As We Drink and Breathe: Adverse Health Effects of Microcystins and Other Harmful Algal Bloom Toxins in the Liver, Gut, Lungs and Beyond
Abstract
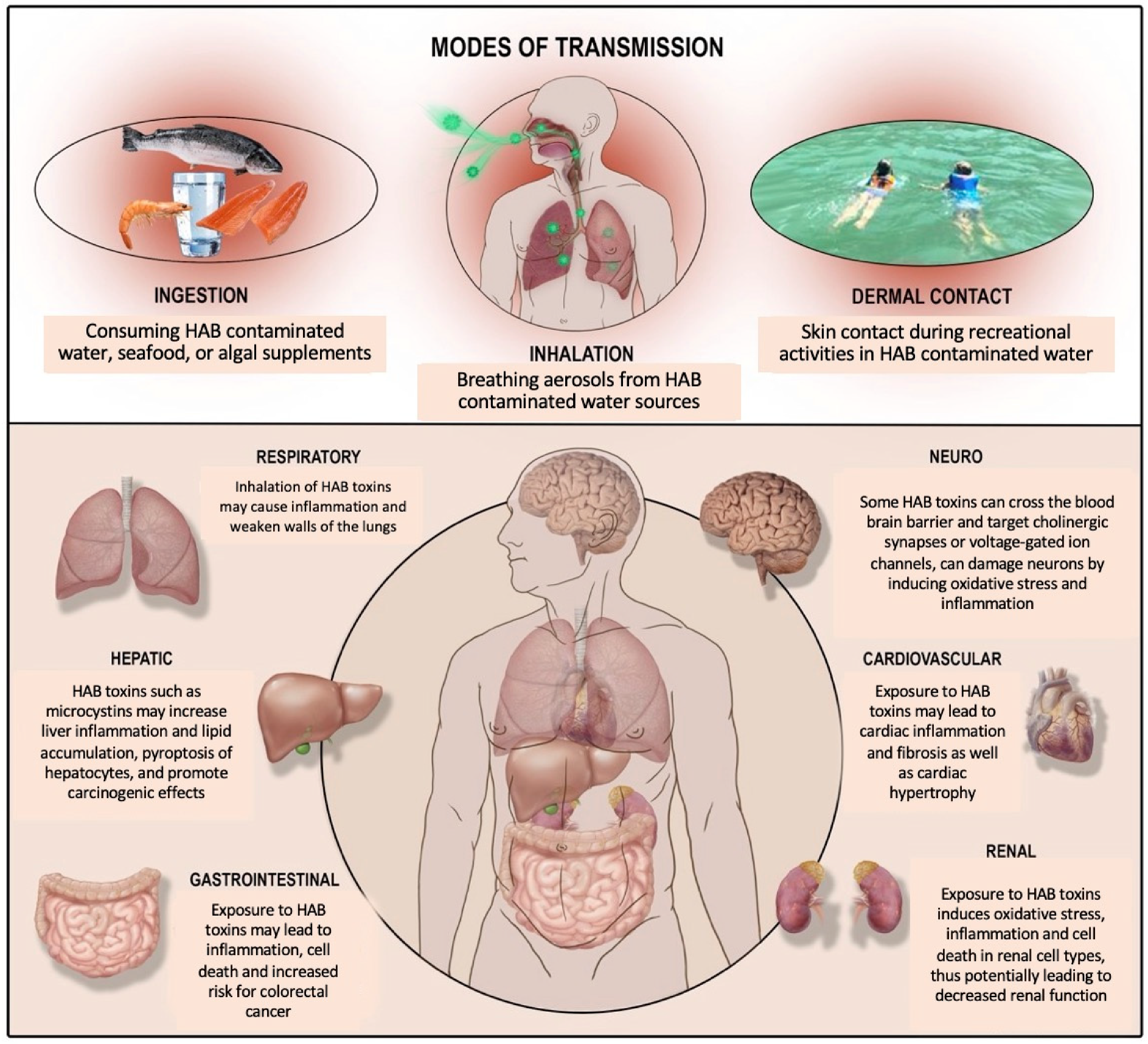
:1. Harmful Algal Blooms and Cyanotoxins
2. Adverse Health Effects of HAB Toxins across Major Organ Systems
3. Microcystins and Liver Health
4. Microcystins and Gut Health
5. Microcystins and Pulmonary Health
6. New Insights into Microcystin Detection
7. Corrective Measures
8. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- National Oceanic and Atmospheric Administration. Available online: https://www.noaa.gov/what-is-harmful-algal-bloom (accessed on 28 January 2022).
