A Preliminary Study on the Safety of Elastography during Pregnancy: Hypoacusia, Anthropometry, and Apgar Score in Newborns
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Reference Population
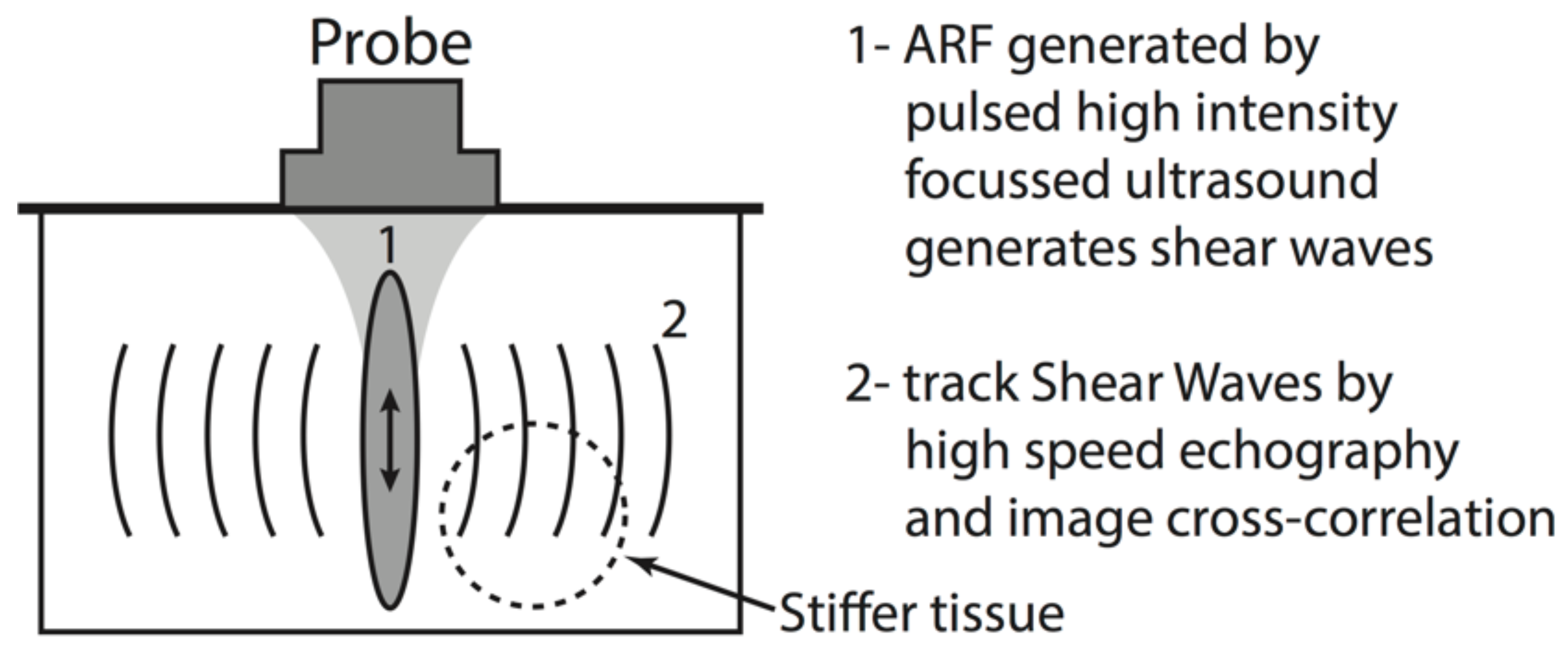
2.3. Acoustic Radiation Force Elastography
2.4. Outcomes
2.5. Statistical Analysis
3. Results
3.1. Characteristics of Population and Apgar Test Score
3.2. Outcomes on Hypoacusia
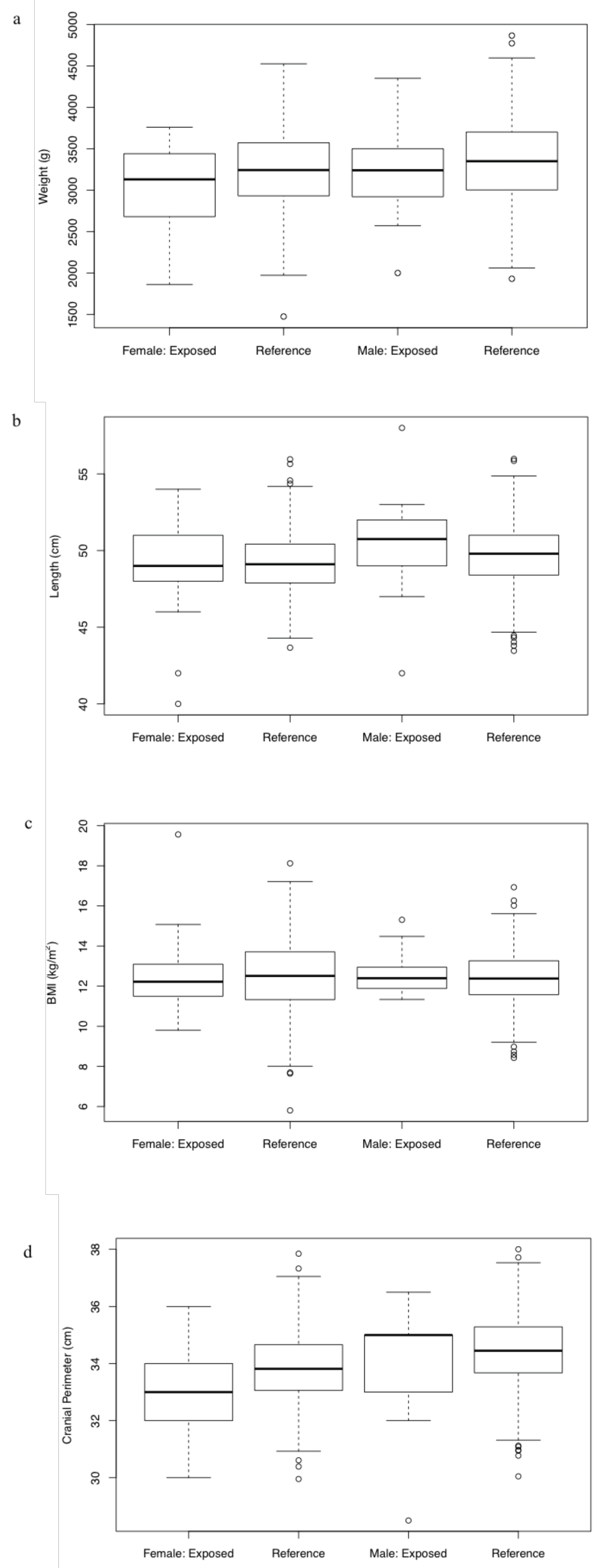
3.3. Outcomes on Anthropometry
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Khalil, M.R.; Thorsen, P.; Uldbjerg, N. Cervical ultrasound elastography may hold potential to predict risk of preterm birth. Dan. Med. 2013, 60, A4570. [Google Scholar]
- Köbbing, K.; Fruscalzo, A.; Hammer, K.; Möllers, M.; Falkenberg, M.; Kwiecien, R.; Klockenbusch, W.; Schmitz, R. Quantitative elastography of the uterine cervix as a predictor of preterm delivery. J. Perinatol. 2014, 34, 774–780. [Google Scholar] [CrossRef] [PubMed]
- Swiatkowska-Freund, M.; Preis, K. Elastography of the uterine cervix: Implications for success of induction of labor. Ultrasound Obstet. Gynecol. 2011, 38, 52–56. [Google Scholar] [CrossRef] [PubMed]
- Pereira, S.; Frick, A.P.; Poon, L.C.; Zamprakou, A.; Nicolaides, K.H. Successful induction of labor: Prediction by preinduction cervical length, angle of progression and cervical elastography. Ultrasound Obstet. Gynecol. 2014, 44, 468–475. [Google Scholar] [CrossRef] [PubMed]
- Wozniak, S.; Czuczwar, P.; Szkodziak, P.; Milart, P.; Wozniakowska, E.; Paszkowski, T. Elastography in predicting preterm delivery in asymptomatic, low-risk women: A prospective observational study. BMC Pregnancy Childbirth 2014, 14, 238. [Google Scholar] [CrossRef] [PubMed]
- Rus Carlborg, G. Nature of acoustic nonlinear radiation stress. Appl. Phys. Lett. 2014, 105, 121904. [Google Scholar]
- Frigoletto, F.; Auerbach, R.; Brickler, A. Consensus conference: The use of diagnostic ultrasound imaging during pregnancy. JAMA 1984, 252, 669–672. [Google Scholar]
- Nyborg, W.L. Biological effects of ultrasound: Development of safety guidelines. Part II: General review. Ultrasound Med. Biol. 2001, 27, 301–333. [Google Scholar] [CrossRef]
- Mundi, R.; Petis, S.; Kaloty, R.; Shetty, V.; Bhandari, M. Low-intensity pulsed ultrasound: Fracture healing. Indian J. Orthop. 2009, 43, 132. [Google Scholar]
