Male Deep-Sea Shrimps Aristeus antennatus at Fishing Grounds: Growth and First Evaluation of Recruitment by Multilocus Genotyping
Abstract
1. Introduction
2. Materials and Methods
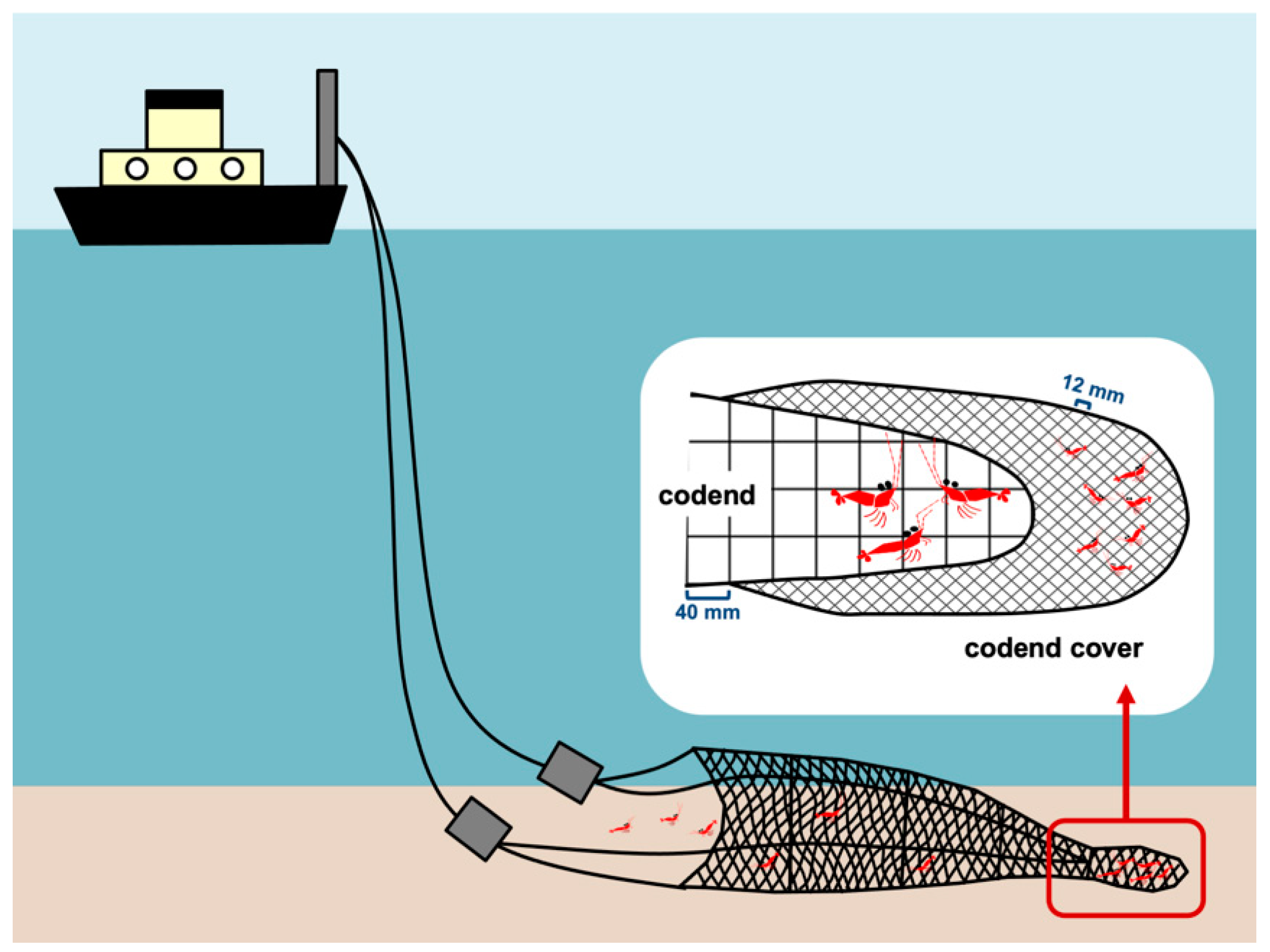
2.1. Sampling and Biological Material
2.2. DNA Extraction and Microsatellite Loci Genotyping
2.3. Statistical and Genetic Analysis
3. Results
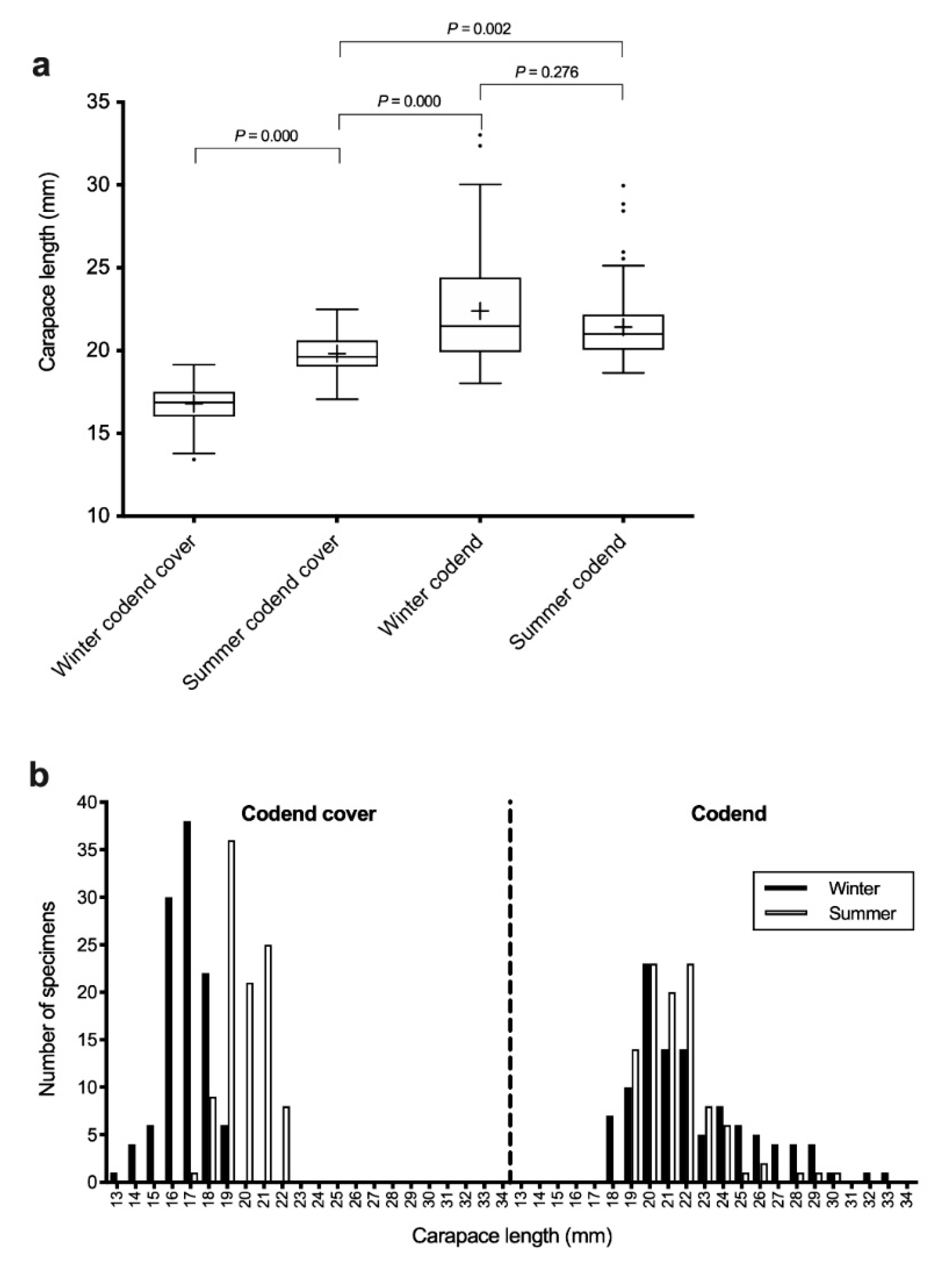
3.1. Comparison of Carapace Length among Male Groups
