Abstract
Manganese nodules are an attractive source of base metals and critical and rare elements and are required to meet a high demand of today’s industry. In previous studies, it has been shown that high concentrations of reducing agent (Fe) in the system are beneficial for the rapid extraction of manganese. However, it is necessary to optimize the operational parameters in order to maximize Mn recovery. In this study, a statistical analysis was carried out using factorial experimental design for the main parameters, including time, MnO2/Fe2O3 ratio, and H2SO4 concentration. After this, Mn recovery tests were carried out over time at different ratios of MnO2/Fe2O3 and H2SO4 concentrations, where the potential and pH of the system were measured. Finally, it is concluded that high concentrations of FeSO4 in the system allow operating in potential and pH ranges (−0.2 to 1.2 V and −1.8 to 0.1) that favor the formation of Fe2+ and Fe3+, which enable high extractions of Mn (73%) in short periods of time (5 to 20 min) operating with an optimum MnO2/Fe2O3 ratio of 1:3 and a concentration of 0.1 mol/L of H2SO4.
1. Introduction
The oxides of Fe and Mn are formed by direct precipitation in ambient seawater and are mainly deposited on the flat parts and the flanks of seamounts, where ocean currents prevent sedimentation [1,2]. These deposits are found in the oceans around the world [3] and among these are the manganese nodules [4].
The economic interest in ferromanganese (Fe-Mn) nodules is due to high grades of base, critical, and rare metals [5]. These metals that provide mineral deposits on the seabed are necessary for the rapid development of high technology application. They also support the growth and quality of life of the middle class in densely populated countries with expanding markets and economies [6]. Manganese is the most abundant marine nodule metal, with an average content of around 24% [7].
In order to dissolve Mn present in marine nodules in acidic media, it is necessary to use a reducing agent [8]. Studies have reported that the acid leaching of manganese nodules with the use of Fe as the reducing agent is efficient at room temperature [8,9,10,11]. In a study conducted by Zakeri et al. [8], ferrous ions were added for the reductive dissolution of manganese nodules. The authors indicated that in a molar H2SO4/MnO2 ratio of 2:1 and a molar Fe2+/MnO2 ratio of 3:1, it was possible to dissolve 90% of Mn in 20 min at 20 °C. Bafgui et al. [9] performed acid leaching of manganese nodules by adding Fe, comparing their results with those previously obtained by Zakeri et al. [8]. The authors concluded that when operating with high Fe/MnO2 ratios, Fe0 is a more efficient reducing agent compared to Fe2+ because it maintains high activity in the system through the regeneration of ferrous ions.
For the acidic leaching of marine nodules with the use of residues (tailings and slags) containing Fe2O3, only two studies have been presented [10,11]. In the studies carried out by Toro et al. [10] and Toro et al. [11], it was shown that variables, such as particle size and agitation speed, do not majorly influence the dissolution of MnO2 and that the most important variable is the Fe2O3 concentration in the system. In the study carried out by Toro et al. [10] with the use of smelter slag, extraction of 70% of Mn was achieved in 40 min when operating at a MnO2/Fe2O3 ratio of 1:2, a particle size of −47 + 38 μm, and a H2SO4 concentration of 1 mol/L. In the later study carried out by Toro et al. [11], involving the use of tailings, it was demonstrated that under the same operating conditions as in Toro et al. [10], greater extraction of Mn (77%) was achieved because tailings are more amenable to leaching. In both studies, the following Reactions (R1)–(R9) involving the use of Fe2O3 were proposed.
(Fe2+ Fe23+)O4(s) + 2H+(aq) = (Fe23+)O3 + Fe2+(aq) + H2O(l)
3(Fe2+ Ti)O3(s) + 6H+(aq) = 3TiO2 + 3Fe2+(aq) + 3H2O(l)
Fe2O3 + H2SO4 = Fe2(SO4)3 + H2O(l)
Fe3O4(s) + 4H2SO4(aq) = FeSO4 + Fe2(SO4)3 + 4H2O(l)
Fe2(SO4)3 + H2O = Fe(OH)3 + Fe(s) + H2(aq) + H2SO4(aq) + O2
FeSO4(s) + H2O(aq) = Fe(s) + H2SO4(aq) + O2
MnO2(s) + Fe2+(aq) + H+(aq) = Fe3+(aq) + Mn2+(aq) + H2O(l)
MnO2(s) + Fe(s) + 8H+(aq) = 2Fe3+(aq) + Mn2+(aq) + 2H2O(l) + 2H2(g)
MnO2(s) + 2/3Fe(s) + 4H+(aq) = Mn2+(aq) + 2/3Fe3+(aq) + 2H2O(l)
However, in previous studies [8,9], thermodynamic aspects were not considered.
Table 1 reports the statistical information of the reactions of interest with iron as a reducing agent and its transformations during manganese leaching. It is emphasized that, unlike previous investigations [11], under these conditions, elemental iron (Fe0) was not formed, since this reaction is not spontaneous (G = 744.22 kJ) and requires a lot of energy. On the other hand, the main reducing agent is ferrous sulfate (FeSO4), which is produced from the reaction between magnetite (Fe3O4, mostly present in tailings) and sulfuric acid (Equation (2)). With this reducing agent, it is possible to reduce manganese present in pyrolusite (Mn4+), obtaining a manganese sulphate (Mn2+), as observed in Equation (5).

Table 1.
Thermodynamic information of the reactions (based on HSC Chemistry 5.1).
The smelting slag is one of the main solid wastes of the copper industry and the produced volume increases day by day [12]. In Chile, the smelters produce 163 tons of slag per day [13] and companies such as Altonorte perform slag flotation for the recovery of Cu. During flotation for each ton of Cu obtained, 151 tons of tailings are generated [14], which are mainly disposed of in tailing dams and represent the most significant environmental liability according to their size and risk of a mining site [15]. Another example is what happened in Lavrio, Greece, due to the intensive mining and metallurgical activities in the last century. This generated huge amount of waste, including acid-generating sulfidic tailings, carbonaceous tailings, and slags. Quantification of the human health risks indicated that direct ingestion of contaminated particles is the most important exposure route for the intake of contaminants by humans [16]. Komnitsas et al. [17] conducted research on waste generated by intensive mining and mineral processing activities in Navodari and Baia, on the Romanian Black Sea coast. Analyzing the experimental results and the associated risks, the authors conclude that it is necessary to rehabilitate the affected areas through removal of toxic and heavy elements from sulphidic tailings and leachates with biosorption and biosolubilisation techniques and development of a vegetative cover on phosphogypsnm stacks and sulphidic tailings dumps. For this reason, it is important to highlight the importance of studying options to reuse waste generated from metallurgical processes.
In Chile, ocean mining is not regulated and is also under-exploited for security reasons [18]. Due to this, it is not possible to carry out a cost-effectiveness study on the extraction of nodules from sea depths. Mining technologies have been developed in the world for the extraction of polymetallic nodules [19]. However, there is no study indicating cost differences between the different methods available on the market. In spite of this, it is necessary to continue investigating processes for the extraction of elements from marine nodules, because technologies are being developed to collect minerals from sea beds and, in the near future, they could be considered as viable alternatives to meet the high demand for metals.
In this investigation, the use of Fe2O3, which is present in tailings, to facilitate reductive leaching of MnO2 from marine nodules for the recovery of Mn is evaluated. The objective is to minimize these environmental liabilities and optimize the most important process variables (time, acid concentration, and MnO2/Fe2O3 ratio).
