MR Imaging in Real Time Guiding of Therapies in Prostate Cancer
Abstract
1. Introduction
2. The Role of MRI
2.1. Detection, Localization and Staging
2.2. Targeting
2.3. Real-Time Treatment Monitoring and MR Temperature Mapping
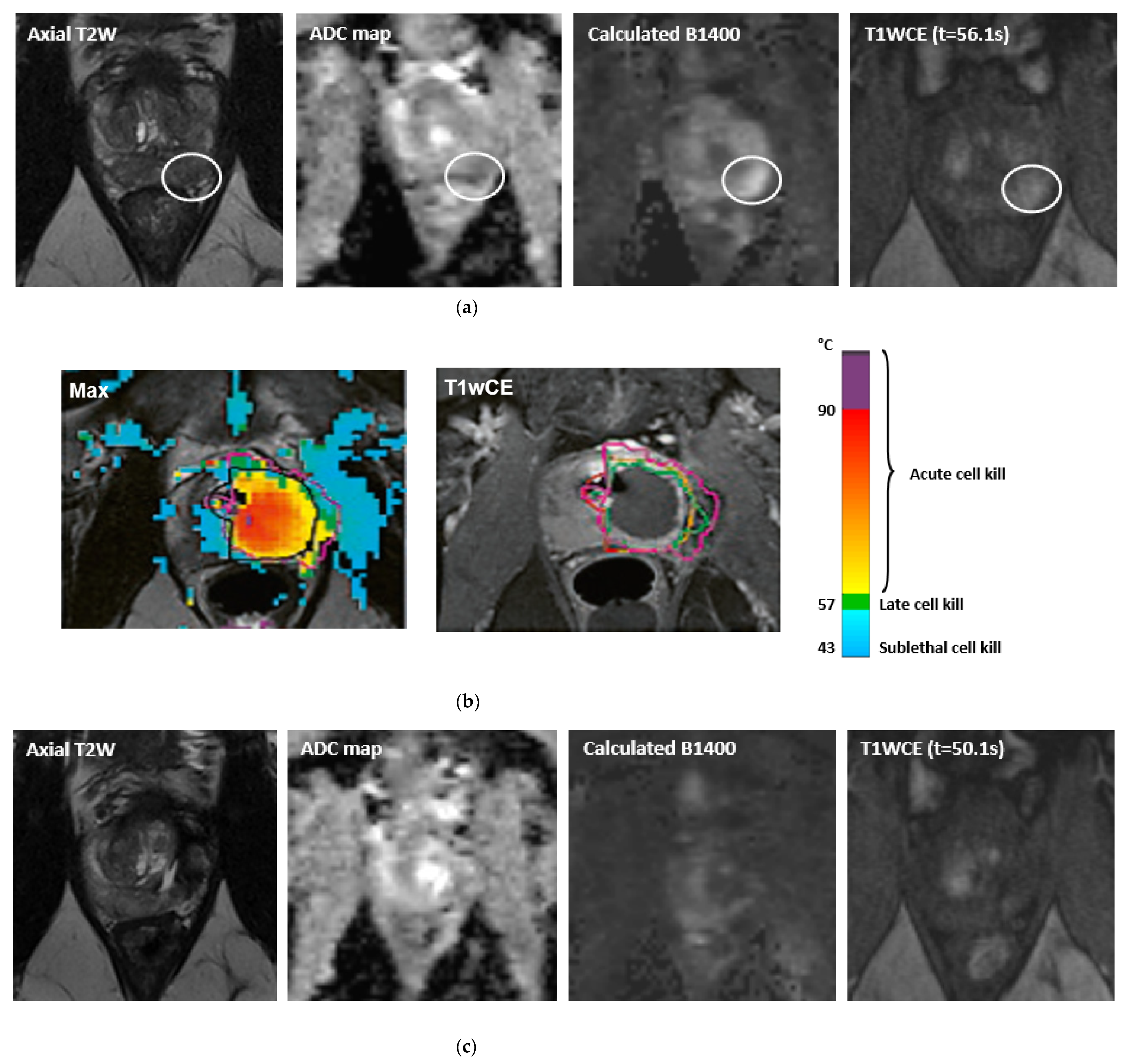
3. The Application of MRI in Real-Time Guided Therapy
3.1. Cryoablation
3.2. Focal Laser Ablation
3.3. High Intensity Focused Ultrasound
3.4. Transurethral Ultrasound Ablation
4. Clinical Follow-Up
5. Discussion and Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Rao, A.R.; Motiwala, H.G.; Karim, O.M. The discovery of prostate-specific antigen. Br. J. Urol. 2007, 101, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Howlader, N.; Noone, A.M.; Krapcho, M.; Miller, D.; Brest, A.; Yu, M.; Ruhl, J.; Tatalovich, Z.; Mariotto, A.; Lewis, D.R.; et al. (Eds.) SEER Cancer Statistics Review, 1975–2017; National Cancer Institute: Bethesda, MD, USA, 2020. Available online: https://seer.cancer.gov/archive/csr/1975_2017/results_merged/sect_23_prostate.pdf (accessed on 6 January 2022).
- Daskivich, T.J.; Chamie, K.; Kwan, L.; Labo, J.; Bs, R.P.; Dash, A.; Greenfield, S.; Litwin, M.S. Overtreatment of men with low-risk prostate cancer and significant comorbidity. Cancer 2010, 117, 2058–2066. [Google Scholar] [CrossRef] [PubMed]
- Ficarra, V.; Novara, G.; Rosen, R.C.; Artibani, W.; Carroll, P.R.; Costello, A.; Menon, M.; Montorsi, F.; Patel, V.R.; Stolzenburg, J.-U.; et al. Systematic Review and Meta-analysis of Studies Reporting Urinary Continence Recovery After Robot-assisted Radical Prostatectomy. Eur. Urol. 2012, 62, 405–417. [Google Scholar] [CrossRef] [PubMed]
- Gaither, T.W.; Awad, M.A.; Osterberg, E.C.; Murphy, G.P.; Allen, I.E.; Chang, A.; Rosen, R.C.; Breyer, B.N. The Natural History of Erectile Dysfunction After Prostatic Radiotherapy: A Systematic Review and Meta-Analysis. J. Sex. Med. 2017, 14, 1071–1078. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Zhao, S.; Liu, Y.; Wang, J.; Luo, L.; Li, E.; Zhang, C.; Luo, J.; Zhao, Z. Risk of secondary rectal cancer and colon cancer after radiotherapy for prostate cancer: A meta-analysis. Int. J. Color. Dis. 2018, 33, 1149–1158. [Google Scholar] [CrossRef] [PubMed]
- Wallis, C.J.D.; Mahar, A.; Choo, R.; Herschorn, S.; Kodama, R.T.; Shah, P.S.; Danjoux, C.; A Narod, S.; Nam, R.K. Second malignancies after radiotherapy for prostate cancer: Systematic review and meta-analysis. BMJ 2016, 352, i851. [Google Scholar] [CrossRef]
- Awad, M.A.; Gaither, T.W.; Osterberg, E.C.; Murphy, G.P.; Baradaran, N.; Breyer, B.N. Prostate cancer radiation and urethral strictures: A systematic review and meta-analysis. Prostate Cancer Prostatic Dis. 2018, 21, 168–174. [Google Scholar] [CrossRef]
- Lardas, M.; Liew, M.; Bergh, R.C.V.D.; De Santis, M.; Bellmunt, J.; Broeck, T.V.D.; Cornford, P.; Cumberbatch, M.G.; Fossati, N.; Gross, T.; et al. Quality of Life Outcomes after Primary Treatment for Clinically Localised Prostate Cancer: A Systematic Review. Eur. Urol. 2017, 72, 869–885. [Google Scholar] [CrossRef]
- Cooperberg, M.R.; Carroll, P.R. Trends in Management for Patients with Localized Prostate Cancer, 1990–2013. JAMA 2015, 314, 80–82. [Google Scholar] [CrossRef]
- Matta, R.; Hird, A.E.; Dvorani, E.; Saskin, R.; Nason, G.J.; Kulkarni, G.; Kodama, R.T.; Herschorn, S.; Nam, R.K. Rates of primary and secondary treatments for patients on active surveillance for localized prostate cancer—A population-based cohort study. Cancer Med. 2020, 9, 6946–6953. [Google Scholar] [CrossRef]
- Ahmed, H.U. The Index Lesion and the Origin of Prostate Cancer. N. Engl. J. Med. 2009, 361, 1704–1706. [Google Scholar] [CrossRef] [PubMed]
- Weinreb, J.C.; Barentsz, J.O.; Choyke, P.L.; Cornud, F.; Haider, M.A.; Macura, K.J.; Margolis, D.; Schnall, M.D.; Shtern, F.; Tempany, C.M.; et al. PI-RADS Prostate Imaging—Reporting and Data System: 2015, Version 2. Eur. Urol. 2016, 69, 16–40. [Google Scholar] [CrossRef] [PubMed]
- Wust, P.; Cho, C.H.; Hildebrandt, B.; Gellermann, J. Thermal monitoring: Invasive, minimal-invasive and non-invasive approaches. Int. J. Hyperth. 2006, 22, 255–262. [Google Scholar] [CrossRef]
- Staruch, R.; Chopra, R.; Hynynen, K. MRI-Controlled Ultrasound Thermal Therapy. IEEE Pulse 2011, 2, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, L.; Ahmed, H.U.; Allen, C.; Barentsz, J.O.; Carey, B.; Futterer, J.J.; Heijmink, S.W.; Hoskin, P.; Kirkham, A.; Padhani, A.; et al. Magnetic Resonance Imaging for the Detection, Localisation, and Characterisation of Prostate Cancer: Recommendations from a European Consensus Meeting. Eur. Urol. 2010, 59, 477–494. [Google Scholar] [CrossRef] [PubMed]