3.2. Genetic Diversity
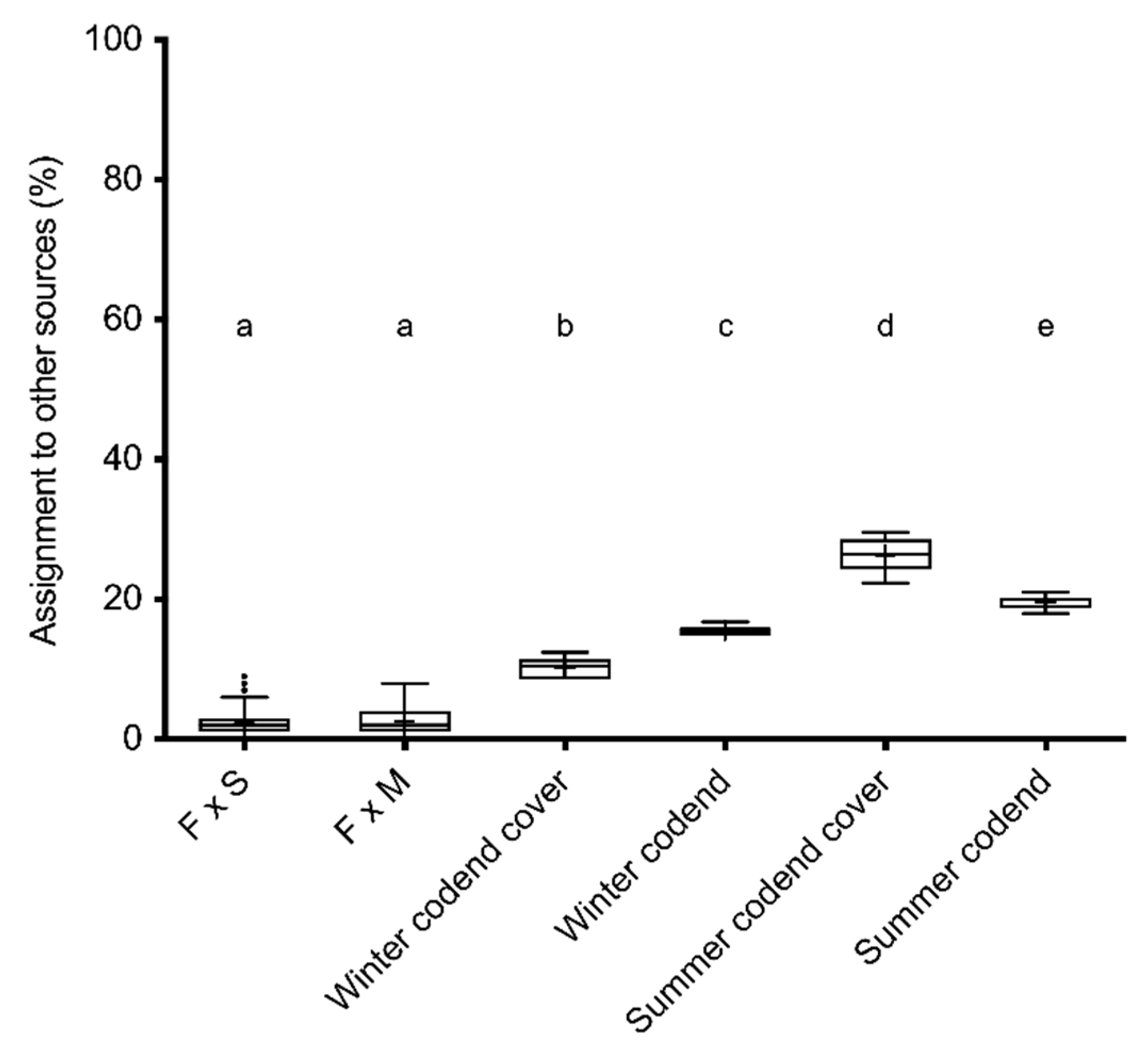
3.3. Genetic Divergence
4. Discussion
4.1. Growth and Recruitment of A. antennatus in Submarine Canyons
4.2. Geographical Origin of Males Recruited into the Fishery
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Moranta, J.; Quetglas, A.; Massutí, E.; Guijarro, B.; Ordines, F.; Valls, M. Research trends on demersal fisheries oceanography in the Mediterranean. In Biological Oceanography Research Trends; Mertens, L.P., Ed.; Nova Science Publishers: New York, NY, USA, 2008; pp. 9–65. [Google Scholar]
- European Parliament and The Council of the European Union Regulation (EU) 2019/1022 of the European Parliament and of the Council of 20 June 2019 establishing a multiannual plan for the fisheries exploiting demersal stocks in the western Mediterranean Sea and amending Regulation (EU) No 508/2014. Off. J. Eur. Union 2019, L172, 1–17.
