4.1. Mineralogy
Phenocryst and groundmass typically comprise equal proportions of the rhyolite (50% phenocrysts, 50% groundmass;
Figure 2). Plagioclase feldspar comprises less than 15% of the rhyolite as subhedral to euhedral, rectangular (smaller than 0.5 × 1.2 cm), albite lathes, commonly with K-feldspar overgrowths. These lathes occur in clusters, as penetrating twins, or as individual phenocrysts, and occasionally K-feldspar mantles have their own albite overgrowths. The albite before the potassium feldspar overgrowth is typically equant without reaction textures, indicating a lack of dissolution before the formation of the K-feldspar. Potassium feldspar constitutes about 45–55% of the rhyolite and is found as 0.75–2 cm phenocrysts with hourglass sector zonation, or as mantles around early formed sodic plagioclase (
Figure 3). Quartz comprises about 25–35% of the rhyolite, occurring as zoned, anhedral to subhedral 0.2–1.5 cm phenocrysts. The core is typically equant, euhedral to subhedral, early beta quartz, pseudomorphed to trigonal, low temperature, alpha quartz. Quartz typically exhibits pseudo-concentric rings (snowball texture), where repeated corrosion and crystallization of later quartz overgrowths occurred. Biotite is annitic and comprises less than 5% of the rhyolite. The annite occurs as interstitial subhedral to anhedral phenocrysts less than 1 mm in size, displaying moderate pleochroism, alternating from a greenish-yellow to dark brown-green, where the edges of are commonly irregular, ragged, and undulatory, filling the space between earlier phenocrysts.
Minor phases comprise less than 10% of the rhyolite, including magnetite, hematite, and zircon. Magnetite and hematite comprise 2–3% of the rhyolite, where hematite is directly related to oxidation. Magnetite occur as euhedral to subhedral 0.1–1.5 cm phenocrysts, and as micro-crysts in the groundmass. Magnetite is typically found as anhedral inclusions in other phenocrysts, ranging from 1–5 mm in size, in close association with other accessary phases, including cassiterite, changbaiite (PbNb2O6), columbite-(Fe) group minerals, zircon and Y ± REE fluorides. Hematite occurs as replacement around the edges of magnetite phenocrysts, and less than 1–2 μm inclusions within the K-feldspar groundmass.
