Distribution and Chemical Speciation of Molybdenum in River and Pond Sediments Affected by Mining Activity in Erdenet City, Mongolia
Abstract
1. Introduction
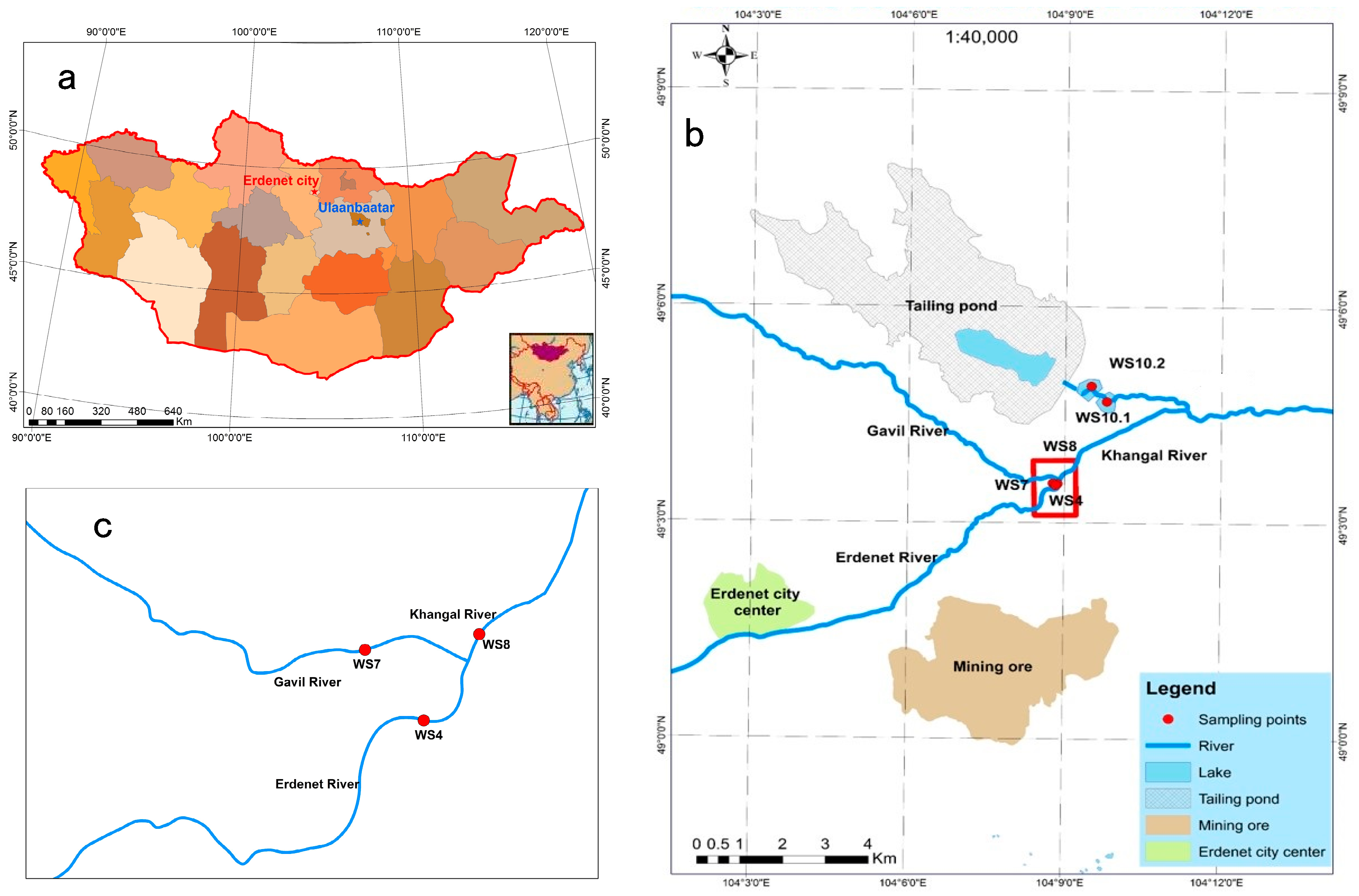
2. Studied Area
3. Materials and Methods
3.1. Sampling Sites
3.2. Water Analysis
3.3. Sediment Analysis
3.3.1. Sequential Extraction
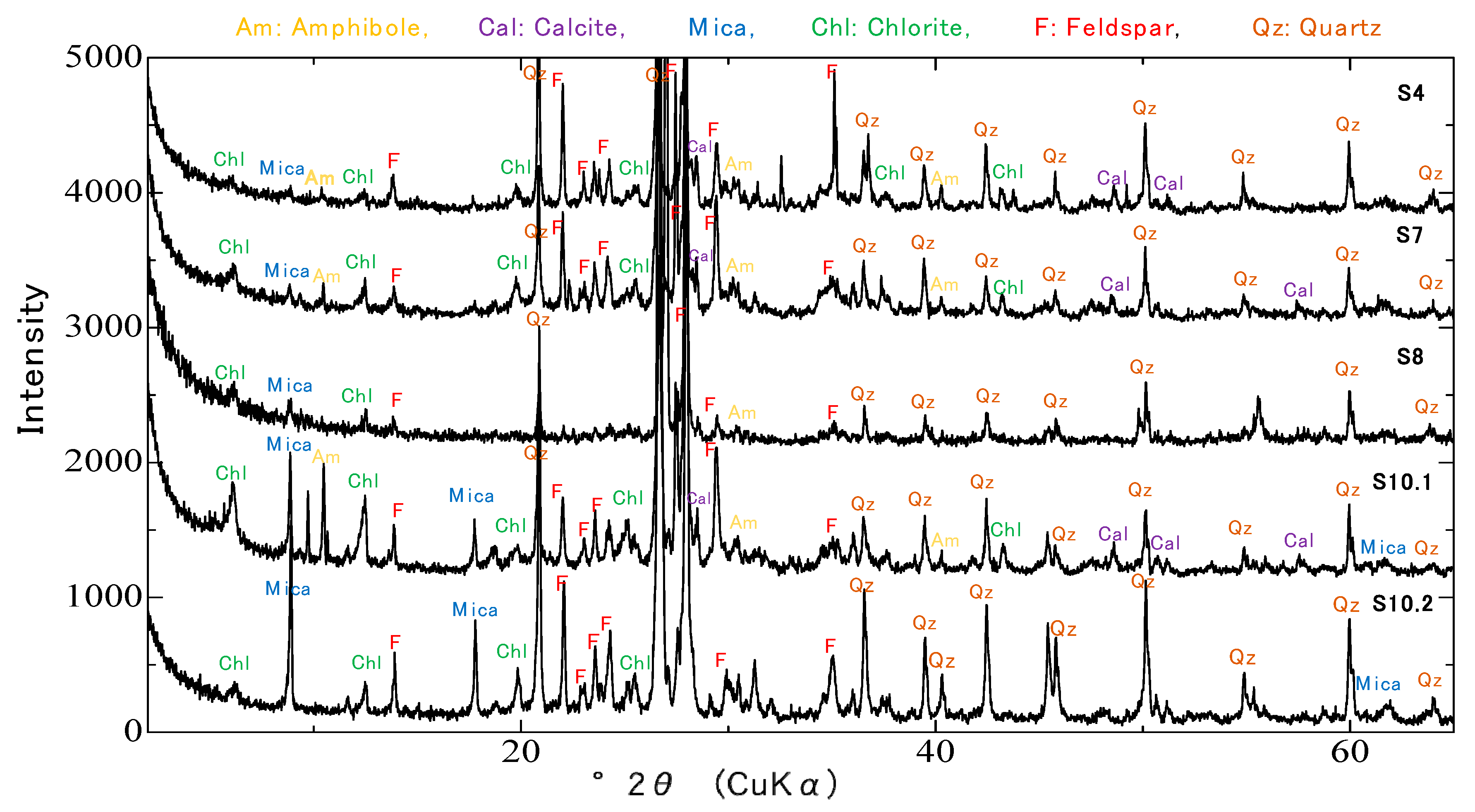
3.3.2. X-ray Diffraction
3.3.3. X-ray Absorption Fine Structure
4. Results
4.1. Water Chemistry
4.2. XRD
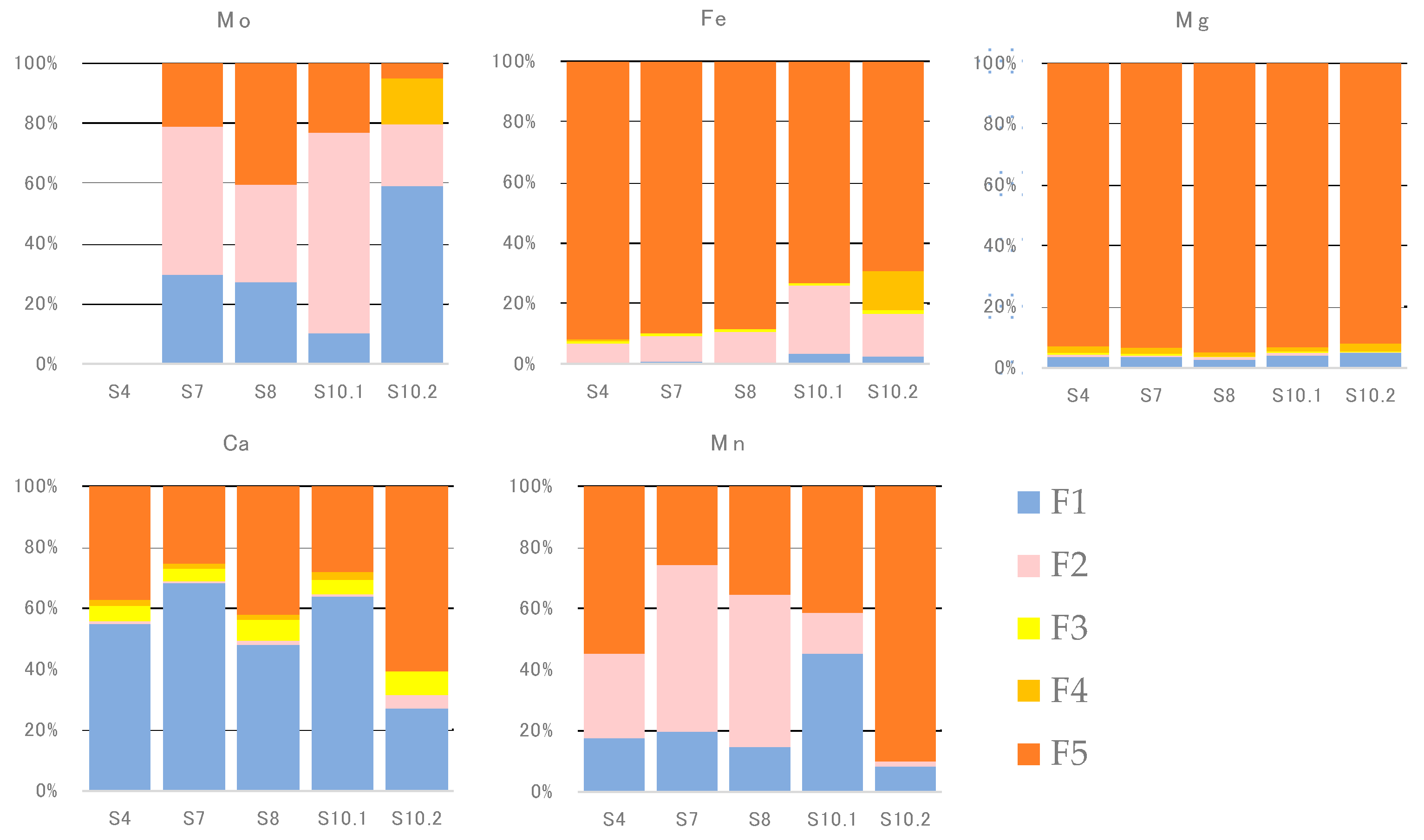
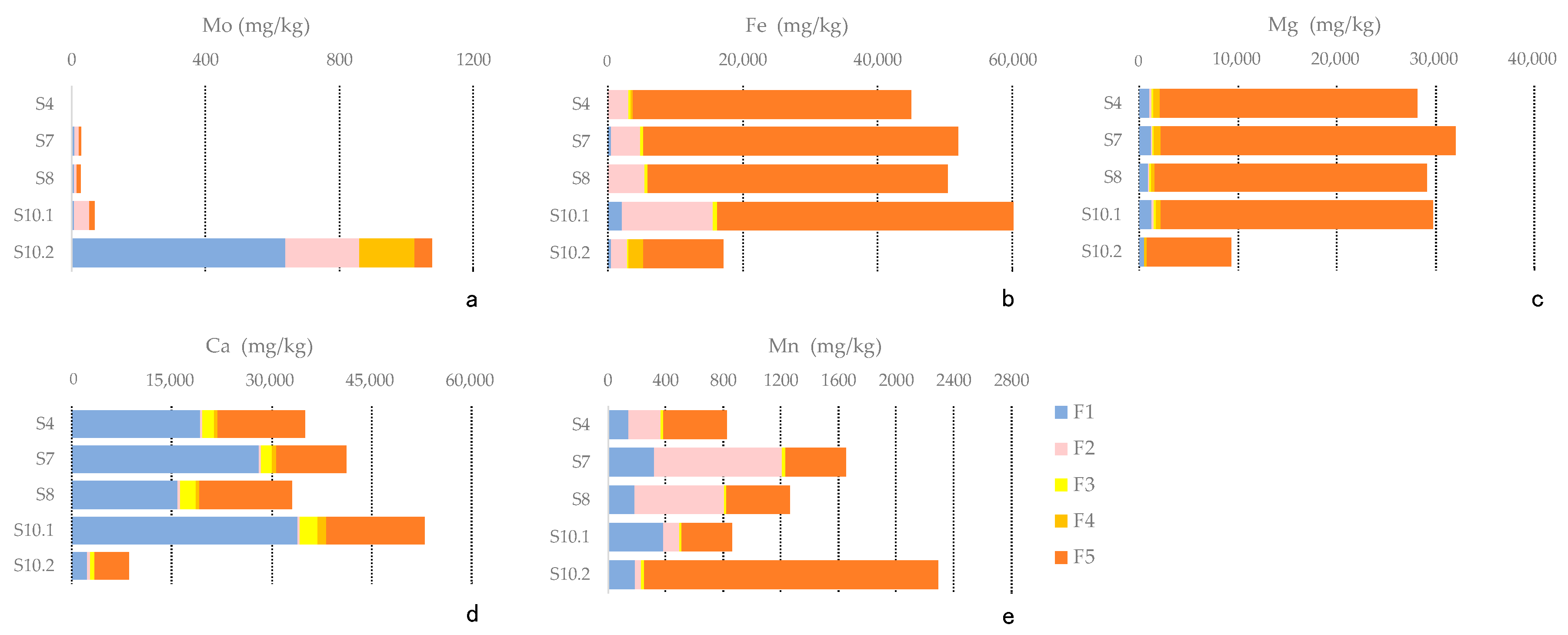
4.3. Sequential Extraction
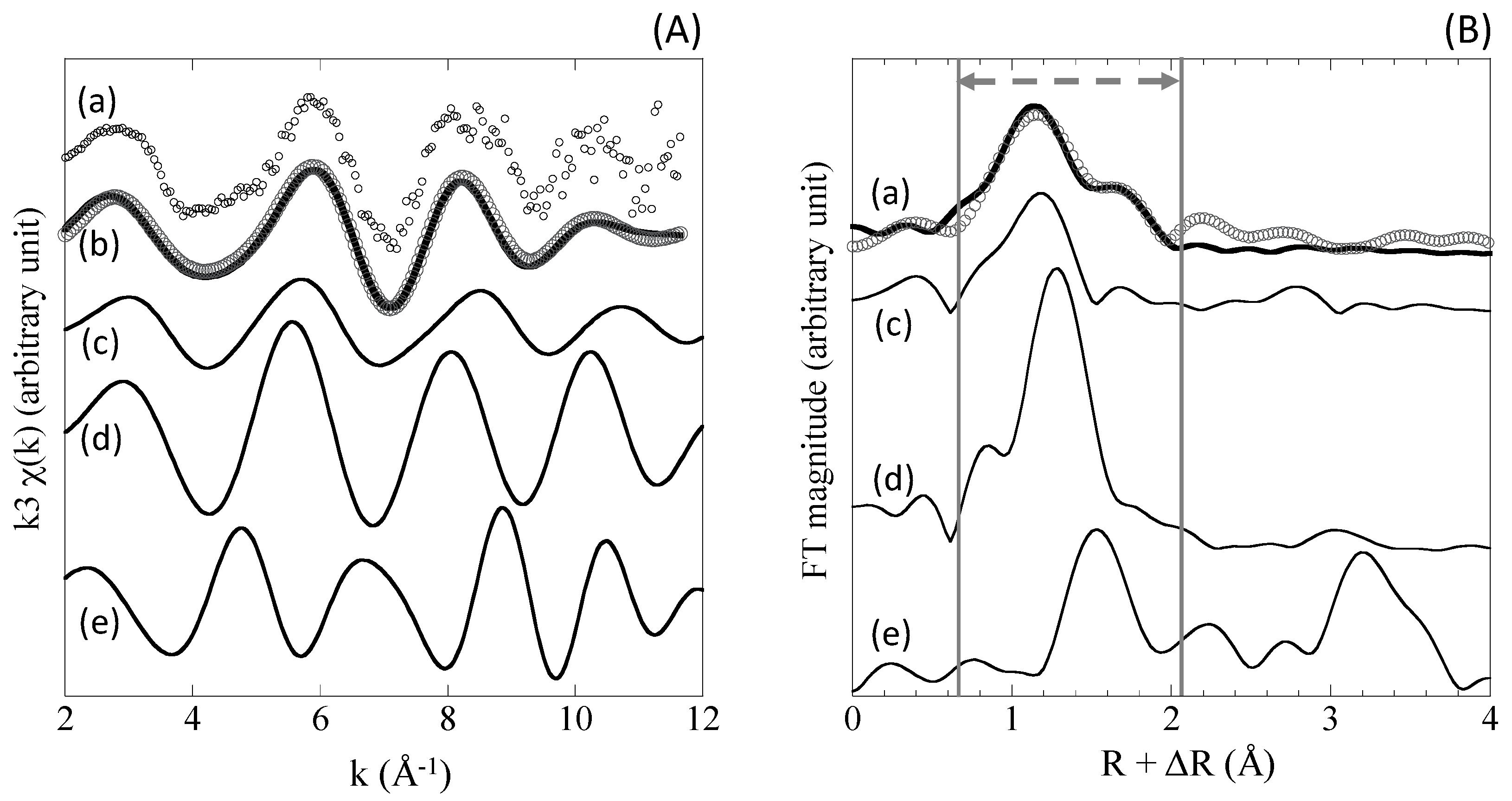
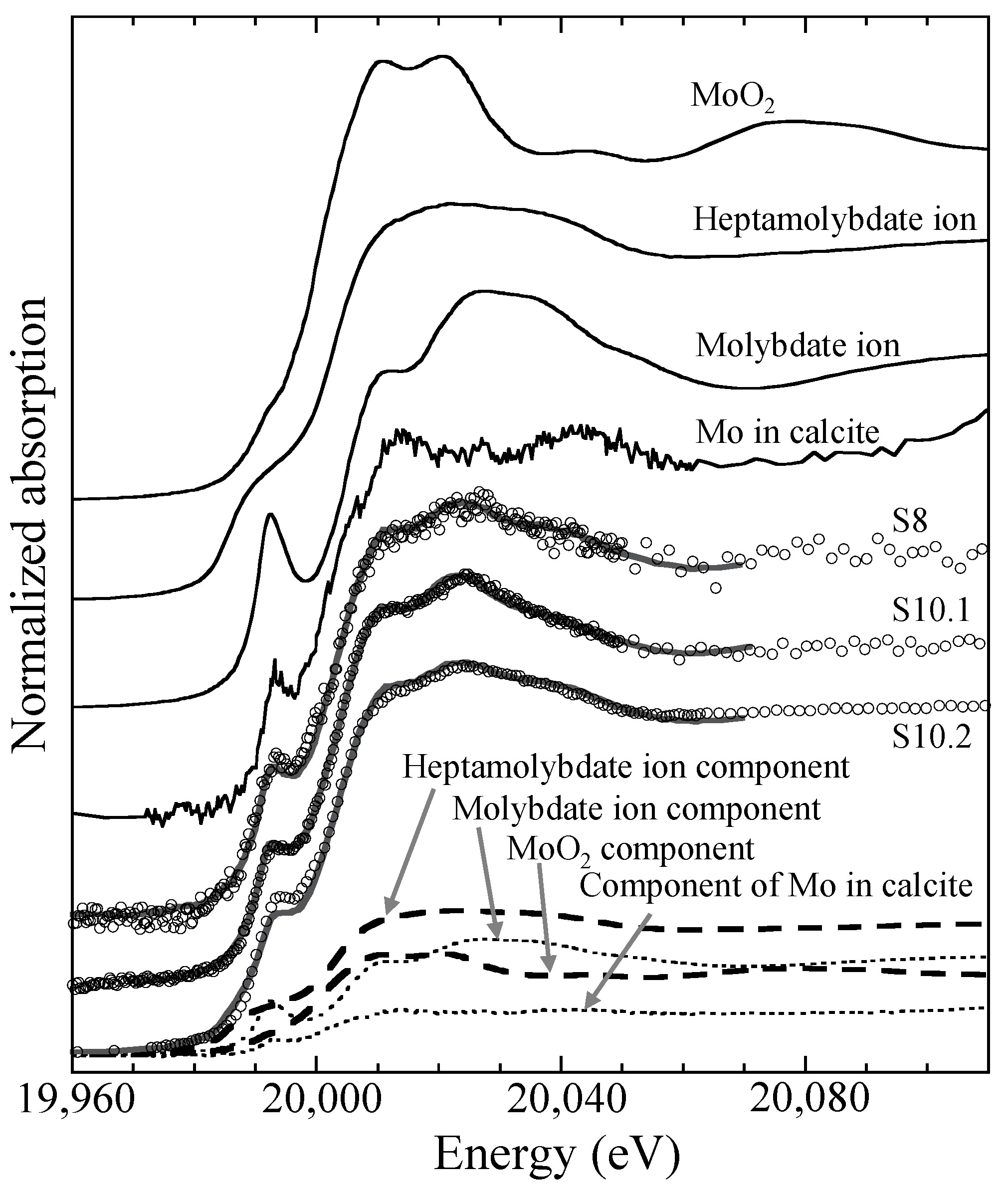
4.4. XAFS
5. Discussion
5.1. Characteristics of Water Chemistry in the Ponds and Rivers
5.2. Chemical Speciation of Mo in Sediments
5.3. Solubility and Mobility of Mo in Erdenet Area
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Duplication 1 | Duplication 2 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fraction | S4 | S7 | S8 | S10.1 | S10.2 | Fraction | S4 | S7 | S8 | S10.1 | S10.2 | |
| Mo mg/kg | F1 | <4.0 | 8.6 | 7.2 | 6.9 | 630 | F1 | <4.0 | 8.6 | <4.0 | 7.0 | 640 |
| F2 | <4.0 | 14 | 9.5 | 44 | 220 | F2 | <4.0 | 14 | 7.7 | 47 | 230 | |
| F3 | <4.0 | <4.0 | <4.0 | <4.0 | <4.0 | F3 | <4.0 | <4.0 | <4.0 | <4.0 | <4.0 | |
| F4 | <4.0 | <4.0 | <4.0 | <4.0 | 160 | F4 | <4.0 | <4.0 | <4.0 | <4.0 | 170 | |
| F5 | <4.0 | 6.4 | 12 | 16 | 53 | F5 | <4.0 | 5.8 | 10 | 15 | 54 | |
| Total 1 | 0 | 29 | 28 | 67 | 1100 | Total | 0 | 29 | 18 | 69 | 1100 | |
| Fe mg/kg | F1 | <200 | 490 | <200 | 2200 | 590 | F1 | <200 | 510 | <200 | 2100 | 640 |
| F2 | 3600 | 4300 | 4800 | 13,000 | 2400 | F2 | 2600 | 4400 | 6200 | 14,000 | 2400 | |
| F3 | 330 | 420 | 350 | 610 | 290 | F3 | 330 | 390 | 390 | 620 | 310 | |
| F4 | 370 | <200 | <200 | <200 | 2500 | F4 | 320 | <200 | <200 | <200 | 1900 | |
| F5 | 43,000 | 47,000 | 47,000 | 48,000 | 12,000 | F5 | 39,000 | 46,000 | 42,000 | 41,000 | 11,000 | |
| Total 1 | 48,000 | 52,000 | 52,000 | 63,000 | 18,000 | Total | 42,000 | 52,000 | 49,000 | 57,000 | 17,000 | |
| Mg mg/kg | F1 | 1000 | 1200 | 810 | 1300 | 460 | F1 | 970 | 1100 | 850 | 1200 | 450 |
| F2 | 210 | 170 | 190 | 210 | 38 | F2 | 210 | 180 | 170 | 230 | 34 | |