- Cox, J.; Mann, M. MaxQuant enables high peptide identification rates, individualized ppb-range mass accuracies and proteome-wide protein quantification. Nat. Biotechnol. 2008, 26, 1367. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J.R.; Wilhelm, S.W.; Boyer, G.L. The Fate of Microcystins in the Environment and Challenges for Monitoring. Toxins 2014, 6, 3354–3387. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davis, T.W.; Gobler, C.J. Preface for Special Issue on “Global expansion of harmful cyanobacterial blooms: Diversity, ecology, causes, and controls”. Harmful Algae 2016, 54, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Hudnell, H.K. The state of US freshwater harmful algal blooms assessments, policy and legislation. Toxicon 2010, 55, 1024–1034. [Google Scholar] [CrossRef] [PubMed]
- Huertas, M.J.; Mallén-Ponce, M.J. Dark side of cyanobacteria: Searching for strategies to blooms control. Microb. Biotechnol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Krausfeldt, L.E.; Farmer, A.T.; Castro, H.F.; Boyer, G.L.; Campagna, S.R.; Wilhelm, S.W. Nitrogen flux into metabolites and microcystins changes in response to different nitrogen sources in Microcystis aeruginosa NIES-843. Environ. Microbiol. 2020, 22, 2419–2431. [Google Scholar] [CrossRef]
- Chaffin, J.D.; Davis, T.W.; Smith, D.J.; Baer, M.M.; Dick, G.J. Interactions between nitrogen form, loading rate, and light intensity on Microcystis and Planktothrix growth and microcystin production. Harmful Algae 2018, 73, 84–97. [Google Scholar] [CrossRef]
- Ladds, M.; Jankowiak, J.; Gobler, C.J. Novel high throughput sequencing-fluorometric approach demonstrates Microcystis blooms across western Lake Erie are promoted by grazing resistance and nutrient enhanced growth. Harmful Algae 2021, 110, 102126. [Google Scholar] [CrossRef]
- Harke, M.J.; Steffen, M.M.; Gobler, C.J.; Otten, T.G.; Wilhelm, S.; Wood, S.A.; Paerl, H.W. A review of the global ecology, genomics, and biogeography of the toxic cyanobacterium, Microcystis spp. Harmful Algae 2016, 54, 4–20. [Google Scholar] [CrossRef] [Green Version]
- Ibelings, B.W.; Mur, L.R.; Walsby, A.E. Diurnal changes in buoyancy and vertical distribution in populations of Microcystis in two shallow lakes. J. Plankton Res. 1991, 13, 419–436. [Google Scholar] [CrossRef]
- Reynolds, C.S.; Rogers, D.A. Seasonal variations in the vertical distribution and buoyancy of Microcystis aeruginosa Kütz. emend. Elenkin in Rostherne Mere, England. Hydrobiologia 1976, 48, 17–23. [Google Scholar] [CrossRef]
- Jones, M.R.; Pinto, E.; Torres, M.A.; Dörr, F.; Mazur-Marzec, H.; Szubert, K.; Tartaglione, L.; Dell’Aversano, C.; Miles, C.O.; Beach, D.G.; et al. CyanoMetDB, a comprehensive public database of secondary metabolites from cyanobacteria. Water Res. 2021, 196, 117017. [Google Scholar] [CrossRef] [PubMed]
- Mallia, V.; Verhaegen, S.; Styrishave, B.; Eriksen, G.S.; Johannsen, M.L.; Ropstad, E.; Uhlig, S. Microcystins and Microcystis aeruginosa PCC7806 extracts modulate steroidogenesis differentially in the human H295R adrenal model. PLoS ONE 2020, 15, e0244000. [Google Scholar] [CrossRef] [PubMed]
- van Santen, J.A.; Poynton, E.F.; Iskakova, D.; McMann, E.; Alsup, T.A.; Clark, T.N.; Fergusson, C.H.; Fewer, D.P.; Hughes, A.H.; McCadden, C.A.; et al. The Natural Products Atlas 2.0: A database of microbially-derived natural products. Nucleic Acids Res. 2022, 50, D1317–D1323. [Google Scholar] [CrossRef]
- Wood, S.A.; Puddick, J.; Hawes, I.; Steiner, K.; Dietrich, D.R.; Hamilton, D.P. Variability in microcystin quotas during a Microcystis bloom in a eutrophic lake. Plos ONE 2021, 16, e0254967. [Google Scholar] [CrossRef]
- Falfushynska, H.; Horyn, O.; Osypenko, I.; Rzymski, P.; Wejnerowski, Ł.; Dziuba, M.K.; Sokolova, I.M. Multibiomarker-based assessment of toxicity of central European strains of filamentous cyanobacteria Aphanizomenon gracile and Raphidiopsis raciborskii to zebrafish Danio rerio. Water Res. 2021, 194, 116923. [Google Scholar] [CrossRef]
- Spoof, L.; Błaszczyk, A.; Meriluoto, J.; Cegłowska, M.; Mazur-Marzec, H. Structures and activity of new anabaenopeptins produced by Baltic Sea cyanobacteria. Mar. Drugs 2016, 14, 8. [Google Scholar] [CrossRef] [Green Version]
- Bittner, M.; Štern, A.; Smutná, M.; Hilscherová, K.; Žegura, B. Cytotoxic and Genotoxic Effects of Cyanobacterial and Algal Extracts—Microcystin and Retinoic Acid Content. Toxins 2021, 13, 107. [Google Scholar] [CrossRef]
