Transcutaneous Auricular Vagus Nerve Stimulation Improves Inflammation but Does Not Interfere with Cardiac Modulation and Clinical Symptoms of Individuals with COVID-19: A Randomized Clinical Trial
Abstract
1. Introduction
2. Material and Methods
2.1. Participants
2.2. Intervention
2.2.1. Experimental Group
2.2.2. Control Group
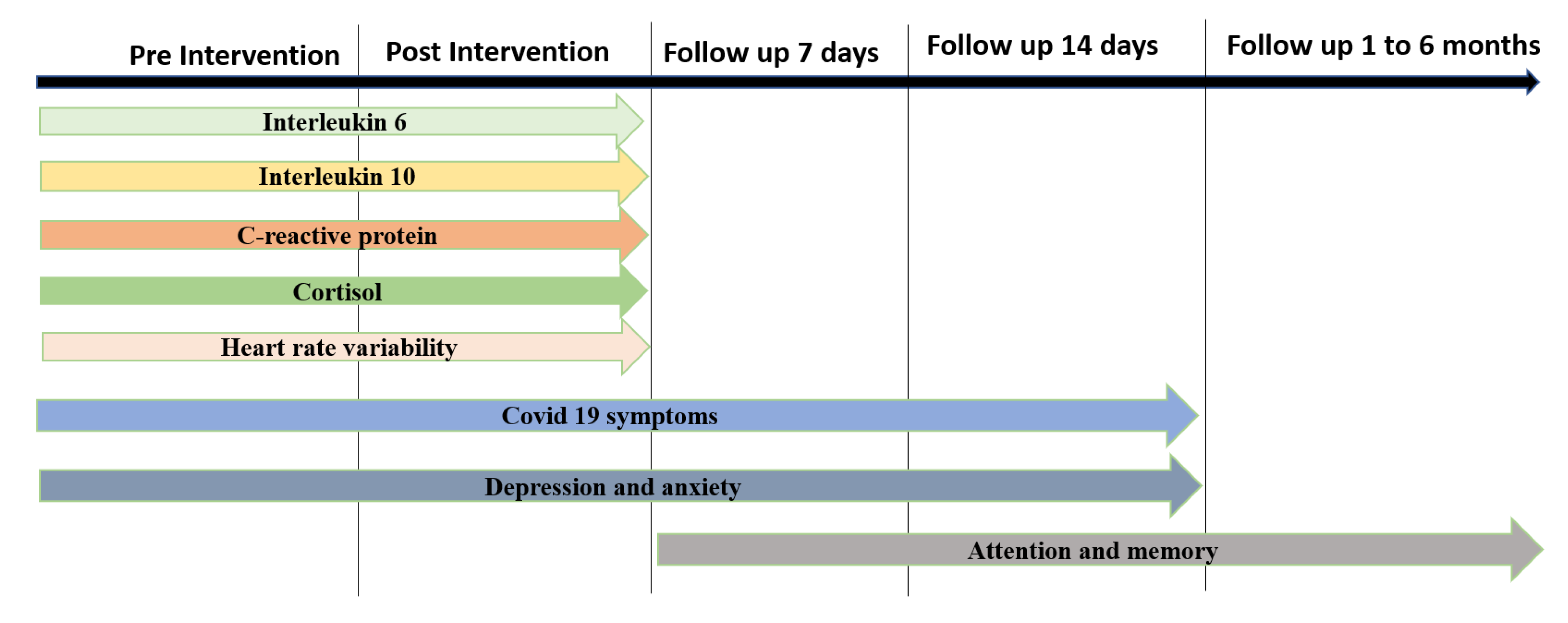
2.3. Outcome Measures
2.3.1. Primary Outcome Measures—Inflammatory Profile
2.3.2. Secondary Outcomes Measures
Cardiac Autonomic Modulation
COVID-19 Clinical Symptoms
Depression and Anxiety Scale
Attention and Memory Levels
2.4. Data Analysis
3. Results
3.1. Effects of Intervention
3.1.1. Primary Outcome–Inflammatory Profile
3.1.2. Secondary Outcomes
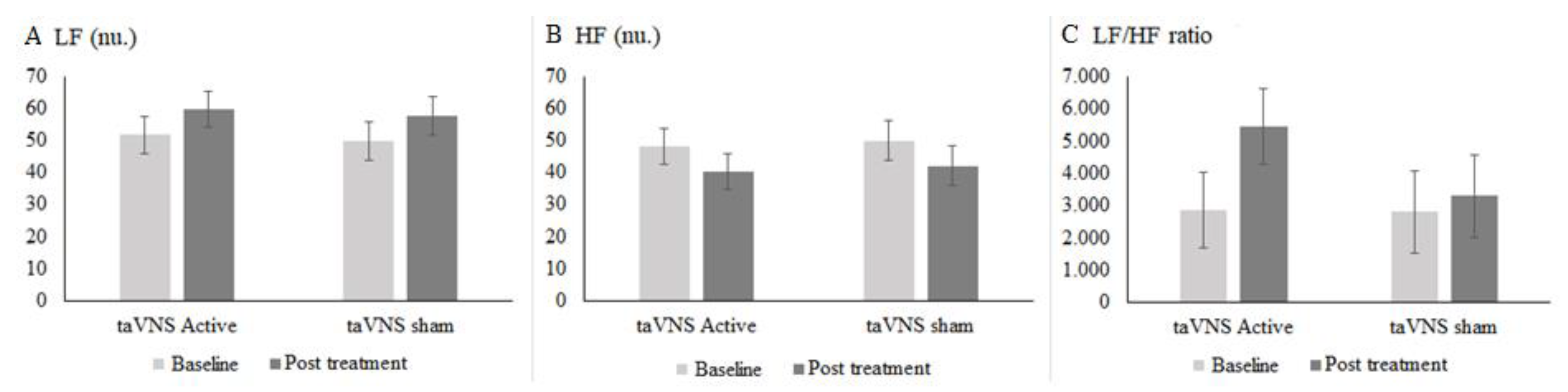
Heart Rate Variability (HRV)
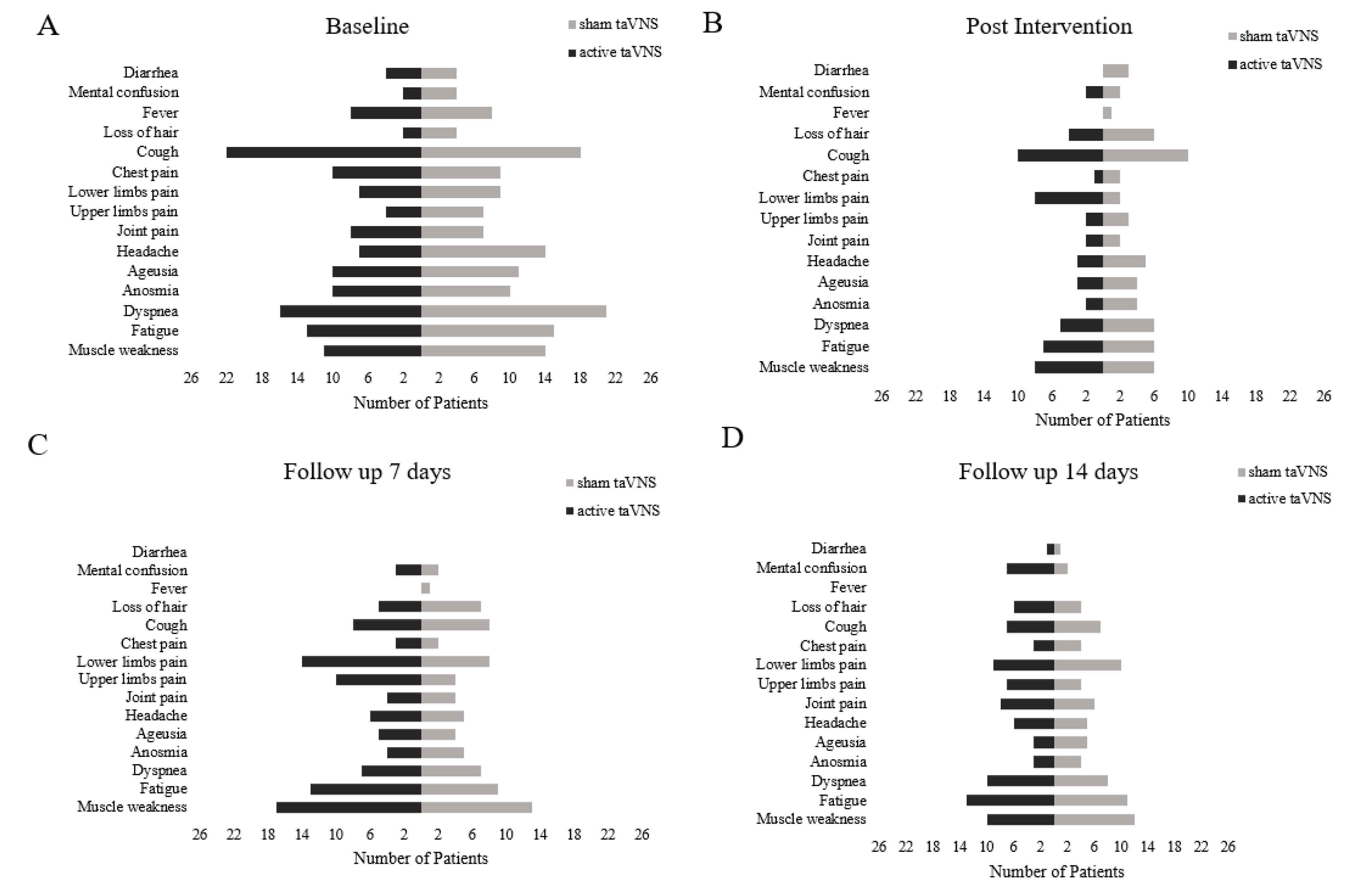
Clinical Symptoms
Depression and Anxiety
Attention and Memory Level
4. Discussion
4.1. Inflammatory Mediators
4.2. Autonomic Modulation
4.3. Clinical Symptoms
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A

Appendix B. Medications Intake during Trial (n = 52)
| Medication | s-taVNS (n = 26) Number (%) | a-taVNS (n = 26) Number (%) | p-Value |
| Antibiotics | 20 (76.9) | 23 (88.5) | 0.23 |
| Nonsteroidal anti-inflammatory drugs (NSAIDs) | 12 (46.2) | 18 (69.2) | 0.09 |
| Corticosteroids | 24 (92.3) | 25 (96.2) | 0.54 |
| Anticoagulants | 21 (80.8) | 25 (96.2) | 0.07 |
| Antihypertensives | 21 (80.8) | 26 (100.0) | 0.08 |
| Bronchodilator | 8 (30.8) | 13 (50.0) | 0.16 |
| Triglyceride Lowering Drugs | 3 (11.5) | 6 (23.1) | 0.30 |
| Antihistaminic | 1 (3.8) | 8 (30.8) | 0.06 |
| Anesthetic drugs | 22 (84.6) | 26 (100.0) | 0.06 |
| Antithyroid drugs | 1 (3.8) | 3 (11.5) | 0.29 |
| Analgesic | 12 (46.2) | 17 (65.4) | 0.16 |
| Hypoglycemic drugs | 10 (38.5) | 16 (61.5) | 0.09 |
| Vasopressors | 6 (23.1) | 12 (46.2) | 0.08 |
| Gastrointestinal drugs * | 10 (38.5) | 26 (100.0) | 0.001 |
| Antipsychotics | 4 (15.4) | 6 (23.1) | 0.46 |
| Expectorants | 0 (0.0) | 2 (7.7) | 0.17 |
| Antidepressants | 2 (7.7) | 3 (11.5) | 0.63 |
| Antifungal drugs | 1 (3.8) | 5 (19.2) | 0.09 |
| Uric acid lowering agent | 0 (0.0) | 2 (7.7) | 0.14 |
| Antiepileptics | 0 (0.0) | 2 (7.7) | 0.14 |
| Antiseptics | 0 (0.0) | 1 (3.8) | 0.30 |
| * laxants, antiflatulent, or proton pump inhibitor. | |||
References
