Silver Carp (Hypophthalmichthys molitrix) (Asian Silver Carp) Presence in Danube Delta and Romania—A Review with Data on Natural Reproduction
Abstract
1. Introduction
2. External Morphology and Biology
3. When and How the Species Entered the Danube Delta and Romania
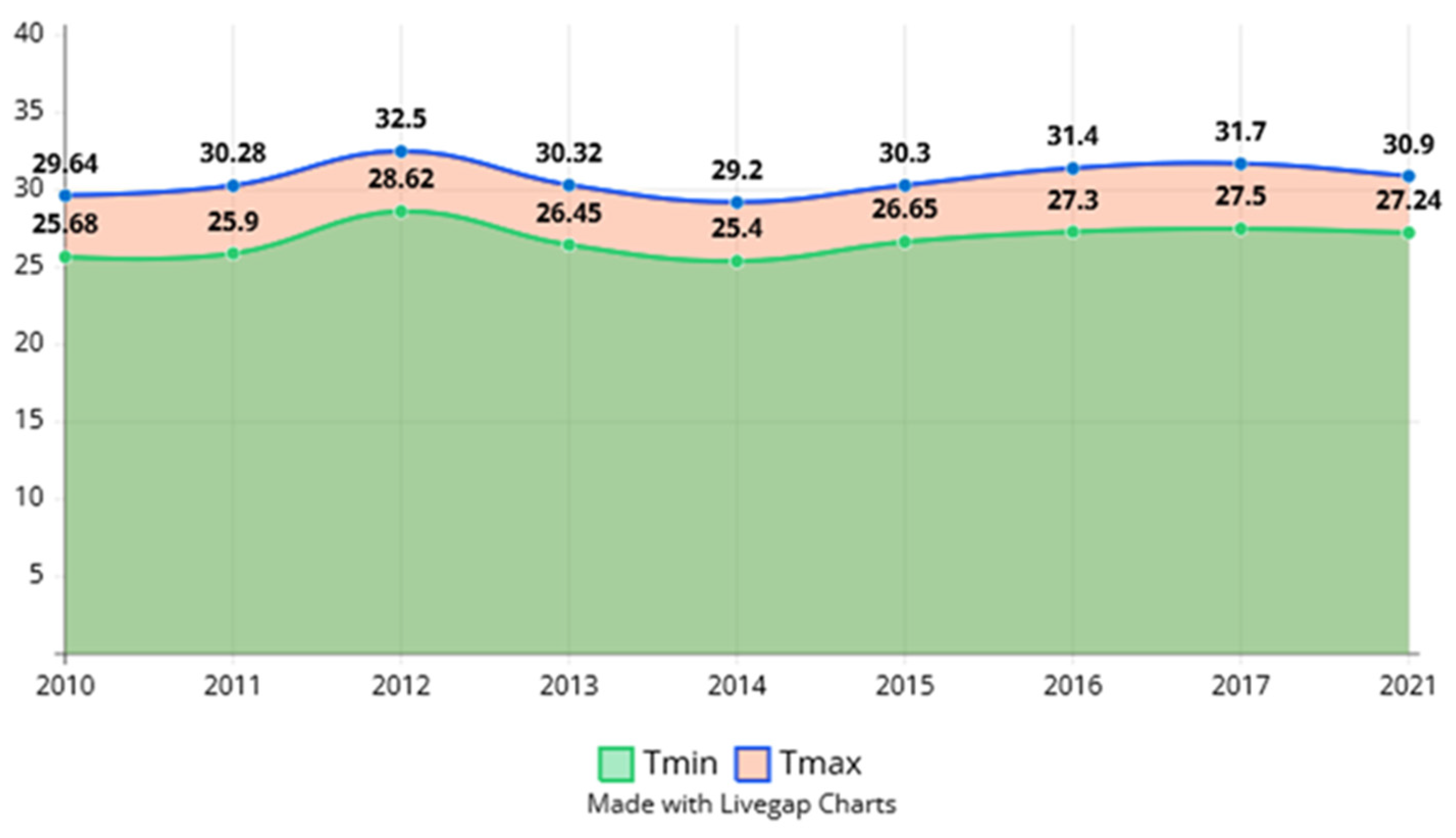
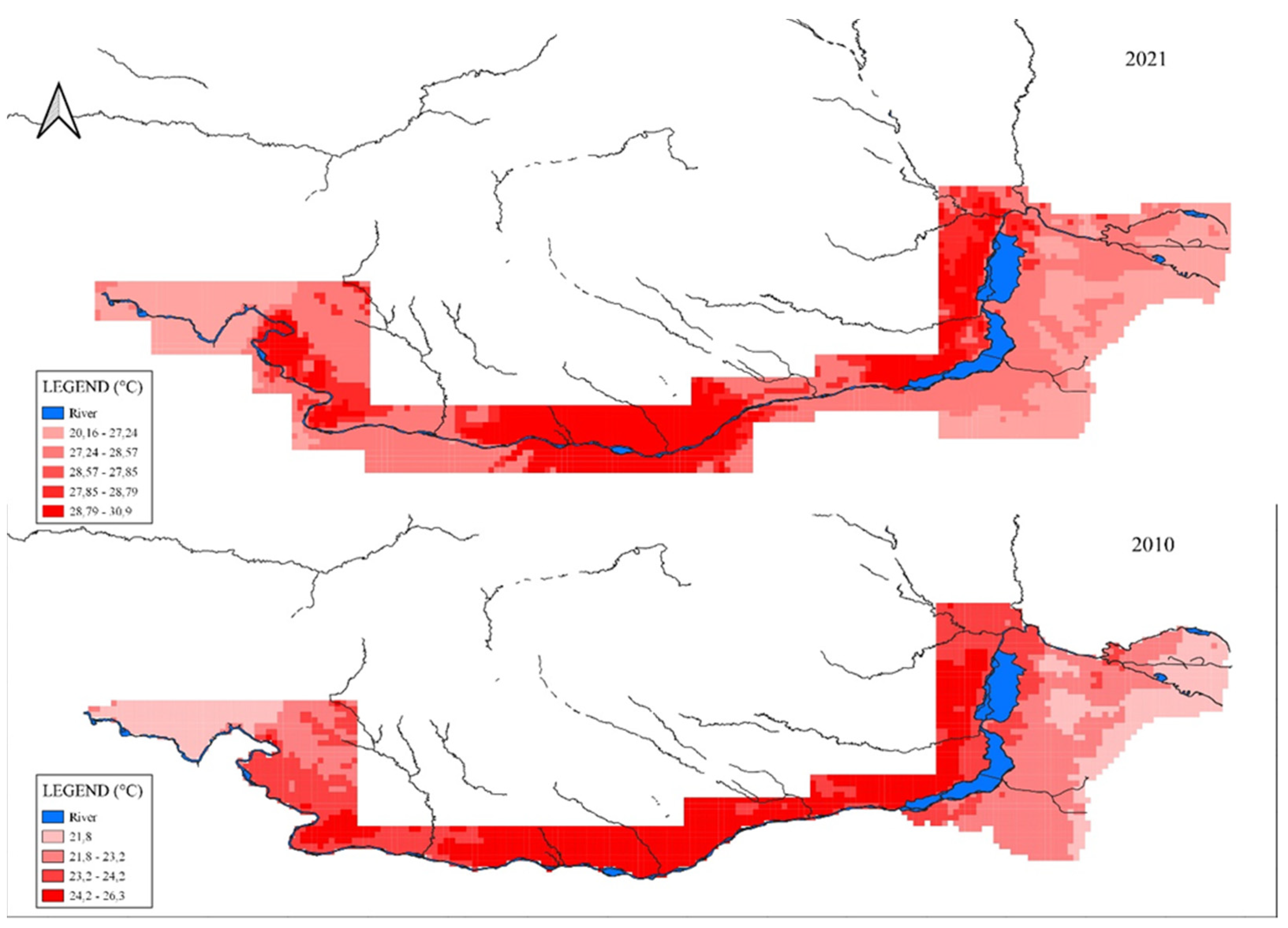
4. Water Temperature Suitable for Reproduction of Hypophthalmichthys molitrix
5. Nature of Nutrition
6. Ecological Effect and Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Panov, V.E.; Alexandrov, B.; Arbačiauskas, K.; Binimelis, R.; Copp, G.H.; Grabowski, M.; Lucy, F.; Leuven, R.S.; Nehring, S.; Paunović, M.; et al. Assessing the Risks of Aquatic Species Invasions via European Inland Waterways: From Concepts to Environmental Indicators. Integr. Environ. Assess. Manag. 2009, 5, 110–126. [Google Scholar] [CrossRef] [PubMed]
- Joint FAO (Food and Agriculture Organization of the United Nations). Caramel Colours. Combined Compendium of Food Additive Specification. Monograph 2011, 11, 1817–7077. [Google Scholar]