- Padilla, F.; Puts, R.; Vico, L.; Raum, K. Stimulation of bone repair with ultrasound: A review of the possible mechanic effects. Ultrasonics 2014, 54, 1125–1145. [Google Scholar] [CrossRef]
- Massó, P.; Rus, G.; Molina, F. Safety of elastography in fetal medicine: Preliminary study on hypoacusis. Ultrasound Obstet. Gynecol. 2017, 50, 660–661. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Zhang, C.; Li, J.; Cao, X.; Song, D. An experimental study of the potential biological effects associated with 2D shear wave elastography on the neonatal brain. Ultrasound Med. Biol. 2016, 42, 1551–1559. [Google Scholar] [CrossRef] [PubMed]
- Kılıç, F.; Kayadibi, Y.; Yüksel, M.A.; Adaletli, İ.; Ustabaşıoğlu, F.E.; Öncül, M.; Madazlı, R.; Yılmaz, M.H.; Mihmanlı, İ.; Kantarcı, F. Shear wave elastography of placenta: In vivo quantitation of placental elasticity in preeclampsia. Diagn. Interv. Radiol. 2015, 21, 202. [Google Scholar] [CrossRef] [PubMed]
- Carlson, L.C.; Feltovich, H.; Palmeri, M.L.; Dahl, J.J.; Munoz del Rio, A.; Hall, T.J. Estimation of shear wave speed in the human uterine cervix. Ultrasound Obstet. Gynecol. 2014, 43, 452–458. [Google Scholar] [CrossRef]
- Sugitani, M.; Fujita, Y.; Yumoto, Y.; Fukushima, K.; Takeuchi, T.; Shimokawa, M.; Kato, K. A new method for measurement of placental elasticity: Acoustic radiation force impulse imaging. Placenta 2013, 34, 1009–1013. [Google Scholar] [CrossRef]
- Tabaru, M.; Yoshikawa, H.; Azuma, T.; Asami, R.; Hashiba, K. Experimental study on temperature rise of acoustic radiation force elastography. J. Med. Ultrason. 2012, 39, 137–146. [Google Scholar] [CrossRef]
- Palmeri, M.L.; Frinkley, K.D.; Nightingale, K.R. Experimental studies of the thermal effects associated with radiation force imaging of soft tissue. Ultrason. Imaging 2004, 26, 100–114. [Google Scholar] [CrossRef]
- Liu, Y.; Herman, B.A.; Soneson, J.E.; Harris, G.R. Thermal safety simulations of transient temperature rise during acoustic radiation force-based ultrasound elastography. Ultrasound Med. Biol. 2014, 40, 1001–1014. [Google Scholar] [CrossRef]
- Fatemi, M.; Alizad, A.; Greenleaf, J.F. Characteristics of the audio sound generated by ultrasound imaging systems. J. Acoust. Soc. Am. 2005, 117, 1448–1455. [Google Scholar] [CrossRef]
- Stratmeyer, M.E.; Greenleaf, J.F.; Dalecki, D.; Salvesen, K.A. Fetal ultrasound: Mechanical effects. J. Ultrasound Med. 2008, 27, 597–605. [Google Scholar] [CrossRef]