- Huang, C.; Xu, X.; Cai, Y.; Ge, Q.; Zeng, G.; Li, X.; Zhang, W.; Ji, C.; Yang, L. Mining the Characteristics of COVID-19 Patients in China: Analysis of Social Media Posts. J. Med. Int. Res. 2020, 22, e19087. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Kaniusas, E.; Szeles, J.C.; Kampusch, S.; Alfageme-Lopez, N.; Yucumá, D.; Li, X.; Mayol, J.; Neumayer, C.; Papa, M.; Panetsos, F. Non-invasive Auricular Vagus Nerve Stimulation as a Potential Treatment for Covid19-Originated Acute Respiratory Distress Syndrome. Front. Physiol. 2020, 11, 890. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Chen, X.; Cai, Y.; Xia, J.; Zhou, X.; Xu, S.; Huang, H.; Zhang, L.; Zhou, X.; Du, C.; et al. Risk Factors Associated With Acute Respiratory Distress Syndrome and Death in Patients With Coronavirus Disease 2019 Pneumonia in Wuhan, China. JAMA Intern. Med. 2020, 180, 934–943, Erratum in JAMA Intern. Med. 2020, 180, 1031.. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, J.; Feng, Y. Critical care response to a hospital outbreak of the 2019-nCoV infection in Shenzhen, China. Crit. Care 2020, 24, 56. [Google Scholar] [CrossRef] [PubMed]
- Beekwilder, J.P.; Beems, T. Overview of the Clinical Applications of Vagus Nerve Stimulation. J. Clin. Neurophysiol. 2010, 27, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Sinkovec, M.; Trobec, R.; Meglic, B. Cardiovascular responses to low-level transcutaneous vagus nerve stimulation. Auton. Neurosci. 2021, 236, 102851. [Google Scholar] [CrossRef]
- Tornero, C.; Pastor, E.; Garzando, M.D.M.; Orduña, J.; Forner, M.J.; Bocigas, I.; Cedeño, D.L.; Vallejo, R.; McClure, C.K.; Czura, C.J.; et al. Non-invasive Vagus Nerve Stimulation for COVID-19: Results From a Randomized Controlled Trial (SAVIOR I). Front. Neurol. 2022, 13, 820864. [Google Scholar] [CrossRef]
- Boezaart, A.P.; Botha, D.A. Treatment of Stage 3 COVID-19 With Transcutaneous Auricular Vagus Nerve Stimulation Drastically Reduces Interleukin-6 Blood Levels: A Report on Two Cases. Neuromodulation Technol. Neural Interface 2020, 24, 166–167. [Google Scholar] [CrossRef]
- Zuanetti, G.; De Ferrari, G.M.; Priori, S.G.; Schwartz, P.J. Protective effect of vagal stimulation on reperfusion arrhythmias in cats. Circ. Res. 1987, 61, 429–435. [Google Scholar] [CrossRef]
- Clancy, J.A.; Mary, D.A.; Witte, K.K.; Greenwood, J.P.; Deuchars, S.A.; Deuchars, J. Non-invasive Vagus Nerve Stimulation in Healthy Humans Reduces Sympathetic Nerve Activity. Brain Stimul. 2014, 7, 871–877. [Google Scholar] [CrossRef]
- Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Heart Rate Variability: Standards of Measurement, Physiological Interpretation and Clinical Use. Circulation 1996, 93, 1043–1065. [Google Scholar] [CrossRef]
- Plaza-Florido, A.; Alcantara, J.M.; Amaro-Gahete, F.J.; Sacha, J.; Ortega, F.B. Cardiovascular Risk Factors and Heart Rate Variability: Impact of the Level of the Threshold-Based Artefact Correction Used to Process the Heart Rate Variability Signal. J. Med. Syst. 2020, 45, 2. [Google Scholar] [CrossRef]
- Yang, X.; Yu, Y.; Xu, J.; Shu, H.; Xia, J.; Liu, H.; Wu, Y.; Zhang, L.; Yu, Z.; Fang, M.; et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: A single-centered, retrospective, observational study. Lancet Respir. Med. 2020, 8, 475–481. [Google Scholar] [CrossRef]
- Umakanthan, S.; Sahu, P.; Ranade, A.V.; Bukelo, M.M.; Rao, J.S.; Abrahao-Machado, L.F.; Dahal, S.; Kumar, H.; Dhananjaya, K.V. Origin, transmission, diagnosis and management of coronavirus disease 2019 (COVID-19). Postgrad. Med. J. 2020, 96, 753–758. [Google Scholar]
- Botega, N.J.; Pondé, M.P.; Medeiros, P.; Lima, M.G.; Guerreiro, C.A.M. Validação da escala hospitalar de ansiedade e depressão (HAD) em pacientes epilépticos ambulatoriais. J. Bras Psiquiatr. 1998, 47, 285–289. [Google Scholar]
- Guy, W. ECDEU Assessment Manual For Psychopharmacology Revised; US Department of Health, Education, and Welfare Publication (ADM): Rockville, MD, USA, 1976; pp. 76–338. [Google Scholar]
- Sproston, N.R.; Ashworth, J.J. Role of C-Reactive Protein at Sites of Inflammation and Infection. Front. Immunol. 2018, 9, 754. [Google Scholar] [CrossRef]
- Sinniger, V.; Pellissier, S.; Fauvelle, F.; Trocmé, C.; Hoffmann, D.; Vercueil, L.; Cracowski, J.; David, O.; Bonaz, B. A 12-month pilot study outcomes of vagus nerve stimulation in Crohn’s disease. Neurogastroenterol. Motil. 2020, 32, e13911. [Google Scholar] [CrossRef]
- Koopman, F.A.; Chavan, S.S.; Miljko, S.; Grazio, S.; Sokolovic, S.; Schuurman, P.R.; Mehta, A.D.; Levine, Y.A.; Faltys, M.; Zitnik, R.; et al. Vagus nerve stimulation inhibits cytokine production and attenuates disease severity in rheumatoid arthritis. Proc. Natl. Acad. Sci. USA 2016, 113, 8284–8289. [Google Scholar] [CrossRef]
- Tarn, J.; Legg, S.; Mitchell, S.; Simon, B.; Ng, W.-F. The Effects of Noninvasive Vagus Nerve Stimulation on Fatigue and Immune Responses in Patients With Primary Sjögren’s Syndrome. Neuromodulation Technol. Neural Interface 2018, 22, 580–585. [Google Scholar] [CrossRef]
- Tracey, K.J. The inflammatory reflex. Nature 2002, 420, 853–859. [Google Scholar] [CrossRef]