Trace minerals include zircon, Y-REE-fluorides, cassiterite, cerianite-(Ce), changbaiite, columbite-(Fe), cryolite and thorite. Zircon is the most common trace mineral, approximately 0.5–1% of the rhyolite at Round Top. Two populations of zircon are distinguished: one has abundant thorite inclusions and in the other these inclusions are rare. Most grains are 0.1–0.4 mm anhedral to subhedral, occasionally euhedral, phenocrysts. Rubin et al. [
21] hypothesized the two zircon populations represent an early magmatic (thorite inclusion-rich), and a later population introduced by migrating hydrothermal fluids (thorite inclusion-poor). Ref. [
21] noted the ubiquitous presence of late-stage hydrothermal zircons within fluorite replacement in surrounding limestone, but not within Round Top rhyolite. Both zircon populations (thorite inclusion-rich and inclusion-poor) were observed in Round Top rhyolite, commonly in close proximity to each other.
Yttrofluorite ((Ca, Y, HREE)F
2), yttrocerite ((Ca, Y, Ce, LREE, HREE)F
2) and variations that include La, Th, Nd, Dy, Yb, Lu, and Tm are the main REE-fluorides identified within Round Top rhyolite (
Figure 4). Additional REE-bearing minerals have been observed and detailed in other papers [
3,
16,
22], including aeschynite-(Ce), synchysite-group minerals, Nb-REE and Ca–Th-REE fluorides. The REE-fluorides and other trace minerals are typically nanoscale, anhedral, interstitial, and late crystallizing.
4.2. Geochemistry
The Round Top laccolith comprises relatively homogenous, high silica, non-topaz bearing rhyolite with low MgO and CaO, high Na
2O and K
2O (
Figure 5;
Table 1), and relatively high Zr (960–1100 ppm), Rb (1660–1930 ppm), Th (160–190 ppm), and U (28–58 ppm;
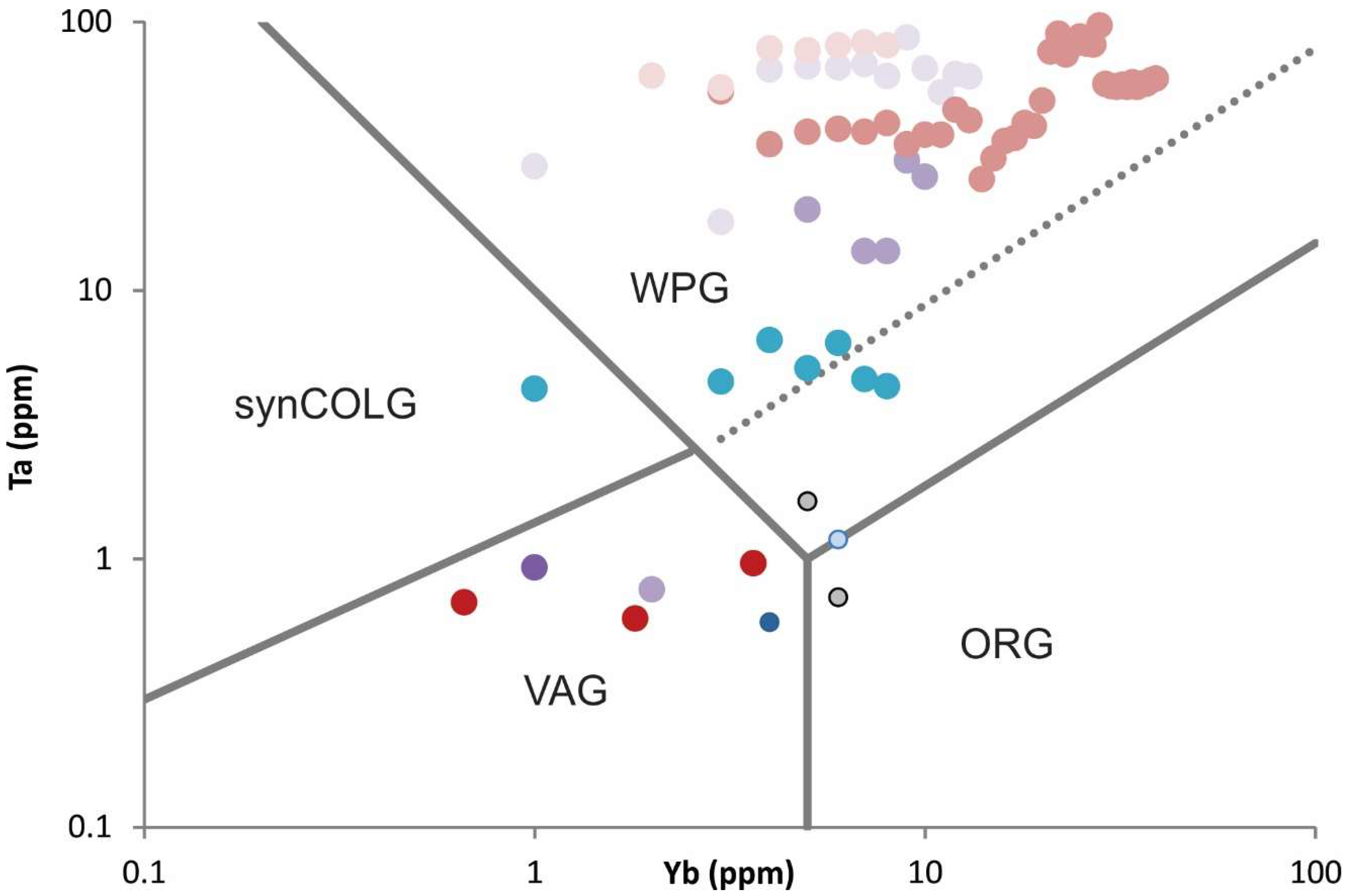
Table 2). Felsic rocks from the Round Top and Sierra Blanca suite of rocks show predominantly Within Plate (WPG) tectonomagmatic affinity (
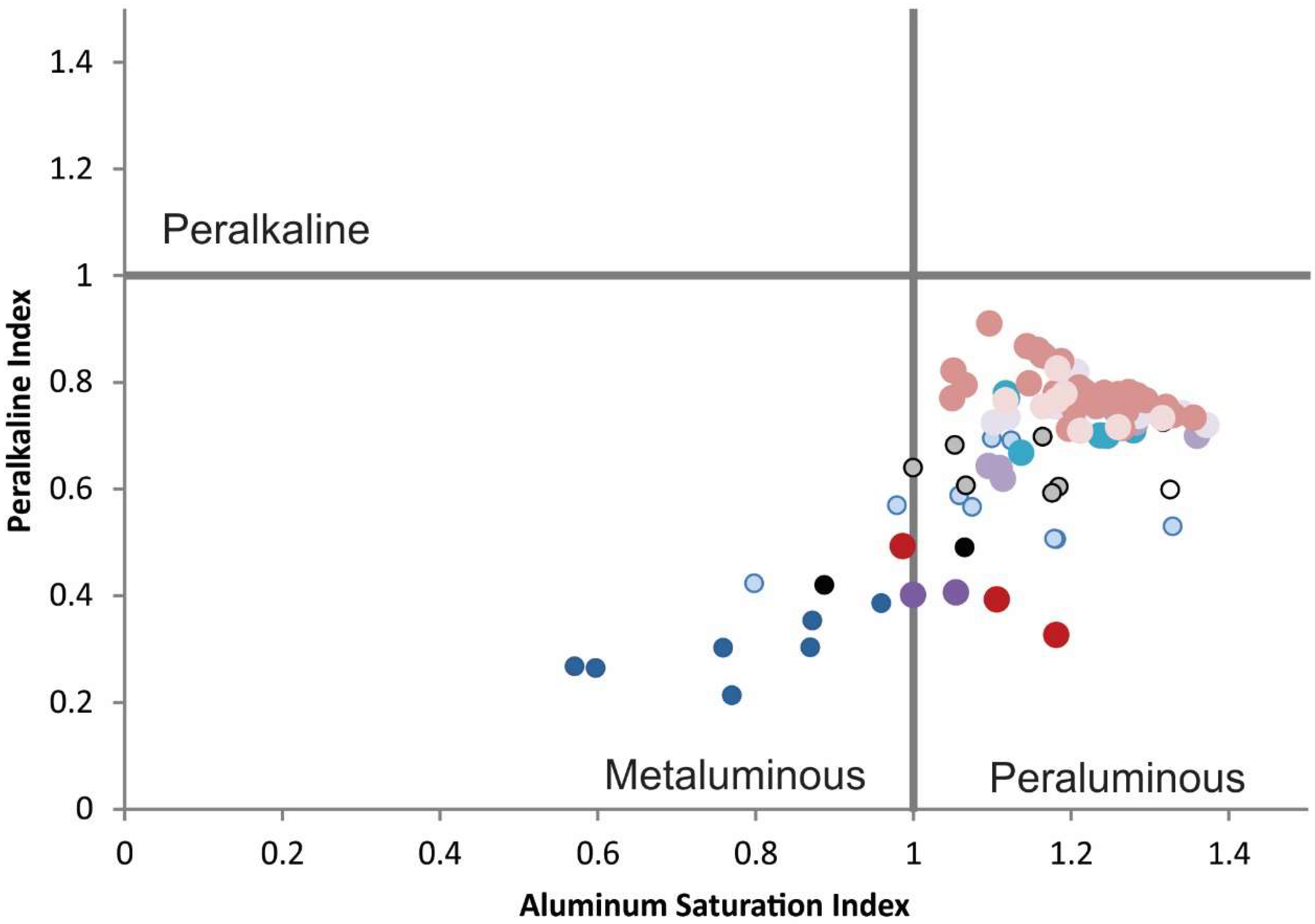
Figure 6), they are F-rich (up to 2.4 wt %), peraluminous with ASI ratios (Aluminum Saturation Index) around 1.2, but not peralkaline as is typical of many REE enriched systems (
Figure 7). Initial Sr
87/Sr
86 ratios for the mafic constituents of the magmatic suite are around 0.702 [
19], where values of felsic members are around 0.728 [
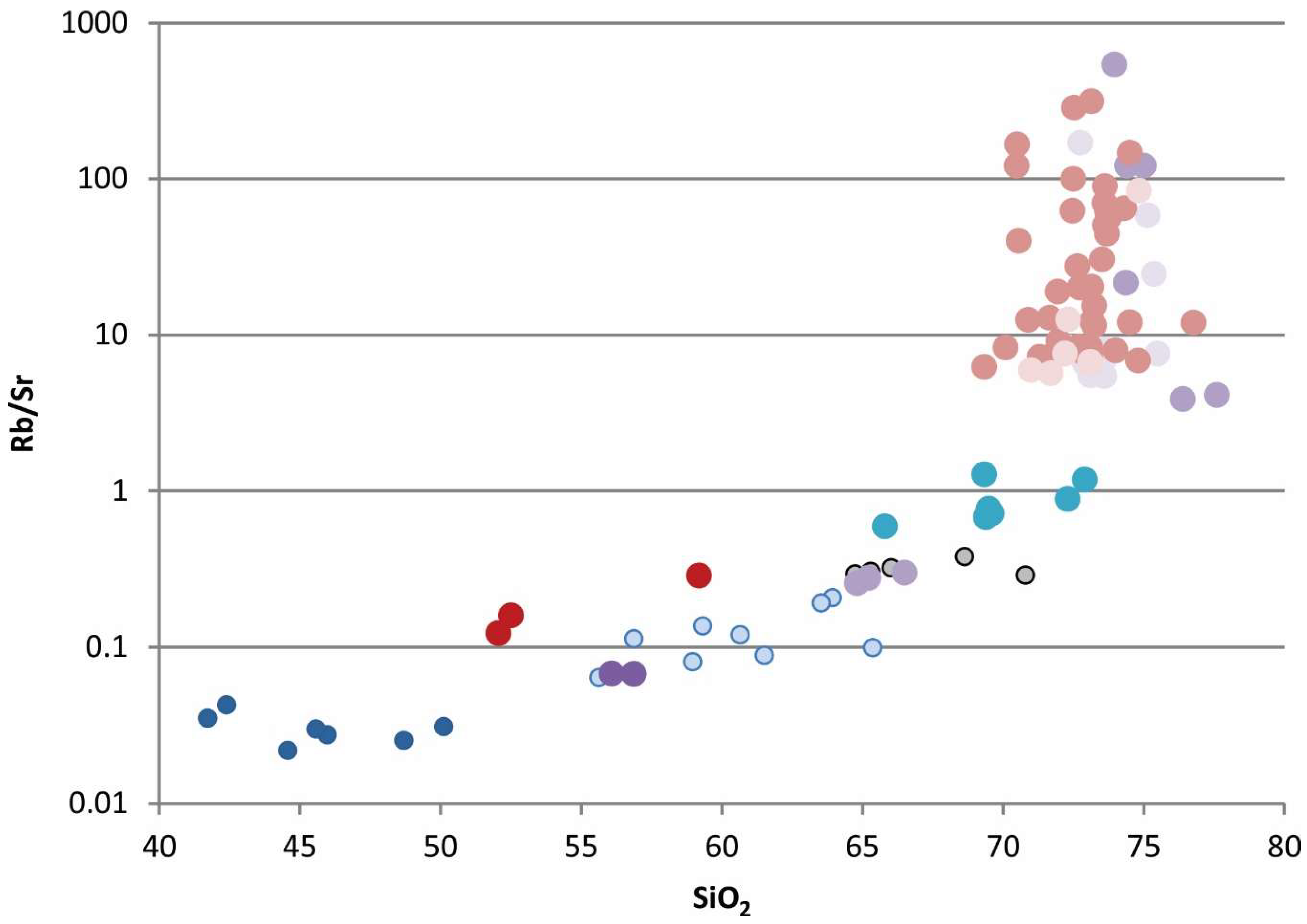
15]. These contrasting ratios imply a more crustal source for the felsic rocks. The Rb/Sr system appears to be affected by late- to post-magmatic processes increasing Rb relative to Sr (
Figure 8). The post magmatic process makes it difficult to rely on Sr in determining crustal contribution to the source of these magmas.
