Abstract
Colorectal cancer (CRC) is one of the most common causes of death and the third most diagnosed cancer worldwide. The tumor microenvironment and cancer stem cells participate in colorectal tumor progression and can dictate malignancy. Nutrition status affects treatment response and the progression or recurrence of the tumor. This review summarizes the main bioactive compounds against the molecular pathways related to colorectal carcinogenesis. Moreover, we focus on the compounds with chemopreventive properties, mainly polyphenols and carotenoids, which are highly studied dietary bioactive compounds present in major types of food, like vegetables, fruits, and seeds. Their proprieties are antioxidant and gut microbiota modulation, important in the intestine because they decrease reactive oxygen species and inflammation, both principal causes of cancer. These compounds can promote apoptosis and inhibit cell growth, proliferation, and migration. Combined with oncologic treatment, a sensitization to first-line colorectal chemotherapy schemes, such as FOLFOX and FOLFIRI, is observed, making them an attractive and natural support in the oncologic treatment of CRC.
1. Introduction
Colorectal cancer (CRC) is third in incidence worldwide and second in mortality. One of the main risk factors for developing CRC is a lifestyle with poor physical activity and a diet high in fat and carbohydrates, which stimulates an inflammatory state that increases reactive oxygen species (ROS) and releases proinflammatory cytokines in the intestinal mucosa [1]. Nutritional status influences part of the response to treatment in cancer patients. For example, malnutrition predicts worse results in response to treatment and survival [2]; also, recurrence, progression, and the presence of metastasis have been attributed to poor nutritional status [3].
The bioactive compounds of food have been of interest since they may contribute to maintaining human health. These compounds are present in small amounts in plants and certain foods such as fruits, vegetables, nuts, and seeds. They can interact with tissue components, potentially promoting multiple health benefits [4]. Bioactive compounds have a wide spectrum of chemical structures, and their main function is as antioxidants; they also modulate the expression and activity of transcription growth factors and inflammatory mediators and are cell cycle intermediaries [1,5]. They can modulate immunoregulator molecules such as PD-L1 by modulating mitochondria activity and hypoxia, ubiquitination, glycosylation, and endoplasmic reticulum degradation, among others [6,7,8,9]. It has been proposed to consider the bioavailability of the compound rather than the amount present in food or dietary supplements to determine the benefit of the compound. It has been reported that the DNA damage repair mechanism can activate the expression of PD-L1 since BRCA2 or Ku70/80 depletion enhances PD-L1 upregulation after DSBs in an ATM/ATR/Chk1-dependent manner [10,11,12]. The consumption of various bioactive compounds, such as phenolic compounds, flavonoids, phenolic acids, carotenoids, and xanthophylls, influences the preservation of cell integrity, the maintenance of cell DNA repair systems, and their action in cell differentiation, proliferation, and apoptosis. This review summarizes the origin of bioactive compounds found in dietary sources such as curcumin, quercetin, resveratrol, lycopene, and cinnamic acid. It also explores their remarkable attributes in regulating the metabolic pathways linked to CRC and their significant roles in chemoprevention and reverse chemoresistance.
2. Generalities of Bioactive Compounds in CRC
Bioactive products have always played a significant role as novel therapeutical agents, irrespective of their source of origin. They are phenolic compounds, flavonoids, phenolic acids, carotenoids, and xanthophylls. Different concentrations are found in vegetables, fruits, grains, spices, and their derived foods and beverages, such as tea, olive oil, fruit juices, wine, chocolate, and beer, as shown in Table 1.

Table 1.
Bioactive compounds and food sources.
2.1. Polyphenols
Polyphenols are compounds in plants with one or more hydroxyl groups attached to an aromatic ring in their structure [13]. Their bioavailability and stability are low, and most of their metabolism occurs in the large intestine, where bacteria convert them to phenolic acid that cells absorb and use. Polyphenols are divided into flavonoids, quercetin, and epigallocatechin-3-gallate (EGCG). Phenolic acids are derivatives of hydroxybenzoic acid or hydroxycinnamic acid, tannins, lignans, and stilbenes.
Their main properties are antioxidant by suppressing the generation of reactive oxygen species and lipid peroxidation. However, they also have anti-inflammatory, immunomodulatory, anticancer, and gastroprotective effects [14].
One of its proposed mechanisms of action is inhibiting the NF-ĸB transcription factor, which is known to participate in the development of colorectal cancer and intestinal inflammatory diseases and is responsible for regulating inflammation, cell growth, and cell survival [15]. Another mechanism proposed for polyphenols as a bioactive compound against cancer is through the inhibition of proinflammatory enzymes, such as cyclooxygenase 2 (COX2) and 5-lipoxygenase [16], whose altered metabolism is common in neoplasms derived from the epithelium. Polymorphisms in COX1 and COX2 have been associated with the development of colorectal cancer, specifically COX2, which controls the cell cycle [17] and its expression increases in response to cytokines and growth factors. COX2 participates in cancer development by increasing cell proliferation, tumor angiogenesis [18], and the production of prostaglandins that seem to increase resistance to apoptosis in epithelial cells. In contrast, in stromal cells, it favors neovascularization present in the development of CRC.
2.2. Flavonoids
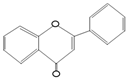
Flavonoids are the most abundant polyphenols and participate as proposals, along with conventional treatments, to eradicate different types of cancer, including colorectal cancer, by modulating various enzymes, such as phosphatidylinositol 3 kinase (PI3K), of which class I is involved in cancer [19]. They also modulate AKT/protein kinase B, involved in cell proliferation, survival, and growth, through its effect on the TSC1/TSC2, mTORC, MAPK, and glucose metabolism [20] pathways that participate in the amplification of molecules favoring cell proliferation, growth, and survival and may explain resistance to first-line treatments in CRC, such as irinotecan, 5-fluorouracil (5-FU), and cisplatin [21]. Resveratrol inhibits the paclitaxel-induced neuropathic pain caused by the PI3K/AKT and SIRT1/PGC1α pathways [22]. Urolithin A, a metabolite produced from the dietary polyphenol ellagic acid, potentiates the effects of 5-FU on colon cancer cells by regulating cyclins A and B1 and activating caspases 8 and 9 to trigger cell cycle arrest and apoptosis [23].
A few randomized controlled trials have evaluated the effect of polyphenols on CRC. Sinicrope et al. (2021) carried out a phase II study where they measured the effectiveness of the Poly E bioactive compound of green tea that contains between 55% and 72% of the active component epigallocatechin on the percentage change in the number of rectal aberrant crypts, which are considered premalignant lesions [24]. With a 6-month intervention with 780 mg oral supplementation of Poly E, they found that it did not significantly reduce the number of crypts, but adenoma recurrence decreased by 29% [24]. The antitumor effect on CRC cells in vitro and in vivo are related in a dose-dependent manner [25]. A report demonstrated that a higher intake of total polyphenols, total flavonoids, total phenolic acids, anthocyanin, and flavanols was associated with a reduction in CRC risk and that stilbenes were inversely associated with colorectal adenomas [26].
2.3. Carotenoids
Carotenoids, a vitamin A (retinol) source, contain a central carbon chain with alternating single and double bonds and present cyclic or acyclic terminal groups [27]. They are found in fruits, vegetables, and certain fish and are represented as B-carotene, lycopene, lutein, zeaxanthin, and curcuma. They function as antioxidants.
Carotenoids have been used as anti-tumor agents for inhibiting tumor growth, inducing apoptosis, modulating gene expression and the immune response, modulating the AP-1 transcriptional complex, WNT/pathway B catenin, and inhibiting mutagenesis [28], although cancer related to inflammation can, understandably, be hindered by carotenoids with anti-inflammatory effects, describing specific signaling pathways. CRC stem cell treatment with β-carotene reduces cell proliferation by regulating miRNA expression, elevating histone H3 and H4 acetylation, downregulating DNMT3A mRNA, and global DNA methylation [29].
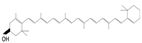
Studies have analyzed the intake impact of dietary carotenoids and CRC [30], finding that high B-carotene intake in men is negatively associated with CRC; hence, it has been proposed as a preventive compound since it can modulate M2 macrophages and fibroblasts, in addition to reducing COX2 [31]. The consumption of lycopene and lutein/zeaxanthin has a non-significant inverse association with CRC. Lycopene, in studies with CRC cell lines [32] inhibits AKT protein phosphorylation and, in the presence of eicosapentaenoic acid, suppresses the P1K3/AKT pathway, blocking mTOR activity [33]. It also improves antitumoral parameters such as catalase, glutathione, and IFN-γ expression [34]. Lycopene reduces the oxidative stress caused by chemotherapeutic 5-FU and oxaliplatin, decreasing the adverse effects of CRC treatment [35].
Similarly, astaxanthin, another carotenoid, can upregulate the expression of the epithelial marker E-cadherin and downregulate the mesenchymal markers vimentin and cortactin. It also increases miR-29a-3p and miR-200a expression, suppressing matrix metalloproteinase-2 (MMP2) and thus inhibiting the metastatic potential of CRC [36]. Moreover, fucoxanthin inhibited the multiplicity of CRC in mice by inducing apoptosis, evidenced by a high number of cleaved caspase-3 cells in colonic adenocarcinoma and mucosal crypts [37]. Terasaki et al. suggest that fucoxanthin prevents CRC by inducing anoikis, apoptosis caused by the disruption of the anchorage to the extracellular matrix or adjacent cells, and by suppressing integrin β1, p-FAK, and p-Paxillin in mice colonic crypt cells [38].
Carotenoids can participate in potentiating the chemotherapy effect when it is combined with 5-FU. Fucoxanthin and phloroglucinol potentiate the cytotoxic effect of chemotherapy in colon cancer cells without harming normal cells [39]. Fucoxanthinol, a fucoxanthin derivative, impeded cell proliferation by downregulating pAkt, PPARβ/δ, and PPARγ in the colonospheres formed by human CRC cells [40]. Crocin, another carotenoid found in saffron, has antiproliferative, antiangiogenic, and antimetastatic properties in CRC via TNF-α, NF-kB, and VEGF pathway suppression in CRC cells [41]. It also increases glutathione synthesis, enhances superoxide dismutase, and eliminates ROS [42]. It is also considered a bioactive compound against the depression [43] present in a high percentage of oncologic patients that can contribute to the adherence and response of oncologic treatment.
