1. Introduction
Yttrium oxide doped with trivalent europium (Y
2O
3:Eu) phosphor—which have the characteristics of optics, electricity, and magnetism—are unsurpassed and non-renewable strategic resources, and have been extensively used in the field of electronic information, energy environment, petrochemical, and metallurgical machinery, especially in rare earth luminescent materials. With the popularization of fluorescent lamps, computers, mobile phones, and other related products, pollutive waste fluorescent powder is sharply increasing. According to statistics, as much as eight thousand tons of waste fluorescent powders were produced in China in 2010; additionally, according to the current market value, rare earth resources in waste fluorescent powder are estimated to be about four billion RMB in value [
1]. If the waste fluorescent powders were recycled efficiently, not only could it reduce the dwindling quantity of rare earth mineral, but it can also be fused to the industrial chain of rare earth recovery, which could greatly improve the utilization of rare earth resources.
In recent years, recycling of rare earth elements from waste fluorescent phosphors has been researched by many foreign and domestic researchers. Tsuyoshi Hirajima et al. [
2] used collecting agents and dispersants to separate rare earth trichromatic fluorescent phosphors by flotation method. Hirajima et al. [
3] used diazomethane as the layered medium and sodium oleate as a surfactant to recycle rare earth fluorescent phosphors by centrifugal separation method. Akira Otsuki et al. [
4] adopted two-step liquid–liquid extraction methods to separate red, green, and blue rare earth fluorescent phosphors. Youming Yang et al. [
5] used hydrochloric acid and hydrogen peroxide to leach red powders, and adopted the sodium carbonate roasting method to leach green and blue phosphors. Guangjun Mei et al. [
6] recovered Y
2O
3 from waste fluorescent powders by the process of leaching, precipitation, and roasting. Hongmin Cui et al. [
7] utilized Aliquat 336 functionalized chelating adsorbent derived from chitosan to enrich and separate Y(III) from diluted solutions. However, rare earth elements are not effectively retrieved by these methods. The above methods are used with mixing power which has pure red, green, and blue phosphors, but waste fluorescent powder in China is obtained by the crushing method.