- Scheltema, M.J.; Tay, K.J.; Postema, A.W.; De Bruin, D.M.; Feller, J.; Futterer, J.J.; George, A.K.; Gupta, R.T.; Kahmann, F.; Kastner, C.; et al. Utilization of multiparametric prostate magnetic resonance imaging in clinical practice and focal therapy: Report from a Delphi consensus project. World J. Urol. 2016, 35, 695–701. [Google Scholar] [CrossRef]
- Muller, B.G.; Fütterer, J.J.; Gupta, R.T.; Katz, A.; Kirkham, A.; Kurhanewicz, J.; Moul, J.W.; Pinto, P.A.; Rastinehad, A.R.; Robertson, C.; et al. The role of magnetic resonance imaging (MRI) in focal therapy for prostate cancer: Recommendations from a consensus panel. Br. J. Urol. 2013, 113, 218–227. [Google Scholar] [CrossRef]
- Muller, B.G.; Bos, W.V.D.; Brausi, M.; Fütterer, J.J.; Ghai, S.; Pinto, P.A.; Popeneciu, I.V.; De Reijke, T.M.; Robertson, C.; De La Rosette, J.J.M.C.H.; et al. Follow-up modalities in focal therapy for prostate cancer: Results from a Delphi consensus project. World J. Urol. 2015, 33, 1503–1509. [Google Scholar] [CrossRef]
- Lebastchi, A.H.; George, A.K.; Polascik, T.J.; Coleman, J.; de la Rosette, J.; Turkbey, B.; Wood, B.J.; Gorin, M.A.; Sidana, A.; Ghai, S.; et al. Standardized Nomenclature and Surveillance Methodologies After Focal Therapy and Partial Gland Ablation for Localized Prostate Cancer: An International Multidisciplinary Consensus. Eur. Urol. 2020, 78, 371–378. [Google Scholar] [CrossRef]
- Zhen, L.; Liu, X.; Yegang, C.; Yongjiao, Y.; Yawei, X.; Jiaqi, K.; Xianhao, W.; Yuxuan, S.; Rui, H.; Wei, Z.; et al. Accuracy of multiparametric magnetic resonance imaging for diagnosing prostate Cancer: A systematic review and meta-analysis. BMC Cancer 2019, 19, 1244. [Google Scholar] [CrossRef]
- Hamoen, E.H.; de Rooij, M.; Witjes, J.A.; Barentsz, J.O.; Rovers, M. Use of the Prostate Imaging Reporting and Data System (PI-RADS) for Prostate Cancer Detection with Multiparametric Magnetic Resonance Imaging: A Diagnostic Meta-analysis. Eur. Urol. 2015, 67, 1112–1121. [Google Scholar] [CrossRef] [PubMed]
- Drost, F.-J.H.; Osses, D.F.; Nieboer, D.; Steyerberg, E.W.; Bangma, C.H.; Roobol, M.J.; Schoots, I.G. Prostate MRI, with or without MRI-targeted biopsy, and systematic biopsy for detecting prostate cancer. Cochrane Database Syst. Rev. 2019, 4, CD012663. [Google Scholar] [CrossRef] [PubMed]
- van der Leest, M.; Cornel, E.; Israël, B.; Hendriks, R.; Padhani, A.R.; Hoogenboom, M.; Zamecnik, P.; Bakker, D.; Setiasti, A.Y.; Veltman, J.; et al. Head-to-head Comparison of Transrectal Ultrasound-guided Prostate Biopsy Versus Multiparametric Prostate Resonance Imaging with Subsequent Magnetic Resonance-guided Biopsy in Biopsy-naïve Men with Elevated Prostate-specific Antigen: A Large Prospective Multicenter Clinical Study. Eur. Urol. 2019, 75, 570–578. [Google Scholar] [CrossRef] [PubMed]
- Kasivisvanathan, V.; Rannikko, A.S.; Borghi, M.; Panebianco, V.; Mynderse, L.A.; Vaarala, M.H.; Briganti, A.; Budäus, L.; Hellawell, G.; Hindley, R.G.; et al. MRI-Targeted or Standard Biopsy for Prostate-Cancer Diagnosis. N. Engl. J. Med. 2018, 378, 1767–1777. [Google Scholar] [CrossRef]
- Rouviere, O.; Puech, P.; Renard-Penna, R.; Claudon, M.; Roy, C.; Mège-Lechevallier, F.; Decaussin-Petrucci, M.; Dubreuil-Chambardel, M.; Magaud, L.; Remontet, L.; et al. Use of prostate systematic and targeted biopsy on the basis of multiparametric MRI in biopsy-naive patients (MRI-FIRST): A prospective, multicentre, paired diagnostic study. Lancet Oncol. 2019, 20, 100–109. [Google Scholar] [CrossRef]
- Ahmed, H.U.; El-Shater Bosaily, A.; Brown, L.C.; Gabe, R.; Kaplan, R.; Parmar, M.K.; Collaco-Moraes, Y.; Ward, K.; Hindley, R.G.; Freeman, A.; et al. Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): A paired validating confirmatory study. Lancet 2017, 389, 815–822. [Google Scholar] [CrossRef]
- Mottet, N.; van den Bergh, R.C.N.; Briers, E.; van den Broeck, T.; Cumberbatch, M.G.; De Santis, M.; Fanti, S.; Fossati, N.; Gandaglia, G.; Gillessen, S.; et al. EAU-EANM-ESTRO-ESUR-SIOG Guidelines on Prostate Cancer—2020 Update. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur. Urol. 2021, 79, 243–262. [Google Scholar] [CrossRef]
- Schieda, N.; Van Der Pol, C.B.; Walker, D.; Tsampalieros, A.K.; Maralani, P.J.; Woo, S.; Davenport, M.S. Adverse Events to the Gadolinium-based Contrast Agent Gadoxetic Acid: Systematic Review and Meta-Analysis. Radiology 2020, 297, 565–572. [Google Scholar] [CrossRef]
- Pesapane, F.; Acquasanta, M.; Di Meo, R.; Agazzi, G.M.; Tantrige, P.; Codari, M.; Schiaffino, S.; Patella, F.; Esseridou, A.; Sardanelli, F. Comparison of Sensitivity and Specificity of Biparametric versus Multiparametric Prostate MRI in the Detection of Prostate Cancer in 431 Men with Elevated Prostate-Specific Antigen Levels. Diagnostics 2021, 11, 1223. [Google Scholar] [CrossRef]