Regarding controversial findings in the literature, CRC risk reduction in the population through a higher dietary intake of polyphenols and carotenoids may be negligible compared to other risk factors; nevertheless, these findings could become significantly beneficial to higher-risk individuals. For example, people with inflammatory bowel disease (IBD), such as ulcerative colitis, who are predisposed to CRC, may have a more significant cancer risk reduction from diet modification than people without IBD. Studies have shown the potential cancer prevention effects of polyphenol-rich strawberries and black raspberries in IBD-related CRC, perhaps an intervention with low toxicity that can supplement current IBD treatment [44].
Research in regulating the metabolic pathways that promote the proliferation of tumor cells through dietary compounds may find more consistent results by focusing their studies on higher-risk populations. Another potential area of research is the synergistic anticancer effects of polyphenols and carotenoids. They are usually studied independently; however, dietary intake commonly includes compounds from both families, meaning their therapeutic benefits could derive from the chemical interactions between polyphenol and carotenoid structures.
3. Regulation of Molecular Pathways in Colorectal Carcinogenesis by Bioactive Compounds
A distinct hallmark of cancer is metabolism pathways, which is why research is focused on knowing how to regulate the metabolism pathways related to CRC, potentially using this knowledge to improve current treatments. The Wnt pathway, best known as Wnt/B catenin, participates in cancer initiation and progression and regulates the pluripotency of cells during development and differentiation [45]. The invasion and prognosis of metastasis are related to the epithelial–mesenchymal transition (EMT), the malignant proliferation of tumor cells, decreased apoptosis, and the regulation of expression of EMT markers. A loss of E-cadherin expression is a molecular key point [46]. Another molecular mechanism for developing CRC is the signal transducer and activator of transcription (STAT). In this regard, seven STAT factors share molecular characteristics that control their action mode; specifically, STAT1 and STAT3 have an important role in CRC progression [47]. Moreover, cytokines such as IFN-γ, IL-6, IL-1, and EGF [48] activate a family of proteins associated with their receptors, the Janus kinases (JAK), which activate STAT molecules. The most studied in CRC is JAK2/STAT3 since they relate to EMT. The JAK/STAT pathway promotes invasion, migration, growth, and chemoresistance [49].
The tumor microenvironment is the different cell types surrounding the tumor, including fibroblasts, endothelial cells, macrophages, dendritic cells, tumor stem cells, adipocytes, and the microbiome. They are key in promoting or restricting tumor development [50,51,52]. The changes that occur in the tumor microenvironment resemble a state of chronic inflammation, which begins with ischemia that continues to interstitial edema, the infiltration of immune cells, and angiogenesis, suggesting that the tumors are infiltrated by inflammatory cells and cytokines PDGF, EGF, IL-1, TNFα, and TGFβ [53] that favor oxidative stress and the presence of ROS [54].
CRC begins with altering the regulatory mechanisms of DNA repair systems and the cell proliferation of mucosa cells lining the colon and rectum. These cells convert into neoplasia that develops polyps that advance to high-grade dysplasia and evolve into invasive tumors. Early genetic mutations include the BRAF and APC genes [55].
CRC develops due to cancer stem cells (CSC), pluripotent stem cells with self-renewal capacity to different lineages found in the colon, promoting carcinogenesis and favoring tumor heterogeneity [56]. Microbiota and metabolome alterations are involved in pro- or anti-cancer actions [57]. Several phytochemicals involved in regulating the Hedgehog, Notch, and Wnt/β-catenin pathways, such as curcumin, quercetin, lycopene, cinnamic acid, resveratrol, sibylline, and EGCG, have been identified. Their importance in regulating pathways as they regulate the maintenance and proliferation of CSCs is summarized below (Table 2). The inflammation process promotes cancer progression through inflammatory cytokines in the tumor microenvironment, such as TNF-α, which is a mediator involved in chronic inflammatory diseases with greater participation during the early stages of carcinogenesis, angiogenesis, invasion, generating reactive oxygen, and nitrogen species [58,59]. Also, cytokines such as IL-2 and IL-6 favor tumor proliferation, inhibit apoptosis, and participate in the conversion of non-cancerous cells to tumor stem cells. Moreover, TGF-b improves invasion and metastasis by inducing the transition epithelium-mesenchymal cells and IFN-γ participate in metastasis [60]. Lastly, Wnt signaling is involved with NF-ĸB and MAPK; together, they can increase oxidative stress and inhibit apoptosis [61,62].

Table 2.
Bioactive compounds and their action in metabolic pathways in CRC.
Curcumin and quercetin are the most studied bioactive compounds regarding CRC. On the one hand, curcumin is a natural and active derivative of turmeric. It is an oily molecule soluble in acetic acid and ketones but appears insoluble in water. It possesses anti-angiogenic, anti-tumor, antiproliferative, and anti-inflammatory properties through various mechanisms, including the suppression of intrinsic and extrinsic apoptotic signaling pathways, cell cycle arrest, and the activation of autophagy. Curcumin, obtained from the dried root of Curcuma longa, can kill only tumor cells without harming healthy cells due to a high expression of a protein known as GADD45a (the gene activated during DNA damage). However, there are limitations in its use as a therapeutic agent since it has low solubility, poor absorption, rapid metabolism, and rapid elimination, which is why the use of nanoparticles has been implemented to encapsulate it so that it can achieve its therapeutic effect within the tumor [89,90] On the other hand, Quercetin is a secondary metabolite of plants. It has an anti-tumor effect by regulating the signal transduction pathways to prevent, inhibit or reverse carcinogenesis. It also has anti-inflammatory and antioxidant effects. It may potentiate cytotoxic effects or reduce the side effects of chemotherapeutic drugs on normal cells or reverse drug susceptibility [90].
Numerous health benefits of these food supplements are recognized by pioneering experimental studies involving both in vitro and in vivo studies in recent decades, including their antioxidant and anti-inflammatory potential, digestive stimulant effects, hypolipidemic actions, antilithogenic properties, anti-diabetic influence, antimutagenic, and anticancer potential [91]. Studies have shown that spices and their bioactive compounds can inhibit or even activate the pathways related to cell division, proliferation, and detoxification, in addition to having immunomodulatory and anti-inflammatory effects [92].
4. Chemosensitive and Chemopreventive Properties by Bioactive Compounds in CRC
There are several applications for polyphenols and carotenoids that have not been sufficiently explored, such as their potential use for predisposition to CRC [93,94], sensitizers to current CRC standard chemotherapy [23,39] candidates for treatment [95,96], and the reduction in its recurrence [97]. Using polyphenols and carotenoids as sensitizers and buffers to chemotherapy and radiotherapy could decrease drug resistance and minimize toxicity by requiring lower doses [98,99]. Further research into the adjuvant treatment of CRC with dietary polyphenols and carotenoids may yield improved outcomes for patients with lower costs and minimal risks.
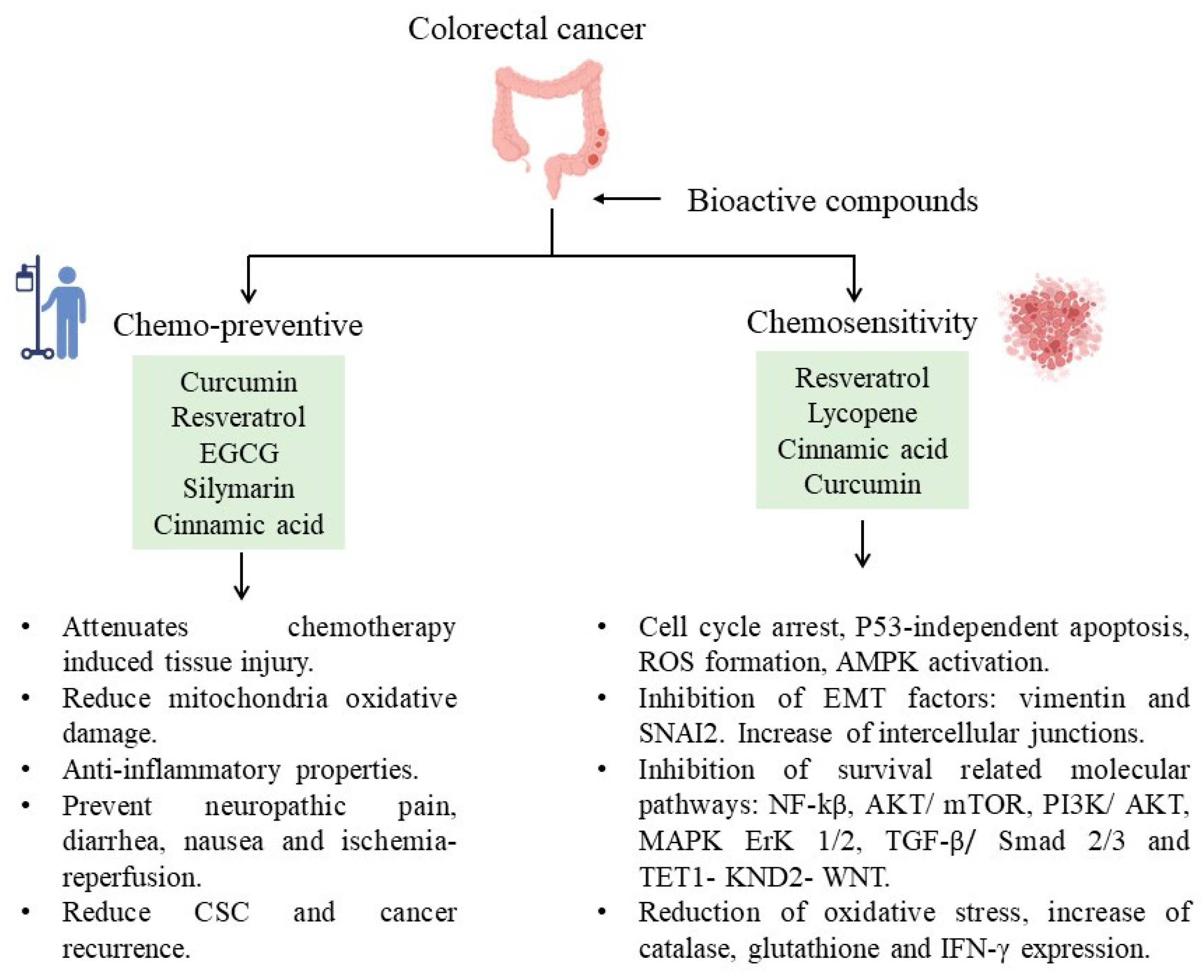
These bioactive compounds have recently been found to protect against chemotherapy side effects or modify susceptibility by reversing chemoresistance (Figure 1). For example, curcumin attenuates oxaliplatin-induced liver injury by activating nuclear erythroid factor 2-related factor 2 (Nrf2) signaling, regulating cellular defense pathways, and the oxidative damage to mitochondria caused by oxaliplatin [100]. Furthermore, curcumin protects against irinotecan-induced intestinal injury inhibiting NF-ĸB activation [101]. It is also active against FOLFIRI and bevacizumab cardiotoxicity by suppressing oxidative stress and preventing mitochondrial dysfunction in cardiac mitochondria [102]. Regarding quercetin, reports from specific cell lines (SW620) exposed to this bioactive compound appeared to be more sensitive to doxorubicin due to the inhibition of ATP-driven transport activity of P-glycoprotein that leads to a higher concentration of doxorubicin at the intracellular level. Also, it has been proposed that quercetin can reverse multidrug resistance by regulating the expression of glutamine transporter 1 member 5 (SLC1A5) [103].
