Preparation of Modified Montmorillonite and Its Application to Rare Earth Adsorption
Abstract
:1. Introduction
2. Experimental
2.1. Materials and Apparatus
2.2. Preparation of Modified Mt
2.3. Analysis and Characterization Methods
2.4. Adsorption and Elution Experiment
2.4.1. Adsorption Experiment
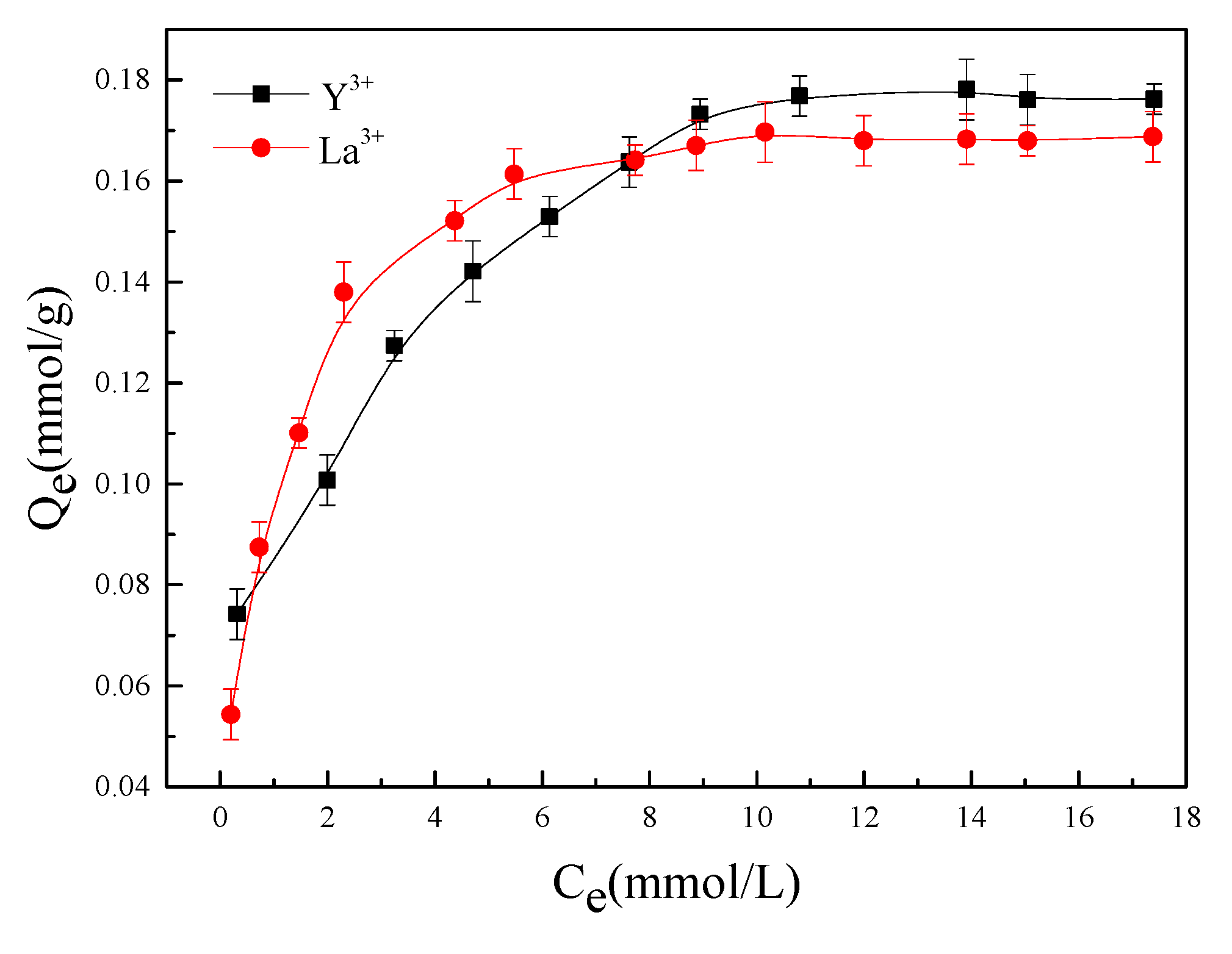
2.4.2. Isothermal Adsorption
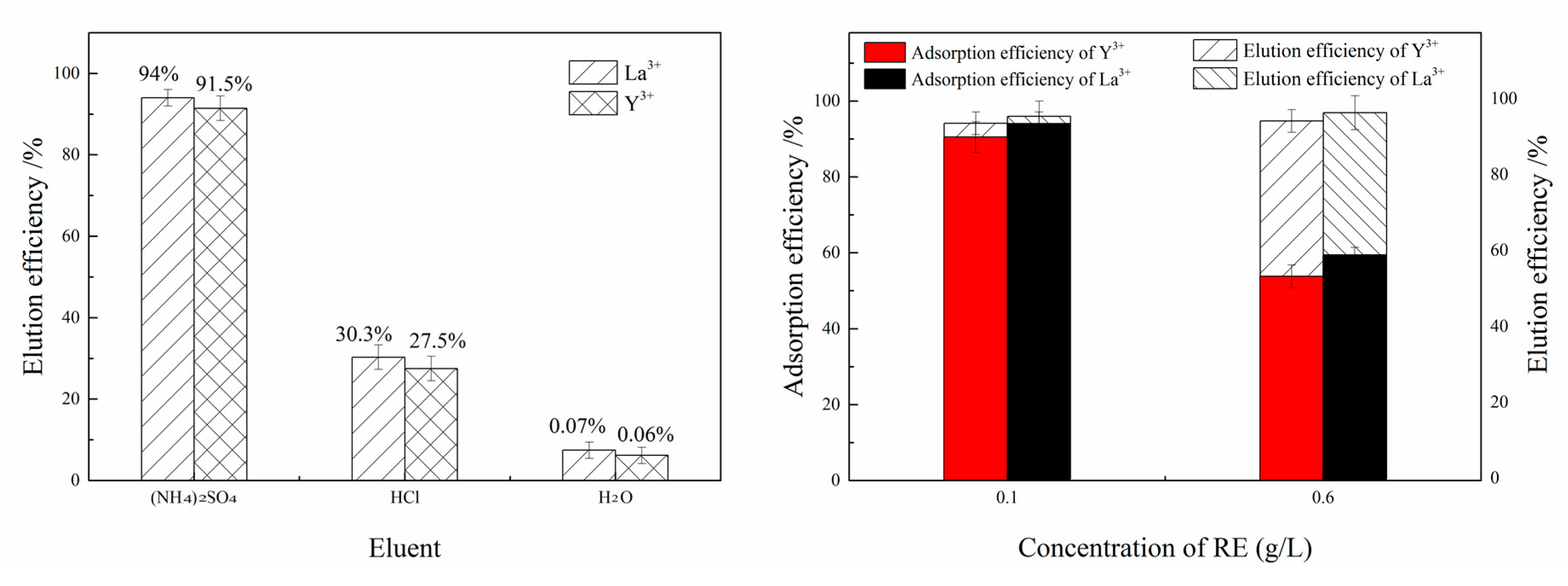
2.4.3. Elution Experiment
3. Results and Discussion
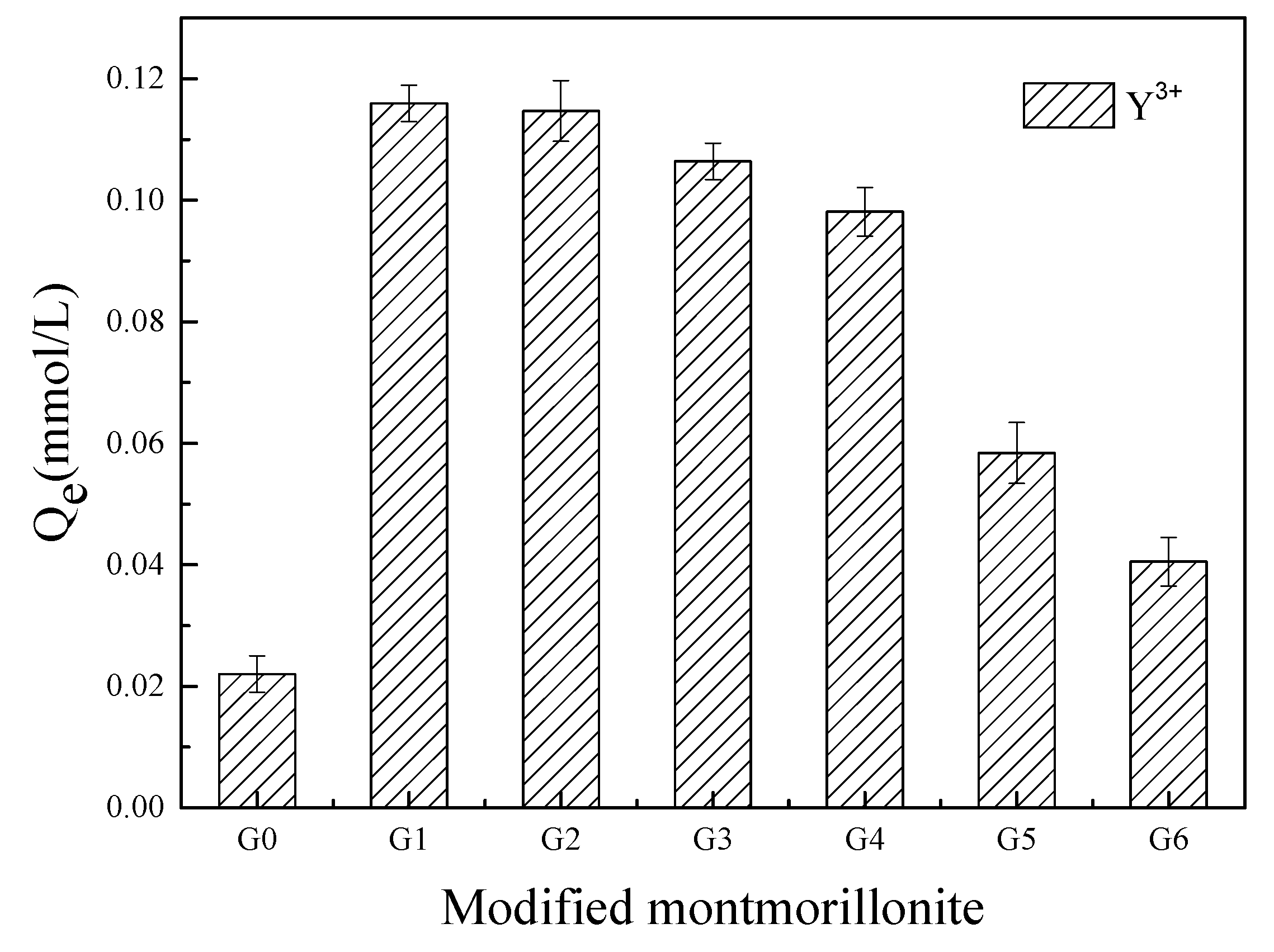
3.1. Selection of Modifier
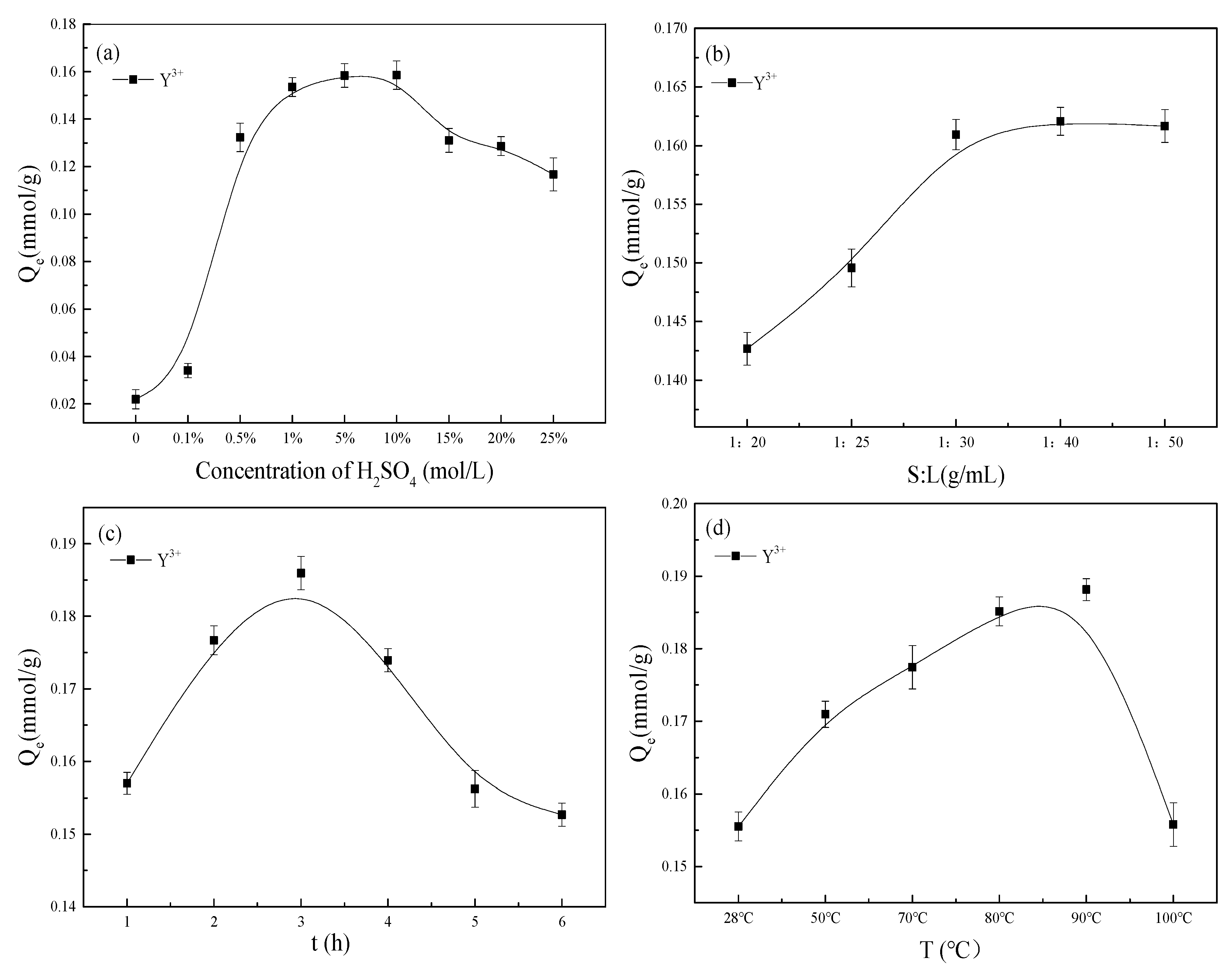
3.2. Effect of Reaction Conditions on the Preparation of Modified Mt
3.3. Characterization of Mt, Modified Mt, Modified Mt Adsorbed Y3+
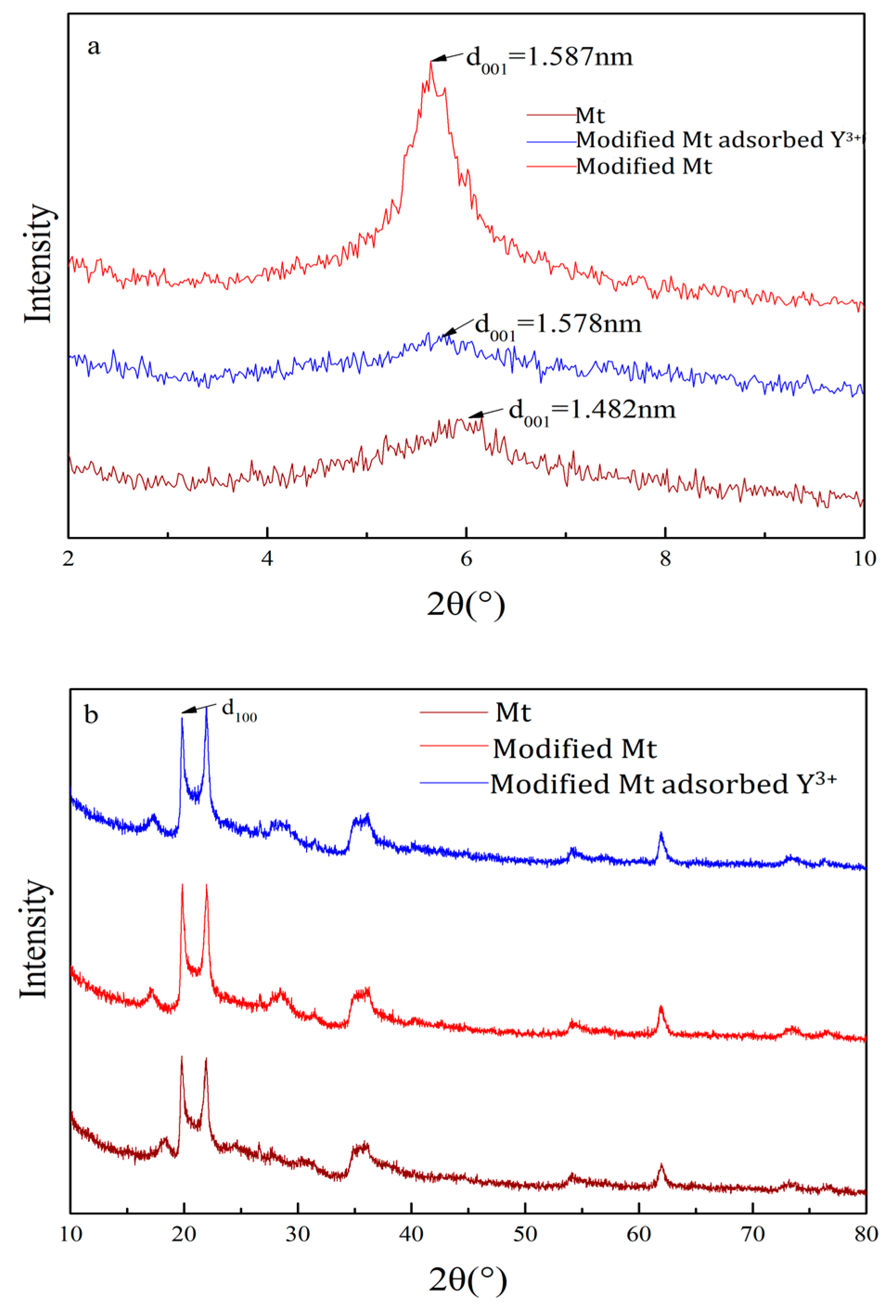
3.3.1. X-Ray Diffraction Analysis (XRD)
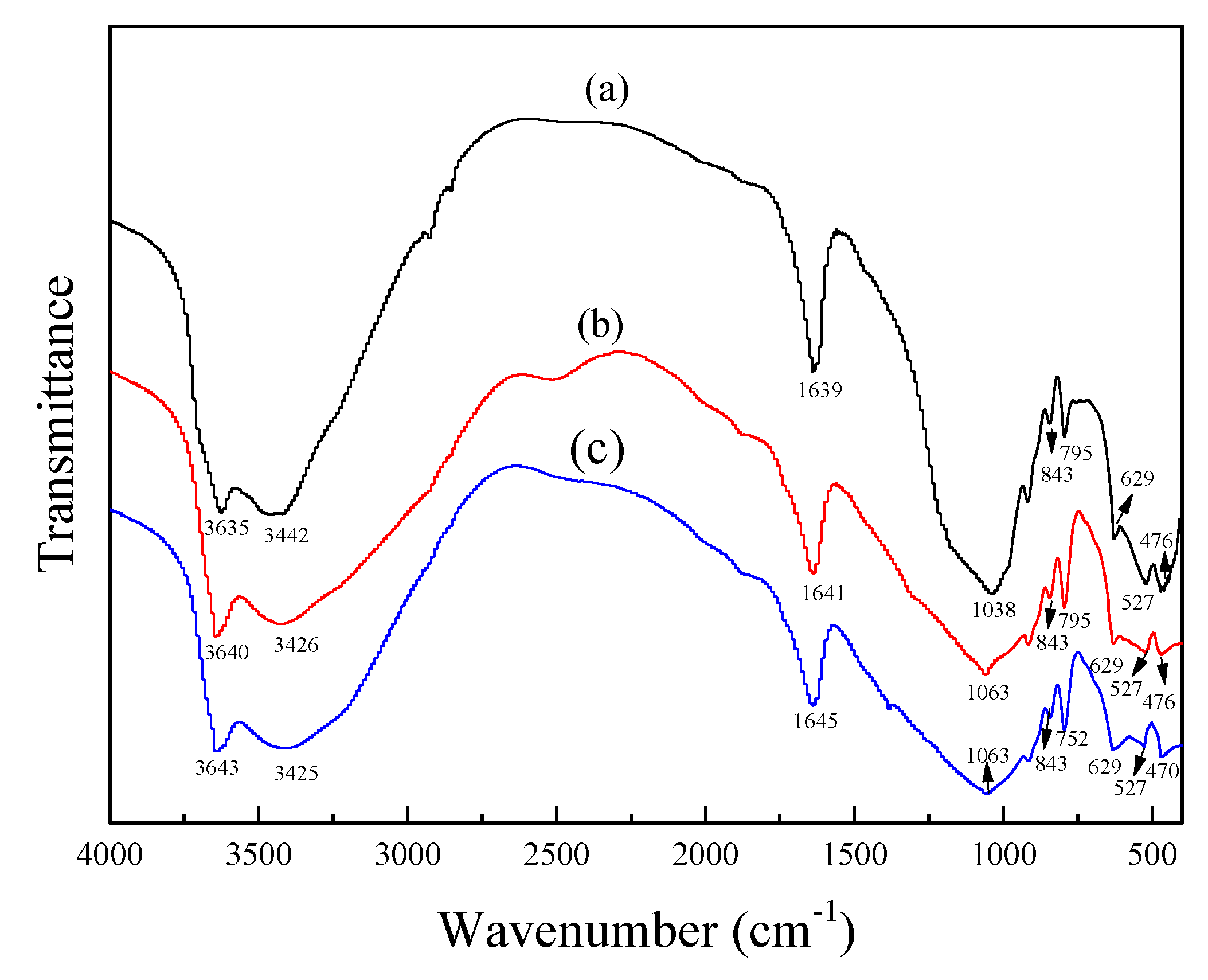
3.3.2. Fourier Transform Infrared Spectrometer (FT-IR)
3.3.3. Scanning Electron Microscopy (SEM)
3.3.4. Specific Surface Area and Pore Diameter Analysis (BET)
3.4. Application of Modified Mt to Recover Rare Earth from Solutions
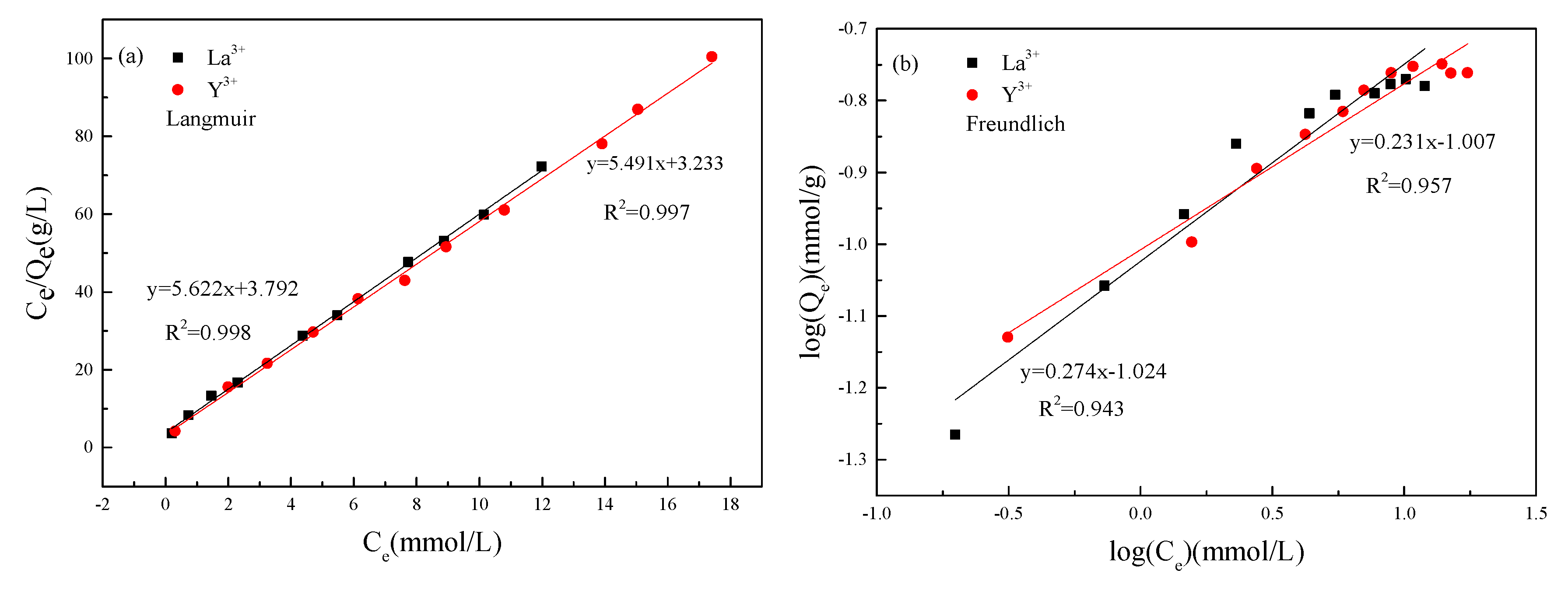
3.4.1. Isothermal Adsorption
3.4.2. Elution Experiment
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Chen, Z. Global Rare earth resources and scenarios of future rare earth industry. Rare-Earth Elem. 2011, 29, 1–6. [Google Scholar] [CrossRef]
- Yang, X.J.; Lin, A.; Li, X.L.; Wu, Y.; Zhou, W.; Chen, Z. China’s ion-adsorption rare earth resources, mining consequences and preservation. Environ. Dev. 2013, 8, 131–136. [Google Scholar] [CrossRef]
