DXA-Derived Visceral Adipose Tissue (VAT) in Elderly: Percentiles of Reference for Gender and Association with Metabolic Outcomes
Abstract
1. Introduction
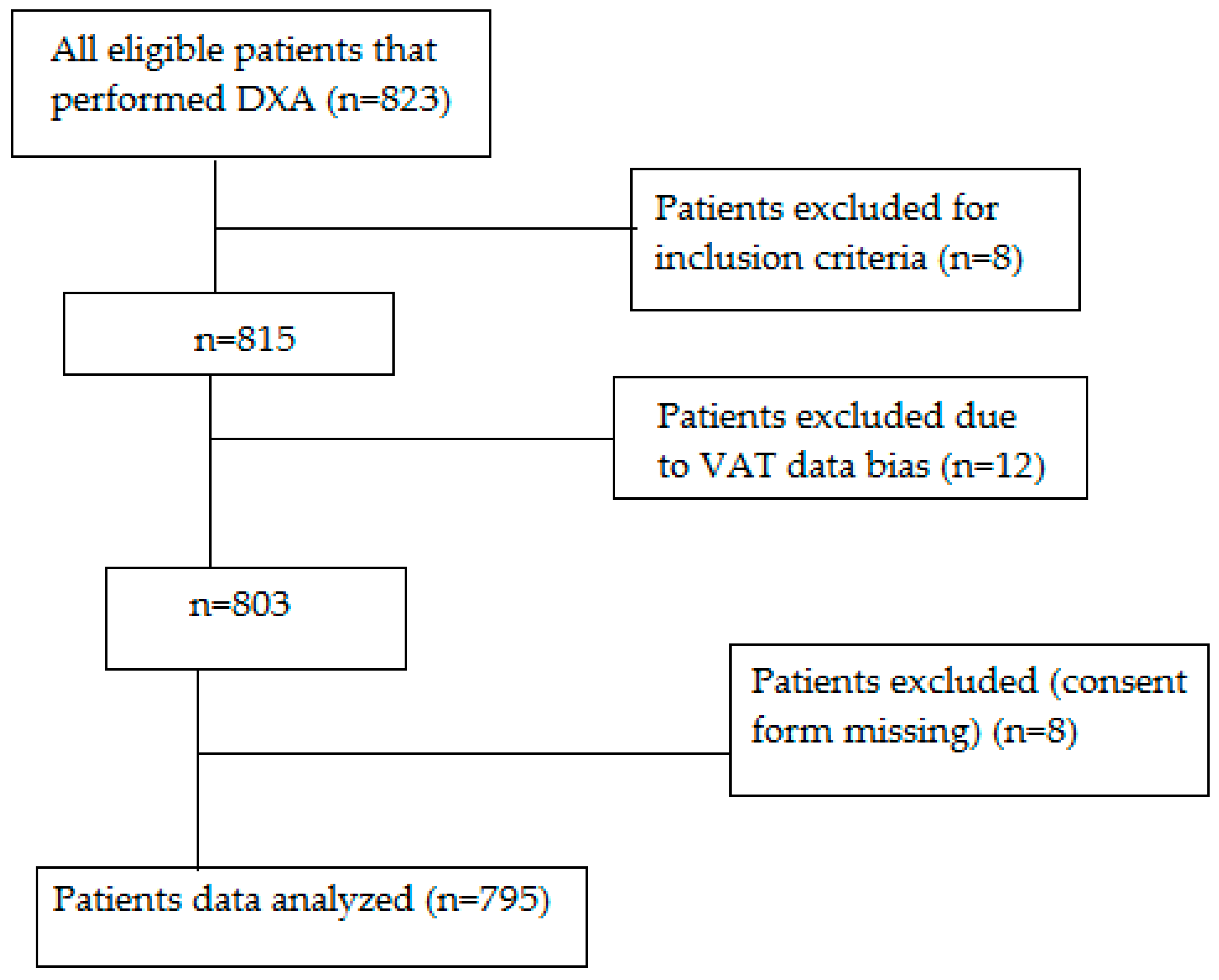
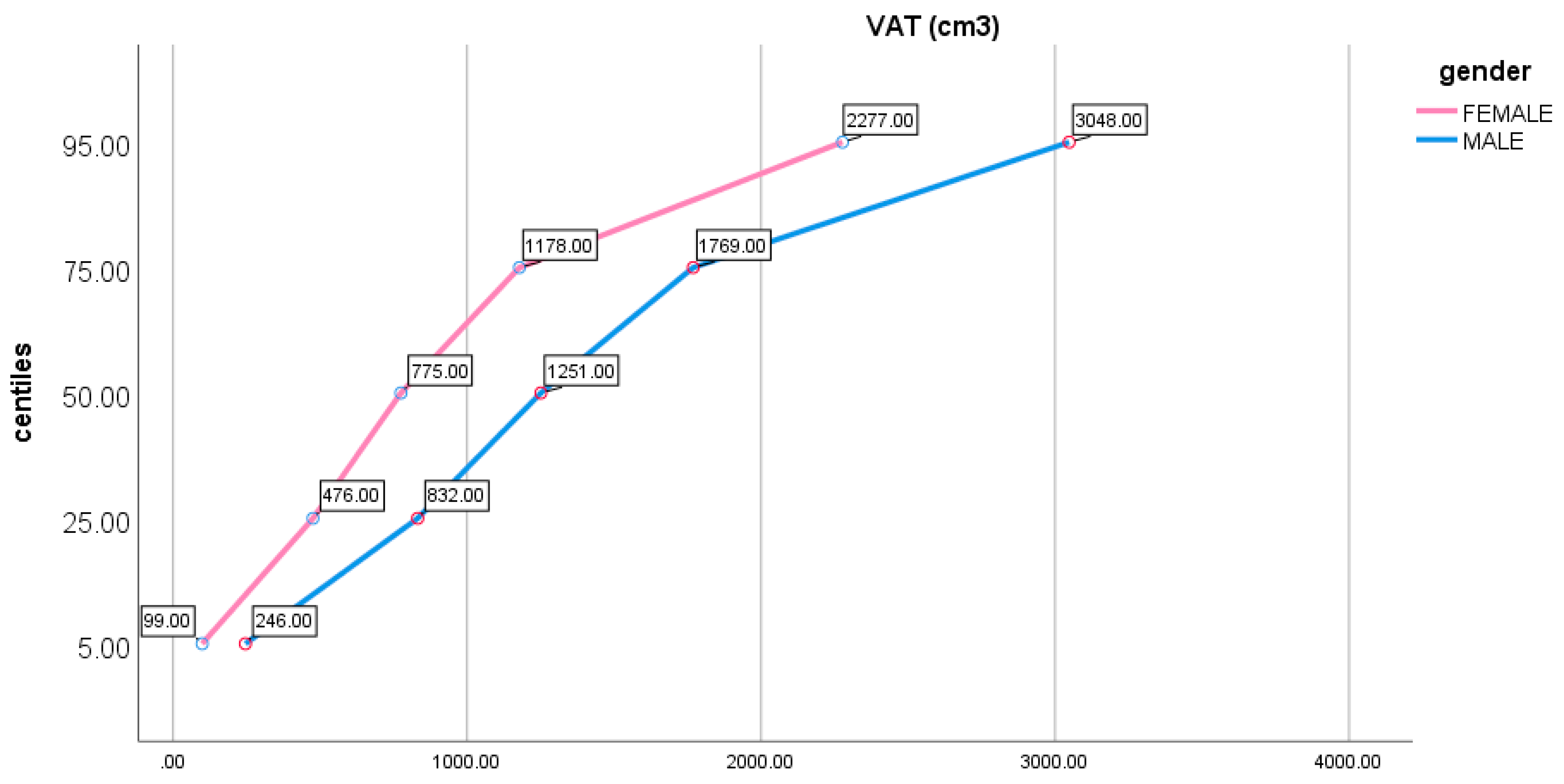
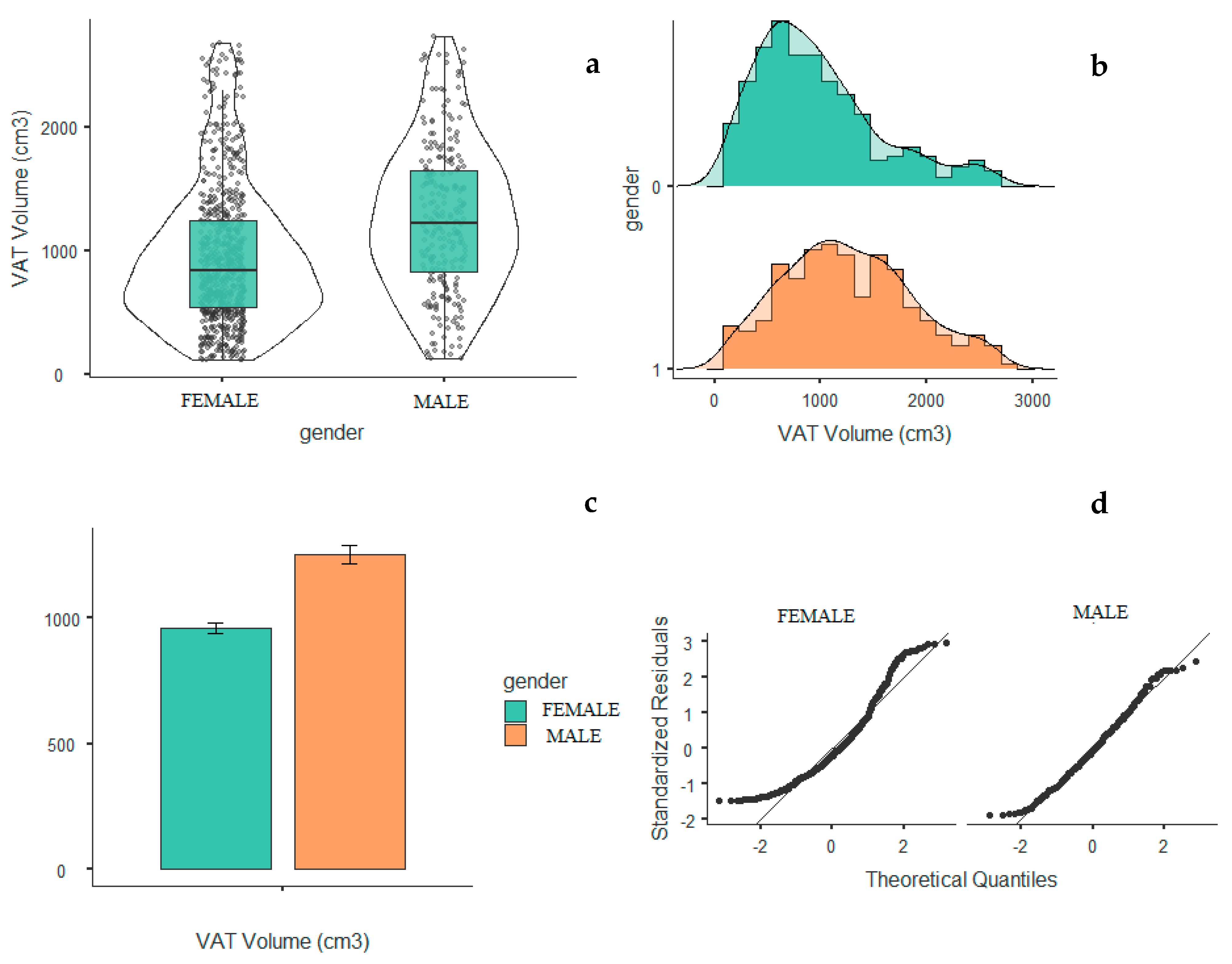
2. Results
3. Discussion
4. Materials and Methods
4.1. Setting
4.2. Study Population
4.3. Observed Variables
4.3.1. Body Composition Assessment and Diagnosis of Visceral Adipose Tissue (VAT)
4.3.2. Anthropometric Data Assessment
4.3.3. Diagnosis of Sarcopenia
4.3.4. Assessment of Bone Mineral Density
4.3.5. Blood Sample Measurements
4.3.6. Screening of Cognitive Status
4.3.7. Assessment of Nutritional Status: (MNA)
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Ethical Statement
Informed Consent
Availability of Data and Materials
References
- Ruderman, N.; Chisholm, D.; Pi-Sunyer, X.; Schneider, S. The metabolically obese, normal-weight individual revisited. Diabetes 1998, 47, 699–713. [Google Scholar] [CrossRef] [PubMed]
- Després, J.P. Is visceral obesity the cause of the metabolic syndrome? Ann. Med. 2006, 38, 52–63. [Google Scholar] [CrossRef] [PubMed]
- Choe, S.S.; Huh, J.Y.; Hwang, I.J.; Kim, J.I.; Kim, J.B. Adipose tissue remodeling: Its role in energy metabolism and metabolic disorders. Front. Endocrinol. 2016, 7, 30. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, T.; Liu, L.F.; Lamendola, C.; Shen, L.; Morton, J.; Rivas, H.; Winer, D.; Tolentino, L.; Choi, O.; Zhang, H.; et al. T-cell profile in adipose tissue is associated with insulin resistance and systemic inflammation in humans. Arter. Thromb. Vasc. Biol. 2014, 34, 2637–2643. [Google Scholar] [CrossRef]
- Wensveen, F.M.; Valentić, S.; Šestan, M.; Turk Wensveen, T.; Polić, B. The “Big Bang” in obese fat: Events initiating obesity-induced adipose tissue inflammation. Eur. J. Immunol. 2015, 45, 2446–2456. [Google Scholar] [CrossRef]
- Camhi, S.M.; Bray, G.A.; Bouchard, C.; Greenway, F.L.; Johnson, W.D.; Newton, R.L.; Ravussin, E.; Ryan, D.H.; Smith, S.R.; Katzmarzyk, P.T. The relationship of waist circumference and BMI to visceral, subcutaneous, and total body fat: Sex and race differences. Obesity 2011, 19, 402–408. [Google Scholar] [CrossRef]
- Onat, A.; Uǧur, M.; Can, G.; Yüksel, H.; Hergenç, G. Visceral adipose tissue and body fat mass: Predictive values for and role of gender in cardiometabolic risk among Turks. Nutrition 2010, 26, 382–389. [Google Scholar] [CrossRef]
- Zuriaga, M.A.; Fuster, J.J.; Farb, M.G.; MacLauchlan, S.; Bretón-Romero, R.; Karki, S.; Hess, D.T.; Apovian, C.M.; Hamburg, N.M.; Gokce, N.; et al. Activation of non-canonical WNT signaling in human visceral adipose tissue contributes to local and systemic inflammation. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef]
- Item, F.; Konrad, D. Visceral fat and metabolic inflammation: The portal theory revisited. Obes. Rev. 2012, 13, 30–39. [Google Scholar] [CrossRef]