- Shi, C.; Cui, J.; Yin, X.; Luo, Y.; Zhou, Z. Grape seed and clove bud extracts as natural antioxidants in silver carp (Hypophthalmichthys molitrix) fillets during chilled storage: Effect on lipid and protein oxidation. Food Control 2014, 40, 134–139. [Google Scholar] [CrossRef]
- Lazăr, M.; Lazăr, R. Studies concerning the biometrics, hematology and biochemistry of meat in carp (Cyprinus carpio). Lucr. Şt. USAMV-Fac. Zoo 2012, 57, 44–49. [Google Scholar]
- Kachele, R.; Zhang, M.; Gao, Z.; Adhikari, B. Effect of vacuum packaging on the shelf-life of silver carp (Hypophthalmichthys molitrix) fillets stored at 4 °C. LWT—Food Sci. Technol. 2017, 80, 163–168. [Google Scholar] [CrossRef]
- Valipour, K.; Ariaii, F.; Khademi Shurmasti, P.D.; Nemati, M. Effect of chitosan edible coating enriched with Eucalyptus essential oil and α-tocopherol on silver carp fillets quality during refrigerated storage. J. Food Saf. 2017, 37, 1–8. [Google Scholar]
- Siddaiah, D.; Sagar Reddy, G.V.; Raju, C.V.; Chandrasekhar, T.C. Changes in lipids, proteins and kamaboko forming ability of silver carp (Hypophthalmichthys molitrix) mince during frozen storage. Food Res. Int. 2001, 34, 47–53. [Google Scholar] [CrossRef]