- Zhou, F.; Feng, J.; Wang, Z.; Xu, Y.; Zhang, Z.; Chi, R. One step purification of impurities in the leachate of weathered crust elution-deposited rere earth ores. Phys. Probl. Miner. Process. 2017, 53, 1188–1199. [Google Scholar]
- Feng, J.; Zhou, F.; Chi, R.; Liu, X.; Xu, Y.; Liu, Q. Effect of a novel compound on leaching process of weathered crust elution-deposited rare earth ore. Miner. Eng. 2018, 129, 63–70. [Google Scholar] [CrossRef]
- Zhou, F.; Feng, J.; Wu, B.H. Comprehensive chemical experiment of montmorillonite adsorbing rare earth ions. Exp. Technol. Manag. 2018, 35, 60–65. (In Chinese) [Google Scholar]
- Tian, J.; Yin, J.; Chi, R.; Rao, G.; Jiang, M.; Ouyang, K. Kinetics on leaching rare earth from the weathered crust elution-deposited rare earth ores with ammonium sulfate solution. Hydrometall 2010, 101, 166–170. [Google Scholar]
- He, Z.; Zhang, Z.; Yu, J.; Zhou, F.; Xu, Y.; Xu, Z.; Chi, R. Kinetics of column leaching of rare earth and aluminum from weathered crust elution-deposited rare earth ore with compound ammonium salt. Hydrometall 2016, 163, 33–39. (In Chinese) [Google Scholar] [CrossRef]
- Tannabe, K.; Anderson, J.R.; Boudar, M. (Eds.) Solid acid and base catalysis. In Catalysis-Science and Technology; Springer: New York, NY, USA, 1981; p. 231. [Google Scholar]
- Borisover, M.; Davis, J. A Chapter 2–Adsorption of Inorganic and Organic Solutes by Clay Minerals. Dev. Clay Sci. 2015, 33–70. [Google Scholar] [CrossRef]
- Zhu, R.; Chen, Q.; Zhou, Q.; Xi, Y.; Zhu, J.; He, H. Adsorbents based on montmorillonite for contaminant removal from water: A review. Appl. Clay Sci. 2016, 123, 239–258. [Google Scholar] [CrossRef]
