Abstract
Insomnia is a common problem in the general population. To treat insomnia, medication therapies and insomnia-related cognitive-behavioral interventions are often applied. The aim of the present study was to investigate the influence of acceptance and commitment therapy (ACT) on sleep quality, dysfunctional sleep beliefs and attitudes, experiential avoidance, and acceptance of sleep problems in individuals with insomnia, compared to a control condition. A total of 35 participants with diagnosed insomnia (mean age: 41.46 years old; 62.9% females) were randomly assigned to the ACT intervention (weekly group therapy for 60–70 min) or to the active control condition (weekly group meetings for 60–70 min without interventional and psychotherapeutic character). At baseline and after eight weeks (end of the study), and again 12 weeks later at follow-up, participants completed self-rating questionnaires on sleep quality, dysfunctional beliefs and attitudes about sleep, emotion regulation, and experiential avoidance. Furthermore, participants in the intervention condition kept a weekly sleep log for eight consecutive weeks (micro-analysis). Every morning, participants completed the daily sleep log, which consisted of items regarding subjective sleep duration, sleep quality, and the feeling of being restored. Sleep quality, dysfunctional beliefs and attitudes towards sleep, emotion regulation, and experiential avoidance improved over time, but only in the ACT condition compared to the control condition. Improvements remained stable until follow-up. Improvements in experiential avoidance were related to a favorable change in sleep and cognitive-emotional processing. Micro-analyses showed that improvements occurred within the first three weeks of treatment. The pattern of results suggests that ACT appeared to have improved experiential avoidance, which in turn improved both sleep quality and sleep-related cognitive-emotional processes at longer-term in adults with insomnia.
1. Introduction
A healthy lifestyle is associated with sufficient and restoring sleep [1]. However, in the general population, insomnia is common—the prevalence rates of insomnia in the general population may range from 9% to 15% [2], from 20% to 40% [3], or from 5% to 25% [4], depending on the underlying definition of and methodology to assess insomnia. Overall, about 30% of the general population reported having experienced insomnia at some point in their life [5]. Importantly, insomnia is related to higher risks of traffic accidents [6,7,8,9,10], more workplace absenteeism, impaired work performance [11,12,13], and to physiological issues such as diabetes [14] and cardiovascular diseases [15]. Further, insomnia predicted incidences of psychiatric disorders such as depression, anxiety, and alcohol abuse [16].
Following the International Classification of Sleep Disorders (ICSD-3) [17] chronic insomnia disorder involves the following symptoms: difficulty in initiating or maintaining sleep, early morning awakening with inability to return to sleep, and resistance to going to sleep. Daytime consequences include the feeling of not being restored, fatigue, excessive daytime sleepiness, and decreased attention, memory, and concentration. These impairments should occur at least three times per week for at least three months.
To treat insomnia, both pharmacological and non-pharmacological interventions are employed. Both the American Sleep Association (https://www.sleepassociation.org/sleep-treatments/cognitive-behavioral-therapy/ (accessed on 7 January 2021)) and the European Sleep Association [18] recommend the specific cognitive-behavioral therapy for insomnia (CBT-i) as first-line treatment. CBT-i can be delivered as group-therapy interventions [19,20] or as internet-delivered interventions [21,22]. Systematic reviews and meta-analyses confirmed that CBT-i improved sleep parameters, without adverse outcomes and side-effects [23]. Further non-pharmacological treatments involve exercise-based and psychotherapeutic interventions. Systematic reviews and meta-analyses showed that exercise-based interventions compared to no interventions led to improvements in some specific sleep parameters [24,25,26,27]. Likewise, mindfulness meditation improved subjective sleep among individuals with insomnia [28] and without insomnia [29]. For pharmacological treatment, compared to no treatment, placebo effects had a considerable impact on subjective improvements [30]. Consequently, alternative and psychotherapeutic interventions are considered. CBT interventions appeared to lead to medium [31] and clinically meaningful effect sizes [23], although risks of relapses were also reported [32]. Here, we considered acceptance and commitment therapy (ACT) as an intervention to treat insomnia.
Acceptance and commitment therapy (ACT) is a psychotherapeutic intervention based on the principles of cognitive-behavioral therapy (CBT) [33,34,35,36,37,38,39,40,41]. Briefly, key elements of ACT include (1) acceptance of all kind of emotions and thoughts as processes of the mind without acting out these emotions (e.g., sadness, anger, disappointments), without taking thoughts as ultimate truths (e.g., “I’m worthless!”; “I’m not worth being loved!”); or for short: “I do not avoid such emotions and cognitions, but I accept them as products of the mind.” (acceptance). The opposite of accepting unpleasant cognitions and emotions is “experiential avoidance.” Following others [42,43], experiential avoidance is the tendency to avoid unpleasant inner states—unpleasant feelings, thoughts, memories, images, impulses, and bodily perceptions are avoided. The disadvantage of such avoidance behavior is that this behavior needs cognitive-emotional resources. This leads to two consequences: First, cognitive-emotional resources are continuously entangled with unpleasantness, and second, cognitive-emotional resources are not available to pursue core values and to pursue what is more important in life. Accepting unpleasant feelings and thoughts, that is, giving-up to avoid inner experiences (experiential avoidance) means accepting unpleasant feelings and thoughts as transient processes of the mind without acting out these emotions and without taking such feelings and thoughts as ultimate truths; (2) identifying core values, i.e., what is really important in one’s life, being committed to these values and related behavior?—“The more I’m aware of my core values, the more my behavior is oriented towards the achievement of these values.”; (3) “focusing my behavior to achieve values equals to be committed both to the values and related behavior” (commitment); and (4) “ACT is both the acronym of the intervention and the chief focus on doing, being active, and behaving actively to move toward my core values.” Authors investigating the underlying psychological mechanisms of ACT underscore the importance of acceptance and not avoiding experiences anymore (experiential avoidance), psychological flexibility, and staying committed to the core values and their actions to turn values into action [33,34,35,36,37,38,39,40,41].
Results from meta-analyses and systematic reviews showed that ACT is always a valuable psychotherapeutic intervention for major depressive disorders [44], anxiety disorder [45], posttraumatic stress disorders [46], and insomnia [47] when compared to no-treatment conditions. In a single case study, ACT impacted favorably on sleep quality [32]. In two previous pilot studies ACT interventions for eight weeks improved subjective sleep quality among four and 15 individuals with insomnia [48,49]. An ACT intervention improved sleep patterns in adults with insomnia (n = 43), compared to a wait-list condition (n = 40) [50]. ACT-interventions improved subjective sleep among ten individuals with insomnia poorly responding to CBT-interventions before [51]. Importantly, improvements appeared to remain stable over a six-month [50] and nine-month period [52]. To explain the favorable impact of ACT on insomnia, aspects of mindfulness [53] and decreased experiential avoidance [54] appeared to decrease psychological arousal.
The current study expanded upon previous research in four ways—First, we introduced an active control condition, i.e., participants in the control condition gathered in small groups once a week in the study center. In doing so, we were able to partial out possible improvements in the ACT condition due to a mere social context. Second, participants in the ACT condition completed a daily log for eight consecutive weeks. In doing so, we were able to observe, if and at which time points ACT improved dimensions of sleep patterns and cognitive-emotional processes. Third, the follow-up occurred 12 weeks after the end of the intervention, which allowed an estimate, if and to what extent ACT interventions could impact cognitive-emotional processes in the longer term. Fourth, we associated experiential avoidance, a key concept to explain ACT-related improvements, with dimensions of sleep quality, emotion regulation, dysfunctional beliefs and attitudes about sleep, and acceptance of sleep problems. To this end, 35 adults with insomnia were randomly assigned to the ACT intervention and to the active control condition, and dimensions of sleep quality and cognitive-emotional processes were self-assessed at baseline, eight weeks later at the end of the study, and again 12 weeks later at follow-up.
The following three hypotheses and one research question were formulated: first, following others [32,47,48,49,50,51], we expected improvements on subjective sleep and cognitive-emotional processes in the intervention-/ACT-condition, compared to an active control condition. Second, following others [34,40,54,55,56] we assumed that improvements in experiential avoidance would be associated with favorable changes in sleep quality and cognitive-emotional processes. Third, following Lappalainen et al. [50] and Daly-Eichenhardt et al. [52], we expected that improvements would remain stable until follow-up 12 weeks after study completion. We treated the study question as exploratory, asking at which time point (week) of the intervention improvements in sleep patterns could be observed, compared to baseline. Answering these research questions is of clinical importance because in clinical practice it may complement the CBT-i, which, although generally effective, may not be effective in all individuals with insomnia. Results are also of practical importance because ACT may particularly improve experiential avoidance, which could be associated with a broad range of further favorable cognitive-emotional processes.
2. Methods
2.1. Study Procedure
Individuals with clinically diagnosed insomnia from the Sleep Disorders Research Center of the Kermanshah University of Medical Sciences (Kermanshah, Iran) were approached between January 2019 and August 2020 to participate in the present interventional study. Eligible participants were fully informed about the study and the secure and anonymous data handling. Thereafter, they signed the written informed consent and they were randomly assigned either to the intervention condition (acceptance and commitment therapy; ACT; see Section 2.6) or to an active control condition (see Section 2.7). At the beginning, eight weeks later at the end of the study, and again 12 weeks later at follow-up participants completed a booklet of questionnaires covering sociodemographic and sleep-related information (see Section 2.5). Further, participants in the ACT condition were keeping a weekly sleep log for eight consecutive weeks (see Section 2.5.3). An experienced and certified psychotherapist on ACT performed the group interventions of ACT once a week for 60–70 min. Meetings of participants in the active control condition were identical as regards frequency and duration as meetings in the ACT condition (see Section 2.7). The Kermanshah University of Medical Science Ethics Committee (IR.KUMS.REC.1396.478) approved the study, which was performed in accordance with the seventh and last revision [57] of the Declaration of Helsinki. Two previous feasibility and pilot studies at a small scale with no control condition [58,59] pawed the grounds for the present study.