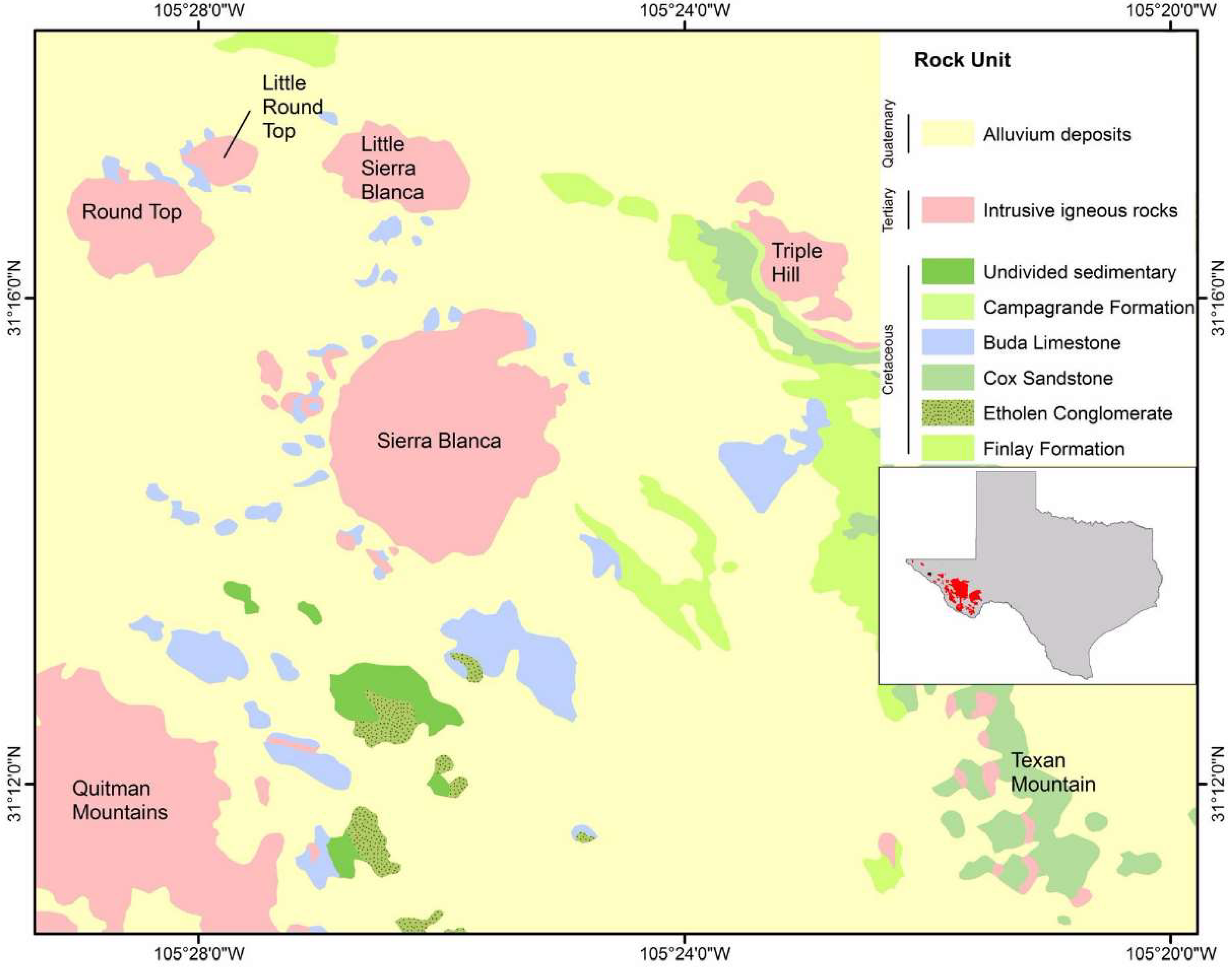
Each of the rhyolite laccoliths of the SBC and other regional magmatic rocks have distinct geochemical characteristics that help to discriminate them from each other [
14,
15,
19]. The Finlay Mountains represent nearby mafic to intermediate compositions (40–65 wt % SiO
2), conforming temporally and compositionally to early gabbro/diorite dikes in the immediate SBC area. The Texan Mountain rhyolite represents the lowest silica content from felsic rocks in the magmatic series (64–70 wt % SiO
2), followed by Triple Hill and Sierra Blanca with Round Top, Little Blanca and Little Round Top representing extremely evolved rhyolitic compositions (70–76 wt % SiO
2). These evolved compositions show an increase in Na
2O relative to K
2O, decreasing Al
2O
3 and very low MgO and P
2O
5 (
Figure 5).
Alkalinity Index versus Aluminum Saturation Index show that all of the igneous rocks in the SBC and the Finlay mountains are alkalic, and all but the more primitive Finlay mountains samples are peraluminous. Tectonomagmatic affinity diagrams including Ta vs. Yb suggest a within-plate tectonic setting for the rhyolites from the SBC (see
Figure 6), including the samples from Round Top. Rb/Sr ratio versus weight percent oxide of silicon shows a significant enrichment in rubidium for late stage, highly evolved rhyolites in the Sierra Blanca magmatic complex, as Rb fractionates preferentially into residual melts (
Figure 8).
