Biological and Mechanical Characterization of the Random Positioning Machine (RPM) for Microgravity Simulations
Abstract
:1. Introduction
2. Materials and Methods
2.1. Biological Characterization
2.1.1. Cell Cultures and Microgravity Simulation Setup
- Two T25 flasks to evaluate microgravity effects at the time point of 24 h after random positioning rotation;
- Two T25 flasks to evaluate microgravity effects at the time point of 72 h after random positioning rotation;
- Two T25 flasks as control (one for 24 h and one for 72 h) to be kept inside the incubator at the reference terrestrial gravity of 1 g.
2.1.2. Sample Collection
MTT Cell Viability Assay
2.1.3. RNA Extraction and Quantitative RT-PCR Analysis
2.2. Mechanical Characterization
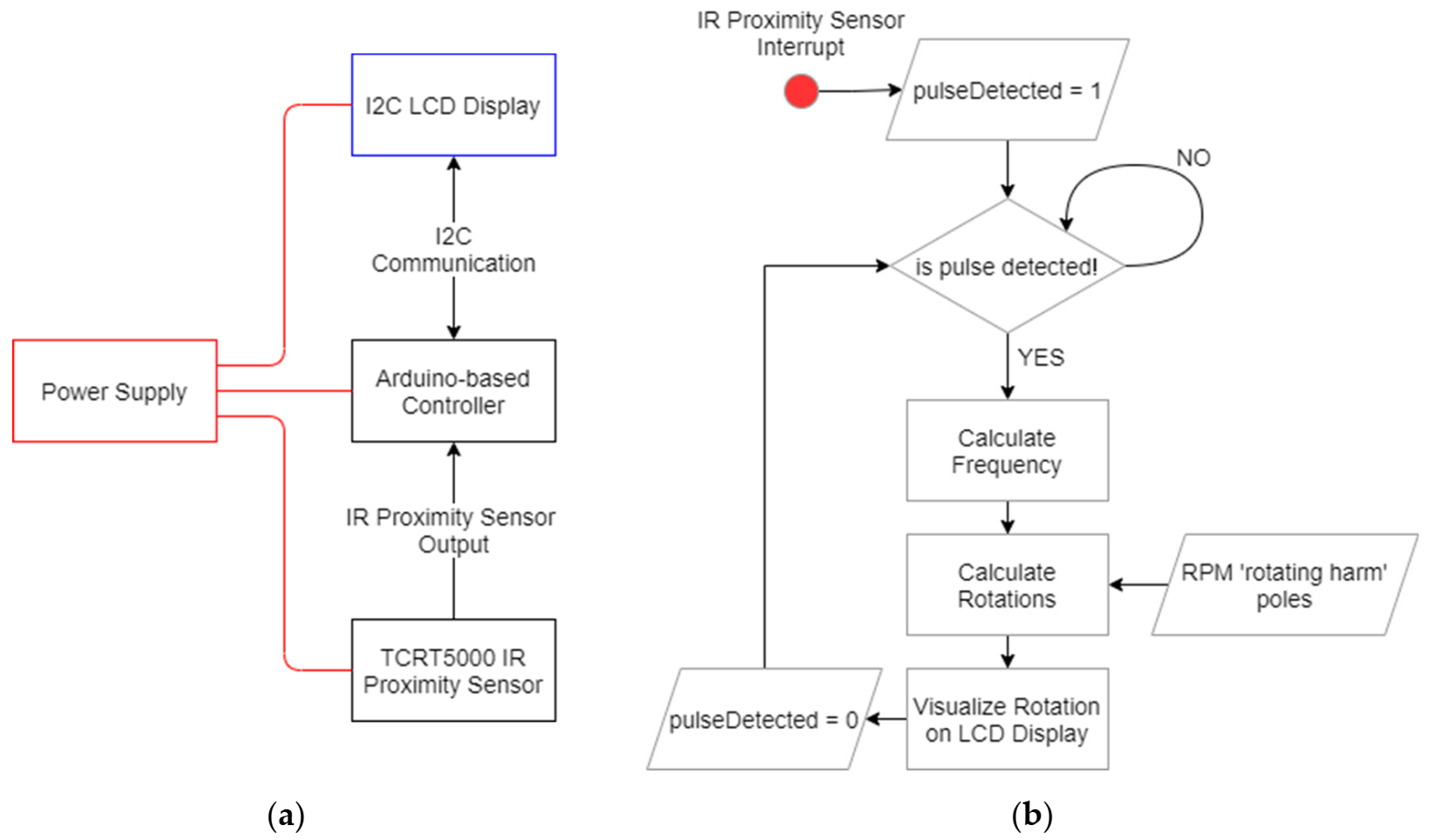
2.2.1. Rotations Measuring Device
- represents the time elapsed between two consecutive reflections (output signals) received by the TCRT5000 (Chipskey Technology Co., Shenzhen, Guangdong, China) proximity sensor;
- is the number of times the rotating part of the RPM is detected at each complete rotation (in our case poles = 2).
2.2.2. Acceleration Measuring Device
- Duration of the sampling (in minutes)—minutes;
- Sampling interval (in ms)—deltaT.