- Chi, R.; Tian, J. (Eds.) Weathered Crust Elution-Deposited Rare Earth Ores; Nova Science Pub Inc.: New York, NY, USA, 2008. [Google Scholar]
- Lagaly, G. The “Layer Charge” of Regular Interstratified 2:1 Clay Minerals. Clays Clay Miner. 1979, 27, 1–10. [Google Scholar] [CrossRef]
- Golubeva, O.Y.; Ula’Yanova, N.Y.; Kostyreva, T.G.; Drozdova, I.A.; Mokeev, M.V. Synthetic nanoclays with the structure of montmorillonite: Preparation, structure, and physico-chemical properties. Galss Phys. Chem. 2013, 5, 533–539. [Google Scholar] [CrossRef]
- Natural Clay Minerals as Environmental Cleaning Agents; Ismadji, S.; Soetaredjo, F.E.; Ayucitra, A. Clay Materials for Environmental Remediation; Springer International Publishing: Berlin, Germany, 2015; pp. 5–37. [Google Scholar]
- Álvarez-Ayuso, E.; Garćia-Sánchez, A. Removal of Heavy Metals from Waste Waters by Natural and Na-exchanged Bentonites. Clays Clay Miner. 2003, 51, 475–480. [Google Scholar] [CrossRef]
- Larraza, I.; Mar, L.-G.; Corrales, T.; Marcelo, G. Hybrid materials: Magnetite–Polyethylenimine–Montmorillonite, as magnetic adsorbents for Cr(VI) water treatment. J. Colloid. Interf. Sci. 2012, 385, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Monvisade, P.; Siriphannon, P. Chitosan intercalated montmorillonite: Preparation, characterization and cationic dye adsorption. Appl. Clay Sci. 2009, 42, 427–431. [Google Scholar] [CrossRef]
