Prevalence of Serum Antibody Titers against Core Vaccine Antigens in Italian Dogs
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Study Protocol
2.2. Detection of Specific Antibodies by VacciCheck
2.3. Statistical Analysis
3. Results
3.1. Dog Population
3.2. Antibody Titers and Protection of the Entire Cohort
3.3. Results According to the Different Variables
3.3.1. Sex and Reproductive Status
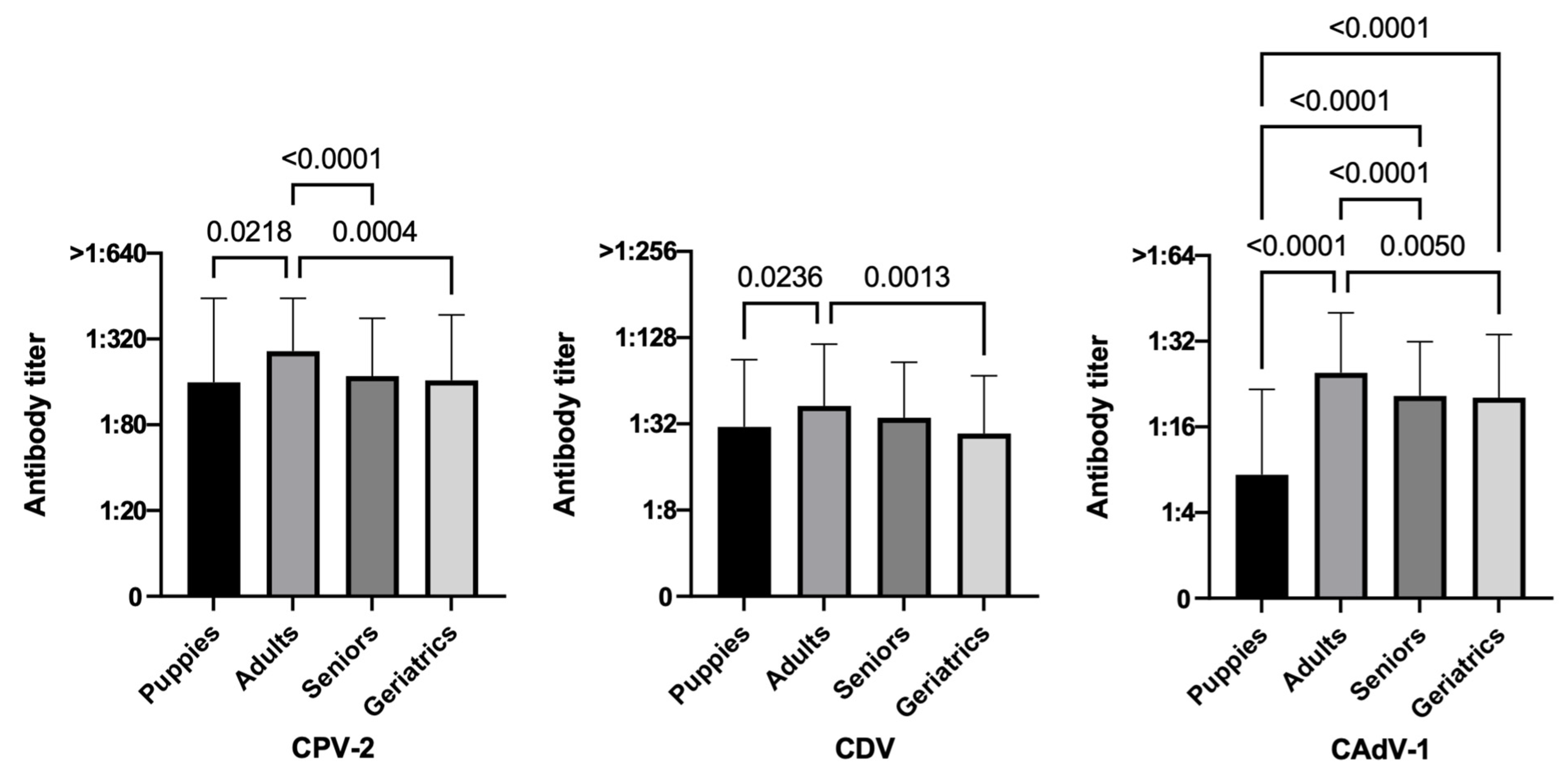
3.3.2. Age
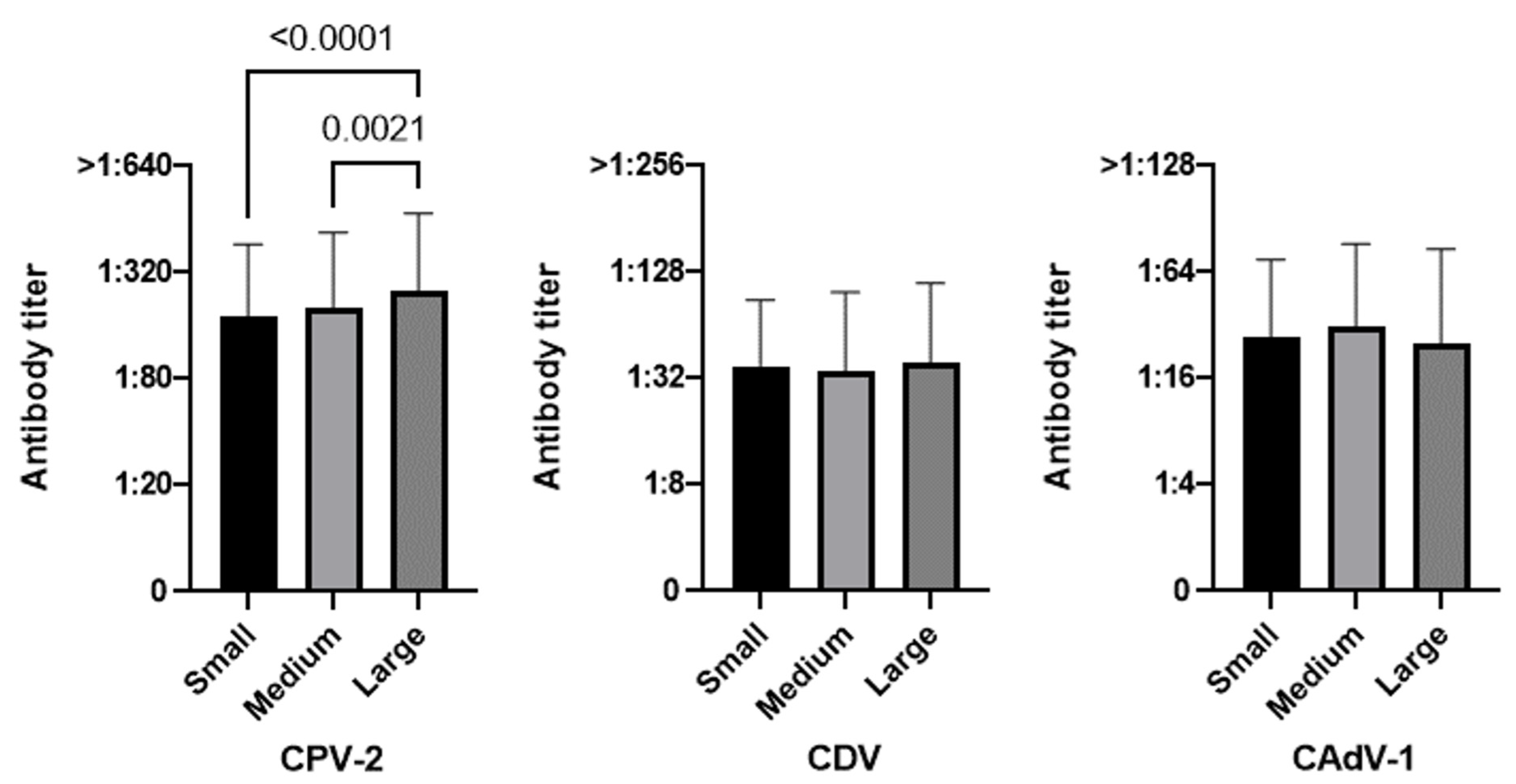
3.3.3. Size
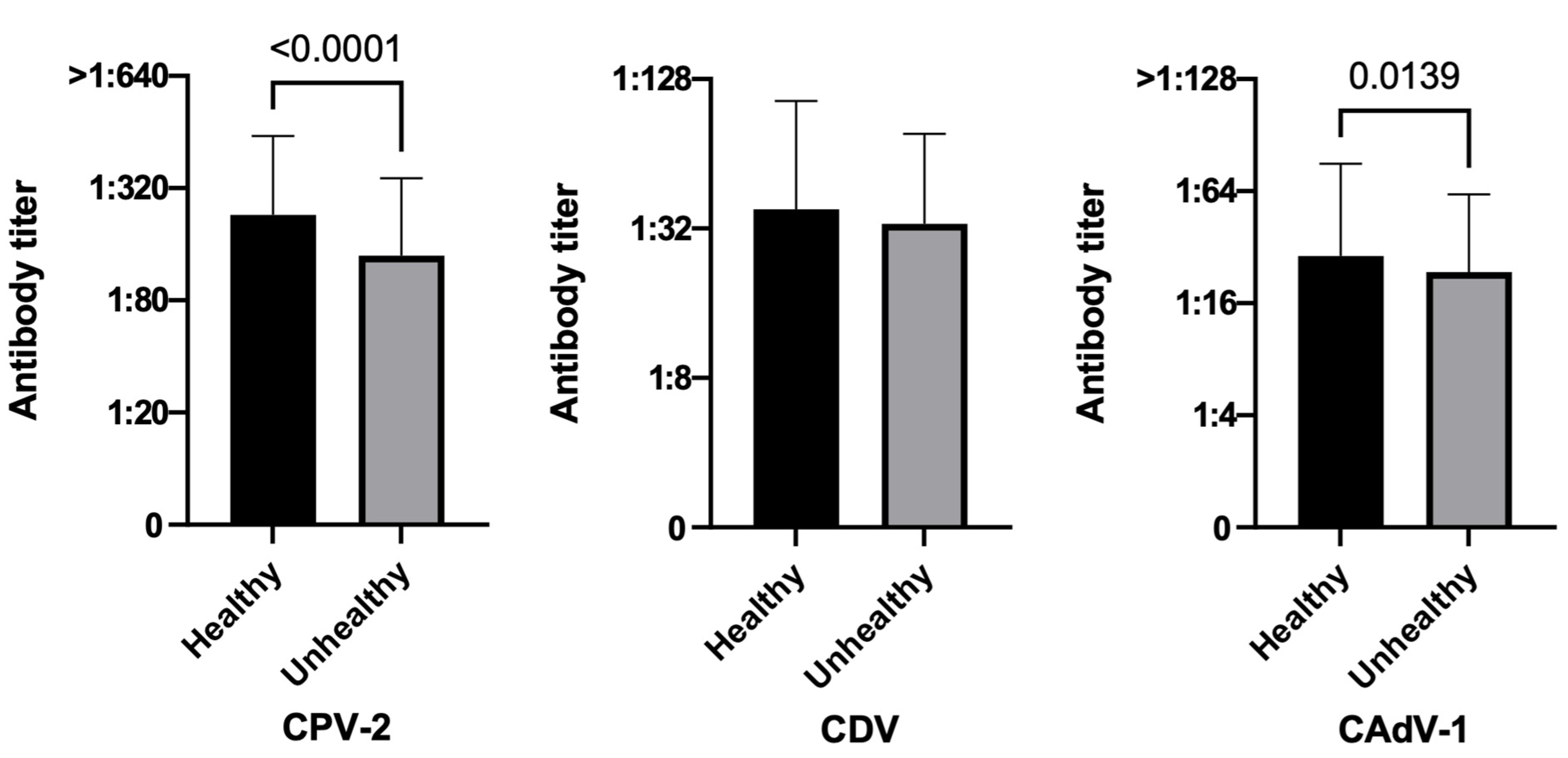
3.3.4. Health Status
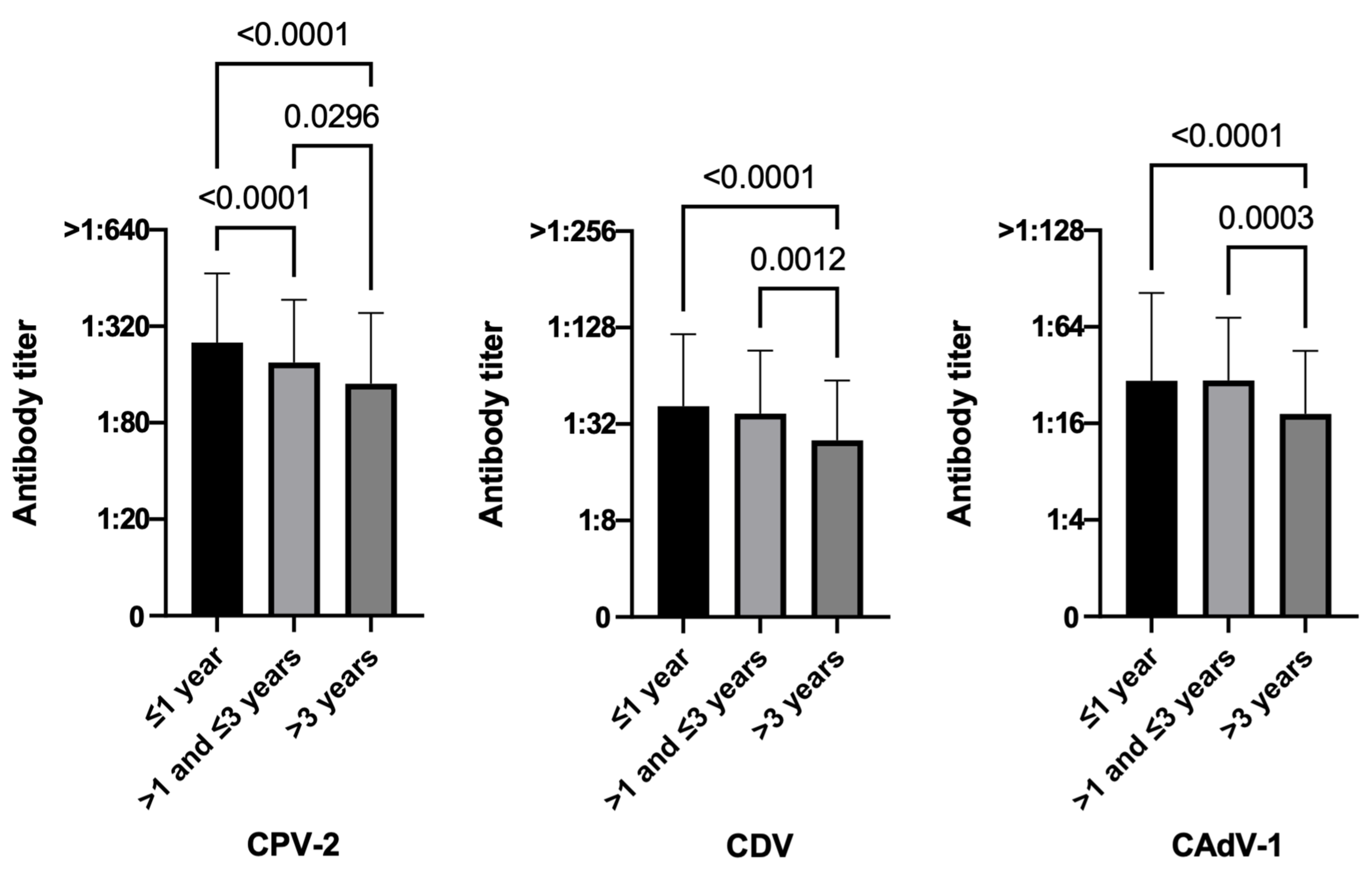
3.3.5. Time Elapsed since the Last Vaccination
3.3.6. Breeds with High Prevalence of Non-Responder Subjects
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Day, M.J.; Horzinek, M.C.; Schultz, R.D.; Squires, R.A. WSAVA Guidelines for the Vaccination of Dogs and Cats. J. Small Anim. Pract. 2016, 57, E1–E45. [Google Scholar] [CrossRef]
- Ellis, J.; Marziani, E.; Aziz, C.; Brown, C.M.; Cohn, L.A.; Lea, C.; Moore, G.E.; Taneja, N. 2022 AAHA Canine Vaccination Guidelines. J. Am. Anim. Hosp. Assoc. 2022, 58, 213–230. [Google Scholar] [CrossRef] [PubMed]
- Australian Veterinary Association (AVA). Vaccination of Dogs and Cats. Available online: https://www.ava.com.au/policy-advocacy/policies/companion-animals-health/vaccination-of-dogs-and-cats/ (accessed on 16 January 2023).