- Gorelli, G.; Company, J.B.; Bahamón, N.; Sardà, F. Improving condend selectivity in the fishery of the deep-sea shrimp Aristeus antennatus in the northwestern Mediterranean Sea. Sci. Mar. 2017, 81, 381–386. [Google Scholar] [CrossRef]
- Sola, I.; Maynou, F.; Sánchez-Lizaso, J.L. Bioeconomic analysis of the EU multiannual management plan for demersal fisheries in the western Mediterranean. Spanish fisheries as a case study. Front. Mar. Sci. 2020, 7, 459. [Google Scholar] [CrossRef]
- Demestre, M.; Lleonart, J. Population dynamics of Aristeus antennatus (Decapoda: Dendrobranchiata) in the northwestern Mediterranean. Sci. Mar. 1993, 57, 183–189. [Google Scholar]
- Sardà, F.; Roldán, M.I.; Heras, S.; Maltagliati, F. Influence of the genetic structure of the red and blue shrimp, Aristeus antennatus (Risso, 1816) on the sustainability of a deep-sea population along a depth gradient in the Western Mediterranean. Sci. Mar. 2010, 74, 569–575. [Google Scholar] [CrossRef]
- Sardà, F.; D’Onghia, G.; Politou, C.Y.; Company, J.B.; Maiorano, P.; Kapiris, K. Deep-sea distribution, biological and ecological aspects of Aristeus antennatus (Risso, 1816) in the Western and Central Mediterranean Sea. Sci. Mar. 2004, 68, 117–127. [Google Scholar] [CrossRef]
- Sardà, F.; Company, J.B. The deep-sea recruitment of Aristeus antennatus (Risso, 1816) (Crustacea: Decapoda) in the Mediterranean Sea. J. Mar. Syst. 2012, 105–108, 145–151. [Google Scholar] [CrossRef]
- Sardà, F.; Company, J.B.; Castellón, A. Intraspecific aggregation structure of a shoal of a Western Mediterranean (Catalan coast) deep-sea shrimp, Aristeus antennatus (Risso, 1816), during the reproductive period. J. Shellfish Res. 2003, 22, 569–579. [Google Scholar]
- Planella, L.; Vera, M.; García-Marín, J.L.; Heras, S.; Roldán, M.I. Mating structure of the blue and red shrimp, Aristeus antennatus (Risso, 1816), revealed by relatedness analysis. Sci. Rep. 2019, 9, 7227. [Google Scholar] [CrossRef]
- D’Onghia, G.; Maiorano, P.; Capezzuto, F.; Carlucci, R.; Battista, D.; Giove, A.; Sion, L.; Tursi, A. Further evidences of deep-sea recruitment of Aristeus antennatus (Crustacea: Decapoda) and its role in the population renewal on the exploited bottoms of the Mediterranean. Fish. Res. 2008, 95, 236–245. [Google Scholar] [CrossRef]
- Demestre, M. Moult activity-related spawning success in the Mediterranean deep-water shrimp Aristeus antennatus (Decapoda: Dendrobranchiata). Mar. Ecol. Prog. Ser. 1995, 127, 57–64. [Google Scholar] [CrossRef]
- D’Onghia, G.; Capezzuto, F.; Mytilineou, C.; Maiorano, P.; Kapiris, K.; Carlucci, R.; Sion, L.; Tursi, A. Comparison of the population structure and dynamics of Aristeus antennatus (Risso, 1816) between exploited and unexploited areas in the Mediterranean Sea. Fish. Res. 2005, 76, 22–38. [Google Scholar] [CrossRef]
- Carbonell, A.; Lloret, J.; Demestre, M. Relationship between condition and recruitment success of red shrimp (Aristeus antennatus) in the Balearic Sea (Northwestern Mediterranean). J. Mar. Syst. 2008, 71, 403–412. [Google Scholar] [CrossRef]
- Guijarro, B.; Massutí, E.; Moranta, J.; Díaz, P. Population dynamics of the red shrimp Aristeus antennatus in the Balearic Islands (western Mediterranean): Short spatio-temporal differences and influence of environmental factors. J. Mar. Syst. 2008, 71, 385–402. [Google Scholar] [CrossRef]
- Demestre, M.; Fortuño, J.M. Reproduction of the deep-water shrimp Aristeus antennatus (Decapoda: Dendrobranchiata). Mar. Ecol. Prog. Ser. 1992, 84, 41–51. [Google Scholar] [CrossRef]
