 19 | 0.564 | 0.0120 | 17 | 0.064 | 0.8078 |
| C3 7 (g/dL) | 14 | −0.199 | 0.4952 | 12 | 0.252 | 0.4299 |
| C4 7 (g/L) | 14 | −0.034 | 0.9072 | 12 | −0.462 | 0.1309 |
| CK 8 (IU/L) | 3 | −0.500 | 0.6667 | 10 | −0.122 | 0.7379 |
| Rheumatoid factor (IU/mL) | 5 | −0.344 | 0.5707 | 7 | −0.079 | 0.8666 |
| NT-proBNP (pg/mL) | 14 | 0.474 | 0.0866 | 13 | 0.319 | 0.2886 |
| Leukocytes (K/µL) | 19 | 0.580 | 0.0093 | 17 | 0.267 | 0.2996 |
| Neutrophils (K/µL) | 19 | 0.781 | 0.0001 | 17 | 0.561 | 0.0191 |
| Variables | Fecal Calprotectin Levels | ||
|---|---|---|---|
| n | rho | p | |
| Age (years) | 36 | 0.180 | 0.2940 |
| Weight (kilograms) | 36 | −0.016 | 0.9257 |
| BMI 1 | 36 | −0.063 | 0.7138 |
| Disease duration (years) | 36 | 0.250 | 0.1413 |
| TP 2 (g/dL) | 10 | −0.018 | 0.9596 |
| Albumins (% TP) | 10 | 0.178 | 0.6229 |
