Comet Pond II: Synergistic Intersection of Concentrated Extraterrestrial Materials and Planetary Environments to Form Procreative Darwinian Ponds
Abstract
1. Introduction and Background
1.1. The Dilution Issue
1.2. Comets and Cometoids
2. New Results and Developments
2.1. Solar System Evolution
2.2. Comets: Chemical Composition and Physical Properties
2.2.1. Organics
2.2.2. Inorganics
Fe and Fe-S
Phosphorus
Trace Minerals and Elements
2.3. Early Earth Environment
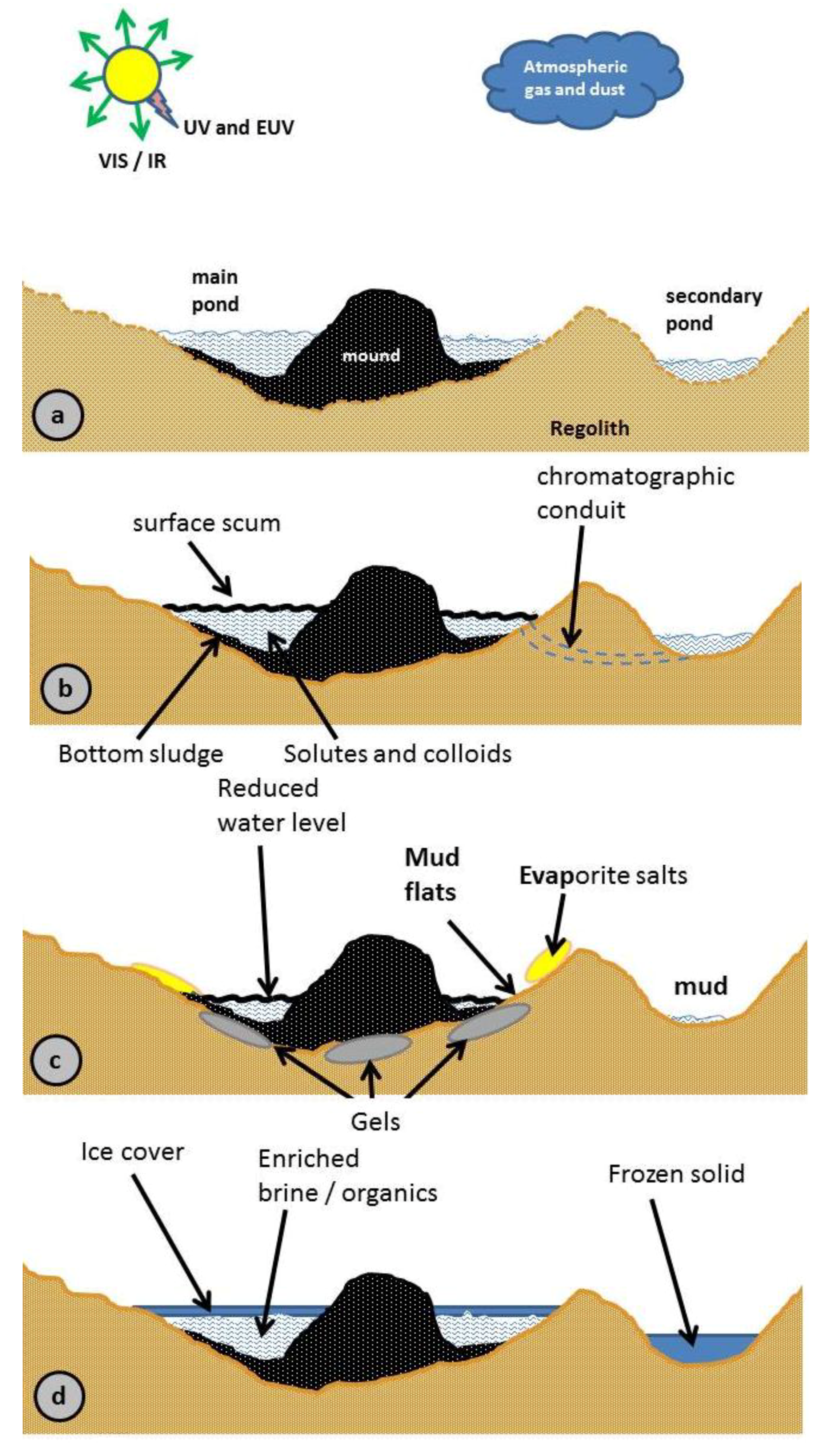
2.4. Comet Pond Description
2.4.1. Comet Pond Description
2.4.2. Geophysical and Physicochemical Environment
3. Emergence of Life in the Pond
3.1. Relevance to Other Theories of Prebiotic Synthesis Leading to Life
3.2. Concept of Macrobiont
4. Advantages and Disadvantages of the Comet Pond Model
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References and Note
- Clark, B.C. Primeval procreative comet pond. Orig. Life Evol. Biosph. 1988, 18, 209–238. [Google Scholar] [CrossRef] [PubMed]
- Darwin, C., in a Letter to Joseph Hooker. Feb. 1, 1871, Darwin Correspondence Project, DCP- LETT-7471.