- Cesari, M.; Kritchevsky, S.B.; Baumgartner, R.N.; Atkinson, H.H.; Penninx, B.W.; Lenchik, L.; Palla, S.L.; Ambrosius, W.T.; Tracy, R.P.; Pahor, M. Sarcopenia, obesity, and inflammation—Results from the Trial of Angiotensin Converting Enzyme Inhibition and Novel Cardiovascular Risk Factors study. Am. J. Clin. Nutr. 2005, 82, 428–434. [Google Scholar] [CrossRef]
- Tzankoff, S.P.; Norris, A.H. Longitudinal changes in basal metabolism in man. J. Appl. Physiol. 1978, 45, 536–539. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.N.; Park, M.S.; Lim, K.I.; Yang, S.J.; Yoo, H.J.; Kang, H.J.; Song, W.; Seo, J.A.; Kim, S.G.; Kim, N.H.; et al. Skeletal muscle mass to visceral fat area ratio is associated with metabolic syndrome and arterial stiffness: The Korean Sarcopenic Obesity Study (KSOS). Diabetes Res. Clin. Pract. 2011, 93, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Bredella, M.A.; Torriani, M.; Ghomi, R.H.; Thomas, B.J.; Brick, D.J.; Gerweck, A.V.; Rosen, C.J.; Klibanski, A.; Miller, K.K. Vertebral Bone Marrow Fat Is Positively Associated With Visceral Fat and Inversely Associated With IGF-1 in Obese Women. Obesity 2011, 19, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Thoma, A.; Lightfoot, A.P. Nf-kb and inflammatory cytokine signalling: Role in skeletal muscle atrophy. In Advances in Experimental Medicine and Biology; Springer: Singapore, 2018; Volume 1088, pp. 267–279. [Google Scholar]
- Rong, Y.D.; Bian, A.L.; Hu, H.Y.; Ma, Y.; Zhou, X.Z. Study on relationship between elderly sarcopenia and inflammatory cytokine IL-6, anti-inflammatory cytokine IL-10. BMC Geriatr. 2018, 18, 308. [Google Scholar] [CrossRef] [PubMed]
- Zamboni, M.; Mazzali, G.; Fantin, F.; Rossi, A.; Di Francesco, V. Sarcopenic obesity: A new category of obesity in the elderly. Nutr. Metab. Cardiovasc. Dis. 2008, 18, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Cauley, J.A. An Overview of Sarcopenic Obesity. J. Clin. Densitom. 2015, 18, 499–505. [Google Scholar] [CrossRef]
- Ormsbee, M.J.; Prado, C.M.; Ilich, J.Z.; Purcell, S.; Siervo, M.; Folsom, A.; Panton, L. Osteosarcopenic obesity: The role of bone, muscle, and fat on health. J. Cachexia Sarcopenia Muscle 2014, 5, 183–192. [Google Scholar] [CrossRef]
- Perna, S.; Spadaccini, D.; Nichetti, M.; Avanzato, I.; Faliva, M.A.; Rondanelli, M. Osteosarcopenic Visceral Obesity and Osteosarcopenic Subcutaneous Obesity, Two New Phenotypes of Sarcopenia: Prevalence, Metabolic Profile, and Risk Factors. J. Aging Res. 2018, 2018, 1–8. [Google Scholar] [CrossRef]
- Zhang, P.; Peterson, M.; Su, G.L.; Wang, S.C. Visceral adiposity is negatively associated with bone density and muscle attenuation. Am. J. Clin. Nutr. 2015, 101, 337–343. [Google Scholar] [CrossRef]
