1. Introduction
The increasing human need for new products and technologies has led to intensive development of industry. On the other side, industrial development is usually followed by high levels of potentially hazardous materials which may have negative influence on the environment as well as human or animal health. In countries with a good economy, strict rules and laws have caused high control of the level of waste materials being released into the environment, and consequently, due to applying adequate techniques for waste purification, the influence of industry on the environment is usually very small or negligible. On the other hand, in developing countries, control and human consciousness are significantly weaker, which often leads to higher quantities of waste materials being released into the environment and a significant increase of ecological problems as a consequence.
Heavy metals are one of the most dangerous pollutants in the nature. They are especially toxic for living organisms because of their stability and tendency of accumulation in the environment [
1]. With the rapid development of industries such as plating facilities, fertilizers, tanneries, batteries, paper and pesticides, heavy metals wastewaters are increasingly discharged into the environment, either directly or indirectly. Heavy metals are not biodegradable and many of their ions are known to be toxic and carcinogenic. Toxic heavy metals which are of particular concern in the treatment of industrial wastewaters are Zn, Cu, Ni, Hg, Cd, Pb and Cr [
2]. Regarding acute toxicity, cadmium and lead along with mercury form “the big three” of heavy metals with the greatest potential hazard to humans and the environment [
3].
Mining presents a significant contribution to economic development in every country, but at the same time it is very often one of the biggest environmental pollutants. Contamination of the environment with heavy metals mainly occurs due to mining activities, i.e., after technological processes of mineral resources where as a result of processing flotation tailings remain [
4].
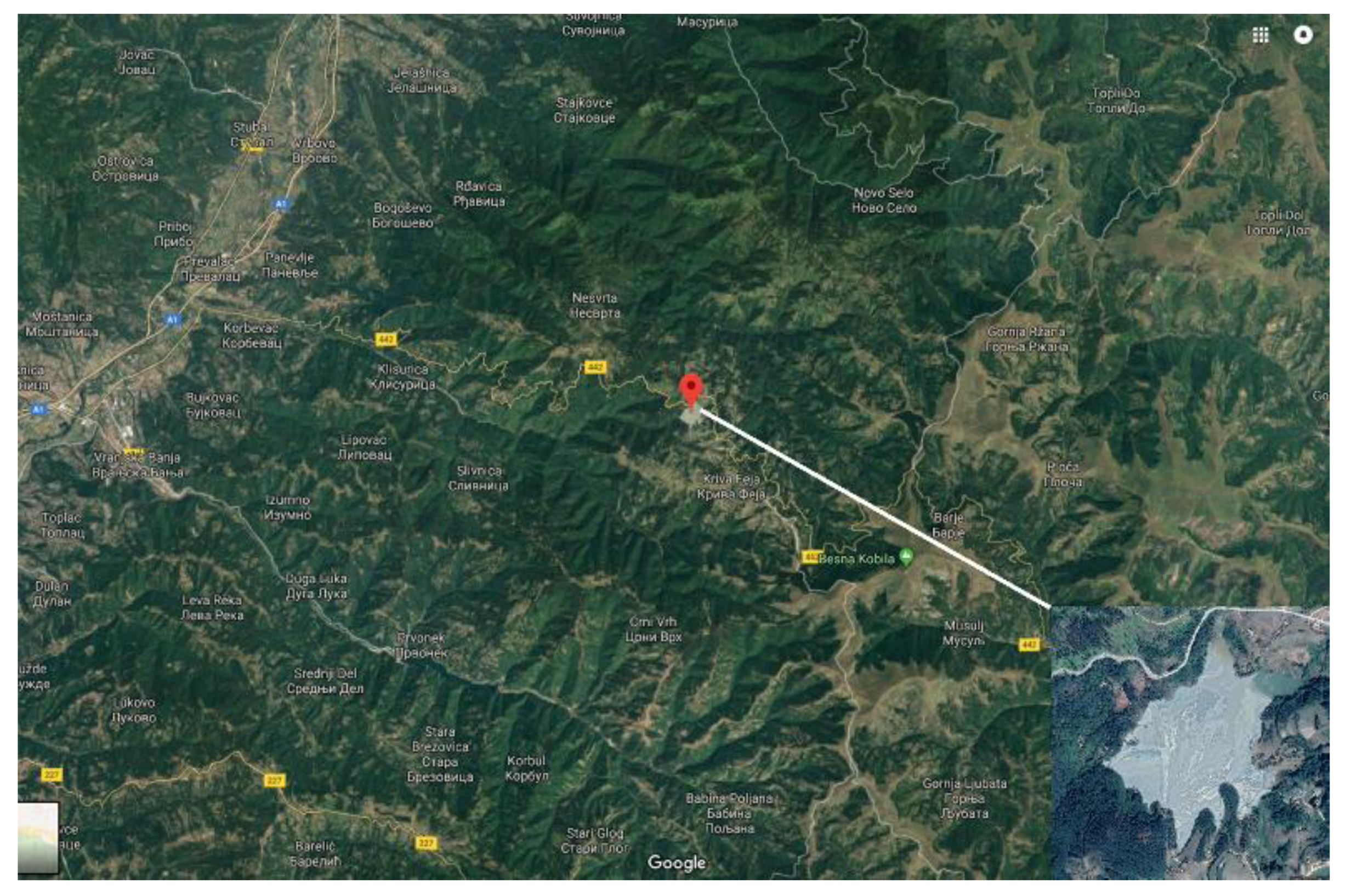
The Grot mine is one of the biggest mines in Serbia, with possibly the most significant influence on the environment. It belongs to the Besna Kobila metallographic region (1823 m) and it is located in the southeast of Serbia, near the town Vranje (
Figure 1). In the Grot mine, the exploitation and processing (flotation) of lead-zinc ore is carried out. Production of ore in the Grot mine is about 14,000 tons of ore monthly, and about 800 tons of lead and zinc concentrate are obtained per month in the flotation of the mine. Tailings of the mine Grot is about 24 ha in area, while the volume of the landfill material in the tailings is about 6 million tons.
