Evaluating the Microbial Habitability of Rogue Planets and Proposing Speculative Scenarios on How They Might Act as Vectors for Panspermia
Abstract
1. Introduction
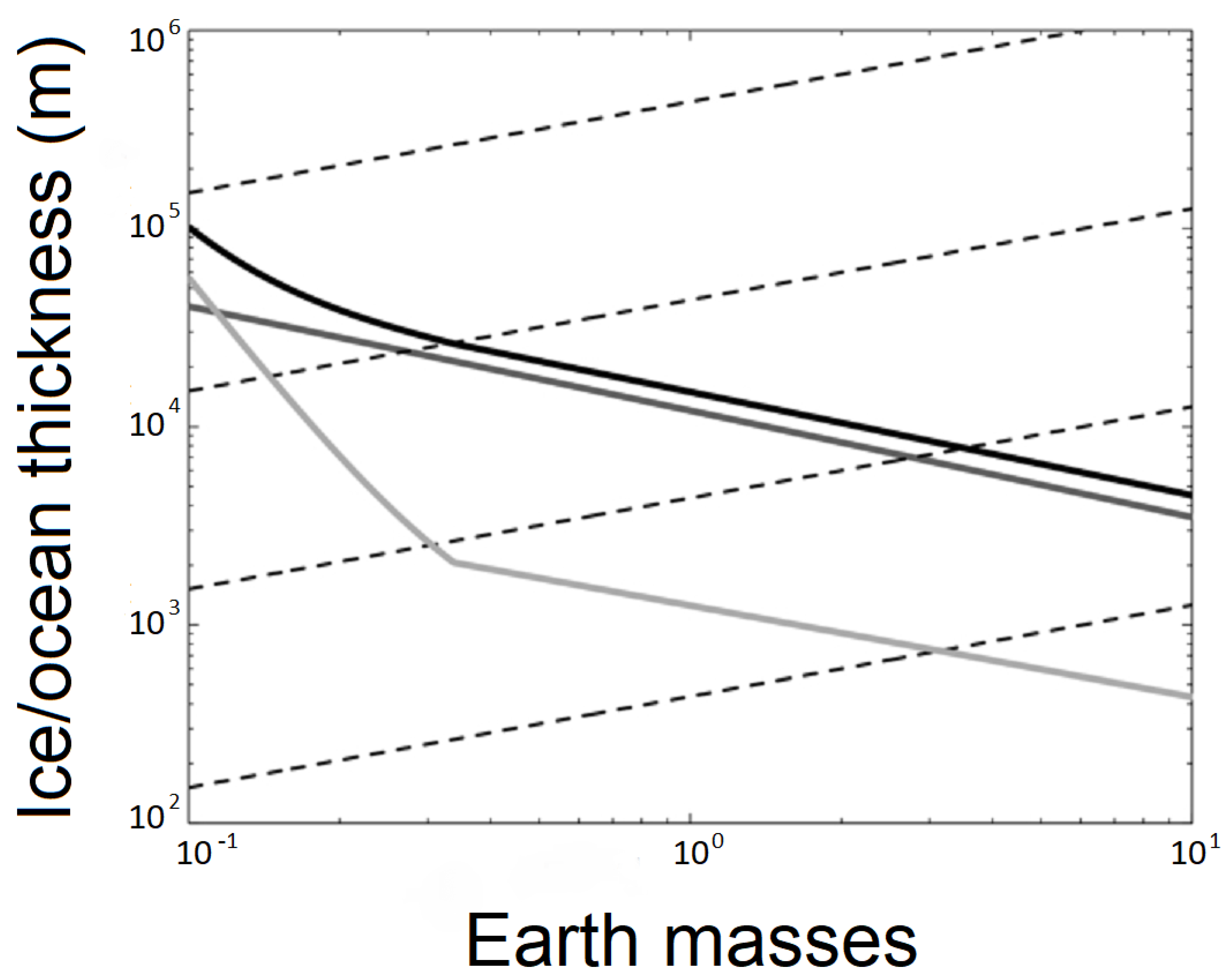
2. Liquid Water on Rogue Planets
3. Energy Sources for Life within Rogue Planets
3.1. Chemical Energy
3.2. Light Energy and Photosynthesis
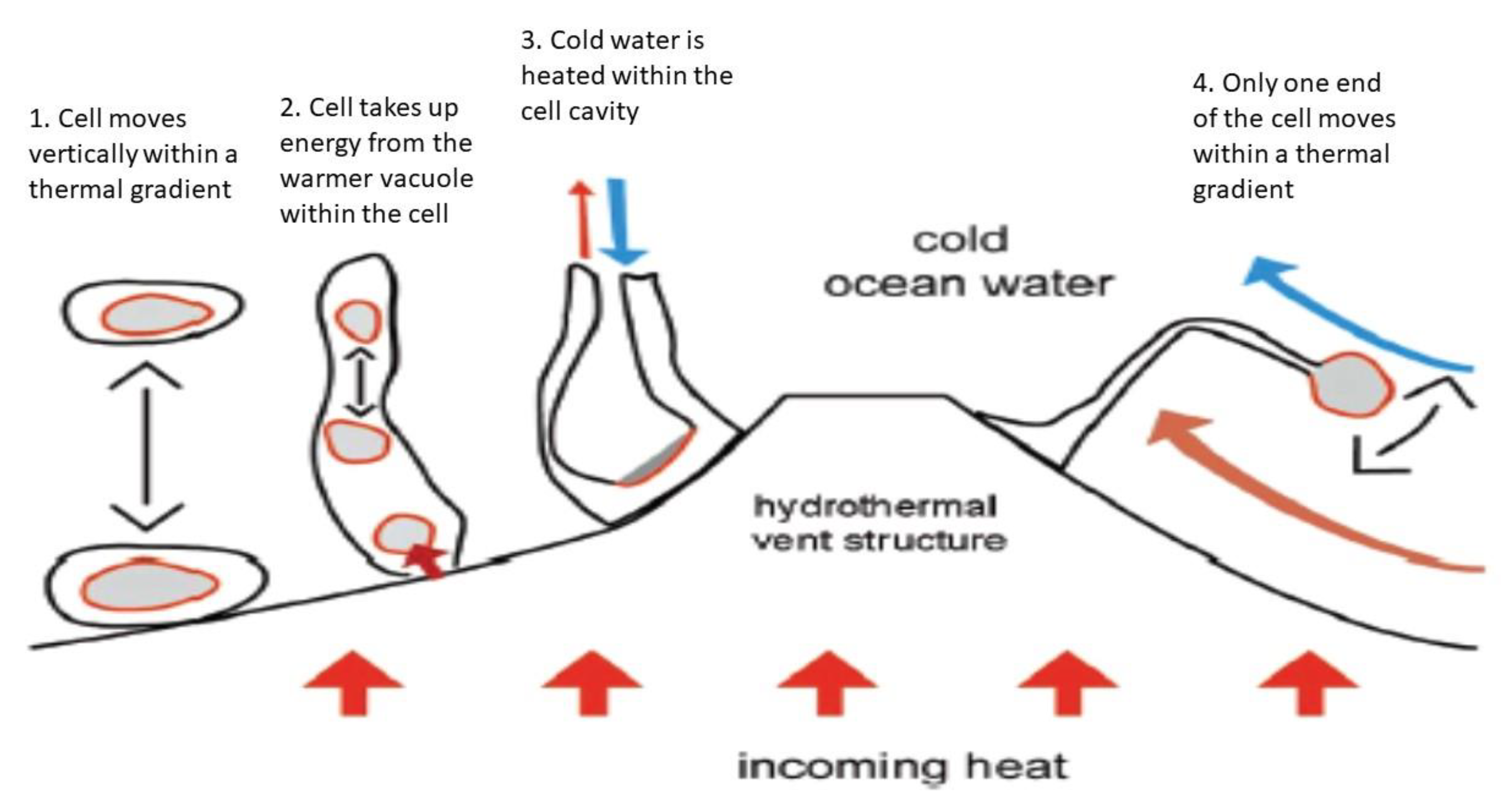
3.3. Thermal Energy and Thermosynthesis
3.4. Osmotic Energy
3.5. Other Potential Energy Sources
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Drake, F.D. Intelligent Life in Space; Macmillan: New York, NY, USA, 1962. [Google Scholar]
- Kok, B.; Radmer, R. Energy requirements of a biosphere. In Chemical Evolution of the Giant Planets; Ponnamperuma, C., Ed.; Academic Press: New York, NY, USA, 1976; pp. 183–197. [Google Scholar]
- Youvan, D.C.; Mars, B.L. Molecular mechanisms of photosynthesis. Sci. Am. 1987, 256, 42–49. [Google Scholar] [CrossRef]
- Caballero, J.A. A review on substellar objects below the deuterium burning mass limit: Planets, brown dwarfs or what? Geosciences 2018, 8, 362. [Google Scholar] [CrossRef]
- Sengupta, S. Brown dwarfs: The missing link between stars and planets. In Worlds Beyond Our Own; Sengupta, S., Ed.; Springer: Cham, Switzerland, 2015; pp. 59–65. [Google Scholar]
- Baker, V.R.; Dohm, J.M.; Fairén, A.G.; Ferré, T.P.A.; Ferris, J.C.; Miyamoto, H.; Schulze-Makuch, D. Extraterrestrial hydrogeology. Hydrogeol. J. 2005, 13, 51–68. [Google Scholar] [CrossRef][Green Version]
- Schulze-Makuch, D.; Irwin, L.N. Life in the Universe: Expectations and Constraints, 3rd ed.; Springer: Berlin, Germany, 2018; 343p. [Google Scholar]
- Delorme, P.; Gagné, J.; Malo, L.; Reylé, C.; Artigau, E.; Albert, L.; Forveille, T.; Delfosse, X.; Allard, F.; Homeier, D. CFBDSIR2149-0403: A 4–7 Jupiter-mass free-floating planet in the young moving group AB Doradus? Astron. Astrophys. 2012, 548, A26. [Google Scholar] [CrossRef]