| F3 | 190 | 140 | 140 | 150 | 27 | F3 | 180 | 130 | 110 | 170 | 29 | |
| F4 | 740 | 700 | 450 | 450 | 230 | F4 | 520 | 650 | 340 | 460 | 200 | |
| F5 | 13,000 | 13,000 | 13,000 | 15,000 | 5700 | F5 | 12000 | 13,000 | 11,000 | 41,000 | 11,000 | |
| Total 1 | 15,000 | 16,000 | 15,000 | 17,000 | 6400 | Total | 14,000 | 15,000 | 12,000 | 14,000 | 6100 | |
| Ca mg/kg | F1 | 19,000 | 28,000 | 14,000 | 33,000 | 2200 | F1 | 20,000 | 29,000 | 18,000 | 35,000 | 2400 |
| F2 | 370 | 350 | 510 | 390 | 420 | F2 | 300 | 270 | 360 | 390 | 380 | |
| F3 | 1900 | 1800 | 2000 | 2700 | 760 | F3 | 1700 | 1500 | 2700 | 2400 | 540 | |
| F4 | 750 | 760 | 670 | 1300 | <30 | F4 | 450 | 580 | 440 | 1400 | <30 | |
| F5 | 14,000 | 11,000 | 16,000 | 17,000 | 6400 | F5 | 12,000 | 10,000 | 11,000 | 13,000 | 4000 | |
| Total 1 | 36,000 | 41,000 | 33,000 | 54,000 | 9800 | Total | 34,000 | 41,000 | 33,000 | 52,000 | 7300 | |
| Mn mg/kg | F1 | 150 | 330 | 160 | 390 | 190 | F1 | 140 | 320 | 210 | 380 | 190 |
| F2 | 210 | 880 | 380 | 110 | 37 | F2 | 240 | 890 | 860 | 120 | 41 | |
| F3 | <30 | <30 | <30 | <30 | <30 | F3 | <30 | <30 | <30 | <30 | <30 | |
| F4 | <30 | <30 | <30 | <30 | <30 | F4 | <30 | <30 | <30 | <30 | <30 | |
| F5 | 480 | 430 | 520 | 400 | 2200 | F5 | 410 | 410 | 360 | 300 | 1900 | |
| Total 1 | 840 | 1600 | 1100 | 890 | 2400 | Total | 786 | 1600 | 1400 | 810 | 2100 | |
References
- España, J.S.; Pamo, E.L.; Santofimia, E.; Aduvire, O.; Reyes, J.; Barettino, D. Acid mine drainage in the Iberian Pyrite Belt (Odiel river watershed, Huelva, SW Spain): Geochemistry, mineralogy and environmental implications. Appl. Geochem. 2005, 20, 1320–1356. [Google Scholar] [CrossRef]
- Fukushi, K.; Sasaki, M.; Sato, T.; Yanase, N.; Amano, H.; Ikeda, H. A natural attenuation of arsenic in drainage from an abandoned arsenic mine dump. Appl. Geochem. 2003, 18, 1267–1278. [Google Scholar] [CrossRef]
- Shevenell, L.; Connors, K.A.; Henry, C.D. Controls on pit lake water quality at sixteen open-pit mines in Nevada. Appl. Geochem. 1999, 14, 669–687. [Google Scholar] [CrossRef]
- World Bank. A Review of Environmental and Social Impacts in the Mining Sector; World Bank: Washington, DC, USA, 2006; p. 44. [Google Scholar]
