Exercise Preconditioning Attenuates the Response to Experimental Colitis and Modifies Composition of Gut Microbiota in Wild-Type Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
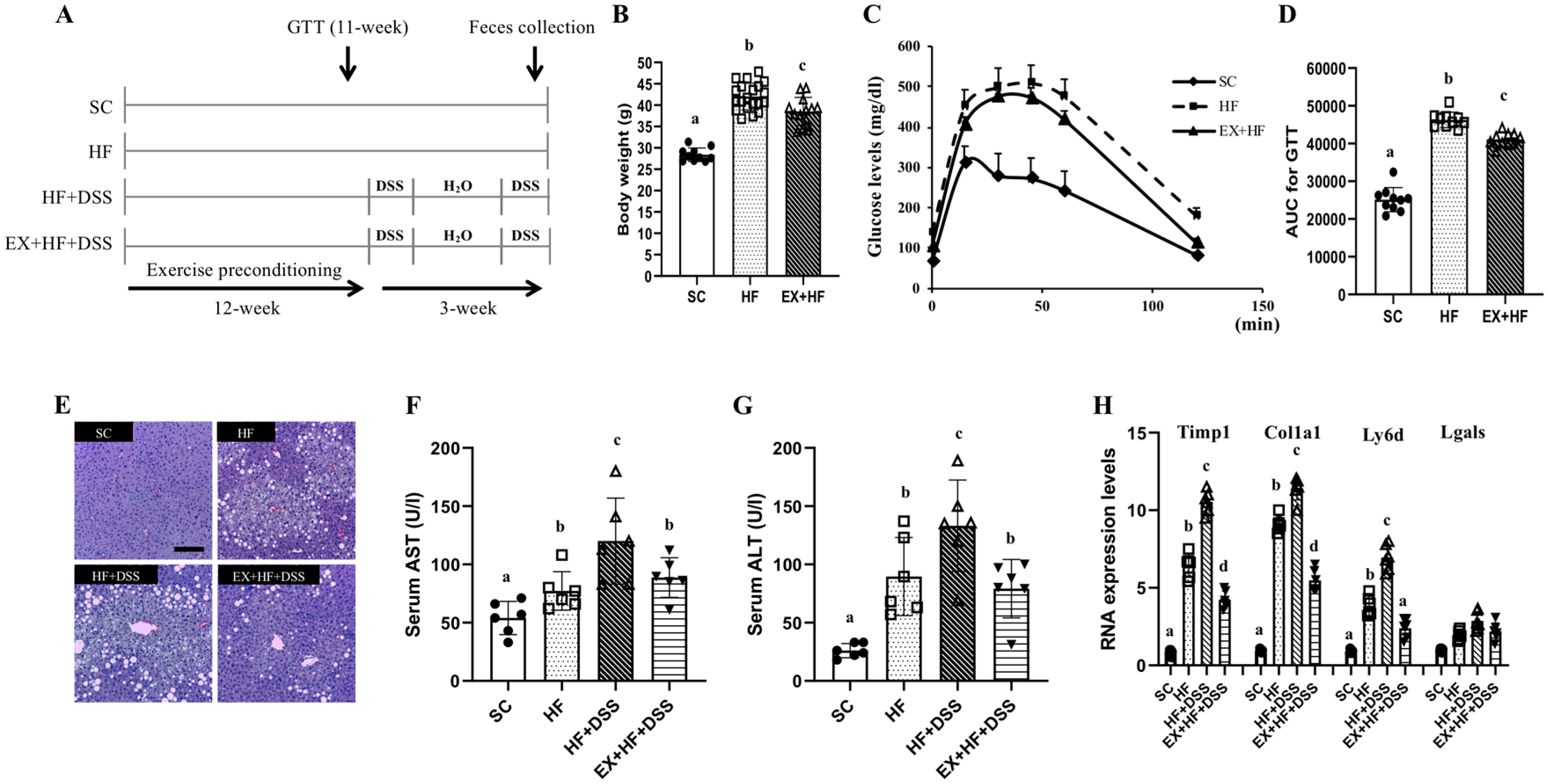
2.2. Experimental Design
2.3. Glucose Tolerance Test
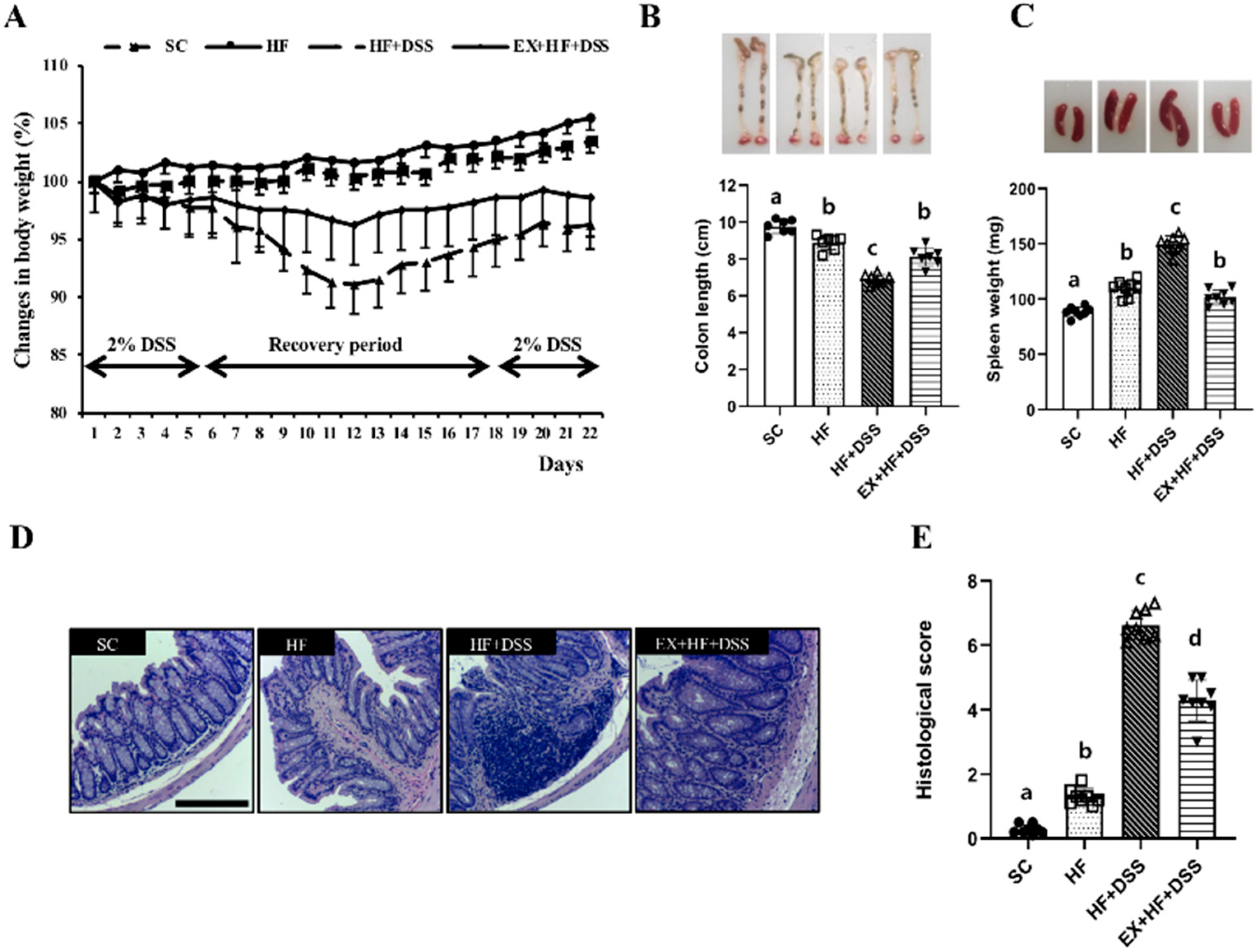
2.4. DSS-Induced Colitis Model
2.5. Blood and Tissue Sampling
2.6. Biochemical Analyses
2.7. Histopathology and Colitis Scoring
2.8. Real-Time PCR
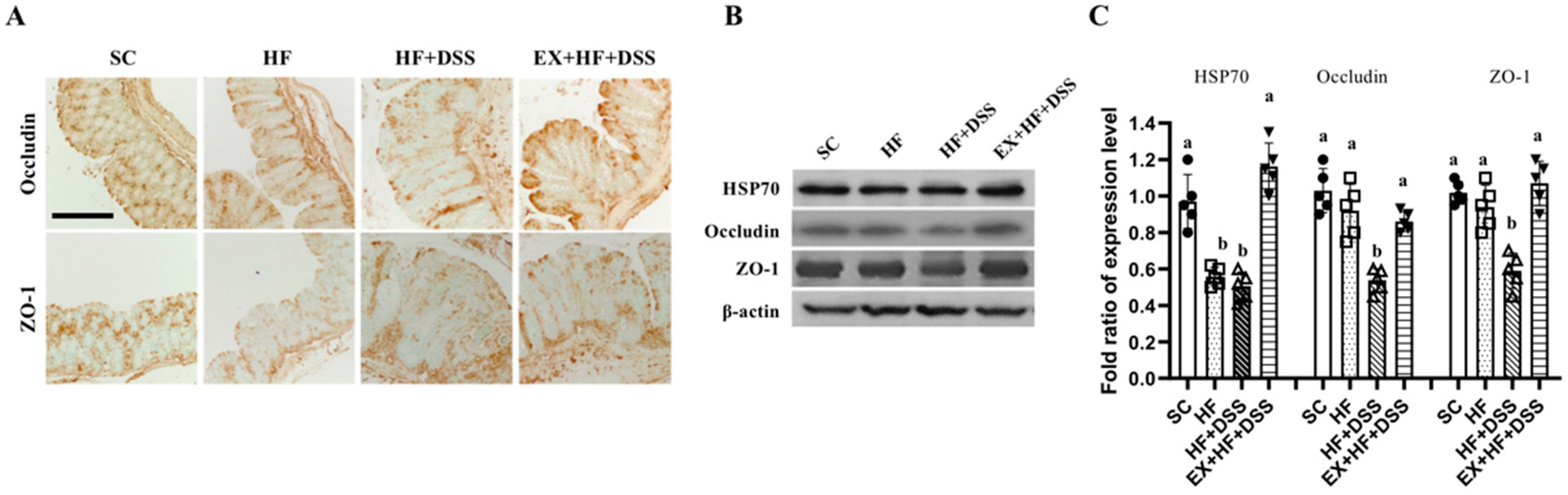
2.9. Western Blot
2.10. Flow Cytometry Analysis
2.11. 16 S rRNA Gene and Metagenome Sequencing
2.12. Metagenome Sequences Analysis
2.13. Statistical Analysis
3. Results
3.1. Exercise Preconditioning Alleviates Metabolic Complications Associated with HF+DSS-induced Colitis
3.2. Exercise Preconditioning Alleviates the Colitis Symptoms and Histological Changes Associated with HF+DSS-induced Colitis
3.3. Exercise Preconditioning Prevents Altered Expression of Gut Barrier Proteins Associated with HF+DSS-induced Colitis
3.4. Exercise Training Alleviates Immune and Inflammatory Responses Associated with HF+DSS-induced Colitis
3.5. Exercise Preconditioning Modifies Composition of Gut Microbiota
3.6. Exercise Preconditioning-induced Modifications in Gut Microbiota and Their Potential Associations with HF+DSS-induced Colitis
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| HF | High fat |
| SC | Standard chow |
| DSS | Dextran sodium sulfate |
| EX | Exercise training |
| IBD | Inflammatory bowel disease |
| HSP | Heat shock protein |
| GTT | Glucose tolerance test |
| ALT | Alanine aminotransferase |
| AST | Aspartate aminotransferase |
| IL-6 | Interleukin-6 |
| MCP | Monocyte chemoattractant protein |
| GRO | Growth-regulated protein |
| ZO | Zonular occudens |
| TLR | Toll-like receptor |
| qPCR | Quantitative polymerase chain reaction |
| PCoA | Principal coordinates analysis |
| OUT | Operational taxonomic unit |
| FDR | False discovery rate |
References
- Barreiro-de Acosta, M.; Alvarez Castro, A.; Souto, R.; Iglesias, M.; Lorenzo, A.; Dominguez-Munoz, J.E. Emigration to western industrialized countries: A risk factor for developing inflammatory bowel disease. J. Crohns Colitis 2011, 5, 566–569. [Google Scholar] [CrossRef] [PubMed]
