Comparison of Heterotrophic Bioleaching and Ammonium Sulfate Ion Exchange Leaching of Rare Earth Elements from a Madagascan Ion-Adsorption Clay
Abstract
1. Introduction
2. Materials and Methods
3. Results
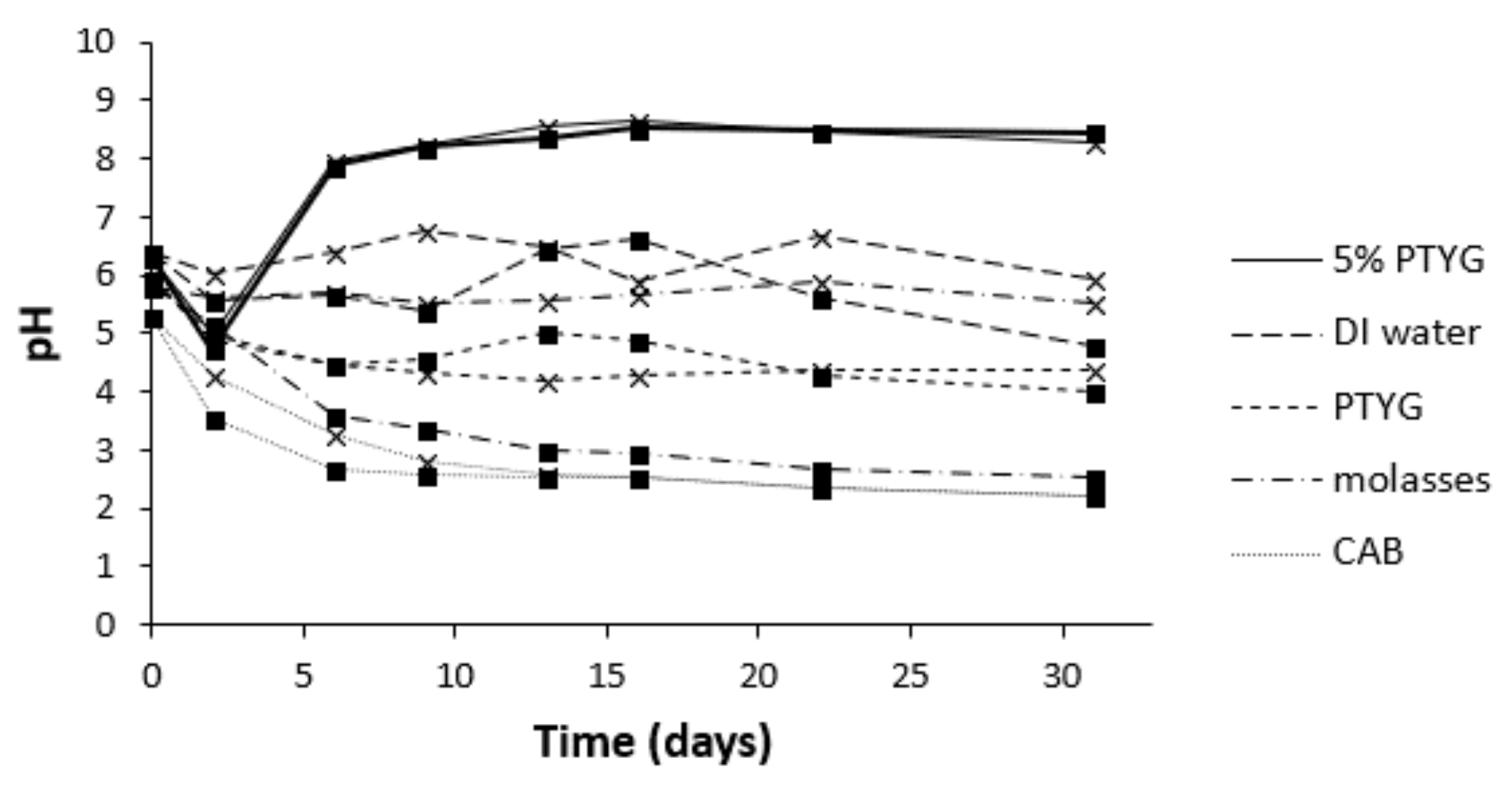
3.1. Selection of Isolates, Growth Medium and IAD
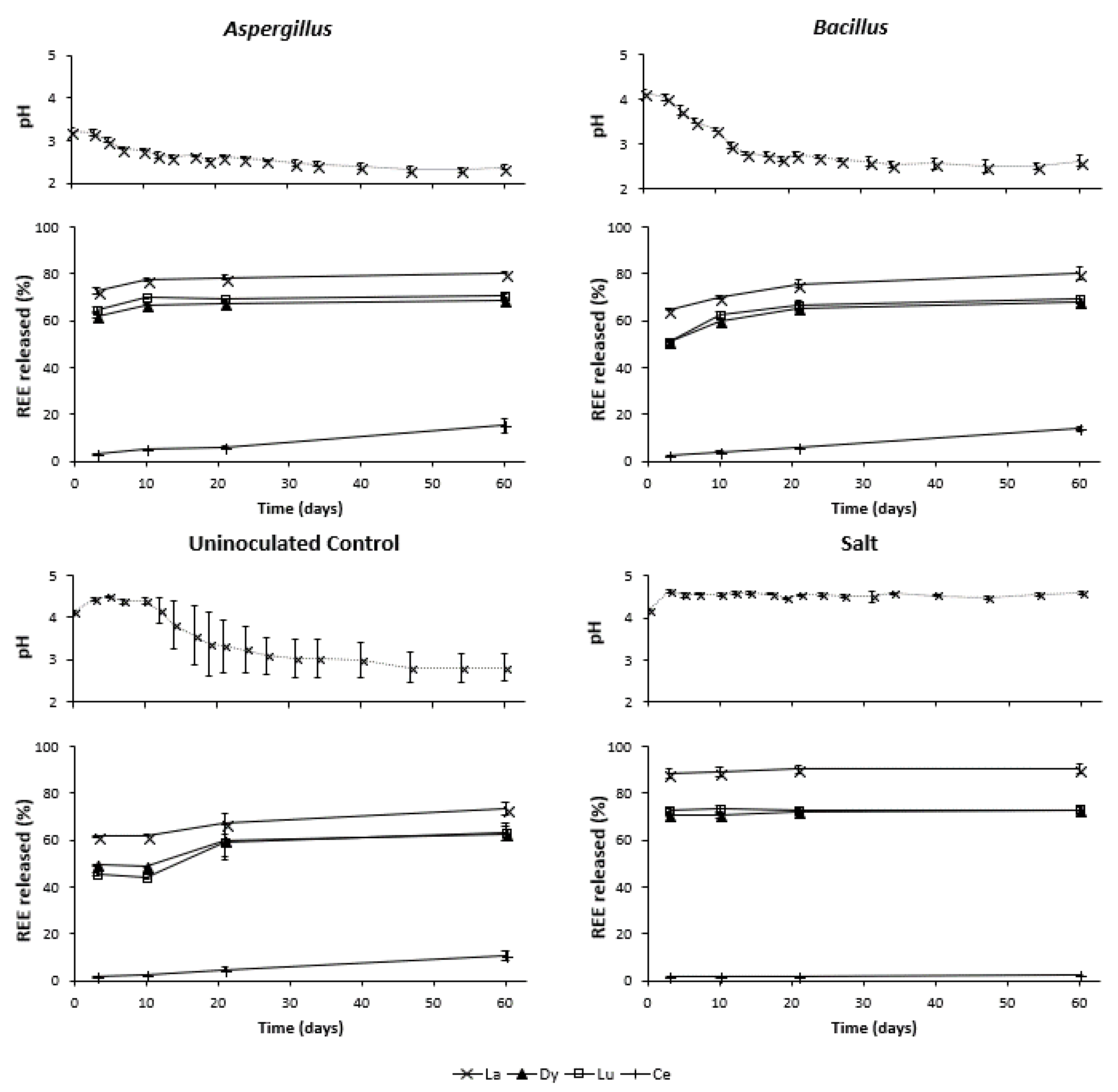
3.2. Changes in pH during Leaching
3.3. REE Released during Bioleaching and Salt Leaching
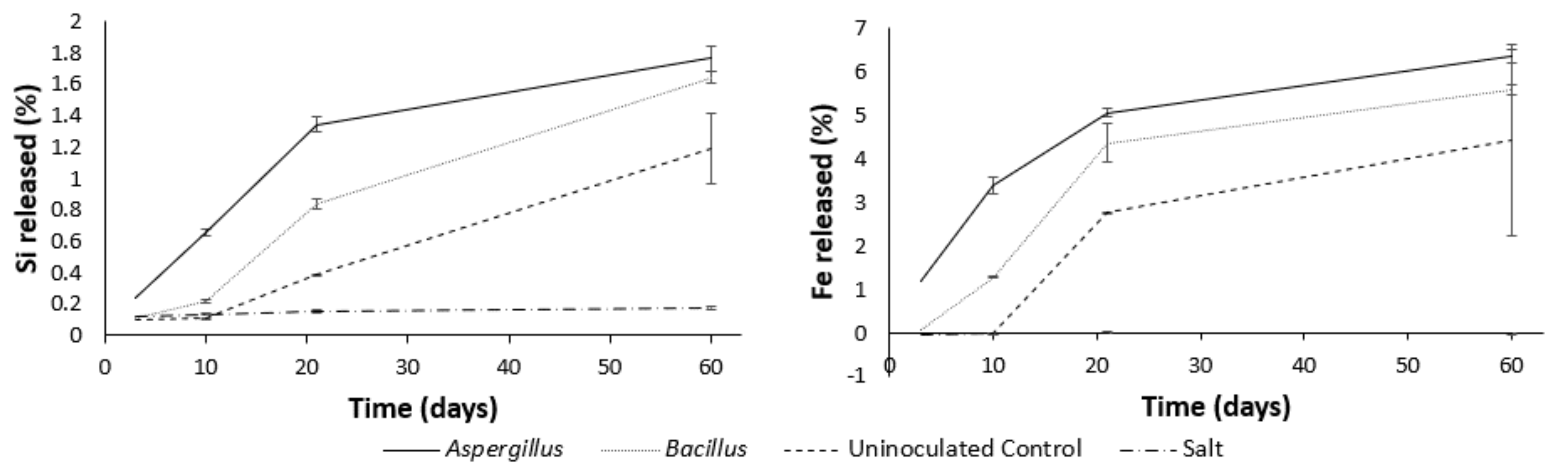
3.4. Major Elements during Leaching
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- EU Commission (EC). Report on Critical Raw Materials for the EU; European Commission: Brussels, Belgium, 2014. [Google Scholar]
- Brown, T.J.; Idoine, N.E.; Raycraft, E.R.; Shaw, R.A.; Deady, E.A.; Hobbs, S.F.; Bide, T. World Mineral Production 2011–2015; British Geological Survey: Keyworth, UK; Nottingham, UK, 2017. [Google Scholar]
- Wall, F. Rare earth elements. In Critical Metals Handbook; Gunn, G., Ed.; John Wiley & Sons Ltd.: New York, NY, USA, 2013; pp. 312–339. [Google Scholar]
- Yang, X.J.; Lin, A.; Li, X.L.; Wu, Y.; Zhou, W.; Chen, Z. China’s ion-adsorption rare earth resources, mining consequences and preservation. Environ. Dev. 2013, 8, 131–136. [Google Scholar] [CrossRef]
- Sanematsu, K.; Watanabe, Y. Characteristics and genesis of ion adsorption-type rare earth element deposits. Rev. Econ. Geol. 2016, 18, 55–79. [Google Scholar]
- Moldoveanu, G.A.; Papangelakis, V.G. Recovery of rare earth elements adsorbed on clay minerals: I. Desorption mechanism. Hydrometallurgy 2012, 117–118, 71–78. [Google Scholar] [CrossRef]
- Bradbury, M.H.; Baeyens, B. Sorption of Eu on Na- and Ca-montmorillonites: Experimental investigations and modelling with cation exchange and surface complexation. Geochim. Cosmochim. Acta 2002, 66, 2325–2334. [Google Scholar] [CrossRef]
- Barmettler, F.; Castelberg, C.; Fabbri, C.; Brandl, H. Microbial mobilization of rare earth elements (REE) from mineral solids—A mini review. AIMS Microbiol. 2016, 2, 190–204. [Google Scholar] [CrossRef]