- Vito IE, D.; Olsina, R.A.; Masi, A.N. Enrichment method for trace amounts of rare earth elements using chemofiltration and XRF determination. Fresenius J. Anal. Chem. 2000, 368, 392–396. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, F.; Li, L.; Zhang, K. Adsorption Rare Earth Metal Ions from Aqueous Solution by Polyamidoamine Dendrimer Functionalized Soy Hull. Waste Biomass Valorization 2016, 7, 1211–1219. [Google Scholar] [CrossRef]
- Hérès, X.; Blet, V.; Di Natale, P.; Ouaattou, A.; Mazouz, H.; Dhiba, D.; Cuer, F. Selective Extraction of Rare Earth Elements from Phosphoric Acid by Ion Exchange. Resins. Met. 2018, 8, 682. [Google Scholar] [CrossRef]
- Chen, L.; Dong, Y. Sorption of Ni(II) to montmorillonite as a function of pH, ionic strength, foreign ions and humic substances. J. Radioanal. Nucl. Chem. 2013, 295, 2117–2123. [Google Scholar] [CrossRef]
- Gu, S.; Kang, X.; Wang, L.; Lichtfouse, E.; Wang, C. Clay mineral adsorbents for heavy metal removal from wastewater: A review. Environ. Chem. Lett. 2019, 17, 629–654. [Google Scholar] [CrossRef]
- Han, H.; Rafiq, M.K.; Zhou, T.; Xu, R.; Mašek, O.; Li, X. A critical review of clay-based composites with enhanced adsorption performance for metal and organic pollutants. J. Hazard. Mater. 2019, 369, 780–796. [Google Scholar] [CrossRef]
- Zhang, H.; Kim, Y.K.; Hunter, T.N.; Brown, A.P.; Lee, J.W.; Harbottle, D. Organically modified clay with potassium copper hexacyanoferrate for enhanced Cs+ adsorption capacity and selective recovery by flotation. J. Mater. Chem. A 2017, 5, 15130–15143. [Google Scholar] [CrossRef]
- Miyamoto, N.; Kawai, R.; Kuroda, K.; Ogawa, M. Adsorption and aggregation of a cationic cyanine dye on layered clay minerals. Appl. Clay Sci. 2000, 16, 161–170. [Google Scholar] [CrossRef]