- Liu, M.C.; Magnier, E.A.; Deacon, N.R.; Allers, K.N.; Dupuy, T.J.; Kotson, M.C.; Aller, K.M.; Burgett, W.S.; Chambers, K.C.; Draper, P.W.; et al. The extremely red, young L dwarf PSO J318.5338–22.8603: A free-floating planetary-mass analog to directly imaged young gas-giant planets. Astrophys. J. Lett. 2013, 777, L20. [Google Scholar] [CrossRef]
- Lund, M.B. Worlds in migration. arXiv 2019, arXiv:1903.12437. [Google Scholar]
- Bromley, B.C.; Kenyon, S.J. The fate of scattered planets. Astrophys. J. 2014, 796, 141. [Google Scholar] [CrossRef]
- Bada, J.L. State-of-the-art instruments for detecting extraterrestrial life. Proc. Natl. Acad. Sci. USA 2001, 98, 797–800. [Google Scholar] [CrossRef] [PubMed]
- Mróz, P.; Poleski, R.; Gould, A.; Udalski, A.; Sumi, T.; Szymański, M.K.; Soszyński, I.; Pietrukowicz, P.; Kozłowski, S.; Skowron, J.; et al. A terrestrial-mass rogue planet candidate detected in the shortest-timescale microlensing event. Astrophys. J. Lett. 2020, 903, L11. [Google Scholar] [CrossRef]
- Fairén, A.G.; Gómez-Elvira, J.; Briones, C.; Prieto-Ballesteros, O.; Rodríguez-Manfredi, J.A.; López Heredero, R.; Belenguer, T.; Moral, A.G.; Moreno-Paz, M.; Parro, V. The Complex Molecules Detector (CMOLD): A fluidic-based instrument suite to search for (bio)chemical complexity on Mars and icy moons. Astrobiology 2020, 20, 1076–1096. [Google Scholar] [CrossRef]
- Ruiz, J.; Fairén, A.G. Seas under ice: Conditions for the stability of liquid-water oceans within icy worlds. Earth Moon Planets 2005, 97, 79–90. [Google Scholar] [CrossRef]
- Lingam, M.; Loeb, A. Subsurface exolife. Int. J. Astrobiol. 2019, 18, 112–141. [Google Scholar] [CrossRef]
- Lingam, M.; Loeb, A. Life in the Cosmos: From Biosignatures to Technosignatures; Harvard University Press: Cambridge, MA, USA, 2021. [Google Scholar]