- Burgstaller, W.; Schinner, F. Leaching of metals with fungi. J. Biotechnol. 1993, 27, 91–116. [Google Scholar] [CrossRef]
- Brisson, V.L.; Zhuang, W.Q.; Alvarez-Cohen, L. Bioleaching of rare earth elements from monazite sand. Biotechnol. Bioeng. 2016, 113, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Hassanien, W.A.G.; Desouky, O.A.N.; Hussien, S.S.E. Bioleaching of some rare earth elements from Egyptian monazite using Aspergillus ficuum and Pseudomonas aeruginosa. Walailak J. Sci. Technol. 2014, 11, 809–823. [Google Scholar]
- Shin, D.; Kim, J.; Kim, B.; Jeong, J.; Lee, J. Use of phosphate solubilizing bacteria to leach rare earth elements from monazite-bearing ore. Minerals 2015, 5, 189–202. [Google Scholar] [CrossRef]
- Hewedy, M.A.; Rushdy, A.A.; Kamal, N.M. Bioleaching of rare earth elements and Uranium from Sinai soil, Egypt using Actinomycetes. Egypt. J. Hosp. Med. 2013, 53, 909–917. [Google Scholar] [CrossRef]
- Qu, Y.; Lian, B.; Mo, B.; Liu, C. Bioleaching of heavy metals from red mud using Aspergillus niger. Hydrometallurgy 2013, 136, 71–77. [Google Scholar] [CrossRef]
- Qu, Y.; Lian, B. Bioleaching of rare earth and radioactive elements from red mud using Penicillium tricolor RM-10. Bioresour. Technol. 2013, 136, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Pourret, O.; Davranche, M.; Gruau, G.; Dia, A. Rare earth elements complexation with humic acid. Chem. Geol. 2007, 243, 128–141. [Google Scholar] [CrossRef]
- Tang, J.; Johannesson, K.H. Speciation of rare earth elements in natural terrestrial waters: Assessing the role of dissolved organic matter from the modeling approach. Geochim. Cosmochim. Acta 2003, 67, 2321–2339. [Google Scholar] [CrossRef]