- Shawabkeh, R.A.; Tutunji, M.F. Experimental study and modeling of basic dye sorption by diatomaceous clay. Appl. Clay Sci. 2003, 24, 111–120. [Google Scholar] [CrossRef]
- He, Z.Y.; Zhang, Z.Y.; Yu, J.X.; Xu, Z.G.; Xu, Y.L.; Zhou, F.; Chi, R. Column leaching process of rare earth and alumioum from weathered crust elution-deposited rare earth ore with ammonium salts. Nonferrous Met. Soc. 2016, 26, 3024–3033. (In Chinese) [Google Scholar] [CrossRef]
- Homaeigohar, S.; Zillohu, A.; Abdelaziz, R.; Hedayati, M.; Elbahri, M. A Novel Nanohybrid Nanofibrous Adsorbent for Water Purification from Dye Pollutants. Materials 2016, 9, 848. [Google Scholar] [CrossRef]
- Yu, B.; Hu, Z.; Zhou, F.; Feng, J.; Chi, R. Lanthanum (III) and Yttrium (III) Adsorption on Montmorillonite: The Role of Aluminum Ion in Solution and Minerals. Miner. Process. Extr. Metall. Rev. 2019, 1–10. [Google Scholar] [CrossRef]
- Vashnia, S.; Tavakoli, H.; Cheraghali, R.; Sepehrian, H. Supporting of Lead Hexacyanoferrate on Mesoporous MCM-41 and its use as Effective Adsorbent for Strontium: Equilibrium, Kinetic, and Thermodynamic Studies. Sep. Sci. Technol. 2014, 49, 241–248. [Google Scholar] [CrossRef]
- Jiang, D.; Lixiong, S.; Deman, L.; Yong, Q.; Wenfeng, T.; Liang, W. Removal of Phosphorus from Leach Liquor of Steel Slag: Adsorption Dephosphorization with Activated Alumina. JOM 2018, 70, 2027–2032. [Google Scholar]
- Homaeigohar, S.; Botcha, N.K.; Zarie, E.S.; Elbahri, M. Ups and Downs of Water Photodecolorization by Nanocomposite Polymer Nanofibers. Nanomaterials 2019, 9, 250. [Google Scholar] [CrossRef]