- Muller, M.; Aït-Belkacem, D.; Hessabi, M.; Gennisson, J.L.; Grangé, G.; Goffinet, F.; Lecarpentier, E.; Cabrol, D.; Tanter, M.; Tsatsaris, V. Assessment of the cervix in pregnant women using shear wave elastography: A feasibility study. Ultrasound Med. Biol. 2015, 41, 2789–2797. [Google Scholar] [CrossRef] [PubMed]
- Diguisto, C.; Simon, E.G.; Callé, S.; Ternifi, R.; Remeniéras, J.P.; Hervé, P.; Perrotin, F. Ultrasonic elastography exploration of the foetal brain: A case of atypical choroid plexus papilloma. J. Obstet. Gynaecol. 2017, 37, 525–527. [Google Scholar] [CrossRef] [PubMed]
- Orenstein, B. The ALARA principle and sonography. Radiol. Today 2011, 12, 11. [Google Scholar]
- Peralta, L.; Molina, F.S.; Melchor, J.; Gómez, L.F.; Massó, P.; Florido, J.; Rus, G. Transient elastography to assess the cervical ripening during pregnancy: A preliminary study. Ultraschall Med. 2017, 38, 395–402. [Google Scholar] [CrossRef]
- WHO International. WHO Global Estimates on Prevalence of Hearing Loss. Mortality and Burden of Diseases and Prevention of Blindness and Deafness, 2012; WHO International: Geneva, Switzerland, 2019. [Google Scholar]
- Bercoff, J.; Tanter, M.; Fink, M. Sonic boom in soft materials: The elastic Cerenkov effect. Appl. Phys. Lett. 2004, 84, 2202–2204. [Google Scholar] [CrossRef]
- Bercoff, J.; Tanter, M.; Fink, M. Supersonic shear imaging: A new technique for soft tissue elasticity mapping. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2004, 51, 396–409. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2012; ISBN 3-900051-07-0. [Google Scholar]
- Spreiz, A.; Müller, D.; Zotter, S.; Albrecht, U.; Baumann, M.; Fauth, C.; Erdel, M.; Zschocke, J.; Utermann, G.; Kotzot, D. Phenotypic variability of a deletion and duplication 6q16. 1? q21 due to a paternal balanced ins (7; 6)(p15; q16. 1q21). Am. J. Med. Genet. Part 2010, 152, 2762–2767. [Google Scholar] [CrossRef]
- Bonaglia, M.C.; Ciccone, R.; Gimelli, G.; Gimelli, S.; Marelli, S.; Verheij, J.; Giorda, R.; Grasso, R.; Borgatti, R.; Pagone, F.; et al. Detailed phenotype–genotype study in five patients with chromosome 6q16 deletion: Narrowing the critical region for Prader–Willi-like phenotype. Eur. J. Hum. Genet. 2008, 16, 1443–1449. [Google Scholar] [CrossRef]
- Iafrate, A.J.; Feuk, L.; Rivera, M.N.; Listewnik, M.L.; Donahoe, P.K.; Qi, Y.; Scherer, S.W.; Lee, C. Detection of large-scale variation in the human genome. Nat. Genet. 2004, 36, 949–951. [Google Scholar] [CrossRef]