- Ananthakrishnan, A.N. Epidemiology and risk factors for IBD. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 205–217. [Google Scholar] [CrossRef] [PubMed]
- Michielan, A.; D’Inca, R. Intestinal permeability in inflammatory bowel disease: Pathogenesis, clinical evaluation, and therapy of leaky gut. Mediat. Inflamm. 2015, 2015, 628157. [Google Scholar] [CrossRef]
- Cheng, L.; Jin, H.; Qiang, Y.; Wu, S.; Yan, C.; Han, M.; Xiao, T.; Yan, N.; An, H.; Zhou, X.; et al. High fat diet exacerbates dextran sulfate sodium induced colitis through disturbing mucosal dendritic cell homeostasis. Int. Immunopharmacol. 2016, 40, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Loftus, E.V., Jr. Clinical epidemiology of inflammatory bowel disease: Incidence, prevalence, and environmental influences. Gastroenterology 2004, 126, 1504–1517. [Google Scholar] [CrossRef] [PubMed]
- Ni, J.; Wu, G.D.; Albenberg, L.; Tomov, V.T. Gut microbiota and IBD: Causation or correlation? Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 573–584. [Google Scholar] [CrossRef] [PubMed]
- Romeo, J.; Warnberg, J.; Pozo, T.; Marcos, A. Physical activity, immunity and infection. Proc. Nutr. Soc. 2010, 69, 390–399. [Google Scholar] [CrossRef]
- Tew, G.A.; Jones, K.; Mikocka-Walus, A. Physical activity habits, limitations, and predictors in people with inflammatory bowel disease: A large cross-sectional online survey. Inflamm. Bowel Dis. 2016, 22, 2933–2942. [Google Scholar] [CrossRef]
- Wischmeyer, P.E.; Kahana, M.; Wolfson, R.; Ren, H.; Musch, M.M.; Chang, E.B. Glutamine induces heat shock protein and protects against endotoxin shock in the rat. J. Appl. Physiol. 2001, 90, 2403–2410. [Google Scholar] [CrossRef]
- Mazur-Bialy, A.I.; Bilski, J.; Wojcik, D.; Brzozowski, B.; Surmiak, M.; Hubalewska-Mazgaj, M.; Chmura, A.; Magierowski, M.; Magierowska, K.; Mach, T. Beneficial effect of voluntary exercise on experimental colitis in mice fed a high-fat diet: The role of irisin, adiponectin and proinflammatory biomarkers. Nutrients 2017, 9, 410. [Google Scholar] [CrossRef]