- Rezaei, F.; Shahbazi, Y. Shelf-life extension and quality attributes of sauced silver carp fillet: A comparison among direct addition, edible coating and biodegradable film. LWT Food Sci. Technol. 2018, 87, 122–133. [Google Scholar] [CrossRef]
- Kolar, C.S.; Chapman, D.C.; Courtenay, J.W.R.; Housel, C.M.; Williams, J.D.; Jennings, D.P. Bigheaded Carps: A Biological Synopsis and Environmental Risk Assessment; American Fisheries Society Special Publication: Bethesda, MD, USA, 2007; Volume 33. [Google Scholar]
- Molnár, T.; Lehoczky, I.; Meleg, E.E.; Boros, G.; Specziár, A.; Mozsár, A.; Vitál, Z.; Józsa, V.; Allele, W.; Urbányi, B.; et al. Comparison of the Genetic Structure of Invasive Bigheaded Carp (Hypophthalmichthys spp.) Populations in Central-European Lacustrine and Riverine Habitats. Animals 2021, 11, 2018. [Google Scholar] [CrossRef]
- National Agricultural Research and Innovation Center Research Institute of Agricultural Economics (NAIK AKI). Statistical Reports. Harvest Reports 2019. Available online: http://repo.aki.gov.hu/3584/1/Lehalaszas_2019_kiadvany.pdf (accessed on 24 June 2021). (In Hungarian)
- Ec.Europa.Eu. 2019. Available online: https://ec.europa.eu/environment/nature/invasivealien/docs/R_2016_1141_Union-list2019-consolidation.pdf (accessed on 12 May 2021).