- Liang, Z.; Hu, R.; Yang, Y.; An, N.; Duo, X.; Liu, Z.; Shi, S.; Liu, X. Is dynamic contrast enhancement still necessary in multiparametric magnetic resonance for diagnosis of prostate cancer: A systematic review and meta-analysis. Transl. Androl. Urol. 2020, 9, 553–573. [Google Scholar] [CrossRef]
- Haghighi, M.; Shah, S.; Taneja, S.S.; Rosenkrantz, A. Prostate Cancer. J. Comput. Assist. Tomogr. 2013, 37, 980–988. [Google Scholar] [CrossRef]
- Hoeks, C.M.A.; Barentsz, J.O.; Hambrock, T.; Yakar, D.; Somford, D.M.; Heijmink, S.W.T.P.J.; Scheenen, T.W.J.; Vos, P.C.; Huisman, H.; Van Oort, I.M.; et al. Prostate Cancer: Multiparametric MR Imaging for Detection, Localization, and Staging. Radiology 2011, 261, 46–66. [Google Scholar] [CrossRef] [PubMed]
- Delongchamps, N.B.; Rouanne, M.; Flam, T.; Beuvon, F.; Liberatore, M.; Zerbib, M.; Cornud, F. Multiparametric magnetic resonance imaging for the detection and localization of prostate cancer: Combination of T2-weighted, dynamic contrast-enhanced and diffusion-weighted imaging. Br. J. Urol. 2010, 107, 1411–1418. [Google Scholar] [CrossRef] [PubMed]
- Haider, M.A.; Van der Kwast, T.; Tanguay, J.; Evans, A.J.; Hashmi, A.-T.; Lockwood, G.; Trachtenberg, J. Combined T2-Weighted and Diffusion-Weighted MRI for Localization of Prostate Cancer. Am. J. Roentgenol. 2007, 189, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Christie, D.R.; Sharpley, C.F. How accurately can multiparametric magnetic resonance imaging measure the tumour volume of a prostate cancer? Results of a systematic review. J. Med. Imaging Radiat. Oncol. 2020, 64, 398–407. [Google Scholar] [CrossRef] [PubMed]
- Eldred-Evans, D.; Tam, H.; Smith, A.P.T.; Winkler, M.; Ahmed, H.U. Use of Imaging to Optimise Prostate Cancer Tumour Volume Assessment for Focal Therapy Planning. Curr. Urol. Rep. 2020, 21, 38. [Google Scholar] [CrossRef] [PubMed]
- Renard-Penna, R.; Rouprêt, M.; Compérat, E.; Ayed, A.; Coudert, M.; Mozer, P.; Xylinas, E.; Bitker, M.-O.; Grenier, P. Accuracy of high resolution (1.5 tesla) pelvic phased array magnetic resonance imaging (MRI) in staging prostate cancer in candidates for radical prostatectomy: Results from a prospective study. Urol. Oncol. Semin. Orig. Investig. 2013, 31, 448–454. [Google Scholar] [CrossRef]
- Haffner, J.; Lemaitre, L.; Puech, P.; Haber, G.-P.; Leroy, X.; Jones, J.S.; Villers, A. Role of magnetic resonance imaging before initial biopsy: Comparison of magnetic resonance imaging-targeted and systematic biopsy for significant prostate cancer detection. Br. J. Urol. 2011, 108, E171–E178. [Google Scholar] [CrossRef]
- Kenigsberg, A.P.; Tamada, T.; Rosenkrantz, A.B.; Llukani, E.; Deng, F.-M.; Melamed, J.; Zhou, M.; Lepor, H. Multiparametric magnetic resonance imaging identifies significant apical prostate cancers. Br. J. Urol. 2017, 121, 239–243. [Google Scholar] [CrossRef]
- Goldberg, H.; Ahmad, A.E.; Chandrasekar, T.; Klotz, L.; Emberton, M.; Haider, M.A.; Taneja, S.; Arora, K.; Fleshner, N.; Finelli, A.; et al. Comparison of Magnetic Resonance Imaging and Transrectal Ultrasound Informed Prostate Biopsy for Prostate Cancer Diagnosis in Biopsy Naïve Men: A Systematic Review and Meta-Analysis. J. Urol. 2020, 203, 1085–1093. [Google Scholar] [CrossRef]
- Wegelin, O.; van Melick, H.H.; Hooft, L.; Bosch, J.R.; Reitsma, H.B.; Barentsz, J.O.; Somford, D.M. Comparing Three Different Techniques for Magnetic Resonance Imaging-targeted Prostate Biopsies: A Systematic Review of In-bore versus Magnetic Resonance Imaging-transrectal Ultrasound fusion versus Cognitive Registration. Is There a Preferred Technique? Eur. Urol. 2017, 71, 517–531. [Google Scholar] [CrossRef] [PubMed]
- Wegelin, O.; Exterkate, L.; van der Leest, M.; Kummer, J.A.; Vreuls, W.; de Bruin, P.C.; Bosch, J.; Barentsz, J.O.; Somford, D.M.; van Melick, H.H. The FUTURE Trial: A Multicenter Randomised Controlled Trial on Target Biopsy Techniques Based on Magnetic Resonance Imaging in the Diagnosis of Prostate Cancer in Patients with Prior Negative Biopsies. Eur. Urol. 2019, 75, 582–590. [Google Scholar] [CrossRef] [PubMed]
- Rieke, V.; Pauly, K.B. MR thermometry. J. Magn. Reson. Imaging 2008, 27, 376–390. [Google Scholar] [CrossRef]
- Blackwell, J.; Kraśny, M.J.; O’Brien, A.; Ashkan, K.; Galligan, J.; Destrade, M.; Colgan, N. Proton Resonance Frequency Shift Thermometry: A Review of Modern Clinical Practices. J. Magn. Reson. Imaging 2020, 55, 389–403. [Google Scholar] [CrossRef] [PubMed]
- Butts, K.; Sinclair, J.; Daniel, B.L.; Wansapura, J.; Pauly, J.M. Temperature quantitation and mapping of frozen tissue. J. Magn. Reson. Imaging 2001, 13, 99–104. [Google Scholar] [CrossRef]