- Miller, S.L. A production of amino acids under possible primitive earth conditions. Science 1953, 117, 528–529. [Google Scholar] [CrossRef] [PubMed]
- Deamer, D. First Life: Discovering the Connections between Stars, cells, and How Life Began; University of California Press: Berkeley, CA, USA, 2012. [Google Scholar]
- Chyba, C.F.; Sagan, C. Comets as a source of prebiotic organic molecules for the Early Earth, 1997. In Comets and the Origin and Evolution of Life, 2nd ed.; Thomas, P.J., Chyba, C.F., McKay, C.P., Eds.; Springer: Heidelberg, Germany, 2006; pp. 147–174. [Google Scholar]
- Cockell, C.S. The origin and emergence of life under impact bombardment. Philos. Trans. R. Soc. B 2006, 361, 1845–1856. [Google Scholar] [CrossRef] [PubMed]
- Damer, B. A field trip to the Archaean in search of Darwin’s Warm Little Pond. Life 2016, 6, 21. [Google Scholar] [CrossRef] [PubMed]
- Deamer, D.W.; Georgiou, C.D. Hydrothermal conditions and the origin of cellular life. Astrobiology 2015, 15, 1091–1095. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, J.D. Studies on the origin of life—The end of the beginning. Nat. Rev. Chem. 2017, 1, 1–7. [Google Scholar] [CrossRef]
- Pearce, B.K.; Pudritz, R.E.; Ralph, E.; Semenov, D.A.; Henning, T.K. Origin of the RNA world: The fate of nucleobases in warm little ponds. Proc. Natl. Acad. Sci. USA 2017, 114, 11327–11332. [Google Scholar] [CrossRef] [PubMed]
- Chyba, C.F.; Thomas, P.J.; Brookshaw, L.; Sagan, C. Cometary delivery of organic molecules to the Early Earth. Science 1990, 249, 336–373. [Google Scholar] [CrossRef]
- Spohn, T.; Knollenberg, J.; Ball, A.J.; Banaszkiewicz, M.; Benkhoff, J.; Grott, M.; Grygorczuk, J.; Hüttig, C.; Hagermann, A.; Kargl, G.; et al. Thermal and mechanical properties of the near-surface layers of comet 67P/Churyumov-Gerasimenko. Science 2015, 349, aab0464. [Google Scholar] [CrossRef] [PubMed]
- O'Brien, D.P.; Izidoro, A.; Jacobson, S.A.; Raymond, S.N.; Rubie, D.C. The delivery of water during terrestrial planet formation. Space Sci. Rev. 2018, 214. [Google Scholar] [CrossRef]
- Prettyman, T.H.; Yamashita, N.; Toplis, M.J.; McSween, H.Y.; Schörghofer, N.; Marchi, S.; Feldman, W.C.; Castillo-Rogez, J.; Forni, O.; Lawrence, D.J.; et al. Extensive water ice within Ceres’ aqueously altered regolith: Evidence from nuclear spectroscopy. Science 2017, 355, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Jewitt, D.; Weaver, H.; Mutchler, M.; Larson, S.; Agarwa, J. Hubble space telescope observations of Main-Belt Comet (596) Scheila. Astrophys. J. Lett. 2011, 733. [Google Scholar] [CrossRef]
- Walsh, K.J.; Morbidelli, A.; Raymond, S.N.; O’Brien, D.P.; Mandell, A.M. A low mass for Mars from Jupiter’s early gas-driven migration. Nature 2011, 475, 206–209. [Google Scholar] [CrossRef] [PubMed]
- Bottke, W.F.; Levison, H.F.; Nesvorn’y, D.; Dones, L. Can planetesimals left over from terrestrial planet formation produce the lunar Late Heavy Bombardment? Icarus 2007, 190, 203–223. [Google Scholar] [CrossRef]
- Morbidelli, A.; Nesvorny, D.; Laurenz, V.; Marchi, S.; Rubie, D.C.; Elkins-Tanton, L.; Wieczorek, M.; Jacobson, S. The timeline of the Lunar bombardment-revisited. Icarus 2018, 305, 262–276. [Google Scholar] [CrossRef]