- Lingam, M.; Loeb, A. Potential for liquid water biochemistry deep under the surface of Moon, Mars, and beyond. Astrophys. J. Lett. 2020, 901, L11. [Google Scholar] [CrossRef]
- Irwin, L.N.; Schulze-Makuch, D. Strategy for modeling putative ecosystems on Europa. Astrobiology 2003, 3, 813–821. [Google Scholar] [CrossRef] [PubMed]
- Abbot, D.; Switzer, E. The Steppenwolf: A proposal for a habitable planet in interstellar space. Astrophys. J. Lett. 2011, 735, L27. [Google Scholar] [CrossRef]
- Stevenson, D. Life-Sustaining Planets in Interstellar Space? Nature 1999, 400, 32. [Google Scholar] [CrossRef]
- Badescu, V. Free-floating planets as potential seats for aqueous and non-aqueous life. Icarus 2011, 216, 485–491. [Google Scholar] [CrossRef]
- Shapiro, R.; Schulze-Makuch, D. The search for alien life in our solar system: Strategies and priorities. Astrobiology 2009, 9, 335–343. [Google Scholar] [CrossRef]
- Lingam, M.; Loeb, A. On the habitable lifetime of terrestrial worlds with high radionuclide abundances. Astrophys. J. Lett. 2020, 889, L20. [Google Scholar] [CrossRef]
- Lyu, Z.; Shao, N.; Akinyemi, T.; Whitman, W.B. Methanogenesis. Curr. Biol. 2018, 28, R727–R732. [Google Scholar] [CrossRef] [PubMed]
- Haldane, J. The origin of life. Ration. Annu. 1929, 148, 3–10. [Google Scholar]
- Fox, S.W.; Dose, K. Molecular Evolution and the Origin of Life; Marcel Dekker: New York, NY, USA, 1977. [Google Scholar]
- Miller, S.L.; Orgel, L.E. The Origins of Life on the Earth; Prentice-Hall: Hoboken, NJ, USA, 1974. [Google Scholar]
- Chyba, C.F.; Thomas, P.J.; Brookshaw, L.; Sagan, C. Cometary delivery of organic molecules to the early Earth. Science 1990, 249, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Stevens, T. Lithoautotrophy in the subsurface. FEMS Microbiol. Rev. 1997, 20, 327–337. [Google Scholar] [CrossRef]