- Wansapura, J.P.; Daniel, B.L.; Vigen, K.K.; Butts, K. In Vivo MR Thermometry of Frozen Tissue Using R2* and Signal Intensity1. Acad. Radiol. 2005, 12, 1080–1084. [Google Scholar] [CrossRef]
- Msc, C.G.O.; Fütterer, J.J.; Scheenen, T.W. 3D MR thermometry of frozen tissue: Feasibility and accuracy during cryoablation at 3T. J. Magn. Reson. Imaging 2016, 44, 1572–1579. [Google Scholar] [CrossRef]
- Overduin, C.; Bomers, J.G.R.; Jenniskens, S.F.M.; Hoes, M.F.; Haken, B.T.; De Lange, F.; Fütterer, J.J.; Scheenen, T.W.J. T1-weighted MR image contrast around a cryoablation iceball: A phantom study and initial comparison within vivofindings. Med. Phys. 2014, 41, 112301. [Google Scholar] [CrossRef]
- Ablin, R.J.; Gonder, M.J.; Soanes, W.A. Alterations of α2 -Globulin and the Clinical Response in Patients with Prostatic Cancer following Cryotherapy. Oncology 1975, 32, 127–144. [Google Scholar] [CrossRef]
- Cytron, S.; Greene, D.; Witzsch, U.; Nylund, P.; Johansen, T.E.B. Cryoablation of the prostate: Technical recommendations. Prostate Cancer Prostatic Dis. 2009, 12, 339–346. [Google Scholar] [CrossRef][Green Version]
- Bomers, J.G.R.; Yakar, D.; Overduin, C.G.; Sedelaar, J.P.M.; Vergunst, H.; Barentsz, J.O.; De Lange, F.; Fütterer, J.J. MR Imaging–guided Focal Cryoablation in Patients with Recurrent Prostate Cancer. Radiology 2013, 268, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Woodrum, D.A.; Kawashima, A.; Karnes, R.J.; Davis, B.; Frank, I.; Engen, D.E.; Gorny, K.R.; Felmlee, J.P.; Callstrom, M.R.; Mynderse, L.A. Magnetic Resonance Imaging-guided Cryoablation of Recurrent Prostate Cancer After Radical Prostatectomy: Initial Single Institution Experience. Urology 2013, 82, 870–875. [Google Scholar] [CrossRef] [PubMed]
- Kinsman, K.A.; White, M.L.; Mynderse, L.A.; Kawashima, A.; Rampton, K.; Gorny, K.R.; Atwell, T.D.; Felmlee, J.P.; Callstrom, M.R.; Woodrum, D.A. Whole-Gland Prostate Cancer Cryoablation with Magnetic Resonance Imaging Guidance: One-Year Follow-Up. Cardiovasc. Interv. Radiol. 2017, 41, 344–349. [Google Scholar] [CrossRef] [PubMed]
- De Marini, P.; Cazzato, R.L.; Garnon, J.; Tricard, T.; Koch, G.; Tsoumakidou, G.; Ramamurthy, N.; Lang, H.; Gangi, A.; De, M.P.; et al. Percutaneous MR-guided whole-gland prostate cancer cryoablation: Safety considerations and oncologic results in 30 consecutive patients. Br. J. Radiol. 2019, 92, 20180965. [Google Scholar] [CrossRef]
- Gangi, A.; Tsoumakidou, G.; Abdelli, O.; Buy, X.; de Mathelin, M.; Jacqmin, D.; Lang, H. Percutaneous MR-guided cryoablation of prostate cancer: Initial experience. Eur. Radiol. 2012, 22, 1829–1835. [Google Scholar] [CrossRef]
- Overduin, C.G.; Jenniskens, S.F.M.; Sedelaar, J.P.M.; Bomers, J.G.R.; Fütterer, J.J. Percutaneous MR-guided focal cryoablation for recurrent prostate cancer following radiation therapy: Retrospective analysis of iceball margins and outcomes. Eur. Radiol. 2017, 27, 4828–4836. [Google Scholar] [CrossRef]
- Bomers, J.G.; Overduin, C.G.; Jenniskens, S.F.; Cornel, E.B.; van Lin, E.N.; Sedelaar, J.M.; Fütterer, J.J. Focal Salvage MR Imaging–Guided Cryoablation for Localized Prostate Cancer Recurrence after Radiotherapy: 12-Month Follow-up. J. Vasc. Interv. Radiol. 2020, 31, 35–41. [Google Scholar] [CrossRef]
- Lomas, D.J.; Woodrum, D.A.; McLaren, R.H.; Gorny, K.R.; Felmlee, J.P.; Favazza, C.; Lu, A.; Mynderse, L.A. Rectal wall saline displacement for improved margin during MRI-guided cryoablation of primary and recurrent prostate cancer. Abdom. Radiol. 2019, 45, 1155–1161. [Google Scholar] [CrossRef]
- Garnon, J.; Cazzato, R.L.; Koch, G.; Uri, I.F.; Tsoumakidou, G.; Caudrelier, J.; Tricard, T.; Gangi, A.; Lang, H. Trans-rectal Ultrasound-Guided Autologous Blood Injection in the Interprostatorectal Space Prior to Percutaneous MRI-Guided Cryoablation of the Prostate. Cardiovasc. Interv. Radiol. 2017, 41, 653–659. [Google Scholar] [CrossRef]
- Raz, O.; Haider, M.A.; Davidson, S.R.; Lindner, U.; Hlasny, E.; Weersink, R.; Gertner, M.R.; Kucharcyzk, W.; McCluskey, S.A.; Trachtenberg, J. Real-Time Magnetic Resonance Imaging–Guided Focal Laser Therapy in Patients with Low-Risk Prostate Cancer. Eur. Urol. 2010, 58, 173–177. [Google Scholar] [CrossRef]
- Woodrum, D.A.; Mynderse, L.A.; Gorny, K.R.; Amrami, K.K.; McNichols, R.J.; Callstrom, M.R. 3.0T MR-Guided Laser Ablation of a Prostate Cancer Recurrence in the Postsurgical Prostate Bed. J. Vasc. Interv. Radiol. 2011, 22, 929–934. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.; Mendhiratta, N.; Sperling, D.; Lepor, H. Focal laser ablation for localized prostate cancer: Principles, clinical trials, and our initial experience. Rev. Urol. 2014, 16, 55–66. [Google Scholar] [PubMed]