- Kapiris, K.; Thessalou-Legaki, M. Comparative reproduction aspects of the deep-water shrimps Aristaeomorpha foliacea and Aristeus antennatus (Decapoda, Aristeidae) in the Greek Ionian Sea (Eastern Mediterranean). Int. J. Zool. 2009, 2009, 979512. [Google Scholar] [CrossRef]
- Sardà, F.; Demestre, M. Shortening of the rostrum and rostral variability in Aristeus antennatus (Risso, 1816). J. Crust. Biol. 1989, 9, 570–577. [Google Scholar] [CrossRef]
- García-Rodríguez, M.; Esteban, A. On the biology and fishery of Aristeus antennatus (Risso, 1816), (Decapoda, Dendrobranchiata) in the Ibiza Channel (Balearic Islands, Spain). Sci. Mar. 1999, 63, 27–37. [Google Scholar] [CrossRef]
- Carretón, M.; Company, J.B.; Planella, L.; Heras, S.; García-Marína, J.L.; Agulló, M.; Clavel-Henry, M.; Rotllant, G.; Dos Santos, A.; Roldán, M.I. Morphological identification and molecular confirmation of the deep-sea blue and red shrimp Aristeus antennatus larvae. PeerJ 2019, 7, e6063. [Google Scholar] [CrossRef]
- Torres, A.P.; Dos Santos, A.; Alemany, F.; Massutí, E. Larval stages of crustacean species of interest for conservation and fishing exploitation in the western Mediterranean. Sci. Mar. 2013, 77, 149–160. [Google Scholar] [CrossRef]
- Clavel-Henry, M.; Solé, J.; Ahumada-Sempoal, M.A.; Bahamon, N.; Briton, F.; Rotllant, G.; Company, J.B. Influence of the summer deep-sea circulations on passive drifts among the submarine canyons in the northwestern Mediterranean Sea. Ocean Sci. 2019, 15, 1745–1759. [Google Scholar] [CrossRef]
- Carbonell, A.; Dos Santos, A.; Alemany, F.; Vélez-Belchi, P. Larvae of the red shrimp Aristeus antennatus (Decapoda: Dendrobranchiata: Aristeidae) in the Baleric Sea: New occurrences fifty years later. Mar. Biodivers. Rec. 2010, 3, e103. [Google Scholar] [CrossRef]
- Cartes, J.E.; López-Pérez, C.; Carbonell, A. Condition and recruitment of Aristeus antennatus at great depths (to 2,300) in the Mediterranean: Relationship with environmental factors. Fish. Oceanogr. 2016, 27, 114–126. [Google Scholar] [CrossRef]
- Sardà, F.; Cartes, J.E. Morphological features and ecological aspects of early juvenile specimens of the aristeid shrimp Aristeus antennatus (Risso, 1816). Mar. Freshw. Res. 1997, 48, 73–77. [Google Scholar] [CrossRef]
- Company, J.B.; Sardà, F.; Puig, P.; Cartes, J.E.; Palanques, A. Duration and timing of reproduction in decapod crustaceans of the NW Mediterranean continental margin: Is there a general pattern? Mar. Ecol. Prog. Ser. 2003, 261, 201–216. [Google Scholar] [CrossRef][Green Version]
- Deval, M.C.; Kapiris, K. A review of biological patterns of the blue-red shrimp Aristeus antennatus in the Mediterranean Sea: A case study of the population of Antalya Bay, eastern Mediterranean Sea. Sci. Mar. 2016, 80, 339–348. [Google Scholar] [CrossRef]
- Fernández, M.V.; Heras, S.; Viñas, J.; Maltagliati, F.; Roldán, M.I. Multilocus comparative phylogeography of two Aristeid shrimps of high commercial interest (Aristeus antennatus and Aristaeomorpha foliacea) reveals different responses to past environmental changes. PLoS ONE 2013, 8, e59033. [Google Scholar] [CrossRef] [PubMed]
- Heras, S.; Planella, L.; Caldarazzo, I.; Vera, M.; García-Marín, J.L.; Roldán, M.I. Development and characterization of novel microsatellite markers by Next Generation Sequencing for the blue and red shrimp Aristeus Antennatus. PeerJ 2016, 4, e2200. [Google Scholar] [CrossRef] [PubMed]
- Goudet, J. FSTAT, A Program to Estimate and Test Gene Diversities and Fixation Indices (Version 2.9.3). 2001. Available online: https://www2.unil.ch/popgen/softwares/fstat.htm (accessed on 19 December 2020).
- Guo, S.W.; Thompson, E.A. Performing the exact test of Hardy-Weinberg proportion for multiple alleles. Biometrics 1992, 48, 361–372. [Google Scholar] [CrossRef]