- Hu, J.; Liu, S.; Ma, T. Research progress of exploring the treatment of sepsis based on cholinergic anti-inflammatory pathway. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue 2021, 33, 122–125. [Google Scholar]
- Tracey, K.J. Reflex control of immunity. Nat. Rev. Immunol. 2009, 9, 418–428. [Google Scholar] [CrossRef]
- García, L.F. Immune Response, Inflammation, and the Clinical Spectrum of COVID-19. Front. Immunol. 2020, 11, 1441. [Google Scholar] [CrossRef]
- Coomes, E.A.; Haghbayan, H. Interleukin-6 in COVID-19: A systematic review and meta-analysis. Rev. Med. Virol. 2020, 30, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Smilowitz, N.R.; Kunichoff, D.; Garshick, M.; Shah, B.; Pillinger, M.; Hochman, J.S.; Berger, J.S. C-reactive protein and clinical outcomes in patients with COVID-19. Eur. Heart J. 2021, 42, 2270–2279. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.; Gwenin, C. Cortisol level dysregulation and its prevalence—Is it nature’s alarm clock? Physiol. Rep. 2021, 8, e14644. [Google Scholar] [CrossRef]
- Lerman, I.; Hauger, R.; Sorkin, L.; Proudfoot, J.; Davis, B.; Huang, A.; Lam, K.; Simon, B.; Baker, D.G. Noninvasive Transcutaneous Vagus Nerve Stimulation Decreases Whole Blood Culture-Derived Cytokines and Chemokines: A Randomized, Blinded, Healthy Control Pilot Trial. Neuromodulation Technol. Neural Interface 2016, 19, 283–291. [Google Scholar] [CrossRef] [PubMed]
- Mol, M.B.A.; Strous, M.T.A.; van Osch, F.H.M.; Vogelaar, F.J.; Barten, D.G.; Farchi, M.; Foudraine, N.A.; Gidron, Y. Heart-rate-variability (HRV), predicts outcomes in COVID-19. PLoS ONE 2021, 16, e0258841. [Google Scholar] [CrossRef] [PubMed]
- Annane, D.; Trabold, F.; Sharshar, T.; Jarrin, I.; Blanc, A.S.; Raphael, J.C.; Gajdos, P. Inappropriate Sympathetic Activation at Onset of Septic Shock. Am. J. Respir. Crit. Care Med. 1999, 160, 458–465. [Google Scholar] [CrossRef]
- Ghosh, R.; Roy, D.; Sengupta, S.; Benito-León, J. Autonomic dysfunction heralding acute motor axonal neuropathy in COVID-19. J. NeuroVirol. 2020, 26, 964–966. [Google Scholar] [CrossRef]
- Kong, J.; Fang, J.; Park, J.; Li, S.; Rong, P. Treating Depression with Transcutaneous Auricular Vagus Nerve Stimulation: State of the Art and Future Perspectives. Front. Psychiatry 2018, 9, 20. [Google Scholar] [CrossRef]