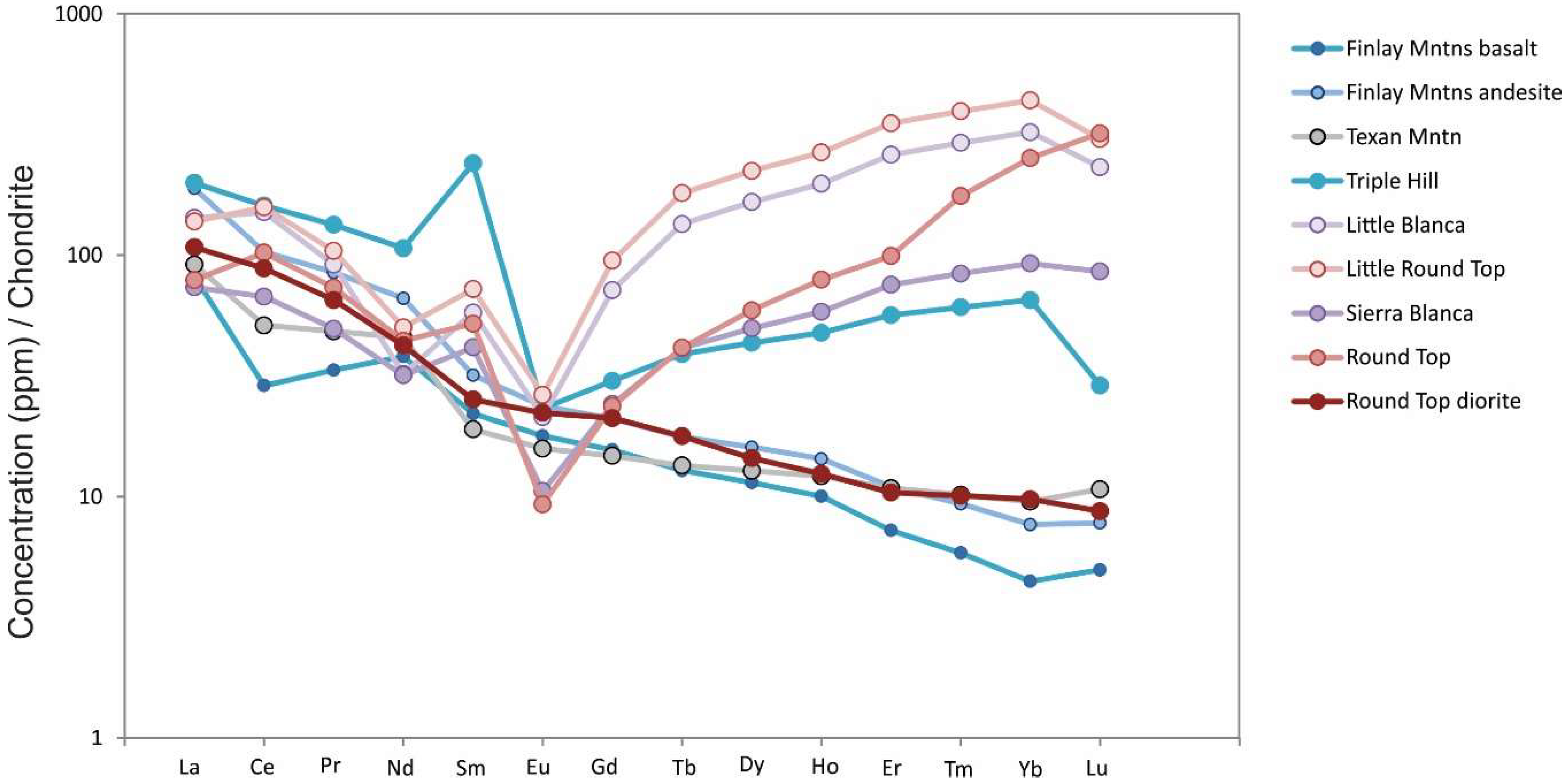
Rare earth element plots show LREE enrichment for felsic, intermediate and low SiO
2 rocks in the SBC. Mafic/Intermediate patterns show little to no Eu anomaly, but felsic members are enriched in HREEs and show a moderate to strongly negative Eu anomaly (Round Top, Sierra Blanca, Little Round Top and Little Blanca rhyolites;
Figure 9).
Finlay Mountain andesitic basalts, gabbroic/dioritic sills in the SBC area and Texan Mountain have LREE-enriched patterns with negligible Eu anomalies (~0.6) and La/Yb ratios ranging from 18–25, 11 and 10, respectively (
Table 3;
Figure 9). Triple Hill, Sierra Blanca and Round Top have increasing HREE, and total REE + Y, concentrations and pronounced negative Eu anomalies. Triple Hill samples have total REE + Y concentration of as high as 373 ppm, with average La/Yb ratio of 3 and Eu/Eu* of 0.09 (Eu/Eu* = Eu/[(Sm + Gd)/2]). Sierra Blanca and Round Top have total REE + Y values as high as 356 and 560, with average La/Yb ratios 0.79 and 0.31, and Eu/Eu* 0.19 and 0.14, respectively. Little Round Top and Little Blanca have similarly elevated HREE concentrations, with total REE + Y as high as 558 and 356 ppm, respectively. Average La/Yb ratios for Little Blanca and Little Round Top are 0.44 and 0.31, with average Eu/Eu* ratio of 0.22 (for both laccoliths).
4.3. Geochemical Modeling