- Arman, T.; Clarke, J. Microcystin Toxicokinetics, Molecular Toxicology, and Pathophysiology in Preclinical Rodent Models and Humans. Toxins 2021, 13, 537. [Google Scholar] [CrossRef]
- Baliu-Rodriguez, D.; Peraino, N.J.; Premathilaka, S.H.; Birbeck, J.A.; Baliu-Rodriguez, T.; Westrick, J.A.; Isailovic, D. Identification of Novel Microcystins Using High-Resolution MS and MS n with Python Code. Environ. Sci. Technol. 2022, 56, 1652–1663. [Google Scholar] [CrossRef]
- Altaner, S.; Puddick, J.; Wood, S.A.; Dietrich, D.R. Adsorption of Ten Microcystin Congeners to Common Laboratory-Ware Is Solvent and Surface Dependent. Toxins 2017, 9, 129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pearson, L.; Mihali, T.; Moffitt, M.; Kellmann, R.; Neilan, B. On the Chemistry, Toxicology and Genetics of the Cyanobacterial Toxins, Microcystin, Nodularin, Saxitoxin and Cylindrospermopsin. Mar. Drugs 2010, 8, 1650–1680. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patočka, J. The toxins of cyanobacteria. Acta Med. 2001, 44, 69–75. [Google Scholar] [CrossRef]
- Campos, A.; Vasconcelos, V. Molecular Mechanisms of Microcystin Toxicity in Animal Cells. Int. J. Mol. Sci. 2010, 11, 268–287. [Google Scholar] [CrossRef] [Green Version]
- Fischer, A.; Hoeger, S.; Stemmer, K.; Feurstein, D.; Knobeloch, D.; Nussler, A.; Dietrich, D. The role of organic anion transporting polypeptides (OATPs/SLCOs) in the toxicity of different microcystin congeners in vitro: A comparison of primary human hepatocytes and OATP-transfected HEK293 cells. Toxicol. Appl. Pharmacol. 2010, 245, 9–20. [Google Scholar] [CrossRef] [Green Version]
- Fischer, W.; Altheimer, S.; Cattori, V.; Meier, P.; Dietrich, D.; Hagenbuch, B. Organic anion transporting polypeptides expressed in liver and brain mediate uptake of microcystin. Toxicol. Appl. Pharmacol. 2005, 203, 257–263. [Google Scholar] [CrossRef] [Green Version]
- Eriksson, J.E.; Grönberg, L.; Nygård, S.; Slotte, J.; Meriluoto, J.A. Hepatocellular uptake of 3H-dihydromicrocystin-LR, a cyclic peptide toxin. Biochim. Biophys. Acta (BBA) Biomembr. 1990, 1025, 60–66. [Google Scholar] [CrossRef]
- Robinson, N.A.; Miura, G.A.; Matson, C.F.; Dinterman, R.E.; Pace, J.G. Characterization of chemically tritiated microcystin-LR and its distribution in mice. Toxicon 1989, 27, 1035–1042. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer. Ingested Nitrate and Nitrite, and Cyanobacterial Peptide Toxins. IARC Monographs on the Evaluation of Carcinogenic risks to Humans, 2010. No. 94. Available online: https://www.ncbi.nlm.nih.gov/books/NBK326544/ (accessed on 2 March 2022).
- Lad, A.; Su, R.C.; Breidenbach, J.D.; Stemmer, P.M.; Carruthers, N.J.; Sanchez, N.K.; Khalaf, F.K.; Zhang, S.; Kleinhenz, A.L.; Dube, P.; et al. Chronic Low Dose Oral Exposure to Microcystin-LR Exacerbates Hepatic Injury in a Murine Model of Non-Alcoholic Fatty Liver Disease. Toxins 2019, 11, 486. [Google Scholar] [CrossRef] [Green Version]
- Poniedziałek, B.; Rzymski, P.; Kokociński, M. Cylindrospermopsin: Water-linked potential threat to human health in Europe. Environ. Toxicol. Pharmacol. 2012, 34, 651–660. [Google Scholar] [CrossRef]
- Griffiths, D.J.; Saker, M.L. The Palm Island mystery disease 20 years on: A review of research on the cyanotoxin cylindrospermopsin. Environ. Toxicol. 2003, 18, 78–93. [Google Scholar] [CrossRef]
- Azevedo, S.M.F.O.; Carmichael, W.W.; Jochimsen, E.M.; Rinehart, K.L.; Lau, S.; Shaw, G.R.; Eaglesham, G.K. Human intoxication by microcystins during renal dialysis treatment in Caruaru—Brazil. Toxicology 2002, 181–182, 441–446. [Google Scholar] [CrossRef]
- Trevino-Garrison, I.; Dement, J.; Ahmed, F.S.; Haines-Lieber, P.; Langer, T.; Menager, H.; Neff, J.; Van Der Merwe, D.; Carney, E. Human Illnesses and Animal Deaths Associated with Freshwater Harmful Algal Blooms—Kansas. Toxins 2015, 7, 353–366. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jetoo, S.; Grover, V.I.; Krantzberg, G. The Toledo Drinking Water Advisory: Suggested Application of the Water Safety Planning Approach. Sustainability 2015, 7, 9787–9808. [Google Scholar] [CrossRef] [Green Version]
- Fawell, J.K.; Mitchell, R.E.; Everett, D.J.; Hill, R.E. The toxicity of cyanobacterial toxins in the mouse: I microcystin-LR. Hum. Exp. Toxicol. 1999, 18, 162–167. [Google Scholar] [CrossRef]