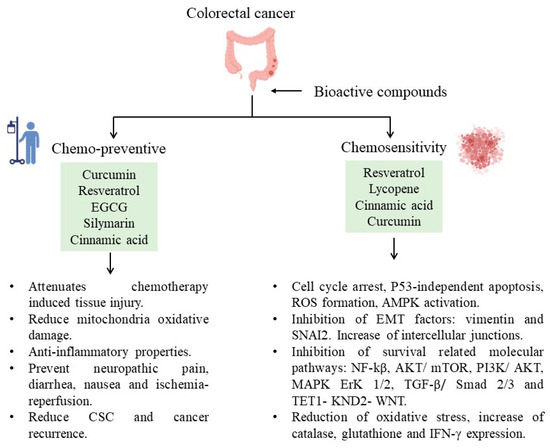
Figure 1.
Bioactive compounds and their participation in decreasing CRC.
The studies of resveratrol against cancer have demonstrated chemopreventive functions related to anti-inflammatory, antioxidant, anti-apoptosis, and anti-proliferative properties [104,105,106,107]. Resveratrol may reduce the side effects of chemotherapy, such as renal toxicity, cardiotoxicity, gastrointestinal toxicity, hepatotoxicity, and UVR-induced skin cancer [108]. In addition, resveratrol has been used as a chemosensitizer in CRC cells (HCT116, HT-29, and SW620) to 5-FU by inducing cell cycle arrest and apoptosis independently of p53 status and inhibiting their endogenous antioxidant capacity, respectively [109,110]. In a study with etoposide-resistant HT-29 cells, resveratrol was a chemosensitizer, inducing cell cycle inhibition, ROS formation, AMPK activation, and apoptosis induction [111]. The activation of AMPK and SIRT1 has long been thought to be the mechanism via which dietary bioactive compounds influence their health benefits. According to Liu et al., resveratrol inhibits proliferation and induces death in ovarian cancer cells (A2780 and SKOV3) by reducing glycolysis and targeting the AMPK/mTOR signaling pathway [112]. In breast cancer, resveratrol regulates EMT by modulating TGF-β1. Furthermore, resveratrol can induce autophagy by upregulating SIRT3 and phosphorylated AMPK, suggesting that the resveratrol-mediated inhibition of tumor progression is attributed to the participation of the SIRT3/AMPK/autophagy signaling axis [113]. Another example is curcumin-induced AMPK and its downstream factor ACC phosphorylation in lung cancer cells [114,115] and prostate cancer [116]. EGCG promotes cell survival by shifting the balance of the mTOR-AMPK pathway during ER stress [117]. The indirect AMPK activators show promise in treating various disorders such as cancer. The effects of systemic and chronic AMPK activation remain unknown.
Resveratrol also sensitizes CRC cells to 5-FU by inhibiting EMT factors (vimentin and SNAI2 proteins), increasing intercellular junctions (desmosomes, gap, and tight junctions, and adhesion molecules such as E-cadherin), and inhibiting the NF-kB pathway. Also, it has been reported that this compound downregulates p-AKT in cancer cell lines; however, it also upregulates the same pathway to prevent paclitaxel-induced neuropathic pain or ischemia-reperfusion injuries [118]. The combined treatment of 5-FU and resveratrol in colorectal cancer sensitizes tumor cells to 5-FU, which induces a further increase in oxidative stress related to the inhibition of the AKT and STAT3 proteins [119] known for their oncogenic potential in colorectal carcinomas [120].When resveratrol is combined with forskolin, it has phosphodiesterase 4D inhibitory effects to inhibit AKT/mTOR signaling in colorectal cancer cells [121]. In the same polyphenol family, EGCG can decrease the probability of recurrence of colorectal adenocarcinoma [122] in combination with cisplatin or oxaliplatin, evidenced by an improved therapeutic effect.
Another chemosensitizer mechanism to 5-FU by resveratrol is through miR-34 regulation. This mechanism was studied in vitro using HCT116 cell lines. The mechanism was mediated by inhibiting the PI3K/Akt and MAPK Erk1/2 signaling pathways that increased miR-34a production and thus indirectly suppressed the SIRT1 gene via E2F3 gene expression [123,124]. Also, a study found that resveratrol sensitizes HT-29 and HCT-116 CRC cells to oxaliplatin, increasing the expression of miR-34c, which inhibits its target, KITLG [125]. Moreover, when resveratrol and oxaliplatin are combined, the cell proliferation of Caco-2 CRC cells decreases due to the induction of apoptosis and necrosis [126].
Previous studies have reported that consuming lycopene during cancer treatment may reduce the oxidative stress caused by 5-FU and oxaliplatin treatments, reducing the associated side effects. It has also been proposed as a chemosensitizer due to the possibility of inducing the expression of apoptotic genes [35]. In addition, lycopene enhances parameters such as catalase, glutathione, and IFN-γ expression in the presence of 5-FU, which may activate antitumor effects and further enhance the effect of 5-FU on cancer elimination [34]. In this regard, in 2021, it was reported that silymarin supplementation could reduce the occurrence of side effects such as diarrhea and nausea in patients with CRC receiving FOLFIRI as first-line treatment plus bevacizumab; however, it does not show a hepatoprotective effect [127].
According to studies, cinnamic acid has cardioprotective [128] and hepatoprotective activity and an analgesic effect during chemotherapy [129]. For example, an in vivo model with rats (Sprague Dawley) treated with oxaliplatin developed side effects; meanwhile, when cinnamic acid was provided, neuropathic pain decreased [130].
There are a few studies of cinnamic acid and CRC as a chemosensitizer; however, it has been recently demonstrated that cinnamic acid alone or combined with FOLFOX reduces side population cells and cancer stem cell markers in the HT29 cell line [73]. However, the chemosensitizer potential of cinnamic acid in chemotherapy has not been explored, so we consider it a promising candidate for study.
Curcumin can reverse chemoresistance by upregulating EMT markers by attenuating the TGF-β/Smad2/3 signaling pathway or downregulating TET1-NKD2-WNT [131]. Curcumin as an adjuvant treatment to FOLFOX chemotherapy in CRC patients did not decrease neurotoxicity or quality of life but improved overall survival in the curcumin group versus those with only chemotherapy [132]. However, in vitro curcumin has been able to sensitize colon cancer stem cells to chemotherapy drugs such as 5-FU, FOLFOX, and irinotecan, further reducing the emergence of chemoresistant cells [70]. In addition, curcumin and resveratrol have been linked to the ability to prevent breast metastasis and gastric cancer, osteosarcoma, and lung cancer through the coordinated action of several molecular pathways such as the SIRT3/AMPK/autophagy signal axis, JAK2/STAT3, and MALAT1/miR-383-5p/DDIT4 [113]. Quercetin involves reduced metastasis by negatively regulating the expression of the protein survivin, which is expressed in the macrophages that regulate the processes of proliferation and cell death. This bioactive compound also regulates cyclin D1, which controls the cell cycle.
5. Conclusions
CRC has a high incidence and is the second most deadly cancer worldwide. However, it is largely preventable and treatable, and nutrition plays a pivotal role. Dietary bioactive compounds in foods such as vegetables, fruits, seeds, nuts, oils, and tea are important in cancer prevention since they may regulate cell differentiation processes, proliferation, and apoptosis; all are affected in cancer cells. Bioactive compounds regulate these molecular pathways related to inflammatory diseases like CRC.
Polyphenols and carotenoids are the most studied bioactive compounds due to their antioxidant, anti-inflammatory properties, and their modulation of gut microbiota. Also, a higher intake of total bioactive compounds, such as phenolic acid, anthocyanin, and flavonoids, has been related to a reduced CRC risk, acting as preventive compounds. The positive evidence about bioactive compounds promotes their study as a strong line of research, and the knowledge obtained could improve the quality of life of patients and reduce all the associated effects of chemotherapy, such as renal, cardio, gastro, and hepatotoxicity, making these compounds an important part of the prevention and treatment of CRC.