- D´Hondt, S.; Rutherford, S.; Spivack, A.J. Metabolic activity of subsurface life in deep-sea sediments. Science 2002, 295, 2067–2070. [Google Scholar] [CrossRef] [PubMed]
- Stevens, T.O.; McKinley, J.P. Lithoautotrophic microbial ecosystems in deep basalt aquifers. Science 1995, 270, 450–455. [Google Scholar] [CrossRef]
- Gold, T. The deep, hot biosphere. Proc. Natl. Acad. Sci. USA 1992, 89, 6045–6049. [Google Scholar] [CrossRef] [PubMed]
- Sleep, N.; Zahnle, K. Refugia from asteroid impacts on early Mars and the early Earth. J. Geophys. Res. 1998, 103, 28529–28544. [Google Scholar] [CrossRef]
- González-Toril, E.; Martínez-Frías, J.; Gómez Gómez, J.M.; Rull, F.; Amils, R. Iron meteorites can support the growth of acidophilic chemolithoautotrophic microorganisms. Astrobiology 2005, 5, 406–414. [Google Scholar] [CrossRef]
- Beatty, J.T.; Overmann, J.; Lince, M.T.; Manske, A.K.; Lang, A.S.; Blankenship, R.E.; Van Dover, C.L.; Martinson, T.A.; Plumley, F.G. An obligately photosynthetic bacterial anaerobe from a deep-sea hydrothermal vent. Proc. Natl. Acad. Sci. USA 2005, 102, 9306–9310. [Google Scholar] [CrossRef]
- Muller, A.W.J. Thermosynthesis by biomembranes: Energy gain from cyclic temperature changes. J. Theor. Biol. 1985, 115, 429–453. [Google Scholar] [CrossRef]
- Muller, A.W.J. Were the first organisms heat engines? A new model for biogenesis and the early evolution of biological energy conversion. Prog. Biophys. Molec. Biol. 1995, 63, 193–231. [Google Scholar] [CrossRef]
- Muller, A.W.J. Finding extraterrestrial organisms living on thermosynthesis. Astrobiology 2003, 3, 555–564. [Google Scholar] [CrossRef]
- Muller, A.W.J.; Schulze-Makuch, D. Thermal energy and the origin of life. Orig. Life Evol. Biosph. 2006, 36, 177–189. [Google Scholar] [CrossRef]
- Schulze-Makuch, D.; Irwin, L.N. Energy cycling and hypothetical organisms in Europa’s ocean. Astrobiology 2002, 2, 105–121. [Google Scholar] [CrossRef]