- Allen, J.M.; Mailing, L.J.; Cohrs, J.; Salmonson, C.; Fryer, J.D.; Nehra, V.; Hale, V.L.; Kashyap, P.; White, B.A.; Woods, J.A. Exercise training-induced modification of the gut microbiota persists after microbiota colonization and attenuates the response to chemically-induced colitis in gnotobiotic mice. Gut Microbes 2018, 9, 115–130. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Huenchullan, S.F.; Ban, L.A.; Olaya-Agudo, L.F.; Maharjan, B.R.; Williams, P.F.; Tam, C.S.; Mclennan, S.V.; Twigg, S.M. Constant-moderate and high-intensity interval training have differential benefits on insulin sensitive tissues in high-fat fed mice. Front. Physiol. 2019, 10, 459. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, L.G.; Leonel, A.J.; Aguilar, E.C.; Batista, N.V.; Alves, A.C.; Coimbra, C.C.; Ferreira, A.V.; de Faria, A.M.; Cara, D.C.; Alvarez Leite, J.I. The combination of high-fat diet-induced obesity and chronic ulcerative colitis reciprocally exacerbates adipose tissue and colon inflammation. Lipids Health Dis. 2011, 10, 204. [Google Scholar] [CrossRef] [PubMed]
- Cooks, T.; Pateras, I.S.; Tarcic, O.; Solomon, H.; Schetter, A.J.; Wilder, S.; Lozano, G.; Pikarsky, E.; Forshew, T.; Rosenfeld, N.; et al. Mutant p53 prolongs NF-kappaB activation and promotes chronic inflammation and inflammation-associated colorectal cancer. Cancer Cell 2013, 23, 634–646. [Google Scholar] [CrossRef]
- Cho, J.; Shin, M.K.; Kim, D.; Lee, I.; Kim, S.; Kang, H. Treadmill running reverses cognitive declines due to Alzheimer disease. Med. Sci. Sports Exerc. 2015, 47, 1814–1824. [Google Scholar] [CrossRef]
- Quail, D.F.; Olson, O.C.; Bhardwaj, P.; Walsh, L.A.; Akkari, L.; Quick, M.L.; Chen, I.C.; Wendel, N.; Ben-Chetrit, N.; Walker, J.; et al. Obesity alters the lung myeloid cell landscape to enhance breast cancer metastasis through IL5 and GM-CSF. Nat. Cell Biol. 2017, 19, 974–987. [Google Scholar] [CrossRef]
- Gulhane, M.; Murray, L.; Lourie, R.; Tong, H.; Sheng, Y.; Wang, R.; Kang, A.; Schreiber, V.; Wong, K.Y.; Magor, G.; et al. High fat diets induce colonic epithelial cell stress and inflammation that is reversed by IL-22. Sci. Rep. 2016, 6, 28990. [Google Scholar] [CrossRef]