3. Results
3.1. Biological Characterization Findings
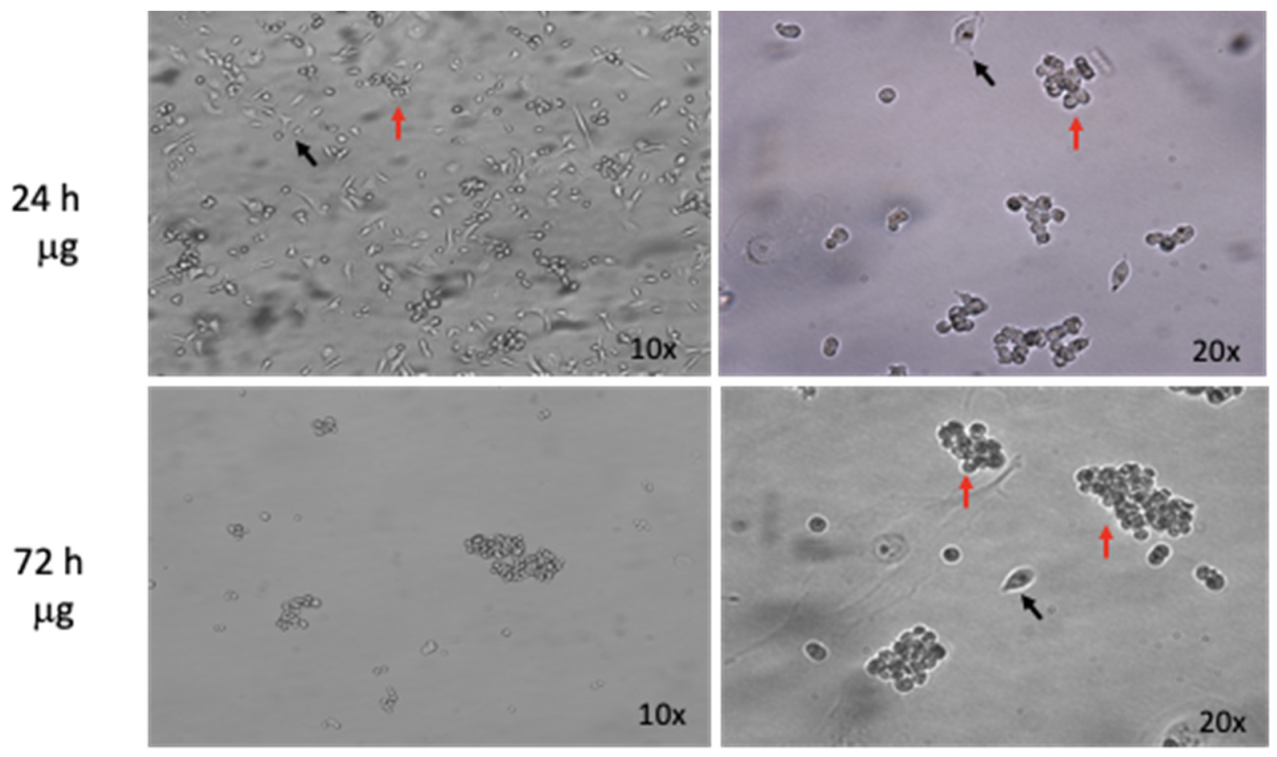
3.1.1. Microgravity Simulations Drive Multicellular Spheroids Formation
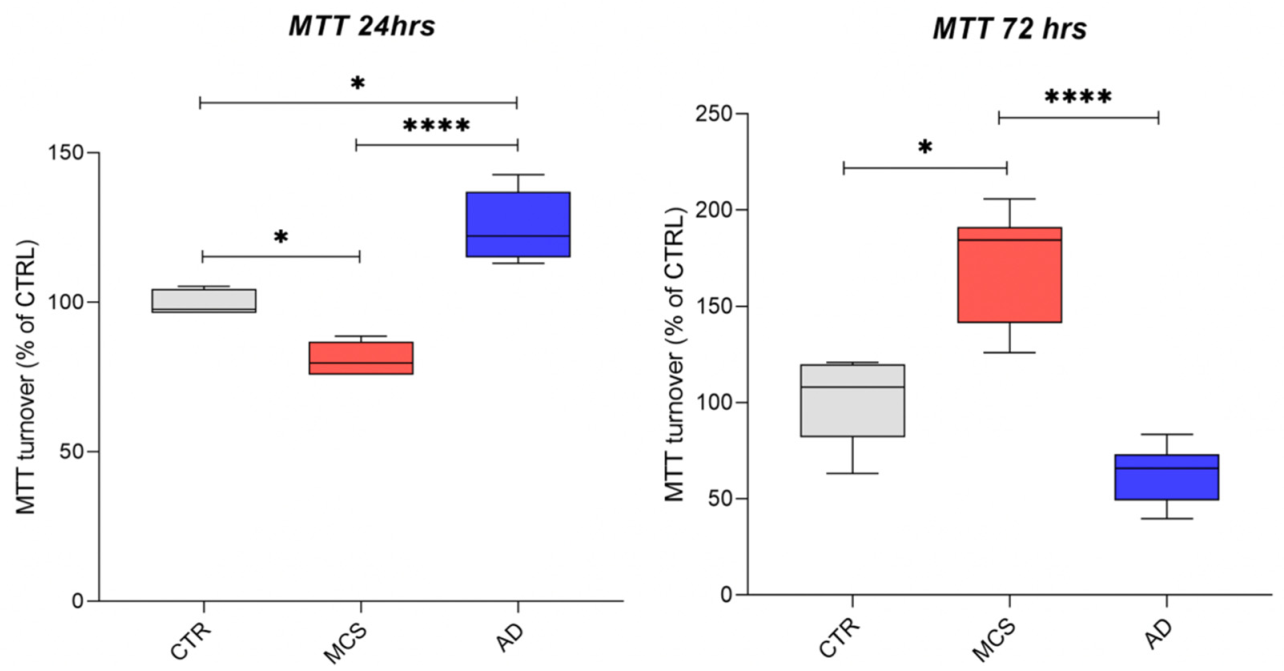
3.1.2. Cell Viability Is Differentially Affected by Simulated Microgravity
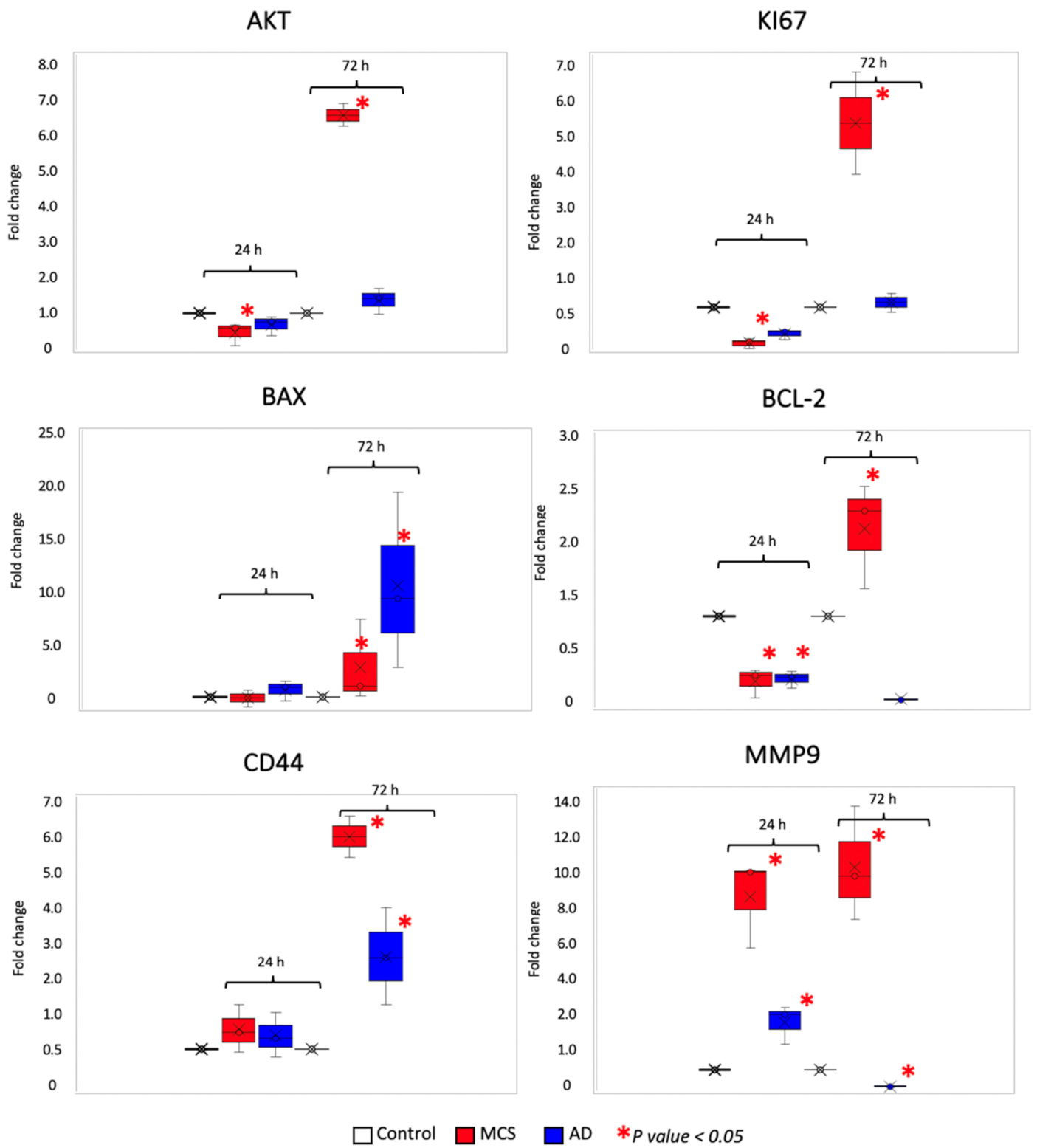
3.1.3. Gravity Unloading Influences Cell Behavior and Gene Expression
3.2. Mechanical Characterization Findings
- In the center of the rotating arm (referred as “centered position”);
- Halfway between the center and the lateral position (referred as “half-lateral position”);
- In lateral position (referred as “full-lateral position”).