- Zolensky, M.; Johnson, J.; Ziegler, K.; Chan, Q.; Kebukawa, Y.; Bottke, W.; Fries, M.; Martinez, J.; Lee, L. Meteoritic evidence for injection of trans-Neptunian objects into the inner Solar System. In Proceedings of the 49th Lunar and Planetary Science Conference, The Woodlands, TX, USA, 19–23 March 2018. [Google Scholar]
- Kring, D.A.; Cohen, B.A. Cataclysmic bombardment throughout the inner solar system 3.9–4.0 Ga. J. Geophys. Res. Planets 2002, 107, 4-1–4-6. [Google Scholar] [CrossRef]
- Draine, B.T. Interstellar dust grains. Annu. Rev. Astron. Astrophys. 2003, 41, 241–289. [Google Scholar] [CrossRef]
- Langevin, Y.; Hilchenbach, M.; Ligiera, N.; Merouane, S.; Hornung, K.; Engrand, C.; Schulz, R.; Kissel, J.; Rynö, J.; Eng, P. COSIMA team. Typology of dust particles collected by the COSIMA mass spectrometer in the inner coma of 67P/Churyumov Gerasimenko. Icarus 2016, 271, 76–97. [Google Scholar]
- Clark, B.C.; Green, S.; Economou, T.; Sandford, S.; Zolensky, M.; McBride, N.; Brownlee, D. Release and fragmentation of aggregates to produce heterogeneous, lumpy coma streams. J. Geophys. Res. Planets 2004, 109. [Google Scholar] [CrossRef]
- Green, S.F.; McDonnell, J.A.M.; McBride, N.; Colwell, M.T.S.H.; Tuzzolino, A.J.; Economou, T.E.; Tsou, P.; Clark, B.C.; Brownlee, D.E. The dust mass distribution of Comet 81P/Wild 2. J. Geophys. Res. Planets 2004, 109. [Google Scholar] [CrossRef]
- Alivisatos, A.P. Semiconductor clusters, nanocrystals, and quantum dots. Science 1996, 271, 933–937. [Google Scholar] [CrossRef]
- Brownlee, D. The Stardust Mission: Analyzing samples from the edge of the Solar System. Annu. Rev. Earth Planet. Sci. 2014, 42, 179–205. [Google Scholar] [CrossRef]
- Kwok, S. Complex organics in space from Solar System to distant galaxies. Astron. Astrophys. Rev. 2016, 24. [Google Scholar] [CrossRef]
- Kwok, S. Abiotic synthesis of complex organics in the universe. Nat. Astron. 2017, 1, 642–643. [Google Scholar] [CrossRef]
- Schmitt-Kopplin, P.; Gabelica, Z.; Gougeon, R.D.; Fekete, A.; Kanawati, B.; Harir, M.; Gebefuegi, I.; Eckel, G.; Hertkorn, N. High molecular diversity of extraterrestrial organic matter in Murchison meteorite revealed 40 years after its fall. Proc. Natl. Acad. Sci. USA 2010, 107, 2763–2768. [Google Scholar] [CrossRef] [PubMed]
- Schmitt-Kopplin, P.; Harir, M.; Kanawati, B.; Gougeon, R.; Moritz, F.; Hertkorn, N.; Clary, S.; Gebefügi, I.; Gabelica, Z. Analysis of extraterrestrial organic matter in Murchison meteorite: A progress report. In Astrobiology: An Evolutionary Approach; Kolb, V.M., Ed.; CRC Press/Taylor &Francis: Boca Raton, FL, USA, 2014; pp. 63–82. [Google Scholar]
- Cooper, G.W.; Onwo, W.M.; Cronin, J.R. Alkyl phosphonic acids and sulfonic acids in the Murchison meteorite. Geochim. Cosmochim. Acta 1992, 56, 4109–4115. [Google Scholar] [CrossRef]
- Mumma, M.J.; Charnley, S.B. The Chemical composition of comets—Emerging taxonomies and natal heritage. Annu. Rev. Astron. Astrophys. 2011, 49, 471–524. [Google Scholar] [CrossRef]
- Elsila, J.E.; Glavin, D.P.; Dworkin, J.P. Cometary glycine detected in samples returned by Stardust. Meteorit. Planet. Sci. 2009, 44, 1323–1330. [Google Scholar] [CrossRef]