- Bomers, J.G.R.; Cornel, E.B.; Fütterer, J.J.; Jenniskens, S.F.M.; Schaafsma, H.E.; Barentsz, J.O.; Sedelaar, J.P.M.; De Kaa, C.A.H.-V.; Witjes, J.A. MRI-guided focal laser ablation for prostate cancer followed by radical prostatectomy: Correlation of treatment effects with imaging. World J. Urol. 2016, 35, 703–711. [Google Scholar] [CrossRef] [PubMed]
- Barqawi, A.; Krughoff, K.; Li, H.; Patel, N.U. Initial Experience of Targeted Focal Interstitial Laser Ablation of Prostate Cancer with MRI Guidance. Curr. Urol. 2015, 8, 199–207. [Google Scholar] [CrossRef]
- Oto, A.; Sethi, I.; Karczmar, G.; McNichols, R.; Ivancevic, M.K.; Stadler, W.M.; Watson, S.; Eggener, S. MR Imaging–guided Focal Laser Ablation for Prostate Cancer: Phase I Trial. Radiology 2013, 267, 932–940. [Google Scholar] [CrossRef]
- Natarajan, S.; Raman, S.; Priester, A.M.; Garritano, J.; Margolis, D.J.; Lieu, P.; Macairan, M.L.; Huang, J.; Grundfest, W.; Marks, L.S. Focal Laser Ablation of Prostate Cancer: Phase I Clinical Trial. J. Urol. 2015, 196, 68–75. [Google Scholar] [CrossRef]
- Mehralivand, S.; George, A.K.; Hoang, A.N.; Rais-Bahrami, S.; Rastinehad, A.R.; Lebastchi, A.H.; Ahdoot, M.; Siddiqui, M.M.; Bloom, J.; Sidana, A.; et al. MRI-guided focal laser ablation of prostate cancer: A prospective single-arm, single-center trial with 3 years of follow-up. Diagn. Interv. Radiol. 2021, 27, 394–400. [Google Scholar] [CrossRef]
- Eggener, S.E.; Yousuf, A.; Watson, S.; Wang, S.; Oto, A. Phase II Evaluation of Magnetic Resonance Imaging Guided Focal Laser Ablation of Prostate Cancer. J. Urol. 2016, 196, 1670–1675. [Google Scholar] [CrossRef]
- Lepor, H.; Llukani, E.; Sperling, D.; Fütterer, J.J. Complications, Recovery, and Early Functional Outcomes and Oncologic Control Following In-bore Focal Laser Ablation of Prostate Cancer. Eur. Urol. 2015, 68, 924–926. [Google Scholar] [CrossRef]
- Chao, B.; Lepor, H. 5-Year Outcomes Following Focal Laser Ablation of Prostate Cancer. Urology 2021, 155, 124–129. [Google Scholar] [CrossRef]
- Walser, E.; Nance, A.; Ynalvez, L.; Yong, S.; Aoughsten, J.S.; Eyzaguirre, E.J.; Williams, S.B. Focal Laser Ablation of Prostate Cancer: Results in 120 Patients with Low- to Intermediate-Risk Disease. J. Vasc. Interv. Radiol. 2019, 30, 401–409.e2. [Google Scholar] [CrossRef] [PubMed]
- Al-Hakeem, Y.; Raz, O.; Gacs, Z.; MacLean, F.; Varol, C. Magnetic resonance image-guided focal laser ablation in clinically localized prostate cancer: Safety and efficacy. ANZ J. Surg. 2018, 89, 1610–1614. [Google Scholar] [CrossRef] [PubMed]
- Magee, D.; Perlis, N.; Corr, K.; Chan, R.; Gertner, M.; Zisman, A.; Jokhu, S.; Ghai, S. Salvage interstitial laser thermal therapy under MRI guidance (MRgFLA) for high-intensity focal ultrasound (HIFU) recurrences: Feasibility study. Clin. Imaging 2021, 76, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Al-Bataineh, O.; Jenne, J.; Huber, P. Clinical and future applications of high intensity focused ultrasound in cancer. Cancer Treat. Rev. 2012, 38, 346–353. [Google Scholar] [CrossRef]
- Azzouz, H.; de la Rosette, J. HIFU: Local Treatment of Prostate Cancer. EAU-EBU Updat. Ser. 2006, 4, 62–70. [Google Scholar] [CrossRef]
- Napoli, A.; Anzidei, M.; De Nunzio, C.; Cartocci, G.; Panebianco, V.; De Dominicis, C.; Catalano, C.; Petrucci, F.; Leonardo, C. Real-time Magnetic Resonance–guided High-intensity Focused Ultrasound Focal Therapy for Localised Prostate Cancer: Preliminary Experience. Eur. Urol. 2012, 63, 395–398. [Google Scholar] [CrossRef]
- Apfelbeck, M.; Clevert, D.-A.; Ricke, J.; Stief, C.; Schlenker, B. Contrast enhanced ultrasound (CEUS) with MRI image fusion for monitoring focal therapy of prostate cancer with high intensity focused ultrasound (HIFU)1. Clin. Hemorheol. Microcirc. 2018, 69, 93–100. [Google Scholar] [CrossRef]
- von Hardenberg, J.; Westhoff, N.; Baumunk, D.; Hausmann, D.; Martini, T.; Marx, A.; Porubsky, S.; Schostak, M.; Michel, M.S.; Ritter, M. Prostate cancer treatment by the latest focal HIFU device with MRI/TRUS-fusion control biopsies: A prospective evaluation. Urol. Oncol. Semin. Orig. Investig. 2018, 36, 401.e1–401.e9. [Google Scholar] [CrossRef]
- Lindner, U.; Ghai, S.; Spensieri, P.; Hlasny, E.; Van Der Kwast, T.H.; McCluskey, S.A.; Haider, M.A.; Kucharczyk, W.; Trachtenberg, J. Focal magnetic resonance guided focused ultrasound for prostate cancer: Initial North American experience. Can. Urol. Assoc. J. 2012, 6, 283–286. [Google Scholar] [CrossRef]
- Ghai, S.; Louis, A.S.; Van Vliet, M.; Lindner, U.; Haider, M.A.; Hlasny, E.; Spensieri, P.; Van Der Kwast, T.H.; McCluskey, S.A.; Kucharczyk, W.; et al. Real-Time MRI-Guided Focused Ultrasound for Focal Therapy of Locally Confined Low-Risk Prostate Cancer: Feasibility and Preliminary Outcomes. Am. J. Roentgenol. 2015, 205, W177–W184. [Google Scholar] [CrossRef]