- British Veterinary Association (BVA). Getting Your Pet Vaccinated. Available online: https://www.bva.co.uk/media/2649/client_leaflet_9_-_getting_your_pet_vaccinated.pdf (accessed on 4 January 2023).
- Vallée, B. Canadian Veterinary Medical Association Adopts a New Position Statement on Vaccination Protocols for Dogs and Cats. Can. Vet. J. 2008, 49, 362–365, quiz 365. [Google Scholar] [PubMed]
- Federation of Veterinarians of Europe (FVE). Joint American Veterinary Medical Association (AVMA)-Federation of Veterinarians of Europe (FVE)-Canadian Veterinary Medical Association (CVMA) Statement on the Benefits of Animal Vaccination Programs in Advancing Animal and Human Health. 16 November 2018. Available online: https://fve.org/cms/wp-content/uploads/AVMA-CVMA-FVE_vacconation_joint-paper.docx.pdf (accessed on 15 January 2023).
- Scherk, M.A.; Ford, R.B.; Gaskell, R.M.; Hartmann, K.; Hurley, K.F.; Lappin, M.R.; Levy, J.K.; Little, S.E.; Nordone, S.K.; Sparkes, A.H. 2013 AAFP Feline Vaccination Advisory Panel Report. J. Feline Med. Surg. 2013, 15, 785–808. [Google Scholar] [CrossRef]
- Stone, A.E.; Brummet, G.O.; Carozza, E.M.; Kass, P.H.; Petersen, E.P.; Sykes, J.; Westman, M.E. 2020 AAHA/AAFP Feline Vaccination Guidelines. J. Feline Med. Surg. 2020, 22, 813–830. [Google Scholar] [CrossRef]
- Advisory Board on Cat Diseases (ABCD). Feline Panleukopenia. Available online: http://www.abcdcatsvets.org/abcd-guidelines-on-feline-panleukopenia-2012-edition/ (accessed on 7 January 2023).
- Advisory Board on Cat Diseases (ABCD). Vaccination Recommended for Cats According to Lifestyle. Available online: http://www.abcdcatsvets.org/wp-content/uploads/2020/03/Tool_Vaccine-recommendations_Feb2020.pdf (accessed on 5 January 2023).
- Hosie, M.J.; Addie, D.D.; Boucraut-Baralon, C.; Egberink, H.; Frymus, T.; Gruffydd-Jones, T.; Hartmann, K.; Horzinek, M.C.; Lloret, A.; Lutz, H.; et al. Matrix Vaccination Guidelines. J. Feline Med. Surg. 2015, 17, 583–587. [Google Scholar] [CrossRef] [PubMed]
- Altman, K.D.; Kelman, M.; Ward, M.P. Are Vaccine Strain, Type or Administration Protocol Risk Factors for Canine Parvovirus Vaccine Failure? Vet. Microbiol. 2017, 210, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Zeng, W.; Zhang, X.; Li, S. The Genetic Evolution of Canine Parvovirus—A New Perspective. PLoS ONE 2017, 12, e0175035. [Google Scholar] [CrossRef]
- Decaro, N.; Buonavoglia, C.; Barrs, V.R. Canine Parvovirus Vaccination and Immunisation Failures: Are We Far from Disease Eradication? Vet. Microbiol. 2020, 247, 108760. [Google Scholar] [CrossRef]
- Hussain, K.; Khan, Y.; Ullah, Q.; Rabbani, A.H.; Naseer, O.; Raza, A.; Shahid, M.; Ali, S.; Ali, A. Canine Parvo Virus: A Review on Current Perspectives in Seroprevalence, Diagnostics and Therapeutics. Glob. Vet. 2021, 23, 113–126. [Google Scholar] [CrossRef]
- Decaro, N.; Buonavoglia, C. Canine Parvovirus—A Review of Epidemiological and Diagnostic Aspects, with Emphasis on Type 2c. Vet. Microbiol. 2012, 155, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Sykes, J.E. Greene’s Infectious Diseases of the Dog and Cat, 5th ed.; Sykes, J.E., Ed.; Elsevier: St Louis, MO, USA, 2022. [Google Scholar]
- Parrish, C.R.; Sykes, J.E. Canine Parvovirus Infections and Other Viral Enteritides. In Greene’s Infectious Diseases of the Dog and Cat; Sikes, J.E., Ed.; Elsevier: St Louis, MO, USA, 2022; pp. 341–351. [Google Scholar]
- Mira, F.; Purpari, G.; di Bella, S.; Vicari, D.; Schirò, G.; di Marco, P.; Macaluso, G.; Battilani, M.; Guercio, A. Update on Canine Distemper Virus (CDV) Strains of Arctic-like Lineage Detected in Dogs in Italy. Vet. Ital. 2018, 54, 225–236. [Google Scholar] [PubMed]
- Duque-Valencia, J.; Sarute, N.; Olarte-Castillo, X.A.; Ruíz-Sáenz, J. Evolution and Interspecies Transmission of Canine Distemper Virus—An Outlook of the Diverse Evolutionary Landscapes of a Multi-Host Virus. Viruses 2019, 11, 582. [Google Scholar] [CrossRef] [PubMed]
- Sykes, J.E.; Vandevelde, M. Canine Distemper Virus Infection. In Greene’s Infectious Diseases of the Dog and Cat; Sykes, J.E., Ed.; Elsevier: St Louis, MO, USA, 2022; pp. 271–388. [Google Scholar]
- Decaro, N.; Campolo, M.; Elia, G.; Buonavoglia, D.; Colaianni, M.L.; Lorusso, A.; Mari, V.; Buonavoglia, C. Infectious Canine Hepatitis: An “Old” Disease Reemerging in Italy. Res. Vet. Sci. 2007, 83, 269–273. [Google Scholar] [CrossRef] [PubMed]
- Decaro, N.; Martella, V.; Buonavoglia, C. Canine Adenoviruses and Herpesvirus. Vet. Clin. N. Am. Small Anim. Pract. 2008, 38, 799–814. [Google Scholar] [CrossRef]
- Mira, F.; Puleio, R.; Schirò, G.; Condorelli, L.; di Bella, S.; Chiaramonte, G.; Purpari, G.; Cannella, V.; Balboni, A.; Randazzo, V.; et al. Study on the Canine Adenovirus Type 1 (CAdV-1) Infection in Domestic Dogs in Southern Italy. Pathogens 2022, 11, 1254. [Google Scholar] [CrossRef]
- Decaro, N. Infectious Canine Hepatitis and Feline Adenovirus Infection. In Greene’s Infectious Diseases of the Dog and Cat; Sykes, J.E., Ed.; Elsevier: St Louis, MO, USA, 2022; pp. 289–300. [Google Scholar]
- Dall’Ara, P. Vaccini e Vaccinazioni Core Del Cane. In Vaccini e Vaccinazioni Degli Animali da Compagnia; Dall’Ara, P., Ed.; EDRA: Milano, Italy, 2020; pp. 153–174. [Google Scholar]
- Möstl, K. Duration of Vaccine-Induced Immunity. EJCAP 2016, 26, 4–8. [Google Scholar]
- Mitchell, S.; Zwijnenberg, R.; Huang, J.; Hodge, A.; Day, M. Duration of Serological Response to Canine Parvovirus-Type 2, Canine Distemper Virus, Canine Adenovirus Type 1 and Canine Parainfluenza Virus in Client-Owned Dogs in Australia. Aust. Vet. J. 2012, 90, 468–473. [Google Scholar] [CrossRef]