The flotation tailings of the mine Grot, Serbia contain Cd, Pb, As, Cr, Ni, Cu, etc. The concentrations of mentioned heavy metals are mostly higher than maximum permissible concentrations (MPC) according to the laws of the Republic of Serbia [
5]. For that reason, the tailings represent a serious problem for nature and the environment, primarily because of the contamination they generate either by scattering particles from the tailings or by contamination of the soil. However, the most important negative effect, especially for human health, is the release of the wastewater. Namely, contaminated wastewater from tailings, is released without any control and with a flow of about 100 m
3/h, contaminating the river Korbevečka and polluting the South Morava, which belongs to the Danube basin. It is very important to note that rivers which are contaminated with tailings wastewater, especially in the surroundings areas of the tailings, where concentration of the heavy metals are the highest, are very often used by local populations to irrigate gardens and fields as well as to meet other water needs. Thus, it is important to apply necessary actions and procedures for purification and treatment which would prevent or at least minimize the pouring of the wastewater from tailings in the rivers and reduce negative effects on the environment.
Besides heavy metals, the contamination of waters with organic pollutants, such as dyes is very frequent, and also represents a serious environmental problem. Dyes are widely used in coloring processes in the leather, textile, paper, plastic and wood industries, as well as in food industries [
6,
7,
8]. Methylene blue (MB) basic cationic dye is the most frequently used dye for leather, cotton, silk and wood [
9,
10,
11]. It is very dangerous for the respiratory system if inhaled, causing mental confusion and mild poisoning if ingested by mouth [
9,
12,
13,
14]. Additionally, MB has been widely used in scientific investigations due to its high level of adsorption on solids, and as a model compound for adsorption characterizations of organic compounds in aqueous solutions [
15,
16,
17].
It is in common for all mentioned pollutants that they must not be found in nature without control, and for that reason, development of the new materials and techniques which may be used for contaminated waters treatment, either before or after discharging them into nature, represents one of the greatest scientific challenges today.
Different treatment methodologies may be used for that purpose such as adsorption, precipitation, ion-exchange, coagulation, membrane filtration and photo-degradation [
18,
19]. Among these, adsorption and ion exchange techniques are upgraded methods in terms of their simplicity, recyclability and mode of waste recovery from the pollutants [
18,
20].
In recent years, a lot of studies have been performed with aim of the selection and/or production of low-cost adsorbents with good adsorption capacities for both organic and inorganic pollutants such as methylene blue or heavy metals. For example, natural materials of both an organic and inorganic nature, such as alginate, chitosan, zeolites, clays, biomaterials, etc. are classified as adsorbents with potential to be widely used for the treatment of waters contaminated with heavy metals. They are available in large quantities and are low-cost, often biodegradable, usually recyclable and show high adsorption ability towards a great number of heavy metal ions [
21,
22,
23,
24]. On the other side, Bukallah et al. (2008) [
25] described the removal of methylene blue from an aqueous solution by adsorption on sand, while Tang et al. (2017) [
26] showed good adsorption properties for MB of the agricultural residue walnut shell. Tahir et al. (2008) followed adsorption of the methylene blue by using bioabsorbents such as Ulva lactuca and Sargassum [
27].