The diverse molecular pathways related to CRC, like JAK/STAT, Wnt/β catenin, EMT, PI3K/AKT, and mTOR, can be regulated by bioactive compounds promoting apoptosis, inhibiting or decreasing cell growth and proliferation, reducing inflammation, eliminating cancer stem cells, and avoiding chemoresistance. Even though all the bioactive compounds have different mechanisms of action, many share mechanisms, such as the regulation of pro- or anti-apoptotic proteins, the modulation of the tumor microenvironment, the regulation of the expression of efflux pumps, and a reduction in oxidative stress.
Since these compounds have an antitumoral effect, their potential has been explored with oncologic treatments, including monoclonal antibodies and CRC first-line chemotherapy, 5-FU.
We consider that good nutrition, represented by consuming bioactive compounds, can benefit oncologic patients. Nevertheless, there are limitations to consider, such as the metabolite status, the administration form, and the physiological status of the patients. These may influence the correct biotransformation, utilization, and beneficial effects. Another limitation is the low number of studies demonstrating the chemosensitivity of these bioactive compounds. Several investigations have only reported in vitro assays with cancer cell lines. There are still important questions about bioactive compounds and their action in cancer, which is why carrying out research with primary culture cells is relevant in order to evaluate the potential of bioactive compounds at different doses and determine their effectiveness.
Author Contributions
Conceptualization, writing—original draft preparation P.D.-G.; writing—review and editing, E.N.G.-T., D.A.d.l.G.K., A.Q.R. and E.A.H.-T. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The authors are grateful for the support provided by Sergio Lozano-Rodríguez (Scientific Publications Support Coordinator, UANL) in reviewing this manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| 5-FU | 5-Fluorouracil |
| ABCB1 | ATP binding cassette gene B1 |
| AKT | Serine/threonine kinase |
| ALDH1A1 | Aldehyde dehydrogenase 1A1 |
| AP-1 | Activator protein-1 |
| APC | Adenomatous polyposis coli |
| BAX | Bcl-2 Associated X-protein |
| BCL-2 | B-cell lymphoma 2 |
| Bim | Bcl-2-interacting mediator |
| BRAF | V-Raf Murine Sarcoma Viral Oncogene Homolog B oncogene |
| COX 1 | Cyclooxygenase 1 |
| COX 2 | Cyclooxygenase 2 |
| CRC | Colorectal Cancer |
| CSC | Cancer Stem Cells |
| DNA | Deoxiribonucleic acid |
| DNMT3A | DNA methyltransferase 3 alpha |
| DR5 | Death Receptor 5 |
| EGCG | Epigallocatechin-3-gallate |
| EMT | Epithelial Mesenchymal Transition |
| ERK | Extracellular Signal-Regulated Kinase |
| FOLFIRI | Folinic acid, Fluorouracil and Irinotecan |
| FOLFOX | Folinic acid, Fluorouracil and Oxaliplatin |
| GSK-3 | Glycogen Synthase Kinase-3 |
| IBD | Inflammatory Bowel Disease |
| IFN-γ | Interferon gamma |
| iNOS | inducible Nitric Oxide Synthase |
| JAK | Janus Kinases |
| KITLG | c-KIT ligand gene |
| MAPK | Mitogen-Activated Protein Kinases |
| MCL-1 | Myeloid Leukemia 1 |
| MiRNA | Micro RNA |
| MMP-2 | Matrix Metalloproteinases 2 |
| MMP-9 | Matrix Metalloproteinases 9 |
| mRNA | Messenger RNA |
| mTORC | mechanistic Target Of Rapamycin Complex 1 |
| NF-kB | Nuclear Factor Kappa-light-chain-enhancer of Activated B cells |
| Nrf2 | Nuclear Erytroid Factor 2 related factor 2 |
| OCT 4 | Octamer-Binding Transcription Factor 4 |
| PARP | The Poly (ADP-ribose) Polymerase |
| PCN | Phenazine-1-carboxamide |
| p-FAK | Phospho- Focal Adhesion Kinase |
| PGC1α | Peroxisome Proliferator-activated receptor-gamma Coactivator α |
| PGE2 | Prostaglandin E2 |
| PI3K | Phosphatidylinositol 3-kinase |
| PPARβ | Perosyxomel Proliferator Activated Receptor β |
| PPARδ | Perosyxomel Proliferator Activated Receptor δ |
| PPARγ | Perosyxomel Proliferator Activated Receptor γ |
| PTEN | Phosphatase and Tensin homolog |
| ROS | Reactive Oxigen Species |
| SIRT1 | Silent Information Regulator 1 |
| SLC1A5 | Solute Carrier Family 1 Member 5 |
| STAT | Signal Transducer and Activator of Transcription |
| STR1 | Stromelysin-1 |
| SUMO | Small Ubiquitin-like Modifier |
| TGF-β | Transforming Growth Factor beta |
| TLR4 | Toll Type Receptor 4 |
| TNF-α | Tumor Necrosis Factor Alpha α |
| TSC1 | Tuberous Sclerosis Complex 1 |
| TSC2 | Tuberous Dclerosis Complex 2 |
| UVR | Ultraviolet Radiation |
| VEGF | Vascular Endothelial Growth Factor |
| WNT | Wingless-related integration site |
References
- Nosrati, N.; Bakovic, M.; Paliyath, G. Molecular mechanisms and pathways as targets for cancer prevention and progression with dietary compounds. Int. J. Mol. Sci. 2017, 18, 2050. [Google Scholar] [CrossRef] [PubMed]
- Salas, S.; Cottet, V.; Dossus, L.; Fassier, P.; Ginhac, J.; Latino-Martel, P.; Romieu, I.; Schneider, S.; Srour, B.; Touillaud, M.; et al. Nutritional Factors during and after Cancer: Impacts on Survival and Quality of Life. Nutrients 2022, 14, 2958. [Google Scholar] [CrossRef] [PubMed]
- Bullock, A.F.; Greenley, S.L.; McKenzie, G.A.G.; Paton, L.W.; Johnson, M.J. Relationship between markers of malnutrition and clinical outcomes in older adults with cancer: Systematic review, narrative synthesis and meta-analysis. Eur. J. Clin. Nutr. 2020, 74, 1519–1535. [Google Scholar] [CrossRef] [PubMed]
- Guaadaoui, A.; Benaicha, S.; Elmajdoub, N.; Bellaoui, M.; Hamal, A. What is a bioactive compound? A combined definition for a preliminary consensus. Int. J. Food Sci. Nutr. 2014, 3, 17–179. [Google Scholar] [CrossRef]
- González, S. Dietary Bioactive Compounds and Human Health and Disease. Nutrients 2020, 12, 348. [Google Scholar] [CrossRef]
- Liu, K.; Sun, Q.; Liu, Q.; Li, H.; Zhang, W.; Sun, C. Focus on immune checkpoint PD-1/PD-L1 pathway: New advances of polyphenol phytochemicals in tumor immunotherapy. Biomed. Pharmacother. 2022, 154, 113618. [Google Scholar] [CrossRef]
- Zheng, C.; Luo, W.; Liu, Y.; Chen, J.; Deng, H.; Zhou, Z.; Shen, J. Killing three birds with one stone: Multi-stage metabolic regulation mediated by clinically usable berberine liposome to overcome photodynamic immunotherapy resistance. Chem. Eng. J. 2023, 454, 140164. [Google Scholar] [CrossRef]
- Lim, S.O.; Li, C.W.; Xia, W.; Cha, J.H.; Chan, L.C.; Wu, Y.; Chang, S.-S.; Lin, W.-C.; Hsu, J.-M.; Hsu, Y.-H.; et al. Deubiquitination and Stabilization of PD-L1 by CSN5. Cancer Cell 2016, 30, 925–939. [Google Scholar] [CrossRef]
- Kang, D.Y.; Sp, N.; Jo, E.S.; Rugamba, A.; Hong, D.Y.; Lee, H.G.; Yoo, J.-S.; Liu, Q.; Jang, K.-J.; Yang, Y.M. The Inhibitory Mechanisms of Tumor PD-L1 Expression by Natural Bioactive Gallic Acid in Non-Small-Cell Lung Cancer (NSCLC) Cells. Cancers 2020, 12, 727. [Google Scholar] [CrossRef]
- Zhou, Z.; Liu, Y.; Jiang, X.; Zheng, C.; Luo, W.; Xiang, X.; Qi, X.; Shen, J. Metformin modified chitosan as a multi-functional adjuvant to enhance cisplatin-based tumor chemotherapy efficacy. Int. J. Biol. Macromol. 2023, 224, 797–809. [Google Scholar] [CrossRef]
- Sato, H.; Niimi, A.; Yasuhara, T.; Permata, T.B.M.; Hagiwara, Y.; Isono, M.; Nuryadi, E.; Sekine, R.; Oike, T.; Kakoti, S.; et al. DNA double-strand break repair pathway regulates PD-L1 expression in cancer cells. Nat. Commun. 2017, 8, 1751. [Google Scholar] [CrossRef]
- Kciuk, M.; Kołat, D.; Kałuzińska-Kołat, Ż.; Gawrysiak, M.; Drozda, R.; Celik, I.; Kontek, R. PD-1/PD-L1 and DNA Damage Response in Cancer. Cells 2023, 12, 530. [Google Scholar] [CrossRef] [PubMed]