- Roth, J.A.; Spickler, A.R. Duration of Immunity Induced by Companion Animal Vaccines. Anim. Health Res. Rev. 2010, 11, 165–190. [Google Scholar] [CrossRef]
- Mouzin, D.E.; Lorenzen, M.J.; Haworth, J.D.; King, V.L. Duration of Serologic Response to Five Viral Antigens in Dogs. J. Am. Vet. Med. Assoc. 2004, 224, 55–60. [Google Scholar] [CrossRef]
- Bohm, M.; Thomson, H.; Weir, A.; Hasted, A.M.; Maxwell, N.S.; Herrtage, M.E. Serum Antibody Titres to Canine Parvovirus, Adenovirus and Distemper Virus in Dogs in the UK Which Had Not Been Vaccinated for at Least Three Years. Vet. Rec. 2004, 154, 457–463. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.; Siedek, E.; Thomas, A.; King, V.; Stirling, C.; Plevová, E.; Salt, J.; Sture, G. Influence of Maternally-Derived Antibodies in 6-Week Old Dogs for the Efficacy of a New Vaccine to Protect Dogs against Virulent Challenge with Canine Distemper Virus, Adenovirus or Parvovirus. Trials Vaccinol. 2014, 3, 107–113. [Google Scholar] [CrossRef]
- Chastant, S.; Mila, H. Passive Immune Transfer in Puppies. Anim. Reprod. Sci. 2019, 207, 162–170. [Google Scholar] [CrossRef]
- Dall’Ara, P.; Lauzi, S.; Filipe, J.; Caseri, R.; Beccaglia, M.; Desario, C.; Cavalli, A.; Aiudi, G.G.; Buonavoglia, C.; Decaro, N. Discrepancy Between In-Clinic and Haemagglutination-Inhibition Tests in Detecting Maternally-Derived Antibodies against Canine Parvovirus in Puppies. Front. Vet. Sci. 2021, 8, 630809. [Google Scholar] [CrossRef] [PubMed]
- Dall’Ara, P. Immune System and Ageing in the Dog: Possible Consequences and Control Strategies. Vet. Res. Commun. 2003, 27, 535–542. [Google Scholar] [CrossRef] [PubMed]
- HogenEsch, H.; Thompson, S. Effect of Ageing on the Immune Response of Dogs to Vaccines. J. Comp. Pathol. 2010, 142, S74–S77. [Google Scholar] [CrossRef]
- Pereira, M.; Valério-Bolas, A.; Saraiva-Marques, C.; Alexandre-Pires, G.; Pereira da Fonseca, I.; Santos-Gomes, G. Development of Dog Immune System: From in Uterus to Elderly. Vet. Sci. 2019, 6, 83. [Google Scholar] [CrossRef]
- Dall’Ara, P. Cosa Influenza l’efficacia Di Un Vaccino. In Vaccini e Vaccinazioni Degli Animali da Compagnia; Dall’Ara, P., Ed.; EDRA: Milano, Italy, 2020; pp. 273–293. [Google Scholar]
- Rashid, A.; Rasheed, K.; Akhtar, M. Factors Influencing Vaccine Efficacy—A General Review. J. Anim. Plant Sci. 2009, 19, 22–25. [Google Scholar]
- Dall’Ara, P. Vaccini e Vaccinazioni Degli Animali Da Compagnia, 1st ed.; Dall’Ara, P., Ed.; EDRA: Milano, Italy, 2020. [Google Scholar]
- Eldredge, D.M.; Carlson, L.D.; Carlson, D.G.; Giffin, J.M.; Adelman, B. Comparative Age of Dogs and Humans. In Dog Owner’s Home Veterinary Handbook; Howell Book House: Howell, NJ, USA, 2007; pp. 575–576. [Google Scholar]
- Kraus, C.; Pavard, S.; Promislow, D.E.L. The Size–Life Span Trade-Off Decomposed: Why Large Dogs Die Young. Am. Nat. 2013, 181, 492–505. [Google Scholar] [CrossRef]
- Day, M.J. Small Animal Vaccination: A Practical Guide for Vets in the UK. Practice 2017, 39, 110–118. [Google Scholar] [CrossRef]
- Waner, T.; Mazar, S.; Keren-Kornblatt, E. Application of a Dot Enzyme-Linked Immunosorbent Assay for Evaluation of the Immune Status to Canine Parvovirus and Distemper Virus in Adult Dogs before Revaccination. J. Vet. Diagn. 2006, 18, 267–270. [Google Scholar] [CrossRef] [PubMed]
- Salomon, K.; de Lange, T.; Calis, A.; Radier, O.; Krosse, J. In-Clinic Canine IgG Antibody Titer Test Comparative Study: Results from Five Clinics. Isr. J. Vet. Med. 2022, 77, 7–14. [Google Scholar]
- Meazzi, S.; Filipe, J.; Fiore, A.; di Bella, S.; Mira, F.; Dall’Ara, P. Agreement between In-Clinics and Virus Neutralization Tests in Detecting Antibodies against Canine Distemper Virus (CDV). Viruses 2022, 14, 517. [Google Scholar] [CrossRef]
- Mende, K.; Stuetzer, B.; Sauter-Louis, C.; Homeier, T.; Truyen, U.; Hartmann, K. Prevalence of Antibodies against Feline Panleukopenia Virus in Client-Owned Cats in Southern Germany. Vet. J. 2014, 199, 419–423. [Google Scholar] [CrossRef]
- Decaro, N.; Buonavoglia, D.; Desario, C.; Amorisco, F.; Colaianni, M.L.; Parisi, A.; Terio, V.; Elia, G.; Lucente, M.S.; Cavalli, A.; et al. Characterisation of Canine Parvovirus Strains Isolated from Cats with Feline Panleukopenia. Res. Vet. Sci. 2010, 89, 275–278. [Google Scholar] [CrossRef] [PubMed]
- Clegg, S.R.; Coyne, K.P.; Dawson, S.; Spibey, N.; Gaskell, R.M.; Radford, A.D. Canine Parvovirus in Asymptomatic Feline Carriers. Vet. Microbiol. 2012, 157, 78–85. [Google Scholar] [CrossRef]
- Clegg, S.R.; Coyne, K.P.; Parker, J.; Dawson, S.; Godsall, S.A.; Pinchbeck, G.; Cripps, P.J.; Gaskell, R.M.; Radford, A.D. Molecular Epidemiology and Phylogeny Reveal Complex Spatial Dynamics in Areas Where Canine Parvovirus Is Endemic. J. Virol. 2011, 85, 7892–7899. [Google Scholar] [CrossRef]
- Carrai, M.; Decaro, N.; van Brussel, K.; Dall’Ara, P.; Desario, C.; Fracasso, M.; Šlapeta, J.; Colombo, E.; Bo, S.; Beatty, J.A.; et al. Canine Parvovirus Is Shed Infrequently by Cats without Diarrhoea in Multi-Cat Environments. Vet. Microbiol. 2021, 261, 109204. [Google Scholar] [CrossRef]
- Dall’Ara, P.; Labriola, C.; Sala, E.; Spada, E.; Magistrelli, S.; Lauzi, S. Prevalence of Serum Antibody Titres against Feline Panleukopenia, Herpesvirus and Calicivirus Infections in Stray Cats of Milan, Italy. Prev. Vet. Med. 2019, 167, 32–38. [Google Scholar] [CrossRef]
- Gonuguntla, H.N. Detection and Typing of CPV with Real-Time PCR and Mini-Sequencing. Adv. Anim. Vet. Sci. 2016, 4, 187–194. [Google Scholar] [CrossRef]
- Decaro, N.; Elia, G.; Desario, C.; Roperto, S.; Martella, V.; Campolo, M.; Lorusso, A.; Cavalli, A.; Buonavoglia, C. A Minor Groove Binder Probe Real-Time PCR Assay for Discrimination between Type 2-Based Vaccines and Field Strains of Canine Parvovirus. J. Virol. Methods 2006, 136, 65–70. [Google Scholar] [CrossRef]