Besides mentioned adsorbents, activated carbons (ACs) are very beneficial materials for different uses such as power transfer, production wearable sensing and sensor application nanofibers [
28,
29,
30,
31,
32,
33,
34,
35]. In addition, their use is particularly significant in inorganic or organic pollutants removal, due to a large number of unique characteristics, such as a highly developed porous structure, large surface area, variability in surface chemistry, and a high level of surface activity [
33,
34,
35]. The major drawback of the conventional activated carbon-based adsorption process is the usage of non-renewable and relatively expensive raw materials (i.e., wood and coal), and therefore, it is still considered to be a high cost process with restricted applicability. To overcome such drawbacks and attain better economic viability, adsorption processes using non-conventional low-cost activated carbons derived from renewable and inexpensive materials such as naturally abundant biomass, saccharides, agricultural and industrial wastes have been examined by different researchers [
36,
37,
38,
39].
In general, for potential practical application of the material for pollutants removal from aqueous solutions, it is very important that the material should possess any kind of universality, i.e., possess the ability to remove different contaminants. For activated carbons, universality and adsorption properties, structure, carbon content, specific surface, etc. are very important [
40] and to get material with the best properties, it is necessary to apply different preparing conditions and perform detailed characterization as well.
In the presented paper, activated carbons were prepared by hydrothermal synthesis from easily accessible, eco-friendly and low-cost materials (glucose and fructose) and activated with different hydroxides in order to get the best properties for use in removal different (inorganic and organic) pollutants from contaminated aqueous solutions. The obtained samples were characterized in detail and then checked for removal of the inorganic (heavy metals) and organic (methylene blue) pollutants from contaminated aqueous solutions. Experiments were also performed on real wastewater samples taken from tailings of the lead and zinc mine, but experiments were performed in laboratory conditions.
2. Materials and Methods
Chemicals that were used in this study were of high purity and supplied from Sigma Aldrich, Darmstadt, Germany.