- Yuh, B.; Liu, A.; Beatty, R.; Jung, A.; Wong, J.Y.C. Focal therapy using magnetic resonance image-guided focused ultrasound in patients with localized prostate cancer. J. Ther. Ultrasound 2016, 4, 8. [Google Scholar] [CrossRef]
- Ghai, S.; Perlis, N.; Lindner, U.; Hlasny, E.; Haider, M.A.; Finelli, A.; Zlotta, A.R.; Kulkarni, G.S.; Van Der Kwast, T.H.; McCluskey, S.A.; et al. Magnetic resonance guided focused high frequency ultrasound ablation for focal therapy in prostate cancer–phase 1 trial. Eur. Radiol. 2018, 28, 4281–4287. [Google Scholar] [CrossRef]
- Tay, K.J.; Cheng, C.W.S.; Lau, W.K.O.; Khoo, J.; Thng, C.H.; Kwek, J.W. Focal Therapy for Prostate Cancer with In-Bore MR–guided Focused Ultrasound: Two-Year Follow-up of a Phase I Trial—Complications and Functional Outcomes. Radiology 2017, 285, 620–628. [Google Scholar] [CrossRef]
- Ghai, S.; Finelli, A.; Corr, K.; Chan, R.; Jokhu, S.; Li, X.; McCluskey, S.; Konukhova, A.; Hlasny, E.; van der Kwast, T.H.; et al. MRI-guided Focused Ultrasound Ablation for Localized Intermediate-Risk Prostate Cancer: Early Results of a Phase II Trial. Radiology 2021, 298, 695–703. [Google Scholar] [CrossRef]
- Ramsay, E.; Mougenot, C.; Staruch, R.; Boyes, A.; Kazem, M.; Bronskill, M.; Foster, H.; Sugar, L.; Haider, M.; Klotz, L.; et al. Evaluation of Focal Ablation of Magnetic Resonance Imaging Defined Prostate Cancer Using Magnetic Resonance Imaging Controlled Transurethral Ultrasound Therapy with Prostatectomy as the Reference Standard. J. Urol. 2017, 197, 255–261. [Google Scholar] [CrossRef]
- Anttinen, M.; Yli-Pietilä, E.; Suomi, V.; Mäkelä, P.; Sainio, T.; Saunavaara, J.; Eklund, L.; Sequeiros, R.B.; Taimen, P.; Boström, P.J. Histopathological evaluation of prostate specimens after thermal ablation may be confounded by the presence of thermally-fixed cells. Int. J. Hyperth. 2019, 36, 914–924. [Google Scholar] [CrossRef]
- Anttinen, M.; Mäkelä, P.; Suomi, V.; Kiviniemi, A.; Saunavaara, J.; Sainio, T.; Horte, A.; Eklund, L.; Taimen, P.; Sequeiros, R.B.; et al. Feasibility of MRI-guided transurethral ultrasound for lesion-targeted ablation of prostate cancer. Scand. J. Urol. 2019, 53, 295–302. [Google Scholar] [CrossRef]
- Anttinen, M.; Mäkelä, P.; Viitala, A.; Nurminen, P.; Suomi, V.; Sainio, T.; Saunavaara, J.; Taimen, P.; Sequeiros, R.B.; Boström, P.J. Salvage Magnetic Resonance Imaging–guided Transurethral Ultrasound Ablation for Localized Radiorecurrent Prostate Cancer: 12-Month Functional and Oncological Results. Eur. Urol. Open Sci. 2020, 22, 79–87. [Google Scholar] [CrossRef]
- Mueller-wolf, M.; Röthke, M.; Hadaschik, B.; Pahernik, S.; Chin, J.; Relle, J.; Bonekamp, D. Transurethral MR-thermometry guided ultrasound ablation of the prostate–the heidelberg experience during phase I of the TULSA-PRO device trial. Magnetom Flash 2016, 66, 130–137. [Google Scholar]
- Chopra, R.; Burtnyk, M.; N’Djin, W.A.; Bronskill, M. MRI-controlled transurethral ultrasound therapy for localised prostate cancer. Int. J. Hyperth. 2010, 26, 804–821. [Google Scholar] [CrossRef]
- Galgano, S.J.; Planz, V.B.; Arora, S.; Rais-Bahrami, S. MR-Guided High-Intensity Directional Ultrasound Ablation of Prostate Cancer. Curr. Urol. Rep. 2021, 22, 3. [Google Scholar] [CrossRef]
- Chopra, R.; Colquhoun, A.; Burtnyk, M.; N’Djin, W.A.; Kobelevskiy, I.; Boyes, A.; Siddiqui, K.; Foster, H.; Sugar, L.; Haider, M.A.; et al. MR Imaging–controlled Transurethral Ultrasound Therapy for Conformal Treatment of Prostate Tissue: Initial Feasibility in Humans. Radiology 2012, 265, 303–313. [Google Scholar] [CrossRef]
- Chin, J.L.; Billia, M.; Relle, J.; Roethke, M.C.; Popeneciu, I.V.; Kuru, T.H.; Hatiboglu, G.; Mueller-Wolf, M.B.; Motsch, J.; Romagnoli, C.; et al. Magnetic Resonance Imaging–Guided Transurethral Ultrasound Ablation of Prostate Tissue in Patients with Localized Prostate Cancer: A Prospective Phase 1 Clinical Trial. Eur. Urol. 2016, 70, 447–455. [Google Scholar] [CrossRef]
- Nair, S.M.; Hatiboglu, G.; Relle, J.; Hetou, K.; Hafron, J.; Harle, C.; Kassam, Z.; Staruch, R.; Burtnyk, M.; Bonekamp, D.; et al. Magnetic resonance imaging-guided transurethral ultrasound ablation in patients with localised prostate cancer: 3-year outcomes of a prospective Phase I study. Br. J. Urol. 2020, 127, 544–552. [Google Scholar] [CrossRef]
- Klotz, L.; Pavlovich, C.P.; Chin, J.; Hatiboglu, G.; Koch, M.; Penson, D.; Raman, S.; Oto, A.; Fütterer, J.; Serrallach, M.; et al. Magnetic Resonance Imaging-Guided Transurethral Ultrasound Ablation of Prostate Cancer. J. Urol. 2021, 205, 769–779. [Google Scholar] [CrossRef]