- Decaro, N.; Elia, G.; Campolo, M.; Desario, C.; Lucente, M.S.; Bellacicco, A.L.; Buonavoglia, C. New Approaches for the Molecular Characterization of Canine Parvovirus Type 2 Strains. J. Vet. Med. Ser. B 2005, 52, 316–319. [Google Scholar] [CrossRef] [PubMed]
- Decaro, N.; Crescenzo, G.; Desario, C.; Cavalli, A.; Losurdo, M.; Colaianni, M.L.; Ventrella, G.; Rizzi, S.; Aulicino, S.; Lucente, M.S.; et al. Long-Term Viremia and Fecal Shedding in Pups after Modified-Live Canine Parvovirus Vaccination. Vaccine 2014, 32, 3850–3853. [Google Scholar] [CrossRef] [PubMed]
- Cavalli, A.; Martella, V.; Desario, C.; Camero, M.; Lanave, G.; Barrs, V.R.; Decaro, N.; Buonavoglia, C. Modified Haemagglutination Inhibition Assay for the Detection of Canine Parvovirus Type 2 Antibodies in Dog Sera. Vet. J. 2021, 274, 105709. [Google Scholar] [CrossRef] [PubMed]
- Balboni, A.; Musto, C.; Kaehler, E.; Verin, R.; Caniglia, R.; Fabbri, E.; Carra, E.; Cotti, C.; Battilani, M.; Delogu, M. Genetic Characterization of Canine Adenovirus Type 1 Detected by Real-Time Polymerase Chain Reaction in an Oral Sample of an Italian Wolf (Canis lupus). J. Wildl. Dis. 2019, 55, 737. [Google Scholar] [CrossRef]
- Balboni, A.; Tryland, M.; Mørk, T.; Killengreen, S.T.; Fuglei, E.; Battilani, M. Unique Genetic Features of Canine Adenovirus Type 1 (CAdV-1) Infecting Red Foxes (Vulpes vulpes) in Northern Norway and Arctic Foxes (Vulpes lagopus) in Svalbard. Vet. Res. Commun. 2019, 43, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Balboni, A.; Terrusi, A.; Urbani, L.; Troia, R.; Stefanelli, S.A.M.; Giunti, M.; Battilani, M. Canine Circovirus and Canine Adenovirus Type 1 and 2 in Dogs with Parvoviral Enteritis. Vet. Res. Commun. 2022, 46, 223–232. [Google Scholar] [CrossRef]
- Dall’Ara, P.; Turin, L. Immunology of the Canine Eye in Health and Disease: A Concise Review. Vet. Med. 2019, 64, 1–17. [Google Scholar] [CrossRef]
- Lechner, E.S.; Crawford, P.C.; Levy, J.K.; Edinboro, C.H.; Dubovi, E.J.; Caligiuri, R. Prevalence of Protective Antibody Titers for Canine Distemper Virus and Canine Parvovirus in Dogs Entering a Florida Animal Shelter. J. Am. Vet. Med. Assoc. 2010, 236, 1317–1321. [Google Scholar] [CrossRef]
- Schultz, R. A Field and Experimental Trial to Assess the Performance of the ImmunoComb Canine VacciCheck Antibody Test Kit; Biogal Galed Labs: Kibbutz Galed, Israel, 2015. [Google Scholar]
- Dodds, J. Vaccination and Antibody Tests: 2018 Update. Available online: https://www.vaccicheck.nl/sites/default/files/documents/serology_testing_clinical_review_dr_dodds_march_2018.pdf (accessed on 8 January 2023).
- den Besten, R. An Analysis of Titer Testing as Part of the Vaccination Guideline for Dogs. Master’s Thesis, Utrecht University, Utrecht, The Netherlands, 2018. [Google Scholar]
- Riedl, M.; Truyen, U.; Reese, S.; Hartmann, K. Prevalence of Antibodies to Canine Parvovirus and Reaction to Vaccination in Client-Owned, Healthy Dogs. Vet. Rec. 2015, 177, 597. [Google Scholar] [CrossRef]
- Killey, R.; Mynors, C.; Pearce, R.; Nell, A.; Prentis, A.; Day, M.J. Long-Lived Immunity to Canine Core Vaccine Antigens in UK Dogs as Assessed by an in-Practice Test Kit. J. Small Anim. Pract. 2018, 59, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Tennant, B.J.; Gaskell, R.M.; Jones, R.C.; Gaskell, C.J. Prevalence of Antibodies to Four Major Canine Viral Diseases in Dogs in a Liverpool Hospital Population. J. Small Anim. Pract. 1991, 32, 175–179. [Google Scholar] [CrossRef]
- McCaw, D.L.; Thomson, M.; Tate, D.; Bonderer, A.; Chen, Y.J. Serum Distemper Virus and Parvovirus Antibody Titers among Dogs Brought to a Veterinary Hospital for Revaccination. J. Am. Vet. Med. Assoc. 1998, 213, 72–75. [Google Scholar] [PubMed]
- Twark, L.; Dodds, W.J. Clinical Use of Serum Parvovirus and Distemper Virus Antibody Titers for Determining Revaccination Strategies in Healthy Dogs. J. Am. Vet. Med. Assoc. 2000, 217, 1021–1024. [Google Scholar] [CrossRef]
- Ottiger, H.-P.; Neimeier-Förster, M.; Bruckner, L.; Stärk, K.D.C.; Duchow, K. Serological Responses of Adult Dogs to Revaccination against Distemper, Parvovirus and Rabies. Vet. Rec. 2006, 159, 7–12. [Google Scholar] [CrossRef]
- Adam, J.N.; Asgarali, Z.; Singh, S.M.; Ezeokoli, C.D. A Serological Study of Canine Parvovirus (CPV-2) and Distemper Virus (CDV) in Stray Dogs in North Trinidad, West Indies. West Indian Vet. J. 2011, 11, 1–4. [Google Scholar]
- Taguchi, M.; Namikawa, K.; Maruo, T.; Orito, K.; Lynch, J.; Sahara, H. Antibody Titers for Canine Parvovirus Type-2, Canine Distemper Virus, and Canine Adenovirus Type-1 in Adult Household Dogs. Can. Vet. J. 2011, 52, 983–986. [Google Scholar]
- Litster, A.; Nichols, J.; Volpe, A. Prevalence of Positive Antibody Test Results for Canine Parvovirus (CPV) and Canine Distemper Virus (CDV) and Response to Modified Live Vaccination against CPV and CDV in Dogs Entering Animal Shelters. Vet. Microbiol. 2012, 157, 86–90. [Google Scholar] [CrossRef]
- Belsare, A.V.; Gompper, M.E. Assessing Demographic and Epidemiologic Parameters of Rural Dog Populations in India during Mass Vaccination Campaigns. Prev. Vet. Med. 2013, 111, 139–146. [Google Scholar] [CrossRef]
- Lund, J.D.; Prior, M.; Madsen, L. Testing Dogs for Immunity against Canine Parvovirus, Canine Distemper Virus and Infectious Canine Hepatitis; VetSurgeon.org. 2013. Available online: https://www.vetsurgeon.org/w/veterinary-research/1100/testing-dogs-for-immunity-against-canine-parvovirus-canine-distemper-virus-and-infectious-canine-hepatitis (accessed on 15 January 2023).