- Cooke, S.L. Anticipating the spread and ecological effects of invasive bigheaded carps (Hypophthalmichthys spp.) in North America: A review of modeling and other predictive studies. Biol. Invasions 2016, 18, 315–344. [Google Scholar] [CrossRef]
- Zhang, G.; Chang, J.; Shu, G. Applications of factor-criteria system reconstruction analysis in the reproduction research on grass carp, black carp, silver carp and bighead in the yangtze river. Int. J. Gen. Syst. 2000, 29, 419–428. [Google Scholar] [CrossRef]
- Lehtonen, H. Alien Freshwater Fishes of Europe. In Invasive Aquatic Species of Europe. Distribution, Impacts and Management; Leppäkoski, E., Gollasch, S., Olenin, S., Eds.; Springer: Dordrecht, The Netherlands, 2002; pp. 153–161. [Google Scholar]
- Milardi, M.; Chapman, D.; Lanzoni, M.; Long, J.M.; Castaldelli, G. First evidence of bighead carp wild recruitment in Western Europe, and its relation to hydrology and temperature. PLoS ONE 2017, 12, e0189517. [Google Scholar] [CrossRef] [PubMed]
- Woynarovich, A.; Moth-Poulsen, T.; Péteri, A. Carp Polyculture in Central and Eastern Europe, the Caucasus and Central Asia: A Manual; FAO Fisheries and Aquaculture Technical Paper, 554; Food and Agriculture Organization of the United Nations: Rome, Italy, 2010; p. 73. [Google Scholar]
- Ciolac, A. The Asian carps from Romanian Danube River. Annals of the University. Dunarea De Jos Galati 2006, 21, 91–95. [Google Scholar]
- Jankovic, D. Natural reproduction by Asiatic herbivorous fishes in the Yugoslav section of the River Danube. Ital. J. Zool. 1998, 65, 227–228. [Google Scholar] [CrossRef]
- Pinter, K. Exotic Fishes in Hungarian Waters: Their Importance in Fishery Utilization of Natural Water Bodies and Fish Farming. Aquac. Res. 1980, 11, 163–167. [Google Scholar] [CrossRef]
- Berry, P.Y.; Low, M.P. Comparative studies on some aspects of the morphology and histology of Ctenopharyngodon idellus, Aristichthys nobilis, and their hybrid (Cyprinidae). Copeia 1970, 4, 708–726. [Google Scholar] [CrossRef]
- Vasile, O. Fish Atlas of the Danube Delta Biosphere Reserve; National Research Institute for the Development of the Danube Delta Tulcea, Publishing House of the Technological Information Center of the Danube Delta: Tulcea, Romania, 2007; pp. 200–203. ISBN 978-973-88117-0-6. [Google Scholar]
- Rose, G. Migration of Freshwater Fishes. Fish Fish. 2002, 3, 361–362. [Google Scholar] [CrossRef]
- Borcea, I. Observations on Black Sea Migratory Fishes. Ann. Univ. Iassy T 1927, XV. [Google Scholar]
- Bamme, V.; Apostolou, A.; Bulat, D.; Dumitrascu, O.C.; Effenberger, M.; Eros, T.; Hortic, S.; Kovác, V.; Simonovic, P. Fish. Chapter 05. In Joint Danube Survey 4 Scientific Report: A Shared Analysis of the Danube River; Liška, I., Wagner, F., Sengl, M., Deutsch, K., Slobodník, J., Paunovi´c, M., Eds.; ICPDR–International Commission for the Protection of the Danube River: Vienna, Austria, 2021; pp. 41–54. [Google Scholar]
- Manea, G. Sturgeons: Biology, Sturgeon Culture, and Sturgeon Culture Facilities; Ceres Publishing House: Bucharest, Romania, 1980. [Google Scholar]
- Marinov, B. Pseudorasbora parva (Schlegel, 1842) (Pisces, Cyprinidae)—A new representative of the ichthyofauna of Bulgaria. Hydrobiology 1979, 8, 75–78, (In Bulgarian, English abstract). [Google Scholar]
- Manolov, Z.; Sivkov, Y. Morphometrische Charakteristik eines für die Bulgarische Ichthyofauna neuen Fisches Pseudorasbora parva (Shlegel). Bull. Du Mus. Natl. De Varna 1977, 13, 219–222, (In Bulgarian, German summary). [Google Scholar]
- Năstase, A. New data concerning fish fauna from lakes of the fluvial Danube delta (Gorgova-Uzlina and SonteaFurtuna lake-complexes, Romania) in 2010. Sci. Ann. Danub. Delta Inst. 2012, 18, 75–88. [Google Scholar]
- Năvodaru, I.; Năstase, A.; Cernisencu, I. Study of ichthyofauna from Roşu-Puiu lake-complex. Sc. Ann. DDI Tulcea Rom. 2007, 1001, 13. [Google Scholar]
- Vasile, A.; Gabriela, M. The Cyprinidae Health Condition from the Anthropical Ecosystem of the Galaţi, Analele Univ. “Al.I. Cuza” Iaşi, secţiunea I Biologie animală, Tom LII, 2006, Ed. Univ. “Al.I. Cuza” Iaşi, 87-93 ISSN: 1224-581X, 93–95. 2007. Available online: http://scientific.thomsonreuters.com/cgibin/jrnlst/jlresults.cgi?PC=MASTER&ISSN (accessed on 20 July 2022).