- Barouni, K.; Bazzi, L.; Salghi, R.; Mihit, M.; Hammouti, B.; Albourine, A.; El Issami, S. Some amino acids as corrosion inhibitors for copper in nitric acid solution. Mater. Lett. 2008, 62, 3325–3327. [Google Scholar] [CrossRef]
- Parolo, M.E.; Pettinari, G.R.; Musso, T.B.; Sánchez-Izquierdo, M.P.; Fernández, L.G. Characterization of organo-modified bentonite sorbents: The effect ofmodification conditions on adsorption performance. Appl. Surf. Sci. 2014, 320, 356–363. [Google Scholar] [CrossRef]
- Kumrić, K.R.; Đukić, A.B.; Trtić-Petrović, T.M.; Vukelić, N.S.; Stojanović, Z.; Grbović Novaković, J.D.; Matović, L.L. Simultaneous Removal of Divalent Heavy Metals from Aqueous Solutions Using Raw and Mechanochemically Treated Interstratified Montmorillonite/Kaolinite Clay. Ind. Eng. Chem. Res. 2013, 52, 7930–7939. [Google Scholar] [CrossRef]
- González-Rodríguez, B.; Trujillano, R.; Rives, V.; Vicente, M.A.; Gil, A.; Korili, S.A. Structural, textural and acidic properties of Cu-, Fe- and Cr-doped Ti-pillared montmorillonites. Appl. Clay Sci. 2015, 118, 124–130. [Google Scholar] [CrossRef]
- Eren, E.; Afsin, B. An investigation of Cu (II) adsorption by raw and acid-activated bentonite: A combined potentiometric, thermodynamic, XRD, IR, DTA study. J. Hazard. Mater 2008, 151, 682–691. [Google Scholar] [CrossRef]
- Önal, M.; Sarıkaya, Y. Preparation and characterization of acid-activated bentonite powders. Powder Technol. 2007, 172, 14–18. [Google Scholar] [CrossRef]
- Steudel, A.; Batenburg, L.F.; Fischer, H.R.; Weidler, P.G.; Emmerich, K. Alteration of swelling clay minerals by acid activation. Appl. Clay Sci. 2009, 44, 100–115. [Google Scholar] [CrossRef]