- Belsare, A.V.; Vanak, A.T.; Gompper, M.E. Epidemiology of Viral Pathogens of Free-Ranging Dogs and Indian Foxes in a Human-Dominated Landscape in Central India. Transbound. Emerg. Dis. 2014, 61, 78–86. [Google Scholar] [CrossRef]
- Curi, N.H.D.A.; Massara, R.L.; Paschoal, A.M.D.O.; Soriano-Araújo, A.; Lobato, Z.I.P.; Demetrio, G.R.; Chiarello, A.G.; Passamani, M. Prevalence and Risk Factors for Viral Exposure in Rural Dogs around Protected Areas of the Atlantic Forest. BMC Vet. Res. 2016, 12, 21. [Google Scholar] [CrossRef] [PubMed]
- Mahon, J.L.; Rozanski, E.A.; Paul, A.L. Prevalence of Serum Antibody Titers against Canine Distemper Virus and Canine Parvovirus in Dogs Hospitalized in an Intensive Care Unit. J. Am. Vet. Med. Assoc. 2017, 250, 1413–1418. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-H.; Yang, D.-K.; Seo, B.-H.; Cho, I.-S. Serosurvey of Rabies Virus, Canine Distemper Virus, Parvovirus, and Influenza Virus in Military Working Dogs in Korea. J. Vet. Med. Sci. 2018, 80, 1424–1430. [Google Scholar] [CrossRef]
- DiGangi, B.A.; Dingman, P.A.; Grijalva, C.J.; Belyeu, M.; Tucker, S.; Isaza, R. Prevalence and Risk Factors for the Presence of Serum Antibodies against Canine Distemper, Canine Parvovirus, and Canine Adenovirus in Communities in Mainland Ecuador. Vet. Immunol. Immunopathol. 2019, 218, 109933. [Google Scholar] [CrossRef]
- Home, C.; Bijoor, A.; Bhatnagar, Y.V.; Vanak, A.T. Serosurvey of Viral Pathogens in Free-Ranging Dog Populations in the High Altitude Trans-Himalayan Region. J. Threat. Taxa 2022, 14, 21025–21031. [Google Scholar] [CrossRef]
- Sadaula, A.; Joshi, J.D.; Lamichhane, B.R.; Gairhe, K.P.; Subedi, N.; Pokheral, C.P.; Thapaliya, S.; Pandey, G.; Rijal, K.R.; Pandey, P. Seroprevalence of Canine Distemper and Canine Parvovirus Among Domestic Dogs in Buffer Zone of Chitwan National Park, Nepal. SSRN Electron. J. 2022. Available online: https://papers.ssrn.com/sol3/papers.cfm?abstract_id= (accessed on 15 January 2023). [CrossRef]
- Netea, M.G.; Domínguez-Andrés, J.; Barreiro, L.B.; Chavakis, T.; Divangahi, M.; Fuchs, E.; Joosten, L.A.B.; van der Meer, J.W.M.; Mhlanga, M.M.; Mulder, W.J.M.; et al. Defining Trained Immunity and Its Role in Health and Disease. Nat. Rev. Immunol. 2020, 20, 375–388. [Google Scholar] [CrossRef] [PubMed]
- Uthayakumar, D.; Paris, S.; Chapat, L.; Freyburger, L.; Poulet, H.; de Luca, K. Non-Specific Effects of Vaccines Illustrated Through the BCG Example: From Observations to Demonstrations. Front. Immunol. 2018, 9, 2869. [Google Scholar] [CrossRef]
- Benn, C.S.; Fisker, A.B.; Rieckmann, A.; Sørup, S.; Aaby, P. Vaccinology: Time to Change the Paradigm? Lancet Infect. Dis. 2020, 20, e274–e283. [Google Scholar] [CrossRef]
- Chastant-Maillard, S.; Mila, H. Canine Colostrum. Vet. Focus 2016, 26, 32–38. [Google Scholar]
- Chastant-Maillard, S.; Freyburger, L.; Marcheteau, E.; Thoumire, S.; Ravier, J.; Reynaud, K. Timing of the Intestinal Barrier Closure in Puppies. Reprod. Domest. Anim. 2012, 47, 190–193. [Google Scholar] [CrossRef]
- Chastant-Maillard, S.; Aggouni, C.; Albaret, A.; Fournier, A.; Mila, H. Canine and Feline Colostrum. Reprod. Domest. Anim. 2017, 52, 148–152. [Google Scholar] [CrossRef]
- Mila, H.; Feugier, A.; Grellet, A.; Anne, J.; Gonnier, M.; Martin, M.; Rossig, L.; Chastant-Maillard, S. Inadequate Passive Immune Transfer in Puppies: Definition, Risk Factors and Prevention in a Large Multi-Breed Kennel. Prev. Vet. Med. 2014, 116, 209–213. [Google Scholar] [CrossRef]
- Mila, H.; Feugier, A.; Grellet, A.; Anne, J.; Gonnier, M.; Martin, M.; Rossig, L.; Chastant-Maillard, S.; Immunoglobulin, G. Concentration in Canine Colostrum: Evaluation and Variability. J. Reprod. Immunol. 2015, 112, 24–28. [Google Scholar] [CrossRef]
- de Cramer, K.G.M.; Stylianides, E.; van Vuuren, M. Efficacy of Vaccination at 4 and 6 Weeks in the Control of Canine Parvovirus. Vet. Microbiol. 2011, 149, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Ellis, J.; Gow, S.; Rhodes, C.; Lacoste, S.; Kong, L.; Musil, K.; Snead, E. Serum Antibody Responses to Vaccinal Antigens in Lean and Obese Geriatric Dogs. Can. Vet. J. 2016, 57, 531–534. [Google Scholar]
- Ginaldi, L.; Loreto, M.F.; Corsi, M.P.; Modesti, M.; de Martinis, M. Immunosenescence and Infectious Diseases. Microbes Infect. 2001, 3, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Mansfield, K.L.; Sayers, R.; Fooks, A.R.; Burr, P.D.; Snodgrass, D. Factors Affecting the Serological Response of Dogs and Cats to Rabies Vaccination. Vet. Rec. 2004, 154, 423–426. [Google Scholar] [CrossRef]
- Kennedy, L.J.; Lunt, M.; Barnes, A.; McElhinney, L.; Fooks, A.R.; Baxter, D.N.; Ollier, W.E.R. Factors Influencing the Antibody Response of Dogs Vaccinated against Rabies. Vaccine 2007, 25, 8500–8507. [Google Scholar] [CrossRef]
- Paolini, A.; Romagnoli, S.; Nardoia, M.; Conte, A.; Salini, R.; Podaliri Vulpiani, M.; Dalla Villa, P. Study on the Public Perception of “Community-Owned Dogs” in the Abruzzo Region, Central Italy. Animals 2020, 10, 1227. [Google Scholar] [CrossRef] [PubMed]
- Carvelli, A.; Scaramozzino, P.; Iacoponi, F.; Condoleo, R.; della Marta, U. Size, Demography, Ownership Profiles, and Identification Rate of the Owned Dog Population in Central Italy. PLoS ONE 2020, 15, e0240551. [Google Scholar] [CrossRef] [PubMed]
- Biogal Galed Labs. Acs. Ltd. VacciCheck® Titer Testing. Available online: https://www.biogal.com/products/vaccicheck/ (accessed on 4 February 2023).