- Freitas, P.M.S.S.; Garcia Rosa, M.L.; Gomes, A.M.; Wahrlich, V.; Di Luca, D.G.; da Cruz Filho, R.A.; da Silva Correia, D.M.; Faria, C.A.; Yokoo, E.M. Central and peripheral fat body mass have a protective effect on osteopenia or osteoporosis in adults and elderly? Osteoporos. Int. 2016, 27, 1659–1663. [Google Scholar] [CrossRef]
- Jiang, Y.; Zhang, Y.; Jin, M.; Gu, Z.; Pei, Y.; Meng, P. Aged-related changes in body composition and association between body composition with bone mass density by body mass index in Chinese Han men over 50-year-old. PLoS ONE 2015, 10, e0130400. [Google Scholar] [CrossRef] [PubMed]
- Atkins, R.C. The epidemiology of chronic kidney disease. Kidney Int. 2005, 67, S14–S18. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.E.; do Carmo, J.M.; da Silva, A.A.; Wang, Z.; Hall, M.E. Obesity, kidney dysfunction and hypertension: Mechanistic links. Nat. Rev. Nephrol. 2019, 15, 367–385. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.R.; Yoo, J.H.; Song, H.C.; Lee, S.S.; Yoo, S.J.; Kim, Y.-D.; Lim, Y.S.; Kim, H.W.; Yang, C.W.; Kim, Y.-S.; et al. Relationship of visceral and subcutaneous adiposity with renal function in people with type 2 diabetes mellitus. Nephrol. Dial. Transplant. 2011, 26, 3550–3555. [Google Scholar] [CrossRef]
- Young, J.A.; Hwang, S.J.; Sarnak, M.J.; Hoffmann, U.; Massaro, J.M.; Levy, D.; Benjamin, E.J.; Larson, M.G.; Vasan, R.S.; O’Donnell, C.J.; et al. Association of visceral and subcutaneous adiposity with kidney function. Clin. J. Am. Soc. Nephrol. 2008, 3, 1786–1791. [Google Scholar] [CrossRef]
- Dekkers, I.A.; de Vries, A.P.J.; Smit, R.A.J.; Rosendaal, F.R.; Rabelink, T.J.; Lamb, H.J.; de Mutsert, R. The Separate Contributions of Visceral Fat and Liver Fat to Chronic Kidney Disease-Related Renal Outcomes. J. Ren. Nutr. 2019, 30, 286–295. [Google Scholar] [CrossRef]
- Kaul, S.; Rothney, M.P.; Peters, D.M.; Wacker, W.K.; Davis, C.E.; Shapiro, M.D.; Ergun, D.L. Dual-energy X-ray absorptiometry for quantification of visceral fat. Obesity 2012, 20, 1313–1318. [Google Scholar] [CrossRef]
- Neeland, I.J.; Grundy, S.M.; Li, X.; Adams-Huet, B.; Vega, G.L. Comparison of visceral fat mass measurement by dual-X-ray absorptiometry and magnetic resonance imaging in a multiethnic cohort: The Dallas Heart Study. Nutr. Diabetes 2016, 6, e221. [Google Scholar] [CrossRef]
- Micklesfield, L.K.; Goedecke, J.H.; Punyanitya, M.; Wilson, K.E.; Kelly, T.L. Dual-energy X-ray performs as well as clinical computed tomography for the measurement of visceral fat. Obesity 2012, 20, 1109–1114. [Google Scholar] [CrossRef]
- Cheung, A.S.; De Rooy, C.; Hoermann, R.; Gianatti, E.J.; Hamilton, E.J.; Roff, G.; Zajac, J.D.; Grossmann, M. Correlation of visceral adipose tissue measured by Lunar Prodigy dual X-ray absorptiometry with MRI and CT in older men. Int. J. Obes. 2016, 40, 1325–1328. [Google Scholar] [CrossRef]