- Toche Tuesta, A.; Curay Carhuamaca, V.L.; Diaz Barrientos, R.; Fernández Rebaza, G.A.; Bonilla Rivera, P.E. Estructura química de compuestos fenólicos del extracto etanólico de hojas de Clinopodium pulchellum (Kunth) Govaerts “panisara”. Rev. Peru Med. Integr. 2017, 2, 803–809. [Google Scholar] [CrossRef][Green Version]
- Chiu, H.F.; Venkatakrishnan, K.; Golovinskaia, O.; Wang, C.K. Gastroprotective Effects of Polyphenols against Various Gastro-Intestinal Disorders: A Mini-Review with Special Focus on Clinical Evidence. Molecules 2021, 26, 2090. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Jiang, Q. Roles of the Polyphenol–Gut Microbiota Interaction in Alleviating Colitis and Preventing Colitis-Associated Colorectal Cancer. Adv. Nutr. 2021, 12, 546–565. [Google Scholar] [CrossRef] [PubMed]
- Gomes, R.N.; Felipe da Costa, S.; Colquhoun, A. Eicosanoids and cancer. Clinics 2018, 73, e530s. [Google Scholar] [CrossRef] [PubMed]
- Gholamalizadeh, M.; Majidi, N.; Tajaddod, S.; Abdollahi, S.; Poorhosseini, S.M.; Ahmadzadeh, M.; Joubani, M.N.; Dahka, S.M.; Shafaei, H.; Hajiesmaeil, M.; et al. Interactions of Colorectal Cancer, Dietary Fats, and Polymorphisms of Arachidonate Lipoxygenase and Cyclooxygenase Genes: A Literature Review. Front. Oncol. 2022, 12, 865208. [Google Scholar] [CrossRef]
- Wasti, H.; Shawana, S. Comparision of BRAF V600E, COX–2 and p53 as Biomarkers for the Early Detection of Colorectal Cancer. J. Bahria Univ. Med. Dent. Coll. 2019, 9, 147–150. [Google Scholar] [CrossRef]
- Aldecoa, F.; Ávila, J. La vía canónica PI3K/AKT/mTOR y sus alteraciones en cáncer. Horiz. Médico. 2021, 21, e1547. [Google Scholar] [CrossRef]
- Miricescu, D.; Totan, A.; Stefani, C.; Stănescu, I.I.; Stanescu, A.M.A.; Rusu, I.R.; Radulescu, R.; Rusu, G.C.; Greabu, M. Structure, activation and biological effects of AKT or protein kinase B. Rom. J. Med. Pract. 2019, 14, 246–250. [Google Scholar] [CrossRef]
- Braicu, C.; Buse, M.; Busuioc, C.; Drula, R.; Gulei, D.; Raduly, L.; Rusu, A.; Irimie, A.; Atanasov, A.G.; Slaby, O.; et al. A Comprehensive Review on MAPK: A Promising Therapeutic Target in Cancer. Cancers 2019, 11, 1618. [Google Scholar] [CrossRef]
- Li, X.; Yang, S.; Wang, L.; Liu, P.; Zhao, S.; Li, H.; Jiang, Y.; Guo, Y.; Wang, X. Resveratrol inhibits paclitaxel-induced neuropathic pain by the activation of PI3K/Akt and SIRT1/PGC1&α; pathway. J. Pain Res. 2019, 12, 879–890. [Google Scholar]
- González-Sarrías, A.; Tomé-Carneiro, J.; Bellesia, A.; Tomás-Barberán, F.A.; Espín, J.C. The ellagic acid-derived gut microbiota metabolite, urolithin A, potentiates the anticancer effects of 5-fluorouracil chemotherapy on human colon cancer cells. Food Funct. 2015, 6, 1460–1469. [Google Scholar] [CrossRef] [PubMed]
- Sinicrope, F.A.; Viggiano, T.R.; Buttar, N.S.; Song, L.M.W.K.; Schroeder, K.W.; Kraichely, R.E.; Kisiel, J.B.; Gostout, C.J.; Kalaiger, A.M. Randomized Phase II Trial of Polyphenon E versus Placebo in Patients at High Risk of Recurrent Colonic Neoplasia. Cancer Prev. Res. 2021, 14, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Herrero de la Parte, B.; Rodeño-Casado, M.; Iturrizaga Correcher, S.; Mar Medina, C.; García-Alonso, I. Curcumin Reduces Colorectal Cancer Cell Proliferation and Migration and Slows In Vivo Growth of Liver Metastases in Rats. Biomedicines 2021, 9, 1183. [Google Scholar] [CrossRef]
- Bahrami, A.; Jafari, S.; Rafiei, P.; Beigrezaei, S.; Sadeghi, A.; Hekmatdoost, A.; Rashidkhani, B.; Hejazi, E. Dietary intake of polyphenols and risk of colorectal cancer and adenoma—A case-control study from Iran. Complement. Ther. Med. 2019, 45, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Alexander, S.P.; Kelly, E.; Marrion, N.; Peters, J.A.; Benson, H.E.; Faccenda, E.; Pawson, A.J.; Sharman, J.L.; Southan, C.; Buneman, O.P.; et al. The Concise Guide to PHARMACOLOGY 2015/16: Overview. Br. J. Pharmacol. 2015, 172, 5729–5743. [Google Scholar] [CrossRef]
- Milani, A.; Basirnejad, M.; Shahbazi, S.; Bolhassani, A. Carotenoids: Biochemistry, pharmacology and treatment. Br. J. Pharmacol. 2017, 174, 1290–1324. [Google Scholar] [CrossRef]
- Kim, D.; Kim, Y.; Kim, Y. Effects of β-carotene on Expression of Selected MicroRNAs, Histone Acetylation, and DNA Methylation in Colon Cancer Stem Cells. J. Cancer Prev. 2019, 24, 224–232. [Google Scholar] [CrossRef]
- Han, X.; Zhao, R.; Zhang, G.; Jiao, Y.; Wang, Y.; Wang, D.; Cai, H. Association of Retinol and Carotenoids Content in Diet and Serum With Risk for Colorectal Cancer: A Meta-Analysis. Front. Nutr. 2022, 9, 918777. [Google Scholar] [CrossRef]
- Desai, S.J.; Prickril, B.; Rasooly, A. Mechanisms of Phytonutrient Modulation of Cyclooxygenase-2 (COX-2) and Inflammation Related to Cancer. Nutr. Cancer 2018, 70, 350–375. [Google Scholar] [CrossRef]
- Costea, T.; Hudiță, A.; Ciolac, O.A.; Gălățeanu, B.; Ginghină, O.; Costache, M.; Ganea, C.; Mocanu, M.M. Chemoprevention of Colorectal Cancer by Dietary Compounds. Int. J. Mol. Sci. 2018, 19, 3787. [Google Scholar] [CrossRef] [PubMed]
- Carini, F.; David, S.; Tomasello, G.; Mazzola, M.; Damiani, P.; Rappa, F.; Battaglia, L.; Geagea, A.G.; Jurjus, R.; Leone, A. Colorectal cancer: An update on the effects of lycopene on tumor progression and cell proliferation. J. Biol. Regul. Homeost. Agents 2017, 31, 769–774. [Google Scholar] [PubMed]
- Alhoshani, N.M.; Al-Zharani, M.; Almutairi, B.; Aljarba, N.H.; AL-Johani, N.S.; Alkeraishan, N.; AlKahtane, A.A.; Alarifi, S.; Ali, D.; Alkahtani, S. Antioxidant and anti-inflammatory activities of lycopene against 5-fluorouracil-induced cytotoxicity in Caco2 cells. Saudi Pharm. J. 2022, 30, 1665–1671. [Google Scholar] [CrossRef] [PubMed]
- Alhoshani, N.M.; Al-Johani, N.S.; Alkeraishan, N.; Alarifi, S.; Alkahtani, S. Effect of lycopene as an adjuvant therapy with 5-florouracil in human colon cancer. Saudi J. Biol. Sci. 2022, 29, 103392. [Google Scholar] [CrossRef] [PubMed]
- Faraone, I.; Sinisgalli, C.; Ostuni, A.; Armentano, M.F.; Carmosino, M.; Milella, L.; Russo, D.; Labanca, F.; Khan, H. Astaxanthin anticancer effects are mediated through multiple molecular mechanisms: A systematic review. Pharmacol. Res. 2020, 155, 104689. [Google Scholar] [CrossRef]
- Terasaki, M.; Uehara, O.; Ogasa, S.; Sano, T.; Kubota, A.; Kojima, H.; Miyashita, K.; Mutoh, M. Alteration of fecal microbiota by fucoxanthin results in prevention of colorectal cancer in AOM/DSS mice. Carcinogenesis 2021, 42, 210–219. [Google Scholar] [CrossRef]
- Terasaki, M.; Iida, T.; Kikuchi, F.; Tamura, K.; Endo, T.; Kuramitsu, Y.; Tanaka, T.; Maeda, H.; Miyashita, K.; Mutoh, M. Fucoxanthin potentiates anoikis in colon mucosa and prevents carcinogenesis in AOM/DSS model mice. J. Nutr. Biochem. 2019, 64, 198–205. [Google Scholar] [CrossRef]
- Lopes-Costa, E.; Abreu, M.; Gargiulo, D.; Rocha, E.; Ramos, A.A. Anticancer effects of seaweed compounds fucoxanthin and phloroglucinol, alone and in combination with 5-fluorouracil in colon cells. J. Toxicol. Environ. Health A 2017, 80, 776–787. [Google Scholar] [CrossRef]
- Terasaki, M.; Maeda, H.; Miyashita, K.; Tanaka, T.; Miyamoto, S.; Mutoh, M. A marine bioofunctional lipid, fucoxanthinol, attenuates human colorectal cancer stemmlike cell tumorigenicity and sphere formation. J. Clin. Biochem. Nutr. 2017, 61, 25–32. [Google Scholar] [CrossRef]
- Bakshi, H.A.; Quinn, G.A.; Nasef, M.M.; Mishra, V.; Aljabali, A.A.A.; El-Tanani, M.; Serrano-Aroca, Á.; Webba Da Silva, M.; McCarron, P.A.; Tambuwala, M.M. Crocin Inhibits Angiogenesis and Metastasis in Colon Cancer via TNF-α/NF-kB/VEGF Pathways. Cells 2022, 11, 1502. [Google Scholar] [CrossRef] [PubMed]
- Veisi, A.; Akbari, G.; Mard, S.A.; Badfar, G.; Zarezade, V.; Mirshekar, M.A. Role of crocin in several cancer cell lines: An updated review. Iran. J. Basic Med. Sci. 2020, 23, 3–12. [Google Scholar] [PubMed]
