Y/X-Chromosome-Bearing Sperm Shows Elevated Ratio in the Left but Not the Right Testes in Healthy Mice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Prediction for Y:X Balance between the Left and the Right Testes
2.2. Experimental Mice
2.3. Sperm Cell Isolation and Staining
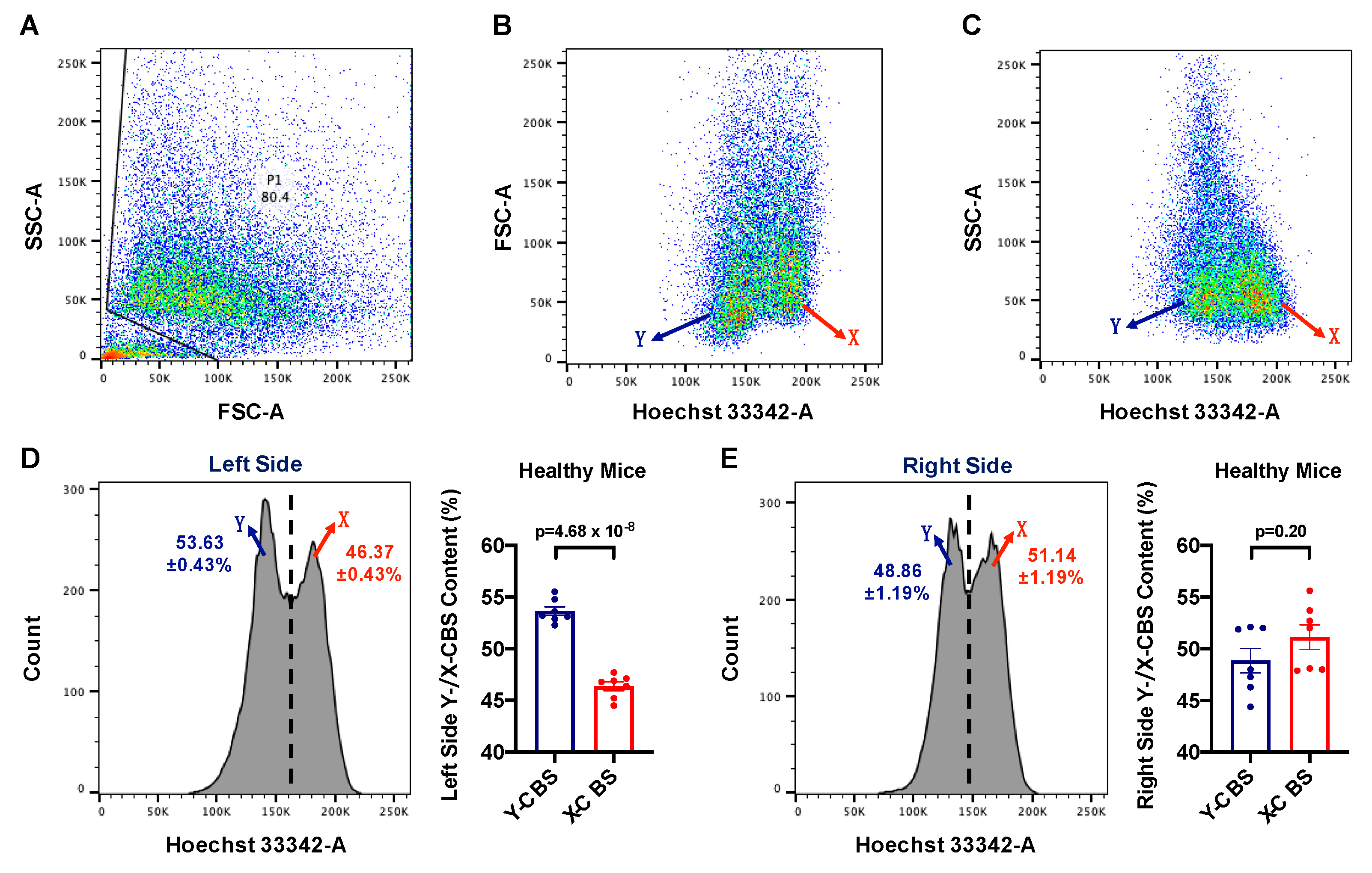
2.4. Flow Cytometry
2.5. Statistical Analysis
3. Results
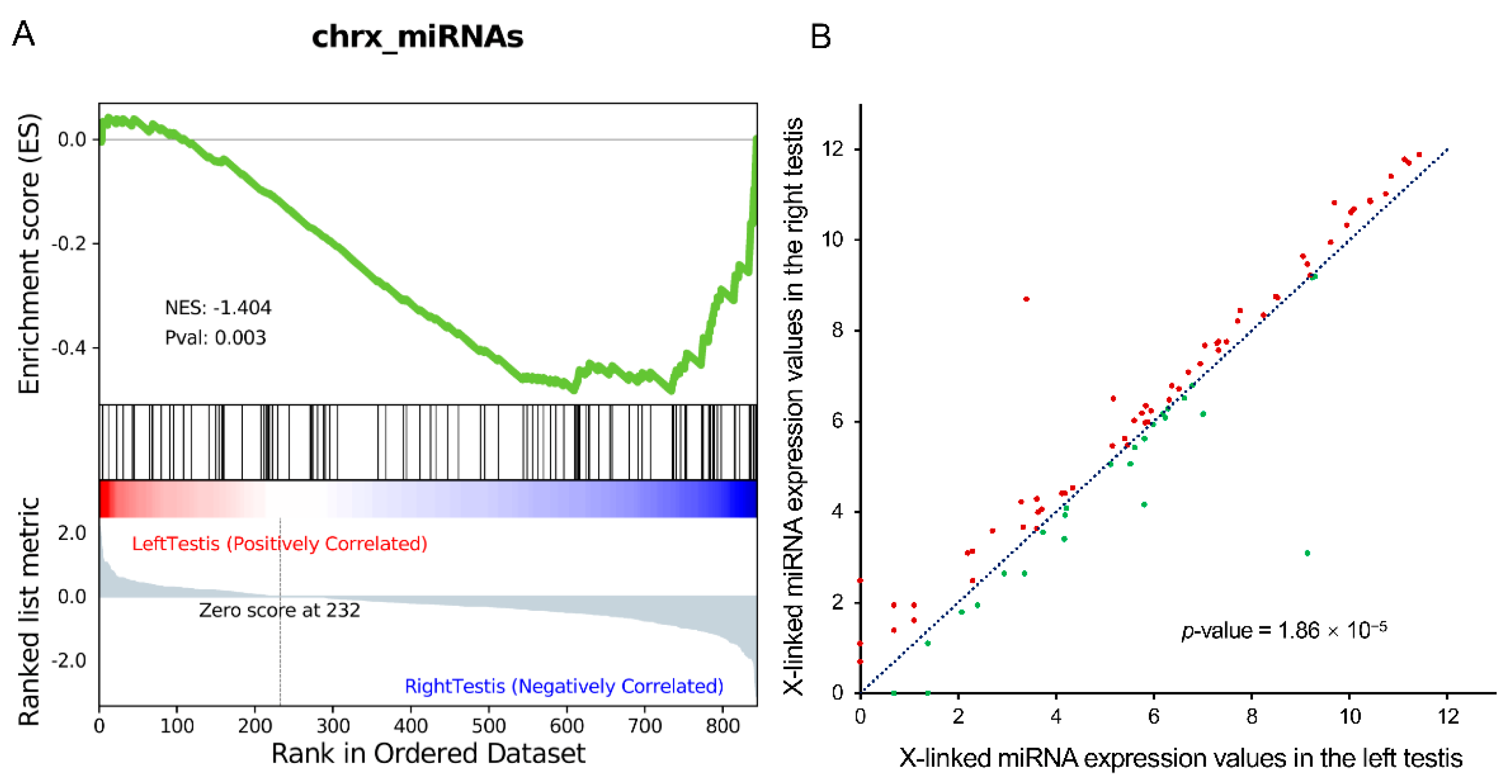
3.1. X-miRNAs Showed Significantly Lower Expression in the Left Testis
3.2. The Y:X Ratio Was Elevated in the Left Testis but Balanced in the Right Testis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dhanoa, J.K.; Mukhopadhyay, C.S.; Arora, J.S. Y-chromosomal genes affecting male fertility: A review. Veter World 2016, 9, 783–791. [Google Scholar] [CrossRef] [Green Version]
- Rahman, M.S.; Pang, M.G. New Biological Insights on X and Y Chromosome-Bearing Spermatozoa. Front. Cell Dev. Biol. 2019, 7, 388. [Google Scholar] [CrossRef]
- Malo, A.F.; Martinez-Pastor, F.; Garcia-Gonzalez, F.; Garde, J.; Ballou, J.D.; Lacy, R.C. A father effect explains sex-ratio bias. Proc. R. Soc. B Boil. Sci. 2017, 284, 20171159. [Google Scholar] [CrossRef] [PubMed]
- Douhard, M.; Geffroy, B. Males can adjust offspring sex ratio in an adaptive fashion through different mechanisms. BioEssays 2021, 43, 2000264. [Google Scholar] [CrossRef] [PubMed]
- You, Y.-A.; Kwon, W.-S.; Rahman, S.; Park, Y.-J.; Kim, Y.-J.; Pang, M.-G. Sex chromosome-dependent differential viability of human spermatozoa during prolonged incubation. Hum. Reprod. 2017, 32, 1183–1191. [Google Scholar] [CrossRef] [PubMed]
- Lavoie, M.D.; Tedeschi, J.N.; Garcia-Gonzalez, F.; Firman, R.C. Exposure to male-dominated environments during development influences sperm sex ratios at sexual maturity. Evol. Lett. 2019, 3, 392–402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Firman, R.C.; Tedeschi, J.N.; Garcia-Gonzalez, F. Sperm sex ratio adjustment in a mammal: Perceived male competition leads to elevated proportions of female-producing sperm. Biol. Lett. 2020, 16, 20190929. [Google Scholar] [CrossRef] [PubMed]