| ESR 3 (mm/h) | 31 | 0.144 | 0.4407 |
| CRP 4 (mg/L) | 36 | 0.331 | 0.0483 |
| C3 5 (g/dL) | 26 | 0.131 | 0.5241 |
| C4 5 (g/L) | 26 | −0.198 | 0.3317 |
| CK 6 (IU/L) | 13 | 0.011 | 0.9715 |
| Rheumatoid factor (IU/mL) | 12 | −0.212 | 0.5082 |
| NT-proBNP (pg/mL) | 27 | 0.370 | 0.0578 |
| FC 7 (after eradication) | 8 | 0.623 | 0.0991 |
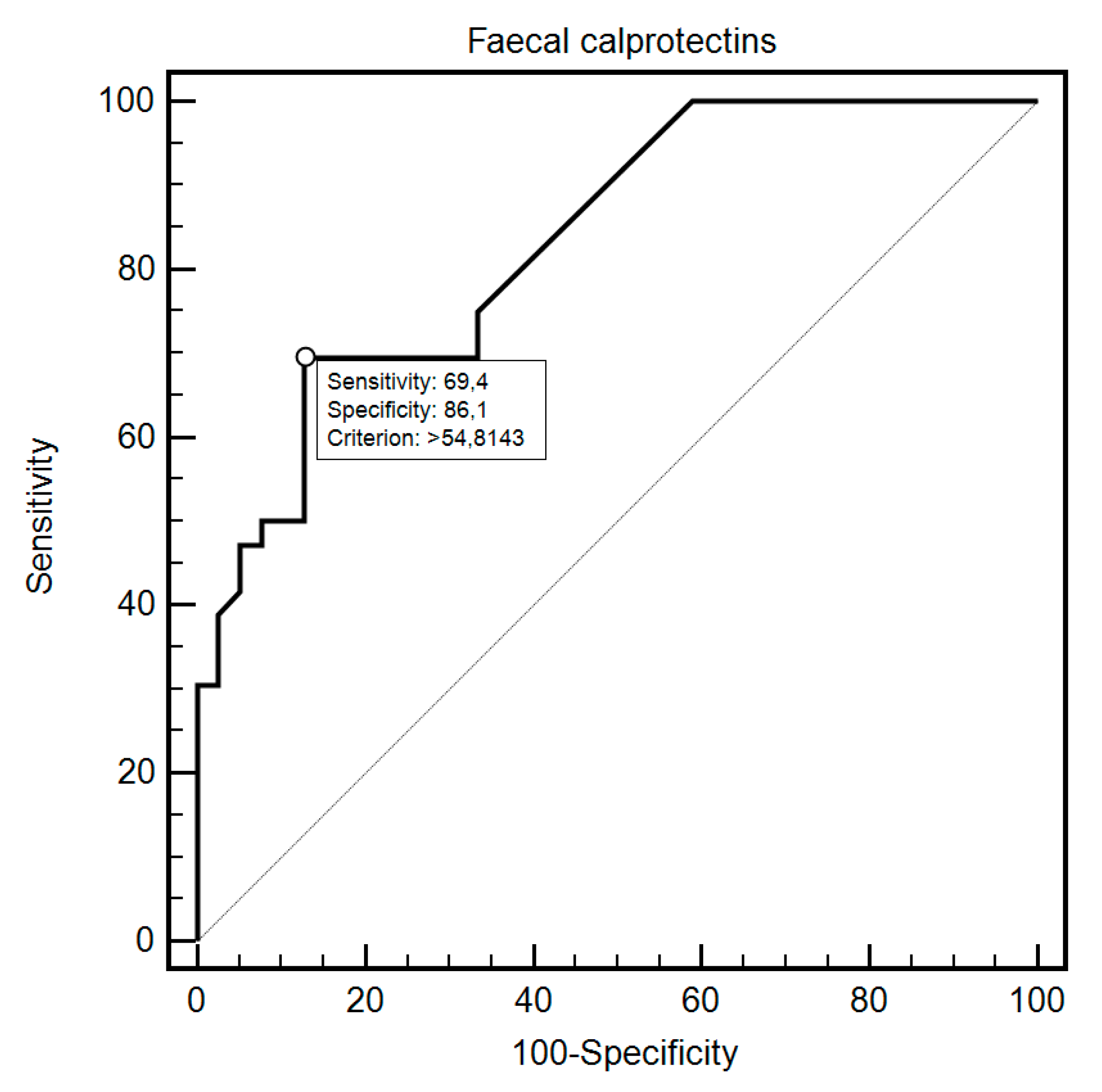
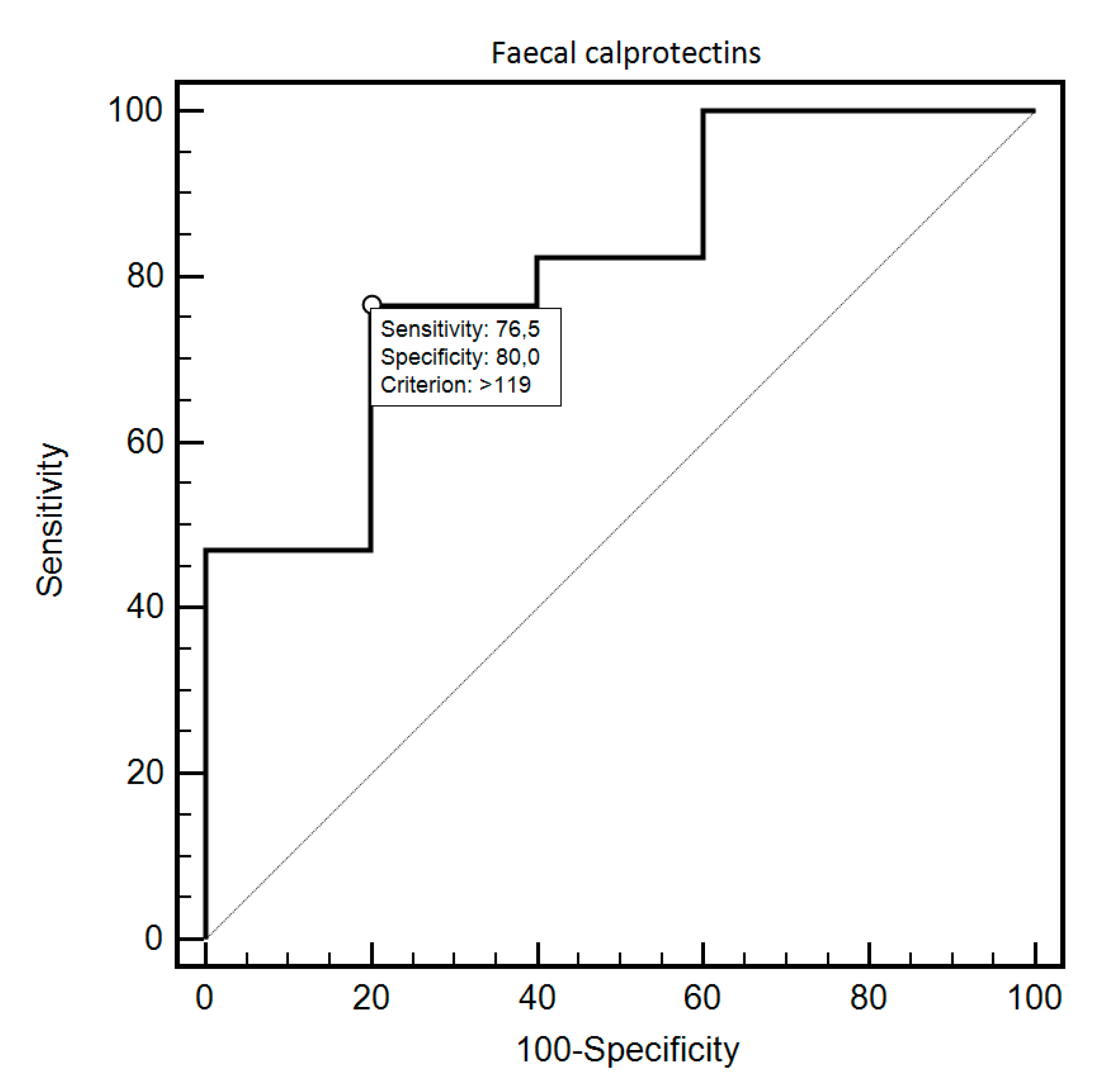
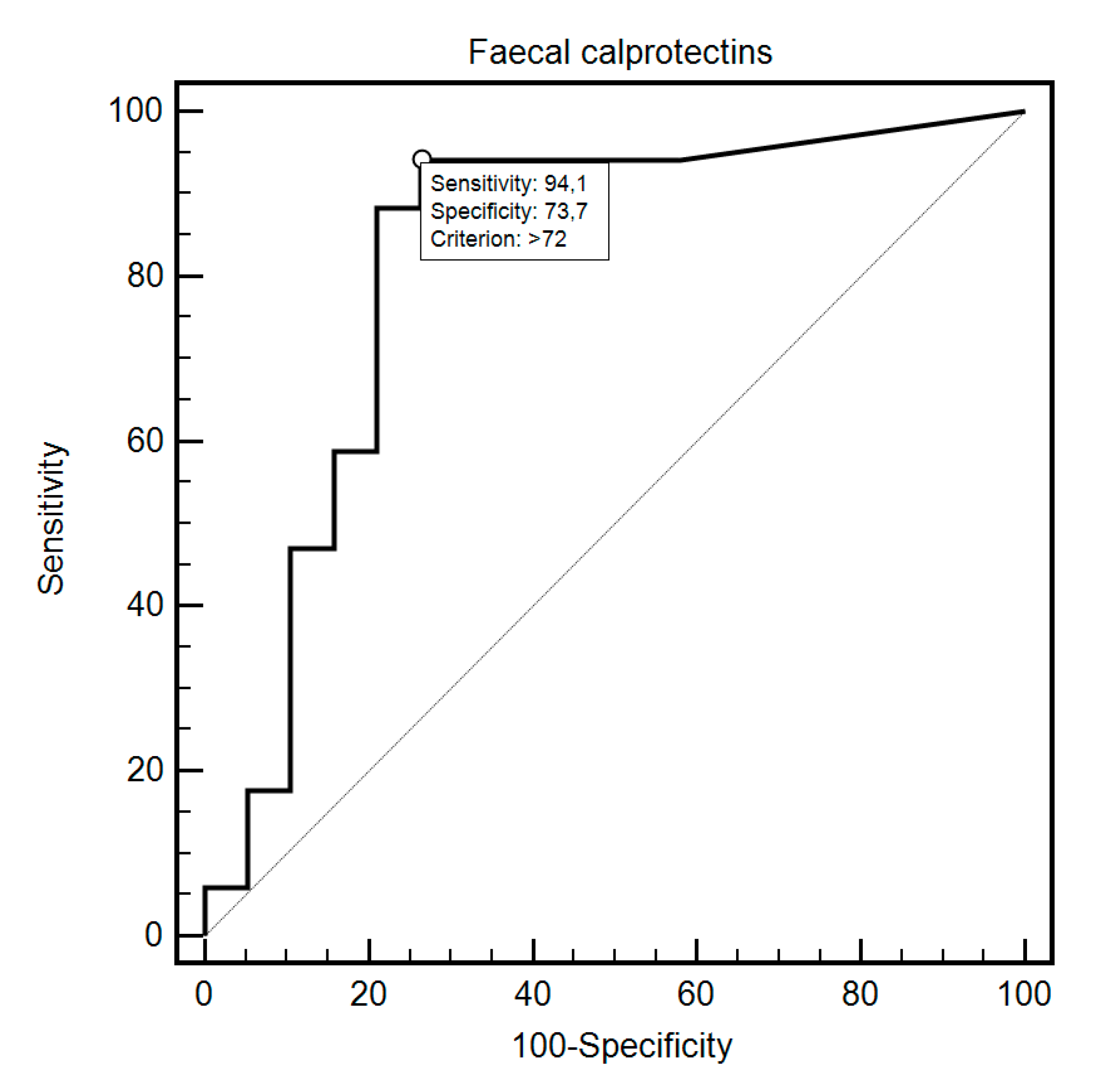
| Fecal Calprotectin | Sensitivity | Specificity | Criterion | AUC (95%CI) | p |
|---|---|---|---|---|---|
| Group Study vs. Control | 69.44% | 87.18% | >60 | 0.83 (0.73–0.91) | <0.0001 |
| SIBO 1 presence Study vs. Control | 76.47% | 80% | >119 | 0.81 (0.59–0.94) | 0.0054 |
| SIBO 1 (study group) Yes No | 94.12% | 73.68% | >72 | 0.82 (0.66–0.93) | <0.0001 |