- Wood, S.A. The aqueous geochemistry of the rare-earth elements: Critical stability constants for complexes with simple carboxylic acids at 25 °C and 1 bar and their application to nuclear waste management. Eng. Geol. 1993, 34, 229–259. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Takahashi, Y.; Shimizu, H. Systematic change in relative stabilities of REE-humic complexes at various metal loading levels. Geochem. J. 2010, 44, 39–63. [Google Scholar] [CrossRef]
- Martell, A.E.; Smith, R.M. Critical Stability Constants. Other Organic Ligands; Plenum: New York, NY, USA, 1977; Volume 3. [Google Scholar]
- Estrade, G.; Salvi, S.; Béziat, D.; Williams-Jones, A.E. The origin of skarn-hosted rare-metal mineralization in the Ambohimirahavavy alkaline complex, Madagascar. Econ. Geol. 2015, 110, 1485–1513. [Google Scholar] [CrossRef]
- Byrne, R.H.; Kim, K.-H. Rare earth precipitation and coprecipitation behavior: The limiting role of PO43− on dissolved rare earth concentrations in seawater. Geochim. Cosmochim. Acta 1993, 57, 519–526. [Google Scholar] [CrossRef]
- Takahashi, Y.; Châtellier, X.; Hattori, K.H.; Kato, K.; Fortin, D. Adsorption of rare earth elements onto bacterial cell walls and its implication for REE sorption onto natural microbial mats. Chem. Geol. 2005, 219, 53–67. [Google Scholar] [CrossRef]
- Moldoveanu, G.A.; Papangelakis, V.G. Recovery of rare earth elements adsorbed on clay minerals: II. Leaching with ammonium sulfate. Hydrometallurgy 2013, 131–132, 158–166. [Google Scholar] [CrossRef]
- Tian, J.; Yin, J.; Chi, R.; Rao, G.; Jiang, M.; Ouyang, K. Kinetics on leaching rare earth from the weathered crust elution-deposited rare earth ores with ammonium sulfate solution. Hydrometallurgy 2010, 101, 166–170. [Google Scholar]
- Moldoveanu, G.A.; Papangelakis, V.G. An overview of rare-earth recovery by ion-exchange leaching from ion-adsorption clays of various origins. Mineral. Mag. 2016, 80, 63–76. [Google Scholar] [CrossRef]
- Gilbertson, J. A Competent Persons Report on the Tantalus Project, Northern Madagascar; Prepared by Tantalus Rare Earths AG; SRK Exploration Services Ltd.: Cardiff, UK, 2015. [Google Scholar]