2. Materials and Methods
2.1. Manganese Nodule Sample
The marine nodules used in this work were the same as those previously used in Toro et al. [11]. They were analyzed by means of atomic emission spectrometry by induction-coupled plasma (ICP-AES), developed in the applied geochemistry laboratory of the department of geological sciences of the Catholic University of the North. They contained 15.96% Mn and 0.45% Fe; Mn was present as MnO2 (29.85%) and Fe as Fe2O3 (26.02%).
2.2. Tailings
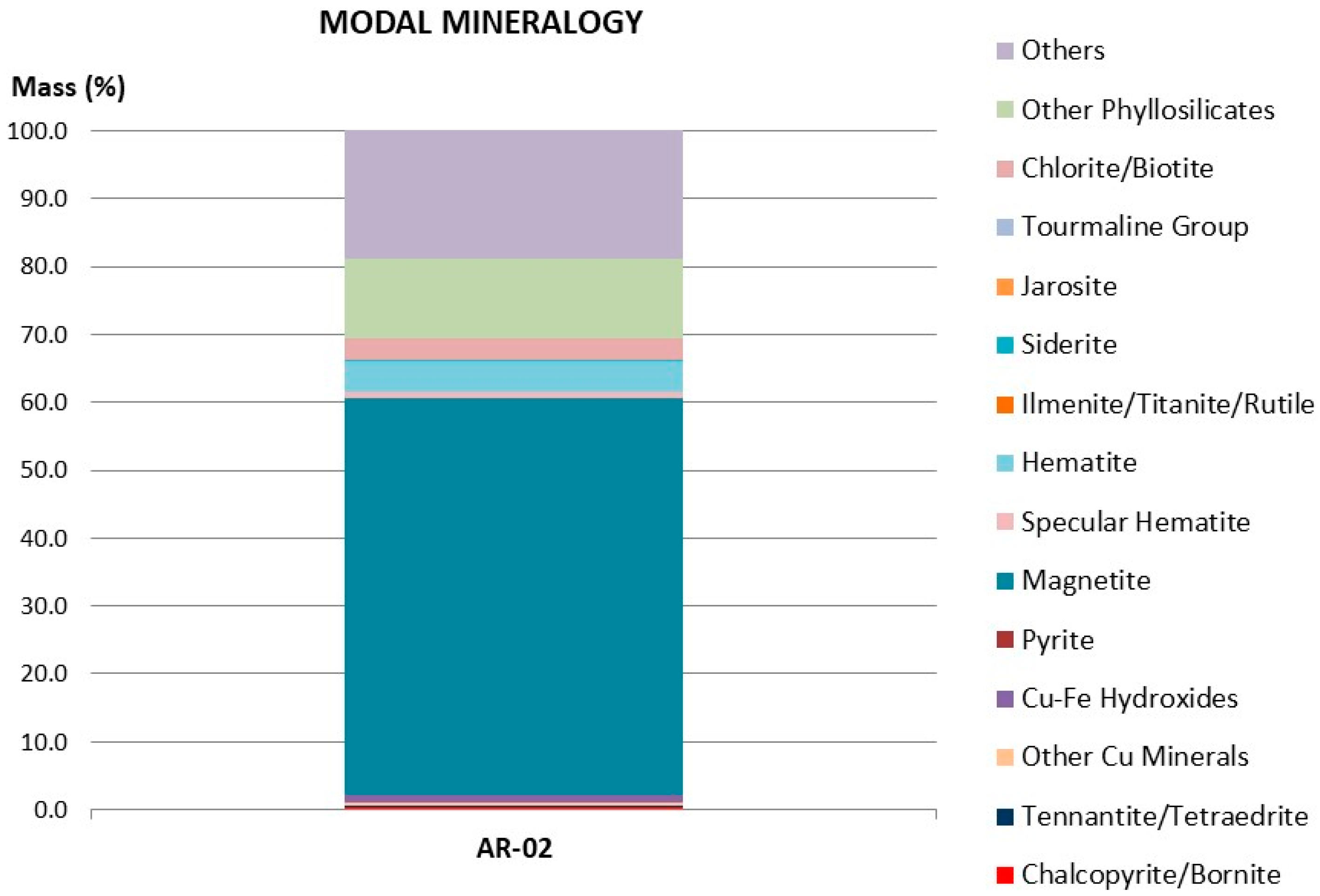
The tailings used for the present investigation were the same as those used in Toro et al. [11]. The methods used to determine their chemical and mineralogical composition are the same as those used for the analysis of the manganese nodules. Figure 1 shows the chemical species determined by QEMSCAN. There were several phases that contained iron (mainly magnetite (58.52%) and hematite (4.47%), while the content of Fe was estimated at 41.90%.
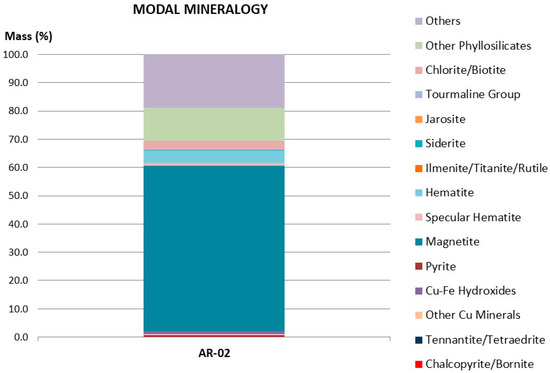
Figure 1.
Detailed modal mineralogy.
2.3. Reagents Used—Leaching Parameters
The sulfuric acid used for the leaching tests was grade PA, with 95–97% purity, a density of 1.84 kg/L, and a molecular weight of 98.80 g/mol. The leaching tests were carried out in a 50 mL glass reactor with a 0.01 solid:liquid ratio. A total of 200 mg of Mn nodules were maintained in suspension with the use of a 5 position magnetic stirrer (IKA ROS, CEP 13087-534, Campinas, Brazil) at a speed of 600 rpm. The tests were conducted at a room temperature of 25 °C, while the studied variables were additives, particle size, and leaching time. Also, the tests were performed in duplicate and measurements (or analyses) were carried out on 5 mL undiluted samples using atomic absorption spectrometry with a coefficient of variation ≤5% and a relative error between 5% and 10%. Measurements of pH and oxidation-reduction potential (ORP) of leach solutions were made using a pH-ORP meter (HANNA HI-4222). The ORP solution was measured using a combination of an ORP electrode cell composed of a platinum operating electrode and a saturated Ag/AgCl reference electrode. The solid waste obtained was analyzed by XRD with the use of a Bruker brand diffractometer; the patterns of the main crystalline phases were obtained using Eva software.
2.4. Estimation of Linear and Interaction Coefficients for Complete Factorial Designs of Experiments of 33
In previous studies [8,9,10,11], in which the dissolution of Mn from marine nodules was investigated with the use of Fe as a reducing agent, it was demonstrated that for high concentrations of Fe in the system (ratios of Fe/MnO2 greater than 1), quite high extractions were obtained (over 70%) in short periods of time (5 to 30 min). The studies conducted by Bafghi et al. [9] and Toro et al. [10] indicated that the concentration of Fe in the system is the most important variable in order to shorten MnO2 dissolution times; it was also found that the concentration of H2SO4 is not an important parameter. However, in these studies, it was not possible to indicate an optimum MnO2/Fe ratio and H2SO4 concentration in relation to time. In order to overcome this and elucidate Mn extraction from marine nodules, three independent variables were selected for the factorial design of 33 experiments, namely time, sulfuric acid concentration, and MnO2/Fe2O3 ratio. This approach allows the determination of the effect of the most relevant factors, as well as their levels, and the development of an experimental model that allows through the determination of coefficients the optimization of the response variable [20,21,22]. A factorial design was applied involving three factors, each one having three levels; thus, 27 experimental tests were carried out. Minitab 18 software was used for the experimental design and development of a multiple regression equation [23].