- Batsaikhan, B.; Kwon, J.S.; Kim, K.H.; Lee, Y.J.; Badarch, M.; Yun, S.T. Hydrochemical evaluation of the influences of mining activities on river water chemistry in central northern Mongolia. Environ. Sci. Pollut. Res. 2017, 24, 2019–2034. [Google Scholar] [CrossRef] [PubMed]
- Battogtokh, B.; Lee, J.M.; Woo, N. Contamination of water and soil by the Erdenet copper-molybdenum mine in Mongolia. Environ. Earth Sci. 2014, 71, 3363–3374. [Google Scholar] [CrossRef]
- Sotnikov, V.I.; Ponomarchuk, V.A.; Shevchenko, D.O.; Berzina, A.P. The Erdenetiyn-Ovoo porphyry Cu-Mo deposit, Northern Mongolia: 40Ar/39Ar geochronology and factors of large-scale mineralization. Geol. Geofiz. 2005, 46, 633–644. [Google Scholar]
- Drever, J.I. The Geochemistry of Natural Waters: Surface and Groundwater, 3rd ed.; Prentice Hall: Upper Saddle River, NJ, USA, 1997. [Google Scholar]
- Kisker, C.; Schindelin, H.; Rees, D.C. Molybdenum-cofactor-containing enzymes: Structure and mechanism. Ann. Rev. 1997, 66, 233–267. [Google Scholar]
- WHO. Guidlines for Drinking Water Quality; WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Smedley, P.L.; Kinniburgh, D.G. Molybdenum in natural waters: A review of occurrence, distributions and controls. Appl. Geochem. 2017, 84, 387–432. [Google Scholar] [CrossRef]
- Franscoli, F.; Hudson-Edwards, A. Geochemistry, mineralogy and microbiology of molybdenum in mining-affected environments. Minerals 2018, 8, 42. [Google Scholar] [CrossRef]
- España, J.S.; Toril, E.G.; Pamo, E.L.; Amils, R.; Ercilla, M.D.; Pastor, E.S.; San Martín-Úriz, P. Biogeochemistry of a hyperacidic and ultraconcentrated pyrite leachate in San Telmo mine (Iberian Pyrite Belt, Spain). Water Air Soil Pollut. 2008, 194, 243–257. [Google Scholar] [CrossRef]
- Skierszkan, E.K.; Mayer, K.U.; Weis, D.; Beckie, R.D. Molybdenum and zinc stable isotope variation in mining waste rock drainage and waste rock at the Antamina mine, Peru. Sci. Total Environ. 2016, 550, 103–113. [Google Scholar] [CrossRef] [PubMed]
- Smuda, J.; Dold, B.; Spangenberg, J.E.; Pfeifer, H.-R. Geochemistry and stable isotope composition of fresh alkaline porphyry copper tailings: Implications on sources and mobility of elements during transport and early stages of deposition. Chem. Geol. 2008, 256, 62–76. [Google Scholar] [CrossRef]