- Sandford, S.A.; Aléon, J.; Alexander, C.M.D.; Araki, T.; Bajt, S.; Baratta, G.A.; Borg, J.; Bradley, J.P.; Brownlee, D.E.; Brucato, J.R.; et al. Organics captured from Comet 81P/Wild 2 by the Stardust spacecraft. Science 2006, 314, 1720–1724. [Google Scholar] [CrossRef] [PubMed]
- Cody, G.D.; Ade, H.; Alexander, M.D.; Araki, T.; Butterworth, A.; Fleckenstein, H.; Flynn, G.; Gilles, M.K.; Jacobsen, C.; Kilcoyne, A.L.D.; et al. Quantitative organic and light-element analysis of Comet 81P/Wild 2 particles Using C-, N-, and O-µ-XANES. Meteorit. Planet. Sci. 2008, 43, 353–365. [Google Scholar] [CrossRef]
- Kissel, J.; Krueger, F.R.; Silén, J.; Clark, B.C. The cometary and interstellar dust analyzer at Comet 81P/Wild 2. Science 2004, 304, 1774–1776. [Google Scholar] [CrossRef] [PubMed]
- Altwegg, K.; Balsiger, H.; Berthelier, J.J.; Bieler, A.; Calmonte, U.; Fuselier, S.A.; Goesmann, F.; Gasc, S.; Gombosi, T.I.; Le Roy, L.; et al. Organics in comet 67P—A first comparative analysis of mass spectra from ROSINA–DFMS, COSAC and Ptolemy. Mon. Not. R. Astron. Soc. 2017, 469, S130–S141. [Google Scholar] [CrossRef]
- Fayolle, E.C.; Öberg, K.I.; Jørgensen, J.K.; Altwegg, K.; Calcutt, H.; Müller, H.S.; Rubin, M.; van der Wiel, M.H.; Bjerkeli, P.; Bourke, T.L.; et al. Protostellar and cometary detection of organohalogens. Nat. Astron. Lett. 2017, 1, 703–708. [Google Scholar] [CrossRef]
- Altwegg, K.; Balsiger, H.; Bar-Nun, A.; Berthelier, J.-J.; Bieler, A.; Bochsler, P.; Briois, C.; Calmonte, U.; Combi, M.R.; Cottin, H.; et al. Prebiotic chemicals—Amino acid and phosphorus—In the coma of comet 67P/Churyumov-Gerasimenko. Sci. Adv. 2016, 2, e1600285. [Google Scholar] [CrossRef] [PubMed]
- Bardyn, A.; Baklouti, D.; Cottin, H.; Fray, N.; Briois, C.; Paquette, J.; Stenzel, O.; Engrand, C.; Fischer, H.; Hornung, K.; et al. Carbon-rich dust in comet 67P/Churyumov-Gerasimenko measured by COSIMA/Rosetta. Mon. Not. R. Astron. Soc. 2017, 469, S712–S722. [Google Scholar] [CrossRef]
- Clark, B.C.; Mason, L.W.; Kissel, J. Systematics of the “CHON” and other light-element particle populations in Comet P/Halley. Astron. Astrophys. 1987, 187, 779–784. [Google Scholar]
- Engrand, C.; Charon, E.; Duprat, J.; Dartois, E.; Leroux, H.; Benzerara, K.; Le Guillou, C.; Bernard, S.; Swaraj, S.; Belkhou, R.; et al. Ultracarbonaceous Antarctic micrometeorites (UCAMMS): Clues for their origin. In Proceedings of the 49th Lunar and Planetary Science Conference, The Woodlands, TX, USA, 19–23 March 2018. [Google Scholar]
- Dartois, E.; Engrand, C.; Duprat, J.; Godard, M.; Charon, E.; Delauche, L.; Sandt, C.; Borondics, F. Dome C ultracarbonaceous Antarctic micrometeorites Infrared and Raman fingerprints. Astron. Astrophys. 2018, 609, A65. [Google Scholar] [CrossRef]
- Flynn, G.J.; Bleuet, P.; Borg, J.; Bradley, J.P.; Brenker, F.E.; Brennan, S.; Bridges, J.; Brownlee, D.E.; Bullock, E.S.; Burghammer, M.; et al. Elemental compositions of Comet 81P/Wild 2 samples collected by Stardust. Science 2006, 314, 1731–1735. [Google Scholar] [CrossRef] [PubMed]
- Ishii, H.A.; Bradley, J.P. Pristine amorphous gems in anhydrous interplanetary dust particles are very underdense. In Proceedings of the 49th Lunar and Planetary Science Conference, The Woodlands, TX, USA, 19–23 March 2018. [Google Scholar]