- For the “centered” configuration the setting (innerRotations, outerRotations) = (120, 40) could be fine;
- For the “half-lateral” configuration it is preferable to move toward (innerRotations, outerRotations) = (115, 45);
- For the “full-lateral” configuration the setting (innerRotations, outerRotations) = (120, 40) could be fine, even if the set of minima of the averaged values seems to move toward (innerRotations, outerRotations) = (115, 35).
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pieters, C.M.; Goswami, J.N.; Clark, R.N.; Annadurai, M.; Boardman, J.; Buratti, B.; Combe, J.-P.; Dyar, M.D.; Green, R.; Head, J.W.; et al. Character and Spatial Distribution of OH/H2O on the Surface of the Moon Seen by M3 on Chandrayaan-1. Science 2009, 326, 568–572. [Google Scholar] [CrossRef] [PubMed]
- Diez, A. Liquid Water on Mars. Science 2018, 361, eaau1829. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Villanueva, M.; Wong, M.; Lu, T.; Zhang, Y.; Wu, H. Interplay of Space Radiation and Microgravity in DNA Damage and DNA Damage Response. NPJ Microgravity 2017, 3, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Michel, E.L.; Johnston, R.S.; Dietlein, L.F. Biomedical Results of the Skylab Program. Life Sci. Space Res. 1976, 14, 3–18. [Google Scholar] [PubMed]
- Montgomery, P.O., Jr.; Cook, J.E.; Reynolds, R.C.; Paul, J.S.; Hayflick, L.; Stock, D.; Schulz, W.W.; Kimsey, S.; Thirolf, R.G.; Rogers, T.; et al. The Response of Single Human Cells to Zero Gravity. In Vitro 1978, 14, 165–173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Naumann, R.J.; Lundquist, C.A.; Tandberg-Hanssen, E.; Horwitz, J.L.; Germany, G.A.; Cruise, J.F.; Lewis, M.L. Spacelab Science Results Study; NASA: Washington, DC, USA, 2009. [Google Scholar]
- Klaus, D.M. Clinostats and Bioreactors. Gravit. Space Biol. Bull. 2001, 14, 55–64. [Google Scholar] [PubMed]
- Becker, J.L.; Souza, G.R. Using Space-Based Investigations to Inform Cancer Research on Earth. Nat. Rev. Cancer 2013, 13, 315–327. [Google Scholar] [CrossRef]
- Borst, A.G.; van Loon, J.J.W.A. Technology and Developments for the Random Positioning Machine, RPM. Microgravity Sci. Technol. 2009, 21, 287–292. [Google Scholar] [CrossRef]
- Hammer, B.E.; Kidder, L.S.; Williams, P.C.; Xu, W.W. Magnetic Levitation of MC3T3 Osteoblast Cells as a Ground-Based Simulation of Microgravity. Microgravity Sci. Technol. 2009, 21, 311–318. [Google Scholar] [CrossRef] [Green Version]
- Anil-Inevi, M.; Yaman, S.; Yildiz, A.A.; Mese, G.; Yalcin-Ozuysal, O.; Cumhur Tekin, H.; Ozcivici, E. Biofabrication of in Situ Self Assembled 3D Cell Cultures in a Weightlessness Environment Generated Using Magnetic Levitation. Sci. Rep. 2018, 8, 7239. [Google Scholar] [CrossRef]
- McPherson, A.; DeLucas, L.J. Microgravity Protein Crystallization. NPJ Microgravity 2015, 1, 15010. [Google Scholar] [CrossRef]
- Senatore, G.; Mastroleo, F.; Leys, N.; Mauriello, G. Effect of Microgravity & Space Radiation on Microbes. Future Microbiol. 2018, 13, 831–847. [Google Scholar]
- Long, J.P.; Pierson, S.; Hughes, J.H. Rhinovirus Replication in HeLa Cells Cultured under Conditions of Simulated Microgravity. Aviat. Space Environ. Med. 1998, 69, 851–856. [Google Scholar]
- Demontis, G.C.; Germani, M.M.; Caiani, E.G.; Barravecchia, I.; Passino, C.; Angeloni, D. Human Pathophysiological Adaptations to the Space Environment. Front. Physiol. 2017, 8, 547. [Google Scholar] [CrossRef]
- Grimm, D.; Wehland, M.; Corydon, T.J.; Richter, P.; Prasad, B.; Bauer, J.; Egli, M.; Kopp, S.; Lebert, M.; Krüger, M. The Effects of Microgravity on Differentiation and Cell Growth in Stem Cells and Cancer Stem Cells. Stem Cells Transl. Med. 2020, 9, 882–894. [Google Scholar] [CrossRef]