2.2. Participants
A total of 45 individuals with insomnia were approached. Inclusion criteria were (1) suffering from insomnia, based on self-reports and on a structured clinical interview for DSM-5 psychiatric disorders [60]; (2) age between 18 years old and 60 years old; (3) willing and able to comply with the study conditions; and (4) signed written informed consent. Exclusion criteria were (1) psychiatric disorders such as major depressive disorders, bipolar disorders, cognitive impairments, or substance use disorder with insomnia as a consequence of the main psychiatric disorder; to exclude psychiatric disorders the same structured clinical interview for DSM-5 psychiatric disorders [60] was employed; (2) insomnia due to shift work or family issues such as small children, children with mental and/or physical impairments that would explain the emergence and maintenance of insomnia; (3) restless legs syndrome or sleep-disordered breathing, as assessed via the clinical interview; (4) intake of medications with sleep- and mood-altering effects; and (5) undergoing interventions such as psychotherapy, including CBTi, relaxation techniques or physical exercising to improve sleep problems.
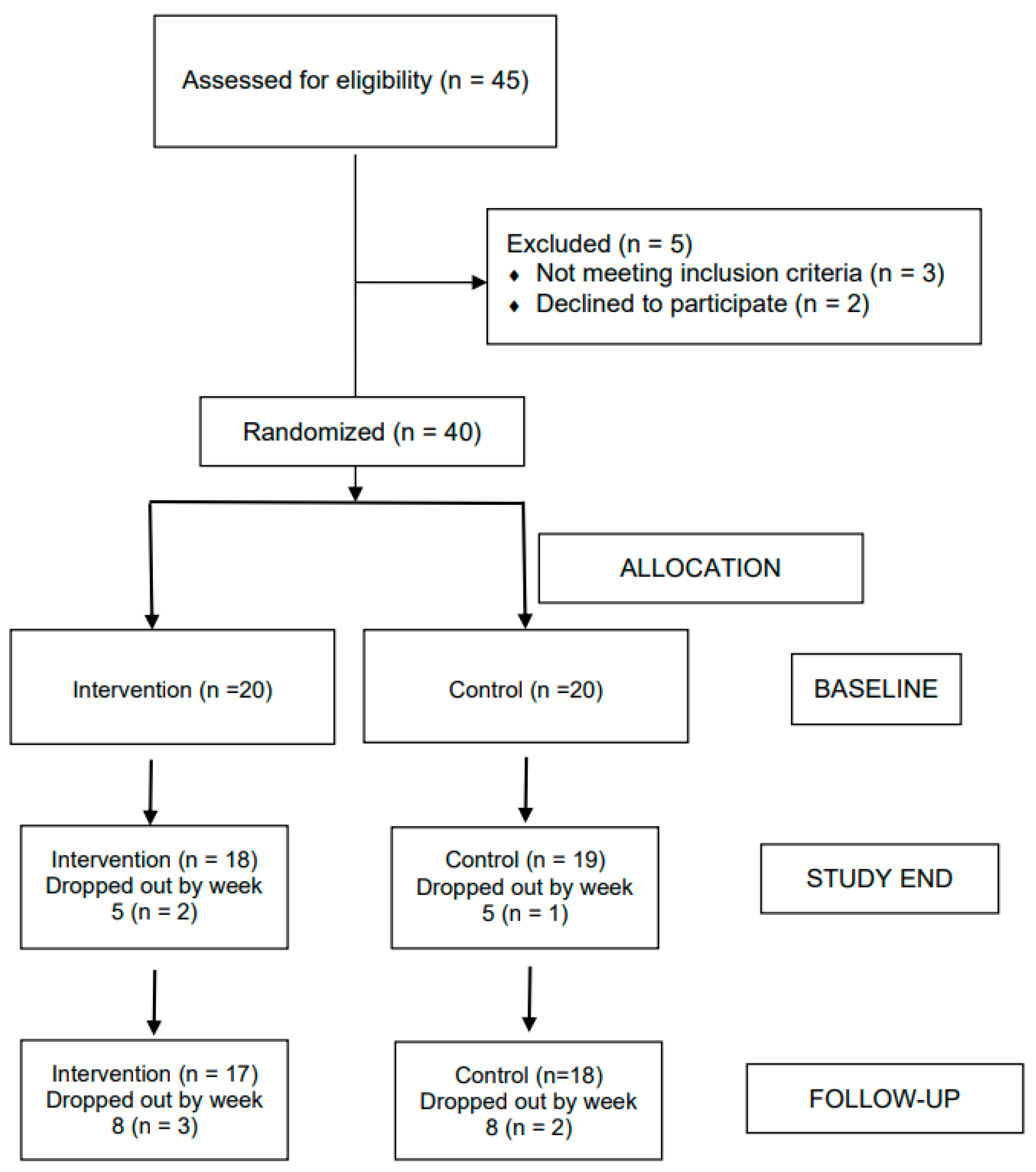
Of the 45 individuals approached, three did not meet the inclusion criteria, and two declined to participate. Hence, 40 individuals (88.89%) were randomly assigned to either the ACT intervention or to the active control condition (see Figure 1). A total of 35 participants accomplished the study from baseline to follow-up. All outcome analyses were performed on completer data.
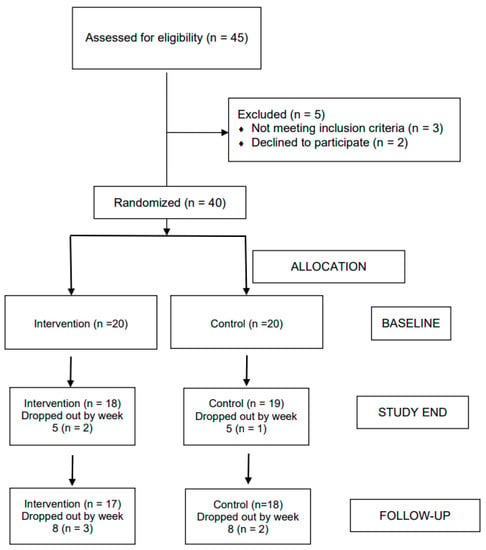
Figure 1.
CONSORT diagram showing the flow of participants through each stage.
2.3. Sample Size Calculation
The sample size calculation was performed with G*Power® [61]. Based on previous results of two pilot studies [58,59], the following parameters were defined: effect size: partial eta-squared: 0.08; Cohen’s f for ANOVAs: 0.29; alpha error probability: 0.05; power: 0.95; number of groups: 2; and number of measurements: 3. These parameters yield a total sample size of 32. However, to allow for dropouts, the sample size was set at 40 participants.
2.4. Randomization
As described elsewhere [62,63,64], randomization was accomplished using the software randomization.com to create a list to assign 40 participants randomly to one of the two study conditions. Thereafter, a psychologist not otherwise involved in the study managed the assignments.
2.5. Measures
2.5.1. Experiential Avoidance
The Experiential Avoidance Questionnaire (EAQ) [55] was used, which consists of 10 items to assess experiential avoidance. Typical items include “I am afraid of my feelings.” and “My thoughts and feelings mess up my life.” Answers are given on 7-point Likert scales ranging from 1 (= never) to 7 (= always). Higher sum scores reflect a higher degree to accept also unpleasant experiences (emotions, cognitions), and accordingly, a lower degree of avoidance (Cronbach’s alpha = 0.85).
2.5.2. Subjective Sleep: Pittsburgh Sleep Quality Index (PSQI)
As described elsewhere [62], the Pittsburgh Sleep Quality Index PSQI [65] is a self-report scale completed in five minutes. It consists of 19 items and contains seven subscales (subjective sleep quality, sleep latency, sleep duration, sleep efficiency, sleep disturbance, sleeping medication, and daytime dysfunction), each weighted equally on a scale from 0 to 3, with higher scores indicating poorer sleep quality. The seven components are then summed to obtain an overall PSQI score, ranging from 0 (good sleep quality) to 21 (poor sleep quality). Total scores of ≥5 reflect poor sleep, associated with considerable sleep complaints. The Farsi versions of PSQI have been validated for adults [66], older adults [67], and adolescents [68] (Cronbach’s alpha = 0.85).
2.5.3. Sleep Log
Participants in the intervention condition completed every morning a sleep log for eight consecutive weeks. Items were taken from a widely used manual on self-treatment of insomnia [69]. The items were (1) time to go to bed—answer in h and min (e.g., 22.30 h); (2) sleep onset duration—answer in min; (3) the number of awakenings after sleep onset; (4) duration of time awake after sleep onset; (5) wake-up time; (6) sleep quality—answer based on a 9-point Likert scale: 1 = very poor sleep quality; 9 = very good sleep quality; (7) feeling of being restored—answer based on a 9-point Likert scale: 1 = not at all restored; 9 = completely restored. Total sleep time (in h and min) was calculated based on the answers 1 to 5. Data from five weekdays were merged to overall scores for one week.
2.5.4. Dysfunctional Beliefs and Attitudes about Sleep (DBAS)
To assess dysfunctional pre-sleep beliefs and attitudes, participants completed the Dysfunctional Beliefs and Attitudes about Sleep (DBAS) questionnaire [70]. The questionnaire consists of 10 items. Typical items include “I need 8 h of sleep to feel refreshed and function well during the day.” and “When I don’t get proper amount of sleep on a given night, I need to catch up on the next day by napping or on the next night by sleeping longer.” Answers are given on 10-point Likert scales, ranging from 1 (= not at all true) to 10 (= completely true), with higher sum scores reflecting more pronounced dysfunctional beliefs and attitudes about sleep (Cronbach’s alpha = 0.79).