- Fernández-García, C.; Coggins, A.J.; Powner, M.A. A chemist’s perspective on the role of phosphorus at the origins of life. Life 2017, 7, 31. [Google Scholar] [CrossRef] [PubMed]
- Pasek, M.A.; Lauretta, D.S. Aqueous corrosion of phosphide minerals from iron meteorites: A highly reactive source of prebiotic phosphorus on the surface of the early Earth. Astrobiology 2005, 5, 515–535. [Google Scholar] [CrossRef] [PubMed]
- Pasek, M.A.; Gull, M.; Herschy, B. Phosphorylation on the early earth. Chem. Geol. 2017, 475, 149–170. [Google Scholar] [CrossRef]
- Goldford, J.E.; Hartman, H.; Smith, T.F.; Segrè, D. Remnants of an ancient metabolism without phosphate. Cell 2017, 168, 1126–1134. [Google Scholar] [CrossRef] [PubMed]
- Degtyarenko, K. Bioinorganic motifs: Towards functional classification of metalloproteins. Bioinformatics 2000, 16, 851–864. [Google Scholar] [CrossRef] [PubMed]
- Crans, D.C.; Smee, J.J.; Gaidamauskas, E.; Yang, L. The chemistry and biochemistry of vanadium and the biological activities exerted by vanadium compounds. Chem. Rev. 2004, 104, 849–902. [Google Scholar] [CrossRef] [PubMed]
- Sagan, C.; Mullen, G. Earth and Mars: Evolution of atmospheres and surface temperatures. Science 1972, 177, 52–56. [Google Scholar] [CrossRef] [PubMed]
- Charnay, B.; Le Hir, G.; Fluteau, F.; Forget, F.; Catling, D.C. A warm or a cold early Earth? New insights from a 3-D climate-carbon model. Earth Planet. Sci. Lett. 2017, 474, 97–109. [Google Scholar] [CrossRef]
- Wordsworth, R.; Pierrehumbert, R. Hydrogen-nitrogen Greenhouse warming in Earth’s early atmosphere. Science 2013, 339, 64–67. [Google Scholar] [CrossRef] [PubMed]
- Tian, F.; Tian, F.; Toon, O.B.; Pavlov, A.A.; De Sterck, H. A hydrogen-rich early Earth atmosphere. Science 2005, 308, 1014–1017. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, R.M.; Kopparapu, R.; Zugger, M.E.; Robinson, T.D.; Freedman, R.; Kasting, J.F. Warming early Mars with CO2 and H2. Nat. Geosci. 2014, 7, 59–63. [Google Scholar] [CrossRef]
- Zahnle, K.; Schaefer, L.; Fegley, B. Earth’s earliest atmosphere. Cold Spring Harb. Perspect. Biol. 2010, 2, a004895. [Google Scholar] [CrossRef] [PubMed]
- Kasting, J.F. Atmospheric composition of Hadean-early Archean earth: The importance of CO. Geol. Soc. Am. Spec. Pap. 2014, 504, 19–28. [Google Scholar]
- Ranjan, S.; Todd, Z.R.; Sutherland, J.D.; Sasselov, D.D. Sulfidic anion concentrations on early Earth for surficial origins-of-life chemistry. Astrobiology 2018. [Google Scholar] [CrossRef] [PubMed]
- Ross, D.S.; Deamer, D. Dry/Wet Cycling and the thermodynamics and kinetics of prebiotic polymer synthesis. Life 2016, 6, 28. [Google Scholar] [CrossRef] [PubMed]
- Van Kranendonk, M.J.; Deamer, D.; Djokic, T. Life springs. Sci. Am. 2017, 317, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Vlassov, A.V.; Johnston, B.H.; Landweber, L.F.; Kazakov, S.A. Ligation activity of fragmented ribozymes in frozen solution: Implication for the RNA World. Nucleic Acids Res. 2004, 32, 2966–2974. [Google Scholar] [CrossRef] [PubMed]
- Mutschler, H.; Wochner, A.; Holliger, P. Freeze-thaw cycles as drivers of complex ribozyme assembly. Nat. Chem. 2017, 7, 502–508. [Google Scholar] [CrossRef] [PubMed]