- Alakangas, L.; Öhander, B.; Lundberg, A. Estimation of temporal changes in oxidation rates of sulphides in copper mine tailings at Laver, Northern Sweden. Sci. Total Environ. 2010, 408, 1386–1392. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Sillanpää, M.; Gjessing, E.T.; Peräniemi, S.; Vogt, R.D. Environmental impact of mining activities on the surface water quality in Tibet: Gyama valley. Sci. Total Environ. 2010, 408, 4177–4184. [Google Scholar] [CrossRef] [PubMed]
- Khorasanipour, M.; Tangestani, M.H.; Naseh, R.; Majmohammadi, H. Hydrochemistry, mineralogy and chemical fractionation of mine and processing wastes associated with porphyry copper mines: A case study from the Sarcheshmeh mine, Iran. Appl. Geochem. 2011, 26, 714–730. [Google Scholar] [CrossRef]
- Bilali, L.E.; Rasmussen, R.E.; Hall, G.E.M.; Fortin, D. Role of sediment composition in trace metal distribution in lake sediments. Appl. Geochem. 2002, 17, 1171–1181. [Google Scholar] [CrossRef]
- Clemente, R.; Dickinson, N.M.; Lepp, N.W. Mobility of metals and metalloids in a multi-element contaminated soil 20 years after cessation of the pollution source activity. Environ. Pollut. 2008, 155, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Appelo, C.A.J.; Postma, D. Geochemistry, Groundwater and Pollution, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Brown, G.E.; Foster, A.L.; Ostergren, J.D. Mineral surfaces and bioavailability of heavy metals: A molecular-scale perspective. In Geology, Mineralogy, and Human Welfare; National Academy of Sciences colloquium, Arnold and Mabel Beckman Center: Irvine, CA, USA, 1999. [Google Scholar]
- Takahashi, Y.; Fan, Q.; Suga, H.; Tanaka, K.; Sakaguchi, A.; Takeichi, Y.; Ono, K.; Mase, K.; Kato, K.; Kanivets, V.V. Comparison of solid-water partitions of radiocesium in river waters in Fukushima and Chernobyl Areas. Sci. Rep. 2017, 7, 12407. [Google Scholar] [CrossRef] [PubMed]
- Harada, T.; Takahashia, Y. Origin of the difference in the distribution behavior of tellurium and selenium in a soil–water system. Geochim. Cosmochim. Acta 2008, 72, 1281–1294. [Google Scholar] [CrossRef]