2.5.5. Sleep Problem Acceptance Questionnaire (SPAQ)
To assess the acceptance to suffer from sleep problems, participants completed the Sleep Problem Acceptance Questionnaire (SPAQ) [71]. The questionnaire consists of eight items. Typical items are “Although things have changed, I am living a normal life despite my sleeping problems.” and “I live a full life even though I have sleeping problems.” Answers are given on 7-point Likert scales, ranging from 1 (= not at all true) to 10 (= completely true), with higher sum scores reflecting a higher acceptance to suffer from sleep problems (Cronbach’s alpha = 0.85).
2.5.6. Difficulties in Emotion Regulation Scale (DERS)
To assess difficulties in emotion regulation, participants completed the Difficulties in Emotion Regulation Scale (DERS) [72]. The questionnaire consists of 36 items. Typical items include “I know my feelings.” and “I pay attention to how I feel.” Answers are given on 5-point Likert scales, ranging from 1 (= not at all true) to 5 (= completely true), In the present study, we considered the overall sum score—a higher sum score reflects a higher degree to regulate their emotions. (Cronbach’s alpha = 0.93).
2.6. Intervention: Acceptance and Commitment Therapy (ACT)
The ACT intervention consisted of eight weekly group sessions lasting for 60–70 min. A group consisted of eight to nine participants. For the content and the structure of the sessions, we followed well-established therapy manuals [42,43,56,73,74]. A clinical psychologist and certified psychotherapist in ACT (AZ) was responsible for the ACT sessions.
Table 1 provides the content of the eight sessions in ACT.

Table 1.
Overview of the content of the eight acceptance and commitment therapy (ACT) sessions.
2.7. Active Control Condition
As described extensively elsewhere [75,76,77,78,79,80], participants in the control condition met in groups with a staff member and social worker once a week for eight consecutive weeks for about 60–70 min. They had group discussions on daily activities and daily problems. The control condition could not be considered as a bona fide intervention [75,76,77,78,79,80], given that the sessions with the staff member and social worker excluded treatment elements that were truly intended to be therapeutic [81]. Participants were just encouraged to exchange daily life experiences. Blood pressure was also checked once per week. The active control condition was not intended to be an active therapy but to control for placebo effects in the intervention condition.
2.8. Statistical Analysis
A series of t-tests and X2-tests was performed to compare sociodemographic and sleep-related data at baseline between participants in the intervention and in the control condition. At baseline, and compared to the active control condition, participants in the intervention-/ACT-condition reported subjectively lower sleep complaints (statistically significantly lower PSQI scores; see Table 2). Given this evidence, PSQI at baseline was introduced as the covariate. Accordingly, a series of ANCOVAs for repeated measures was performed with the factors time (baseline, study end, follow-up), group (intervention versus control condition) and the time by group interaction; subjective sleep quality, dysfunctional beliefs and attitudes about sleep, sleep problems acceptance, difficulties in emotion regulation, and experiential avoidance were dependent variables. In case of deviation from sphericity, ANCOVAs were computed using Greenhouse–Geisser corrected degrees of freedom, though the original degrees of freedom are reported with the relevant Greenhouse–Geisser epsilon value (ε). Post-hoc tests were performed with Bonferroni–Holm corrections for p-values. For t-tests, Cohen’s d effect sizes are reported. For F-tests, partial eta-squared’s () effect sizes are reported. Cut-off values for Cohen’s d’s were d < 0.19 = trivial effect size (T); 0.20 < d < 0.49 = small effect size (S); 0.50 < d < 0.79 = medium effect size (M); d > 0.80 = large effect size (L). Cut-off values for partial eta-squared were: < 0.019 = trivial effect size (T); < 0.059 = small effect size (S); 0.06 < < 0.139 = medium effect size (M); > 0.14 = large effect size (L).

Table 2.
Descriptive and inferential statistical overview of sociodemographic, sleep, and psychological functioning at baseline, separately for individuals with insomnia in the intervention (n = 17) and the control (n = 18) conditions.
For micro-analysis of weekly change in total sleep time, sleep quality, and the feeling of being restored, we followed Becker [82] and compared standard mean changes with Cohen’s d’s effect sizes.
With a series of Pearson’s correlations associations between improvements in experiential avoidance (that is, less avoidance of unpleasant experiences at the end of the study after the intervention) and sleep quality, dysfunctional beliefs and attitudes about sleep, sleep problems acceptance, and difficulties in emotion regulation were calculated.
All statistical computations were performed with SPSS® 25.0 (IBM Corporation, Armonk, NY, USA) for Apple Mac®.
3. Results
3.1. Sociodemographic and Sleep-Related Information at Baseline
Table 2 provides the descriptive and statistical overview of sociodemographic and sleep-related information at baseline between the participants in the intervention and control condition. Statistical indices are not repeated in the text anymore.
Compared to participants in the control condition, participants in the intervention condition reported higher educational levels, a higher acceptance of sleep problems, and a lower subjective sleep disturbance (lower PSQI score). Given this evidence, the PSQI score at baseline was introduced as the covariate. There were no descriptive and statistically significant mean differences for age, sex distribution, current job position, civil status, subjective sleep quality, dysfunctional beliefs and attitudes about sleep, difficulties in emotion regulation, and experiential avoidance.
3.2. Changes in Subjective Sleep Quality, Dysfunctional Beliefs and Attitudes about Sleep, Sleep Problems Acceptance, Difficulties in Emotion Regulation, and Experiential Avoidance over Time between and within the Intervention and Control Conditions
Table 3 and Table 4 provide the descriptive and inferential statistical overview of subjective sleep quality, dysfunctional beliefs and attitudes about sleep, sleep problems acceptance, difficulties in emotion regulation, and experiential avoidance over time between and within the intervention and control conditions and always controlling for PSQI baseline scores.

Table 3.
Descriptive statistical overview of sleep-related indices and psychological functioning at baseline, at the end of the eight-week study and at follow-up after 12 weeks, separately for individuals with insomnia in the intervention (n = 17) and the control (n = 18) conditions.

Table 4.
Inferential statistical indices of sleep-related indices and psychological functioning, always controlling for PSQI scores at baseline.
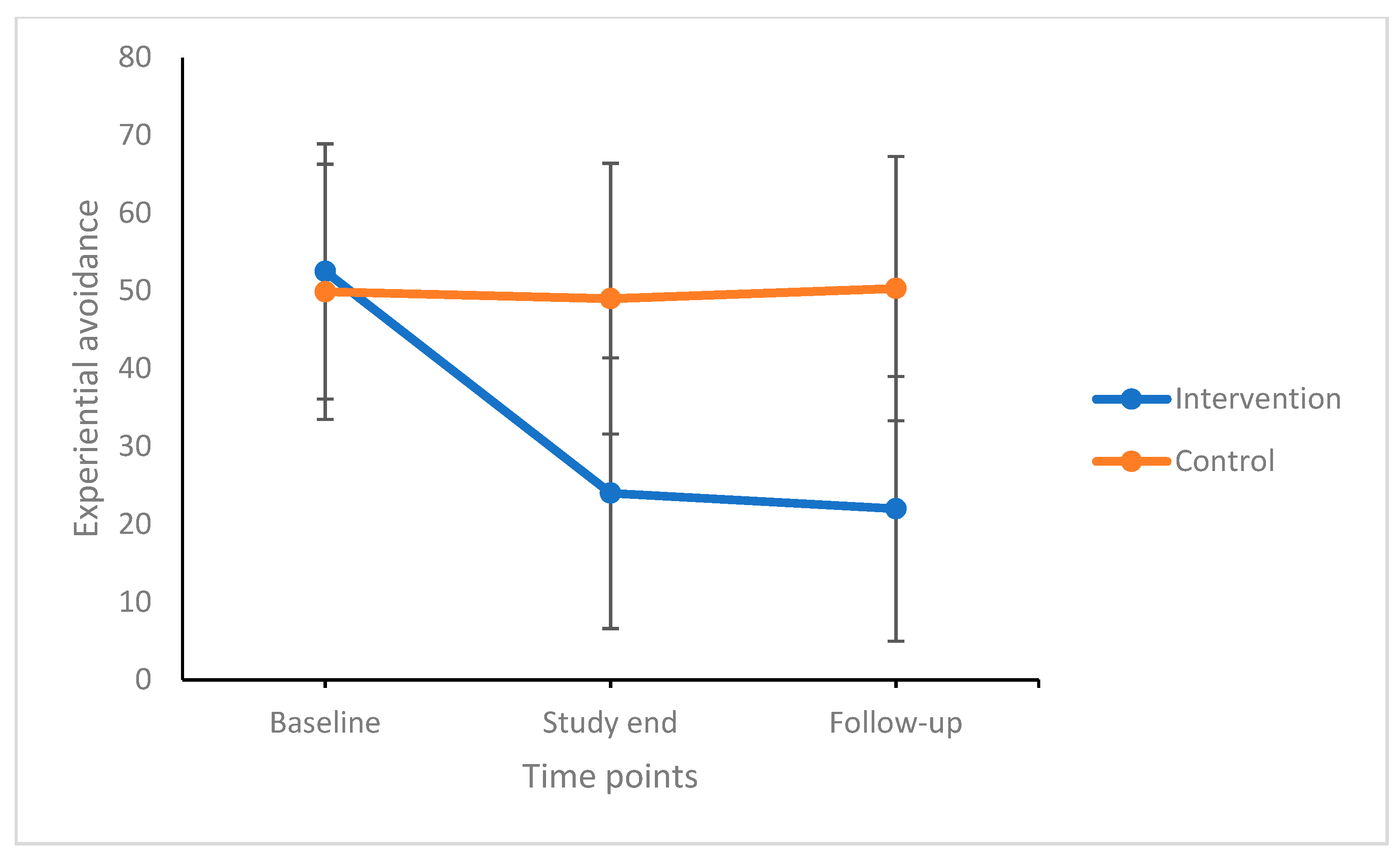
Experiential avoidance and dysfunctional beliefs and attitudes about sleep decreased over time (the significant factor was time). Subjective sleep quality, sleep problems acceptance, difficulties in emotion regulation did not statistically change over time. Significant time by group interactions showed that experiential avoidance, dysfunctional beliefs and attitudes about sleep, subjective sleep quality, sleep problems acceptance, and difficulties in emotion regulation decreased over time in the intervention-/ACT condition but not in the active control condition. Post-hoc comparisons with Bonferroni–Holm corrections for p-values showed that compared to the control condition, participants in the intervention condition reported more favorable scores in all dimensions (the significant factor was group). Importantly, improvements remained stable 12 weeks after the end of the intervention. Figure 2 illustrates mean changes over time between and within groups for experiential avoidance.