Then, the response variable was expressed based on the linear effect of the variables of interest and considering the effects of interaction and curvature, as shown in Equation (6).
where α is an overall constant, xi is the value of the level “i” of the factor x, βi is the coefficient of the linear factor xi, βij is the coefficient of the interactions xi × xj, n is the level of the factor, and Mn extraction is the dependent variable.
Mn Extraction (%) = α + β1 × x1 + β2 × x2 + β3 × x3 + β12 × x1 × x2 + β13 × x1 × x3 + β23 × x2 × x3 + β11 × x12 + β22 × x22 + β33 × x32,
Table 2 shows the values of the levels for each factor, while Table 3 shows the recovery obtained for each configuration.

Table 2.
Experimental conditions.

Table 3.
Experimental configuration and Mn extraction.
3. Results and Discussion
3.1. Effect of Variables
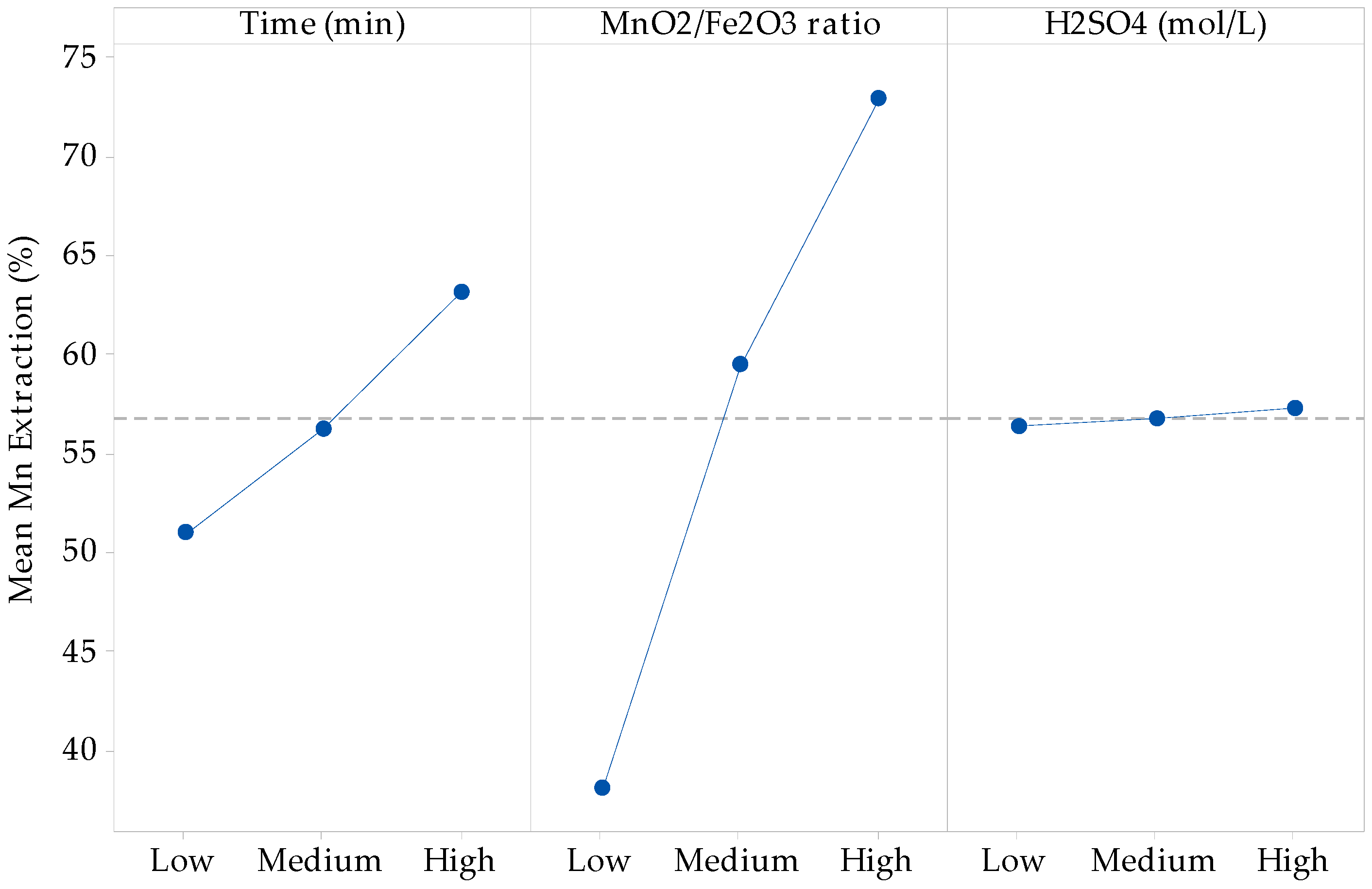
From the principal components analysis, it is seen that there is no main effect of the sulfuric acid concentration factor, which means that the average response is the same across all levels of the factor, while the time and MnO2/Fe2O3 ratio factors have a main effect since the variation between the different levels affects the response differently, as shown in main effects plot for Mn extraction of Figure 2. Developing the ANOVA test and the multiple linear regression adjustment, the recovery according to the predictive variables of time and MnO2/Fe2O3 ratio is given by Equation (7).
where x1 represents the time factor and x2 represents the MnO2/Fe2O3 ratio (previous coding). Then, it is seen that the double and triple interaction factors, together with the curvature of time and H2SO4 concentration factors, do not contribute to the explanation of the variability of the model.
Mn Extraction (%) = 53.90 + 6.12 × x1 − 17.40 × x2 − 4.00 × x22,
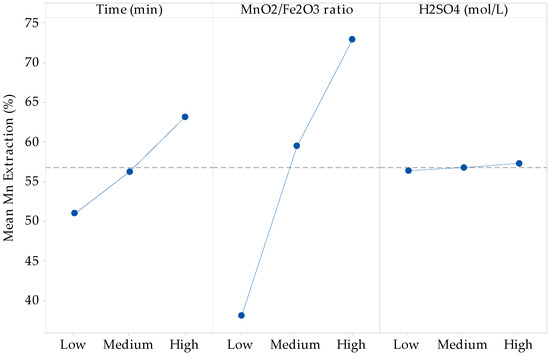
Figure 2.
Linear effect plot for Mn extraction (%).