- Beversdorf, L.J.; Weirich, C.A.; Bartlett, S.L.; Miller, T. Variable cyanobacterial toxin and metabolite profiles across six eutrophic lakes of differing physio-chemical characteristics. Toxins 2017, 9, 62. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sedan, D.; Giannuzzi, L.; Rosso, L.; Marra, C.A.; Andrinolo, D. Biomarkers of prolonged exposure to microcystin-LR in mice. Toxicon 2013, 68, 9–17. [Google Scholar] [CrossRef]
- Sedan, D.; Laguens, M.; Copparoni, G.; Aranda, J.O.; Giannuzzi, L.; Marra, C.A.; Andrinolo, D. Hepatic and intestine alterations in mice after prolonged exposure to low oral doses of Microcystin-LR. Toxicon 2015, 104, 26–33. [Google Scholar] [CrossRef]
- Roberts, V.A.; Vigar, M.; Backer, L.; Veytsel, G.E.; Hilborn, E.D.; Hamelin, E.I.; Esschert, K.L.V.; Lively, J.Y.; Cope, J.R.; Hlavsa, M.C.; et al. Surveillance for harmful algal bloom events and associated human and animal illnesses—One health harmful algal bloom system, United States, 2016–2018. Morb. Mortal. Wkly. Rep. 2020, 69, 1889. [Google Scholar] [CrossRef]
- Roy-Lachapelle, A.; Solliec, M.; Bouchard, M.F.; Sauvé, S. Detection of Cyanotoxins in Algae Dietary Supplements. Toxins 2017, 9, 76. [Google Scholar] [CrossRef] [Green Version]
- Marsan, D.W.; Conrad, S.M.; Stutts, W.L.; Parker, C.H.; Deeds, J.R. Evaluation of microcystin contamination in blue-green algal dietary supplements using a protein phos-phatase inhibition-based test kit. Heliyon 2018, 4, e00573. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vichi, S.; Lavorini, P.; Funari, E.; Scardala, S.; Testai, E. Contamination by Microcystis and microcystins of blue–green algae food supplements (BGAS) on the italian market and possible risk for the exposed population. Food Chem. Toxicol. 2012, 50, 4493–4499. [Google Scholar] [CrossRef] [PubMed]
- Milutinović, A.; Sedmak, B.; Horvat-Znidarsic, I.; Suput, D. Renal injuries induced by chronic intoxication with microcystins. Cell. Mol. Biol. Lett. 2002, 7, 139–141. [Google Scholar] [PubMed]
- Lin, H.; Liu, W.; Zeng, H.; Pu, C.; Zhang, R.; Qiu, Z.; Chen, J.-A.; Wang, L.; Tan, Y.; Zheng, C.; et al. Determination of Environmental Exposure to Microcystin and Aflatoxin as a Risk for Renal Function Based on 5493 Rural People in Southwest China. Environ. Sci. Technol. 2016, 50, 5346–5356. [Google Scholar] [CrossRef]
- Lun, Z.; Hai, Y.; Kun, C. Relationship between microcystin in drinking water and colorectal cancer. Biomed. Environ. Sci. 2002, 15, 166–171. [Google Scholar]
- Botha, N.; van de Venter, M.; Downing, T.G.; Shephard, E.G.; Gehringer, M.M. The effect of intraperitoneally administered microcystin-LR on the gastrointestinal tract of Balb/c mice. Toxicon 2004, 43, 251–254. [Google Scholar] [CrossRef]
- Chorus, I.; Welker, M. Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management; Taylor & Francis: London, UK, 2021. [Google Scholar]
- Hachim, M.Y.; Hachim, I.Y.; Elemam, N.M.; Hamoudi, A.R. Toxicogenomic analysis of publicly available transcriptomic data can predict food, drugs, and chemical-induced asthma. Pharm. Pers. Med. 2019, 12, 181–199. [Google Scholar] [CrossRef] [Green Version]
- Martins, N.D.; Colvara, W.A.; Rantin, F.T.; Kalinin, A.L. Microcystin-LR: How it affects the cardio-respiratory responses to hypoxia in Nile tilapia, Oreochromis niloticus. Chemosphere 2011, 84, 154–159. [Google Scholar] [CrossRef]
- Qiu, T.; Xie, P.; Liu, Y.; Li, G.; Xiong, Q.; Hao, L.; Li, H. The profound effects of microcystin on cardiac antioxidant enzymes, mitochondrial function and cardiac toxicity in rat. Toxicology 2009, 257, 86–94. [Google Scholar] [CrossRef]
- Martin, R.M.; Bereman, M.S.; Marsden, K.C. Exposure to a mixture of BMAA and MCLR synergistically modulates behavior in larval zebrafish while exacerbating molecular changes related to neurodegeneration. bioRxiv 2020. [Google Scholar]
- Doyle, M.E. Toxins from Cyanobacteria. Ph.D. Thesis, University of Wisconsin, Madison, WI, USA, 2011; p. 9. Available online: https://fri.wisc.edu/files/Briefs_File/FRI_Brief_CyanobacteriaToxins_Aug2011.pdf (accessed on 28 January 2022).
- McLellan, N.L.; Manderville, R.A. Toxic mechanisms of microcystins in mammals. Toxicol. Res. 2017, 6, 391–405. [Google Scholar] [CrossRef] [Green Version]
- D’Anglada, L.V.; Strong, J. Health Effects Support Document for the Cyanobacterial Toxin Microcystins; D’Anglada, L.V., Ed.; U.S. Environmental Protection Agency: Washington, DC, USA, 2015; p. 138.