- Anttinen, M.; Mäkelä, P.; Nurminen, P.; Yli-Pietilä, E.; Suomi, V.; Sainio, T.; Saunavaara, J.; Taimen, P.; Sequeiros, R.B.; Boström, P.J. Palliative MRI-guided transurethral ultrasound ablation for symptomatic locally advanced prostate cancer. Scand. J. Urol. 2020, 54, 481–486. [Google Scholar] [CrossRef]
- Kara, T.; Akata, D.; Akyol, F.; Karcaaltincaba, M.; Ozmen, M.N. The role of dynamic contrast enhanced MRI in the detection of recurrent prostate carcinoma after external beam radiotherapy-correlation with transrectal ultrasound and pathological findings. Diagn. Interv. Radiol. 2009, 17, 38–43. [Google Scholar] [CrossRef]
- Haider, M.A.; Chung, P.; Sweet, J.; Toi, A.; Jhaveri, K.; Ménard, C.; Warde, P.; Trachtenberg, J.; Lockwood, G.; Milosevic, M. Dynamic Contrast-Enhanced Magnetic Resonance Imaging for Localization of Recurrent Prostate Cancer After External Beam Radiotherapy. Int. J. Radiat. Oncol. 2007, 70, 425–430. [Google Scholar] [CrossRef]
- Roy, C.; Foudi, F.; Charton, J.; Jung, M.; Lang, H.; Saussine, C.; Jacqmin, D. Comparative Sensitivities of Functional MRI Sequences in Detection of Local Recurrence of Prostate Carcinoma After Radical Prostatectomy or External-Beam Radiotherapy. Am. J. Roentgenol. 2013, 200, W361–W368. [Google Scholar] [CrossRef]
- Cirillo, S.; Petracchini, M.; Scotti, L.; Gallo, T.; Macera, A.; Bona, M.C.; Ortega, C.; Gabriele, P.; Regge, D. Endorectal magnetic resonance imaging at 1.5 Tesla to assess local recurrence following radical prostatectomy using T2-weighted and contrast-enhanced imaging. Eur. Radiol. 2008, 19, 761–769. [Google Scholar] [CrossRef]
- Kitajima, K.; Hartman, R.P.; Froemming, A.T.; Hagen, C.E.; Takahashi, N.; Kawashima, A. Detection of Local Recurrence of Prostate Cancer After Radical Prostatectomy Using Endorectal Coil MRI at 3 T: Addition of DWI and Dynamic Contrast Enhancement to T2-Weighted MRI. Am. J. Roentgenol. 2015, 205, 807–816. [Google Scholar] [CrossRef] [PubMed]
- Casciani, E.; Polettini, E.; Carmenini, E.; Floriani, I.; Masselli, G.; Bertini, L.; Gualdi, G.F. Endorectal and Dynamic Contrast-Enhanced MRI for Detection of Local Recurrence After Radical Prostatectomy. Am. J. Roentgenol. 2008, 190, 1187–1192. [Google Scholar] [CrossRef]
- Lindner, U.; Weersink, R.; Haider, M.; Gertner, M.; Davidson, S.; Atri, M.; Wilson, B.; Fenster, A.; Trachtenberg, J. Image Guided Photothermal Focal Therapy for Localized Prostate Cancer: Phase I Trial. J. Urol. 2009, 182, 1371–1377. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, S.; Jones, T.A.; Priester, A.M.; Geoghegan, R.; Lieu, P.; Delfin, M.; Felker, E.; Margolis, D.J.; Sisk, A.; Pantuck, A.; et al. Focal Laser Ablation of Prostate Cancer: Feasibility of Magnetic Resonance Imaging-Ultrasound Fusion for Guidance. J. Urol. 2017, 198, 839–847. [Google Scholar] [CrossRef] [PubMed]
- Valerio, M.; Shah, T.T.; Shah, P.; Mccartan, N.; Emberton, M.; Arya, M.; Ahmed, H.U. Magnetic resonance imaging-transrectal ultrasound fusion focal cryotherapy of the prostate: A prospective development study. Urol. Oncol. Semin. Orig. Investig. 2017, 35, 150.e1–150.e7. [Google Scholar] [CrossRef] [PubMed]
- Rosenhammer, B.; Niessen, C.; Rotzinger, L.; Reiss, J.; Schnabel, M.J.; Burger, M.; Bründl, J. Oncological Outcome and Value of Postoperative Magnetic Resonance Imaging after Focal High-Intensity Focused Ultrasound Therapy for Prostate Cancer. Urol. Int. 2019, 103, 270–278. [Google Scholar] [CrossRef]
- Mortezavi, A.; Krauter, J.; Gu, A.; Sonderer, J.; Bruhin, J.; Reeve, K.A.; Held, L.; Donati, O.F.; Rupp, N.J.; Moch, H.; et al. Extensive Histological Sampling following Focal Therapy of Clinically Significant Prostate Cancer with High Intensity Focused Ultrasound. J. Urol. 2019, 202, 717–724. [Google Scholar] [CrossRef]
- Apfelbeck, M.; Chaloupka, M.; Schlenker, B.; Stief, C.; Clevert, D.-A. Follow-up after focal therapy of the prostate with high intensity focused ultrasound (HIFU) using contrast enhanced ultrasound (CEUS) in combination with MRI image fusion. Clin. Hemorheol. Microcirc. 2019, 73, 135–143. [Google Scholar] [CrossRef]
- Tokuda, J.; Wang, Q.; Tuncali, K.; Seethamraju, R.T.; Tempany, C.M.; Schmidt, E.J. Temperature-Sensitive Frozen-Tissue Imaging for Cryoablation Monitoring Using STIR-UTE MRI. Investig. Radiol. 2020, 55, 310–317. [Google Scholar] [CrossRef]
- Abreu, A.L.D.C.; Ma, Y.; Shoji, S.; Marien, A.; Leslie, S.; Gill, I.; Ukimura, O. Denonvilliers’ space expansion by transperineal injection of hydrogel: Implications for focal therapy of prostate cancer. Int. J. Urol. 2013, 21, 416–418. [Google Scholar] [CrossRef]