2.1. Synthesis of the Samples
Synthesis of activated carbon materials was performed in two steps. At first, required amounts of glucose (G) and fructose (F) were dissolved in deionized water in order to prepare solutions with a concentration of 1.0 mol/dm3. Hydrothermal synthesis was carried out in the PTFE (polytetrafluoroethylene) chamber placed in a stainless steel autoclave. Prepared solutions were placed into autoclave and heated into an oven under autogenous pressure at 240 °C for 24 h. Afterward, samples were filtrated, washed with distilled water and dried for 1 h at 100 °C.
The second part of experiments was activation of carbon containing materials with selected hydroxides (KOH, NaOH and LiOH) as activation agents. Chemical activation was performed by dry mixing of selected precursor and hydroxide at room temperature with the ratio 1:3. Subsequently, the mixed samples were activated in a horizontal tube furnace (Protherm Furnaces, model PTF 16/38/250, Ankara, Turkey) at 750 °C, 200 cm3/min nitrogen flow and the heating rate of 5 °C/min, with a retention time of 1 h.
Activated carbons obtained by this procedure were designated as AC@G–T–N2–t–XOH and AC@F–T–N2–t–XOH, where G and F are precursors, N2 is activation gas, T and t are temperature and time of activation while XOH represent activation agents (X = Li, Na, K). During activations the following yields were obtained: 6.0; 7.9 and 13.6 for LiOH, NaOH and KOH activated glucose, respectively, and 6.4; 7.4 and 13.5 for LiOH, NaOH and KOH activated fructose, respectively.
2.2. Characterization Methods
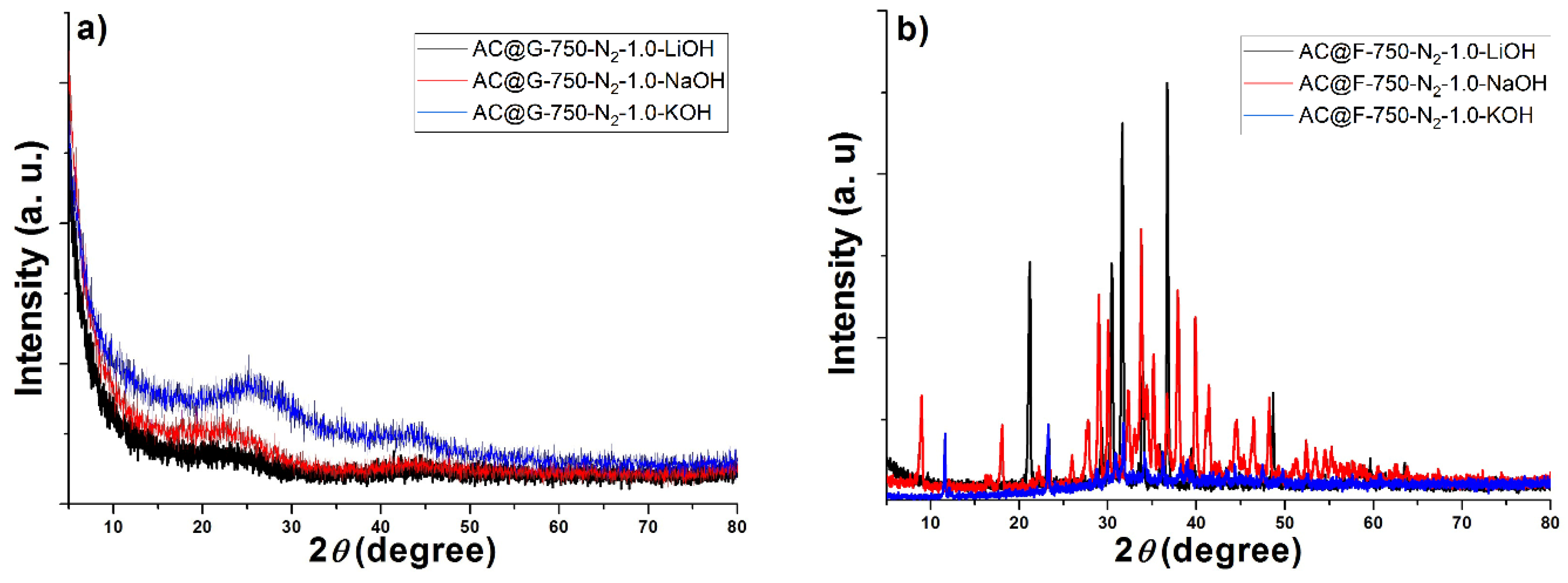
The crystal structure of the obtained samples was characterized by x-ray powder diffraction (XRPD) analysis using Ultima IV Rigaku diffractometer (Rigaku, The Woodlands, TX, USA), equipped with Cu Kα 1,2 radiation source (a generator voltage of 40.0 kV and a generator current of 40.0 mA). All samples were recorded in the range of 10–90° 2θ, with a scanning step of 0.02° and at a scan rate of 2°/min.
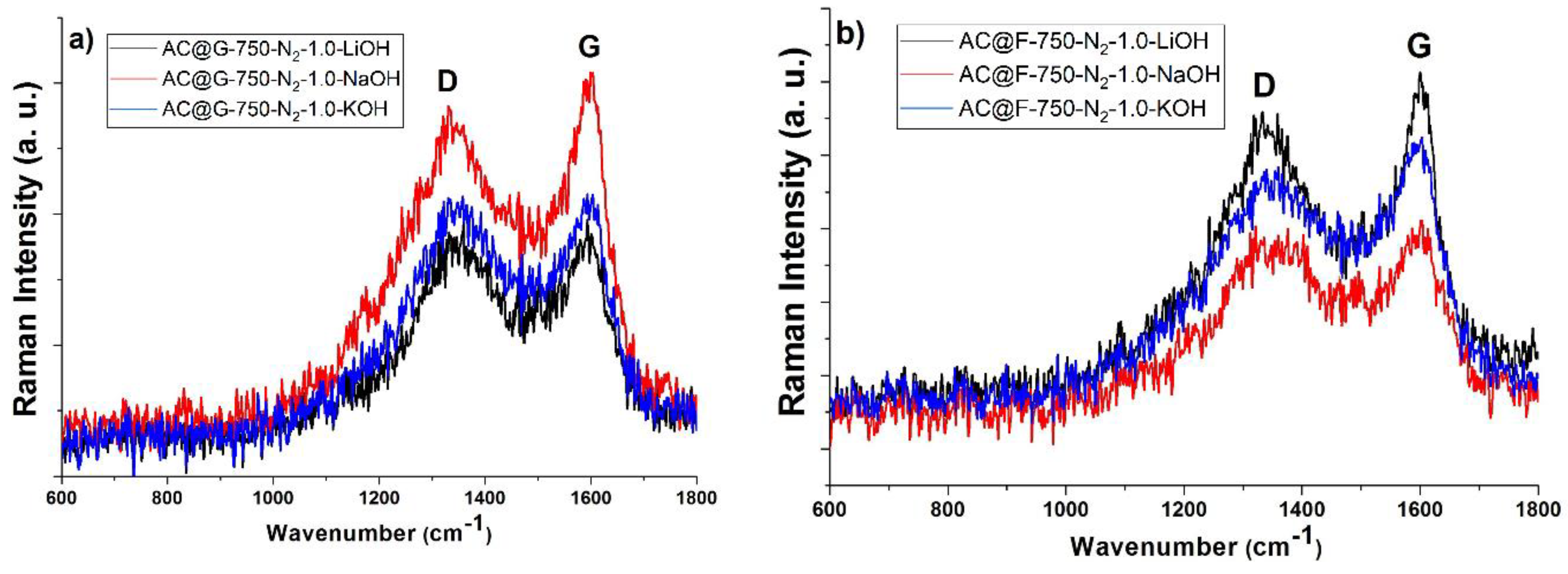
Raman spectra of activated carbons were collected on a DXR Raman microscope (Thermo Scientific, Waltham USA) equipped with an Olympus optical microscope and a CCD detector, with a diode pumped solid state high-brightness laser (532 nm) and a 10× objective. The powdered sample was placed on X–Y motorized sample stage. The analysis of the scattered light was carried out by the spectrograph with a grating 900 lines/mm. The laser power was 1 mW.