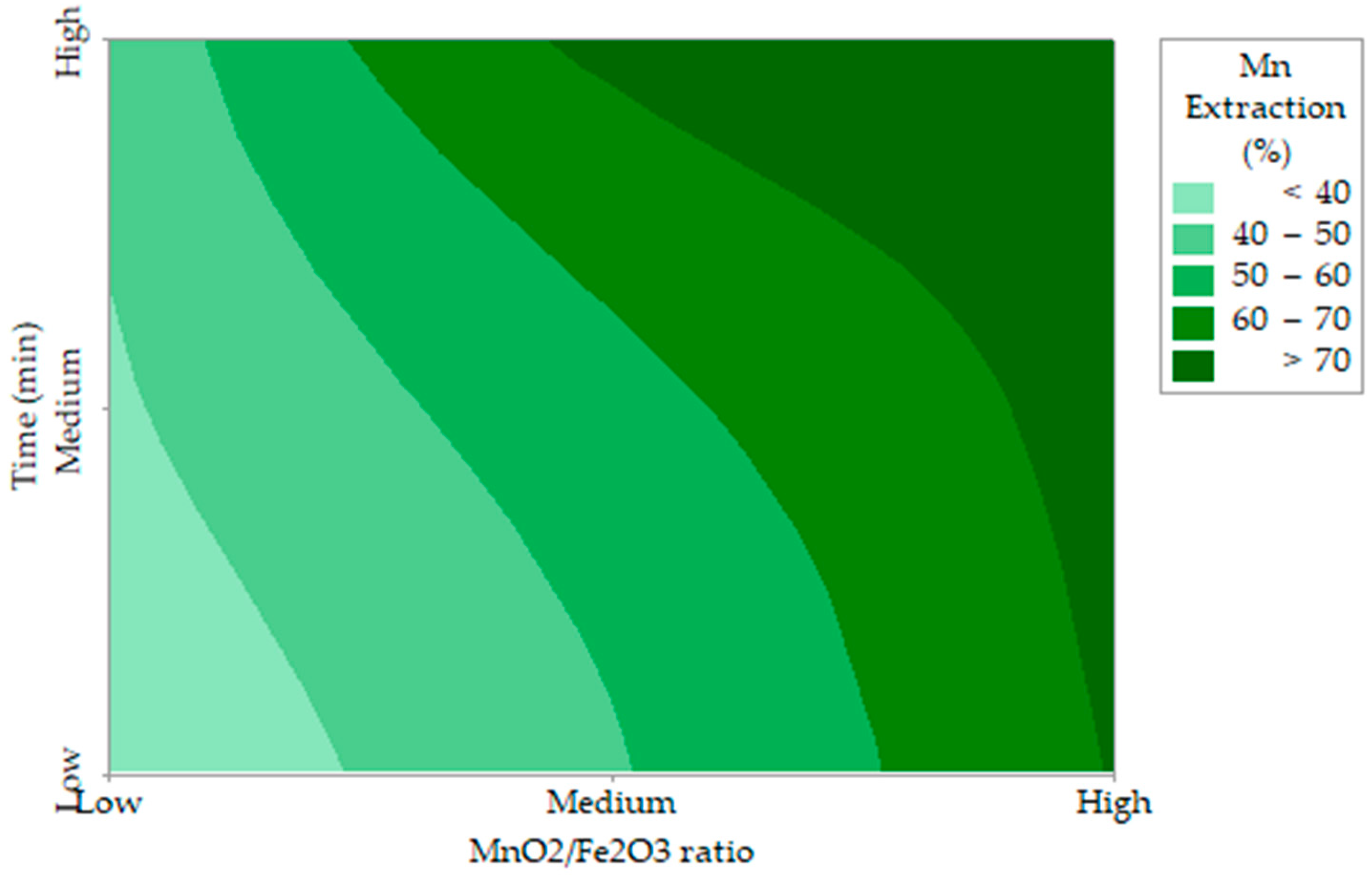
A gradient analysis of manganese extraction, ∇Mn Extraction (x1, x2) = (6.12, −23.40), indicates an increase in the positive direction of the predictor variables. The response decreases faster with respect to the variable x2 than with respect to the variable x1, as shown in Figure 3.
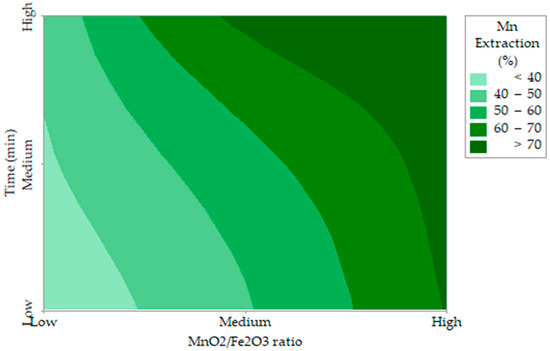
Figure 3.
Contour plot of Mn extraction (%) versus MnO2/Fe2O3, time (min).
The ANOVA test indicates that the model adequately represents manganese extraction for the set of sampled values. Also, the model does not require additional adjustments and is validated by the following goodness-of-fit statistics. The p-value of the model (p < 0.05) indicates that it is statistically significant. The value of the R2 statistic is 94.94%, which indicates that approximately 95% of the total variability is explained by the model, while the predictive R2 is 92.79%, indicating that the model can adequately predict the response to new observations. The F test indicates the significance of the model, given that FRegression(143.89) >> (FTable = F3,23(3.03)), while the residual normality test indicates that these are distributed with media −7.41 × 10−7, with a standard deviation of 3.5. The p-value of the Kolmogorov-Smirnov test is greater than the level of significance, so it is not possible to reject the assumption of the regression model, which is that the residuals are normally distributed.
3.2. Effect on Acid Concentration and MnO2/Fe2O3 Ratio
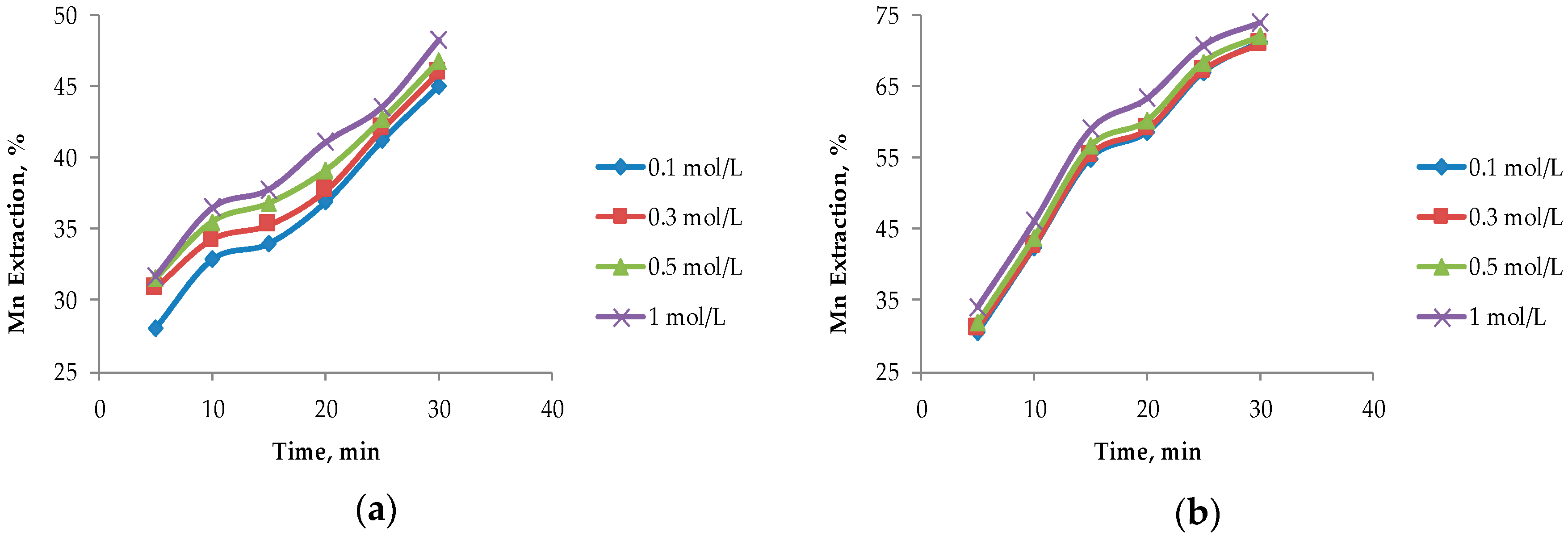
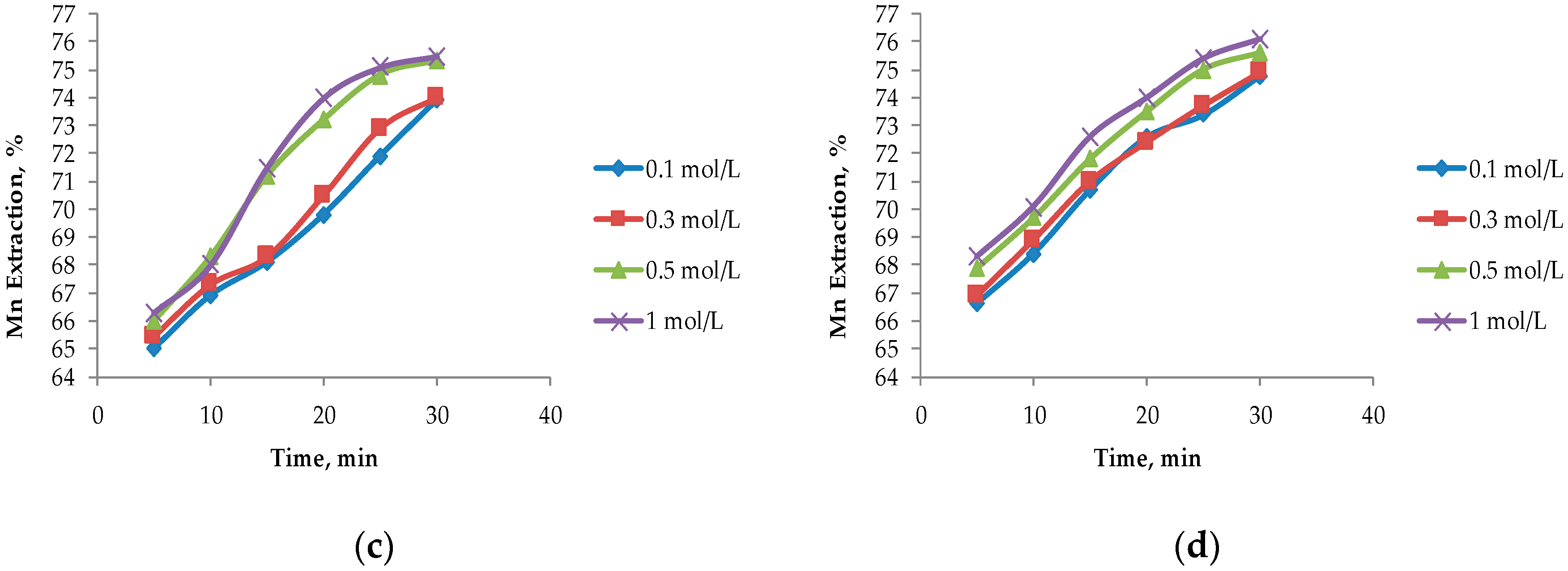
In Figure 4, it can be seen that the largest extractions of Mn from marine nodules are obtained when operating at MnO2/Fe2O3 ratios less than 1:1, which agrees with the theories proposed by Kanungo et al. [24], Zakeri et al. [8], Bafghi et al. [9], and Toro et al. [10], which mentioned that the presence of more Fe than MnO2 in the system improved Mn dissolution in short periods of time. The highest Mn dissolution (76.10%) was obtained by operating at a MnO2/Fe2O3 ratio of 1:3 with a H2SO4 concentration of 1 mol/L at 30 min. However, this extraction is not far from that obtained when operating at a MnO2/Fe2O3 ratio of 1:2 (75.50%) at the same acid concentration. For the MnO2/Fe2O3 ratios described in Figure 4b–d, it