- Chen, H.H.; Thirumavalavan, M.; Ma, Y.-J.; Lee, J.F. The influence of structural and processing parameters of modifiers on the interlayer structure of modified montmorillonite. RSC Adv. 2015, 5, 83217–83224. [Google Scholar] [CrossRef]
- Wang, H.; Wu, T.; Chen, J.; Zheng, Q.; He, C.; Zhao, Y. Sorption of Se (IV) on Fe-and Al-modified bentonite. Radioanal. Nucl. Chem. 2015, 303, 107–113. [Google Scholar] [CrossRef]
- Wang, L.; Wang, A. Adsorption properties of Congo Red from aqueous solution onto surfactant-modified montmorillonite. J. Hazard. Mater 2008, 160, 173–180. [Google Scholar] [CrossRef]
- Alshameri, A.; He, H.; Zhu, J.; Xi, Y.; Zhu, R.; Ma, L.; Tao, Q. Adsorption of ammonium by different natural clay minerals: Characterization, kinetics and adsorption isotherms. Appl. Clay Sci. 2018, 159, 83–93. [Google Scholar] [CrossRef]
- Xiao, Y.; Huang, L.; Long, Z.; Feng, Z.; Wang, L. Adsorption ability of rare earth elements on clay minerals and its practical performance. J. Rare Earth 2016, 34, 543–548. [Google Scholar] [CrossRef]
- Gupta, V.K.; Ali, I.; Saini, V.K. Adsorption studies on the removal of Vertigo Blue 49 and Orange DNA13 from aqueous solutions using carbon slurry developed from a waste material. J. Colloid Interface Sci. 2007, 315, 87–93. [Google Scholar] [CrossRef]

| Modified Mt | Modifier | Volume (mL)/Mass (g) | Temperature (°C) | Time (h) | |
|---|---|---|---|---|---|
| Acid-Mt | G0 | - | - | - | - |
| G1 | HCl (1:1) + H2O4 (1:4) | 40 mL + 40 mL | 90 | 4 | |
| G2 | HCl (1:1) | 20 mL | room temperature | 2 | |
| G3 | H2SO4 (1:4) | 80 mL | 90 | 4 | |