- Rathje, C.C.; Johnson, E.E.P.; Drage, D.; Patinioti, C.; Silvestri, G.; Affara, N.A.; Ialy-Radio, C.; Cocquet, J.; Skinner, B.M.; Ellis, P.J.I. Differential Sperm Motility Mediates the Sex Ratio Drive Shaping Mouse Sex Chromosome Evolution. Curr. Biol. 2019, 29, 3692–3698.e4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tiido, T.; Rignell-Hydbom, A.; Jönsson, B.; Giwercman, Y.L.; Rylander, L.; Hagmar, L.; Giwercman, A. Exposure to persistent organochlorine pollutants associates with human sperm Y:X chromosome ratio. Hum. Reprod. 2005, 20, 1903–1909. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kvist, L.; Giwercman, Y.L.; Jönsson, B.A.; Lindh, C.; Bonde, J.-P.; Toft, G.; Struciński, P.; Pedersen, H.S.; Zvyezday, V.; Giwercman, A. Serum levels of perfluorinated compounds and sperm Y:X chromosome ratio in two European populations and in Inuit from Greenland. Reprod. Toxicol. 2012, 34, 644–650. [Google Scholar] [CrossRef] [Green Version]
- Cui, C.; Huang, C.; Liu, K.; Xu, G.; Yang, J.; Zhou, Y.; Feng, Y.; Kararigas, G.; Geng, B.; Cui, Q. Large-scale in silico identification of drugs exerting sex-specific effects in the heart. J. Transl. Med. 2018, 16, 236. [Google Scholar] [CrossRef]
- Cui, C.; Yang, W.; Shi, J.; Zhou, Y.; Yang, J.; Cui, Q.; Zhou, Y. Identification and Analysis of Human Sex-biased MicroRNAs. Genom. Proteom. Bioinform. 2018, 16, 200–211. [Google Scholar] [CrossRef]
- Guo, S.; Zhou, Y.; Zeng, P.; Xu, G.; Wang, G.; Cui, Q. Identification and analysis of the human sex-biased genes. Briefings Bioinform. 2016, 19, 188–198. [Google Scholar] [CrossRef]
- Shi, J.; Hu, C.; Zhou, Y.; Cui, C.; Yang, J.; Cui, Q. MicroRNA Profiling in Paired Left and Right Eyes, Lungs, and Testes of Normal Mice. Mol. Ther. Nucleic Acids 2020, 21, 687–695. [Google Scholar] [CrossRef] [PubMed]
- Kozomara, A.; Birgaoanu, M.; Griffiths-Jones, S. miRBase: From microRNA sequences to function. Nucleic Acids Res. 2019, 47, D155–D162. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Cui, Q. sTAM: An Online Tool for the Discovery of miRNA-Set Level Disease Biomarkers. Mol. Ther. Nucleic Acids 2020, 21, 670–675. [Google Scholar] [CrossRef]