- Rousset, F. Genepop’007: A complete reimplementation of the Genepop software for Windows and Linux. Mol. Ecol. Res. 2008, 8, 103–106. [Google Scholar] [CrossRef]
- Weir, B.S.; Cockerham, C.C. Estimating F-statistics for the analysis of population structure. Evolution 1984, 38, 1358–1370. [Google Scholar]
- Cannas, R.; Sacco, F.; Follesa, M.C.; Sabatini, A.; Arculeo, M.; Lo Brutto, S.; Maggio, T.; Deianam, A.M.; Cau, A. Genetic variability of the blue and red shrimp Aristeus antennatus in the Western Mediterranean Sea inferred by DNA microsatellite loci. Mar. Ecol. 2012, 33, 350–363. [Google Scholar] [CrossRef]
- Heras, S.; Planella, L.; García-Marín, J.L.; Vera, M.; Roldán, M.I. Genetic structure and population connectivity of the blue and red shrimp Aristeus antennatus. Sci. Rep. 2019, 9, 13531. [Google Scholar] [CrossRef]
- Brookfield, J.F.Y. A simple new method for estimating null allele frequency from heterozygote deficiency. Mol. Ecol. 1996, 5, 453–455. [Google Scholar] [CrossRef] [PubMed]
- Van Oosterhout, C.; Hutchinson, W.F.D.; Willis, D.P.; Shipley, P. Micro-checker: Software for identifying and correcting genotyping errors in microsatellite data. Mol. Ecol. Notes 2004, 4, 535–538. [Google Scholar] [CrossRef]
- Nielsen, E.E.; Bach, L.A.; Kotlicki, P. HYBRIDLAB (version 1.0): A program for generating simulated hybrids from population samples. Mol. Ecol. Notes 2006, 6, 971–973. [Google Scholar] [CrossRef]
- Rannala, B.; Mountain, J.L. Detecting immigration by using multilocus genotypes. Proc. Natl. Acad. Sci. USA 1997, 94, 9197–9201. [Google Scholar] [CrossRef] [PubMed]
- Piry, S.; Alapetite, A.; Cornuet, J.M.; Paetkau, D.; Baudouin, L.; Estoup, A. GeneClass2: A software for genetic assignment and first-generation migrant detection. J. Hered. 2004, 95, 536–539. [Google Scholar] [CrossRef] [PubMed]
- Cartes, J.E.; Demestre, M. Estimating secondary production in the deep-water shrimp, Aristeus antennatus (Risso, 1816) in the Catalano-Balearic basin (Western Mediterranean). J. Northw. Atl. Fish. Sci. 2003, 31, 355–361. [Google Scholar] [CrossRef]
- Chandler, J.C.; Elizur, A.; Ventura, T. The decapod researcher’s guide to the galaxy of sex determination. Hydrobiologia 2017, 825, 61–80. [Google Scholar] [CrossRef]
- Sardà, F.; Demestre, M. Estudio biológico de la gamba Aristeus antennatus (Risso, 1816) en el Mar Catalán (NE de España). Inv. Pesq. 1987, 51 (Suppl. S1), 213–232. [Google Scholar]
- Sardà, F.; Maynou, F.; Talló, L. Seasonal and spatial mobility patterns of rose shrimp Aristeus antennatus in the Western Mediterranean: Results of a long-term study. Mar. Ecol. Prog. Ser. 1997, 159, 133–141. [Google Scholar] [CrossRef]
- Maggio, T.; Lo Brutto, S.; Cannas, R.; Deiana, A.M.; Arculeo, M. Environmental features of deep-sea habitats linked to the genetic population structure of a crustacean species in the Mediterranean Sea. Mar. Ecol. 2009, 30, 354–365. [Google Scholar] [CrossRef]
- Roldán, M.I.; Heras, S.; Patellani, R.; Maltagliati, F. Analysis of genetic structure of the red shrimp Aristeus antennatus from the Western Mediterranean employing two mitochondrial regions. Genetica 2009, 136, 1–4. [Google Scholar] [CrossRef]
- Fernández, M.V.; Heras, S.; Maltagliati, F.; Turco, A.; Roldán, M.I. Genetic structure in the blue and red shrimp Aristeus antennatus and the role played by hydrographical and oceanographical barriers. Mar. Ecol. Prog. Ser. 2011, 421, 163–171. [Google Scholar] [CrossRef]
- Marra, A.; Mona, S.; Sà, R.M.; D’Onghia, G.; Maiorano, P. Population genetic history of Aristeus antennatus (Crustacea: Decapoda) in the Western and Central Mediterranean Sea. PLoS ONE 2015, 10, e0117272. [Google Scholar] [CrossRef]