- Rosenfeld, J.A.; Amrom, D.; Andermann, E.; Andermann, F.; Veilleux, M.; Curry, C.; Fisher, J.; Deputy, S.; Aylsworth, A.S.; Powell, C.M.; et al. Genotype–phenotype correlation in interstitial 6q deletions: A report of 12 new cases. Neurogenetics 2012, 13, 31–47. [Google Scholar] [CrossRef]
- Joint Committee on Infant Hearing. Year 2007 position statement: Principles and guidelines for early hearing detection and intervention programs. Am. Acad. Pediatr. 2007, 120, 898–921.
- Trinidad-Ramos, G.; de Aguilar, V.A.; Jaudenes-Casaubón, C.; Núñez-Batalla, F.; Sequí-Canet, J.M. Recomendaciones de la Comisión para la Detección Precoz de la Hipoacusia (CODEPEH) para 2010. Acta Otorrinolaringol. Ógica Esp. 2010, 61, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Sadler, T.W. Langman’s Medical Embryology; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2006; Volume 333. [Google Scholar]
- Yang, H.; Sung, C.; Shin, D.; Cho, Y.; Jang, C.; Cho, H.H. Newborn hearing screening in prematurity: Fate of screening failures and auditory maturation. Clin. Otolaryngol. 2017, 42, 661–667. [Google Scholar] [CrossRef] [PubMed]
- Cubillana-Herrero, J.D.; Pelegrín-Hernández, J.P.; Soler-Valcarcel, A.; Mínguez-Merlos, N.; Cubillana-Martínez, M.J.; Barrios, Á.N.; Medina-Banegas, A.; Hernandez, J.A.F. The assessment of the Newborn Hearing Screening Program in the Region of Murcia from 2004 to 2012. Int. J. Pediatr. Otorhinolaryngol. 2016, 88, 228–232. [Google Scholar] [CrossRef]
- Vohr, B.R.; Carty, L.M.; Moore, P.E.; Letourneau, K. The Rhode Island hearing assessment program: Experience with statewide hearing screening (1993–1996). J. Pediatr. 1998, 133, 353–357. [Google Scholar] [CrossRef]
- Stewart, D.L.; Mehl, A.; Hall, J.W.; Thomson, V.; Carroll, M.; Hamlett, J. Universal newborn hearing screening with automated auditory brainstem response: A multisite investigation. J. Perinatol. 2000, 20, S128–S131. [Google Scholar] [CrossRef]
- Norton, S.J.; Gorga, M.P.; Widen, J.E.; Folsom, R.C.; Sininger, Y.; Cone-Wesson, B.; Vohr, B.R.; Mascher, K.; Fletcher, K. Identification of neonatal hearing impairment: Evaluation of transient evoked otoacoustic emission, distortion product otoacoustic emission, and auditory brain stem response test performance. Ear Hear. 2000, 21, 508–528. [Google Scholar] [CrossRef]
| N % | |||
|---|---|---|---|
| Mothers | |||