- Lazo, D.E.; Dyer, L.G.; Alorro, R.D. Silicate, phosphate and carbonate mineral dissolution behaviour in the presence of organic acids: A review. Miner. Eng. 2017, 100, 115–123. [Google Scholar] [CrossRef]
- Stumm, W.; Morgan, J.J. Kinetics at the solid-water interface: Adsoprtion, dissolution of minerals, nucleation, and crystal growth. In Aquatic Chemistry, Chemical Equilibria and Rates in Natural Waters, 3rd ed.; Stumm, W., Morgan, J.J., Eds.; John Wiley & Sons Inc.: New York, NY, USA, 1996; p. 1022. [Google Scholar]
- Jenneman, G.E.; McInerney, M.J.; Crocker, M.E.; Knapp, R.M. Effect of sterilization by dry heat or autoclaving on bacterial penetration through Berea sandstone. Appl. Environ. Microbiol. 1986, 51, 39–43. [Google Scholar] [PubMed]
- Behera, S.K.; Panda, P.P.; Singh, S.; Pradhan, N.; Sukla, L.B.; Mishra, B.K. Study on reaction mechanism of bioleaching of nickel and cobalt from lateritic chromite overburdens. Int. Biodeterior. Biodegrad. 2011, 65, 1035–1042. [Google Scholar] [CrossRef]
- Arenas, L.F.; de León, C.P.; Walsh, F.C. Electrochemical redox processes involving soluble cerium species. Electrochim. Acta 2016, 205, 226–247. [Google Scholar] [CrossRef]
- Papagianni, M. Advances in citric acid fermentation by Aspergillus niger: Biochemical aspects, membrane transport and modeling. Biotechnol. Adv. 2007, 25, 244–263. [Google Scholar] [CrossRef] [PubMed]
- Andersen, M.R.; Lehmann, L.; Nielsen, J. Systemic analysis of the response of Aspergillus niger to ambient pH. Genome Biol. 2009, 10, R47. [Google Scholar] [CrossRef] [PubMed]
- Kubicek, C.P.; Schreferl-Kunar, G.; Wöhrer, W.; Röhr, M. Evidence for a cytoplasmic pathway of oxalate biosynthesis in Aspergillus niger. Appl. Environ. Microbiol. 1988, 54, 633–637. [Google Scholar] [PubMed]
- Young, J.M.; Rawlence, N.J.; Weyrich, L.S.; Cooper, A. Limitations and recommendations for successful DNA extraction from forensic soil samples: A review. Sci. Justice 2014, 54, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Bakhiet, S.E.A.; Al-Mokhtar, E.A.I. Production of citric acid by Aspergillus niger using sugarcane molasses as substrate. Jordan J. Biol. Sci. 2015, 8, 211–215. [Google Scholar]