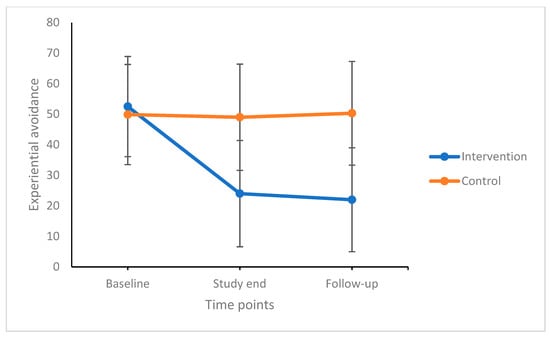
Figure 2.
Experiential avoidance decreased over time, but more so in the intervention condition compared to the control condition. Improvements at the end of the study remained stable until follow-up 12 weeks later. Points are means and bars are standard deviations.
3.3. Micro-Analysis; Weekly Changes in Total Sleep Time, Sleep Quality, and the Feeling of Being Restored in the Intervention Group
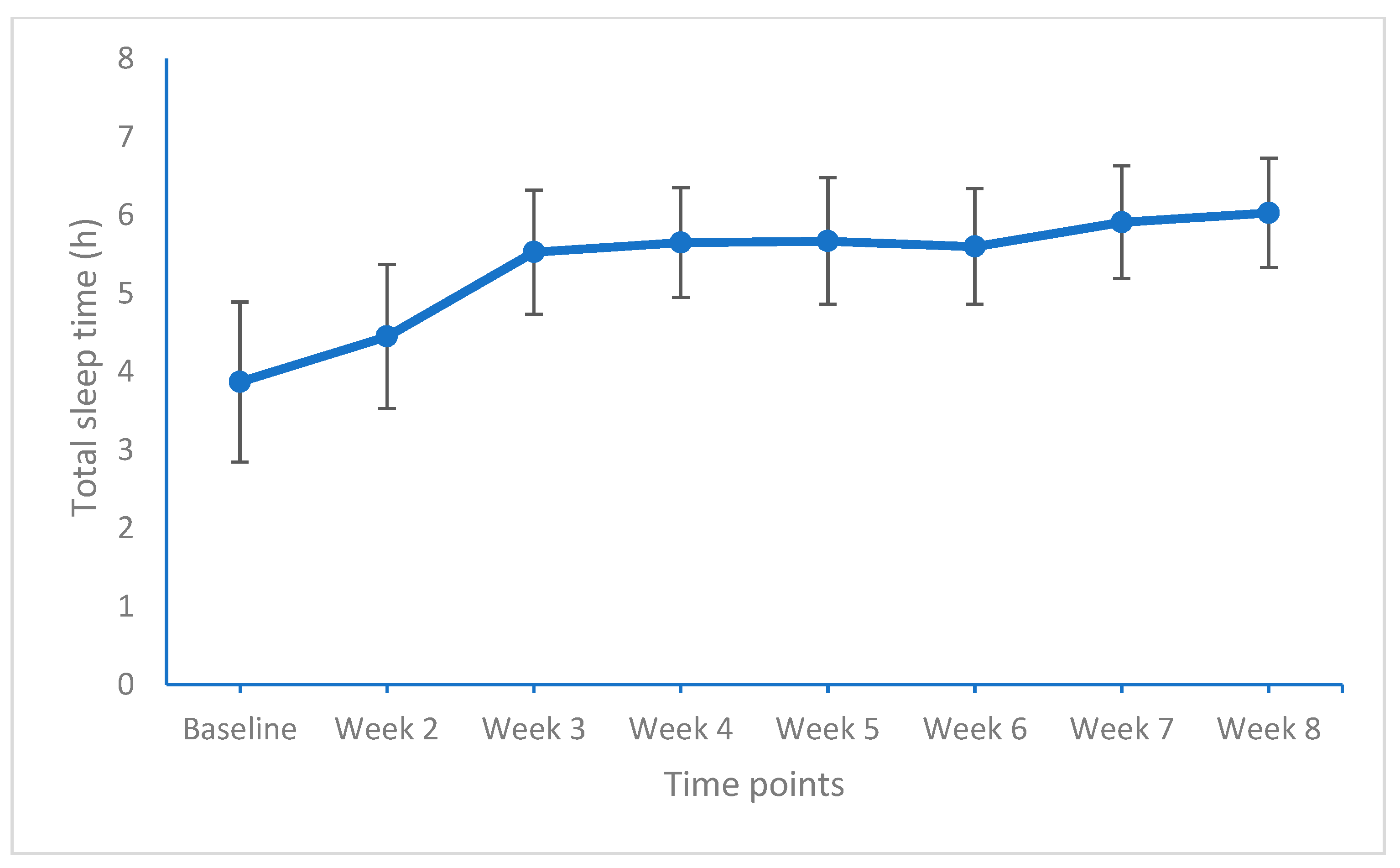
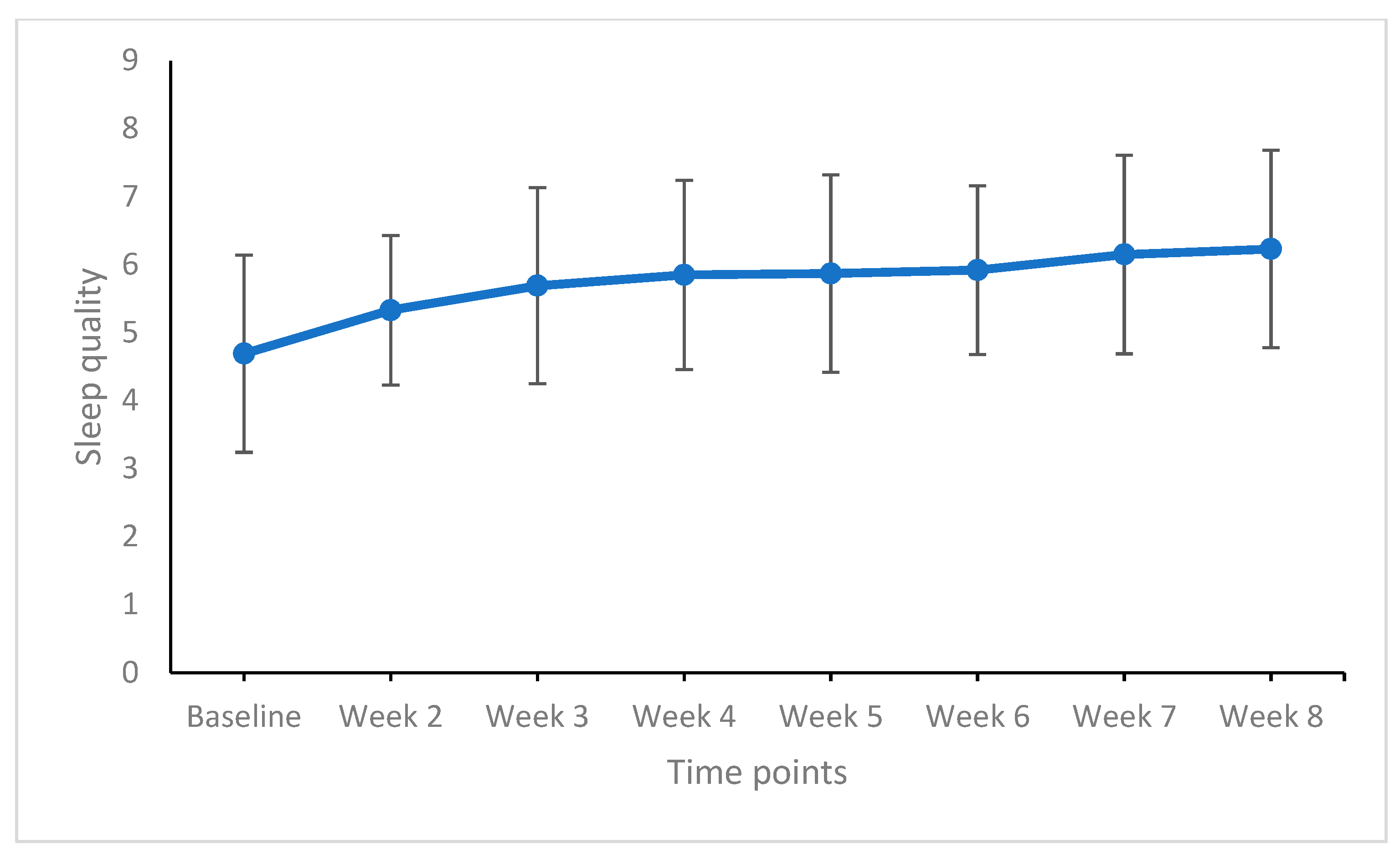
Table 5 provides the effect sizes of weekly comparisons of sleep duration, sleep quality, and the feeling of being restored, and Figure 3, Figure 4 and Figure 5 show the graphical solutions.

Table 5.
Descriptive and inferential statistical overview of sleep diary data over all eight weeks in the intervention condition (n = 17).
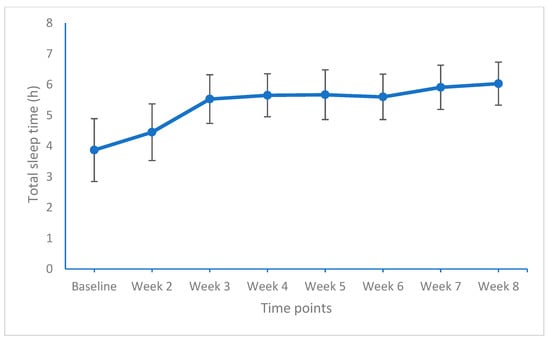
Figure 3.