- Tao, W.; Ruan, J.; Wu, R.; Zhao, M.; Zhao, T.; Qi, M.; Yau, S.S.; Yao, G.; Zhang, H.; Hu, Y.; et al. A natural carotenoid crocin exerts antidepressant action by promoting adult hippocampal neurogenesis through Wnt/β-catenin signaling. J. Adv. Res. 2023, 43, 219–231. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Shi, N.; Afzali, A. Chemopreventive Effects of Strawberry and Black Raspberry on Colorectal Cancer in Inflammatory Bowel Disease. Nutrients 2019, 11, 1261. [Google Scholar] [CrossRef]
- La Vecchia, S.; Sebastián, C. Metabolic pathways regulating colorectal cancer initiation and progression. Semin. Cell Dev. Biol. 2020, 98, 63–70. [Google Scholar] [CrossRef]
- Yang, Y.; Feng, M.; Bai, L.L.; Liao, W.; Zhou, K.; Zhang, M.; Wu, Q.; Wen, F.; Lei, W.; Zhang, P.; et al. Comprehensive analysis of EMT-related genes and lncRNAs in the prognosis, immunity, and drug treatment of colorectal cancer. J. Transl. Med. 2021, 19, 391. [Google Scholar] [CrossRef]
- Erdogan, F.; Radu, T.B.; Orlova, A.; Qadree, A.K.; de Araujo, E.D.; Israelian, J.; Valent, P.; Mustjoki, S.M.; Herling, M.; Moriggl, R.; et al. JAK-STAT core cancer pathway: An integrative cancer interactome analysis. J. Cell Mol. Med. 2022, 26, 2049–2062. [Google Scholar] [CrossRef]
- Ni, Y.; Low, J.T.; Silke, J.; O’Reilly, L.A. Digesting the Role of JAK-STAT and Cytokine Signaling in Oral and Gastric Cancers. Front. Immunol. 2022, 13, 835997. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Y.; Song, H.; Yin, H.; Jiang, T.; Xu, Y.; Liu, L.; Wang, H.; Gao, H.; Wang, R.; et al. The circular RNA circSPARC enhances the migration and proliferation of colorectal cancer by regulating the JAK/STAT pathway. Mol. Cancer 2021, 20, 81. [Google Scholar] [CrossRef]
- Kovács, T.; Mikó, E.; Ujlaki, G.; Sári, Z.; Bai, P. The Microbiome as a Component of the Tumor Microenvironment; Springer: Berlin/Heidelberg, Germany, 2020; pp. 137–153. [Google Scholar]
- Hanus, M.; Parada-Venegas, D.; Landskron, G.; Wielandt, A.M.; Hurtado, C.; Alvarez, K.; Hermoso, M.A.; López-Köstner, F.; De la Fuente, M. Immune System, Microbiota, and Microbial Metabolites: The Unresolved Triad in Colorectal Cancer Microenvironment. Front. Immunol. 2021, 12, 612826. [Google Scholar] [CrossRef]
- Laplane, L.; Duluc, D.; Bikfalvi, A.; Larmonier, N.; Pradeu, T. Beyond the tumour microenvironment. Int. J. Cancer 2019, 145, 2611–2618. [Google Scholar] [CrossRef] [PubMed]
- Aboelella, N.S.; Brandle, C.; Kim, T.; Ding, Z.C.; Zhou, G. Oxidative Stress in the Tumor Microenvironment and Its Relevance to Cancer Immunotherapy. Cancers 2021, 13, 986. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Han, Z.P.; Zhang, S.S.; Jing, Y.Y.; Bu, X.X.; Wang, C.Y.; Sun, K.; Jiang, G.-C.; Zhao, X.; Li, R.; et al. Effects of inflammatory factors on mesenchymal stem cells and their role in the promotion of tumor angiogenesis in colon cancer. J. Biol. Chem. 2011, 286, 25007–25015. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.H.; Goel, A.; Chung, D.C. Pathways of Colorectal Carcinogenesis. Gastroenterology 2020, 158, 291–302. [Google Scholar] [CrossRef]
- Walcher, L.; Kistenmacher, A.K.; Suo, H.; Kitte, R.; Dluczek, S.; Strauß, A.; Blaudszun, A.-R.; Yevsa, T.; Fricke, S.; Kossatz-Boehlert, U. Cancer Stem Cells—Origins and Biomarkers: Perspectives for Targeted Personalized Therapies. Front. Immunol. 2020, 11, 1280. [Google Scholar] [CrossRef]
- Yang, Y.; Misra, B.B.; Liang, L.; Bi, D.; Weng, W.; Wu, W.; Cai, S.; Qin, H.; Goel, A.; Li, X.; et al. Integrated microbiome and metabolome analysis reveals a novel interplay between commensal bacteria and metabolites in colorectal cancer. Theranostics 2019, 9, 4101–4114. [Google Scholar] [CrossRef]
- Landskron, G.; De La Fuente, M.; Thuwajit, P.; Thuwajit, C.; Hermoso, M.A. Chronic inflammation and cytokines in the tumor microenvironment. J. Immunol. Res. 2014, 2014, 149185. [Google Scholar] [CrossRef]
- Salomon, B.L.; Leclerc, M.; Tosello, J.; Ronin, E.; Piaggio, E.; Cohen, J.L. Tumor Necrosis Factor α and Regulatory T Cells in Oncoimmunology. Front. Immunol. 2018, 9, 444. [Google Scholar] [CrossRef]
- Jorgovanovic, D.; Song, M.; Wang, L.; Zhang, Y. Roles of IFN-γin tumor progression and regression: A review. Biomark. Res. 2020, 8, 49. [Google Scholar] [CrossRef]
- Duchartre, Y.; Kim, Y.M.; Kahn, M. The Wnt signaling pathway in cancer. Crit. Rev. Oncol./Hematol. 2016, 99, 141–149. [Google Scholar] [CrossRef]
- Wu, C.H.; Hsu, F.T.; Chao, T.L.; Lee, Y.H.; Kuo, Y.C. Revealing the suppressive role of protein kinase C delta and p38 mitogen-activated protein kinase (MAPK)/NF-κB axis associates with lenvatinib-inhibited progression in hepatocellular carcinoma in vitro and in vivo. Biomed. Pharmacother. 2022, 145, 112437. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Tan, A.H.; Li, J. Curcumin Represses Colorectal Cancer Cell Proliferation by Triggering Ferroptosis via PI3K/Akt/mTOR Signaling. Nutr. Cancer 2023, 75, 726–733. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Xu, T.; Chen, C. The critical roles of miR-21 in anti-cancer effects of curcumin. Ann. Transl. Med. 2015, 4, 330. [Google Scholar]
- Xiang, T.; Fang, Y.; Wang, S.X. Quercetin suppresses HeLa cells by blocking PI3K/Akt pathway. J. Huazhong Univ. Sci. Technol. (Med. Sci.) 2014, 34, 740–744. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Si, Y.; Wang, Z.; Wang, J.; Guo, Y.; Zhang, X. Quercetin inhibits the growth of human gastric cancer stem cells by inducing mitochondrial-dependent apoptosis through the inhibition of PI3K/Akt signaling. Int. J. Mol. Med. 2016, 38, 619–626. [Google Scholar] [CrossRef]
- Liu, Y.Z.; Wu, K.; Huang, J.; Liu, Y.; Wang, X.; Meng, Z.J.; Yuan, S.-X.; Wang, D.-X.; Luo, J.-Y.; Zuo, G.-W.; et al. The PTEN/PI3K/Akt and Wnt/β-catenin signaling pathways are involved in the inhibitory effect of resveratrol on human colon cancer cell proliferation. Int. J. Oncol. 2014, 45, 104–112. [Google Scholar] [CrossRef]
- Li, D.; Wang, G.; Jin, G.; Yao, K.; Zhao, Z.; Bie, L.; Guo, Y.; Li, N.; Deng, W.; Chen, X.; et al. Resveratrol suppresses colon cancer growth by targeting the AKT/STAT3 signaling pathway. Int. J. Mol. Med. 2019, 43, 630–640. [Google Scholar] [CrossRef]
- Wang, R.; Lu, X.; Yu, R. Lycopene Inhibits Epithelial–Mesenchymal Transition and Promotes Apoptosis in Oral Cancer via PI3K/AKT/m-TOR Signal Pathway. Drug Des. Dev. Ther. 2020, 14, 2461–2471. [Google Scholar] [CrossRef]
- Toden, S.; Okugawa, Y.; Jascur, T.; Wodarz, D.; Komarova, N.L.; Buhrmann, C.; Shakibaei, M.; Boland, C.R.; Goel, A. Curcumin mediates chemosensitization to 5-fluorouracil through miRNA-induced suppression of epithelialto-mesenchymal transition in chemoresistant colorectal cancer. Carcinogenesis 2014, 36, 355–367. [Google Scholar] [CrossRef]
- Han, M.; Song, Y.; Zhang, X. Quercetin suppresses the migration and invasion in human colon cancer caco-2 cells through regulating toll-like receptor 4/nuclear factor-kappa B pathway. Pharmacogn. Mag. 2016, 12, S237. [Google Scholar]
- Qi, W.J.; Sheng, W.S.; Peng, C.; Xiaodong, M.; Yao, T.Z. Investigating into anti-cancer potential of lycopene: Molecular targets. Biomed. Pharmacother. 2021, 138, 111546. [Google Scholar] [CrossRef] [PubMed]
- Soltanian, S.; Riahirad, H.; Pabarja, A.; Jafari, E.; Khandani, B.K. Effect of Cinnamic acid and FOLFOX in diminishing side population and downregulating cancer stem cell markers in colon cancer cell line HT-29. DARU. J. Pharm. Sci. 2018, 26, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Vallée, A.; Lecarpentier, Y.; Vallée, J.N. Curcumin: A therapeutic strategy in cancers by inhibiting the canonical WNT/β-catenin pathway. J. Exp. Clin. Cancer Res. 2019, 38, 323. [Google Scholar] [CrossRef] [PubMed]