- Gomes, A.F.S.; Lopez, D.L.; Ladeira, A.C.Q. Characterization and assessment of chemical modifications of metal-bearing sludges arising from unsuitable disposal. J. Hazard. Mater. 2012, 199–200, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Yanase, N.; Nightingale, T.; Payne, T.; Duerden, P. Uranium distribution in mineral phases of rock by sequential extraction procedure. Radiochim. Acta 1991, 52–53, 387–393. [Google Scholar] [CrossRef]
- Žemberyová, B.; Barteková, J.; Hagarová, I. The utilization of modified BCR three-step sequential extraction procedure for the fractionation of Cd, Cr, Cu, Ni, Pb and Zn in soil reference materials of different origins. Talanta 2006, 70, 973–978. [Google Scholar] [CrossRef] [PubMed]
- Favas, P.J.C.; Pratas, J.; Gomes, M.E.P.; Cala, V. Selective chemical extraction of heavy metals in tailings and soils contaminated by mining activity: Environmental implications. J. Geochem. Explor. 2011, 111, 160–171. [Google Scholar] [CrossRef]
- Qin, H.B.; Takeichi, Y.; Nitani, H.; Terada, Y.; Takahashi, Y. Tellurium distribution and speciation in contaminated soils from abandoned mine tailings: Comparison with selenium. Environ. Sci. Technol. 2017, 51, 6027–6035. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, P.E.R.; Hayes, J.R.; Grosvenor, A.P. Investigating the geochemical model for molybdenum mineraliation in the JEB tailings management facility at McClean Lake, Saskatchewan: An X-ray absorption spectroscopy study. Environ. Sci. Technol. 2015, 49, 6504–6509. [Google Scholar] [CrossRef] [PubMed]
- Kavalieris, I.; Khashgerel, B.E.; Morgan, L.E.; Undrakhtamir, A.; Borohul, A. Characteristics and 40Ar/39Ar geochronology of the Erdenet Cu-Mo deposit, Mongolia. Econ. Geol. 2017, 112, 1033–1053. [Google Scholar] [CrossRef]
- Kashiwabara, T.; Takahashi, Y.; Tanimizu, M.; Usui, A. Molecular-scale mechanisms of distribution and isotopic fractionation of molybdenum between seawater and ferromanganese oxides. Geochim. Cosmochim. Acta 2011, 75, 5762–5784. [Google Scholar] [CrossRef]
- Schwertmann, U.; Cornell, R.M. Iron Oxides in the Laboratory: Preparation and Characterization; VCH: Vancouver, BC, Canada, 1991. [Google Scholar]
- Rodríguez, L.; Ruiz, E.; Alonso-Azcárate, J.; Rincón, J. Heavy metal distribution and chemical speciation in tailings and soils around a Pb-Zn mine in Spain. J. Environ. Manag. 2009, 90, 1106–1116. [Google Scholar] [CrossRef] [PubMed]