The rhyolite of the Round Top laccolith does not have any direct source material indicators (autoliths, xenoliths, etc.) and identification of protolith compositions is determined from indirect geochemical and isotopic constraints, and petrogenetic modeling of probable regional source rocks. The Finlay Mountain andesitic to basaltic compositions represent earlier more primitive magmas that could be differentiated to form rhyolite similar to Round Top. The variation in Sr isotope ratios between mafic sills in the SBC (Sr
87/Sr
86 = 0.702) [
19] and Round Top (Sr
87/Sr
86 = 0.728) [
15], may be the result of late- to post-magmatic and hydrothermal processes that effected the Rb/Sr system. It is possible that crustal melts contributed, at least partially, to the initial melts that produced the Round Top rhyolite. Granulitic crustal rocks in the Trans-Pecos region provide possible source material at depth. Mafic, intermediate, and felsic xenolith compositions have been identified in the Trans-Pecos region and represent crustal lithologies [
24].
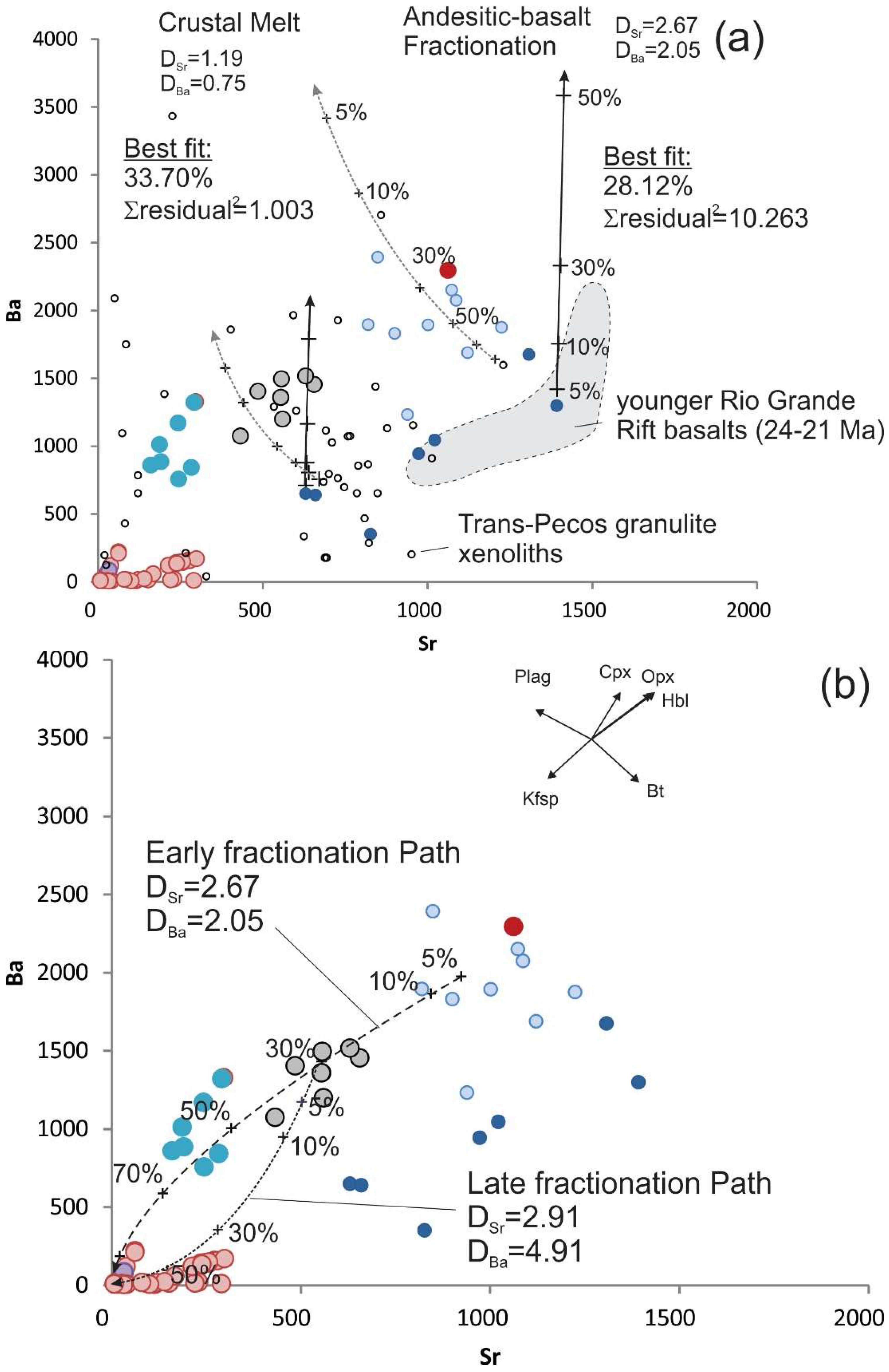
The petrogenetic model evaluates starting melts of a composition similar to the andesitic basalts of the Finlay Mountains, differentiated by closed-system Rayleigh fractional crystallization (FC) to produce early Sierra Blanca Complex melts—compositions similar to Texan Mountain and Triple Hill, and crustal melts represented by xenoliths from the Trans-Pecos region (
Figure 10a). Solid lines represent the differentiation sequence of a range of regional basaltic compositions, dotted line represents batch melting (BM) of a range of regional crustal xenolith sources. Both products of regional melted crust and differentiated mafic magma compositions can produce appropriate melts similar to the mafic diorite sills that precede the Round Top rhyolite in the Sierra Blanca Complex, though major element least sum-residual-squared regression [