- Su, R.C.; Blomquist, T.M.; Kleinhenz, A.L.; Khalaf, F.K.; Dube, P.; Lad, A.; Breidenbach, J.D.; Mohammed, C.J.; Zhang, S.; Baum, C.E.; et al. Exposure to the Harmful Algal Bloom (HAB) Toxin Microcystin-LR (MC-LR) Prolongs and Increases Severity of Dextran Sulfate Sodium (DSS)-Induced Colitis. Toxins 2019, 11, 371. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Testai, E.; Scardala, S.; Vichi, S.; Buratti, F.M.; Funari, E. Risk to human health associated with the environmental occurrence of cyanobacterial neurotoxic alkaloids anatoxins and saxitoxins. Crit. Rev. Toxicol. 2016, 46, 385–419. [Google Scholar] [CrossRef] [PubMed]
- Pathmalal, M. Cyanotoxins: A Hidden Cause of Chronic Kidney Disease of Unknown Etiology (CKDu) in Sri Lanka-A Review; University of Sri Jayewardenepura: Gangodawila, Sri Lanka, 2019. [Google Scholar]
- Brown, G.T.; Kleiner, D.E. Histopathology of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Metabolism 2016, 65, 1080–1086. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cobbina, E.; Akhlaghi, F. Non-alcoholic fatty liver disease (NAFLD)–pathogenesis, classification, and effect on drug metabolizing enzymes and transporters. Drug Metab. Rev. 2017, 49, 197–211. [Google Scholar] [CrossRef] [PubMed]
- Le, M.; Devaki, P.; Ha, N.B.; Jun, D.W.; Te, H.S.; Cheung, R.C.; Nguyen, M.H. Prevalence of non-alcoholic fatty liver disease and risk factors for advanced fibrosis and mortality in the United States. PLoS ONE 2017, 12, e0173499. [Google Scholar] [CrossRef] [Green Version]
- Puri, P.; Sanyal, A.J. Nonalcoholic fatty liver disease: Definitions, risk factors, and workup. Clin. Liver Dis. 2012, 1, 99–103. [Google Scholar] [CrossRef]
- Fazel, Y.; Koenig, A.B.; Sayiner, M.; Goodman, Z.D.; Younossi, Z.M. Epidemiology and natural history of non-alcoholic fatty liver disease. Metabolism 2016, 65, 1017–1025. [Google Scholar] [CrossRef] [Green Version]
- Neuman, M.G.; Cohen, L.B.; Nanau, R.M. Biomarkers in nonalcoholic fatty liver disease. Can. J. Gastroenterol. Hepatol. 2014, 28, 607–618. [Google Scholar] [CrossRef]
- Clarke, J.D.; Dzierlenga, A.; Arman, T.; Toth, E.; Li, H.; Lynch, K.D.; Tian, D.-D.; Goedken, M.; Paine, M.F.; Cherrington, N. Nonalcoholic fatty liver disease alters microcystin-LR toxicokinetics and acute toxicity. Toxicon 2019, 162, 1–8. [Google Scholar] [CrossRef]
- Zhang, F.; Lee, J.; Liang, S.; Shum, C.K. Cyanobacteria blooms and non-alcoholic liver disease: Evidence from a county level ecological study in the United States. Environ. Health 2015, 14, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Albadrani, M.; Alhasson, F.; Dattaroy, D.; Chandrashekaran, V.; Seth, R.; Nagarkatti, M.; Nagarkatti, P.; Chatterjee, S. Micrcystin exposure Exacerbates Non-alcoholic Fatty Liver Disease (NAFLD) via NOX2 Dependent Activation of miR21-induced Inflammatory Pathways. Free Radic. Biol. Med. 2017, 112, 61. [Google Scholar] [CrossRef]
- Canet, M.J.; Hardwick, R.N.; Lake, A.D.; Dzierlenga, A.L.; Clarke, J.; Cherrington, N.J. Modeling Human Nonalcoholic Steatohepatitis-Associated Changes in Drug Transporter Expression Using Experimental Rodent Models. Drug Metab. Dispos. 2014, 42, 586–595. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arman, T.; Lynch, K.D.; Montonye, M.L.; Goedken, M.; Clarke, J.D. Sub-Chronic Microcystin-LR Liver Toxicity in Preexisting Diet-Induced Nonalcoholic Steatohepatitis in Rats. Toxins 2019, 11, 398. [Google Scholar] [CrossRef] [Green Version]
- Batista, T.; de Sousa, G.; Suput, J.S.; Rahmani, R.; Šuput, D. Microcystin-LR causes the collapse of actin filaments in primary human hepatocytes. Aquat. Toxicol. 2003, 65, 85–91. [Google Scholar] [CrossRef]
- He, J.; Li, G.; Chen, J.; Lin, J.; Zeng, C.; Chen, J.; Deng, J.; Xie, P. Prolonged exposure to low-dose microcystin induces nonalcholic steatohepatitis in mice: A systems toxicology study. Arch. Toxicol. 2017, 91, 465–480. [Google Scholar] [CrossRef]
- Díez-Quijada, L.; Hercog, K.; Štampar, M.; Filipič, M.; Cameán, A.M.; Jos, Á.; Žegura, B. Genotoxic Effects of Cylindrospermosin, Microcystin-LR and Their Binary Mixture in Human Hepatocellular Carcinoma (HepG2) Cell Line. Toxins 2020, 12, 778. [Google Scholar] [CrossRef]
- Arman, T.; Baron, J.A.; Lynch, K.D.; White, L.A.; Aldan, J.; Clarke, J.D. MCLR-elicited hepatic fibrosis and carcinogenic gene expression changes persist in rats with diet-induced nonalcoholic steatohepatitis through a 4-week recovery period. Toxicology 2021, 464, 153021. [Google Scholar] [CrossRef]
- Su, R.C.; Lad, A.; Breidenbach, J.D.; Kleinhenz, A.L.; Modyanov, N.; Malhotra, D.; Haller, S.T.; Kennedy, D.J. Assessment of diagnostic biomarkers of liver injury in the setting of microcystin-LR (MC-LR) hepatotoxicity. Chemosphere 2020, 257, 127111. [Google Scholar] [CrossRef]