- Staats, P.; Giannakopoulos, G.; Blake, J.; Liebler, E.; Levy, R.M. The Use of Non-invasive Vagus Nerve Stimulation to Treat Respiratory Symptoms Associated With COVID-19: A Theoretical Hypothesis and Early Clinical Experience. Neuromodulation Technol. Neural Interface 2020, 23, 784–788. [Google Scholar] [CrossRef]
- Lai, Y.-H.; Huang, Y.-C.; Huang, L.-T.; Chen, R.-M.; Chen, C. Cervical Noninvasive Vagus Nerve Stimulation for Migraine and Cluster Headache: A Systematic Review and Meta-Analysis. Neuromodulation Technol. Neural Interface 2020, 23, 721–731. [Google Scholar] [CrossRef]
- Paterson, R.W.; Brown, R.L.; Benjamin, L.; Nortley, R.; Wiethoff, S.; Bharucha, T.; Jayaseelan, D.L.; Kumar, G.; Raftopoulos, R.E.; Zambreanu, L.; et al. The emerging spectrum of COVID-19 neurology: Clinical, radiological and laboratory findings. Brain 2020, 143, 3104–3120. [Google Scholar] [CrossRef]
- Kremer, S.; Lersy, F.; De Sèze, J.; Ferré, J.C.; Maamar, A.; Carsin-Nicol, B.; Collange, O.; Bonneville, F.; Adam, G.; Martin-Blondel, G.; et al. Brain MRI Findings in Severe COVID-19: A Retrospective Observational Study. Radiology 2020, 297, E242–E251. [Google Scholar] [CrossRef]
- Bridwell, R.; Long, B.; Gottlieb, M. Neurologic complications of COVID-19. Am. J. Emerg. Med. 2020, 38, 1549.e3–1549.e7. [Google Scholar] [CrossRef]
- Beurel, E.; Toups, M.; Nemeroff, C.B. The Bidirectional Relationship of Depression and Inflammation: Double Trouble. Neuron 2020, 107, 234–256. [Google Scholar] [CrossRef]
- Dowlati, Y.; Herrmann, N.; Swardfager, W.; Liu, H.; Sham, L.; Reim, E.K.; Lanctôt, K.L. A Meta-Analysis of Cytokines in Major Depression. Biol. Psychiatry 2010, 67, 446–457. [Google Scholar] [CrossRef]
- Howren, M.B.; Lamkin, D.M.; Suls, J. Associations of Depression With C-Reactive Protein, IL-1, and IL-6: A Meta-Analysis. Psychosom. Med. 2009, 71, 171–186. [Google Scholar] [CrossRef]
- Guo, Z.-P.; Sörös, P.; Zhang, Z.-Q.; Yang, M.-H.; Liao, D.; Liu, C.-H. Use of Transcutaneous Auricular Vagus Nerve Stimulation as an Adjuvant Therapy for the Depressive Symptoms of COVID-19: A Literature Review. Front. Psychiatry 2021, 12, 765106. [Google Scholar] [CrossRef]
- Farmer, A.D.; Strzelczyk, A.; Finisguerra, A.; Gourine, A.V.; Gharabaghi, A.; Hasan, A.; Burger, A.M.; Jaramillo, A.M.; Mertens, A.; Majid, A.; et al. International Consensus Based Review and Recommendations for Minimum Reporting Standards in Research on Transcutaneous Vagus Nerve Stimulation (Version 2020). Front. Hum. Neurosci. 2021, 14, 568051. [Google Scholar] [CrossRef]