- Egerer, A.; Schaefer, Z.; Larson, L. A Point-of-Care Dot Blot ELISA Assay for Detection of Protective Antibody against Canine Adenovirus, Canine Parvovirus, and Canine Distemper Virus Is Diagnostically Accurate. J. Am. Vet. Med. Assoc. 2022, 260, 1928–1933. [Google Scholar] [CrossRef] [PubMed]


| Protective Antibody Titers (PATs)-% (n. of Dogs) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| WHOLE POPULATION (1027 Dogs) | VACCINATED DOGS (951 Dogs) | UNVACCINATED DOGS (76 Dogs) | |||||||
| CPV-2 | CDV | CAdV-1 | CPV-2 | CDV | CAdV-1 | CPV-2 | CDV | CAdV-1 | |
| OVERALL VALUE | 90.8 (933/1027) | 68.6 (705/1027) | 79.8 (820/1027) | 93.0 (884/951) | 70.0 (666/951) | 81.8 (778/951) | 64.5 (49/76) | 51.3 (39/76) | 55.3 (42/76) |
| Sex and reproductive status | |||||||||
| Intact females | 91.3 (358/392) | 65.8 (258/392) | 77.3 (303/392) | 94.0 (344/366) | 67.8 (248/366) | 79.0 (289/366) | 53.8 (14/26) | 38.5 (10/26) | 53.8 (14/26) |
| Neutered females | 92.3 (155/168) | 74.4 (125/168) | 89.3 (150/168) | 93.2 (150/161) | 74.5 (120/161) | 90.7 (146/161) | 71.4 (5/7) | 71.4 (5/7) | 57.1 (4/7) |
| Intact males | 88.9 (351/395) | 68.6 (271/395) | 77.5 (306/395) | 91.3 (324/355) | 69.9 (248/355) | 79.7 (283/355) | 67.5 (27/40) | 57.5 (23/40) | 57.5 (23/40) |
| Neutered males | 95.8 (69/72) | 70.8 (51/72) | 84.7 (61/72) | 95.7 (66/69) | 72.5 (50/69) | 87.0 (60/69) | 100.0 (3/3) | 33.3 (1/3) | 33.3 (1/3) |
| Age | |||||||||
| Puppies (4 months–<1 year) | 74.4 (96/129) | 53.5 (69/129) | 31.8 (41/129) | 79.2 (84/106) | 54.7 (58/106) | 26.4 (28/106) | 52.2 (12/23) | 47.8 (11/23) | 56.5 (13/23) |
| Adults | 96.2 (511/531) | 74.0 (393/531) | 90.4 (480/531) | 98.2 (488/497) | 75.1 (373/497) | 92.8 (461/497) | 67.6 (23/34) | 58.8 (20/34) | 55.9 (19/34) |
| Seniors | 90.8 (246/271) | 69.7 (189/271) | 82.3 (223/271) | 90.9 (239/263) | 70.7 (186/263) | 83.7 (220/263) | 87.5 (7/8) | 37.5 (3/8) | 37.5 (3/8) |
| Geriatrics | 83.3 (80/96) | 56.3 (54/96) | 79.2 (76/96) | 85.9 (73/85) | 57.6 (49/85) | 81.2 (69/85) | 63.6 (7/11) | 45.5 (5/11) | 63.6 (7/11) |
| Size | |||||||||
| Small (<10 kg) | 91.0 (272/299) | 71.9 (215/299) | 81.9 (245/299) | 92.4 (267/289) | 73.7 (213/289) | 84.1 (243/289) | 50.0 (5/10) | 20.0 (2/10) | 20.0 (2/10) |
| Medium (≥10–<25 kg) | 88.6 (319/360) | 65.3 (235/360) | 83.6 (301/360) | 92.1 (292/317) | 66.6 (211/317) | 86.4 (274/317) | 62.8 (27/43) | 55.8 (24/43) | 62.8 (27/43) |
| Large (≥25 kg) | 92.9 (342/368) | 69.3 (255/368) | 74.5 (274/368) | 94.2 (325/345) | 70.1 (242/345) | 75.7 (261/345) | 73.9 (17/23) | 56.5 (13/23) | 56.5 (13/23) |
| Health status | |||||||||
| Healthy | 91.4 (805/881) | 68.1 (600/881) | 79.5 (700/881) | 93.8 (758/808) | 69.4 (561/808) | 81.6 (659/808) | 64.4 (47/73) | 53.4 (39/73) | 56.2 (41/73) |
| Unhealthy | 87.7 (128/146) | 71.9 (105/146) | 82.2 (120/146) | 88.1 (126/143) | 73.4 (105/143) | 83.2 (119/143) | 66.7 (2/3) | 0.0 (0/0) | 33.3 (1/3) |
| Time after vaccination * | |||||||||
| ≤1 year | // | // | // | 93.3 (456/489) | 71.4 (349/489) | 77.9 (381/489) | // | // | // |
| >1–≤3 years | // | // | // | 95.4 (309/324) | 74.4 (241/324) | 91.4 (296/324) | // | // | // |
| >3 years | // | // | // | 87.0 (100/115) | 53.9 (62/115) | 68.7 (79/115) | // | // | // |
| CPV-2 | CDV | CAdV-1 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| PROTECTED | p-Value * | PROTECTED | p-Value * | PROTECTED | p-Value * | |||||
| Statistical Variable (Number) | YES | NO | YES | NO | YES | NO | ||||
| Sex | Intact females (366) | 94.0 (344) | 6.0 (22) | 0.4043 | 67.8 (248) | 32.2 (118) | 0.4479 | 79.0 (289) | 21.0 (77) | 0.0051 |
| Neutered females (161) | 93.2 (150) | 6.8 (11) | 74.5 (120) | 25.5 (41) | 90.7 (146) | 9.3 (15) | ||||
| Intact males (355) | 91.3 (324) | 8.7 (31) | 69.9 (248) | 30.1 (107) | 79.7 (283) | 20.3 (72) | ||||
| Neutered males (69) | 95.7 (66) | 4.3 (3) | 72.5 (50) | 27.5 (19) | 87.0 (60) | 13.0 (9) | ||||
| Age | Puppies (106) | 79.2 (84) | 20.8 (22) | <0.0001 | 54.7 (58) | 45.3 (48) | <0.0001 | 26.4 (28) | 73.6 (78) | <0.0001 |
| Adults (497) | 98.2 (488) | 1.8 (9) | 75.1 (373) | 24.9 (124) | 92.8 (461) | 7.2 (36) | ||||
| Seniors (263) | 90.9 (239) | 9.1 (24) | 70.7 (186) | 29.3 (77) | 83.7 (220) | 16.3 (43) | ||||
| Geriatrics (85) | 85.9 (73) | 14.1 (12) | 57.6 (49) | 42.4 (36) | 81.2 (69) | 18.8 (16) | ||||
| Size | Small (289) | 92.4 (267) | 7.6 (22) | 0.5207 | 73.7 (213) | 26.3 (76) | 0.1591 | 83.4 (243) | 15.6 (46) | 0.0008 |