- Zabinsky, S.I.; Rehr, J.J.; Ankudinov, A.; Albers, R.C.; Eller, M. Multiple-scattering calculations of X-ray-absorption spectra. Phys. Rev. B 1995, 52, 2995–3009. [Google Scholar] [CrossRef]
- Bailey, S.W. Classification and structures of the micas. Rev. Mineral. Geochem. 1984, 13, 1–12. [Google Scholar]
- Bailey, S.W. Chlorites: Structures and crystal chemistry: In hydrous phyllosilicates (Exclusive of Micas). Rev. Mineral. 1988, 19, 347–403. [Google Scholar]
- Cornell, M.; Schwertmann, U. The Iron Oxides: Structure, Properties, Reactions, Occurences and Uses, 2nd ed.; VCH: Vancouver, BC, Canada, 2004. [Google Scholar]
- Busenberg, E.; Plummer, L.N. Thermodynamics of magnesian calcite solid-solutions at 25 °C and 1 atm total pressure. Geochim. Cosmochim. Acta 1989, 53, 1189–1208. [Google Scholar] [CrossRef]
- O’Day, P.A.; Rehr, J.J.; Zabinsky, S.I.; Brown, G.E.J. Extended X-ray absorption fine structure (EXAFS) analysis of disorder and multiple-scattering in complex crystalline solids. J. Am. Chem. Soc. 1994, 116, 2938–2949. [Google Scholar] [CrossRef]
- Capitelli, F.; Selim, M.D.; Mukherjea, K.K. Synthesis and crystal structure determination of sodium molybdate dihydrate. Asian J. Chem. 2006, 18, 2856–2860. [Google Scholar]
- Evans, H.T.; Gatehouse, M.B.; Leverett, P. Crystal structure of the heptamolybdate(VI) (paramolybdate) ion, [Mo7O24]6−, in the ammonium and potassium tetrahydrate salts. J. Chem. Soc. Dalton Trans. 1975, 6, 505–514. [Google Scholar] [CrossRef]
- Bolzan, A.A.; Kennedy, B.J.; Howard, C.J. Neutron Powder diffraction study of molybdenum and tungsten dioxides. Aust. J. Chem. 1995, 48, 1473–1477. [Google Scholar] [CrossRef]
- Akcil, A.; Koldas, S. Acid mine drainage (AMD): Causes, treatment and case studies. J. Clean. Prod. 2006, 14, 1139–1145. [Google Scholar] [CrossRef]
- Webster, J.G.; Swedlund, P.J.; Webster, K.S. Trace metal adsorption onto an acid mine drainage iron(III) oxy hydroxy sulfate. Environ. Sci. Technol. 1998, 32, 1361–1368. [Google Scholar] [CrossRef]
- Ribbe, P.H. Chemistry, structure, and nomenclature of feldspars. Rev. Mineral. 1975, 2, 1–12. [Google Scholar]
- Goldberg, S.; Johnston, C.T.; Suarez, D.L.; Lesch, S.M. Mechanism of molybdenum adsorption on soils and soil minerals evaluated using vibrational spectroscopy and surface complexation modeling. Dev. Earth Environ. Sci. 2007, 7, 235–266. [Google Scholar]