- Luca, C.; Manea, A.; Luca, A.; Dinischiotu, A.; Costache, M. Molecular phylogeny of the Romanian cyprinids from the Danube River. Rom. Biotechnol. Lett. 2008, 13, 3970–3975. [Google Scholar]
- Bulat, D.; Toderas, I.; Usatii, M.; Biletchi, L.; Ene, A.; Bogdevici, O.; Denga, Y. Ichthyofauna diversity of the Prut River and the major threats. In Proceedings of the International Conference “Environmental Challenges in Lower Danube Euroregion”, Galati, Romania, 25–26 June 2015. [Google Scholar]
- Costiniuc, C.D.; Gorgan, D.L. Researches about Tansa—Belcesti Lake’s Ihtiofauna; Analele Ştiinţifice ale Universităţi “Al. I. Cuza”, Biologie animală, Tom L: Iaşi, Romania, 2004; pp. 215–221. ISSN 1224-581X. [Google Scholar]
- Năstase, A.; Iani, M.; Honț, Ș. Paraschiv, M. Fish fauna from Lower Romanian Mureș River. Sci. Ann. Danub. Delta Inst. 2020, 25. [Google Scholar]
- Bulat, D.; Bulat, D.; Ungureanu, L.; Dadu, A. Invasive ichthyofauna of the Lower Prut River ecosystem. In Environmental Challenges in Lower Danube Euroregion. 25–26 iunie 2015, Galaţi; Casa Cărţii de Ştiinţă: Cluj-Napoca, Romania, 2015; p. 36. [Google Scholar]
- Lujić, J.; Kostić, D.; Šipoš, Š.; Miljanović, B. Fish as a biological indicator in assesing [i.e. assessing] water quality of the river Tamiš [Serbia]. In Proceedings of the 5th International Conference Aquaculture and Fishery, Belgrade, Serbia, 1–3 June 2011. [Google Scholar]
- Teclean, I.C.; Cupsa, D. Fishfauna from the Lowland Mures River (Romania) and the Floodplain Natural Park Area (Western Romania); University of Oradea Publishing House: Oradea, Romania, 2013. [Google Scholar]
- Buchtova, H.; Ježek, F. A new look at the assessment of the silver carp (Hypophthalmichthys molitrix Val.) as a food fish. Czech J. Food Sci. 2011, 29, 487–497. [Google Scholar] [CrossRef]
- Luca, C.; Kevorkian, S.; Elvira, M.; Dinischiotu, A.; Costache, M. Romanian Cyprinids phylogeny based on 16s ARN mitochondrial genes. Biotechnol. Anim. Husb. 2007, 23, 405–411. [Google Scholar] [CrossRef]
- Bulat, D.; Bulat, D.; Davideanu, A.; Popescu, I.E.; Davideanu, G. Romania—Republic of Moldova joint study concerning the fish fauna in Stanca-Costesti reservoir. Aquac. Aquar. Conserv. Legis. 2016, 9, 550–563. [Google Scholar]
- Năstase, A.; Honț, S.; Iani, M.; Paraschiv, M.; Cernișencu, I.; Năvodaru, I. Ecological status of fish fauna from Razim Lake and the adjacent area, the Danube Delta Biosphere Reserve, Romania. Acta Ichthyol. Piscat. 2022, 52, 43–52. [Google Scholar] [CrossRef]
- Năstase, A.; Oţel, V.; Năvodaru, I. Ecological status of fish fauna in Arms of the Danube Delta (Danube Delta Biosphere Reserve, Romania) at the beginning of the third millennium. Acta Zool. Bulg. 2017, 69, 349–360. [Google Scholar]