- Bravatà, V.; Cammarata, F.P.; Minafra, L.; Pisciotta, P.; Scazzone, C.; Manti, L.; Savoca, G.; Petringa, G.; Cirrone, G.A.P.; Cuttone, G.; et al. Proton-Irradiated Breast Cells: Molecular Points of View. J. Radiat. Res. 2019, 60, 451–465. [Google Scholar] [CrossRef]
- Cammarata, F.P.; Forte, G.I.; Broggi, G.; Bravatà, V.; Minafra, L.; Pisciotta, P.; Calvaruso, M.; Tringali, R.; Tomasello, B.; Torrisi, F.; et al. Molecular Investigation on a Triple Negative Breast Cancer Xenograft Model Exposed to Proton Beams. Int. J. Mol. Sci. 2020, 21, 6337. [Google Scholar] [CrossRef]
- Bravatà, V.; Cammarata, F.P.; Minafra, L.; Musso, R.; Pucci, G.; Spada, M.; Fazio, I.; Russo, G.; Forte, G.I. Gene Expression Profiles Induced by High-Dose Ionizing Radiation in MDA-MB-231 Triple-Negative Breast Cancer Cell Line. Cancer Genom. Proteom. 2019, 16, 257–266. [Google Scholar] [CrossRef] [Green Version]
- Lebert, J.M.; Lester, R.; Powell, E.; Seal, M.; McCarthy, J. Advances in the Systemic Treatment of Triple-Negative Breast Cancer. Curr. Oncol. 2018, 25, S142–S150. [Google Scholar] [CrossRef] [Green Version]
- Savoca, G.; Calvaruso, M.; Minafra, L.; Bravatà, V.; Cammarata, F.P.; Iacoviello, G.; Abbate, B.; Evangelista, G.; Spada, M.; Forte, G.I.; et al. Local Disease-Free Survival Rate (LSR) Application to Personalize Radiation Therapy Treatments in Breast Cancer Models. J. Pers. Med. 2020, 10, 177. [Google Scholar] [CrossRef]
- Hybel, T.E.; Dietrichs, D.; Sahana, J.; Corydon, T.J.; Nassef, M.Z.; Wehland, M.; Krüger, M.; Magnusson, N.E.; Bauer, J.; Utpatel, K.; et al. Simulated Microgravity Influences VEGF, MAPK, and PAM Signaling in Prostate Cancer Cells. Int. J. Mol. Sci. 2020, 21, 1263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vicario, N.; Bernstock, J.D.; Spitale, F.M.; Giallongo, C.; Giunta, M.A.S.; Li Volti, G.; Gulisano, M.; Leanza, G.; Tibullo, D.; Parenti, R.; et al. Clobetasol Modulates Adult Neural Stem Cell Growth via Canonical Hedgehog Pathway Activation. Int. J. Mol. Sci. 2019, 20, 1991. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Warnke, E.; Pietsch, J.; Wehland, M.; Bauer, J.; Infanger, M.; Görög, M.; Hemmersbach, R.; Braun, M.; Ma, X.; Sahana, J.; et al. Spheroid Formation of Human Thyroid Cancer Cells under Simulated Microgravity: A Possible Role of CTGF and CAV1. Cell Commun. Signal. 2014, 12, 32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pfeffer, C.; Singh, A. Apoptosis: A Target for Anticancer Therapy. Int. J. Mol. Sci. 2018, 19, 448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chancellor, J.C.; Blue, R.S.; Cengel, K.A.; Auñón-Chancellor, S.M.; Rubins, K.H.; Katzgraber, H.G.; Kennedy, A.R. Limitations in Predicting the Space Radiation Health Risk for Exploration Astronauts. NPJ Microgravity 2018, 4, 8. [Google Scholar] [CrossRef]
- Bailey, S.M.; Luxton, J.J.; McKenna, M.J.; Taylor, L.E.; George, K.A.; Jhavar, S.G.; Swanson, G.P. Ad Astra—Telomeres in Space! Int. J. Radiat. Biol. 2021, 26, 1–9. [Google Scholar] [CrossRef]
- Ong, J.; Lee, A.G.; Moss, H.E. Head-Down Tilt Bed Rest Studies as a Terrestrial Analog for Spaceflight Associated Neuro-Ocular Syndrome. Front. Neurol. 2021, 12, 648958. [Google Scholar] [CrossRef]
- Basner, M.; Dinges, D.F.; Howard, K.; Moore, T.M.; Gur, R.C.; Mühl, C.; Stahn, A.C. Continuous and Intermittent Artificial Gravity as a Countermeasure to the Cognitive Effects of 60 Days of Head-Down Tilt Bed Rest. Front. Physiol. 2021, 12, 643854. [Google Scholar] [CrossRef]
- Topal, U.; Zamur, C. Microgravity, Stem Cells, and Cancer: A New Hope for Cancer Treatment. Stem Cells Int. 2021, 2021, 5566872. [Google Scholar] [CrossRef]
- Lüönd, F.; Tiede, S.; Christofori, G. Breast Cancer as an Example of Tumour Heterogeneity and Tumour Cell Plasticity during Malignant Progression. Br. J. Cancer 2021, 125, 164–175. [Google Scholar] [CrossRef]