- Zhang, L.-F.; Tan-Tai, W.-J.; Li, X.-H.; Liu, M.-F.; Shi, H.-J.; Martin-DeLeon, P.A.; Wai-Sum, O.; Chen, H. PHB regulates meiotic recombination via JAK2-mediated histone modifications in spermatogenesis. Nucleic Acids Res. 2020, 48, 4780–4796. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Ru, Y.; Shi, H.-J.; Wang, Y.; Wu, B.; Upur, H.; Zhang, Y.-L. Cholecystokinin receptors regulate sperm protein tyrosine phosphorylation via uptake of HCO3−. Reproduction 2015, 150, 257–268. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Y.; Wu, F.; Zhang, M.; Xiong, Z.; Yin, Q.; Ru, Y.; Shi, H.; Li, J.; Mao, S.; Li, Y.; et al. EMC10 governs male fertility via maintaining sperm ion balance. J. Mol. Cell Biol. 2018, 10, 503–514. [Google Scholar] [CrossRef] [Green Version]
- Bennett, D.; Boyse, E.A. Sex ratio in progeny of mice inseminated with sperm treated with H-Y antiserum. Nature 1973, 246, 308–309. [Google Scholar] [CrossRef]
- Lin, S.-P.; Lee, R.K.-K.; Tsai, Y.-J.; Hwu, Y.-M.; Lin, M.-H. Separating X-Bearing Human Spermatozoa Through a Discontinuous Percoll Density Gradient Proved to Be Inefficient by Double-Label Fluorescent in situ Hybridization. J. Assist. Reprod. Genet. 1998, 15, 565–569. [Google Scholar] [CrossRef]
- Aleahmad, F.; Gourabi, H.; Zeinali, B.; Ashtiani, S.K.; Baharvand, H. Separation of X- and Y-bearing human spermatozoa by sperm isolation medium gradients evaluated by FISH. Reprod. Biomed. Online 2009, 18, 475–478. [Google Scholar] [CrossRef]
- Engelmann, U.; Krassnigg, F.; Schatz, H.; Schill, W.-B. Separation of human X and Y spermatozoa by free-flow electrophoresis. Gamete Res. 1988, 19, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Sang, L.; Yang, W.; Han, L.; Liang, A.; Hua, G.; Xiong, J.; Huo, L.; Yang, L. An immunological method to screen sex-specific proteins of bovine sperm. J. Dairy Sci. 2011, 94, 2060–2070. [Google Scholar] [CrossRef] [PubMed]
- Ellis, P.J.I.; Yu, Y.; Zhang, S. Transcriptional dynamics of the sex chromosomes and the search for offspring sex-specific antigens in sperm. Reproduction 2011, 142, 609–619. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, W.-C.; Sang, L.; Xiao, Y.; Zhang, H.-L.; Tang, K.-Q.; Yang, L.-G. Tentative identification of sex-specific antibodies and their application for screening bovine sperm proteins for sex-specificity. Mol. Biol. Rep. 2014, 41, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Garner, D.; Seidel, G. History of commercializing sexed semen for cattle. Theriogenology 2008, 69, 886–895. [Google Scholar] [CrossRef]