- Daban, C.; Martinez-Aran, A.; Cruz, N.; Vieta, E. Safety and efficacy of Vagus Nerve Stimulation in treatment-resistant depression. A systematic review. J. Affect. Disord. 2008, 110, 1–15. [Google Scholar] [CrossRef]
- Fang, J.; Rong, P.; Hong, Y.; Fan, Y.; Liu, J.; Wang, H.; Zhang, G.; Chen, X.; Shi, S.; Wang, L.; et al. Transcutaneous Vagus Nerve Stimulation Modulates Default Mode Network in Major Depressive Disorder. Biol. Psychiatry 2015, 79, 266–273. [Google Scholar] [CrossRef]
- Nemeroff, C.B.; Mayberg, H.S.; E Krahl, S.; McNamara, J.; Frazer, A.; Henry, T.; George, M.S.; Charney, D.S.; Brannan, S.K. VNS Therapy in Treatment-Resistant Depression: Clinical Evidence and Putative Neurobiological Mechanisms. Neuropsychopharmacology 2006, 31, 1345–1355. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Feng, X.L.; Wang, X.H.; Van Ijzendoorn, M.H. Coping with COVID-19: Exposure to COVID-19 and Negative Impact on Livelihood Predict Elevated Mental Health Problems in Chinese Adults. Int. J. Environ. Res. Public Health 2020, 17, 3857. [Google Scholar] [CrossRef] [PubMed]
- Botega, N.J.; Zomignani, M.A.; Garcia Junior, C.; Bio, M.R.; Pereira, W.A.B. Morbidade psiquiátrica no hospital geral: Utilização da edição revisada da “Clinical Interview Schedule-CIS-R”. ABP-APAL 1994, 16, 5762. [Google Scholar]
- Deitos, T.F.H.; Nascimento, C.A.M.D.; Noal, M.H.O. Depressao no hospital geral: Uma revisao bibliografica. J. Bras. De Psiquiatr. 1992, 7, 327–331. [Google Scholar]
- Evans, D.L.; Charney, D.S.; Lewis, L.; Golden, R.N.; Gorman, J.M.; Krishnan, K.R.R.; Nemeroff, C.B.; Bremner, J.D.; Carney, R.M.; Coyne, J.C.; et al. Mood Disorders in the Medically Ill: Scientific Review and Recommendations. Biol. Psychiatry 2005, 58, 175–189. [Google Scholar] [CrossRef] [PubMed]
- Lai, J.; Ma, S.; Wang, Y.; Cai, Z.; Hu, J.; Wei, N.; Wu, J.; Du, H.; Chen, T.; Li, R.; et al. Factors associated with mental health outcomes among health care workers exposed to coronavirus disease 2019. JAMA Netw. Open 2020, 3, e203976. [Google Scholar] [CrossRef]
- Harrison, N.A. Brain Structures Implicated in Inflammation-Associated Depression. Inflamm. Assoc. Depress. Evid. Mech. Implic. 2016, 31, 221–248. [Google Scholar] [CrossRef]
- Mehta, N.D.; Haroon, E.; Xu, X.; Woolwine, B.J.; Li, Z.; Felger, J.C. Inflammation negatively correlates with amygdala-ventromedial prefrontal functional connectivity in association with anxiety in patients with depression: Preliminary results. Brain Behav. Immun. 2018, 73, 725–730. [Google Scholar] [CrossRef]