- Telcean, I.C.; Cupşa, D. The backwaters and drainage canals as natural refuges for the lowland rivers’ fishfauna (Someş, Crişuri, and Mureş Rivers-north-western Romania). Biharean Biol. 2009, 3, 37–44. [Google Scholar]
- Bouroş, G.; Ionescu, D.T.; Hodor, C. Observation of Eurasian Otter’s Diel Activity Using Camera Trapping in Central-Eastern Romania. Vestnik Zool. 2019, 53, 47–56. [Google Scholar] [CrossRef]
- Cojocaru, C.D. Fish parasites of romania-an update. In Proceedings of the Sixth International Symposium on Aquatic Animal Health, Global Strategies for a Changing Environment, Timişoara, Romania, 5–9 September 2010. [Google Scholar] [CrossRef]
- László, A.; Béla, H.K.; Sándor, N. Changes in fish assemblage in the Hungarian section of River Szamos/Someş after a massive cyanide and heavy metal pollution. North-West. J. Zool. 2013, 9, 131–138. [Google Scholar]
- Diop, B.; Sanz, N.; Duplan, Y.J.J.; Guene, E.H.M.; Blanchard, F.; Pereau, J.-C.; Doyen, L. Maximum Economic Yield Fishery Management in the Face of Global Warming. Ecol. Econ. 2018, 154, 52–61. [Google Scholar] [CrossRef]
- Ionita, M.; Rimbu, N.; Chelcea, S.; Patrut, S. Multidecadal variability of summer temperature over Romania and its relation with Atlantic Multidecadal Oscillation. Theor. Appl. Climatol. 2013, 113, 305–315. [Google Scholar] [CrossRef]
- Rimbu, N.; Stefan, S.; Necula, C. The variability of winter high temperature extremes in Romania and its relationship with large-scale atmospheric circulation. Theor. Appl. Climatol. 2015, 121, 121–130. [Google Scholar] [CrossRef]
- Cheng, J.; Xu, L.; Feng, W.; Fan, H.; Jiang, J. Changes in Water Level Regimes in China’s Two Largest Freshwater Lakes: Characterization and Implication. Water 2019, 11, 917. [Google Scholar] [CrossRef]
- Simionov, I.A.; Petrea, S.M.; Mogodan, A.; Nica, A.; Cristea, D.; Neculita, M. Effect of changes in the Romanian lower sector Danube River hydrological and hydrothermal regime on fish diversity. Sci. Pap. Ser. E. Land Reclam. Earth Obs. Surv. Environ. Eng. 2020, 106–111. [Google Scholar]
- Ciolac, A. Migration of fishes in romanian danube river (Nº 1). Appl. Ecol. Environ. Res. 2004, 2, 143–163. [Google Scholar] [CrossRef]
- Fick, S.E.; Hijmans, R.J. WorldClim 2: New 1-km spatial resolution climate surfaces for global land areas. Int. J. Climatol. 2017, 37, 4302–4315. [Google Scholar] [CrossRef]
- Harris, I.; Jones, P.D.; Osborn, T.J.; Lister, D.H. Updated high-resolution grids of monthly climatic observations—The CRU TS3.10 Dataset. Int. J. Climatol. 2014, 34, 623–642. [Google Scholar] [CrossRef]
- Available online: https://www.worldclim.org/data/monthlywth.html (accessed on 5 August 2022).