25] of Tran-Pecos crustal xenolith (Σrisidual
2 = 1.003 at 33.70% melt) and Finlay Mountain andesitic-basalt (Σr
2 = 10.265 at 28.12% fractionation) mineralogies suggests a crustal melt is more appropriate for the rhyolite source.
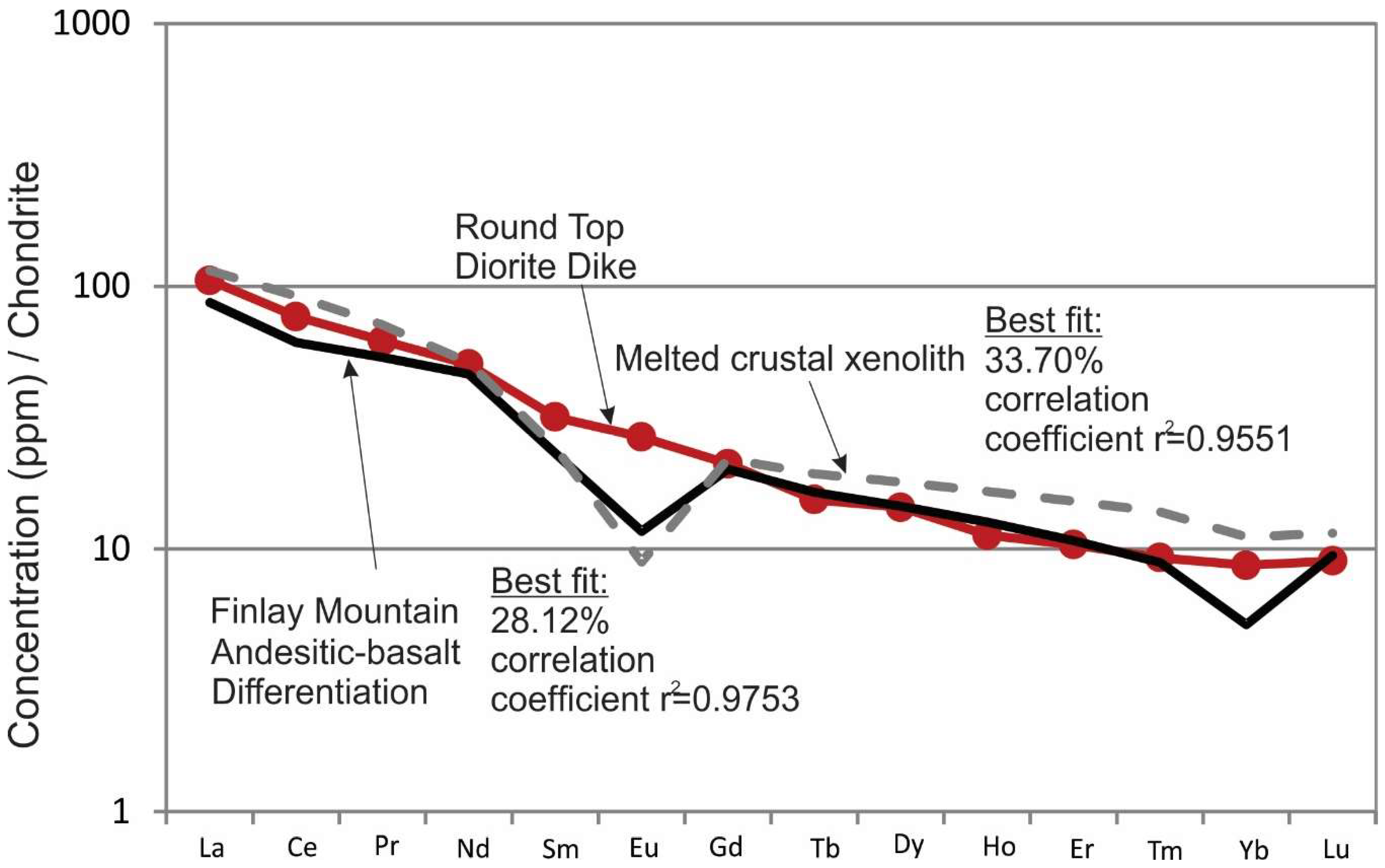
The sum residual squared from differentiated andesitic basalt, or melted crust, shows good correlation when compared with rare earth patterns for Round Top diorite compositions (i.e., The diorite compositions correlate well with melted xenolith (r
2 = 0.9551) and fractionated basalt compositions (r
2 = 0.9753);
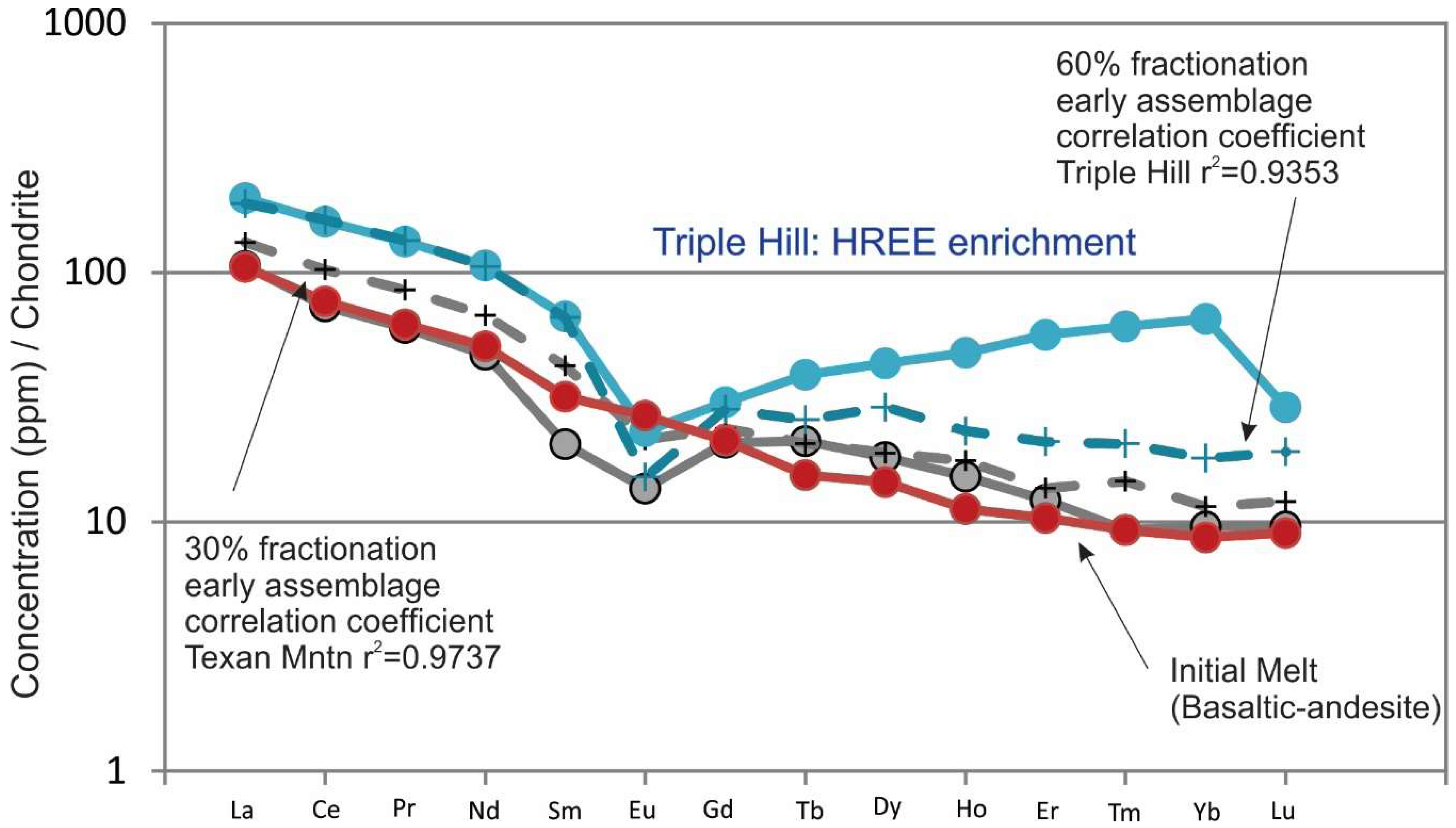
Figure 11). The FC of the observed early phenocryst assemblage from melt products in the melting model produce compositions similar to Texan Mountain and Triple Hill at 60% FC (
Figure 10b and
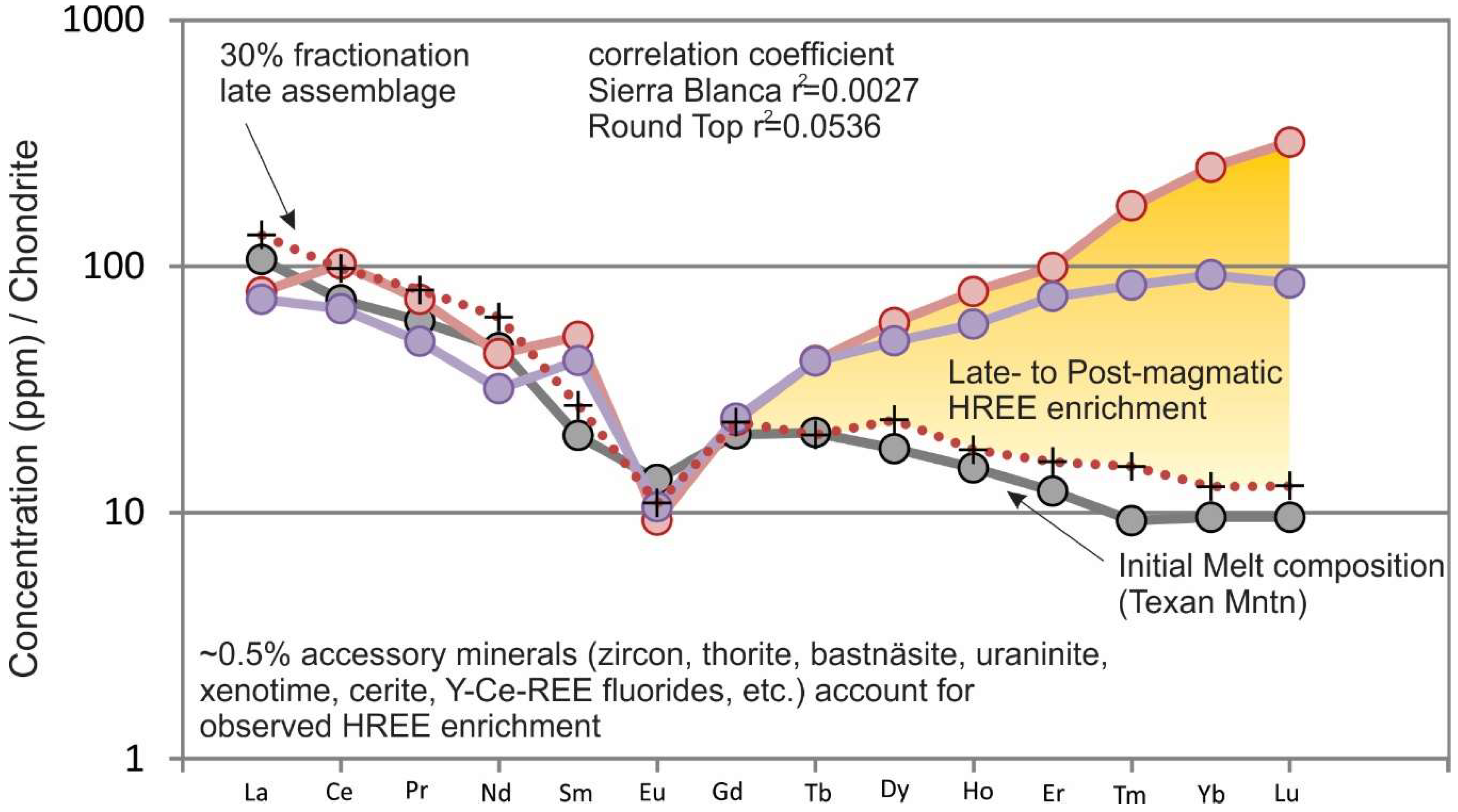
Figure 12). Melt compositions similar to Texan Mountain fractionating observed mineral phases become the highly evolved compositions of Round Top, Sierra Blanca, Little Round Top and Little Blanca between 50% and 90% FC (
Figure 10b and
Figure 13). The REE model correlates well with early FC modeling, but HREE enrichment starts to effect correlation (r
2 = 0.9353) when modeling compositions from Triple Hill through the most evolved compositions, even though LREEs fit very well.