- Nusslock, R.; Brody, G.H.; Armstrong, C.C.; Carroll, A.L.; Sweet, L.H.; Yu, T.; Barton, A.W.; Hallowell, E.S.; Chen, E.; Higgins, J.P.; et al. Higher Peripheral Inflammatory Signaling Associated With Lower Resting-State Functional Brain Connectivity in Emotion Regulation and Central Executive Networks. Biol. Psychiatry 2019, 86, 153–162. [Google Scholar] [CrossRef]

| Variables | a-taVNS (N = 26) | s-taVNS (N = 26) |
|---|---|---|
| Participants | ||
| Sex, male n (%) | 16 (61%) | 10 (38%) |
| Age (years), mean (SD) | 53 (17) | 57 (16) |
| Weight (kg), mean (SD) | 83 (13) | 86 (17) |
| Height (m), mean (SD) | 1.67 (7) | 1.66 (7) |
| BMI (Kg/m2), mean (SD) | 30 (4) | 31 (6) |
| SBP (mmHg), mean (SD) | 124 (17) | 126 (29) |
| DBP (mmHg), mean (SD) | 78 (10) | 75 (12) |
| HR (bpm), mean (SD) | 83 (13) | 80 (16) |
| IL-6 (pg/mL), mean (SD) | 32 (28) | 43 (38) |
| IL-10 (pg/mL), mean (SD) | 10 (9) | 8 (6) |
| CRP (mg/dL), mean (SD) | 9 (7) | 11 (10) |
| Cortisol (ug/dL), mean (SD) | 21 (17) | 25 (21) |
| LF (n.u), mean (SD) | 52 (27) | 50 (31) |
| HF (n.u), mean (SD) | 48 (27) | 50 (31) |
| HF/LF ratio, mean (SD) | 3 (5) | 3 (4) |
| Anxiety, mean (SD) | 8 (3) | 5 (3) |
| Depression, mean (SD) | 5 (4) | 3 (3) |
| Onset of Symptoms (days), mean (SD) | 9 (2) | 9 (2) |
| Death (n) | 4 | 6 |
| Comorbidities | ||
| AH, n (%) | 12 (46%) | 15 (58%) |
| Diabetes mellitus, n (%) | 7 (27%) | 7 (27%) |
| Obesity, n (%) | 11 (42%) | 13 (50%) |
| COPD, n (%) | 1 (4%) | 7 (27%) |
| Smokers, n (%) | 1 (4%) | 1 (4%) |
| Vaccinated, n (%) | 21 (81%) | 15 (58%) |
| Pfizer, n (%) | ||
| 1st dose | 2 (8%) | 1 (4%) |
| 2nd dose | 1 (4%) | 0 (0%) |
| CoronaVAC, n (%) | ||
| 1st dose | 12 (46%) | 10 (38%) |
| 2nd dose | 9 (35%) | 10 (38%) |
| AstraZeneca, n (%) | ||
| 1st dose | 6 (23%) | 3 (12%) |
| 2nd dose | 4 (15%) | 2 (8%) |
| Janssen, n (%) | 1 (4%) | 1 (4%) |
| a-taVNS (N = 26) | s-taVNS (N = 26) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Variable | Baseline | Post | Raw Changes within the Group | Adjusted Changes within the Group # | Baseline | Post | Raw Changes within the Group | Adjusted Changes within the Group # | Adjusted Difference between Groups ## |
| Anxiety | 8.00 (3.5) | 5.3 (3.7) | −2.7 (4.3) | 2.5 (4.6) | 5.5 (3.4) | 4.7 (4.6) | −1.0 (3.7) | −1.2 (4.6) | −1.27 |
| (−4.1 to 1.5) | |||||||||
| Depression | 4.6 (4.0) | 4.0 (5.0) * | −1.0 (3.8) | −1.8 (4.2) | 3.2 (3.0) | 3.1 (3.8) | 0.2 (3.6) | 1.0 (4.2) | −2.85 |
| (−5.4 to −0.3) | |||||||||