| Medium (317) | 92.1 (292) | 7.9 (25) | 66.6 (211) | 33.4 (106) | 86.4 (274) | 13.6 (43) | ||||
| Large (345) | 94.2 (325) | 5.8 (20) | 70.1 (242) | 29.9 (103) | 75.7 (261) | 24.3 (84) | ||||
| Health status | Healthy (808) | 93.8 (758) | 6.2 (50) | 0.0203 | 69.4 (561) | 30.6 (247) | 0.3733 | 81.6 (659) | 18.4 (149) | 0.7245 |
| Unhealthy (143) | 88.1 (126) | 11.9 (17) | 73.4 (105) | 26.6 (38) | 83.2 (119) | 16.8 (24) | ||||
| Time after vaccination | ≤1 year (489) | 93.3 (456) | 6.7 (33) | 0.0087 | 71.4 (349) | 28.6 (140) | 0.0001 | 77.9 (381) | 22.1 (108) | <0.0001 |
| >1-≤3 years (324) | 95.4 (309) | 4.6 (15) | 74.4 (241) | 25.6 (83) | 91.4 (296) | 8.6 (28) | ||||
| >3 years (115) | 87.0 (100) | 13.0 (15) | 53.9 (62) | 46.1 (53) | 68.9 (79) | 31.3 (36) | ||||
| NEGATIVE DOGS | WHOLE POPULATION (1027 Dogs) % (Number & Health) | VACCINATED DOGS (951 Dogs) % (Number & Health) | UNVACCINATED DOGS (76 Dogs) % (Number & Health) |
|---|---|---|---|
| Only for CPV-2 | 1.7 (17–15H + 2U) | 1.6 (15–13H + 2U) | 2.6 (2–2H) |
| Only for CDV | 1.8 (18–17H + 1U) | 1.6 (15–14H + 1U) | 3.9 (3–3H) |
| Only for CAdV-1 | 2.7 (28–25H + 3U) | 2.6 (25–22H + 3U) | 3.9 (3–3H) |
| For CPV-2 and CDV | 0.5 (5–3H + 2U) | 0.2 (2–2U) | 3.9 (3–3H) |
| For CPV-2 and CAdV-1 | 1.0 (10–10H) | 0.4 (4–4H) | 7.9 (6–6H) |
| For CDV and CAdV-1 | 0.4 (4–3H + 1U) | 0.3 (3–2H + 1U) | 1.3 (1–1H) |
| For CPV-2, CDV and CAdV-1 | 0.8 (8–8H) | 0.2 (2–2H) | 7.9 (6–6H) |
| Total | 8.8 (90–81H + 9U) | 6.9 (66–57H + 9U) | 31.6 (24–24H) |
| % of Protection | ||||||
|---|---|---|---|---|---|---|
| Authors (Year) | [Reference] | Country | No. Dogs | CPV-2 | CDV | CAdV-1 |
| Tennant et al. (1991) | [68] | UK | 190 | 70.0 | 84.0 | // |
| McCaw et al. (1998) | [69] | USA | 122 | 73.0 | 79.0 | // |
| Twark et al. (2000) | [70] | USA and Canada | 1441 | 95.1 | 97.6 | // |
| Bohm et al. (2004) | [31] | UK | 144 | 95.0 | 71.5 | 82.0 |
| Mouzin et al. (2004) | [30] | USA and Canada | 322 | 98.1 | 98.1 | 98.4 |
| Ottiger et al. (2006) | [71] | Switzerland and Germany | 260 | 64.0 | 83.0 | // |
| Lechner et al. (2010) | [62] | USA | 431 * | 67.0 | 43.2 | // |
| Adam et al. (2011) | [72] | Trinidad and Tobago | 92 * | 78.3 | 39.1 | // |
| Taguchi et al. (2011) | [73] | Japan | 1031 | 86.0 | 72.0 | 71.0 |
| Mitchell et al. (2012) | [28] | Australia | 235 | 97.4 | 84.1 | 95.7 |
| Litster et al. (2012) | [74] | USA | 102 | 97.9 | 93.8 | // |
| Belsare et al. (2013) | [75] | India | 77 | 88.0 | 73.0 | 68.0 |
| Lund et al. (2013) | [76] | Denmark | 322 | 88.5 | 87.3 | 85.1 |
| Belsare et al. (2014) | [77] | India | 219 * | 88.0 | 72.0 | 77.0 |
| Riedl et al. (2015) | [66] | Germany | 100 | 86.0 | // | // |
| Curi et al. (2016) | [78] | Brazil | 320 * | 97.0 | 15.0 | 27.8 |
| Mahon et al. (2017) | [79] | USA | 80 | 81.0 | 50.0 | // |
| Killey et al. (2018) | [67] | UK | 486 | 98.5 | 95.7 | 97.3 |
| den Besten (2018) | [65] | Netherland | 929 | 97.5 | 90.7 | 91.6 |
| Kim et al. (2018) | [80] | Korea | 78 | 100 | 94.8 | // |
| DiGangi et al. (2019) | [81] | Ecuador | 154 | 95.0 | 66.0 | 60.0 |
| Home et al. (2022) | [82] | India | 97 * | 100.0 | 54.0 | 66.0 |
| Sadauda et al. (2022) | [83] | Nepal | 163 * | 17.0 | 33.0 | // |
| This study (2023) | // | Italy | 1027 | 90.8 | 68.6 | 79.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dall’Ara, P.; Lauzi, S.; Zambarbieri, J.; Servida, F.; Barbieri, L.; Rosenthal, R.; Turin, L.; Scarparo, E.; Filipe, J. Prevalence of Serum Antibody Titers against Core Vaccine Antigens in Italian Dogs. Life 2023, 13, 587. https://doi.org/10.3390/life13020587
Dall’Ara P, Lauzi S, Zambarbieri J, Servida F, Barbieri L, Rosenthal R, Turin L, Scarparo E, Filipe J. Prevalence of Serum Antibody Titers against Core Vaccine Antigens in Italian Dogs. Life. 2023; 13(2):587. https://doi.org/10.3390/life13020587
Chicago/Turabian StyleDall’Ara, Paola, Stefania Lauzi, Jari Zambarbieri, Francesco Servida, Laura Barbieri, Roberto Rosenthal, Lauretta Turin, Elena Scarparo, and Joel Filipe. 2023. "Prevalence of Serum Antibody Titers against Core Vaccine Antigens in Italian Dogs" Life 13, no. 2: 587. https://doi.org/10.3390/life13020587
APA StyleDall’Ara, P., Lauzi, S., Zambarbieri, J., Servida, F., Barbieri, L., Rosenthal, R., Turin, L., Scarparo, E., & Filipe, J. (2023). Prevalence of Serum Antibody Titers against Core Vaccine Antigens in Italian Dogs. Life, 13(2), 587. https://doi.org/10.3390/life13020587