- Su, R.C.; Lad, A.; Breidenbach, J.D.; Blomquist, T.M.; Gunning, W.T.; Dube, P.; Kleinhenz, A.L.; Malhotra, D.; Haller, S.T.; Kenedy, D.J. Hyperglycemia induces key genetic and phenotypic changes in human liver epithelial HepG2 cells which parallel the Leprdb/J mouse model of non-alcoholic fatty liver disease (NAFLD). PLoS ONE 2019, 14, e0225604. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.J.; Zhang, J.Y.; Hong, Y.; Chen, Y.X. Evaluation of organ distribution of microcystins in the freshwater phytoplantivorous fish Hypophthal-michthys molitrix. J. Zhejiang Univ. Sci. B 2007, 8, 116–120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gaudin, J.; Huet, S.; Jarry, G.; Fessard, V. In vivo DNA damage induced by the cyanotoxin microcystin-LR: Comparison of intraperitoneal and oral administrations by use of the comet assay. Mutat. Res. Toxicol. Environ. Mutagen. 2008, 652, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Greer, B.; Meneely, J.P.; Elliott, C.T. Uptake and accumulation of Microcystin-LR based on exposure through drinking water: An animal model assessing the human health risk. Sci. Rep. 2018, 8, 4913. [Google Scholar] [CrossRef] [Green Version]
- Ito, E.; Kondo, F.; Harada, K.-I. First report on the distribution of orally administered microcystin-LR in mouse tissue using an immunostaining method. Toxicon 1999, 38, 37–48. [Google Scholar] [CrossRef]
- Dahlhamer, J.M.; Zammitti, E.P.; Ward, B.W.; Wheaton, A.G.; Croft, J.B. Prevalence of inflammatory bowel disease among adults aged ≥ 18 years—United States, 2015. Morb. Mortal. Wkly. Rep. 2016, 65, 1166–1169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Molodecky, A.N.; Kaplan, G.G. Environmental risk factors for inflammatory bowel disease. Gastroenterol. Hepatol. 2010, 6, 339–346. [Google Scholar]
- Su, R.C.; Breidenbach, J.D.; Alganem, K.; Khalaf, F.K.; French, B.W.; Dube, P.; Malhotra, D.; McCullumsmith, R.; Presloid, J.B.; Wooten, R.M.; et al. Microcystin-LR (MC-LR) Triggers Inflammatory Responses in Macrophages. Int. J. Mol. Sci. 2021, 22, 9939. [Google Scholar] [CrossRef]
- Danese, S.; Sans, M.; Fiocchi, C. The CD40/CD40L costimulatory pathway in inflammatory bowel disease. Gut 2004, 53, 1035–1043. [Google Scholar] [CrossRef] [Green Version]
- Su, R.C.; Warner, E.A.; Breidenbach, J.D.; Lad, A.; Blomquist, T.M.; Kleinhenz, A.L.; Modyanov, N.; Malhotra, D.; Kennedy, D.J.; Haller, S.T. CD40 Receptor Knockout Protects against Microcystin-LR (MC-LR) Prolongation and Exacerbation of Dextran Sulfate Sodium (DSS)-Induced Colitis. Biomedicines 2020, 8, 149. [Google Scholar] [CrossRef]
- Su, R.C.; Meyers, C.M.; Warner, E.A.; Garcia, J.A.; Refsnider, J.M.; Lad, A.; Breidenbach, J.D.; Modyanov, N.; Malhotra, D.; Haller, S.T.; et al. Harmful Algal Bloom Toxicity in Lithobates catesbeiana Tadpoles. Toxins 2020, 12, 378. [Google Scholar] [CrossRef]
- Olson, N.E.; Cooke, M.; Shi, J.H.; Birbeck, J.A.; Westrick, J.A.; Ault, A.P. Harmful Algal Bloom Toxins in Aerosol Generated from Inland Lake Water. Environ. Sci. Technol. 2020, 54, 4769–4780. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.S.; Yue, Z.; Irvin, C.M.; Kirkpatrick, B.; Backer, L.C. Characterization of aerosols containing microcystin. Mar. Drugs 2007, 5, 136–150. [Google Scholar] [CrossRef] [PubMed]
- Backer, L.C.; McNeel, S.V.; Barber, T.; Kirkpatrick, B.; Williams, C.; Irvin, M.; Zhou, Y.; Johnson, T.B.; Nierenberg, K.; Aubel, M.; et al. Recreational exposure to microcystins during algal blooms in two California lakes. Toxicon 2010, 55, 909–921. [Google Scholar] [CrossRef] [PubMed]
- May, N.W.; Olson, N.E.; Panas, M.G.; Axson, J.L.; Tirella, P.S.; Kirpes, R.M.; Craig, R.L.; Gunsch, M.J.; China, S.; Laskin, A.; et al. Aerosol Emissions from Great Lakes Harmful Algal Blooms. Environ. Sci. Technol. 2018, 52, 397–405. [Google Scholar] [CrossRef]
- Giannuzzi, L.; Sedan, D.; Echenique, R.; Andrinolo, D. An Acute Case of Intoxication with Cyanobacteria and Cyanotoxins in Recreational Water in Salto Grande Dam, Argentina. Mar. Drugs 2011, 9, 2164–2175. [Google Scholar] [CrossRef] [Green Version]
- Schaefer, A.M.; Yrastorza, L.; Stockley, N.; Harvey, K.; Harris, N.; Grady, R.; Sullivan, J.; McFarland, M.; Reif, J.S. Exposure to microcystin among coastal residents during a cyanobacteria bloom in Florida. Harmful Algae 2020, 92, 101769. [Google Scholar] [CrossRef]
- Benson, J.M.; Hutt, J.A.; Rein, K.; Boggs, S.E.; Barr, E.B.; Fleming, L.E. The toxicity of microcystin LR in mice following 7 days of inhalation exposure. Toxicon 2005, 45, 691–698. [Google Scholar] [CrossRef] [Green Version]