The significant HREE enrichment in Round Top and Sierra Blanca are difficult to account for with the observed late stage crystallizing phases. A model that incorporates just 0.5% of REE bearing accessory minerals known to be present, but in the matrix as pervasive micro to nano-crystalline crystals, accounts for the observed enrichment. The HREE enrichment is occurring at the transition between late stage magmatic crystallization (with an exsolving vapor phase) and alteration/precipitation related to high-temperature hydrothermal fluids, but with high enough fluorine to retain a large amount of silicate melt component in the fluid. The Round Top rare earth geochemistry is modeled and compared to similar highly evolved systems (Kymi complex in southern Finland [
28], Erzgebirge and adjacent Fichtelgebirge and Oberpfalz rocks in Germany [
29,
30]) to understand the timing and processes that form economically viable rare earth element deposits in highly evolved magmatic rocks (
Figure 14,
Figure 15 and
Figure 16).
Irber [
30] discusses quantification of the tetrad effect and ligand dependent differences in the ionic radii of REEs [
32]. The variation in ionic radii are based on variable expansion of the electron cloud (nephelauxetic- or tetrad-effect), type of complexing and internal atomic structure. The inference is that the tetrad effect can be generated during REE partitioning at the transition between silicate melt and high-temperature hydrothermal systems, or between coexisting melt, aqueous high-temperature late stage fluid and crystallizing minerals, in both cases with strong complexing of fluorine and REE.
The tetrad-effect integrates a quantitative measurement of the observed variation in La–Nd, Sm–Gd, Gd–Ho, and Er–Lu trends. The tetrad effects
TE1,3 are quantified using chondrite normalized values in Irbers equation [
30],
TE1,3 = (
t1
× t3)
1/2, where
t1
= (Ce/Ce
t × Pr/Pr
t)
1/2 and
t3
= (Tb/Tb
t × Dy/Dy
t)
1/2; Ce/Ce
t = Ce
CN/(La
CN2/3 × Nd
CN1/3), Pr/Pr
t = Pr
CN/La
CN1/3 × Nd
CN2/3), Tb/Tb
t = Tb
CN/(Gd
CN2/3 × Ho
CN1/3) and Dy/Dy
t = Dy
CN/(Gd
CN1/3 × Ho