- Barrera, G.; Bunout, D.; Gattás, V.; De La Maza, M.P.; Leiva, L.; Hirsch, S. A high body mass index protects against femoral neck osteoporosis in healthy elderly subjects. Nutrition 2004, 20, 769–771. [Google Scholar] [CrossRef] [PubMed]
- De Laet, C.; Kanis, J.A.; Odén, A.; Johanson, H.; Johnell, O.; Delmas, P.; Eisman, J.A.; Kroger, H.; Fujiwara, S.; Garnero, P.; et al. Body mass index as a predictor of fracture risk: A meta-analysis. Osteoporos. Int. 2005, 16, 1330–1338. [Google Scholar] [CrossRef] [PubMed]
- Bales, C.W.; Ritchie, C.S. Sarcopenia, Weight loss, and Nutritional Frailty in the Elderly. Annu. Rev. Nutr. 2002, 22, 309–323. [Google Scholar] [CrossRef] [PubMed]
- Wajchenberg, B.L.; Giannella-Neto, D.; Da Silva, M.E.R.; Santos, R.F. Depot-specific hormonal characteristics of subcutaneous and visceral adipose tissue and their relation to the metabolic syndrome. Horm. Metab. Res. 2002, 34, 616–621. [Google Scholar] [CrossRef] [PubMed]
- Gastaldelli, A.; Cusi, K.; Pettiti, M.; Hardies, J.; Miyazaki, Y.; Berria, R.; Buzzigoli, E.; Sironi, A.M.; Cersosimo, E.; Ferrannini, E.; et al. Relationship Between Hepatic/Visceral Fat and Hepatic Insulin Resistance in Nondiabetic and Type 2 Diabetic Subjects. Gastroenterology 2007, 133, 496–506. [Google Scholar] [CrossRef]
- Jung, S.H.; Ha, K.H.; Kim, D.J. Visceral fat mass has stronger associations with diabetes and prediabetes than other anthropometric obesity indicators among Korean adults. Yonsei Med. J. 2016, 57, 674–680. [Google Scholar] [CrossRef]
- Ginsberg, H.N.; Zhang, Y.L.; Hernandez-Ono, A. Regulation of plasma triglycerides in insulin resistance and diabetes. Arch. Med. Res. 2005, 36, 232–240. [Google Scholar] [CrossRef]
- Seyed-Sadjadi, N.; Berg, J.; Bilgin, A.A.; Grant, R. Visceral fat mass: Is it the link between uric acid and diabetes risk? Lipids Health Dis. 2017, 16, 1–9. [Google Scholar] [CrossRef]
- Yamada, A.; Sato, K.K.; Kinuhata, S.; Uehara, S.; Endo, G.; Hikita, Y.; Fujimoto, W.Y.; Boyko, E.J.; Hayashi, T. Association of Visceral Fat and Liver Fat with Hyperuricemia. Arthritis Care Res. 2016, 68, 553–561. [Google Scholar] [CrossRef]
- Takahashi, S.; Yamamoto, T.; Tsutsumi, Z.; Moriwaki, Y.; Yamakita, J.; Higashino, K. Close correlation between visceral fat accumulation and uric acid metabolism in healthy men. Metabolism 1997, 46, 1162–1165. [Google Scholar] [CrossRef]
- Vianna, H.R.; Soares, C.M.B.M.; Tavares, M.S.; Teixeira, M.M.; Silva, A.C.S. Inflammation in chronic kidney disease: The role of cytokines. J. Braz. Nephrol. 2011, 33, 351–364. [Google Scholar] [CrossRef]
- Rosa, E.C.; Zanella, M.T.; Ribeiro, A.B.; Kohlmann Junior, O. Visceral obesity, hypertension and cardio-renal risk: A review. Arq. Bras. Endocrinol. Metabol. 2005, 49, 196–204. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, Q.; Zheng, D.; Liu, J.; Fang, L.; Li, Q. Skeletal muscle mass to visceral fat area ratio is an important determinant associated with type 2 diabetes and metabolic syndrome. Diabetes Metab. Syndr. Obes. Targets Ther. 2019, 12, 1399–1407. [Google Scholar] [CrossRef] [PubMed]