- Higgs, P.S. The Effect of limited diffusion and wet-dry cycling on reversible polymerization reactions: Implications for prebiotic synthesis of nucleic acids. Life 2016, 6, 24. [Google Scholar] [CrossRef] [PubMed]
- Solomons, T.W.G.; Fryhle, C.B.; Snyder, S.A. Organic Chemistry, 11th ed.; Wiley: Hoboken, NJ, USA, 2014; pp. 605–607. [Google Scholar]
- Benner, S.A.; Kim, H.-J.; Carrigan, M.A. Asphalt, water, and the prebiotic synthesis of ribose, ribonucleosides, and RNA. Acct. Chem. Res. 2012, 45, 2025–2034. [Google Scholar] [CrossRef] [PubMed]
- Benner, S.A. Paradoxes in the origin of life. Orig. Life Evol. Biosph. 2014, 44, 339–343. [Google Scholar] [CrossRef] [PubMed]
- Clark, B.C. Macrobiont—The pond as proto-organism. In Prebiological Self Organization of Matter; Ponnamperuma, C., Erlich, F.R., Eds.; A Deepak Publishing: Hampton, VA, USA, 1990; pp. 115–116. [Google Scholar]
- Clark, B.C. Comet pond as macrobiont. In Book of Abstracts, The World Space Congress. Proceedings of the 29th Plenary Meeting of Committee on Space Research COSPAR, Washington, DC, USA, 28 August–5 September 1992; Pergamon Press: Oxford, UK, 1992; pp. 577–578. [Google Scholar]
- Clark, B.C.; Morris, R.V.; McLennan, S.M.; Gellert, R.; Jolliff, B.; Knoll, A.H.; Squyres, S.W.; Lowenstein, T.K.; Ming, D.W.; Tosca, N.J. Chemistry and mineralogy of outcrop at Meridiani Planum, Mars. Earth Planet. Sci. Lett. 2005, 240, 73–94. [Google Scholar] [CrossRef]
- Knoll, A.H.; Carr, M.; Clark, B.; Des Marais, D.J.; Farmer, J.D.; Fischer, W.W.; Grotzinger, J.P.; McLennan, S.M.; Malin, M.; Schröder, C.; et al. Astrobiological implications of Meridiani sediments. Earth Planet. Sci. Lett. 2005, 240, 179–189. [Google Scholar] [CrossRef]
- Joyce, G.F.; Orgel, L.E. Prospects for understanding of RNA world. In The RNA world: The Nature of Modern RNA Suggests a Prebiotic RNA World; Gesteland, R.F., Atkins, J.F., Eds.; Cold Spring Harbor University Press: Cold Spring Harbor, NY, USA, 1993; pp. 1–25. [Google Scholar]
- Orgel, L.E. Prebiotic chemistry and the origin of the RNA world. Crit. Rev. Biochem. Mol. Biol. 2004, 9, 99–123. [Google Scholar]
- Robertson, M.P.; Joyce, G.F. The origins of the RNA World. Cold Spring Harb. Perspect. Biol. 2012, 4, a003608. [Google Scholar] [CrossRef] [PubMed]
- Pressman, A.; Blanco, C.; Chen, I.A. The RNA World as a model system to study the origin of life. Curr. Biol. 2015, 25, R953–R963. [Google Scholar] [CrossRef] [PubMed]
- Joyce, G.F. Forty years of in vitro evolution. Angew. Chem. Int. Ed. 2007, 46, 6420–6436. [Google Scholar] [CrossRef] [PubMed]
- Szostak, J.W. The narrow road to the deep past: In search of the chemistry of the origin of life. Angew. Chem. Int. Ed. 2017, 56, 1107–11043. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.L.; Orgel, L.E. The Origins of Life on the Earth; Prentice Hall: Englewood Cliffs, NJ, USA, 1974. [Google Scholar]
- Peretó, J. Out of fuzzy chemistry: From prebiotic chemistry to metabolic networks. Chem. Soc. Rev. 2012, 41, 5394–5403. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Mirazo, K.; Briones, C.; de la Escosura, A. Prebiotic systems chemistry: New perspectives for the origins of life. Chem. Rev. 2014, 114, 285–366. [Google Scholar] [CrossRef] [PubMed]