- Sui, N.; Huang, K.; Lin, J.; Li, X.; Wang, X.; Xiao, C.; Liu, H. Removal of Al, Fe and Si from complex rare-earth leach solution: A three-liquid-phase partitioning approach. Sep. Purif. Technol. 2014, 127, 97–106. [Google Scholar] [CrossRef]
| Description | Inoculum | Fluid |
|---|---|---|
| Aspergillus | Aspergillus sp. Isolate | Modified CAB |
| Bacillus | Bacillus sp. Isolate | Modified CAB |
| Uninoculated Control | None | Modified CAB |
| Salt | None | 0.5 M ammonium sulfate |
| Element (mg·kg−1) | Si | Al | Fe | La | Ce | Dy | Lu |
|---|---|---|---|---|---|---|---|
| IAD | 188,569 | 175,298 | 37,358 | 1594 | 387 | 53.3 | 5.32 |
| Isolate | IAD Sample Depth | pH Drop | Solubilize Inorganic Phosphate | Urease Activity | Siderophore | Closest Match (Accession Number) |
|---|---|---|---|---|---|---|
| BP18 | 0–0.8 m, 3.8–4.8 m | + | − | − | − | Bacillus thuringiensis (KP813752) |
| BP23 | 0–0.8 m | − | + | − | − | Burkholderia sp. (AB911072) |
| BP25 | 0.8–3.8 m | + | − | + | − | Leifsonia sp. (LN876290) |
| BP32 | 0–0.8, 0.8–3.8 m | + | − | − | + | Cunninghamella bainieri (KF201293) |
| BP38 | 0.8–3.8, 3.8–4.8 m | + | − | − | − | Shewanella sp. (GU143896) |
| BP46 | 3.8–4.8 m | + | − | − | − | Uncultured bacterium (AM180664) |
| BP57 | 4.8–5.8 m | − | + | − | + | Bacillus aryabhattai (AB9304966) |
| TD01 | 0.9–1.1 m | + | − | − | * | Bacillus thuringiensis (HQ83480) |
| TD02 | 0.9–1.1 m | + | − | − | − | Bacillus sp. (KC893975) |
| TD17 | 0.9–1.1 m | − | − | − | − | Bacillus sp. (KU315821) |
| TD20 | 0.9–1.1 m | + | − | + | − | Bacterium (KT692625) |
| TD25 | 3.4–3.6 m | + | + | − | + | Aspergillus niger (KM516789) |
| TD29 | 3.4–3.6 m | + | + | − | − | Bacillus sp. (KJ584025) |
| TD31 | 3.4–3.6 m | + | − | + | − | Staphlococcus sp. (KX079771) |
| TD37 | 4.3–4.5 m | + | − | − | + | Bacillus sp. (KP670302) |