- Buhrmann, C.; Shayan, P.; Brockmueller, A.; Shakibaei, M. Resveratrol suppresses cross-talk between colorectal cancer cells and stromal cells in multicellular tumor microenvironment: A bridge between in vitro and in vivo tumor microenvironment study. Molecules 2020, 25, 4292. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, X.Q.; Zhang, Q.; Zhu, J.Y.; Li, Y.; Xie, C.F.; Li, X.-T.; Wu, J.-S.; Geng, S.-S.; Zhong, C.-Y.; et al. (-)-Epigallocatechin-3-gallate inhibits colorectal cancer stem cells by suppressing Wnt/β-catenin pathway. Nutrients 2017, 9, 572. [Google Scholar] [CrossRef]
- Afzal, M.; Safer, A.M.; Menon, M. Green tea polyphenols and their potential role in health and disease. Inflammopharmacology 2015, 23, 151–161. [Google Scholar] [CrossRef]
- Oh, S.; Gwak, J.; Park, S.; Yang, C.S. Green tea polyphenol EGCG suppresses Wnt/β-catenin signaling by promoting GSK-3β- and PP2A-independent β-catenin phosphorylation/degradation. BioFactors 2014, 40, 586–595. [Google Scholar] [CrossRef]
- Cha, J.H.; Kim, W.K.; Ha, A.W.; Kim, M.H.; Chang, M.J. Anti-inflammatory effect of lycopene in SW480 human colorectal cancer cells. Nutr. Res. Pract. 2017, 11, 90. [Google Scholar] [CrossRef]
- Raina, K.; Agarwal, C.; Agarwal, R. Effect of silibinin in human colorectal cancer cells: Targeting the activation of NF-κB signaling. Mol. Carcinog. 2013, 52, 195–206. [Google Scholar] [CrossRef]
- Özgöçmen, M.; Bayram, D.; Armağan, İ.; Türel, G.Y.; Sevimli, M.; Şenol, N. Is Quercetin Beneficial for Colon Cancer? A Cell Culture Study, Using the Apoptosis Pathways. Anticancer Agents Med. Chem. 2021, 22, 193–200. [Google Scholar] [CrossRef]
- Rajagopal, C.; Lankadasari, M.B.; Aranjani, J.M.; Harikumar, K.B. Targeting oncogenic transcription factors by polyphenols: A novel approach for cancer therapy. Pharmacol. Res. 2018, 130, 273–291. [Google Scholar] [CrossRef] [PubMed]
- Qin, H.; Zhang, H.; Zhang, X.; Zhang, S.; Zhu, S.; Wang, H. Resveratrol attenuates radiation enteritis through the SIRT1/FOXO3a and PI3K/AKT signaling pathways. Biochem. Biophys. Res. Commun. 2021, 554, 199–205. [Google Scholar] [CrossRef]
- Ismail, N.I.; Othman, I.; Abas, F.; HLajis, N.; Naidu, R. Mechanism of Apoptosis Induced by Curcumin in Colorectal Cancer. Int. J. Mol. Sci. 2019, 20, 2454. [Google Scholar] [CrossRef]
- Lim, J.Y.; Wang, X.D. Mechanistic understanding of β-cryptoxanthin and lycopene in cancer prevention in animal models. Biochim. Biophys. Acta (BBA)-Mol. Cell Biol. Lipids 2020, 1865, 158652. [Google Scholar] [CrossRef] [PubMed]
- Urrego, D.; Tomczak, A.P.; Zahed, F.; Stühmer, W.; Pardo, L.A. Potassium channels in cell cycle and cell proliferation. Philos. Trans. R. Soc. B Biol. Sci. 2014, 369, 20130094. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; He, L.Y.; Chen, Y.; Wang, W.Y.; Zhao, X.H.; Wu, M.Y. Quercetin affects leptin and its receptor in human gastric cancer MGC-803 cells and JAK-STAT pathway. Chin. J. Cell Mol. Immunol. 2012, 28, 12–16. [Google Scholar]
- Cerezo-Guisado, M.I.; Zur, R.; Lorenzo, M.J.; Risco, A.; Martín-Serrano, M.A.; Alvarez-Barrientos, A.; Cuenda, A.; Centeno, F. Implication of Akt, ERK1/2 and alternative p38MAPK signalling pathways in human colon cancer cell apoptosis induced by green tea EGCG. Food Chem. Toxicol. 2015, 84, 125–132. [Google Scholar] [CrossRef]
- Weng, W.; Goel, A. Curcumin and colorectal cancer: An update and current perspective on this natural medicine. Semin. Cancer Biol. 2022, 80, 73–86. [Google Scholar] [CrossRef]
- Vemuri, S.K.; Banala, R.R.; Mukherjee, S.; Uppula, P.; GPV, S.; AV, G.R.; Malarvilli, T. Novel biosynthesized gold nanoparticles as anti-cancer agents against breast cancer: Synthesis, biological evaluation, molecular modelling studies. Mater. Sci. Eng. C 2019, 99, 417–429. [Google Scholar] [CrossRef]
- Mechchate, H.; Es-safi, I.; Conte, R.; Hano, C.; Amaghnouje, A.; Jawhari, F.Z.; Radouane, N.; Bencheikh, N.; Grafov, A.; Bousta, D. In Vivo and In Vitro Antidiabetic and Anti-Inflammatory Properties of Flax (Linum usitatissimum L.) Seed Polyphenols. Nutrients 2021, 13, 2759. [Google Scholar] [CrossRef]
- Samtiya, M.; Aluko, R.E.; Dhewa, T.; Moreno-Rojas, J.M. Potential Health Benefits of Plant Food-Derived Bioactive Components: An Overview. Foods 2021, 10, 839. [Google Scholar] [CrossRef] [PubMed]
- Zamora-Ros, R.; Cayssials, V.; Jenab, M.; Rothwell, J.A.; Fedirko, V.; Aleksandrova, K.; Tjønneland, A.; Kyrø, C.; Overvad, K.; Boutron-Ruault, M.C.; et al. Dietary intake of total polyphenol and polyphenol classes and the risk of colorectal cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort. Eur. J. Epidemiol. 2018, 33, 1063–1075. [Google Scholar] [CrossRef] [PubMed]
- Saini, R.K.; Keum, Y.S.; Daglia, M.; Rengasamy, K.R. Dietary carotenoids in cancer chemoprevention and chemotherapy: A review of emerging evidence. Pharmacol. Res. 2020, 157, 104830. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Hosokawa, M.; Kasajima, H.; Hatanaka, K.; Kudo, K.; Shimoyama, N.; Miyashita, K. Anticancer effects of fucoxanthin and fucoxanthinol on colorectal cancer cell lines and colorectal cancer tissues. Oncol. Lett. 2015, 10, 1463–1467. [Google Scholar] [CrossRef] [PubMed]
- Cui, K.; Wu, H.; Fan, J.; Zhang, L.; Li, H.; Guo, H.; Yang, R.; Li, Z. The Mixture of Ferulic Acid and P-Coumaric Acid Suppresses Colorectal Cancer through lncRNA 495810/PKM2 Mediated Aerobic Glycolysis. Int. J. Mol. Sci. 2022, 23, 12106. [Google Scholar] [CrossRef]
- Goncalves, P.; Araujo, J.; Pinho, M.J.; Martel, F. In Vitro Studies on the Inhibition of Colon Cancer by Butyrate and Polyphenolic Compounds. Nutr. Cancer 2011, 63, 282–294. [Google Scholar] [CrossRef]
- Mileo, A.M.; Nisticò, P.; Miccadei, S. Polyphenols: Immunomodulatory and Therapeutic Implication in Colorectal Cancer. Front. Immunol. 2019, 10, 729. [Google Scholar] [CrossRef]
- Bracci, L.; Fabbri, A.; Del Cornò, M.; Conti, L. Dietary Polyphenols: Promising Adjuvants for Colorectal Cancer Therapies. Cancers 2021, 13, 4499. [Google Scholar] [CrossRef]
- Lu, Y.; Wu, S.; Xiang, B.; Li, L.; Lin, Y. Curcumin attenuates oxaliplatin-induced liver injury and oxidative stress by activating the Nrf2 pathway. Drug Des. Dev. Ther. 2020, 14, 73–85. [Google Scholar] [CrossRef]
- Ouyang, M.; Luo, Z.; Zhang, W.; Zhu, D.; Lu, Y.; Wu, J.; Yao, X. Protective effect of curcumin against irinotecan-induced intestinal mucosal injury via attenuation of NF-κB activation, oxidative stress and endoplasmic reticulum stress. Int. J. Oncol. 2019, 54, 1376–1386. [Google Scholar] [CrossRef]
- Sabet, N.S.; Atashbar, S.; Khanlou, E.M.; Kahrizi, F.; Salimi, A. Curcumin Attenuates Bevacizumab-Induced Toxicity via Suppressing Oxidative Stress and Preventing Mitochondrial Dysfunction in Heart Mitochondria. Naunyn-Schmiedeberg's Arch. Pharmacol. 2020, 393, 1447–1457. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, J.; Wang, K.; Han, W.; Wang, X.; Gao, M.; Wang, Z.; Sun, Y.; Yan, H.; Zhang, H.; et al. Quercetin overcomes colon cancer cells resistance to chemotherapy by inhibiting solute carrier family 1, member 5 transporter. Eur. J. Pharmacol. 2020, 881, 173185. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.; Namkoong, K.; Shin, M.; Park, J.; Yang, E.; Ihm, J.; Thu, V.T.; Kim, H.K.; Han, J.; Ghaffar, S.; et al. Cardiovascular Protective Effects and Clinical Applications of Resveratrol. J. Med. Food 2017, 20, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Riba, A.; Deres, L.; Sumegi, B.; Toth, K.; Szabados, E.; Halmosi, R. Cardioprotective effect of resveratrol in a postinfarction heart failure model. Oxid. Med. Cell. Longev. 2017, 2017, 6819281. [Google Scholar] [CrossRef] [PubMed]