Total sleep time (h) from baseline to week 8 (end of the study). Points are means and bars are standard deviations.
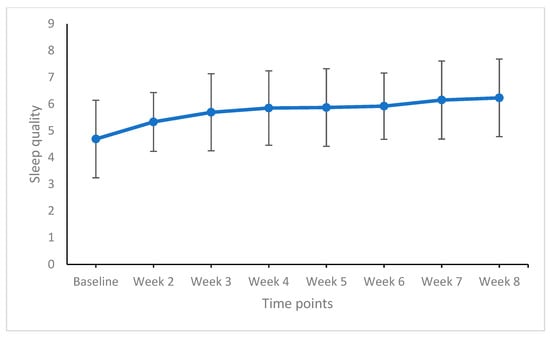
Figure 4.
Sleep quality from baseline to week 8 (end of the study). Points are means and bars are standard deviations. Higher means reflect a higher sleep quality.
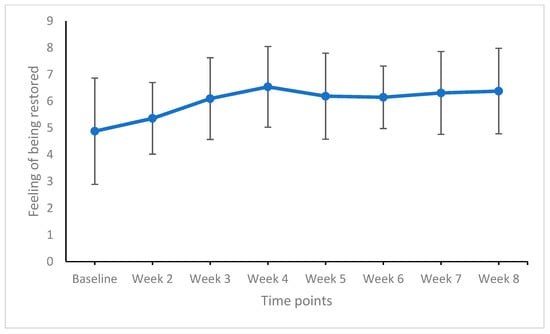
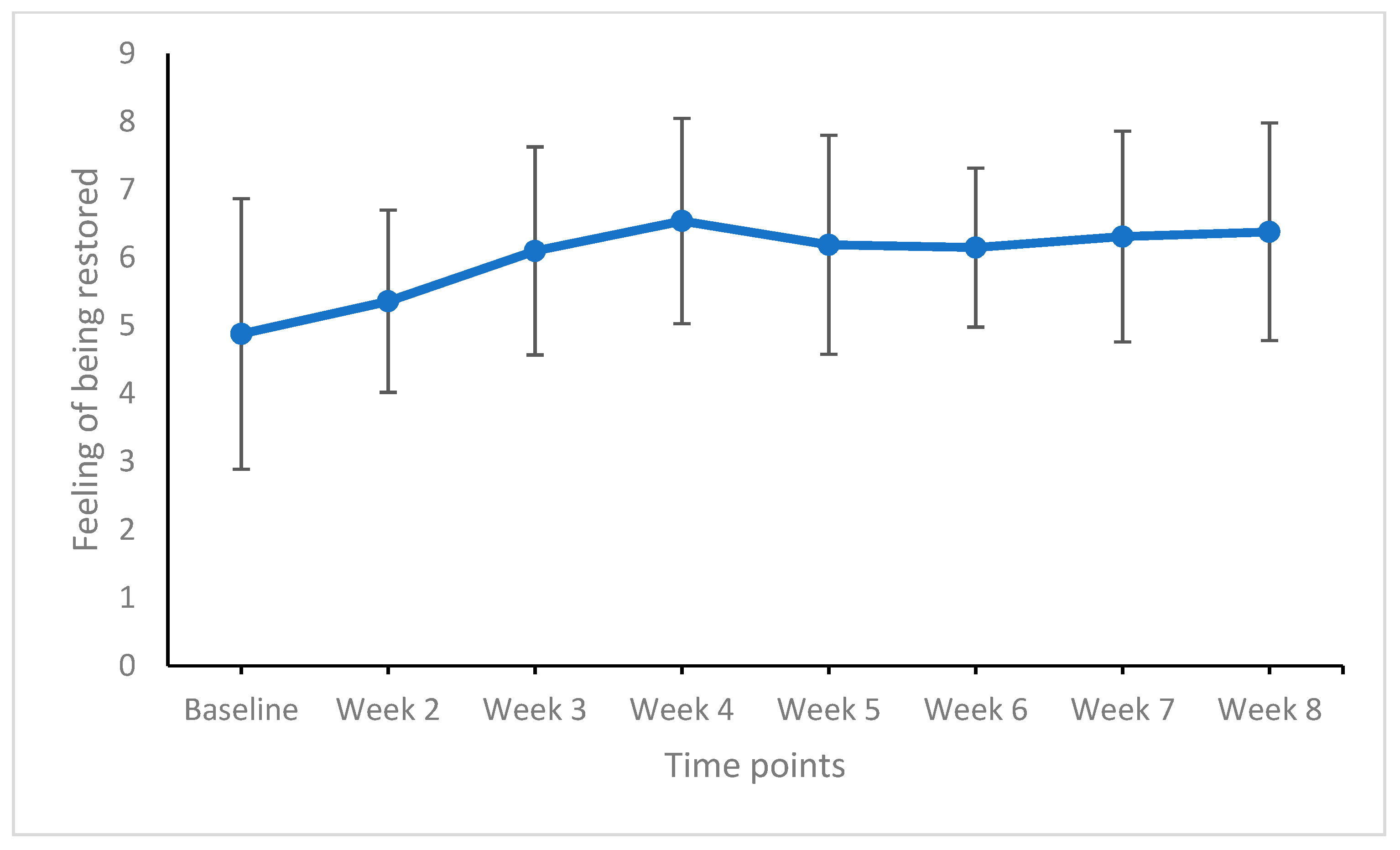
Figure 5.
Feeling of being restored from baseline to week 8 (end of the study). Points are means and bars are standard deviations. Higher means reflect a higher feeling of being restored.
For total sleep time, significant improvements (medium and large effect sizes) were observed from baseline to week 2, from week 2 to week 3, and again from week 6 to week 7 (medium effect size).
For subjective sleep quality, there were no significant improvements from week to week (trivial to small effect sizes). Subjective sleep quality, however, improved from baseline to weeks 3 to 8 (always large effect sizes).
For the feeling of being restored, significant improvements were observed from baseline to week 2 and from week 2 to week 3 (medium effect sizes). From week 3, week-to-week comparisons yielded no significant improvements (trivial to small effect sizes).
Overall, significant improvements were found within the first three weeks after the beginning of the intervention.
3.4. Changes in Experiential Avoidance and Associations with Sleep Quality, Dysfunctional Beliefs and Attitudes about Sleep, Sleep Problems Acceptance, and Difficulties in Emotion Regulation at the End of the Study in the Intervention Condition
To improve sleep, the psychotherapeutic intervention was ACT; to operationalize ACT, we employed the Experiential Avoidance Questionnaire (AAQ). Improvements in avoiding unpleasant experiences (that is, lower scores in experiential avoidance) should be associated with improvements in sleep quality, dysfunctional beliefs and attitudes about sleep, sleep problems acceptance, and difficulties in emotion regulation at the end of the study.
A higher change in experiential avoidance over time, that is, a lower attitude to avoid unpleasant experiences, was associated with lower dysfunctional beliefs and attitudes about sleep (DBAS, r = −0.45, p < 0.05), fewer difficulties in emotion regulation (DERS; r = −0.14, ns), a higher degree to accept sleep problems (SPAQ; r = −0.14, ns), and a higher sleep quality (PSQI; r = −0.59, p < 0.05).
4. Discussion
The key findings of the present study were that among a sample of individuals with insomnia an eight-week acceptance and commitment therapy (ACT) group intervention improved experiential avoidance, sleep quality, dysfunctional beliefs and attitudes about sleep, sleep problems acceptance, and difficulties in emotion regulation, as compared to an active control condition. Improvements were above all observed within the first three weeks of intervention. Further, improvements remained stable for further 12 weeks until follow-up. The present pattern of results was comparable and fit well with those reported recently in a systematic review on the influence of ACT on insomnia and sleep quality [47]. However, the present results expand upon previous research in that ACT interventions affected the sleep quality and the underlying dysfunctional cognitive-emotional processes. As such, it is conceivable that such improvements in cognitive-emotional processes may also explain the therapeutic effect in the long-term.
Three hypotheses and one research question were formulated and each of them is considered as follows.
With the first hypothesis, we assumed that subjective sleep and cognitive-emotional processes would improve over time in the ACT condition, compared to the active control condition, and data did support this. Accordingly, the present data are in accordance with previous results [32,47,50]. However, the present results expand upon previous studies in three ways—(1) improvements at the end of the interventions remained stable for 12 additional weeks until follow-up; (2) unlike previous studies [32,47,50,58,59], we employed an active control condition to partial out possible treatment effects due to the social context of group therapy; and (3) the micro-analysis allowed a fine-grained measurement and assessment of sleep-related improvements.
With the second hypothesis, we expected that improvements in experiential avoidance would be associated with favorable changes in sleep quality and cognitive-emotional processes, and data did confirm this. Accordingly, the present data both replicated previous findings [34,40,54,55,56] and expanded on previous results in three ways—(1) we employed a questionnaire to assess experiential avoidance, which by definition, is one of the key concepts of ACT, namely, accepting prevalently unpleasant feelings and dysfunctional thoughts, or the other way around, not avoiding such unpleasant feelings and dysfunctional thoughts anymore. Table 3 and Table 4 display that experiential avoidance improved over time in the ACT condition but not in the active control condition, thereby suggesting that improvements in experiential avoidance were associated with ACT-related interventions; 2) following the transdiagnostic approach [83,84,85,86,87,88], it appears plausible that improvements in sleep quality are associated with favorable changes in the underling cognitive-emotional processes; and (3) improvements in sleep quality and quantity were above all observed within the first three weeks of treatment.