- Shida, T.; Akiyama, K.; Oh, S.; Sawai, A.; Isobe, T.; Okamoto, Y.; Ishige, K.; Mizokami, Y.; Yamagata, K.; Onizawa, K.; et al. Skeletal muscle mass to visceral fat area ratio is an important determinant affecting hepatic conditions of non-alcoholic fatty liver disease. J. Gastroenterol. 2018, 53, 535–547. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.S.; Kim, K.J.; Kim, K.M.; Hur, N.W.; Rhee, Y.; Han, D.S.; Lee, E.J.; Lim, S.K. Relationship between visceral adiposity and bone mineral density in korean adults. Calcif. Tissue Int. 2010, 87, 218–225. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.-T.; Broe, K.E.; Zhou, Y.; Boyd, S.K.; Cupples, L.A.; Hannan, M.T.; Lim, E.; McLean, R.R.; Samelson, E.J.; Bouxsein, M.L.; et al. Visceral Adipose Tissue Is Associated With Bone Microarchitecture in the Framingham Osteoporosis Study. J. Bone Miner. Res. 2017, 32, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Machado, L.G.; Domiciano, D.S.; Figueiredo, C.P.; Caparbo, V.F.; Takayama, L.; Oliveira, R.M.; Lopes, J.B.; Menezes, P.R.; Pereira, R.M.R. Visceral fat measured by DXA is associated with increased risk of non-spine fractures in nonobese elderly women: A population-based prospective cohort analysis from the São Paulo Ageing & Health (SPAH) Study. Osteoporos. Int. 2016, 27, 3525–3533. [Google Scholar]
- Shapses, S.A.; Cifuentes, M. Body weight/composition and weight change: Effects on bone health. In Nutrition and Bone Health; Springer: New York, NY, USA, 2015; pp. 561–583. ISBN 9781493920013. [Google Scholar]
- Scott, D.; Seibel, M.; Cumming, R.; Naganathan, V.; Blyth, F.; Le Couteur, D.G.; Handelsman, D.J.; Waite, L.M.; Hirani, V. Sarcopenic Obesity and Its Temporal Associations With Changes in Bone Mineral Density, Incident Falls, and Fractures in Older Men: The Concord Health and Ageing in Men Project. J. Bone Miner. Res. 2017, 32, 575–583. [Google Scholar] [CrossRef]
- Bonaccorsi, G.; Cafarelli, F.P.; Cervellati, C.; De Guio, F.; Greco, P.; Giganti, M.; Guglielmi, G. A new corrective model to evaluate TBS in obese post-menopausal women: A cross-sectional study. Aging Clin. Exp. Res. 2019, 32, 1303–1308. [Google Scholar] [CrossRef]
- Swainson, M.G.; Batterham, A.M.; Hind, K. Age- and sex-specific reference intervals for visceral fat mass in adults. Int. J. Obes. 2020, 44, 289–296. [Google Scholar] [CrossRef]
- Farhangi, M.A.; Keshavarz, S.A.; Eshraghian, M.; Ostadrahimi, A.; Saboor-Yaraghi, A.A. White blood cell count in women: Relation to inflammatory biomarkers, haematological profiles, visceral adiposity, and other cardiovascular risk factors. J. Health Popul. Nutr. 2013, 31, 58. [Google Scholar] [CrossRef] [PubMed]