can be seen that, for leaching times of 30 min, very similar values are obtained, but much higher dissolution kinetics are seen in 1:2 and 1:3 ratios where higher than 65% recoveries of Mn are obtained after 5 min. For a MnO2/Fe2O3 ratio of 2:1 (Figure 4a), much lower dissolution is obtained compared to the other cases under the same operating conditions; the maximum dissolution achieved is 48.30% after 30 min when the H2SO4 concentration is 1 mol/L.
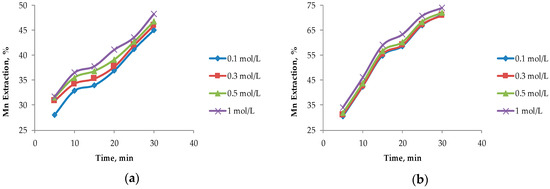
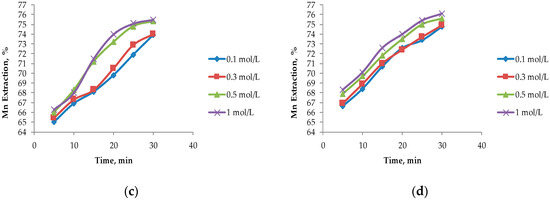
Figure 4.
Effect on the acid concentration at different ratios of MnO2/Fe2O3: (a) Ratio 2:1; (b) ratio 1:1; (c) ratio 1:2; and (d) ratio 1:3 (25 °C, 600 rpm, −75 + 53 μm).
The concentration of H2SO4 in the system is not significant when MnO2/Fe2O3 ratios are 1:2 and 1:3; this finding agrees with those of Toro et al. [10], who mention that high concentrations of Fe2O3 in the system are independent of the acid concentration. However, it can be observed that this factor has a greater impact as long as there is a lower concentration of reducing agent (FeSO4) in the system. For a MnO2/Fe2O3 ratio of 2:1, there is a difference of 3.30% between 0.1 and 1 mol/L of H2SO4.
For these two variables analyzed under the exposed operational parameters, it can be observed that when operating at a MnO2/Fe2O3 ratio of 1:2, at a concentration of H2SO4 of 0.5 mol/L at 20 min, similar results like those obtained when operating in a MnO2/Fe2O3 ratio of 1:3 (73.50% approximately) are obtained. This is consistent with what was proposed by Toro et al. [11], who indicated that, when operating at high concentrations of Fe2O3 in the system, the dissolution kinetics of MnO2 were drastically increased and significant differences were only observed in short periods of time (5 to 10 min). However, for the second case mentioned in Figure 4d, better results are obtained at low acid concentrations (0.1 mol/L). For this reason, it can be concluded that it is more convenient to operate at MnO2/Fe2O3 ratios of 1:3 and low concentrations of acid (0.1 mol/L) at 20 min. This is because the tailings are wastes that do not have a commercial value and their reuse is beneficial, while the increase of the acid concentration in the system results in a direct increase in the cost of the process.