- Madigan, M.T.; Martinko, J.M.; Parker, J. Brock Biology of Microorganisms; Prentice Hall: Hoboken, NJ, USA, 2000. [Google Scholar]
- Shihira-Ishikawa, I.; Nawata, T. The structure and physiological properties of the cytoplasm in intact Valonia cell. Jpn. J. Phycol. 1992, 40, 151–159. [Google Scholar]
- Wilmer, P.; Stone, G.; Johnston, I. Environmental Physiology of Animals; Blackwell Science: Oxford, UK, 2000. [Google Scholar]
- Heinz, J.; Krahn, T.; Schulze-Makuch, D. A new record for microbial perchlorate tolerance: Fungal growth in NaClO4 Brines and its implications for putative life on Mars. Life 2020, 10, 53. [Google Scholar] [CrossRef]
- Breuer, U.; Harms, H. Debaryomyces hansenii—An extremophilic yeast with biotechnological potential. Yeast 2006, 23, 415–437. [Google Scholar] [CrossRef]
- Irwin, L.N.; Schulze-Makuch, D. The astrobiology of alien worlds: Known and unknown forms of life. Universe 2020, 6, 130. [Google Scholar] [CrossRef]
- Blakemore, R.P. Magnetotactic bacteria. Annu. Rev. Microbiol. 1982, 36, 217–238. [Google Scholar] [CrossRef]
- Gould, J.L. Animal navigation: The evolution of magnetic orientation. Curr. Biol. 2018, 18, R482–R484. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.I.; Markwardt, C.B.; Swank, J.H.; Ransom, S.; Roberts, M.; Kaspi, V.; Woods, P.M.; Safi-Harb, S.; Balman, S.; Parke, W.C.; et al. Discovery of a transient magnetar: XTE J1810-197. Astrophys. J. Lett. 2004, 609, L21. [Google Scholar] [CrossRef]
- Debes, J.H.; Sigurdsson, S. The survival rate of ejected terrestrial planets with moons. Astrophys. J. 2007, 668, L167–L170. [Google Scholar] [CrossRef]
- Sauvage, J.F.; Flinders, A.; Spivack, A.J.; Pockalny, R.; Dunlea, A.G.; Anderson, C.H.; Smith, D.C.; Murray, R.W.; D’Hondt, S. The contribution of water radiolysis to marine sedimentary life. Nat. Commun. 2021, 12, 1297. [Google Scholar] [CrossRef]