- Staras, M.; Otel, V. The evidence regarding natural spawning of silver carp species Hypophthalmichthys molitrix (Valenciennes 1844) Val. in the Danube river. Sci. Ann. Danub. Delta Inst. 1999, 7, 183–187. [Google Scholar]
- Staras, M.; Cernisencu, I.; Constantin, G. New data concerning presence of silver carp Hypophtalmichthys molitrix, Val in the Danube river and Delta. Sci. Ann. Danub. Delta Inst. 1993, 2, 259–262. [Google Scholar]
- Smith, D.W. The feeding selectivity of silver carp, Hypophthalmichthys molitrix (Valenciennes 1844) Val. J. Fish Biol. 1989, 34, 819–828. [Google Scholar] [CrossRef]
- Komarovsky, F.Y.; Karasina, F.M.; Chernina, Z.V. Bioaccumulation of the persistent pesticides in fish of the Danube. Water Resour. 1993, 20, 520–522. [Google Scholar]
- Trichkova, T.; Todorov, M.; Kenderov, M.; Hubenov, Z.; Botev, I.; Stefanov, T.; Georgiev, D.; Jurajda, P. Invasive Alien Species of Benthic Macroinvertebrates and Fish in the Bulgarian Sector of the Danube River—Results of the Joint Danube Survey 4 (JDS4). Water 2022, 14, 2299. [Google Scholar] [CrossRef]
- Attayde, J.L.; van Nes, E.H.; Araujo, A.I.; Corso, G.; Scheffer, M. Omnivory by planktivores stabilizes plankton dynamics, but may either promote or reduce algal biomass. Ecosystems 2010, 13, 410–420. [Google Scholar] [CrossRef][Green Version]
- Currie, W.J.; Cuddington, K.M.; Stewart, T.J.; Zhang, H.; Koops, M.A. Modelling Spread, Establishment and Impact of Bighead and Silver Carps in the Great Lakes. No. 2011/113; DFO: Ottawa, ON, Canada, 2012. [Google Scholar]
- Liu, Q.-G.; Chen, Y.; Li, J.-L.; Chen, L.-Q. The food web structure and ecosystem properties of a filter-feeding carps dominated deep reservoir ecosystem. Ecol. Model. 2007, 203, 279–289. [Google Scholar] [CrossRef]
- Xie, P. Gut contents of silver carp, Hypophthalmichthys molitrix (Valenciennes 1844), and the disruption of a centric diatom, Cyclotella, on passage through the esophagus and intestine. Aquaculture 1999, 180, 295–305. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jawdhari, A.; Mihăilescu, D.F.; Fendrihan, S.; Jujea, V.; Stoilov-Linu, V.; Negrea, B.-M. Silver Carp (Hypophthalmichthys molitrix) (Asian Silver Carp) Presence in Danube Delta and Romania—A Review with Data on Natural Reproduction. Life 2022, 12, 1582. https://doi.org/10.3390/life12101582
Jawdhari A, Mihăilescu DF, Fendrihan S, Jujea V, Stoilov-Linu V, Negrea B-M. Silver Carp (Hypophthalmichthys molitrix) (Asian Silver Carp) Presence in Danube Delta and Romania—A Review with Data on Natural Reproduction. Life. 2022; 12(10):1582. https://doi.org/10.3390/life12101582
Chicago/Turabian StyleJawdhari, Abdulhusein, Dan Florin Mihăilescu, Sergiu Fendrihan, Valentin Jujea, Valeriu Stoilov-Linu, and Bogdan-Mihai Negrea. 2022. "Silver Carp (Hypophthalmichthys molitrix) (Asian Silver Carp) Presence in Danube Delta and Romania—A Review with Data on Natural Reproduction" Life 12, no. 10: 1582. https://doi.org/10.3390/life12101582
APA StyleJawdhari, A., Mihăilescu, D. F., Fendrihan, S., Jujea, V., Stoilov-Linu, V., & Negrea, B.-M. (2022). Silver Carp (Hypophthalmichthys molitrix) (Asian Silver Carp) Presence in Danube Delta and Romania—A Review with Data on Natural Reproduction. Life, 12(10), 1582. https://doi.org/10.3390/life12101582