- Agulló, M.; Heras, S.; García-Marín, J.L.; Vera, M.; Planella, L.; Roldán, M.I. Genetic analyses reveal temporal stability and connectivity pattern in blue and red shrimp Aristeus antennatus populations. Sci. Rep. 2020, 10, 21505. [Google Scholar] [CrossRef]
- Orsi Relini, L.; Mannini, A.; Relini, G. Updating knowledge on growth, population dynamics, and ecology of the blue and red shrimp, Aristeus antennatus (Risso, 1816), on the basis of the study of its instars. Mar. Ecol. 2013, 34, 90–102. [Google Scholar] [CrossRef]
- Relini, M.; Maiorano, P.; D’Onghia, G.; Orsi Relini, L.; Tursi, A.; Panza, M. Recaptures of tagged deep-sea shrimp Aristeus antennatus (Risso, 1816) in the Mediterranean. Rap. Com. Int. Mer. Médit. 2004, 37, 424. [Google Scholar]
- Millot, C. Circulation in the Western Mediterranean Sea. J. Mar. Syst. 1999, 20, 423–442. [Google Scholar] [CrossRef]
- Rubio, A.; Arnau, P.A.; Espino, M.; Flexas, M.M.; Jordà, G.; Salat, J.; Puigdefàbregas, J.; Arcilla, A.S. A field study of the behaviour of an anticyclonic eddy on the Catalan continental shelf (NW Mediterranean). Prog. Oceanogr. 2005, 66, 142–156. [Google Scholar] [CrossRef]
- Sardà, F.; Company, J.B.; Bahamón, N.; Rotllant, G.; Flexas, M.M.; Sánchez, J.D.; Zúñiga, D.; Coenjaerts, J.; Orellana, D.; Jordà, G.; et al. Relationship between environment and the occurrence of the deep-water rose shrimp Aristeus antennatus (Risso, 1816) in the Blanes submarine canyon (NW Mediterranean). Prog. Oceanogr. 2009, 82, 227–238. [Google Scholar] [CrossRef]
- Demestre, M.; Martín, P. Optimum exploitation of a demersal resource in the western Mediterranean: The fishery of the deep-water shrimp Aristeus antennatus (Risso, 1816). Sci. Mar. 1993, 57, 175–182. [Google Scholar]
- Ahumada-Sempoal, M.A.; Flexas, M.M.; Bernardello, R.; Bahamon, N.; Cruzado, A. Northern Current variability and its impact on the Blanes Canyon circulation: A numerical study. Prog. Oceanogr. 2013, 118, 61–70. [Google Scholar] [CrossRef]
- Dharmarajan, G.; Beatty, W.S.; Rhodes, O.E. Heterozygote deficiencies caused by a Wahlund effect: Dispelling unfounded expectations. J. Wildl. Manag. 2013, 77, 226–234. [Google Scholar] [CrossRef]
- Puig, P.; Company, J.B.; Sardà, F.; Palanques, A. Responses of deep-water shrimp populations to intermediate nepheloid layer detachments on the Northwestern Mediterranean continental margin. Deep Sea Res. Part I Oceanogr. Res. Pap. 2001, 48, 2195–2207. [Google Scholar] [CrossRef]
| Cohort | Aa123 | Aa138 | Aa1444 | Aa667 | Aa681 | Aa751 | Aa956 | Aa1061 | Aa1195 | Aa818 | Average | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Winter codend cover | NA | 4 | 19 | 13 | 9 | 20 | 3 | 6 | 8 | 4 | 4 | 9.0 |
| AR | 3.913 | 18.777 | 12.752 | 8.842 | 19.567 | 3.000 | 5.988 | 7.884 | 3.895 | 4.000 | 8.862 | |
| HO | 0.485 | 0.829 | 0.490 | 0.392 | 0.637 | 0.144 | 0.571 | 0.429 | 0.591 | 0.352 | 0.495 | |
| HE | 0.614 | 0.880 | 0.754 | 0.702 | 0.795 | 0.302 | 0.666 | 0.682 | 0.608 | 0.582 | 0.660 | |
| FIS | 0.210 | 0.059 | 0.350 * | 0.442 * | 0.199 | 0.521 * | 0.142 | 0.371 * | 0.028 | 0.394 * | 0.251 | |
| Nu | 0.091 | 0.043 | 0.155 | 0.181 | 0.071 | 0.122 | 0.054 | 0.173 | 0.147 | |||
| Winter codend | NA | 5 | 20 | 12 | 7 | 22 | 3 | 6 | 8 | 5 | 6 | 9.4 |
| AR | 4.930 | 19.904 | 11.937 | 6.918 | 21.436 | 3.000 | 6.000 | 7.792 | 4.991 | 5.995 | 9.290 | |
| HO | 0.485 | 0.812 | 0.490 | 0.449 | 0.545 | 0.126 | 0.644 | 0.475 | 0.654 | 0.356 | 0.506 | |
| HE | 0.610 | 0.896 | 0.715 | 0.699 | 0.856 | 0.271 | 0.665 | 0.697 | 0.596 | 0.625 | 0.665 | |