| Age (years) | 32.2(17-42) | 5.4 | |
| Delivery mode: | |||
| Vaginal | 29 (69.0) | ||
| Cesarean section | 13 (31.0) | ||
| Gestational age at delivery (wk) | 39.6 (33.3-41.9) | 1.9 | |
| Gestational age at ARFE test (wk) | 27.5 (6.0-41.0) | 10.8 | |
| Total | 41 | ||
| Newborns | |||
| Sex: | |||
| Female | 25 (59.5) | ||
| Male | 17 (40.5) | ||
| 1-min Apgar score | 8.7 (4-10) | 1.0 | |
| 5-min Apgar score | 9.7 (7-10) | 0.7 | |
| Preterm birth | 1 (2.4) | ||
| Total | 42 |
| Hypoacusia % | Normal Audition N | Total N | |
|---|---|---|---|
| ARFE exposed population | 0 * | 41 | 41 |
| Reference population | 0.5 | - | - |
| OR | REF % | -Test | p-Value |
|---|---|---|---|
| 0 | −0.5 | 0.38 | 0.538 |
| Anthropometric Scores at Birth | Mean/ Range | SD | Z-Scores | WHO Percentile Range | p-Value | ||||
|---|---|---|---|---|---|---|---|---|---|
| F | M | F | M | F | M | F | M | ||
| Weight (g) | 3052.4 (1860–3760) | 3278.8 (2000–4350) | 503.8 | 588.6 | −0.36 | −0.11 | 25–50 | 25–50 | 0.1233 |
| Length (cm) | 49.1 (40–54) | 50.5 (42–58) | 3.1 | 3.6 | 0.16 | 0.18 | 50–75 | 50–75 | 0.1582 |
| BMI (kg/m) | 12.6 (9.8–19.6) | 12.7 (11.3–15.3) | 2.0 | 1.2 | −0.38 | −0.64 | 25–50 | 25–50 | 0.94860 |
| Cranial perimeter (cm) | 33.1 (30–36) | 34.2 (28.5–36.5) | 1.5 | 2.1 | −0.51 | −0.14 | 25–50 | 25–50 | 0.06608 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Massó, P.; Melchor, J.; Rus, G.; Molina, F.S. A Preliminary Study on the Safety of Elastography during Pregnancy: Hypoacusia, Anthropometry, and Apgar Score in Newborns. Diagnostics 2020, 10, 967. https://doi.org/10.3390/diagnostics10110967
Massó P, Melchor J, Rus G, Molina FS. A Preliminary Study on the Safety of Elastography during Pregnancy: Hypoacusia, Anthropometry, and Apgar Score in Newborns. Diagnostics. 2020; 10(11):967. https://doi.org/10.3390/diagnostics10110967
Chicago/Turabian StyleMassó, Paloma, Juan Melchor, Guillermo Rus, and Francisca Sonia Molina. 2020. "A Preliminary Study on the Safety of Elastography during Pregnancy: Hypoacusia, Anthropometry, and Apgar Score in Newborns" Diagnostics 10, no. 11: 967. https://doi.org/10.3390/diagnostics10110967
APA StyleMassó, P., Melchor, J., Rus, G., & Molina, F. S. (2020). A Preliminary Study on the Safety of Elastography during Pregnancy: Hypoacusia, Anthropometry, and Apgar Score in Newborns. Diagnostics, 10(11), 967. https://doi.org/10.3390/diagnostics10110967