- De Duve, C. Clues from present-day biology: The thioester world. In The Molecular Origins of Life: Assembling Pieces of the Puzzle; Brack, A., Ed.; Cambridge University Press: Cambridge, UK, 2000; pp. 219–236. [Google Scholar]
- Wächtershäuser, G. From volcanic origins of chemoautotrophic life to Bacteria, Archaea and Eukarya. Philos. Trans. R. B Biol. Sci. 2006, 361, 1787–1808. [Google Scholar] [CrossRef] [PubMed]
- Des Marais, D.J. When did photosynthesis emerge on Earth? Science 2000, 289, 1703–1705. [Google Scholar] [CrossRef]
- Camacho, A.; Walter, X.A.; Picazo, A.; Zopfi, J. Photoferrotrophy: Remains of an ancient photosynthesis in modern environments. Front. Microbiol. 2017, 8, 323. [Google Scholar] [CrossRef] [PubMed]
- Ferris, J.P. Catalyzed RNA synthesis for the RNA world. In The Molecular Origins of Life: Assembling Pieces of the Puzzle; Brack, A., Ed.; Cambridge University Press: Cambridge, UK, 2000; pp. 255–268. [Google Scholar]
- Kolb, V.; Zhang, S.; Xu, Y.; Arrhenius, G. Mineral induced phosphorylation of glycolate ion—A metaphor in chemical evolution. Orig. Life Evol. Biosph. 1997, 27, 485–503. [Google Scholar] [CrossRef] [PubMed]
- Cleaves, H.J. Prebiotic chemistry: Geochemical context and reaction screening. Life 2013, 3, 331–345. [Google Scholar] [CrossRef] [PubMed]
- Cleaves, H.J. Prebiotic synthesis of biochemical compounds: An overview. In Astrobiology: An Evolutionary Approach; Kolb, V.M., Ed.; CRC Press/Taylor &Francis: Boca Raton, FL, USA, 2014; pp. 83–117. [Google Scholar]
- Benner, S.A. Understanding nucleic acids using synthetic chemistry. Acc. Chem. Res. 2004, 37, 784–797. [Google Scholar] [CrossRef] [PubMed]
- Ricardo, A.; Carrigan, M.A.; Olcott, A.N.; Benner, S.A. Borate minerals stabilize ribose. Science 2004, 303, 196. [Google Scholar] [CrossRef] [PubMed]
- Gasda, P.J.; Haldeman, E.B.; Wiens, R.C.; Rapin, W.; Bristow, T.F.; Bridges, J.C.; Schwenzer, S.P.; Clark, B.; Herkenhoff, K.; Frydenvang, J. In situ detection of boron by ChemCam on Mars. Geophys. Res. Lett. 2017, 44, 8739–8748. [Google Scholar] [CrossRef]
- Powner, M.W.; Sutherland, J.D.; Szostak, J.W. The origins of nucleotides. Synlett 2011, 14, 1956–1964. [Google Scholar] [CrossRef]
- Patel, B.H.; Percivalle, C.; Ritson, D.J.; Duffy, C.D.; Sutherland, J.D. Common origins of RNA, protein and lipid precursors in a cyanosulfidic protometabolism. Nat. Chem. 2015, 7, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Powner, M.W.; Gerland, B.; Sutherland, J.D. Synthesis of activated pyrimidine ribonucleotides in prebiotically plausible conditions. Nature 2009, 459, 239–242. [Google Scholar] [CrossRef] [PubMed]
- Coggins, A.J.; Powner, M.W. Prebiotic synthesis of phosphoenol pyruvate by α-phosphorylation-controlled triose glycolysis. Nat. Chem. 2017, 9, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Islam, S.; Powner, M.W. Prebiotic systems chemistry: Complexity overcoming clutter. Chem 2017, 2, 470–501. [Google Scholar] [CrossRef]