| FIS | 0.205 | 0.094 | 0.316 * | 0.358 * | 0.364 * | 0.534 * | 0.032 | 0.318 * | −0.097 | 0.430 * | 0.239 | |
| Nu | 0.075 | 0.048 | 0.135 | 0.152 | 0.186 | 0.128 | 0.129 | 0.153 | ||||
| Summer codend cover | NA | 5 | 19 | 13 | 6 | 19 | 3 | 6 | 6 | 5 | 6 | 8.8 |
| AR | 4.959 | 18.915 | 12.937 | 5.979 | 18.875 | 3.000 | 5.999 | 6.000 | 4.918 | 5.998 | 8.758 | |
| HO | 0.418 | 0.837 | 0.500 | 0.479 | 0.541 | 0.188 | 0.561 | 0.459 | 0.622 | 0.347 | 0.496 | |
| HE | 0.625 | 0.911 | 0.762 | 0.694 | 0.828 | 0.287 | 0.682 | 0.696 | 0.628 | 0.689 | 0.681 | |
| FIS | 0.330 * | 0.082 | 0.344 * | 0.310 * | 0.347 * | 0.347 | 0.177 | 0.340 * | 0.008 | 0.497 * | 0.272 * | |
| Nu | 0.116 | 0.040 | 0.152 | 0.131 | 0.166 | 0.080 | 0.057 | 0.136 | 0.195 | |||
| Summer codend | NA | 6 | 25 | 12 | 7 | 20 | 3 | 6 | 7 | 6 | 6 | 9.8 |
| AR | 5.933 | 24.513 | 11.955 | 6.958 | 19.806 | 3.000 | 6.000 | 6.998 | 5.877 | 5.999 | 9.704 | |
| HO | 0.520 | 0.800 | 0.531 | 0.542 | 0.640 | 0.096 | 0.610 | 0.485 | 0.610 | 0.306 | 0.517 | |
| HE | 0.647 | 0.910 | 0.735 | 0.690 | 0.809 | 0.281 | 0.684 | 0.747 | 0.612 | 0.618 | 0.675 | |
| FIS | 0.196 | 0.120 | 0.278 * | 0.215 | 0.209 * | 0.660 * | 0.108 | 0.351 * | 0.004 | 0.504 * | 0.235 | |
| Nu | 0.069 | 0.060 | 0.120 | 0.083 | 0.091 | 0.155 | 0.151 | 0.185 |
| Winter Codend Cover | Summer Codend Cover | Winter Codend | |
|---|---|---|---|
| Summer codend cover | 0.0008 1 | ||
| Winter codend | 0.0010 1 | 0.0000 | |
| Summer codend | 0.0004 | 0.0004 | 0.0000 |
| Replicates | F × S F1 Baseline 1 | F × M F1 Baseline 1 | Other Sources | |
|---|---|---|---|---|
| F × S F1 | 250 | 69.7 (16.3) | 28.1 (15.9) | 2.2 (1.8) |
| F × M F1 | 250 | 47.2 (16.9) | 50.2 (16.8) | 2.6 (1.8) |
| Winter codend cover | 10 | 56.6 (16.4) | 33.2 (15.7) | 10.2 (1.3) |
| Summer codend cover | 10 | 44.4 (13.9) | 29.3 (13.4) | 26.3 (2.2) |
| Winter codend | 10 | 54.5 (13.3) | 30.0 (13.3) | 15.5 (0.6) |
| Summer codend | 10 | 49.10 (12.5) | 31.3 (12.3) | 19.6 (1.0) |
| Baselines | |||||
|---|---|---|---|---|---|
| Palamós Winter Codend | Palamós Winter Codend Cover | Roses Winter Codend | Blanes Winter Codend | Other Sources | |
| Palamós winter codend | 67.3 | 7.9 | 4.0 | 13.9 | 6.9 |
| Palamós winter codend cover | 21.9 | 43.8 | 10.5 | 20.0 | 3.8 |
| Palamós summer codend | 41.0 | 13.0 | 14.0 | 25.0 | 7.0 |
| Palamós summer codend cover | 39.8 | 18.4 | 5.1 | 24.5 | 12.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abras, A.; García-Marín, J.-L.; Heras, S.; Vera, M.; Agulló, M.; Planella, L.; Roldán, M.I. Male Deep-Sea Shrimps Aristeus antennatus at Fishing Grounds: Growth and First Evaluation of Recruitment by Multilocus Genotyping. Life 2021, 11, 116. https://doi.org/10.3390/life11020116
Abras A, García-Marín J-L, Heras S, Vera M, Agulló M, Planella L, Roldán MI. Male Deep-Sea Shrimps Aristeus antennatus at Fishing Grounds: Growth and First Evaluation of Recruitment by Multilocus Genotyping. Life. 2021; 11(2):116. https://doi.org/10.3390/life11020116
Chicago/Turabian StyleAbras, Alba, Jose-Luis García-Marín, Sandra Heras, Manuel Vera, Melania Agulló, Laia Planella, and María Inés Roldán. 2021. "Male Deep-Sea Shrimps Aristeus antennatus at Fishing Grounds: Growth and First Evaluation of Recruitment by Multilocus Genotyping" Life 11, no. 2: 116. https://doi.org/10.3390/life11020116
APA StyleAbras, A., García-Marín, J.-L., Heras, S., Vera, M., Agulló, M., Planella, L., & Roldán, M. I. (2021). Male Deep-Sea Shrimps Aristeus antennatus at Fishing Grounds: Growth and First Evaluation of Recruitment by Multilocus Genotyping. Life, 11(2), 116. https://doi.org/10.3390/life11020116