- Ito, E.; Kondo, F.; Harada, K.-I. Intratracheal administration of microcystin-LR, and its distribution. Toxicon 2000, 39, 265–271. [Google Scholar] [CrossRef]
- Oliveira, V.R.; Mancin, V.G.; Pinto, E.F.; Soares, R.M.; Azevedo, S.M.; Macchione, M.; Carvalho, A.; Zin, W.A. Repeated intranasal exposure to microcystin-LR affects lungs but not nasal epithelium in mice. Toxicon 2015, 104, 14–18. [Google Scholar] [CrossRef]
- Picanço, M.; Soares, R.; Cagido, V.; Azevedo, S.; Rocco, P.; Zin, W. Toxicity of a cyanobacterial extract containing microcystins to mouse lungs. Braz. J. Med. Biol. Res. 2004, 37, 1225–1229. [Google Scholar] [CrossRef] [Green Version]
- Soares, R.M.; Cagido, V.R.; Ferraro, R.B.; Meyer-Fernandes, J.R.; Rocco, P.R.; Zin, W.A.; Azevedo, S.M. Effects of microcystin-LR on mouse lungs. Toxicon 2007, 50, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Stewart, I.; Webb, P.M.; Schluter, P.J.; Fleming, L.E.; Burns, J.W.; Gantar, M.; Backer, L.C.; Shaw, G.R. Epidemiology of recreational exposure to freshwater cyanobacteria–an international prospective cohort study. BMC Public Health 2006, 6, 93. [Google Scholar] [CrossRef] [Green Version]
- May, N.W.; Gunsch, M.J.; Olson, N.E.; Bondy, A.L.; Kirpes, R.M.; Bertman, S.B.; China, S.; Laskin, A.; Hopke, P.K.; Ault, A.P.; et al. Unexpected Contributions of Sea Spray and Lake Spray Aerosol to Inland Particulate Matter. Environ. Sci. Technol. Lett. 2018, 5, 405–412. [Google Scholar] [CrossRef]
- Oudra, B.; Loudiki, M.; Vasconcelos, V.; Sabour, B.; Sbiyyaa, B.; Oufdou, K.; Mezrioui, N. Detection and quantification of microcystins from cyanobacteria strains isolated from reservoirs and ponds in Morocco. Environ. Toxicol. 2002, 17, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, J.; Codd, G.A. Analysis of Cyanobacterial Toxins by Immunological Methods. Chem. Res. Toxicol. 2003, 16, 103–112. [Google Scholar] [CrossRef]
- Mountfort, D.O.; Holland, P.; Sprosen, J. Method for detecting classes of microcystins by combination of protein phos-phatase inhibition assay and ELISA: Comparison with LC-MS. Toxicon 2005, 45, 199–206. [Google Scholar] [CrossRef]
- Poon, K.F.; Lam MH, W.; Lam, P.K.; Wong, B.S. Determination of microcystins in cyanobacterial blooms by solid-phase microextraction-high-performance liquid chromatography. Environ. Toxicol. Chem. Int. J. 2001, 20, 1648–1655. [Google Scholar]
- Li, Y.-W.; Zhan, X.-J.; Xiang, L.; Deng, Z.-S.; Huang, B.-H.; Wen, H.-F.; Sun, T.-F.; Cai, Q.-Y.; Li, H.; Mo, C.-H. Analysis of Trace Microcystins in Vegetables Using Solid-Phase Extraction Followed by High Performance Liquid Chromatography Triple-Quadrupole Mass Spectrometry. J. Agric. Food Chem. 2014, 62, 11831–11839. [Google Scholar] [CrossRef]
- Palagama, D.S.W.; West, R.E., III; Isailovic, D. Improved solid-phase extraction protocol and sensitive quantification of six microcystins in water using an HPLC-orbitrap mass spectrometry system. Anal. Methods 2017, 9, 2021–2030. [Google Scholar] [CrossRef]
- Yu, H.; Clark, K.D.; Anderson, J.L. Rapid and sensitive analysis of microcystins using ionic liquid-based in situ dispersive liquid–liquid microextraction. J. Chromatogr. A 2015, 1406, 10–18. [Google Scholar] [CrossRef]
- Rapala, J.; Erkomaa, K.; Kukkonen, J.; Sivonen, K.; Lahti, K. Detection of microcystins with protein phosphatase inhibition assay, high-performance liquid chromatog-raphy–UV detection and enzyme-linked immunosorbent assay: Comparison of methods. Anal. Chim. Acta 2002, 466, 213–231. [Google Scholar] [CrossRef]
- Xu, L.; Lam, P.; Chen, J.; Xu, J.; Wong, B.; Zhang, Y.; Wu, R.; Harada, K. Use of protein phosphatase inhibition assay to detect microcystins in Donghu Lake and a fish pond in China. Chemosphere 2000, 41, 53–58. [Google Scholar] [CrossRef]
- Metcalf, J.S.; Bell, S.G.; Codd, G.A. Colorimetric Immuno-Protein Phosphatase Inhibition Assay for Specific Detection of Microcystins and Nodularins of Cyanobacteria. Appl. Environ. Microbiol. 2001, 67, 904–909. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bateman, K.P.; Thibault, P.; Douglas, D.J.; White, R.L. Mass spectral analyses of microcystins from toxic cyanobacteria using on-line chromatographic and elec-trophoretic separations. J. Chromatogr. A 1995, 712, 253–268. [Google Scholar] [CrossRef]
- Li, P.C.; Hu, S.; Lam, P.K. Development of a Capillary Zone Electrophoretic Method for the Rapid Separation and Detection of Hepatotoxic Microcystins. Mar. Pollut. Bull. 1999, 39, 250–254. [Google Scholar] [CrossRef]
- Palagama, D.S.; Baliu-Rodriguez, D.; Lad, A.; Levison, B.S.; Kennedy, D.J.; Haller, S.T.; Westrick, J.; Hensley, K.; Isailovic, D. Development and applications of solid-phase extraction and liquid chromatography-mass spectrometry methods for quantification of microcystins in urine, plasma, and serum. J. Chromatogr. A 2018, 1573, 66–77. [Google Scholar] [CrossRef]