| TD47 | 3.4–3.6 m | + | − | − | − | Curvularia verruculosa (HF934909) |
| Organic Ligand (g·L−1) | pH | Eh | |||
|---|---|---|---|---|---|
| Oxalate | Citrate | Gluconate | mV | ||
| Aspergillus | 0.695 ± 0.318 | 10.2 ± 1.3 | 21.1 ± 1.2 | 2.38 ± 0.07 | 579 ± 5 |
| Bacillus | 0.563 ± 0.464 | 3.19 ± 3.73 | 21.7 ± 1.7 | 2.62 ± 0.13 | 577 ± 53 |
| Uninoculated Control | 1.03 ± 0.18 | 0.858 ± 0.888 | 9.45 ± 7.92 | 2.81 ± 0.34 | 640 ± 100 |
| Original medium | <0.05 | <0.05 | <0.05 | 4.38 | 4.38 |
| Aspergillus (×10−3) | Bacillus (×10−3) | Uninoculated Control (×10−3) | Salt (×10−3) | |
|---|---|---|---|---|
| Day 3 | 2.98 ± 0.01 | 2.61 ± 0.04 | 2.47 ± 0.04 | 2.75 ± 0.05 |
| Day 10 | 3.01 ± 0.02 | 2.99 ± 0.05 | 2.36 ± 0.01 | 2.74 ± 0.00 |
| Day 21 | 2.94 ± 0.03 | 2.94 ± 0.02 | 2.95 ± 0.40 | 2.68 ± 0.03 |
| Day 60 | 2.93 ± 0.05 | 2.88 ± 0.04 | 2.88 ± 0.13 | 2.69 ± 0.01 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barnett, M.J.; Palumbo-Roe, B.; Gregory, S.P. Comparison of Heterotrophic Bioleaching and Ammonium Sulfate Ion Exchange Leaching of Rare Earth Elements from a Madagascan Ion-Adsorption Clay. Minerals 2018, 8, 236. https://doi.org/10.3390/min8060236
Barnett MJ, Palumbo-Roe B, Gregory SP. Comparison of Heterotrophic Bioleaching and Ammonium Sulfate Ion Exchange Leaching of Rare Earth Elements from a Madagascan Ion-Adsorption Clay. Minerals. 2018; 8(6):236. https://doi.org/10.3390/min8060236
Chicago/Turabian StyleBarnett, Megan J., Barbara Palumbo-Roe, and Simon P. Gregory. 2018. "Comparison of Heterotrophic Bioleaching and Ammonium Sulfate Ion Exchange Leaching of Rare Earth Elements from a Madagascan Ion-Adsorption Clay" Minerals 8, no. 6: 236. https://doi.org/10.3390/min8060236
APA StyleBarnett, M. J., Palumbo-Roe, B., & Gregory, S. P. (2018). Comparison of Heterotrophic Bioleaching and Ammonium Sulfate Ion Exchange Leaching of Rare Earth Elements from a Madagascan Ion-Adsorption Clay. Minerals, 8(6), 236. https://doi.org/10.3390/min8060236