- Garner, D.L.; Evans, K.M.; Seidel, G.E. Sex-Sorting Sperm Using Flow Cytometry/Cell Sorting. Methods Mol. Biol. 2013, 927, 279–295. [Google Scholar] [CrossRef]
- Amann, R. Issues affecting commercialization of sexed sperm. Theriogenology 1999, 52, 1441–1457. [Google Scholar] [CrossRef]
- Pinkel, D.; Lake, S.; Gledhill, B.L.; Van Dilla, M.A.; Stephenson, D.; Watchmaker, G. High resolution DNA content measurements of mammalian sperm. Cytometry 1982, 3, 1–9. [Google Scholar] [CrossRef]
- Fugger, E. Clinical experience with flow cytometric separation of human X- and Y-chromosome bearing sperm. Theriogenology 1999, 52, 1435–1440. [Google Scholar] [CrossRef]
- Ortega-Ferrusola, C.; Gil, M.; Rodríguez-Martínez, H.; Anel, L.; Peña, F.J.; Martín-Muñoz, P. Flow cytometry in Spermatology: A bright future ahead. Reprod. Domest. Anim. 2017, 52, 921–931. [Google Scholar] [CrossRef] [PubMed]
- Noda, T.; Sakurai, N.; Nozawa, K.; Kobayashi, S.; Devlin, D.J.; Matzuk, M.M.; Ikawa, M. Nine genes abundantly expressed in the epididymis are not essential for male fecundity in mice. Andrology 2019, 7, 644–653. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, C.; Shi, J.; Chi, Y.; Yang, J.; Cui, Q. Y/X-Chromosome-Bearing Sperm Shows Elevated Ratio in the Left but Not the Right Testes in Healthy Mice. Life 2021, 11, 1219. https://doi.org/10.3390/life11111219
Hu C, Shi J, Chi Y, Yang J, Cui Q. Y/X-Chromosome-Bearing Sperm Shows Elevated Ratio in the Left but Not the Right Testes in Healthy Mice. Life. 2021; 11(11):1219. https://doi.org/10.3390/life11111219
Chicago/Turabian StyleHu, Chengqing, Jiangcheng Shi, Yujing Chi, Jichun Yang, and Qinghua Cui. 2021. "Y/X-Chromosome-Bearing Sperm Shows Elevated Ratio in the Left but Not the Right Testes in Healthy Mice" Life 11, no. 11: 1219. https://doi.org/10.3390/life11111219
APA StyleHu, C., Shi, J., Chi, Y., Yang, J., & Cui, Q. (2021). Y/X-Chromosome-Bearing Sperm Shows Elevated Ratio in the Left but Not the Right Testes in Healthy Mice. Life, 11(11), 1219. https://doi.org/10.3390/life11111219