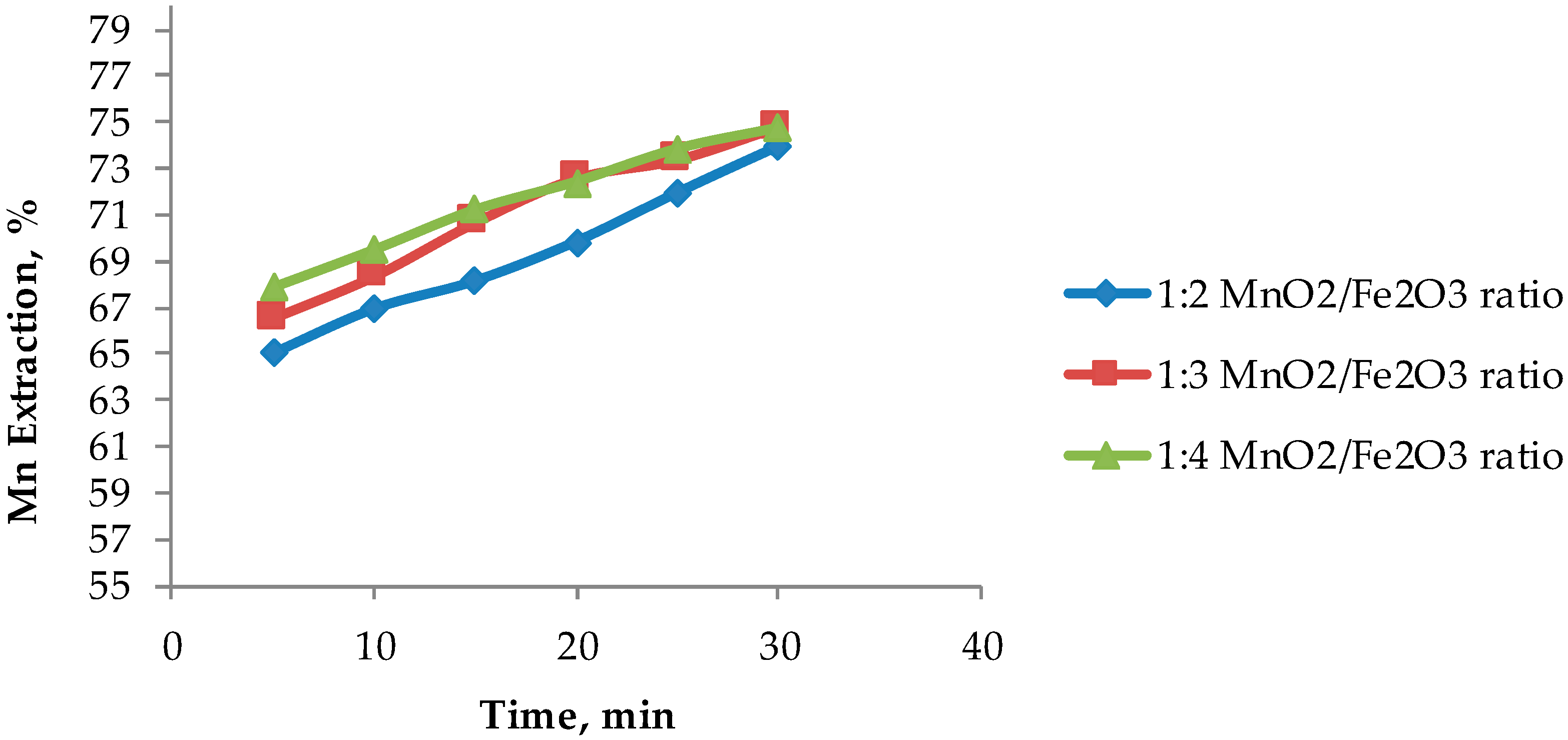
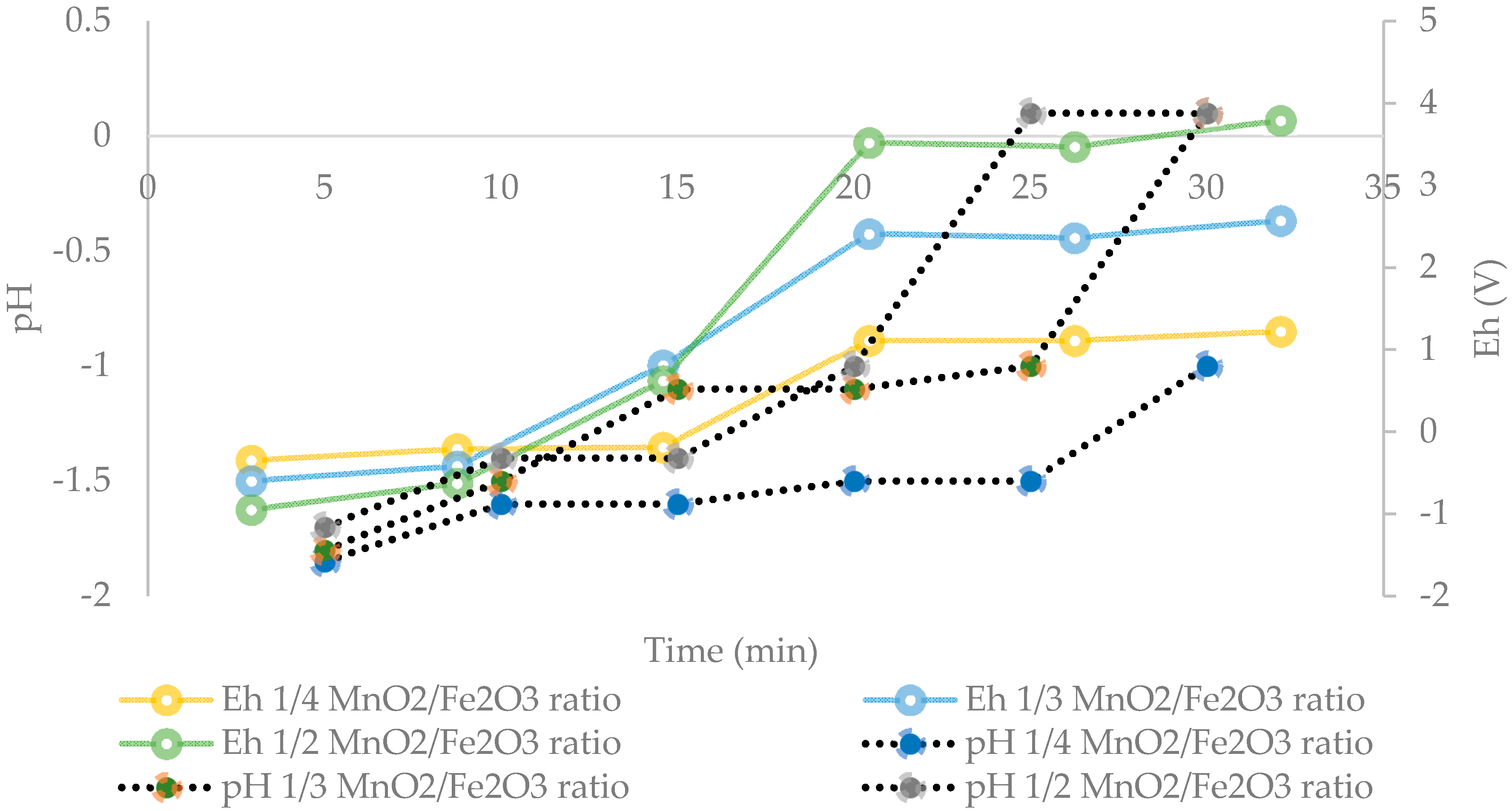
Based on the results presented in Figure 4, Figure 5 shows that an optimum MnO2/Fe2O3 ratio can be determined. It can be seen that there is no difference in manganese extractions when operating at 1:3 and 1:4 ratios. For short periods of time (5 to 20 min), it can be observed that there are greater extractions for ratios higher than 1:2, achieving dissolutions of Mn over 70% at 15 min. However, it can be seen that at 30 min the results converge in extractions of approximately 75%. Finally, it can be indicated that for times between 15 to 25 min, it is convenient to operate at a MnO2/Fe2O3 ratio of 1:3, while at 30 min, the optimum ratio is 1:2. Figure 6 shows the potential and pH values obtained in the tests presented in Figure 5, which vary between (−0.2 V to 1.2 V) and (−1.8 to 0.1), respectively.