| a-taVNS (N = 20) | s-taVNS (N = 20) | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Outcome | 7 d | 14 d | 30 d | 60 d | 90 d | 120 d | 150 d | 180 d | 7 d | 14 d | 30 d | 60 d | 90 d | 120 d | 150 d | 180 d | Difference between Groups SD (95% CI) |
| Memory | 4.15 (0.33) | 3.95 (0.33) | 4 (0.33) | 3.45 (0.33) | 3.1 (0.33) | 3.4 (0.33) | 4.05 (0.33) | 4.15 (0.33) | 4.05 (0.33) | 4.10 (0.33) | 3.5 (0.33) | 3.75 (0.33) | 3.9 (0.33) | 3.85 (0.33) | 3.85 (0.33) | 3.95 (0.33) | −0.90 (−0.42 to 0.24) |
| Attention | 3.9 (0.34) | 3.9 (0.34) | 3.85 (0.34) | 3.55 (0.34) | 3.35 (0.34) | 3.6 (0.34) | 4.25 (0.34) | 4.3 (0.34) | 3.85 (0.34) | 3.7 (0.34) | 3.5 (0.34) | 3.6 (0.34) | 3.75 (0.34) | 3.8 (0.34) | 3.75 (0.34) | 3.65 (0.34) | 0.14 (−0.20 to 0.47) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corrêa, F.I.; Souza, P.H.L.; Uehara, L.; Ritti-Dias, R.M.; Oliveira da Silva, G.; Segheto, W.; Pacheco-Barrios, K.; Fregni, F.; Corrêa, J.C.F. Transcutaneous Auricular Vagus Nerve Stimulation Improves Inflammation but Does Not Interfere with Cardiac Modulation and Clinical Symptoms of Individuals with COVID-19: A Randomized Clinical Trial. Life 2022, 12, 1644. https://doi.org/10.3390/life12101644
Corrêa FI, Souza PHL, Uehara L, Ritti-Dias RM, Oliveira da Silva G, Segheto W, Pacheco-Barrios K, Fregni F, Corrêa JCF. Transcutaneous Auricular Vagus Nerve Stimulation Improves Inflammation but Does Not Interfere with Cardiac Modulation and Clinical Symptoms of Individuals with COVID-19: A Randomized Clinical Trial. Life. 2022; 12(10):1644. https://doi.org/10.3390/life12101644
Chicago/Turabian StyleCorrêa, Fernanda Ishida, Paulo Henrique Leite Souza, Laura Uehara, Raphael Mendes Ritti-Dias, Gustavo Oliveira da Silva, Wellington Segheto, Kevin Pacheco-Barrios, Felipe Fregni, and João Carlos Ferrari Corrêa. 2022. "Transcutaneous Auricular Vagus Nerve Stimulation Improves Inflammation but Does Not Interfere with Cardiac Modulation and Clinical Symptoms of Individuals with COVID-19: A Randomized Clinical Trial" Life 12, no. 10: 1644. https://doi.org/10.3390/life12101644
APA StyleCorrêa, F. I., Souza, P. H. L., Uehara, L., Ritti-Dias, R. M., Oliveira da Silva, G., Segheto, W., Pacheco-Barrios, K., Fregni, F., & Corrêa, J. C. F. (2022). Transcutaneous Auricular Vagus Nerve Stimulation Improves Inflammation but Does Not Interfere with Cardiac Modulation and Clinical Symptoms of Individuals with COVID-19: A Randomized Clinical Trial. Life, 12(10), 1644. https://doi.org/10.3390/life12101644