- Atri, M.; Gertner, M.R.; Haider, M.A.; Weersink, R.A.; Trachtenberg, J. Contrast-enhanced ultrasonography for real-time monitoring of interstitial laser thermal therapy in the focal treatment of prostate cancer. Can. Urol. Assoc. J. 2013, 3, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Bomers, J.G.R.; Bosboom, D.G.H.; Tigelaar, G.H.; Sabisch, J.; Fütterer, J.J.; Yakar, D. Feasibility of a 2nd generation MR-compatible manipulator for transrectal prostate biopsy guidance. Eur. Radiol. 2016, 27, 1776–1782. [Google Scholar] [CrossRef]
- Schouten, M.G.; Ansems, J.; Renema, W.K.J.; Bosboom, D.; Scheenen, T.W.J.; Fütterer, J.J. The accuracy and safety aspects of a novel robotic needle guide manipulator to perform transrectal prostate biopsies. Med. Phys. 2010, 37, 4744–4750. [Google Scholar] [CrossRef]
- Schouten, M.G.; Bomers, J.G.R.; Yakar, D.; Huisman, H.; Rothgang, E.; Bosboom, D.; Scheenen, T.W.J.; Misra, S.; Fütterer, J.J. Evaluation of a robotic technique for transrectal MRI-guided prostate biopsies. Eur. Radiol. 2011, 22, 476–483. [Google Scholar] [CrossRef] [PubMed]
- Armato, S.G.; Huisman, H.; Drukker, K.; Hadjiiski, L.; Kirby, J.; Petrick, N.; Redmond, G.; Giger, M.L.; Cha, K.; Mamonov, A.; et al. PROSTATEx Challenges for computerized classification of prostate lesions from multiparametric magnetic resonance images. J. Med. Imaging 2018, 5, 044501. [Google Scholar] [CrossRef] [PubMed]
- Ciris, P.A.; Chiou, J.-Y.G.; Glazer, D.I.; Chao, T.-C.; Tempany-Afdhal, C.M.; Madore, B.; Maier, S.E. Accelerated Segmented Diffusion-Weighted Prostate Imaging for Higher Resolution, Higher Geometric Fidelity, and Multi-b Perfusion Estimation. Investig. Radiol. 2019, 54, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Czarniecki, M.; Caglic, I.; Grist, J.T.; Gill, A.B.; Lorenc, K.; Slough, R.A.; Priest, A.N.; Barrett, T. Role of PROPELLER-DWI of the prostate in reducing distortion and artefact from total hip replacement metalwork. Eur. J. Radiol. 2018, 102, 213–219. [Google Scholar] [CrossRef]
- Winkel, D.J.; Heye, T.J.; Benz, M.R.; Glessgen, C.G.; Wetterauer, C.; Bubendorf, L.; Block, T.K.; Boll, D.T. Compressed Sensing Radial Sampling MRI of Prostate Perfusion: Utility for Detection of Prostate Cancer. Radiology 2019, 290, 702–708. [Google Scholar] [CrossRef]
- Liu, W.; Turkbey, B.; Sénégas, J.; Remmele, S.; Xu, S.; Kruecker, J.; Bernardo, M.; Wood, B.J.; Pinto, P.A.; Choyke, P.L. AcceleratedT2mapping for characterization of prostate cancer. Magn. Reson. Med. 2011, 65, 1400–1406. [Google Scholar] [CrossRef]
- Yu, A.C.; Badve, C.; Ponsky, L.E.; Pahwa, S.; Dastmalchian, S.; Rogers, M.; Jiang, Y.; Margevicius, S.; Schluchter, M.; Tabayoyong, W.; et al. Development of a Combined MR Fingerprinting and Diffusion Examination for Prostate Cancer. Radiology 2017, 283, 729–738. [Google Scholar] [CrossRef]
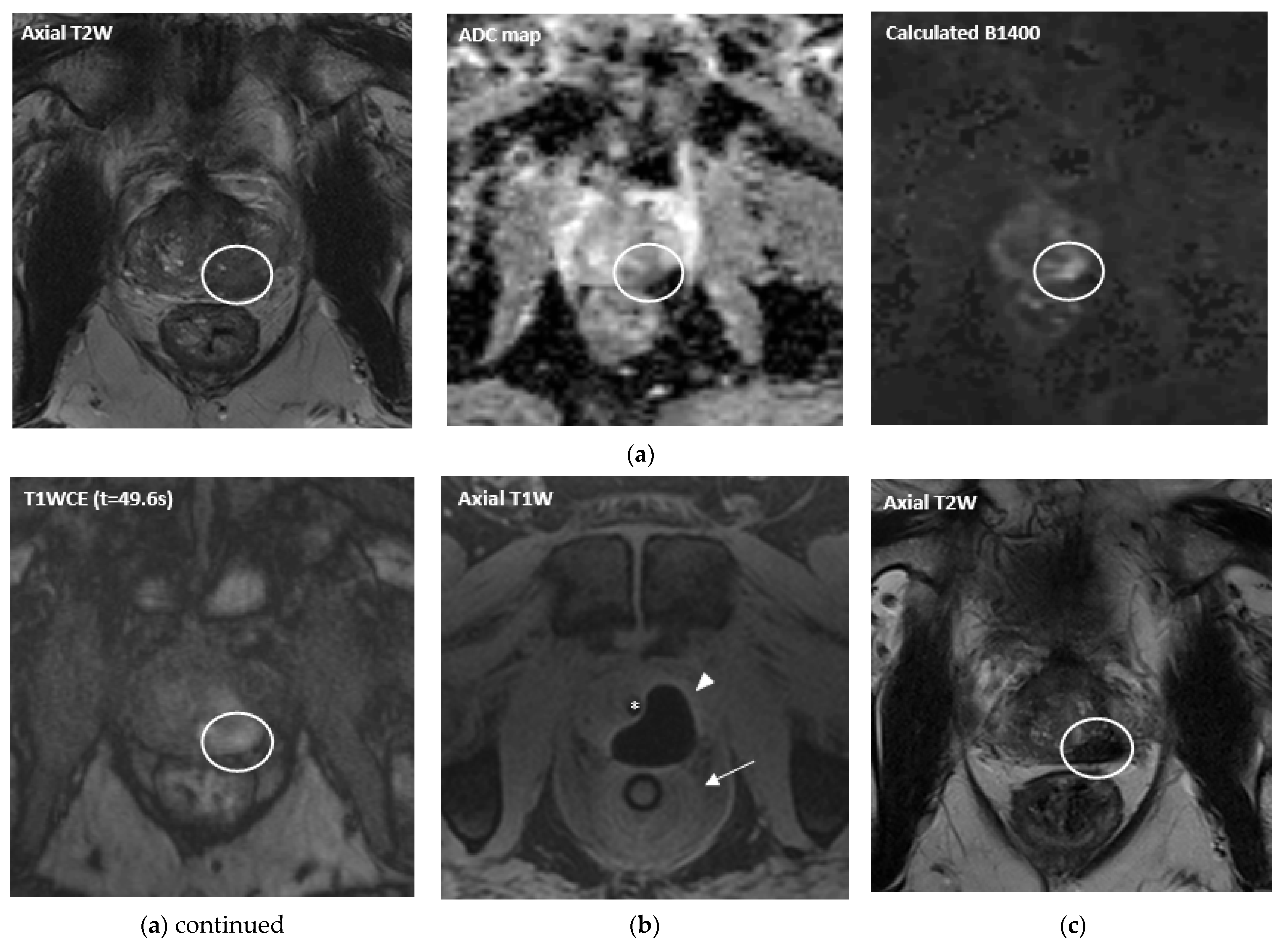

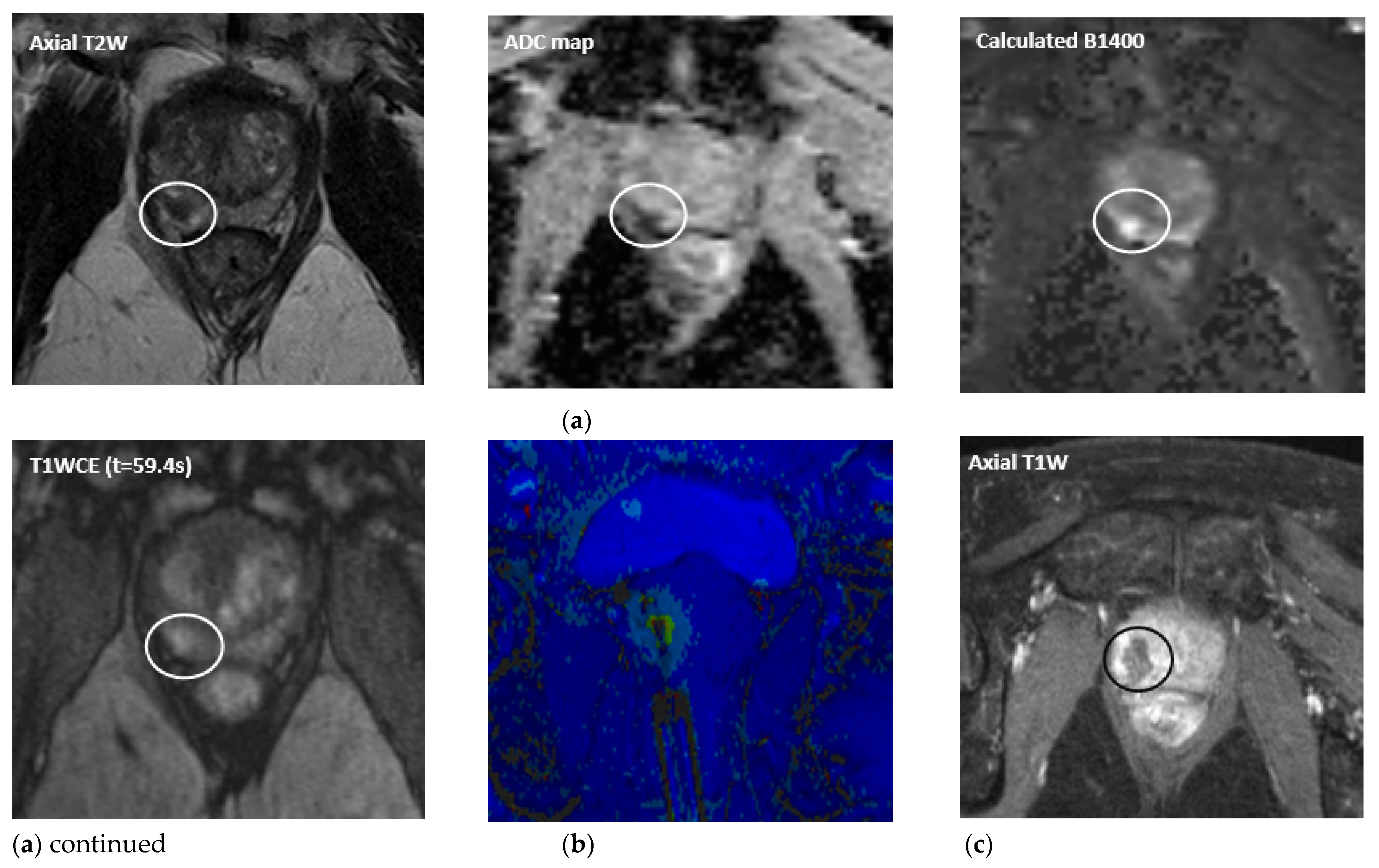
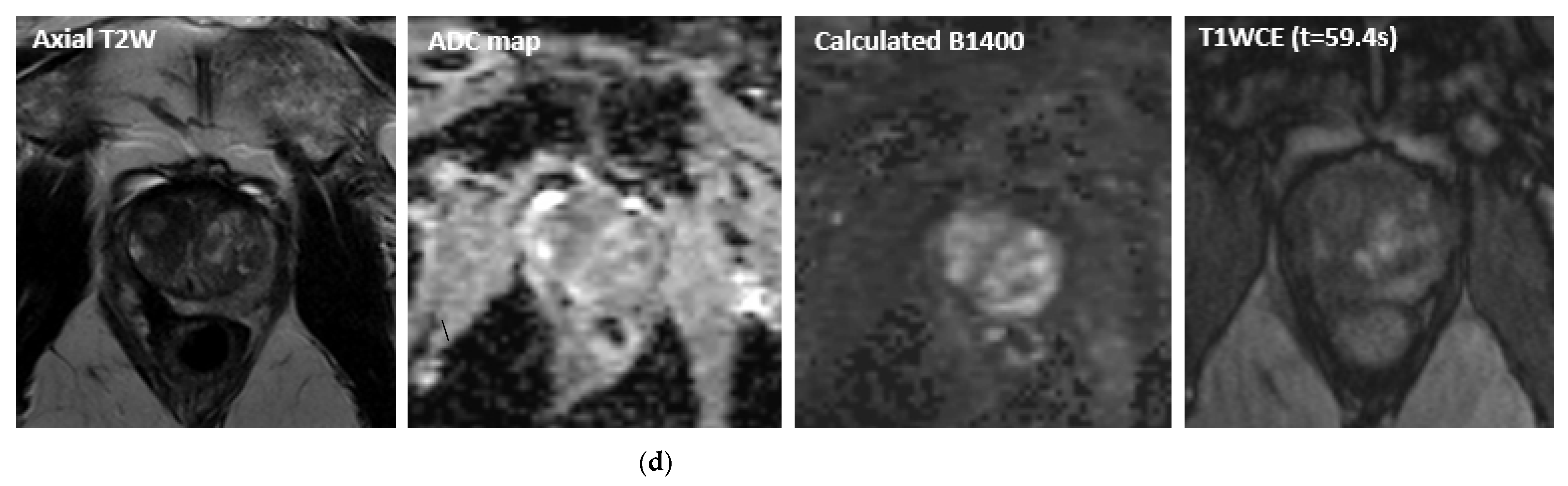
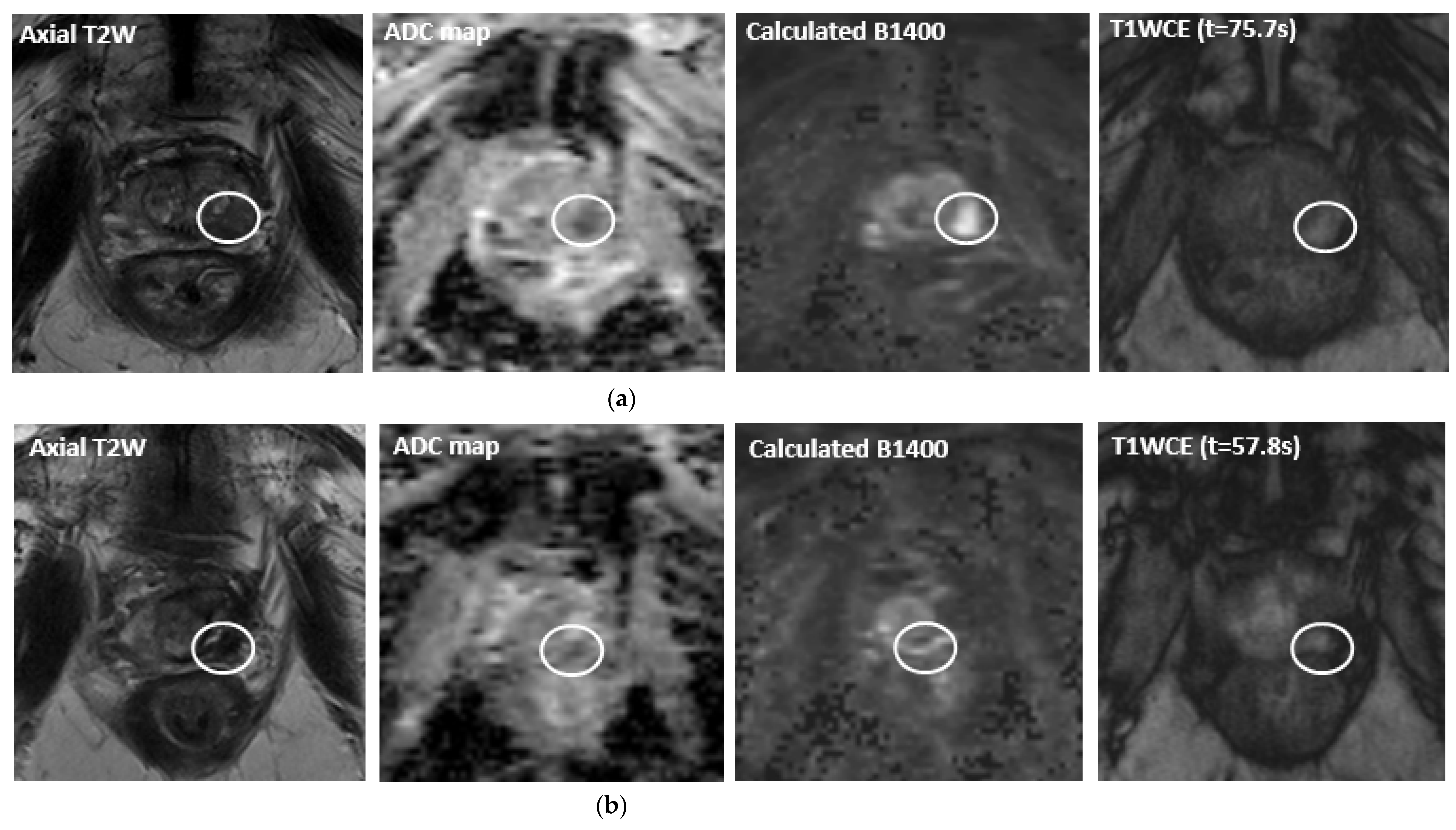
| Ablation Modality | Mechanism of Activity | Pros and Cons |
|---|---|---|
| Cryoablation | Cryoablation induces irreversible localized cell destruction by freezing followed by thawing | Cons: Real-time temperature mapping is not applicable Smaller lesions are more difficult to treat |
| FLA | FLA causes photothermal injury which leads to coagulative necrosis | Pros: Possibility to perform using the transperineal, transrectal or transgluteal approach Ability to perform under local anesthesia in an outpatient setting Real-time temperature mapping Cons: Only feasible for small lesions |
| HIFU | HIFU uses ultrasound energy focused by an acoustic lens to cause tissue damage | Pros: Real-time temperature mapping Cons: May not be suitable in patients with prostate calcifications Less suitable for ventral lesions |
| TULSA | TULSA delivers high-intensity directional (but unfocused) ultrasound energy which causes thermal damage | Pros: Ability to perform focal-, hemi- or whole gland ablation Real-time temperature mapping Cons: May not be suitable in patients with prostate calcifications |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wimper, Y.; Fütterer, J.J.; Bomers, J.G.R. MR Imaging in Real Time Guiding of Therapies in Prostate Cancer. Life 2022, 12, 302. https://doi.org/10.3390/life12020302
Wimper Y, Fütterer JJ, Bomers JGR. MR Imaging in Real Time Guiding of Therapies in Prostate Cancer. Life. 2022; 12(2):302. https://doi.org/10.3390/life12020302
Chicago/Turabian StyleWimper, Yvonne, Jurgen J. Fütterer, and Joyce G. R. Bomers. 2022. "MR Imaging in Real Time Guiding of Therapies in Prostate Cancer" Life 12, no. 2: 302. https://doi.org/10.3390/life12020302
APA StyleWimper, Y., Fütterer, J. J., & Bomers, J. G. R. (2022). MR Imaging in Real Time Guiding of Therapies in Prostate Cancer. Life, 12(2), 302. https://doi.org/10.3390/life12020302