- Minafra, L.; Porcino, N.; Bravatà, V.; Gaglio, D.; Bonanomi, M.; Amore, E.; Cammarata, F.P.; Russo, G.; Militello, C.; Savoca, G.; et al. Radiosensitizing Effect of Curcumin-Loaded Lipid Nanoparticles in Breast Cancer Cells. Sci. Rep. 2019, 9, 11134. [Google Scholar] [CrossRef] [Green Version]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [Green Version]
- Timmins, N.E.; Nielsen, L.K. Generation of Multicellular Tumor Spheroids by the Hanging-Drop Method. Methods Mol. Med. 2007, 140, 141–151. [Google Scholar]
- Rijal, G.; Li, W. A Versatile 3D Tissue Matrix Scaffold System for Tumor Modeling and Drug Screening. Sci Adv. 2017, 3, e1700764. [Google Scholar] [CrossRef] [Green Version]
- Langhans, S.A. Three-Dimensional in Vitro Cell Culture Models in Drug Discovery and Drug Repositioning. Front. Pharmacol. 2018, 9, 6. [Google Scholar] [CrossRef]
- Ahn, C.B.; Lee, J.-H.; Han, D.G.; Kang, H.-W.; Lee, S.-H.; Lee, J.-I.; Son, K.H.; Lee, J.W. Simulated Microgravity with Floating Environment Promotes Migration of Non-Small Cell Lung Cancers. Sci. Rep. 2019, 9, 14553. [Google Scholar] [CrossRef]
- Zhao, T.; Li, R.; Tan, X.; Zhang, J.; Fan, C.; Zhao, Q.; Deng, Y.; Xu, A.; Lukong, K.E.; Genth, H.; et al. Simulated Microgravity Reduces Focal Adhesions and Alters Cytoskeleton and Nuclear Positioning Leading to Enhanced Apoptosis via Suppressing FAK/RhoA-Mediated mTORC1/NF-κB and ERK1/2 Pathways. Int. J. Mol. Sci. 2018, 19, 1994. [Google Scholar] [CrossRef] [Green Version]
- Song, M.; Bode, A.M.; Dong, Z.; Lee, M.-H. AKT as a Therapeutic Target for Cancer. Cancer Res. 2019, 79, 1019–1031. [Google Scholar] [CrossRef] [Green Version]
- Pascual, J.; Turner, N.C. Targeting the PI3-Kinase Pathway in Triple-Negative Breast Cancer. Ann. Oncol. 2019, 30, 1051–1060. [Google Scholar] [CrossRef] [Green Version]
- Wu, Q.; Ma, G.; Deng, Y.; Luo, W.; Zhao, Y.; Li, W.; Zhou, Q. Prognostic Value of Ki-67 in Patients with Resected Triple-Negative Breast Cancer: A Meta-Analysis. Front. Oncol. 2019, 9, 1068. [Google Scholar] [CrossRef] [Green Version]
- Däster, S.; Amatruda, N.; Calabrese, D.; Ivanek, R.; Turrini, E.; Droeser, R.A.; Zajac, P.; Fimognari, C.; Spagnoli, G.C.; Iezzi, G.; et al. Induction of Hypoxia and Necrosis in Multicellular Tumor Spheroids Is Associated with Resistance to Chemotherapy Treatment. Oncotarget 2017, 8, 1725–1736. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peng, F.; Xiong, L.; Xie, X.; Tang, H.; Huang, R.; Peng, C. Isoliquiritigenin Derivative Regulates miR-374a/BAX Axis to Suppress Triple-Negative Breast Cancer Tumorigenesis and Development. Front. Pharmacol. 2020, 11, 378. [Google Scholar] [CrossRef] [PubMed]
- Inao, T.; Iida, Y.; Moritani, T.; Okimoto, T.; Tanino, R.; Kotani, H.; Harada, M. Bcl-2 Inhibition Sensitizes Triple-Negative Human Breast Cancer Cells to Doxorubicin. Oncotarget 2018, 9, 25545–25556. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Misra, S.; Hascall, V.C.; Markwald, R.R.; Ghatak, S. Interactions between Hyaluronan and Its Receptors (CD44, RHAMM) Regulate the Activities of Inflammation and Cancer. Front. Immunol. 2015, 6, 201. [Google Scholar] [CrossRef] [Green Version]
- Zou, W.; Yang, Y.; Zheng, R.; Wang, Z.; Zeng, H.; Chen, Z.; Yang, F.; Wang, J. Association of CD44 and CD24 Phenotype with Lymph Node Metastasis and Survival in Triple-Negative Breast Cancer. Int. J. Clin. Exp. Pathol. 2020, 13, 1008–1016. [Google Scholar]
- Barillari, G. The Impact of Matrix Metalloproteinase-9 on the Sequential Steps of the Metastatic Process. Int. J. Mol. Sci. 2020, 21, 4526. [Google Scholar] [CrossRef]
- Huang, H. Matrix Metalloproteinase-9 (MMP-9) as a Cancer Biomarker and MMP-9 Biosensors: Recent Advances. Sensors 2018, 18, 3249. [Google Scholar] [CrossRef] [Green Version]