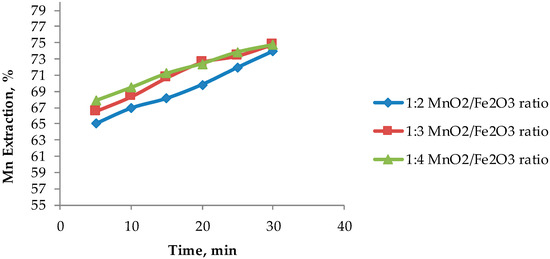
Figure 5.
Effect of the MnO2/Fe2O3 ratio on manganese extraction (25 °C, 600 rpm, −75 + 53 μm, acid concentration of 0.1 mol/L).
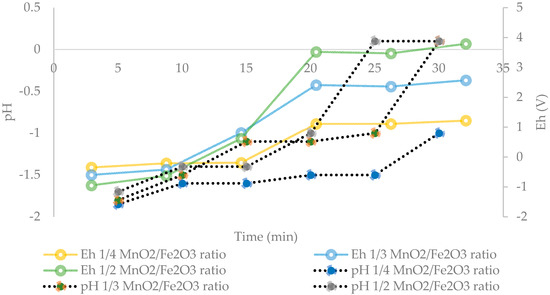
Figure 6.
Effect of the potential and pH in solution at different MnO2/Fe2O3 ratios (25 °C, 600 rpm, −75 + 53 μm, acid concentration of 0.1 mol/L).
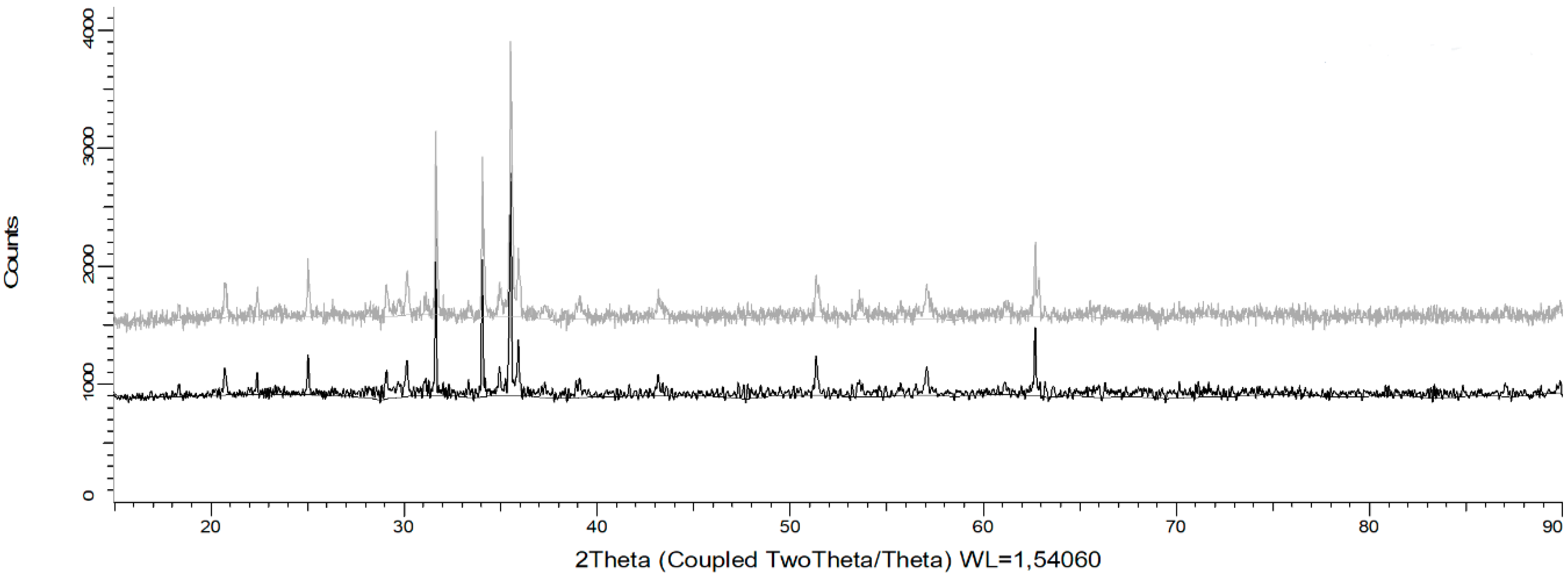
Senanayake [25] stated that during reductive leaching of MnO2 with the use of FeSO4 as a reducing agent, the values of potential and pH must be in the range of −0.4 to 1.4 V and −2 to 0.1 in order to dissolve Mn. In addition, it is indicated that the divalent Fe (II), produced by the partial acid dissolution of Fe3O4, acts as a reducing agent for MnO2. Under these conditions, Mn ions remain in solution and do not precipitate through oxidation-reduction reactions by the presence of Fe2+ and Fe3+ ions [26]. This can be seen in Figure 7, when analyzing the residues of the present study by XRD analysis and mainly the presence of fayalite (Fe2+2 SiO4), magnetite (Fe3O4), and gypsum (CaSO4·2H2O) is observed. It is concluded that, in these residues, no Fe precipitates were generated from the solution when tailings were added. In future studies, it may be interesting to perform a kinetic study to elucidate the effect of temperature in order to determine the Mn dissolution mechanisms from marine nodules in very short periods of time (5 min).
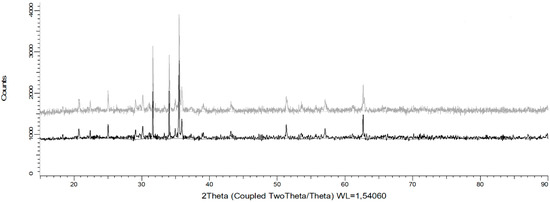
Figure 7.
X-ray diffractogram of solid waste after leaching for 30 min at 25 °C using a MnO2/Fe2O3 ratio of 1:2, 600 rpm, −75 + 53 μm, and an acid concentration of 0.1 mol/L.
4. Conclusions
The present study shows results by means of a statistical model as well as extraction curves versus time to investigate the extraction of Mn from MnO2 present in manganese nodules using tailings obtained from slag flotation when operating in an acid medium and a room temperature of 25 °C. FeSO4 proves to be a good reducing agent, shortening the dissolution time of MnO2. The main findings are the following:
- At 30 min, the optimum MnO2/Fe2O3 ratio is 1:2, with a H2SO4 concentration of 0.1 mol/L, achieving a Mn extraction of 74%.
- For short periods of time (5 to 20 min), the optimum MnO2/Fe2O3 ratio is 1:3, with a H2SO4 concentration of 0.1 mol/L, achieving a Mn extraction between 68% and 73%.
- When operating at MnO2/Fe2O3 ratios lower than 1:1, the concentration of acid in the system is not an important factor.
- High concentrations of FeSO4 in the system allow the operation in potential and pH ranges, which favor the generation of Fe2+ and Fe3+; thus, the formation of Fe precipitates is avoided.
The reductive leaching of marine nodules in an acidic medium with the addition of tailings is an attractive and cost efficient alternative and results in high extraction of Mn in short periods of time with the use of low concentrations of acid. In the future, a study should be carried out to improve the economic viability of the process.
Author Contributions
N.T. contributed in project administration, investigation and wrote paper, M.S. and E.G. contributed in the data curation and software, M.C. and E.T. contributed in validation and supervision and J.C and P.C.H. performed the experiments, review and editing.
Funding
This research received no external funding.