CN2/3). Trace element ratios and tetrad calculations help to determine if the system mineralization was largely charge and radius controlled (CHARAC), derived from magmatic hydrothermal fluids, or transitional non-charge and radius-controlled ligand complexing processes (Non-CHARAC) [
30,
31,
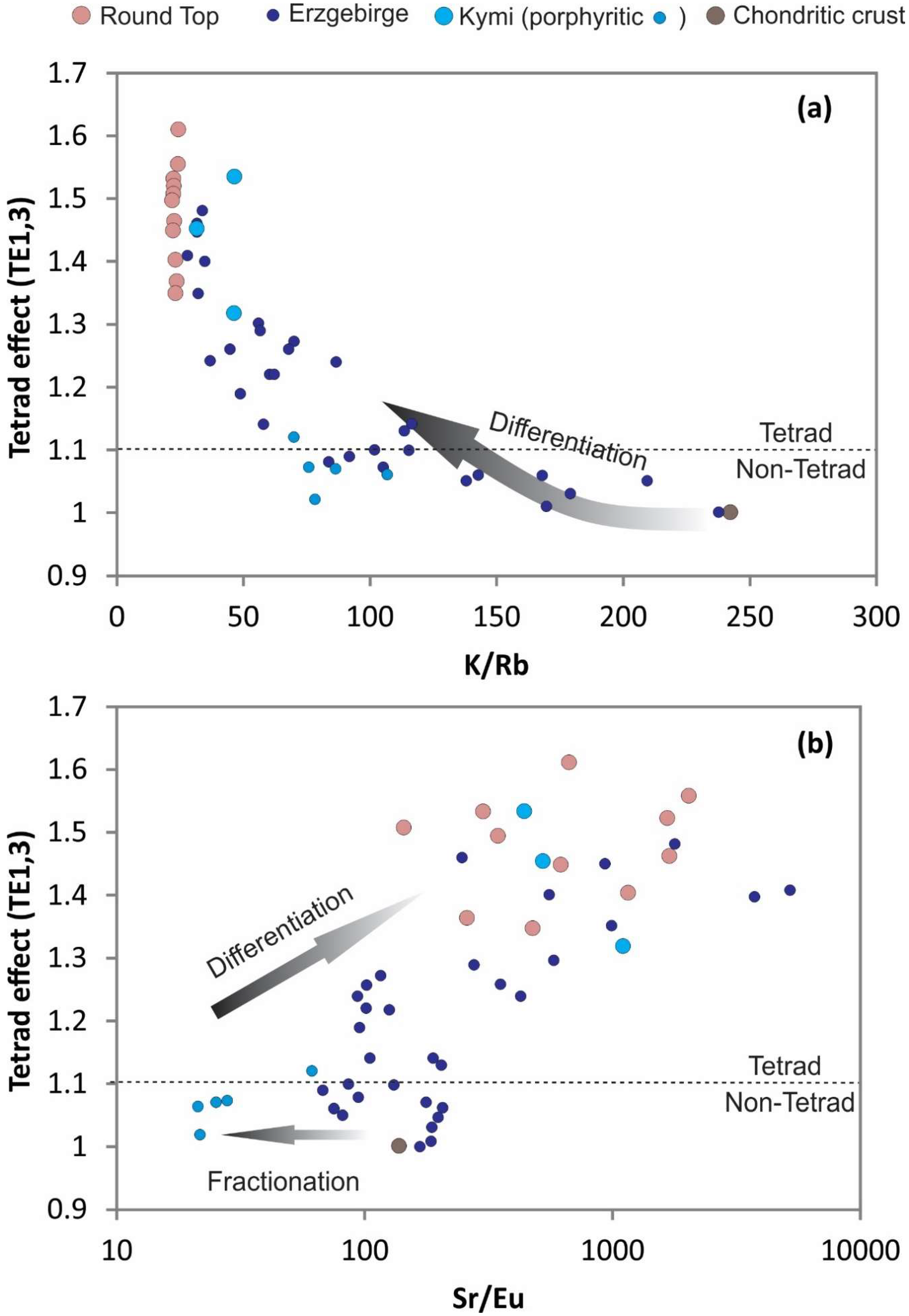
33]. Rb preferentially fractionates into residual melt, and extremely low K/Rb ratios (<50) commonly indicate interaction with an aqueous fluid phase (
Figure 14a). Eu and Sr should have similar behavior in granitic systems. The observed decoupled behavior, where Sr is elevated relative to Eu, is a result of strong Eu depletion with early feldspar crystallization (
Figure 14b). Zr/Hf ratios shift toward lower ratios with more evolved compositions of silicate melts. Round Top ratios represent increased Hf in late stage zircons transitioning to very low Zr/Hf ratios of pegmatitic systems (e.g., [
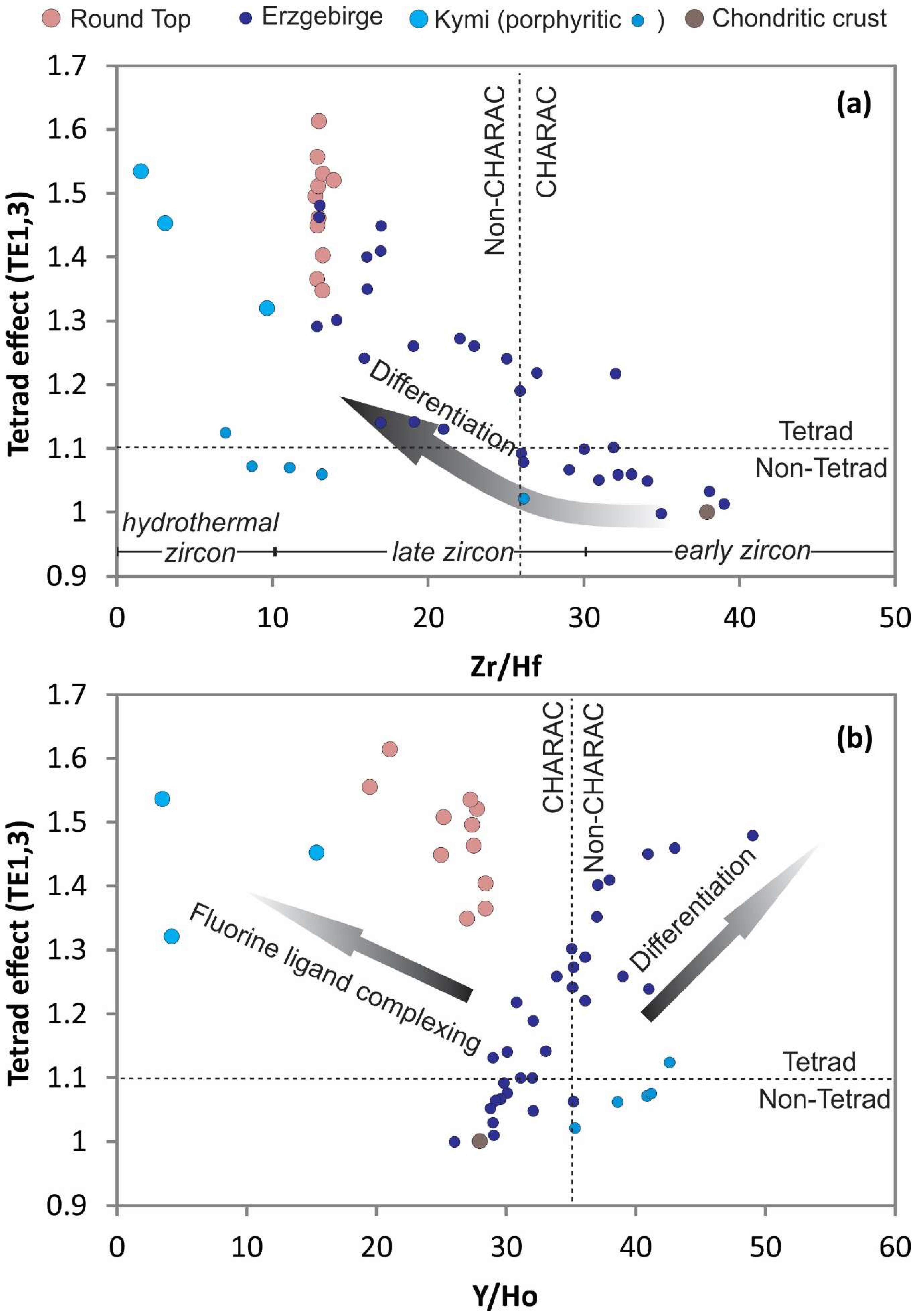
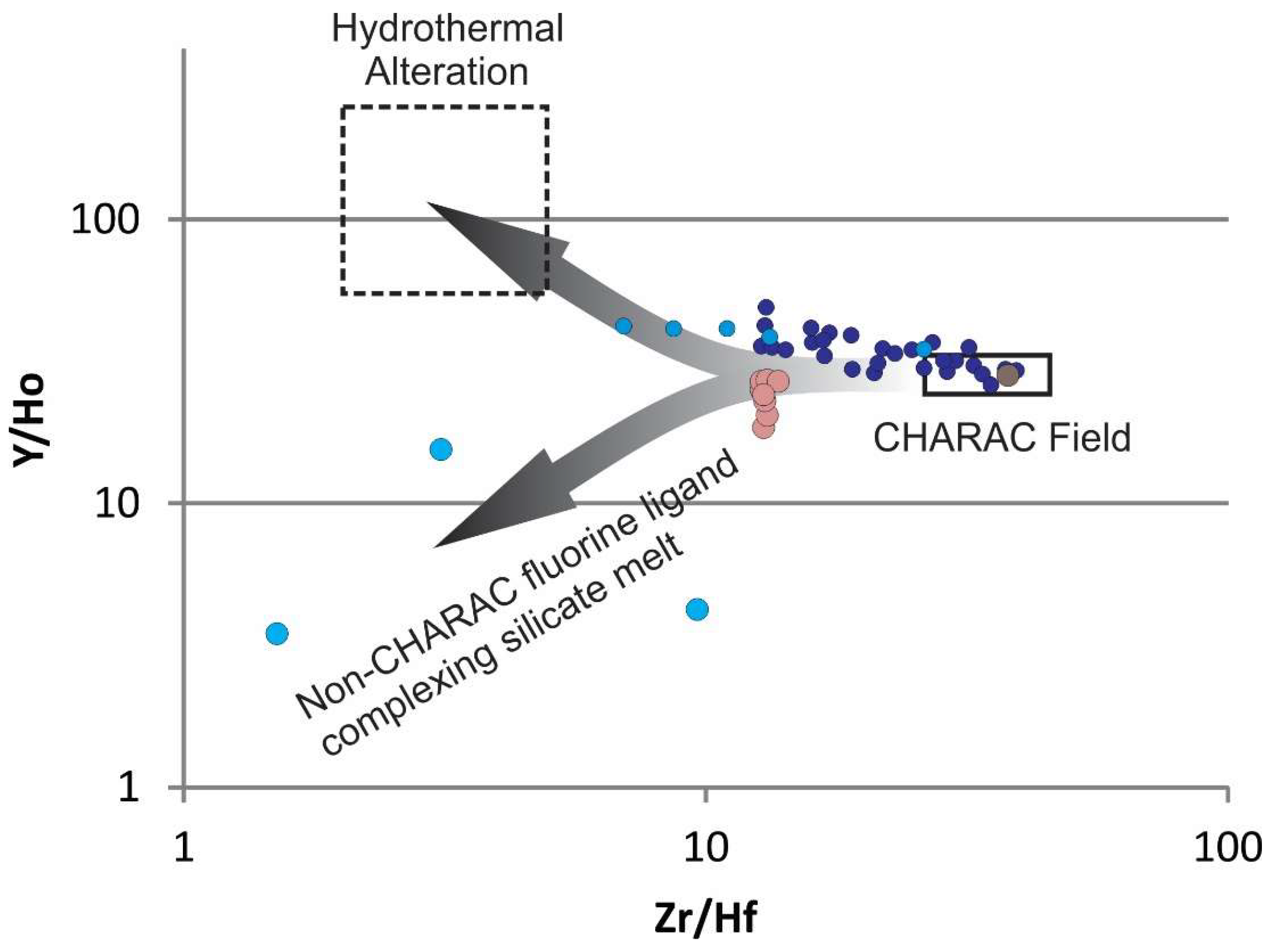
34]). Y/Ho ratios have near chondritic values, but only coincidently due to an original path of differentiation and non-CHARAC behavior followed by fluorine ligand complexing in late silicate melt before aqueous fluid transition. Y/Ho ratios combined with Zr/Hf ratios show that the Round Top samples do not have the elevated Y and Hf relative to Ho and Zr, respectively, that would be typical of hydrothermal alteration processes in these types of rocks (cf. [
35];
Figure 15 and
Figure 16). Y and HREEs were retained in the melt due to the F-rich nature of the magmas (cf. [
36,
37,
38]).
The observed mineral textures trend toward late melt phase REE mineralization in a rapid nucleation environment with a moderate to high degree of undercooling. Geochemical modeling suggests that the Round Top rhyolite HREE enrichment is a result of thorough and long-lived differentiation processes in a fluorine-rich magmatic system, where fluorine complexing with HREE and similar trace elements was pervasive and transitional between low temperature silicate melt and magmatic hydrothermal fluids.