- Mehner, C.; Hockla, A.; Miller, E.; Ran, S.; Radisky, D.C.; Radisky, E.S. Tumor Cell-Produced Matrix Metalloproteinase 9 (MMP-9) Drives Malignant Progression and Metastasis of Basal-like Triple Negative Breast Cancer. Oncotarget 2014, 5, 2736–2749. [Google Scholar] [CrossRef] [Green Version]
- Vorselen, D.; Roos, W.H.; MacKintosh, F.C.; Wuite, G.J.L.; van Loon, J.J.W.A. The Role of the Cytoskeleton in Sensing Changes in Gravity by Nonspecialized Cells. FASEB J. 2014, 28, 536–547. [Google Scholar] [CrossRef] [Green Version]
- Arun, R.P.; Sivanesan, D.; Patra, B.; Varadaraj, S.; Verma, R.S. Simulated Microgravity Increases Polyploid Giant Cancer Cells and Nuclear Localization of YAP. Sci. Rep. 2019, 9, 10684. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.; Kwon, H.; Shin, Y.K.; Song, G.; Lee, T.; Kim, Y.; Jeong, W.; Lee, U.; Zhang, X.; Nam, G.; et al. MAML1/2 Promote YAP/TAZ Nuclear Localization and Tumorigenesis. Proc. Natl. Acad. Sci. USA 2020, 117, 13529–13540. [Google Scholar] [CrossRef]
- Kim, Y.J.; Jeong, A.J.; Kim, M.; Lee, C.; Ye, S.-K.; Kim, S. Time-Averaged Simulated Microgravity (taSMG) Inhibits Proliferation of Lymphoma Cells, L-540 and HDLM-2, Using a 3D Clinostat. Biomed. Eng. Online 2017, 16, 48. [Google Scholar] [CrossRef] [Green Version]
- Deng, B.; Liu, R.; Tian, X.; Han, Z.; Chen, J. Simulated Microgravity Inhibits the Viability and Migration of Glioma via FAK/RhoA/Rock and FAK/Nek2 Signaling. In Vitro Cell. Dev. Biol. Anim. 2019, 55, 260–271. [Google Scholar] [CrossRef]
- Masiello, M.G.; Cucina, A.; Proietti, S.; Palombo, A.; Coluccia, P.; D’Anselmi, F.; Dinicola, S.; Pasqualato, A.; Morini, V.; Bizzarri, M. Phenotypic Switch Induced by Simulated Microgravity on MDA-MB-231 Breast Cancer Cells. Biomed Res. Int. 2014, 2014, 652434. [Google Scholar] [CrossRef]
- Wuest, S.L.; Stern, P.; Casartelli, E.; Egli, M. Fluid Dynamics Appearing during Simulated Microgravity Using Random Positioning Machines. PLoS ONE 2017, 12, e0170826. [Google Scholar] [CrossRef]
- Hauslage, J.; Cevik, V.; Hemmersbach, R. Pyrocystis Noctiluca Represents an Excellent Bioassay for Shear Forces Induced in Ground-Based Microgravity Simulators (clinostat and Random Positioning Machine). NPJ Microgravity 2017, 3, 12. [Google Scholar] [CrossRef] [Green Version]



| Gene Symbol | Forward Primer 5′> 3′ | Reverse Primer 5′ > 3′ |
|---|---|---|
| AKT | caaggacgggcacattaaga | gccgtagtcattgtcctcca |
| Ki67 | cgtcccagtggaagagttgt | accccgctccttttgatagt |
| BAX | gaaccatcatgggctgga | cgtcccaaagtaggagag |
| Bcl2 | ttgacagaggatcatgctgta | atctttatttcatgaggcacgtt |
| CD44 | aacatggtccattcacct | agaggaagggtgtgctc |
| MMP9 | gagaccggtgagctggata | tacacgcgagtgaaggtgag |
| RN18S1(rRNA18S) | cggacaggattgacagattga | agagtctcgttcgttatcgga |
| “Centered” Position | |||||||||||
| (inner, outer) = (110, 30) | (inner, outer) = (110, 40) | (inner, outer) = (110, 50) | |||||||||
| accX | accY | accZ | accX | accY | accZ | accX | accY | accZ | |||
| mean | 0.0567 | 0.0256 | 0.0859 | mean | 0.0571 | 0.0207 | 0.0854 | mean | 0.0579 | 0.0228 | 0.0874 |
| SD | 0.5002 | 0.2679 | 0.2732 | SD | 0.4974 | 0.2709 | 0.275 | SD | 0.4977 | 0.269 | 0.2736 |
| (inner, outer) = (120, 30) | (inner, outer) = (120, 40) | (inner, outer) = (120, 50) | |||||||||
| accX | accY | accZ | accX | accY | accZ | accX | accY | accZ | |||
| mean | 0.0499 | 0.0252 | 0.0877 | mean | 0.0565 | 0.0173 | 0.0838 | mean | 0.0639 | 0.0233 | 0.0869 |
| SD | 0.4995 | 0.2659 | 0.2718 | SD | 0.4797 | 0.2845 | 0.2723 | SD | 0.5011 | 0.2661 | 0.2707 |
| (inner, outer) = (130, 30) | (inner, outer) = (130, 40) | (inner, outer) = (130, 50) | |||||||||
| accX | accY | accZ | accX | accY | accZ | accX | accY | accZ | |||
| mean | 0.0537 | 0.0316 | 0.0761 | mean | 0.0557 | 0.0229 | 0.0863 | mean | 0.0615 | 0.0236 | 0.0794 |