- Kim, I.W.; Myung, S.J.; Do, M.Y.; Ryu, Y.M.; Kim, M.J.; Do, E.J.; Park, S.; Yoon, S.M.; Ye, B.D.; Byeon, J.S.; et al. Western-style diets induce macrophage infiltration and contribute to colitis-associated carcinogenesis. J. Gastroenterol. Hepatol. 2010, 25, 1785–1794. [Google Scholar] [CrossRef]
- Qin, L.; Yao, Z.Q.; Chang, Q.; Zhao, Y.L.; Liu, N.N.; Zhu, X.S.; Liu, Q.Q.; Wang, L.F.; Yang, A.G.; Gao, C.F.; et al. Swimming attenuates inflammation, oxidative stress, and apoptosis in a rat model of dextran sulfate sodium-induced chronic colitis. Oncotarget 2017, 8, 7391–7404. [Google Scholar] [CrossRef]
- Walsh, N.P.; Gleeson, M.; Shephard, R.J.; Gleeson, M.; Woods, J.A.; Bishop, N.C.; Fleshner, M.; Green, C.; Pedersen, B.K.; Hoffman-Goetz, L.; et al. Position statement. Part one: Immune function and exercise. Exerc. Immunol. Rev. 2011, 17, 6–63. [Google Scholar]
- Matsumoto, M.; Inoue, R.; Tsukahara, T.; Ushida, K.; Chiji, H.; Matsubara, N.; Hara, H. Voluntary running exercise alters microbiota composition and increases n-butyrate concentration in the rat cecum. Biosci. Biotechnol. Biochem. 2008, 72, 572–576. [Google Scholar] [CrossRef] [PubMed]
- Cook, M.D.; Allen, J.M.; Pence, B.D.; Wallig, M.A.; Gaskins, H.R.; White, B.A.; Woods, J.A. Exercise and gut immune function: Evidence of alterations in colon immune cell homeostasis and microbiome characteristics with exercise training. Immunol. Cell Biol. 2016, 94, 158–163. [Google Scholar] [CrossRef] [PubMed]
- Evans, C.C.; LePard, K.J.; Kwak, J.W.; Stancukas, M.C.; Laskowski, S.; Dougherty, J.; Moulton, L.; Glawe, A.; Wang, Y.; Leone, V.; et al. Exercise prevents weight gain and alters the gut microbiota in a mouse model of high fat diet-induced obesity. PLoS ONE 2014, 9, e92193. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Kang, H. Exercise training modifies gut microbiota with attenuated host responses to sepsis in wild-type mice. FASEB J. 2019, 33, 5772–5781. [Google Scholar] [CrossRef]
- Liu, W.L.; Chen, S.J.; Chen, Y.; Sun, L.M.; Zhang, W.; Zeng, Y.M.; Zhou, T.H.; Si, J.M. Protective effects of heat shock protein70 induced by geranylgeranylacetone in atrophic gastritis in rats. Acta Pharmacol. Sin. 2007, 28, 1001–1006. [Google Scholar] [CrossRef] [PubMed]