| Eradication (study group) Yes No | 90% | 75% | ≤254 | 0.80 (0.51–0.96) | 0.0509 |
| Disease subtype dcSCC 2 IcSCC 3 | 75.86% | 66.67% | ≤162 | 0.72 (0.54–0.86) | 0.0477 |
| Antibodies type ACA 4 Scl-70 5, unknown, ACA 4/Scl-70 5 | 90% | 61.54% | ≤162 | 0.74 (0.56–0.88) | 0.0096 |
| Antibodies type Scl-70 5 ACA 4, unknown, ACA 4/Scl-70 5 | 54.55% | 86.36% | >206 | 0.66 (0.48–0.82) | 0.1131 |
| Gastrointestinal diseases Yes No | 66.67% | 75% | >152 | 0.68 (0.47–0.85) | 0.2076 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Polkowska-Pruszyńska, B.; Gerkowicz, A.; Rawicz-Pruszyński, K.; Krasowska, D. The Role of Fecal Calprotectin in Patients with Systemic Sclerosis and Small Intestinal Bacterial Overgrowth (SIBO). Diagnostics 2020, 10, 587. https://doi.org/10.3390/diagnostics10080587
Polkowska-Pruszyńska B, Gerkowicz A, Rawicz-Pruszyński K, Krasowska D. The Role of Fecal Calprotectin in Patients with Systemic Sclerosis and Small Intestinal Bacterial Overgrowth (SIBO). Diagnostics. 2020; 10(8):587. https://doi.org/10.3390/diagnostics10080587
Chicago/Turabian StylePolkowska-Pruszyńska, Beata, Agnieszka Gerkowicz, Karol Rawicz-Pruszyński, and Dorota Krasowska. 2020. "The Role of Fecal Calprotectin in Patients with Systemic Sclerosis and Small Intestinal Bacterial Overgrowth (SIBO)" Diagnostics 10, no. 8: 587. https://doi.org/10.3390/diagnostics10080587
APA StylePolkowska-Pruszyńska, B., Gerkowicz, A., Rawicz-Pruszyński, K., & Krasowska, D. (2020). The Role of Fecal Calprotectin in Patients with Systemic Sclerosis and Small Intestinal Bacterial Overgrowth (SIBO). Diagnostics, 10(8), 587. https://doi.org/10.3390/diagnostics10080587