| G4 | HCl (1:10) | 20 mL | room temperature | 2 | |
| Organo-Mt | G5 | HDTMA + DDTC (mole ratio is 1:1) | 0.413 g | 70 room temperature | 2 |
| 24 | |||||
| G6 | HDTMA | 0.510 g | 70 | 2 | |
| Samples | Specific Surface Area (m2/g) | Pore Volume (cm3/g) | Pore Size (nm) |
|---|---|---|---|
| Mt | 37.198 | 0.109 | 11.689 |
| Modified Mt | 107.074 | 0.227 | 8.479 |
| Modified Mt adsorbed Y3+ | 82.947 | 0.223 | 10.764 |
| Rare Earth Iron | Langmuir Model Parameters | Freundlich Model Parameters | ||||
|---|---|---|---|---|---|---|
| Qmax (mmol/g) | KL (L/mol) | R2 | 1/n | KF (L/mol) | R2 | |
| Y3+ | 0.182 | 1.718 | 0.997 | 0.231 | 0.098 | 0.957 |
| La3+ | 0.178 | 1.482 | 0.998 | 0.274 | 0.095 | 0.943 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, X.; Zhou, F.; Chi, R.; Feng, J.; Ding, Y.; Liu, Q. Preparation of Modified Montmorillonite and Its Application to Rare Earth Adsorption. Minerals 2019, 9, 747. https://doi.org/10.3390/min9120747
Liu X, Zhou F, Chi R, Feng J, Ding Y, Liu Q. Preparation of Modified Montmorillonite and Its Application to Rare Earth Adsorption. Minerals. 2019; 9(12):747. https://doi.org/10.3390/min9120747
Chicago/Turabian StyleLiu, Xu, Fang Zhou, Ruan Chi, Jian Feng, Yinying Ding, and Qi Liu. 2019. "Preparation of Modified Montmorillonite and Its Application to Rare Earth Adsorption" Minerals 9, no. 12: 747. https://doi.org/10.3390/min9120747
APA StyleLiu, X., Zhou, F., Chi, R., Feng, J., Ding, Y., & Liu, Q. (2019). Preparation of Modified Montmorillonite and Its Application to Rare Earth Adsorption. Minerals, 9(12), 747. https://doi.org/10.3390/min9120747