- Yeo, M.; Park, H.K.; Kim, D.K.; Cho, S.W.; Kim, Y.S.; Cho, S.Y.; Paik, Y.K.; Hahm, K.B. Restoration of heat shock protein70 suppresses gastric mucosal inducible nitric oxide synthase expression induced by Helicobacter pylori. Proteomics 2004, 4, 3335–3342. [Google Scholar] [CrossRef] [PubMed]
- Lollo, P.C.; Moura, C.S.; Morato, P.N.; Amaya-Farfan, J. Differential response of heat shock proteins to uphill and downhill exercise in heart, skeletal muscle, lung and kidney tissues. J. Sports Sci. Med. 2013, 12, 461–466. [Google Scholar]
- Luo, B.; Xiang, D.; Nieman, D.C.; Chen, P. The effects of moderate exercise on chronic stress-induced intestinal barrier dysfunction and antimicrobial defense. Brain Behav. Immun. 2014, 39, 99–106. [Google Scholar] [CrossRef]
- Dokladny, K.; Lobb, R.; Wharton, W.; Ma, T.Y.; Moseley, P.L. LPS-induced cytokine levels are repressed by elevated expression of HSP70 in rats: Possible role of NF-kappaB. Cell Stress Chaperones 2010, 15, 153–163. [Google Scholar] [CrossRef]
- Hoffman-Goetz, L.; Pervaiz, N.; Packer, N.; Guan, J. Freewheel training decreases pro- and increases anti-inflammatory cytokine expression in mouse intestinal lymphocytes. Brain Behav. Immun. 2010, 24, 1105–1115. [Google Scholar] [CrossRef]
- Kasimay, O.; Guzel, E.; Gemici, A.; Abdyli, A.; Sulovari, A.; Ercan, F.; Yeğen, B.C. Colitis-induced oxidative damage of the colon and skeletal muscle is ameliorated by regular exercise in rats: The anxiolytic role of exercise. Exp. Physiol. 2006, 91, 897–906. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.A.; Gu, W.; Lee, I.A.; Joh, E.H.; Kim, D.H. High fat diet-induced gut microbiota exacerbates inflammation and obesity in mice via the TLR4 signaling pathway. PLoS ONE 2012, 7, e47713. [Google Scholar] [CrossRef] [PubMed]
- Saxena, A.; Fletcher, E.; Larsen, B.; Baliga, M.S.; Durstine, J.L.; Fayad, R. Effect of exercise on chemically-induced colitis in adiponectin deficient mice. J. Inflamm. 2012, 9, 30. [Google Scholar] [CrossRef] [PubMed]
- Holland, A.M.; Hyatt, H.W.; Smuder, A.J.; Sollanek, K.J.; Morton, A.B.; Roberts, M.D.; Kavazis, A.N. Influence of endurance exercise training on antioxidant enzymes, tight junction proteins, and inflammatory markers in the rat ileum. BMC Res. Notes 2015, 8, 514. [Google Scholar] [CrossRef]