- Lollar, B.S.; Heuer, V.B.; McDermott, J.; Tille, S.; Warr, O.; Moran, J.J.; Telling, J.; Hinrichs, K.U. A window into the abiotic carbon cycle—Acetate and formate in fracture waters in 2.7 billion years-old host rocks of the Canadian Shield. Geochim. Cosmochim. Acta 2021, 294, 295–314. [Google Scholar] [CrossRef]
- Pirbadian, S.; Barchinger, S.E.; Leung, K.M.; Byun, H.S.; Jangir, Y.; Bouhenni, R.A.; Reed, S.B.; Romine, M.F.; Saffarini, D.A.; Shi, L.; et al. Shewanella oneidensis MR-1 nanowires are outer membrane and periplasmic extensions of the extracellular electron transport components. Proc. Natl. Acad. Sci. USA 2014, 111, 12883–12885. [Google Scholar] [CrossRef] [PubMed]
- Rowe, A.R.; Chellamuthu, P.; Lam, B.; Okamoto, A.; Nealson, K.H. Marine sediments microbes capable of electrode oxidations as a surrogate for lithotrophic insoluble substrate metabolism. Front. Microbiol. 2015, 5, 784. [Google Scholar] [CrossRef]
- Strigari, L.; Barnabè, M.; Marshall, P.; Blandford, R. Nomads of the Galaxy. Mon. Not. R. Astron. Soc. 2012, 423, 1856–1865. [Google Scholar] [CrossRef]
- Meech, K.J.; Weryk, R.; Micheli, M.; Kleyna, J.T.; Hainaut, O.R.; Jedicke, R.; Wainscoat, R.J.; Chambers, K.C.; Keane, J.V.; Petric, A.; et al. A brief visit from a red and extremely elongated interstellar asteroid. Nature 2017, 552, 378–381. [Google Scholar] [CrossRef] [PubMed]
- Opitom, C.; Fitzsimmons, A.; Jehin, E.; Moulane, Y.; Hainaut, O.; Meech, K.J.; Yang, B.; Snodgrass, C.; Micheli, M.; Keane, J.V.; et al. 2I/Borisov: A C2-depleted interstellar comet. Astron. Astrophys. 2019, 631, L8. [Google Scholar] [CrossRef]
- Bayaz, R. Trillions of Rogue Planets with Life Roam Milky Way; Outnumber Stars by Factors of Thousands; Before Its News: Mill Valley, CA, USA, 2012; Available online: http://beforeitsnews.com/space/2012/05/trillions-of-rogue-planets-with-life-roam-milky-way-outnumber-stars-by-factors-of-thousands-2124171.html (accessed on 11 May 2012).