| SD | 0.4842 | 0.282 | 0.261 | SD | 0.4970 | 0.2621 | 0.272 | SD | 0.499 | 0.2629 | 0.2718 |
| “Half-Lateral” Position | |||||||||||
| (inner, outer) = (110, 30) | (inner, outer) = (110, 40) | (inner, outer) = (110, 50) | |||||||||
| accX | accY | accZ | accX | accY | accZ | accX | accY | accZ | |||
| mean | 0.1194 | 0.0152 | 0.0821 | mean | 0.098 | 0.008 | 0.0807 | mean | 0.0774 | 0.0091 | 0.0952 |
| SD | 0.5006 | 0.2626 | 0.2725 | SD | 0.4995 | 0.2644 | 0.2748 | SD | 0.5017 | 0.2626 | 0.2716 |
| (inner, outer) = (120, 30) | (inner, outer) = (120, 40) | (inner, outer) = (120, 50) | |||||||||
| accX | accY | accZ | accX | accY | accZ | accX | accY | accZ | |||
| mean | 0.119 | 0.0104 | 0.0833 | mean | 0.0965 | 0.0086 | 0.0922 | mean | 0.0707 | 0.0067 | 0.0947 |
| SD | 0.5011 | 0.2646 | 0.273 | SD | 0.5058 | 0.2554 | 0.2749 | SD | 0.4998 | 0.2651 | 0.2732 |
| (inner, outer) = (130, 30) | (inner, outer) = (130, 40) | (inner, outer) = (130, 50) | |||||||||
| accX | accY | accZ | accX | accY | accZ | accX | accY | accZ | |||
| mean | 0.1202 | 0.0105 | 0.0880 | mean | 0.0948 | 0.0089 | 0.0926 | mean | 0.0815 | 0.0021 | 0.0964 |
| SD | 0.4999 | 0.2628 | 0.2729 | SD | 0.4994 | 0.2611 | 0.2723 | SD | 0.4969 | 0.2655 | 0.2737 |
| “Full-Lateral” Position | |||||||||||
| (inner, outer) = (110, 30) | (inner, outer) = (110, 40) | (inner, outer) = (110, 50) | |||||||||
| accX | accY | accZ | accX | accY | accZ | accX | accY | accZ | |||
| mean | 0.0076 | 0.0167 | 0.0901 | mean | −0.0181 | 0.0187 | 0.0895 | mean | −0.0614 | 0.0242 | 0.089 |
| SD | 0.4973 | 0.2695 | 0.273 | SD | 0.4999 | 0.2682 | 0.2751 | SD | 0.4991 | 0.2666 | 0.2704 |
| (inner, outer) = (120, 30) | (inner, outer) = (120, 40) | (inner, outer) = (120, 50) | |||||||||
| accX | accY | accZ | accX | accY | accZ | accX | accY | accZ | |||
| mean | 0.0071 | 0.0132 | 0.0923 | mean | −0.018 | 0.0151 | 0.0898 | mean | −0.0618 | 0.0202 | 0.0934 |
| SD | 0.4995 | 0.2718 | 0.271 | SD | 0.4961 | 0.2743 | 0.2700 | SD | 0.4976 | 0.2725 | 0.2729 |
| (inner, outer) = (130, 30) | (inner, outer) = (130, 40) | (inner, outer) = (130, 50) | |||||||||
| accX | accY | accZ | accX | accY | accZ | accX | accY | accZ | |||
| mean | 0.0024 | 0.0182 | 0.0936 | mean | −0.0216 | 0.0225 | 0.0918 | mean | −0.0615 | 0.0199 | 0.0923 |
| SD | 0.4997 | 0.2667 | 0.2713 | SD | 0.4957 | 0.2679 | 0.2733 | SD | 0.4981 | 0.2654 | 0.271 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calvaruso, M.; Militello, C.; Minafra, L.; La Regina, V.; Torrisi, F.; Pucci, G.; Cammarata, F.P.; Bravatà, V.; Forte, G.I.; Russo, G. Biological and Mechanical Characterization of the Random Positioning Machine (RPM) for Microgravity Simulations. Life 2021, 11, 1190. https://doi.org/10.3390/life11111190
Calvaruso M, Militello C, Minafra L, La Regina V, Torrisi F, Pucci G, Cammarata FP, Bravatà V, Forte GI, Russo G. Biological and Mechanical Characterization of the Random Positioning Machine (RPM) for Microgravity Simulations. Life. 2021; 11(11):1190. https://doi.org/10.3390/life11111190
Chicago/Turabian StyleCalvaruso, Marco, Carmelo Militello, Luigi Minafra, Veronica La Regina, Filippo Torrisi, Gaia Pucci, Francesco P. Cammarata, Valentina Bravatà, Giusi I. Forte, and Giorgio Russo. 2021. "Biological and Mechanical Characterization of the Random Positioning Machine (RPM) for Microgravity Simulations" Life 11, no. 11: 1190. https://doi.org/10.3390/life11111190
APA StyleCalvaruso, M., Militello, C., Minafra, L., La Regina, V., Torrisi, F., Pucci, G., Cammarata, F. P., Bravatà, V., Forte, G. I., & Russo, G. (2021). Biological and Mechanical Characterization of the Random Positioning Machine (RPM) for Microgravity Simulations. Life, 11(11), 1190. https://doi.org/10.3390/life11111190