Elemental analyses of samples were performed on a Vario EL III C, H, N, S/O Elemental Analyzer (Elementar, Langenselbold, Germany). The amounts of C, N and H were determined directly while the amount of oxygen was calculated as: 100%–ash%–C%–N%–H%.
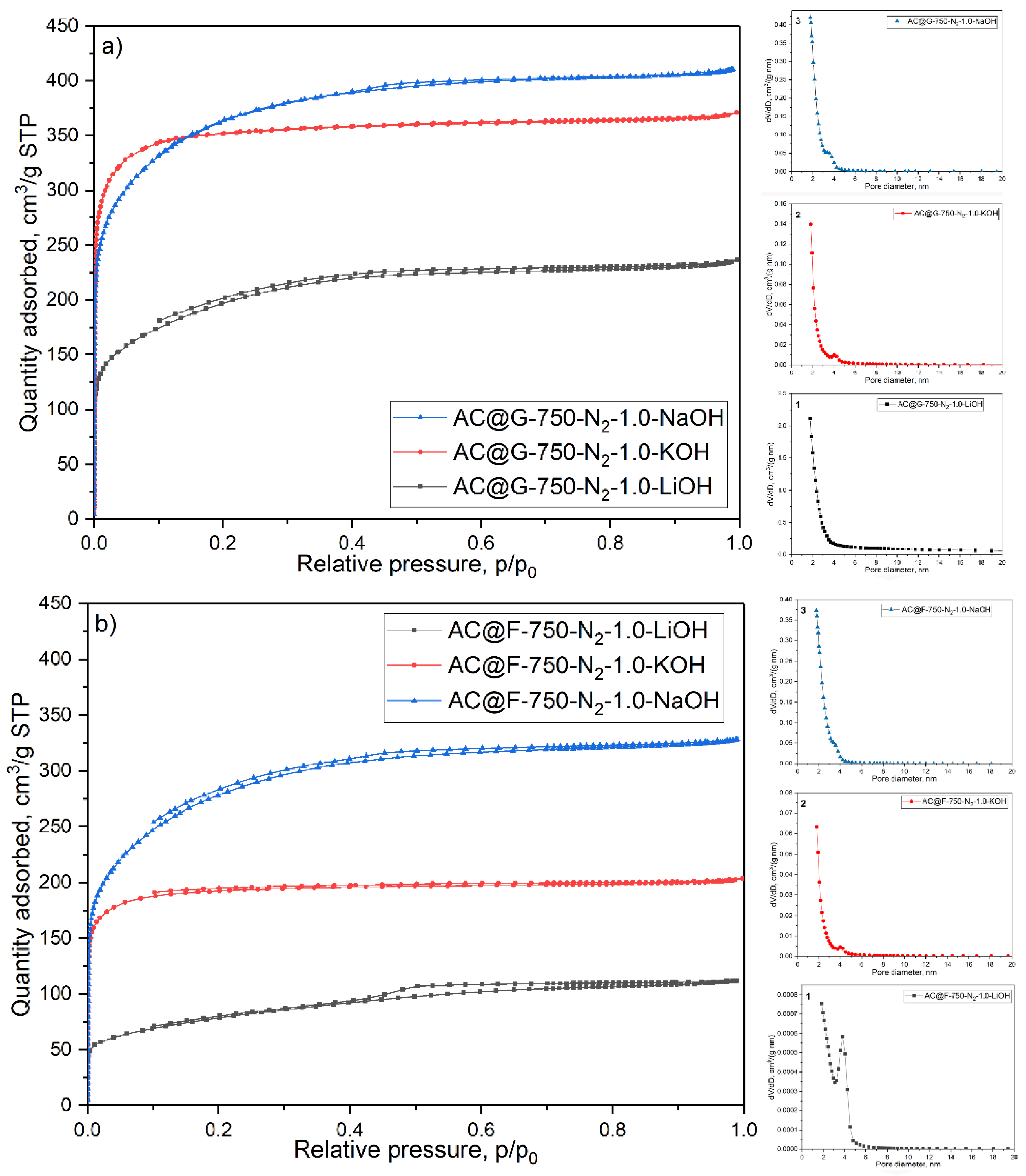
The adsorption/desorption isotherms of activated carbons based on adsorption/desorption of N
2 at 77 K were determined using a Micromeritics, model TriStar II 3020 (Micromeritics, Norcross, GA, USA). Degassing of samples under a N
2 stream (purity 6.0) and programmed bi-level heating (the first heating stage was at 90 °C during 60 min; the second heating stage was at 250 °C during 240 min). The specific surface area (S
BET) of obtained samples was calculated by the law of BET (Brunauer, Emmet, Teller) theory [
41] from the best linear fit in the region
p/
po < 0.01 (
p and
po represent the equilibrium and saturation pressures of nitrogen at a fixed temperature). Using BJH (Barrett, Joyner and Halenda) method [
41], for every sample from desorption branch of the nitrogen isotherm pore-size distributions were calculated. In addition, mesopore surface (S
meso) and micropore volume (V
micro) were also calculated from the isotherms. By using the high-resolution α
s–plot method [
42,
43,
44,
45], mesopore surface area (S
meso) and micropore volume (V
micro) were determined. Micropore surface (S
micro) was calculated by subtracting S
meso from the S
BET.