- Baliu-Rodriguez, D.; Kucheriavaia, D.; Palagama, D.S.W.; Lad, A.; O’Neill, G.M.; Birbeck, J.A.; Kennedy, D.J.; Haller, S.T.; Westrick, J.A.; Isailovic, D. Development and Application of Extraction Methods for LC-MS Quantification of Microcystins in Liver Tissue. Toxins 2020, 12, 263. [Google Scholar] [CrossRef] [Green Version]
- Devasurendra, A.M.; Palagama, D.S.; Rohanifar, A.; Isailovic, D.; Kirchhoff, J.R.; Anderson, J.L. Solid-phase extraction, quantification, and selective determination of microcystins in water with a gold-polypyrrole nanocomposite sorbent material. J. Chromatogr. A 2018, 1560, 1–9. [Google Scholar] [CrossRef]
- Kucheriavaia, D.; Veličković, D.; Peraino, N.; Lad, A.; Kennedy, D.J.; Haller, S.T.; Westrick, J.A.; Isailovic, D. Toward Revealing Microcystin Distribution in Mouse Liver Tissue Using MALDI-MS Imaging. Toxins 2021, 13, 709. [Google Scholar] [CrossRef]
- Burford, M.A.; Gobler, C.J.; Hamilton, D.P.; Visser, P.M.; Lurling, M.; Codd, G.A. Solutions for Managing Cyanobacterial Blooms: A Scientific Summary for Policy Makers; UNESCO-IOC: Paris, France, 2019. [Google Scholar]
- Zuo, J.; Hu, L.; Shen, W.; Zeng, J.; Li, L.; Song, L.; Gan, N. The involvement of α-proteobacteria Phenylobacterium in maintaining the dominance of toxic Microcystis blooms in Lake Taihu, China. Environ. Microbiol. 2020, 23, 1066–1078. [Google Scholar] [CrossRef]
- Zhao, J.; Tian, F.; Zhai, Q.; Yu, R.; Zhang, H.; Gu, Z.; Chen, W. Protective effects of a cocktail of lactic acid bacteria on microcystin-LR-induced hepatotoxicity and oxidative damage in BALB/c mice. RSC Adv. 2017, 7, 20480–20487. [Google Scholar] [CrossRef] [Green Version]
- Nybom, S.M.; Salminen, S.J.; Meriluoto, J.A. Removal of microcystin-LR by strains of metabolically active probiotic bacteria. FEMS Microbiol. Lett. 2007, 270, 27–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nybom, S.M.; Salminen, S.J.; Meriluoto, J.A. Specific strains of probiotic bacteria are efficient in removal of several different cyanobacterial toxins from solution. Toxicon 2008, 52, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Nybom, S.; Dziga, D.; Heikkilä, J.; Kull, T.; Salminen, S.; Meriluoto, J. Characterization of microcystin-LR removal process in the presence of probiotic bacteria. Toxicon 2012, 59, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Nybom, S. Biodegradation of cyanobacterial toxins. Environmental Biotechnology-New Approaches and Prospective Applications; InTech: Rijeka, Croatia, 2013; pp. 147–170. [Google Scholar]
- Thees, A.; Atari, E.; Birbeck, J.; Westrick, J.A.; Huntley, J.F. Isolation and characterization of Lake Erie bacteria that degrade the cyanobacterial microcystin toxin MC-LR. J. Great Lakes Res. 2018, 45, 138–149. [Google Scholar] [CrossRef]
- McCartney, A.T.; Yeo, J.Y.; Blomquist, T.M.; Huntley, J.F. Whole-Genome Sequencing of Bacterial Isolates That Degrade the Cyanobacterial Toxin Microcys-tin-LR. Microbiol. Resour. Announc. 2020, 9, e00959-20. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lad, A.; Breidenbach, J.D.; Su, R.C.; Murray, J.; Kuang, R.; Mascarenhas, A.; Najjar, J.; Patel, S.; Hegde, P.; Youssef, M.; et al. As We Drink and Breathe: Adverse Health Effects of Microcystins and Other Harmful Algal Bloom Toxins in the Liver, Gut, Lungs and Beyond. Life 2022, 12, 418. https://doi.org/10.3390/life12030418
Lad A, Breidenbach JD, Su RC, Murray J, Kuang R, Mascarenhas A, Najjar J, Patel S, Hegde P, Youssef M, et al. As We Drink and Breathe: Adverse Health Effects of Microcystins and Other Harmful Algal Bloom Toxins in the Liver, Gut, Lungs and Beyond. Life. 2022; 12(3):418. https://doi.org/10.3390/life12030418
Chicago/Turabian StyleLad, Apurva, Joshua D. Breidenbach, Robin C. Su, Jordan Murray, Rebecca Kuang, Alison Mascarenhas, John Najjar, Shivani Patel, Prajwal Hegde, Mirella Youssef, and et al. 2022. "As We Drink and Breathe: Adverse Health Effects of Microcystins and Other Harmful Algal Bloom Toxins in the Liver, Gut, Lungs and Beyond" Life 12, no. 3: 418. https://doi.org/10.3390/life12030418
APA StyleLad, A., Breidenbach, J. D., Su, R. C., Murray, J., Kuang, R., Mascarenhas, A., Najjar, J., Patel, S., Hegde, P., Youssef, M., Breuler, J., Kleinhenz, A. L., Ault, A. P., Westrick, J. A., Modyanov, N. N., Kennedy, D. J., & Haller, S. T. (2022). As We Drink and Breathe: Adverse Health Effects of Microcystins and Other Harmful Algal Bloom Toxins in the Liver, Gut, Lungs and Beyond. Life, 12(3), 418. https://doi.org/10.3390/life12030418