Acknowledgments
The authors are grateful for the contribution of the Scientific Equipment Unit- MAINI of the Universidad Católica del Norte for aiding in generating data by automated electronic microscopy QEMSCAN®, and for facilitating the chemical analysis of the solutions. We are also grateful to the Altonorte Mining Company for supporting this research and providing slag for this study, and we thank Marina Vargas Aleuy, María Barraza Bustos and Carolina Ossandón Cortés of the Universidad Católica del Norte for supporting the experimental tests.
Conflicts of Interest
The authors declare they have no conflict of interest.
References
- Konstantinova, N.; Cherkashov, G.; Hein, J.R.; Mirão, J.; Dias, L.; Madureira, P.; Kuznetsov, V.; Maksimov, F. Composition and characteristics of the ferromanganese crusts from the western Arctic Ocean. Ore Geol. Rev. 2017, 87, 88–99. [Google Scholar] [CrossRef]
- Hein, J.R.; Koschinsky, A. Deep-Ocean Ferromanganese Crusts and Nodules. In Treatise on Geochemistry; Elsevier: Amsterdam, The Netherlands, 2014; Volume 13, pp. 273–291. [Google Scholar]
- Marino, E.; González, F.J.; Somoza, L.; Lunar, R.; Ortega, L.; Vázquez, J.T.; Reyes, J.; Bellido, E. Strategic and rare elements in Cretaceous-Cenozoic cobalt-rich ferromanganese crusts from seamounts in the Canary Island Seamount Province (northeastern tropical Atlantic). Ore Geol. Rev. 2017, 87, 41–61. [Google Scholar] [CrossRef]
- Josso, P.; Pelleter, E.; Pourret, O.; Fouquet, Y.; Etoubleau, J.; Cheron, S.; Bollinger, C. A new discrimination scheme for oceanic ferromanganese deposits using high fi eld strength and rare earth elements. Ore Geol. Rev. 2017, 87, 3–15. [Google Scholar] [CrossRef]
- Cronan, D.S. Cobalt-rich ferromanganese crusts in the Pacific. In Handbook of Marine Mineral Deposits; CRC Press: Boca Raton, FL, USA, 2000; pp. 239–279. [Google Scholar]
- Hein, J.R.; Cherkashov, G.A. Preface for Ore Geology Reviews Special Issue: Marine Mineral Deposits: New resources for base, precious, and critical metals. Ore Geol. Rev. 2017. [Google Scholar] [CrossRef]
- Sharma, R. Environmental Issues of Deep-Sea Mining. Procedia Earth Planet. Sci. 2015, 11, 204–211. [Google Scholar] [CrossRef]
- Zakeri, A.; Bafghi, M.S.; Shahriari, S.; Das, S.C.; Sahoo, P.K.; Rao, P.K. Dissolution kinetics of manganese dioxide ore in sulfuric acid in the presence of ferrous ion. Hydrometallurgy 2007, 8, 22–27. [Google Scholar]
- Bafghi, M.S.; Zakeri, A.; Ghasemi, Z.; Adeli, M. Reductive dissolution of manganese ore in sulfuric acid in the presence of iron metal. Hydrometallurgy 2008, 90, 207–212. [Google Scholar] [CrossRef]
- Toro, N.; Herrera, N.; Castillo, J.; Torres, M.C.; Sepúlveda, R.S. Initial Investigation into the Leaching of Manganese from Nodules at Room Temperature with the Use of Sulfuric Acid and the Addition of Foundry Slag—Part I. Minerals 2018, 8, 565. [Google Scholar] [CrossRef]
- Toro, N.; Saldaña, M.; Castillo, J.; Higuera, F.; Acosta, R. Leaching of Manganese from Marine Nodules at Room Temperature with the Use of Sulfuric Acid and the Addition of Tailings. Minerals 2019, 9, 289. [Google Scholar] [CrossRef]
- Alejandra, C.; Estay, S. Utilización de Escorias de Fundición para la Producción de Compuestos de Hierro; Universidad de Chile: Santiago, Chile, 2006. [Google Scholar]
- Vásquez, M. En Chile Diariamente se Desecha Cobre Avaluado en una Cifra Cercana a Los 450 Mil dólares. 2019. Available online: https://www.pucv.cl/uuaa/vriea/noticias/nuestros-investigadores/en-chile-diariamente-se-desecha-cobre-avaluado-en-una-cifra-cercana-a/2016-08-05/124009.html (accessed on 7 April 2019).
- COCHILCO. Sulfuros primarios: Desafíos y oportunidades I Comisión Chilena del Cobre; COCHILCO: San Diego, Chile, 2017. [Google Scholar]
- Medvinsky-Roa, G.; Caroca, V.; Vallejo, J. Informe sobre la situación de los Relaves Mineros en Chile para ser presentado en el cuarto informe periódico de Chile para el Comité de Derechos Económicos, Sociales y Culturales, perteneciente al consejo Económico Social de la Naciones Unidas. Providencia. 2015. Available online: https://tbinternet.ohchr.org/Treaties/CESCR/Shared Documents/CHL/INT_CESCR_CSS_CHL_20605_S.pdf (accessed on 7 April 2019).
- Xenidis, A.; Papassiopi, N.; Komnitsas, K. Carbonate-rich mining tailings in Lavrion: Risk assessment and proposed rehabilitation schemes. Adv. Environ. Res. 2003, 7, 207–222. [Google Scholar] [CrossRef]
- Komnitsas, K.; Kontopoulos, A.; Lazar, I.; Cambridge, M. Risk assessment and proposed remedial actions in coastal tailings disposal sites in Romania. Miner. Eng. 1998, 11, 1179–1190. [Google Scholar] [CrossRef]
- SERNAGEOMIN. Anuario de la Mineria de Chile 2017; SERNAGEOMIN: San Diego, Chile, 2017. [Google Scholar]
- ISA. Polymetallic Nodule Mining Technology: Current Trends and Challenges Ahead; ISA: Chennai, India, 2008; p. 276.
- Douglas, C. Montgomery: Design and Analysis of Experiments, 8th ed.; John Wiley & Sons: New York, NY, USA, 2012. [Google Scholar]
- Ghosh, M.K.; Barik, S.P.; Anand, S. Sulphuric acid leaching of polymetallic nodules using paper as a reductant. Trans. Indian Inst. Met 2008, 61, 477–481. [Google Scholar] [CrossRef]
- Mitić, M.; Tošić, S.; Pavlović, A.; Mašković, P.; Kostić, D.; Mitić, J.; Stevanović, V. Optimization of the extraction process of minerals from Salvia officinalis L. using factorial design methodology. Microchem. J. 2019, 145, 1224–1230. [Google Scholar] [CrossRef]
- Mathews, P.G.; William, A. Design of Experiments with MINITAB; William A. Tony: Milwaukee, WI, USA, 2005. [Google Scholar]
- Kanungo, S.B. Rate process of the reduction leaching of manganese nodules in dilute HCl in presence of pyrite. Part I. Dissolution behaviour of iron and sulphur species during leaching. Hydrometallurgy 1999, 52, 313–330. [Google Scholar] [CrossRef]
- Senanayake, G. Acid leaching of metals from deep-sea manganese nodules—A critical review of fundamentals and applications. Miner. Eng. 2011, 24, 1379–1396. [Google Scholar] [CrossRef]
- Komnitsas, K.; Bazdanis, G.; Bartzas, G.; Sahinkaya, E.; Zaharaki, D. Removal of heavy metals from leachates using organic/inorganic permeable reactive barriers. Desalin. Water Treat. 2013, 51, 3052–3059. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).