With the third hypothesis, we assumed that ACT-related improvements on sleep remained stable also for 12 weeks later at follow-up, and again data did confirm this. Accordingly, the present results both replicated [50,52] and expanded upon previous studies in the following two ways—(1) improvements in the ACT-condition were always compared to an active control condition, and (2) we measured if and to what extent experiential avoidance remained stable over time as a proxy of the learning process and interiorization of the acquired ACT-related skills during the intervention.
Lastly, we took the study question as exploratory, asking at which time point (week) of the intervention improvements in sleep patterns could be observed. As shown in Table 5 and in Figure 3, Figure 4 and Figure 5, improvements in sleep duration, sleep quality, and the feelings of being restored occurred within the first three weeks of intervention, followed by further descriptive but statistically nonsignificant improvements. To our knowledge, no such data have been found before. A closer look at the specific interventions employed within the first four weeks reveals (see Table 1) that participants acquired key concepts of ACT—accepting unpleasant feelings and thoughts as simple products of the mind, to which we are not obliged to react (acceptance, cognitive diffusion); thinking about personal core values and how to act and move to get closer to such core values, while staying committed to them (commitment); and techniques of mindfulness. In contrast, the content of the sessions in weeks five to eight might be more considered as consolidation of what has been acquired before [42]. As such, it appears plausible that the learning curve changed from a rapid and steep improvement to a rather asymptotic process.
Despite the novelty of the results, the following limitations warrant against overgeneralization. First, the sample size was small, though, we focused on effect size calculations, which by definition are not sensitive to sample sizes. Second, participants were prevalently women (62.9%); given this fact, it is conceivable that a balanced sex ratio would have yielded a modified pattern of results. Third, we assessed a highly selected group of adults with insomnia self-reporting a sleep duration of four hours per night. In contrast, clinical and everyday experiences show that adults with insomnia usually report further psychological issues such as symptoms of depression, anxiety, and rumination. As such, it is conceivable that the present sample does not appropriately reflect every day and clinical reality. Fourth, except for the diagnosis, we fully relied on self-reports; therefore, experts’ ratings might have strengthened the validity of the results. Fifth, similarly, psychophysiological indices such as cortisol concentrations as a proxy of psychophysiological arousal could have informed us if subjective sleep improvements were accompanied by psychophysiological changes. Sixth, by definition, individuals with insomnia claim an insufficiently short sleep duration (see Table 5), while objective sleep-EEG assessments do not confirm such claims. Given this background, it would have been important to see if and to what extent subjective and objective sleep duration do match or not, and if ACT interventions would have yielded a more accurate sleep duration estimation when compared to objective measurements; simply put, it would have been interesting to see if improvements in experiential avoidance were associated with a more accurate objective sleep duration estimation. Seventh, while we claimed that improvements in experiential avoidance as the key factor of ACT was the driver of improved sleep, most of the change in sleep occurred within the first three weeks of treatment; given this fact, the change in experiential avoidance should have had to occur even earlier in order to establish an adequate timeline between outcome and presumed change mechanism. Eighth, it would have been interesting to know if participants’ family members observed participants’ behavioral and sleep-related changes. Future studies might introduce the so-called one-for-many procedure, where family members and peers are asked to evaluate the behavior of study participants [89].
5. Conclusions
The results of the present randomized intervention study suggested that ACT had the potential to favorably impact subjective sleep and cognitive-emotional processes related to insomnia among adults with insomnia. Improvements were observable in the first three to four weeks; improvements remained stable for 12 additional weeks after the end of the study.
Author Contributions
Conceptualization: A.Z., H.K., M.R., S.L., M.E., K.D., A.B.B., D.S.-B., S.B.; Data gathering: A.Z., H.K., M.R., M.E., D.S.-B.; Data entry: A.Z., H.K., M.R., M.E., K.D., D.S.-B.; Statistics: S.L., K.D., A.B.B., D.S.-B., S.B.; Manuscript draft: A.Z., H.K., M.R., S.L., M.E., K.D., A.B.B., D.S.-B., S.B.; Final manuscript, including revisions: A.Z., H.K., M.R., S.L., M.E., K.D., A.B.B., D.S.-B., S.B. All authors have read and agreed to the published version of the manuscript.
Funding
We thank the Iran National Science Foundation (INSF) and Kermanshah University of Medical Sciences for their financial support. The University of Basel (Basel, Switzerland) financially supported the APC.
Institutional Review Board Statement
The Kermanshah University of Medical Science Ethics Committee (IR.KUMS.REC.1396.478) approved the study, which was performed in accordance with the seventh and last revision [57] of the Declaration of Helsinki.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
Data are available upon request for experts in the field.
Acknowledgments
The authors are grateful to the participants for their participation and engagement in the study.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Kyle, S.D.; Morgan, K.; Espie, C.A. Insomnia and health-related quality of life. Sleep Med. Rev. 2010, 14, 69–82. [Google Scholar] [CrossRef]
- Ohayon, M.M. Epidemiology of insomnia: What we know and what we still need to learn. Sleep Med. Rev. 2002, 6, 97–111. [Google Scholar] [CrossRef]
- Moree, W.J.; Li, B.-F.; Jovic, F.; Coon, T.; Yu, J.; Gross, R.S.; Tucci, F.; Marinkovic, D.; Zamani-Kord, S.; Malany, S. Characterization of novel selective H1-antihistamines for clinical evaluation in the treatment of insomnia. J. Med. Chem. 2009, 52, 5307–5310. [Google Scholar] [CrossRef]
- Morin, C.M.; LeBlanc, M.; Daley, M.; Gregoire, J.; Merette, C. Epidemiology of insomnia: Prevalence, self-help treatments, consultations, and determinants of help-seeking behaviors. Sleep Med. 2006, 7, 123–130. [Google Scholar] [CrossRef]
- Saddichha, S. Diagnosis and treatment of chronic insomnia. Ann. Indian Acad. Neurol. 2010, 13, 94. [Google Scholar] [CrossRef]
- Bioulac, S.; Franchi, J.M.; Arnaud, M.; Sagaspe, P.; Moore, N.; Salvo, F.; Philip, P. Risk of Motor Vehicle Accidents Related to Sleepiness at the Wheel: A Systematic Review and Meta-Analysis. Sleep 2017, 40. [Google Scholar] [CrossRef]
- Garbarino, S.; Magnavita, N.; Guglielmi, O.; Maestri, M.; Dini, G.; Bersi, F.M.; Toletone, A.; Chiorri, C.; Durando, P. Insomnia is associated with road accidents. Further evidence from a study on truck drivers. PLoS ONE 2017, 12, e0187256. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, M.; Amici, R.; Lucas, R.; Akerstedt, T.; Cirignotta, F.; Horne, J.; Leger, D.; McNicholas, W.T.; Partinen, M.; Teran-Santos, J.; et al. Sleepiness at the wheel across Europe: A survey of 19 countries. J. Sleep Res. 2015, 24, 242–253. [Google Scholar] [CrossRef]
- Gupta, R.; Pandi-Perumal, S.R.; Almeneessier, A.S.; BaHammam, A.S. Hypersomnolence and Traffic Safety. Sleep Med. Clin. 2017, 12, 489–499. [Google Scholar] [CrossRef] [PubMed]
- Leger, D.; Bayon, V.; Ohayon, M.M.; Philip, P.; Ement, P.; Metlaine, A.; Chennaoui, M.; Faraut, B. Insomnia and accidents: Cross-sectional study (EQUINOX) on sleep-related home, work and car accidents in 5293 subjects with insomnia from 10 countries. J. Sleep Res. 2014, 23, 143–152. [Google Scholar] [CrossRef]
- Godet-Cayré, V.; Pelletier-Fleury, N.; Le Vaillant, M.; Dinet, J.; Massuel, M.A.; Léger, D. Insomnia and absenteeism at work. Who pays the cost? Sleep 2006, 29, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Leger, D. Working with poor sleep. Sleep 2014, 37, 1401–1403. [Google Scholar] [CrossRef][Green Version]
- Teixeira, L.R.; Fischer, F.M.; Lowden, A. Sleep deprivation of working adolescents--A hidden work hazard. Scand. J. Work Environ. Health 2006, 32, 328–330. [Google Scholar] [CrossRef][Green Version]
- Kawakami, N.; Takatsuka, N.; Shimizu, H. Sleep disturbance and onset of type 2 diabetes. Diabetes Care 2004, 27, 282–283. [Google Scholar] [CrossRef]