The morphology of the investigated activated carbons was examined by scanning electron microscope Tescan MIRA3 XM FESEM (Tescan, Brno—Kohoutovice, Czech Republic).
2.3. Adsorption Experiments
Experiments on testing availability of the selected samples to be used in area of environmental protection and purification of waters contaminated with heavy metals were performed under batch conditions for the single component solutions of lead(II)-nitrate, cadmium(II)-nitrate and zinc(II)-nitrate of known heavy metal ions concentrations (~700 mg/dm
3). For that purpose, 0.5 g of the adsorbents were mixed with 50 cm
3 of heavy metals solutions at 350 rpm, 24 h at initial pH 4.2 and room temperature. Initial pH was adjusted by adding very small quantities (few drops) of 0.1 mol/dm
3 HNO
3. After reaction time the suspensions were centrifuged, and concentrations of the heavy metals, as well as concentrations of potassium, sodium and lithium ions, were determined from the supernatants using an atomic absorption spectrometer (AAS) Analytic Jena 300 (Analytic Jena, Jena, Germany). Final pH was also measured. In the second step, adsorption experiments were performed on wastewater taken from tailings of the lead and zinc flotation facility Grot, Vranje, Serbia. The samples were taken from the outlet pipe of the flotation facility (OF) and hydro-cyclone overflow (PHC) in accordance with standard procedure [
46]. Collected samples were kept in the capped container and left at room temperature in order to separate liquid from the solid phase. After few days, the liquid phase was decanted and filtered through qualitative filter paper in order to remove large particles and impurities. The adsorption experiments were performed under the following batch conditions: 0.5 g of the adsorbent was mixed with 50 cm
3 of the contaminated water for 24 h. According to SRPS ISO 5667-10:2007, during sampling, the wastewater is mixed with a solution of nitric acid in order to stabilize it, so the pH of the solution after sampling and before materials testing was ~4.2. For that reason, all experiments related to heavy metals removal were performed at initial pH = 4.2 and final pHs were measured. After 24 h the suspensions were centrifuged, and the initial and non-adsorbed concentrations of heavy metals were determined using an inductively coupled plasma optical emission spectrometry (ICP-OES) on an ICP spectrometer Spectroflame (Spectro Analytical Instruments, Kleve, Germany).