- Facchini, F.S.; Carantoni, M.; Jeppesen, J.; Reaven, G.M. Hematocrit and hemoglobin are independently related to insulin resistance and compensatory hyperinsulinemia in healthy, non-obese men and women. Metabolism 1998, 47, 831–835. [Google Scholar] [CrossRef]
- Mohammad, A.; De Lucia Rolfe, E.; Sleigh, A.; Kivisild, T.; Behbehani, K.; Wareham, N.J.; Brage, S.; Mohammad, T. Validity of visceral adiposity estimates from DXA against MRI in Kuwaiti men and women. Nutr. Diabetes 2017, 7, e238. [Google Scholar] [CrossRef] [PubMed]
- WHO. Obesity: Preventing and Managing the Global Epidemic; WHO: Geneva, Switzerland, 2000. [Google Scholar]
- Kvamme, J.M.; Holmen, J.; Wilsgaard, T.; Florholmen, J.; Midthjell, K.; Jacobsen, B.K. Body mass index and mortality in elderly men and women: The Tromsø and HUNT studies. J. Epidemiol. Community Health 2012, 66, 611–617. [Google Scholar] [CrossRef]
- Sergi, G.; Perissinotto, E.; Pisent, C.; Buja, A.; Maggi, S.; Coin, A.; Grigoletto, F.; Enzi, G. An adequate threshold for body mass index to detect underweight condition in elderly persons: The Italian Longitudinal Study on Aging (ILSA). J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2005, 60, 866–871. [Google Scholar] [CrossRef]
- Janssen, I.; Mark, A.E. Elevated body mass index and mortality risk in the elderly. Obes. Rev. 2007, 8, 41–59. [Google Scholar] [CrossRef]
- Fess, E.; Moran, C. Clinical Assessment Recommendations, 2nd ed.; American Society of Hand Therapists: Chicago, IL, USA, 1992. [Google Scholar]
- Fess, E. Clinical Assessment Recommendations, 3rd ed.; American Society Hand Therapists: Mount Laurel, NJ, USA, 1992. [Google Scholar]
- Wang, Z.M.; Visser, M.; Ma, R.; Baumgartner, R.N.; Kotler, D.; Gallagher, D.; Heymsfield, S.B. Skeletal muscle mass: Evaluation of neutron activation and dual-energy X-ray absorptiometry methods. J. Appl. Physiol. 1996, 80, 824–831. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef]
- Kanis, J.A.; Oden, A.; Johansson, H.; Borgström, F.; Ström, O.; McCloskey, E. FRAX®and its applications to clinical practice. Bone 2009, 44, 734–743. [Google Scholar] [CrossRef]
- Cockcroft, D.W.; Gault, M.H. Prediction of creatinine clearance from serum creatinine. Nephron 1976, 16, 31–41. [Google Scholar] [CrossRef]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Vellas, B.; Guigoz, Y.; Garry, P.J.; Nourhashemi, F.; Bennahum, D.; Lauque, S.; Albarede, J.L. The mini nutritional assessment (MNA) and its use in grading the nutritional state of elderly patients. Nutrition 1999, 15, 116–122. [Google Scholar] [CrossRef]
| Variable | r | Variable | r |
|---|---|---|---|
| Gender | 0.30 ** | Weight | 0.72 ** |
| Age | −0.11 ** | BMI | 0.63 ** |
| White blood cells | 0.07 * | MNA | 0.33 ** |
| Hemoglobin | 0.10 ** | Femoral Neck BMD | 0.37 ** |
| Hematocrit | 0.08 * | Femoral Neck T-score | 0.34 ** |
| Lymphocytes % | 0.09 * | Hip FRAX | −0.24 ** |
| Triglycerides | 0.24 ** | FFM | 0.38 ** |
| HDL cholesterol | −0.22 ** | FM | 0.67 ** |