- Mallon, L.; Broman, J.E.; Hetta, J. Sleep complaints predict coronary artery disease mortality in males: A 12-year follow-up study of a middle-aged Swedish population. J. Intern. Med. 2002, 251, 207–216. [Google Scholar] [CrossRef]
- Hertenstein, E.; Feige, B.; Gmeiner, T.; Kienzler, C.; Spiegelhalder, K.; Johann, A.; Jansson-Fröjmark, M.; Palagini, L.; Rücker, G.; Riemann, D. Insomnia as a predictor of mental disorders: A systematic review and meta-analysis. Sleep Med. Rev. 2019, 43, 96–105. [Google Scholar] [CrossRef]
- American Academy of Sleep Medicine. The International Classification of Sleep Disorders, 3rd ed.; American Academy of Sleep Medicine: Darien, IL, USA, 2014. [Google Scholar]
- Riemann, D.; Baglioni, C.; Bassetti, C.; Bjorvatn, B.; Dolenc Groselj, L.; Ellis, J.G.; Espie, C.A.; Garcia-Borreguero, D.; Gjerstad, M.; Gonçalves, M.; et al. European guideline for the diagnosis and treatment of insomnia. J. Sleep Res. 2017, 26, 675–700. [Google Scholar] [CrossRef] [PubMed]
- Chung, K.F.; Lee, C.T.; Yeung, W.F.; Chan, M.S.; Chung, E.W.; Lin, W.L. Sleep hygiene education as a treatment of insomnia: A systematic review and meta-analysis. Fam. Pr. 2018, 35, 365–375. [Google Scholar] [CrossRef] [PubMed]
- Davidson, J.R.; Dickson, C.; Han, H. Cognitive behavioural treatment for insomnia in primary care: A systematic review of sleep outcomes. Br. J. Gen. Pract. J. R. Coll. Gen. Pract. 2019, 69, e657–e664. [Google Scholar] [CrossRef]
- Seyffert, M.; Lagisetty, P.; Landgraf, J.; Chopra, V.; Pfeiffer, P.N.; Conte, M.L.; Rogers, M.A. Internet-Delivered Cognitive Behavioral Therapy to Treat Insomnia: A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0149139. [Google Scholar] [CrossRef]
- Zachariae, R.; Lyby, M.S.; Ritterband, L.M.; O’Toole, M.S. Efficacy of internet-delivered cognitive-behavioral therapy for insomnia-A systematic review and meta-analysis of randomized controlled trials. Sleep Med. Rev. 2015, 30, 1–10. [Google Scholar] [CrossRef]
- Trauer, J.M.; Qian, M.Y.; Doyle, J.S.; Rajaratnam, S.M.; Cunnington, D. Cognitive Behavioral Therapy for Chronic Insomnia: A Systematic Review and Meta-analysis. Ann. Intern. Med. 2015, 163, 191–204. [Google Scholar] [CrossRef]
- Banno, M.; Harada, Y.; Taniguchi, M.; Tobita, R.; Tsujimoto, H.; Tsujimoto, Y.; Kataoka, Y.; Noda, A. Exercise can improve sleep quality: A systematic review and meta-analysis. PeerJ 2018, 6, e5172. [Google Scholar] [CrossRef]
- Cristini, J.; Weiss, M.; De Las Heras, B.; Medina-Rincón, A.; Dagher, A.; Postuma, R.B.; Huber, R.; Doyon, J.; Rosa-Neto, P.; Carrier, J.; et al. The effects of exercise on sleep quality in persons with Parkinson’s disease: A systematic review with meta-analysis. Sleep Med. Rev. 2020, 55, 101384. [Google Scholar] [CrossRef] [PubMed]
- Mercier, J.; Savard, J.; Bernard, P. Exercise interventions to improve sleep in cancer patients: A systematic review and meta-analysis. Sleep Med. Rev. 2017, 36, 43–56. [Google Scholar] [CrossRef]
- Yang, P.Y.; Ho, K.H.; Chen, H.C.; Chien, M.Y. Exercise training improves sleep quality in middle-aged and older adults with sleep problems: A systematic review. J. Physiother. 2012, 58, 157–163. [Google Scholar] [CrossRef]
- Rusch, H.L.; Rosario, M.; Levison, L.M.; Olivera, A.; Livingston, W.S.; Wu, T.; Gill, J.M. The effect of mindfulness meditation on sleep quality: A systematic review and meta-analysis of randomized controlled trials. Ann. N. Y. Acad. Sci. 2019, 1445, 5–16. [Google Scholar] [CrossRef]
- Brand, S.; Holsboer-Trachsler, E.; Naranjo, J.R.; Schmidt, S. Influence of mindfulness practice on cortisol and sleep in long-term and short-term meditators. Neuropsychobiology 2012, 65, 109–118. [Google Scholar] [CrossRef]
- Yeung, V.; Sharpe, L.; Glozier, N.; Hackett, M.L.; Colagiuri, B. A systematic review and meta-analysis of placebo versus no treatment for insomnia symptoms. Sleep Med. Rev. 2018, 38, 17–27. [Google Scholar] [CrossRef]
- Harvey, A.G.; Tang, N.K. Cognitive behaviour therapy for primary insomnia: Can we rest yet? Sleep Med. Rev. 2003, 7, 237–262. [Google Scholar] [CrossRef]
- Dalrymple, K.L.; Fiorentino, L.; Politi, M.C.; Posner, D. Incorporating principles from acceptance and commitment therapy into cognitive-behavioral therapy for insomnia: A case example. J. Contemp. Psychother. 2010, 40, 209–217. [Google Scholar] [CrossRef]
- Biglan, A.; Hayes, S.C.; Pistorello, J. Acceptance and commitment: Implications for prevention science. Prev. Sci. 2008, 9, 139–152. [Google Scholar] [CrossRef]
- Eustis, E.H.; Hayes-Skelton, S.A.; Roemer, L.; Orsillo, S.M. Reductions in experiential avoidance as a mediator of change in symptom outcome and quality of life in acceptance-based behavior therapy and applied relaxation for generalized anxiety disorder. Behav. Res. 2016, 87, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Harris, R. Embracing your demons: An overview of acceptance and commitment therapy. Psychother. Aust. 2006, 12, 2–8. [Google Scholar]
- Hayes, S.C. Acceptance and commitment therapy: Towards a unified model of behavior change. World Psychiatry Off. J. World Psychiatr. Assoc. (Wpa) 2019, 18, 226–227. [Google Scholar] [CrossRef] [PubMed]
- Hayes, S.C. Acceptance and Commitment Therapy, Relational Frame Theory, and the Third Wave of Behavioral and Cognitive Therapies - Republished Article. Behav. Ther. 2016, 47, 869–885. [Google Scholar] [CrossRef]
- Hayes, S.C.; Luoma, J.B.; Bond, F.W.; Masuda, A.; Lillis, J. Acceptance and commitment therapy: Model, processes and outcomes. Behav Res 2006, 44, 1–25. [Google Scholar] [CrossRef]
- Stockton, D.; Kellett, S.; Berrios, R.; Sirois, F.; Wilkinson, N.; Miles, G. Identifying the Underlying Mechanisms of Change During Acceptance and Commitment Therapy (ACT): A Systematic Review of Contemporary Mediation Studies. Behav. Cogn. Psychother. 2019, 47, 332–362. [Google Scholar] [CrossRef]
- Wicksell, R.K.; Olsson, G.L.; Hayes, S.C. Psychological flexibility as a mediator of improvement in Acceptance and Commitment Therapy for patients with chronic pain following whiplash. Eur. J. Pain 2010, 14, 1059.e1051–1059.e1011. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.Q.; Leeming, E.; Smith, P.; Chung, P.K.; Hagger, M.S.; Hayes, S.C. Acceptance and Commitment Therapy for Health Behavior Change: A Contextually-Driven Approach. Front Psychol. 2017, 8, 2350. [Google Scholar] [CrossRef]
- Harris, R. ACT Made Simple: An Easy-to-Read Primer on Acceptance and Commitment Therapy; New Harbinger Press: Oakland, CA, USA, 2019. [Google Scholar]
- Eifert, G.H. Acceptance and Commitment Therapy (ACT); Hogrefe: Göttingen, Germany, 2011. [Google Scholar]
- Folke, F.; Parling, T.; Melin, L. Acceptance and commitment therapy for depression: A preliminary randomized clinical trial for unemployed on long-term sick leave. Cogn. Behav. Pract. 2012, 19, 583–594. [Google Scholar] [CrossRef]
- Swain, J.; Hancock, K.; Hainsworth, C.; Bowman, J. Acceptance and commitment therapy in the treatment of anxiety: A systematic review. Clin. Psychol. Rev. 2013, 33, 965–978. [Google Scholar] [CrossRef] [PubMed]
- Twohig, M.P. Acceptance and commitment therapy for treatment-resistant posttraumatic stress disorder: A case study. Cogn. Behav. Pract. 2009, 16, 243–252. [Google Scholar] [CrossRef]
- Salari, N.; Khazaie, H.; Hosseinian-Far, A.; Khaledi-Paveh, B.; Ghasemi, H.; Mohammadi, M.; Shohaimi, S. The effect of acceptance and commitment therapy on insomnia and sleep quality: A systematic review. BMC Neurol 2020, 20, 300. [Google Scholar] [CrossRef]
- Khazaie, H.; Zakiei, A.; Rezaei, M.; Hoseini, S.M.; Alikhani, M. Emotional dysregulation leads to reduced sleep quality when the level of repetitive negative thoughts is high: Findings of a structural equation model. Iran. J. Psychiatry Behav. Sci. 2019, 13. [Google Scholar] [CrossRef]
- Khazaie, H.; Zakiei, A.; Komasi, S. A simultaneous cluster analysis of cognitive, emotional, and personality factors and insomnia and sleep quality among earthquake victims. Disaster Med. Public Health Prep. 2019, 13, 745–752. [Google Scholar] [CrossRef] [PubMed]
- Lappalainen, P.; Langrial, S.; Oinas-Kukkonen, H.; Muotka, J.; Lappalainen, R. ACT for sleep-internet-delivered self-help ACT for sub-clinical and clinical insomnia: A randomized controlled trial. J. Contextual Behav. Sci. 2019, 12, 119–127. [Google Scholar] [CrossRef]
- Hertenstein, E.; Thiel, N.; Lüking, M.; Külz, A.K.; Schramm, E.; Baglioni, C.; Spiegelhalder, K.; Riemann, D.; Nissen, C. Quality of Life Improvements after Acceptance and Commitment Therapy in Nonresponders to Cognitive Behavioral Therapy for Primary Insomnia. Psychother. Psychosom. 2014, 83, 371–373. [Google Scholar] [CrossRef]