- Rath, H.C.; Herfarth, H.H.; Ikeda, J.S.; Grenther, W.B.; Hamm, T.E., Jr.; Balish, E.; Taurog, J.D.; Hammer, R.E.; Wilson, K.H.; Sartor, R.B. Normal luminal bacteria, especially Bacteroides species, mediate chronic colitis, gastritis, and arthritis in HLA-B27/human beta2 microglobulin transgenic rats. J. Clin. Investig. 1996, 98, 945–953. [Google Scholar] [CrossRef]
- Funke, G.; Hany, A.; Altwegg, M. Isolation of Escherichia fergusonii from four different sites in a patient with pancreatic carcinoma and cholangiosepsis. J. Clin. Microbiol. 1993, 31, 2201–2203. [Google Scholar] [CrossRef]
- Zhang, Z.; Wu, X.; Cao, S.; Wang, L.; Wang, D.; Yang, H.; Feng, Y.; Wang, S.; Li, L. Caffeic acid ameliorates colitis in association with increased Akkermansia population in the gut microbiota of mice. Oncotarget 2016, 7, 31790–31799. [Google Scholar] [CrossRef]
- Munukka, E.; Ahtiainen, J.P.; Puigbo, P.; Jalkanen, S.; Pahkala, K.; Keskitalo, A.; Kujala, U.M.; Pietilä, S.; Hollmén, M.; Elo, L.; et al. Six-week endurance exercise alters gut metagenome that is not reflected in systemic metabolism in over-weight women. Front. Microbiol. 2018, 9, 2323. [Google Scholar] [CrossRef]
- Mailing, L.J.; Allen, J.M.; Buford, T.W.; Fields, C.J.; Woods, J.A. Exercise and the gut microbiome: A review of the evidence, potential mechanisms, and implications for human health. Exerc. Sport Sci. Rev. 2019, 47, 75–85. [Google Scholar] [CrossRef]


| Total Reads per Group | Coverage (%) | Observed OTUs | Chao1 | Shannon | Simpson | |
|---|---|---|---|---|---|---|
| SC | 389582.000 ± 7299.597 | 0.990 ± 0.000 | 190.667 ± 9.838 | 229.720 ± 25.765 | 3.903 ± 0.247 | 0.847 ± 0.019 |
| HF | 363285.333 ± 4693.449 | 0.990 ± 0.000 | 233.333 ± 11.348 | 263.300 ± 12.401 | 4.870 ± 0.125 | 0.913 ± 0.009 |
| HF+DSS | 374658.000 ± 19939.972 | 0.990 ± 0.000 | 147.000 ± 11.358 | 160.053 ± 14.209 | 2.823 ± 0.194 | 0.683 ± 0.026 |
| HF+EX+DSS | 343162.000 ± 12509.490 | 0.990 ± 0.000 | 149.000 ± 9.866 | 166.183 ± 11.907 | 3.740 ± 0.437 | 0.830 ± 0.070 |
| Phylum | Family | Abundance (%) | Abundance Relative to SC (%) | p Value | |||||