| β Globulins | 0.16 ** | Handgrip | 0.21 ** |
| Creatinine | 0.21 ** | ASM | 0.30 ** |
| eGFR | 0.29 ** | ASM/h2 | 0.20 ** |
| Amylase | −0.08 * | FFM% | −0.52 ** |
| Uric acid | 0.20 ** | FM% | 0.52 ** |
| Glycemia | 0.15 ** | Osteoporosis (N = 0/Y = 1) | −0.25 ** |
| Height | 0.29 ** | Diabetes (N = 0/Y = 1) | 0.18 ** |
| Variable | β | Confidence Interval 95% | Variable | β | Confidence Interval 95% |
|---|---|---|---|---|---|
| Gender (F = 0/M = 1) | 0.423 ** | 595.06; 741.79 | Fe (µg/dL) | −0.018 | −1.42; 0.63 |
| Age (years) | 0.010 | −3.89; 6.17 | Creatinine (mg/dL) | 0.066 ** | 27.66; 156.41 |
| Height (cm) | 0.066 | −8.92; 0.29 | eGFR (mL/min) | −0.056 δ.* | −3.60; −0.21 |
| Weight (kg) | 0.047 | −3.04; 8.13 | Amylase (U/L) | 0.002 | −1.29; 1.41 |
| BMI (kg/m2) | 0.062 | −3.93; 22.55 | Glycemia (mg/dL) | 0.063 ** | 0.33; 1.94 |
| MMSE (pts) | −0.029 | −8.71; 2.61 | Uric Acid (mg/dL) | 0.052 π.* | 1.84; 34.23 |
| MNA (pts) | 0.012 | −9.17; 14.27 | ESR (mm/hr) | −0.035 | −1.95; 0.30 |
| White Blood Cells (K/uL) | −0.026 | −16.40; 4.53 | CRP (mg/dL) | −0.009 | −14.08; 9.58 |
| Red Blood Cells (M/uL) | −0.031 | −82.16; 17.18 | AST (U/L) | 0.014 | −1.48; 2.86 |
| Hemoglobin (g/dL) | −0.011 | −24.80; 14.99 | ALT (U/L) | 0.004 | −1.75; 2.07 |
| Hematocrit (%) | −0.025 | −10.73; 3.21 | γGT (U/L) | 0.027 | −0.33; 1.28 |
| Platelets (K/uL) | −0.010 | −0.43; 0.27 | FM (g) | 0.700 ** | 0.05; 0.05 |
| Lymphocytes (%) | 0.035 | −0.00; 0.00 | FM% | −0.047 | −11.02; 5.19 |
| Total Cholesterol (mg/dL) | −0.040 | −1.46; 0.15 | FFM (g) | 0.006 | −0.01; 0.01 |
| LDL Cholesterol (mg/dL) | −0.033 | −1.60; 0.27 | FFM % | 0.047 | −5.20; 11.02 |
| HDL Cholesterol (mg/dL) | −0.043 π,* | −4.25; −0.16 | T-score (sd) | −0.002 | −28.09; 25.54 |
| Triglycerides (mg/dL) | 0.101 ** | 0.73; 1.94 | Hip FRAX (%) | 0.048 | −0.34; 10.24 |
| Total blood proteins (g/dL) | −0.040 | −96.81; 5.15 | ASM/h2 | −0.005 | −31.58; 25.57 |
| Albumin (g/dL) | −0.013 | −88.84; 50.85 | Handgrip (kg) | −0.027 | −8.96; 2.90 |
| α 1 Globulin (%) | −0.007 | −26.65; 19.93 | Osteoporosis (N = 0/Y = 1) | 0.003 | −66,88; 74.30 |
| α 2 Globulin (%) | −0.029 | −24.27; 5.38 | Diabetes (N = 0/Y = 1) | 0.054 Ω.* | 16.27; 178.28 |
| β Globulins (%) | 0.065 ** | 7.34; 40.07 | Sarcopenia (N = 0/Y = 1) | 0.000 | −73.01; 71.59 |
| Variable | β | Confidence Interval 95% | Variable | β | Confidence Interval 95% |
|---|---|---|---|---|---|
| Gender (F = 0/M = 1) | 0.432 ** | 577.79; 783.44 | Fe (µg/dL) | −0.034 | −1.75; 0.23 |
| Age (years) | 0.006 | −4.16; 5.58 | Creatinine (mg/dL) | 0.082 ** | 49.75; 172.03 |
| Height (cm) | −0.635 * | −68.12; −31.87 | eGFR (mL/min) | −0.085 ** | −4.61; −1.16 |
| Weight (kg) | 1.260 ** | 44.06; 90.43 | Amylase (U/L) | 0.003 | −1.22; 1.41 |
| BMI (kg/m2) | −0.797 * | −170.84; −61.55 | Glycemia (mg/dL) | 0.084 ** | 0.74; 2.31 |
| MMSE (pts) | 0.160 | −9.46; 1.56 | Uric Acid (mg/dL) | 0.081 ** | 12.75; 43.13 |