- Gustafsson, J.P. Modelling molybdate and tungstate adsorption to ferrihydrite. Chem. Geol. 2003, 200, 105–115. [Google Scholar] [CrossRef]
- Arai, Y. X-ray absorption spectroscopic investigation of molybdenum multinuclear sorption mechanism at the goethite-water interface. Environ. Sci. Technol. 2010, 44, 8491–8496. [Google Scholar] [CrossRef] [PubMed]
- Davantès, A.; Lefèvre, G. In situ characterization of (poly)molybdate and (poly)tungstate ions sorbed onto iron (hydr)oxides by ATR-FTIR spectroscopy. Eur. Phys. J. Spec. Top. 2015, 224, 1977–1983. [Google Scholar] [CrossRef]
- Tsubaki, H.; Saito, T.; Murakami, T. Size distribution of ferrihydrite aggregate and its implication for metal adsorption and transport. J. Mineral. Petrol. Sci. 2012, 107, 244–249. [Google Scholar] [CrossRef]
- Fukushi, K.; Aoyama, K.; Yang, C.; Kitadai, N.; Nakashima, S. Surface complexation modeling for sulfate adsorption on ferrihydrite consistent with in situ infrared spectroscopic observations. Appl. Geochem. 2013, 36, 92–103. [Google Scholar] [CrossRef]
- Dahl, T.W.; Chappaz, A.; Fitts, J.P.; Lyons, T.W. Molybdenum reduction in a sulfidic lake: Evidence from X-ray absorption fine-structure spectroscopy and implications for the Mo paleoproxy. Geochim. Cosmochim. Acta 2013, 103, 213–231. [Google Scholar] [CrossRef]
| Extraction Steps | Reagents | Temp. | Targets |
|---|---|---|---|
| Fraction 1 | CH3COONa·3H2O (1 mol/L) with CH3COOH at pH 5 | Room Temp. | Exchangeable Ca and Mg carbonates |
| Fraction 2 | C2H8N2O4 (0.2 mol/L) with C2H2O4 at pH 3 | Low crystalline Fe and Mn oxides | |
| Fraction 3 | NH2OH·HCl (0.5 mol/L) with HNO3 at pH 2 | Crystalline Fe and Mn oxides | |
| Fraction 4 | H2O2 (8.8 mol/L), CH3COONH4 (1 mol/L) with HNO3 at pH 2 | 85 °C | Organic matter and sulfides |
| Fraction 5 | HF (48%) with HNO3 60%, HCl (30%) | 120 °C | Residues |
| W4 | W7 | W8 | W10.1 | W10.2 | ||
|---|---|---|---|---|---|---|
| T | °C | 1.5 | 1.3 | 3.1 | 1.2 | 6.8 |
| pH | (-) | 7.4 | 8.2 | 7.3 | 7.1 | 6.7 |
| ORP | mV | 200 | 180 | 180 | 170 | 230 |
| EC | mS/m | 100 | 160 | 110 | 200 | 220 |
| DO | mg/L | 7.5 | 6.5 | 7.7 | 5.1 | 2.3 |
| Na+ | mg/L | 54 | 54 | 55 | 140 | 95 |