|---|---|---|---|---|---|---|---|---|---|
| SC | HF | HF+DSS | HF+EX+DSS | HF | HF+DSS | HF+EX+DSS | |||
| Actinobacteria | Bifidobacteriaceae | 1.81 | 0.00 | 0.00 | 0.00 | −1.81 * | −1.81 * | −1.81 * | 0.027 |
| Bacteroidetes | Bacteroidaceae | 14.00 | 18.06 | 58.53 | 30.60 | +4.06 | +44.53 * | +16.60 | 0.026 |
| Firmicutes | Streptococcaceae | 0.01 | 1.04 | 0.26 | 0.30 | +1.03 * | +0.25 | +0.29 | 0.013 |
| Firmicutes | Christensenellaceae | 0.01 | 0.07 | 0.02 | 0.03 | +0.06 * | +0.01 | +0.02 | 0.006 |
| Firmicutes | Clostridiaceae | 1.99 | 0.65 | 0.00 | 0.02 | −1.34 * | −1.99 * | −1.97 * | 0.005 |
| Firmicutes | Lachnospiraceae | 0.69 | 18.67 | 3.85 | 8.84 | +17.97 * | +3.15 | +8.14 * | <0.001 |
| Firmicutes | Oscillospiraceae | 0.05 | 3.76 | 0.18 | 0.28 | +3.71 * | +0.12 | +0.22 | 0.001 |
| Firmicutes | Peptococcaceae | 0.38 | 6.44 | 0.78 | 1.02 | +6.06 * | +0.40 | +0.64 | 0.005 |
| Firmicutes | Peptostreptococcaceae | 0.01 | 0.11 | 0.86 | 0.18 | +0.09 | +0.85 * | +0.17 | 0.005 |
| Firmicutes | Ruminococcaceae | 1.44 | 5.53 | 0.56 | 1.75 | +4.09 * | −0.88 | +0.30 | <0.001 |
| Proteobacteria | Rhodospirillaceae | 0.00 | 0.05 | 0.00 | 2.86 | +0.05 | 0.00 | +2.86 * | 0.021 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cho, J.; Kim, D.; Kang, H. Exercise Preconditioning Attenuates the Response to Experimental Colitis and Modifies Composition of Gut Microbiota in Wild-Type Mice. Life 2020, 10, 200. https://doi.org/10.3390/life10090200
Cho J, Kim D, Kang H. Exercise Preconditioning Attenuates the Response to Experimental Colitis and Modifies Composition of Gut Microbiota in Wild-Type Mice. Life. 2020; 10(9):200. https://doi.org/10.3390/life10090200
Chicago/Turabian StyleCho, Jinkyung, Donghyun Kim, and Hyunsik Kang. 2020. "Exercise Preconditioning Attenuates the Response to Experimental Colitis and Modifies Composition of Gut Microbiota in Wild-Type Mice" Life 10, no. 9: 200. https://doi.org/10.3390/life10090200
APA StyleCho, J., Kim, D., & Kang, H. (2020). Exercise Preconditioning Attenuates the Response to Experimental Colitis and Modifies Composition of Gut Microbiota in Wild-Type Mice. Life, 10(9), 200. https://doi.org/10.3390/life10090200