| MNA (pts) | −0.026 | −16.96; 6.08 | ESR (mm/hr) | 0.011 | −0.81; 1.34 |
| White Blood Cells (K/uL) | 0.001 | −10.02; 10.34 | CRP (mg/dL) | 0.012 | −8.56; 14.61 |
| Red Blood Cells (M/uL) | −0.036 | −85.46; 8.04 | AST (U/L) | 0.011 | −1.61; 2.65 |
| Hemoglobin (g/dL) | −0.033 | −32.38; 4.71 | ALT (U/L) | 0.020 | −1.00; 2.77 |
| Hematocrit (%) | −0.033 | −11.34; 1.80 | γGT (U/L) | 0.039 | −1.00; 1.51 |
| Platelets (K/uL) | 0.018 | −0.20; 0.47 | FM (g) | 0.492 ** | 0.03; 0.05 |
| Lymphocytes (%) | 0.015 | −2.24; 4.50 | FM% | 0.118 Σ | −2.85; 17.40 |
| Total Cholesterol (mg/dL) | −0.004 | −0.81; 0.68 | FFM (g) | −0.050 *,Σ | −0.03; −0.01 |
| LDL Cholesterol (mg/dL) | −0.020 | −1.29; 0.51 | FFM % | −0.118 Σ | −17.40; 2.85 |
| HDL Cholesterol (mg/dL) | −0.065 ** | −5.16; −0.88 | T-score (sd) | −0.004 | −27.89; 23.56 |
| Triglycerides (mg/dL) | 0.127 ** | 1.05; 2.19 | Hip FRAX (%) | 0.029 | −2.22; 7.71 |
| Total blood proteins (g/dL) | −0.025 | −75.35; 20.91 | ASM/h2 | −0.171 **,Σ | −137.46; −57.17 |
| Albumin (g/dL) | −0.029 | −104.97; 24.24 | Handgrip (kg) | −0.040 Σ | −10.68; 1.52 |
| α 1 Globulin (%) | 0.024 | −10.35; 34.71 | Osteoporosis (N=0/Y=1) | 0.009 | −55.31; 82.51 |
| α 2 Globulin (%) | 0.012 | −10.68; 18.40 | Diabetes (N = 0/Y = 1) | 0.068 ** | 43.33; 201.80 |
| β Globulins (%) | 0.061 ** | 5.98; 37.45 | Sarcopenia (N = 0/Y = 1) | −0.045 Σ | −8.24; 147.46 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spadaccini, D.; Perna, S.; Peroni, G.; D’Antona, G.; Iannello, G.; Faragli, A.; Infantino, V.; Riva, A.; Petrangolini, G.; Negro, M.; et al. DXA-Derived Visceral Adipose Tissue (VAT) in Elderly: Percentiles of Reference for Gender and Association with Metabolic Outcomes. Life 2020, 10, 163. https://doi.org/10.3390/life10090163
Spadaccini D, Perna S, Peroni G, D’Antona G, Iannello G, Faragli A, Infantino V, Riva A, Petrangolini G, Negro M, et al. DXA-Derived Visceral Adipose Tissue (VAT) in Elderly: Percentiles of Reference for Gender and Association with Metabolic Outcomes. Life. 2020; 10(9):163. https://doi.org/10.3390/life10090163
Chicago/Turabian StyleSpadaccini, Daniele, Simone Perna, Gabriella Peroni, Giuseppe D’Antona, Giancarlo Iannello, Alessandro Faragli, Vittoria Infantino, Antonella Riva, Giovanna Petrangolini, Massimo Negro, and et al. 2020. "DXA-Derived Visceral Adipose Tissue (VAT) in Elderly: Percentiles of Reference for Gender and Association with Metabolic Outcomes" Life 10, no. 9: 163. https://doi.org/10.3390/life10090163
APA StyleSpadaccini, D., Perna, S., Peroni, G., D’Antona, G., Iannello, G., Faragli, A., Infantino, V., Riva, A., Petrangolini, G., Negro, M., Gasparri, C., & Rondanelli, M. (2020). DXA-Derived Visceral Adipose Tissue (VAT) in Elderly: Percentiles of Reference for Gender and Association with Metabolic Outcomes. Life, 10(9), 163. https://doi.org/10.3390/life10090163