- Daly-Eichenhardt, A.; Scott, W.; Howard-Jones, M.; Nicolaou, T.; McCracken, L.M. Changes in Sleep Problems and Psychological Flexibility following Interdisciplinary Acceptance and Commitment Therapy for Chronic Pain: An Observational Cohort Study. Front Psychol 2016, 7, 1326. [Google Scholar] [CrossRef]
- Garland, S.N.; Zhou, E.S.; Gonzalez, B.D.; Rodriguez, N. The quest for mindful sleep: A critical synthesis of the impact of mindfulness-based interventions for insomnia. Curr. Sleep Med. Rep. 2016, 2, 142–151. [Google Scholar] [CrossRef]
- McCracken, L.M.; Williams, J.L.; Tang, N.K. Psychological flexibility may reduce insomnia in persons with chronic pain: A preliminary retrospective study. Pain Med. 2011, 12, 904–912. [Google Scholar] [CrossRef] [PubMed]
- Hayes, S.C.; Strosahl, K.; Wilson, K.G.; Bissett, R.T.; Pistorello, J.; Toarmino, D.; Polusny, M.A.; Dykstra, T.A.; Batten, S.V.; Bergan, J. Measuring experiential avoidance: A preliminary test of a working model. Psychol. Rec. 2004, 54, 553–578. [Google Scholar] [CrossRef]
- Hayes, S.C.; Strosahl, K.D.; Wilson, K.G. Acceptance and Commitment Therapy; The Guilford Press: New York NY, USA, 2012. [Google Scholar]
- World Medical Association. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA 2013, 310, 2191–2194. [Google Scholar] [CrossRef]
- Khazaie, H.; Zakiei, A. Efficacy of Acceptance and Commitment Therapyon Emotional Dysregulation and Sleep Quality in Patientswith Chronic Insomnia. J. Maz. Univ. Med Sci. 2019, 29, 52–63. [Google Scholar]
- Zakiei, A.; Khazaie, H. The Effectiveness of Acceptance and Commitment Therapy on Insomnia Patients (A Single-arm Trial Plan). J. Turk. Sleep Med. 2019, 6, 65–73. [Google Scholar] [CrossRef]
- First, M. Structured Clinical Interview for the DSM (SCID). In The Encyclopedia of Clinical Psychology; Wiley: Marblehead, MA, USA, 2015; pp. 1–6. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Lang, A.G.; Buchner, A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Alikhani, M.; Ebrahimi, A.; Farnia, V.; Khazaie, H.; Radmehr, F.; Mohamadi, E.; Davarinejad, O.; Dürsteler, K.; Sadeghi Bahmani, D.; Brand, S. Effects of treatment of sleep disorders on sleep, psychological and cognitive functioning and biomarkers in individuals with HIV/AIDS and under methadone maintenance therapy. J. Psychiatr. Res. 2020, 130, 260–272. [Google Scholar] [CrossRef]
- Farnia, V.; Alikhani, M.; Ebrahimi, A.; Golshani, S.; Sadeghi Bahmani, D.; Brand, S. Ginseng treatment improves the sexual side effects of methadone maintenance treatment. Psychiatry Res. 2019, 276, 142–150. [Google Scholar] [CrossRef]
- Jahangard, L.; Sadeghi, A.; Ahmadpanah, M.; Holsboer-Trachsler, E.; Sadeghi Bahmani, D.; Haghighi, M.; Brand, S. Influence of adjuvant omega-3-polyunsaturated fatty acids on depression, sleep, and emotion regulation among outpatients with major depressive disorders - Results from a double-blind, randomized and placebo-controlled clinical trial. J. Psychiatr. Res. 2018, 107, 48–56. [Google Scholar] [CrossRef]
- Buysse, D.J.; Reynolds, C.F., 3rd; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef]
- Farrahi Moghaddam, J.; Nakhaee, N.; Sheibani, V.; Garrusi, B.; Amirkafi, A. Reliability and validity of the Persian version of the Pittsburgh Sleep Quality Index (PSQI-P). Sleep Breath. Schlaf Atm. 2012, 16, 79–82. [Google Scholar] [CrossRef]
- Chehri, A.; Nourozi, M.; Eskandari, S.; Khazaie, H.; Hemati, N.; Jalali, A. Validation of the Persian version of the Pittsburgh Sleep Quality Index in elderly population. Sleep Sci. (Sao PauloBraz.) 2020, 13, 119–124. [Google Scholar] [CrossRef]
- Chehri, A.; Brand, S.; Goldaste, N.; Eskandari, S.; Brühl, A.; Sadeghi Bahmani, D.; Khazaie, H. Psychometric Properties of the Persian Pittsburgh Sleep Quality Index for Adolescents. Int. J. Environ. Res. Public Health 2020, 17, 7095. [Google Scholar] [CrossRef]
- Backhaus, J.; Riemann, D. Schlafstörungen bewältigen [Treating Insomnia]; Beltz: Weinheim, Germany; Basel, Switzerland, 1996. [Google Scholar]
- Morin, C.M.; Vallières, A.; Ivers, H. Dysfunctional beliefs and attitudes about sleep (DBAS): Validation of a brief version (DBAS-16). Sleep 2007, 30, 1547–1554. [Google Scholar] [CrossRef]
- Bothelius, K.; Jernelöv, S.; Fredrikson, M.; McCracken, L.M.; Kaldo, V. Measuring acceptance of sleep difficulties: The development of the sleep problem acceptance questionnaire. Sleep 2015, 38, 1815–1822. [Google Scholar] [CrossRef] [PubMed]
- Gratz, K.L.; Roemer, L. Multidimensional Assessment of Emotion Regulation and Dysregulation: Development, Factor Structure, and Initial Validation of the Difficulties in Emotion Regulation Scale. J. Psychopathol. Behav. Assess. 2004, 26, 41–54. [Google Scholar] [CrossRef]
- McKay, M.; Forsyth, J.P.; Eifert, G.H. Your Life on Purpose. How to Find What Matters and Create the Life You Want; New Harbinger Publications: Oakland, CA, USA, 2010. [Google Scholar]
- Wengenroth, M. Acceptance and Commitment Therapy (ACT); Therapy Tools; Beltz: Weinheim, Germany; Basel, Switzerland, 2017. [Google Scholar]
- Sadeghi Bahmani, D.; Motl, R.W.; Razazian, N.; Khazaie, H.; Brand, S. Aquatic exercising may improve sexual function in females with multiple sclerosis—An exploratory study. Mult. Scler. Relat. Disord. 2020, 43, 102106. [Google Scholar] [CrossRef] [PubMed]
- Sadeghi Bahmani, D.; Razazian, N.; Farnia, V.; Alikhani, M.; Tatari, F.; Brand, S. Compared to an active control condition, in persons with multiple sclerosis two different types of exercise training improved sleep and depression, but not fatigue, paresthesia, and intolerance of uncertainty. Mult. Scler. Relat. Disord. 2019. [Google Scholar] [CrossRef] [PubMed]
- Sadeghi Bahmani, D.; Razazian, N.; Motl, R.W.; Farnia, V.; Alikhani, M.; Pühse, U.; Gerber, M.; Brand, S. Physical activity interventions can improve emotion regulation and dimensions of empathy in persons with multiple sclerosis: An exploratory study. Mult. Scler. Relat. Disord. 2020, 37, 101380. [Google Scholar] [CrossRef] [PubMed]
- Jasbi, M.; Sadeghi Bahmani, D.; Karami, G.; Omidbeygi, M.; Peyravi, M.; Panahi, A.; Mirzaee, J.; Holsboer-Trachsler, E.; Brand, S. Influence of adjuvant mindfulness-based cognitive therapy (MBCT) on symptoms of post-traumatic stress disorder (PTSD) in veterans-results from a randomized control study. Cogn. Behav. 2018. [Google Scholar] [CrossRef]
- Norouzi, E.; Gerber, M.; Masrur, F.; Vaezmousavi, M.; Pühse, U.; Brand, S. Implementation of a Mindfulness-Based Stress Reduction (MBSR) program to reduce stress, anxiety and depression and to improve psychological well-being among retired Iranian football players. Psychol. Sport Exerc. 2019, 36. [Google Scholar] [CrossRef]
- Cordier, D.; Gerber, M.; Brand, S. Effects of two types of exercise training on psychological well-being, sleep, quality of life and physical fitness in patients with high-grade glioma (WHO III and IV): Study protocol for a randomized controlled trial. Cancer Commun. (Lond. Engl.) 2019, 39, 46. [Google Scholar] [CrossRef] [PubMed]
- Marcus, D.K.; O’Connell, D.; Norris, A.L.; Sawaqdeh, A. Is the Dodo bird endangered in the 21st century? A meta-analysis of treatment comparison studies. Clin. Psychol. Rev. 2014, 34, 519–530. [Google Scholar] [CrossRef]
- Becker, B.J. Synthesizing standardized mean-change measures. Br. J. Math. Stat. Psychol. 1988, 41, 257–278. [Google Scholar] [CrossRef]
- Harvey, A.G. A transdiagnostic approach to treating sleep disturbance in psychiatric disorders. Cogn. Behav. 2009, 38 (Suppl. 1), 35–42. [Google Scholar] [CrossRef]
- Leventhal, A.M.; Zvolensky, M.J. Anxiety, depression, and cigarette smoking: A transdiagnostic vulnerability framework to understanding emotion-smoking comorbidity. Psychol. Bull. 2015, 141, 176–212. [Google Scholar] [CrossRef] [PubMed]
- Norton, P.J.; Barrera, T.L.; Mathew, A.R.; Chamberlain, L.D.; Szafranski, D.D.; Reddy, R.; Smith, A.H. Effect of transdiagnostic cbt for anxiety disorders on comorbid diagnoses. Depress Anxiety 2013, 30, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Norton, P.J.; Paulus, D.J. Toward a Unified Treatment for Emotional Disorders: Update on the Science and Practice. Behav. Ther. 2016, 47, 854–868. [Google Scholar] [CrossRef]
- Norton, P.J.; Roberge, P. Transdiagnostic Therapy. Psychiatr. Clin. North Am. 2017, 40, 675–687. [Google Scholar] [CrossRef]
- Pearl, S.B.; Norton, P.J. Transdiagnostic versus diagnosis specific cognitive behavioural therapies for anxiety: A meta-analysis. J. Anxiety Disord. 2017, 46, 11–24. [Google Scholar] [CrossRef]
- Holtzman, N.S.; Strube, M.J. Above and beyond short-term mating, long-term mating is uniquely tied to human personality. Evol. Psychol. Int. J. Evol. Approaches Psychol. Behav. 2013, 11, 1101–1129. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).