- Berger, J.A.; Schmidt, M.E.; Gellert, R.; Boyd, N.I.; Desouza, E.D.; Flemming, R.L.; Izawa, M.R.; Ming, D.W.; Perrett, G.M.; Rampe, E.B.; et al. Zinc and germanium in the sedimentary rocks of Gale Crater on Mars indicate hydrothermal enrichment followed by diagenetic fractionation. J. Geophys. Res. Planets 2017, 122, 1747–1772. [Google Scholar] [CrossRef]
- Menor-Salván, C.; Marin-Yaseli, M.R. Prebiotic chemistry in eutectic solutions at the water-ice matrix. Chem. Soc. Rev. 2012, 41, 5404–5415. [Google Scholar] [CrossRef] [PubMed]
- Kolb, V.M. Prebiotic chemistry: In water and in the solid state. In Astrobiology: An Evolutionary Approach; Kolb, V.M., Ed.; CRC Press/Taylor &Francis: Boca Raton, FL, USA, 2014; pp. 199–216. [Google Scholar]
- Dass, A.V.; Jaber, M.; Brack, A.; Foucher, F.; Kee, T.P.; Georgelin, T.; Westall, F. Potential role of inorganic confined environments in prebiotic phosphorylation. Life 2018, 8, 7. [Google Scholar] [CrossRef] [PubMed]
- Deamer, D.W. Membrane compartments in prebiotic evolution. In The Molecular Origins of Life: Assembling Pieces of the Puzzle; Brack, A., Ed.; Cambridge University Press: Cambridge, UK, 2000; pp. 189–205. [Google Scholar]
- Colmana, D.R.; Poudela, S.; Stamps, B.W.; Boyda, E.S.; Spear, J.R. The deep, hot biosphere: Twenty-five years of retrospection. Proc. Natl. Acad. Sci. USA 2017, 114, 6895–6903. [Google Scholar] [CrossRef] [PubMed]
- McCollom, T.M. Abiotic methane formation during experimental serpentinization of olivine. Proc. Natl. Acad. Sci. USA 2016, 113, 13965–13970. [Google Scholar] [CrossRef] [PubMed]
- Drake, M.J.; Righter, K. Determining the composition of the Earth. Nature 2002, 416, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Schwartzman, D.W.; Lineweaver, C.H. The hyperthermophilic origin of life revisited. Biochem. Soc. Trans. 2004, 32, 168–171. [Google Scholar] [CrossRef] [PubMed]
- Feller, G. Cryosphere and psychrophiles: Insights into a cold origin of life? Life 2017, 7, 25. [Google Scholar] [CrossRef] [PubMed]
- Kwon, J.Y.; Bercovici, H.L.; Cunningham, K.; Varnum, M.E.W. How will we react to the discovery of extraterrestrial life? Front. Psychol. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Lauretta, D.S. OSIRIS-REX: The Journey to Asteroid Bennu and Back. Elements 2017, 13, 212. [Google Scholar] [CrossRef][Green Version]



© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clark, B.C.; Kolb, V.M. Comet Pond II: Synergistic Intersection of Concentrated Extraterrestrial Materials and Planetary Environments to Form Procreative Darwinian Ponds. Life 2018, 8, 12. https://doi.org/10.3390/life8020012
Clark BC, Kolb VM. Comet Pond II: Synergistic Intersection of Concentrated Extraterrestrial Materials and Planetary Environments to Form Procreative Darwinian Ponds. Life. 2018; 8(2):12. https://doi.org/10.3390/life8020012
Chicago/Turabian StyleClark, Benton C., and Vera M. Kolb. 2018. "Comet Pond II: Synergistic Intersection of Concentrated Extraterrestrial Materials and Planetary Environments to Form Procreative Darwinian Ponds" Life 8, no. 2: 12. https://doi.org/10.3390/life8020012
APA StyleClark, B. C., & Kolb, V. M. (2018). Comet Pond II: Synergistic Intersection of Concentrated Extraterrestrial Materials and Planetary Environments to Form Procreative Darwinian Ponds. Life, 8(2), 12. https://doi.org/10.3390/life8020012