| K+ | mg/L | 2.4 | 20 | 3.9 | 4.6 | 2.1 |
| Mg2+ | mg/L | 43 | 79 | 46 | 94 | 83 |
| Ca2+ | mg/L | 100 | 180 | 110 | 300 | 260 |
| SO42− | mg/L | 250 | 320 | 250 | 880 | 900 |
| Alkalinity | meq/L | 4.4 | 5.8 | 3.4 | 2.9 | 3.1 |
| NO2− | mg/L | 7.4 | 8.7 | 7.0 | 9.0 | 6.5 |
| NO3− | mg/L | 24 | 24 | 24 | 1.7 | <1 |
| Si | mg/L | 8.1 | 11 | 8.3 | 8.2 | 8.1 |
| Cl− | mg/L | 23 | 29 | 26 | 36 | 33 |
| Fe | mg/L | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 |
| Mn | mg/L | <0.5 | <0.5 | <0.5 | <0.5 | <0.5 |
| Cu | µg/L | <10 | <10 | <10 | <10 | <10 |
| Mo | µg/L | 27 | 7.6 | 24 | 780 | 1500 |
| Molybdate on Ferrihydrite (%) | Heptamolybdate (%) | MoO2 (%) | |
|---|---|---|---|
| S8 | 48 | 28 | 24 |
| S10.1 | 50 | 13 | 37 |
| S10.2 | 40 | 35 | 25 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Solongo, T.; Fukushi, K.; Altansukh, O.; Takahashi, Y.; Akehi, A.; Baasansuren, G.; Ariuntungalag, Y.; Enkhjin, O.; Davaajargal, B.; Davaadorj, D.; et al. Distribution and Chemical Speciation of Molybdenum in River and Pond Sediments Affected by Mining Activity in Erdenet City, Mongolia. Minerals 2018, 8, 288. https://doi.org/10.3390/min8070288
Solongo T, Fukushi K, Altansukh O, Takahashi Y, Akehi A, Baasansuren G, Ariuntungalag Y, Enkhjin O, Davaajargal B, Davaadorj D, et al. Distribution and Chemical Speciation of Molybdenum in River and Pond Sediments Affected by Mining Activity in Erdenet City, Mongolia. Minerals. 2018; 8(7):288. https://doi.org/10.3390/min8070288
Chicago/Turabian StyleSolongo, Tsetsgee, Keisuke Fukushi, Ochir Altansukh, Yoshio Takahashi, Akitoshi Akehi, Gankhurel Baasansuren, Yunden Ariuntungalag, Odgerel Enkhjin, Boldbaatar Davaajargal, Davaasuren Davaadorj, and et al. 2018. "Distribution and Chemical Speciation of Molybdenum in River and Pond Sediments Affected by Mining Activity in Erdenet City, Mongolia" Minerals 8, no. 7: 288. https://doi.org/10.3390/min8070288
APA StyleSolongo, T., Fukushi, K., Altansukh, O., Takahashi, Y., Akehi, A., Baasansuren, G., Ariuntungalag, Y., Enkhjin, O., Davaajargal, B., Davaadorj, D., & Hasebe, N. (2018). Distribution and Chemical Speciation of Molybdenum in River and Pond Sediments Affected by Mining Activity in Erdenet City, Mongolia. Minerals, 8(7), 288. https://doi.org/10.3390/min8070288