- Sarubbo, F.; Moranta, D.; Asensio, V.J.; Miralles, A.; Esteban, S. Effects of Resveratrol and Other Polyphenols on the Most Common Brain Age-Related Diseases. Curr. Med. Chem. 2017, 24, 4245–4266. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Liu, W.; Lu, F.; Kong, W.; Zhou, X.; Miao, P.; Lei, C.; Wang, Y. Resveratrol attenuates neurological deficit and neuroinflammation following intracerebral hemorrhage. Exp. Ther. Med. 2018, 15, 4131–4138. [Google Scholar] [CrossRef]
- Xiao, Q.; Zhu, W.; Feng, W.; Lee, S.S.; Leung, A.W.; Shen, J.; Gao, L.; Xu, C. A review of resveratrol as a potent chemoprotective and synergistic agent in cancer chemotherapy. Front. Pharmacol. 2019, 9, 1534. [Google Scholar] [CrossRef]
- Moutabian, H.; Majdaeen, M.; Ghahramani-Asl, R.; Yadollahi, M.; Gharepapagh, E.; Ataei, G.; Falahatpour, Z.; Bagheri, H.; Farhood, B. A systematic review of the therapeutic effects of resveratrol in combination with 5-fluorouracil during colorectal cancer treatment: With a special focus on the oxidant, apoptotic, and anti-inflammatory activities. Cancer Cell Int. 2022, 22, 142. [Google Scholar] [CrossRef]
- Santandreu, F.M.; Valle, A.; Oliver, J.; Roca, P. Resveratrol Potentiates the Cytotoxic Oxidative Stress Induced by Chemotherapy in Human Colon Cancer Cells. Cell. Physiol. Biochem. 2011, 28, 219–228. [Google Scholar] [CrossRef]
- Cocetta, V.; Quagliariello, V.; Fiorica, F.; Berretta, M.; Montopoli, M. Resveratrol as Chemosensitizer Agent: State of Art and Future Perspectives. Int. J. Mol. Sci. 2021, 22, 2049. [Google Scholar] [CrossRef]
- Liu, Y.; Tong, L.; Luo, Y.; Li, X.; Chen, G.; Wang, Y. Resveratrol inhibits the proliferation and induces the apoptosis in ovarian cancer cells via inhibiting glycolysis and targeting AMPK/mTOR signaling pathway. J. Cell. Biochem. 2018, 119, 6162–6172. [Google Scholar] [CrossRef]
- Wang, J.; Huang, P.; Pan, X.; Xia, C.; Zhang, H.; Zhao, H.; Yuan, Z.; Liu, J.; Meng, C.; Liu, F. Resveratrol reverses TGF-β1-mediated invasion and metastasis of breast cancer cells via the SIRT3/AMPK/autophagy signal axis. Phytother. Res. 2023, 37, 211–230. [Google Scholar] [CrossRef]
- Hardie, D.G. AMP-activated/SNF1 protein kinases: Conserved guardians of cellular energy. Nat. Rev. Mol. Cell Biol. 2007, 8, 774–785. [Google Scholar] [CrossRef]
- Xiao, K.; Jiang, J.; Guan, C.; Dong, C.; Wang, G.; Bai, L.; Sun, J.; Hu, C.; Bai, C. Curcumin Induces Autophagy via Activating the AMPK Signaling Pathway in Lung Adenocarcinoma Cells. J. Pharmacol. Sci. 2013, 123, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Shen, G.; Khor, T.O.; Kim, J.H.; Kong, A.N.T. Curcumin inhibits Akt/mammalian target of rapamycin signaling through protein phosphatase-dependent mechanism. Mol. Cancer Ther. 2008, 7, 2609–2620. [Google Scholar] [CrossRef] [PubMed]
- Holczer, M.; Besze, B.; Zámbó, V.; Csala, M.; Bánhegyi, G.; Kapuy, O. Epigallocatechin-3-Gallate (EGCG) Promotes Autophagy-Dependent Survival via Influencing the Balance of mTOR-AMPK Pathways upon Endoplasmic Reticulum Stress. Oxid. Med. Cell. Longev. 2018, 2018, 6721530. [Google Scholar] [CrossRef]
- Khan, K.; Quispe, C.; Javed, Z.; Iqbal, M.J.; Sadia, H.; Raza, S.; Irshad, A.; Salehi, B.; Reiner, Ž.; Sharifi-Rad, J. Resveratrol, curcumin, paclitaxel and miRNAs mediated regulation of PI3K/Akt/mTOR pathway: Go four better to treat bladder cancer. Cancer Cell Int. 2020, 20, 560. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.S.; Dutta, P.; Austin, D.; Wang, P.; Awad, A.; Vadgama, J.V. Combination of Resveratrol and 5-Flurouracil Enhanced Anti-Telomerase Activity and Apoptosis by Inhibiting STAT3 and Akt Signaling Pathways in Human Colorectal Cancer Cells. 2018. Available online: www.oncotarget.com (accessed on 28 June 2023).
- Ko, J.H.; Sethi, G.; Um, J.Y.; Shanmugam, M.K.; Arfuso, F.; Kumar, A.P.; Bishayee, A.; Ahn, K.S. The Role of Resveratrol in Cancer Therapy. Int. J. Mol. Sci. 2017, 18, 2589. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.U.; Nam, J.; Cha, M.D.; Kim, S.W. Inhibition of phosphodiesterase 4D decreases the malignant properties of DLD-1 colorectal cancer cells by repressing the AKT/mTOR/Myc signaling pathway. Oncol. Lett. 2019, 17, 3589–3598. [Google Scholar] [CrossRef]
- Celiberto, F.; Aloisio, A.; Girardi, B.; Pricci, M.; Iannone, A.; Russo, F.; Riezzo, G.; D’Attoma, B.; Ierardi, E.; Losurdo, G.; et al. Fibres and Colorectal Cancer: Clinical and Molecular Evidence. Int. J. Mol. Sci. 2023, 24, 13501. [Google Scholar] [CrossRef]
- Redondo-Blanco, S.; Fernández, J.; Gutiérrez-del-Río, I.; Villar, C.J.; Lombó, F. New Insights toward Colorectal Cancer Chemotherapy Using Natural Bioactive Compounds. Front. Pharmacol. 2017, 8, 109. [Google Scholar] [CrossRef]
- Kumazaki, M.; Noguchi, S.; Yasui, Y.; Iwasaki, J.; Shinohara, H.; Yamada, N.; Akao, Y. Anti-cancer effects of naturally occurring compounds through modulation of signal transduction and miRNA expression in human colon cancer cells. J. Nutr. Biochem. 2013, 24, 1849–1858. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Li, W.; Dong, F.; Sun, H.; Wu, B.; Tan, J.; Zou, W.; Zhou, D. KITLG is a novel target of miR-34c that is associated with the inhibition of growth and invasion in colorectal cancer cells. J. Cell. Mol. Med. 2014, 18, 2092–2102. [Google Scholar] [CrossRef] [PubMed]
- Kaminski, B.M.; Weigert, A.; Scherzberg, M.C.; Ley, S.; Gilbert, B.; Brecht, K.; Brüne, B.; Steinhilber, D.; Stein, J.; Ulrich-Rückert, S. Resveratrol-induced potentiation of the antitumor effects of oxaliplatin is accompanied by an altered cytokine profile of human monocyte-derived macrophages. Apoptosis 2014, 19, 1136–1147. [Google Scholar] [CrossRef]
- Chang, T.K.; Yin, T.C.; Su, W.C.; Tsai, H.L.; Huang, C.W.; Chen, Y.C.; Li, C.-C.; Chen, P.-J.; Ma, C.-J.; Chuang, K.-H.; et al. A Pilot Study of Silymarin as Supplementation to Reduce Toxicities in Metastatic Colorectal Cancer Patients Treated with First-Line FOLFIRI Plus Bevacizumab. Oncol. Res. 2020, 28, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Koczurkiewicz-Adamczyk, P.; Klaś, K.; Gunia-Krzyżak, A.; Piska, K.; Andrysiak, K.; Stępniewski, J.; Lasota, S.; Wójcik-Pszczoła, K.; Dulak, J.; Madeja, Z.; et al. Cinnamic Acid Derivatives as Cardioprotective Agents against Oxidative and Structural Damage Induced by Doxorubicin. Int. J. Mol. Sci. 2021, 22, 6217. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, M.; Yang, T.; Qin, L.; Hu, Y.; Zhao, D.; Wu, L.; Liu, T. Cinnamic Acid Ameliorates Nonalcoholic Fatty Liver Disease by Suppressing Hepatic Lipogenesis and Promoting Fatty Acid Oxidation. Evid.-Based Complement. Altern. Med. 2021, 2021, 9561613. [Google Scholar] [CrossRef]
- Chae, H.K.; Kim, W.; Kim, S.K. Phytochemicals of Cinnamomi Cortex: Cinnamic Acid, but not Cinnamaldehyde, Attenuates Oxaliplatin-Induced Cold and Mechanical Hypersensitivity in Rats. Nutrients 2019, 11, 432. [Google Scholar] [CrossRef]
- Lu, Y.; Zhang, R.; Zhang, X.; Zhang, B.; Yao, Q. Curcumin may reverse 5-fluorouracil resistance on colonic cancer cells by regulating TET1-NKD-Wnt signal pathway to inhibit the EMT progress. Biomed. Pharmacother. 2020, 129, 110381. [Google Scholar] [CrossRef]
- Howells, L.M.; Iwuji, C.O.O.; Irving, G.R.B.; Barber, S.; Walter, H.; Sidat, Z.; Griffin-Teall, N.; Singh, R.; Foreman, N.; Patel, S.R.; et al. Curcumin Combined with FOLFOX Chemotherapy is Safe and Tolerable in Patients with Metastatic Colorectal Cancer in a Randomized Phase IIa Trial. J. Nutr. 2019, 149, 1133–1139. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).