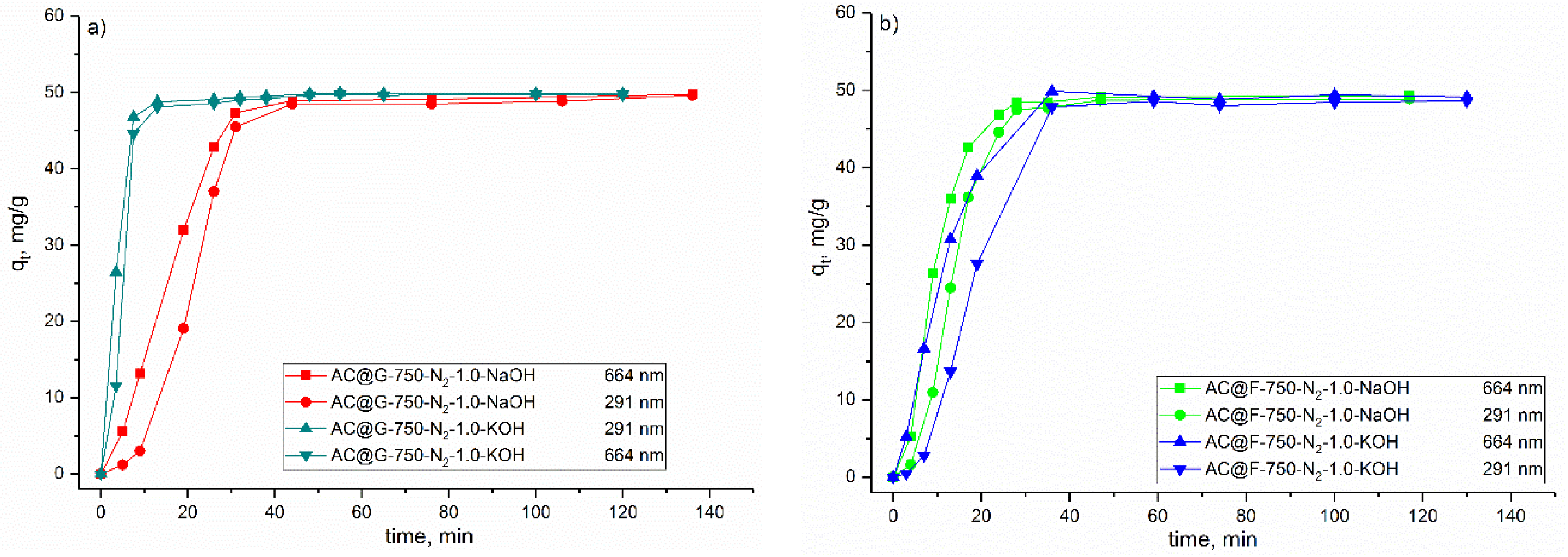
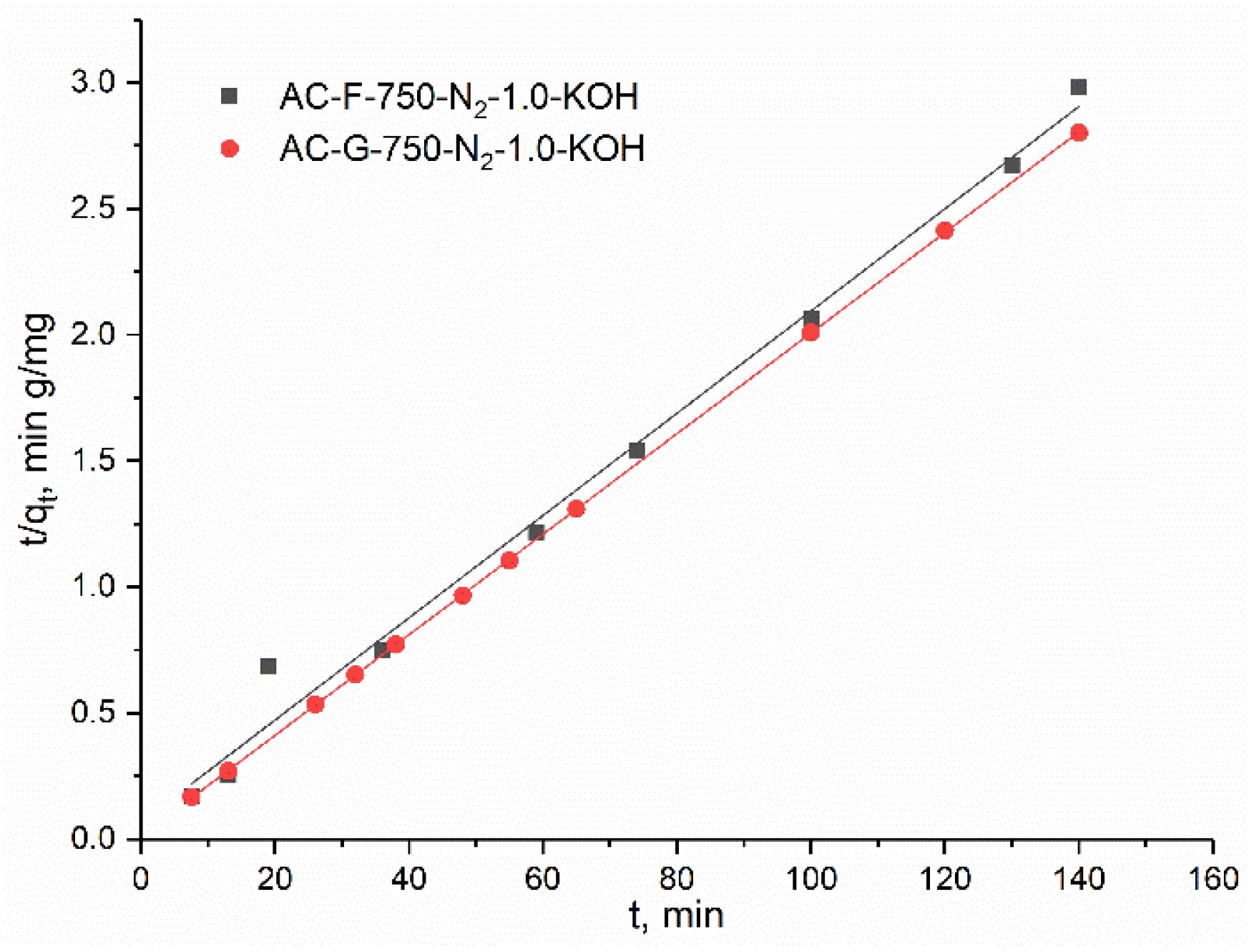
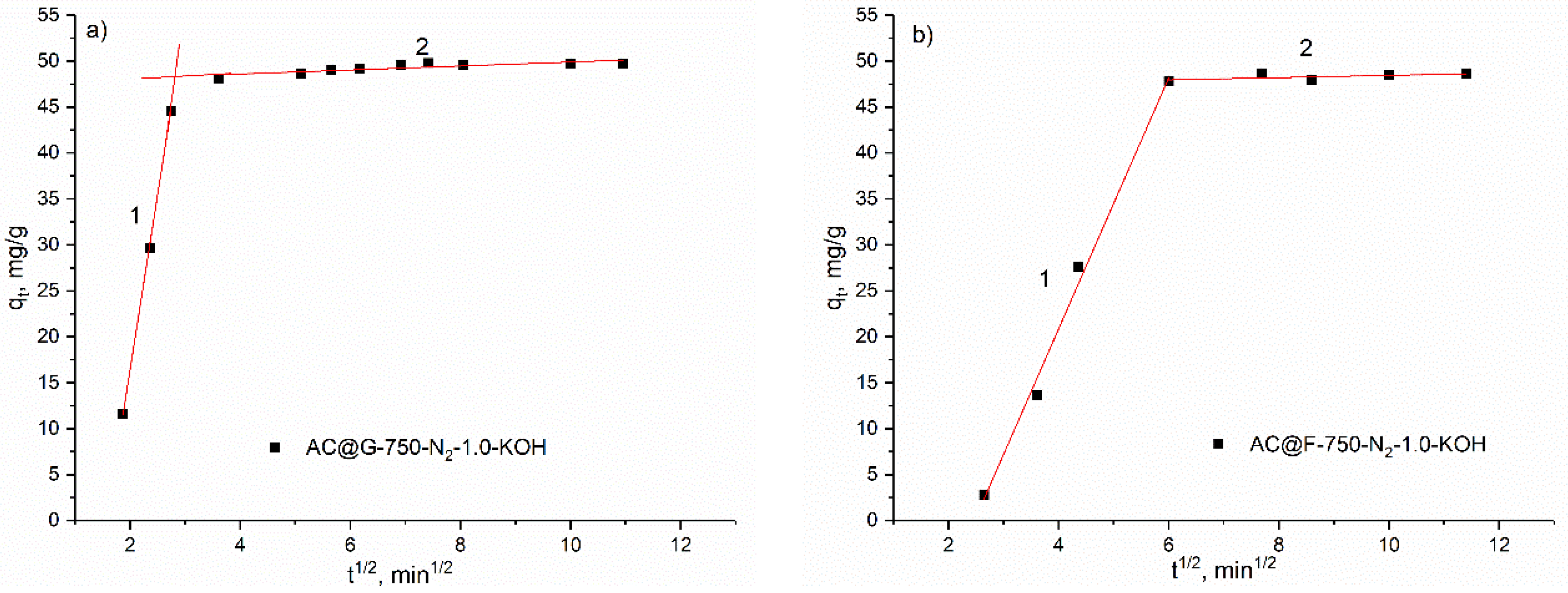
The adsorption experiments for dyes were carried out in magnetically stirred (σ = 400 rpm) glass vessel. Testing of the methylene blue (MB) adsorption was performed for initial MB concentration of 200 mg/dm3 in distilled water at room temperature and under conditions protected from light. In order to simulate real conditions, the initial pH (6.6) as well as final pH were not adjusted and were only measured. The solid/liquid ratio applied in these experiments was 25 mg/25 cm3. The contact time was 24 h. The kinetic experiments were performed with the concentration of the MB of 50 mg/dm3, and other conditions as in previous experiments (250 mg of the adsorbents were mixed with a 250 cm3 MB solution). At a specific time, 1 cm3 of the solutions were taken, centrifuged and analyzed on Agilent 8435 UV-VIS spectrophotometer with a diode array detector (Agilent, Santa Clara, CA, USA). The two maximum wavelengths for MB were used, the first in the ultraviolet (UV) and the second one in the visible (VIS) range of electromagnetic spectrum. Thus, the absorption spectra were collected at 291 nm and 664 nm.