- Chyba, C.F.; Phillips, C.B. Possible ecosystems and the search for life on Europa. Proc. Natl. Acad. Sci USA 2001, 98, 801–804. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, R.; Geissler, P.; Tufts, B.R.; Hoppa, G.V. Habitability of Europa´s crust: The role of tidal-tectonic processes. J. Geophys. Res. Planets 2000, 105, 17551–17562. [Google Scholar] [CrossRef]
- Roads, E.; Longton, R.E.; Convey, P. Millennial timescale regeneration in a moss from Antarctica. Curr. Biol. 2014, 24, R222–R223. [Google Scholar] [CrossRef]
- Shmakova, L.; Malavin, S.; Iakovenko, N.; Vishnivetskaya, T.; Shain, D.; Plewka, M.; Rivkina, E. A living bdelloid rotifer from 24,000-year-old Arctic permafrost. Curr. Biol. 2021, 31, R712–R713. [Google Scholar] [CrossRef]
- Shatilovich, A.V.; Tchesunov, A.V.; Neretina, T.V.; Grabarnik, I.P.; Gubin, S.V.; Vishnivetskaya, T.A.; Onstott, T.C.; Rivkina, E.M. Viable nematodes from late Pleistocene permafrost of the Kolyma river lowland. Dokl. Biol. Sci. 2018, 480, 100–102. [Google Scholar] [CrossRef] [PubMed]
- Yashina, S.; Gubin, S.; Maksimovich, S.; Yashina, A.; Gakhova, E.; Gilichinsky, D. Regeneration of whole fertile plants from 30,000-y-old fruit tissue buried in Siberian permafrost. Proc. Natl. Acad. Sci. USA 2012, 109, 4008–4013. [Google Scholar] [CrossRef] [PubMed]
- Kirschvink, J.L.; Weiss, B.P. Mars, panspermia, and the origin of life: Where did it all begin? Palaeontol. Electron. 2002, 4, 8–15. Available online: http://palaeo-electronica.org/paleo/2001_2/editor/mars.html (accessed on 30 July 2021).
- Melosh, H.J. Exchange of meteorites (and life?) between stellar systems. Astrobiology 2003, 3, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Schulze-Makuch, D.; Irwin, L.N.; Fairén, A.G. Drastic environmental change and its effects on a planetary biosphere. Icarus 2013, 225, 775–780. [Google Scholar] [CrossRef]
- Wickramasinghe, N.C.; Wallis, J.; Wallis, D.H.; Schild, R.E.; Gibson, C.H. Life-bearing primordial planets in solar vicinity. Astrophys. Space Sci. 2012, 341, 295–299. [Google Scholar] [CrossRef]
- Wallis, M.K.; Wickramasinghe, N.C. Interstellar transfer of planetary microbiota. Mon. Not. R. Astron. Soc. Lett. 2004, 348, 52–61. [Google Scholar] [CrossRef]
- Wickramasinghe, J.T.; Wickramasinghe, N.-C.; Napier, W.M. Comets and the Origin of Life; World Scientific: Singapore, 2010. [Google Scholar]
- Hartmann, W.K.; Davis, D.R. Satellite-sized planetesimals and lunar origin. Icarus 1975, 24, 504–515. [Google Scholar] [CrossRef]
- Jacobsen, S.B. The Hf-W isotopic system and the origin of the Earth and Moon. Annu. Rev. Earth Planet. Sci. 2005, 33, 531–570. [Google Scholar] [CrossRef]
- Benz, W.; Slattery, W.L.; Cameron, A.G.W. Collisional stripping of Mercury´s mantle. Icarus 1988, 74, 516–528. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schulze-Makuch, D.; Fairén, A.G. Evaluating the Microbial Habitability of Rogue Planets and Proposing Speculative Scenarios on How They Might Act as Vectors for Panspermia. Life 2021, 11, 833. https://doi.org/10.3390/life11080833
Schulze-Makuch D, Fairén AG. Evaluating the Microbial Habitability of Rogue Planets and Proposing Speculative Scenarios on How They Might Act as Vectors for Panspermia. Life. 2021; 11(8):833. https://doi.org/10.3390/life11080833
Chicago/Turabian StyleSchulze-Makuch, Dirk, and Alberto G. Fairén. 2021. "Evaluating the Microbial Habitability of Rogue Planets and Proposing Speculative Scenarios on How They Might Act as Vectors for Panspermia" Life 11, no. 8: 833. https://doi.org/10.3390/life11080833
APA StyleSchulze-Makuch, D., & Fairén, A. G. (2021). Evaluating the Microbial Habitability of Rogue Planets and Proposing Speculative Scenarios on How They Might Act as Vectors for Panspermia. Life, 11(8), 833. https://doi.org/10.3390/life11080833