For the synthesis of Y
2O
3:Eu nanoparticles, several processes such as the gas-phase condensation technique, sol–gel route, polyol method, spray pyrolysis, and the hydrothermal method were studied by researchers [
8]. Jitao Chen et al. [
9] reported the self-assembled Y
2O
3:Eu
3+ 3D architectures synthesized by a simple hydrothermal process, followed by calcination treatment. They found that some of the nano lamellas wrinkled into the petals of flowerlike architectures, which may be potentially applied in optoelectronics due to their strong red emission. Guowei Chen et al. [
10] synthesized Y
2O
3:Eu phosphor powder via a facile Pechini sol–gel method using pluronic F127 as a template agent. Under the excitation of 245 nm UV radiation, the mesoporous Y
2O
3:Eu particles show stronger PL intensity than that of particles without special structure, due to the advantage of surface area and pores. Arunkumar Paulraj et al. [
11] compared the hydrothermal method with the combustion method and solid-state metathesis reaction.
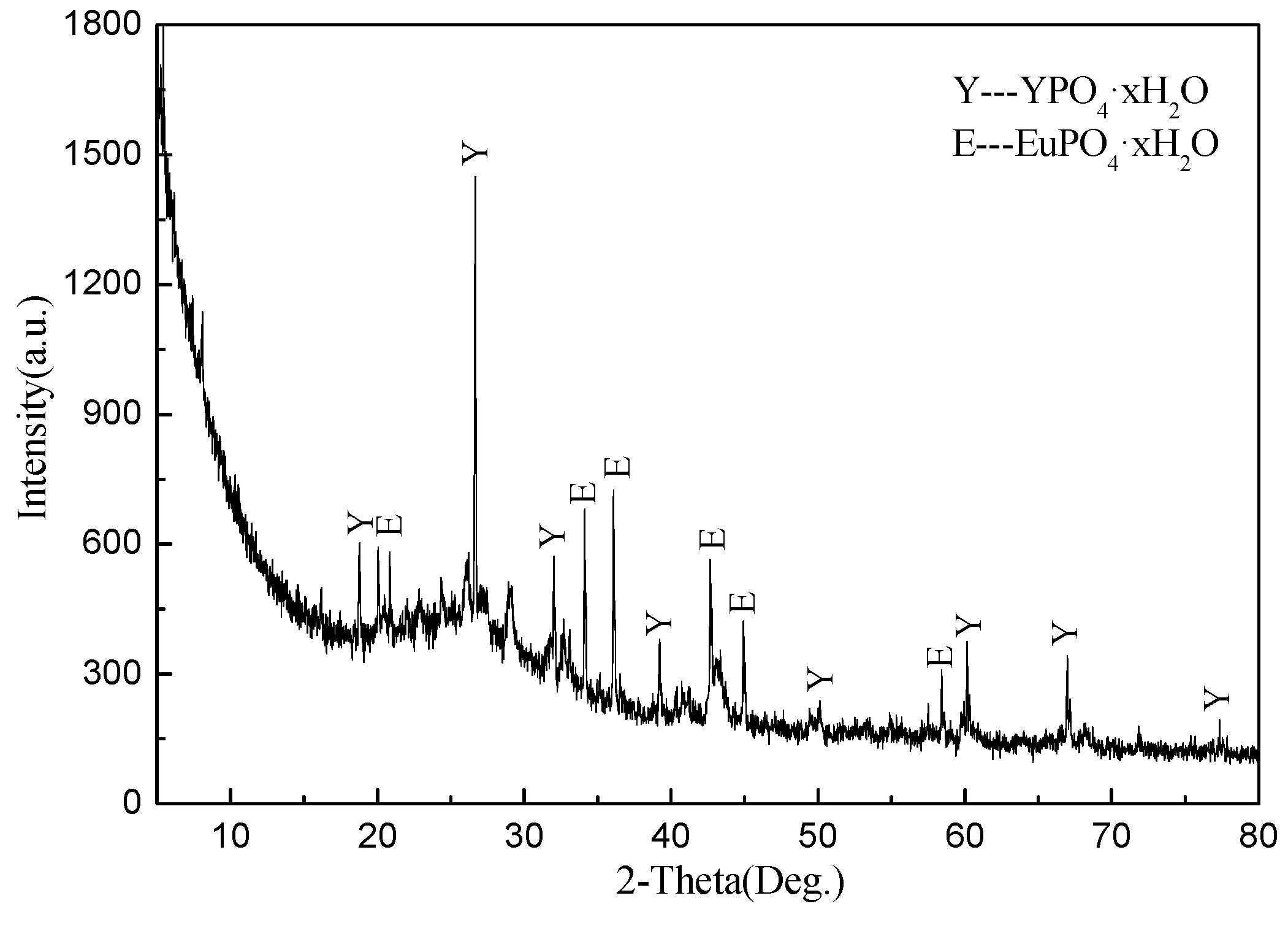
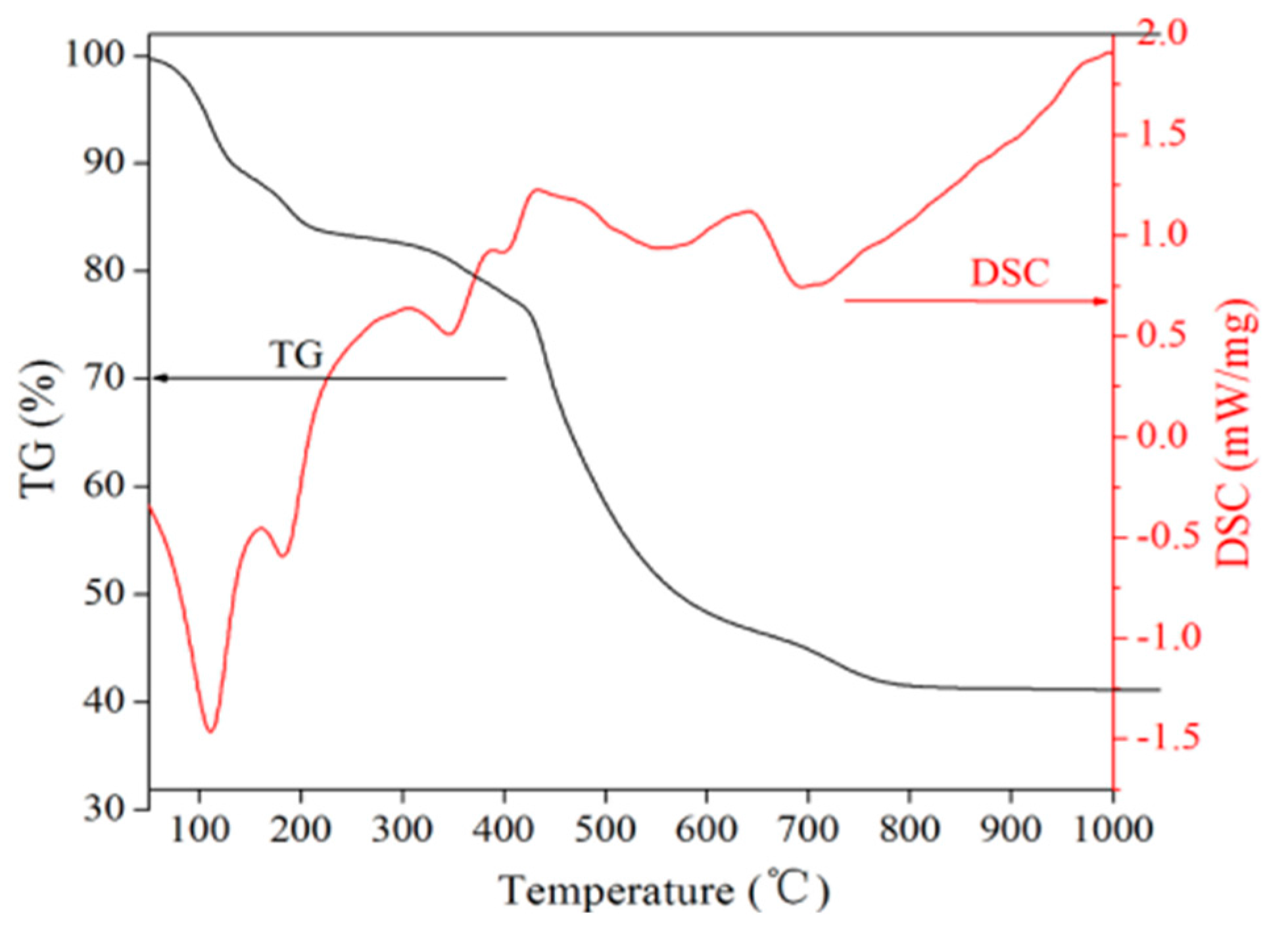
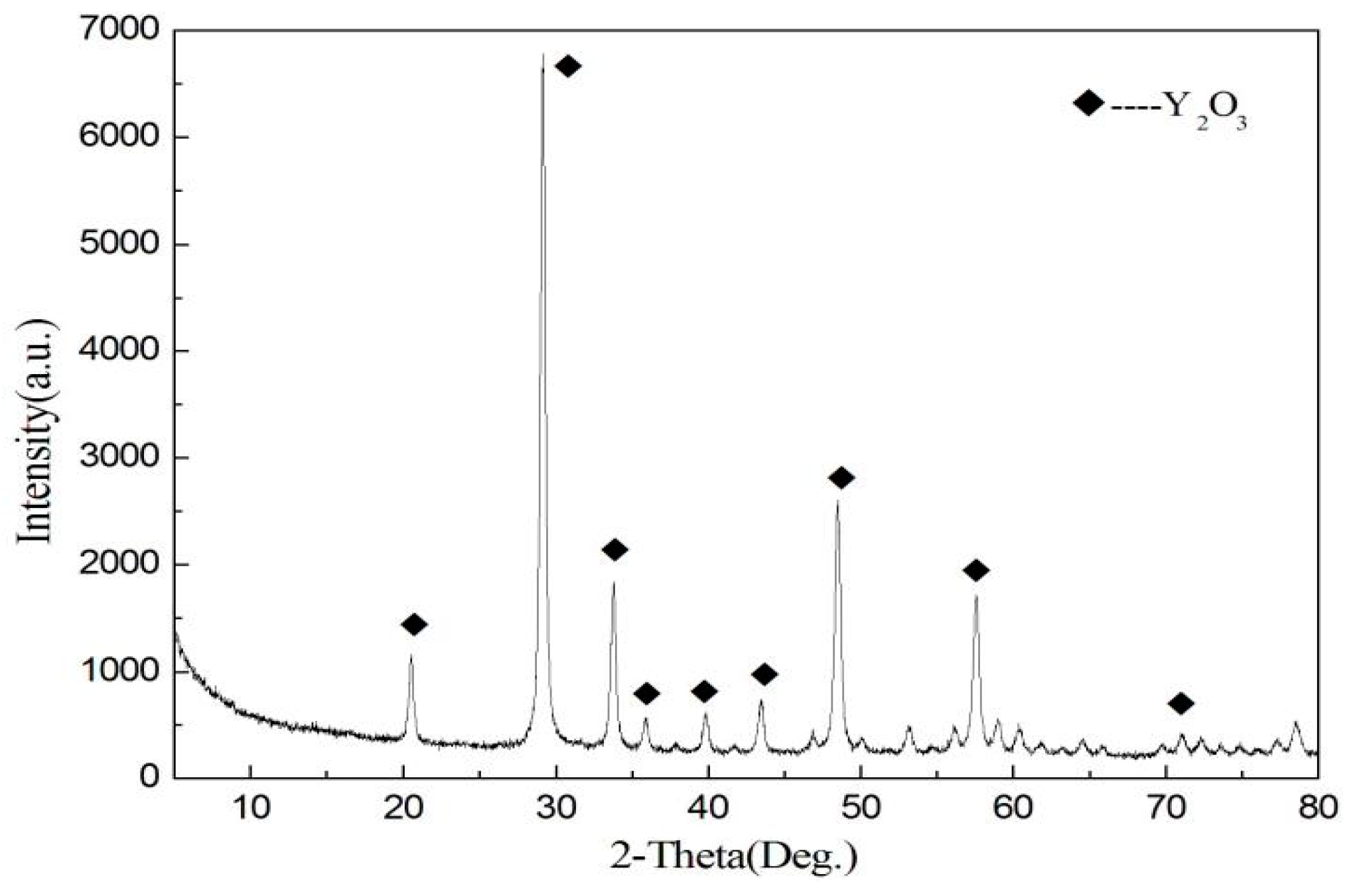
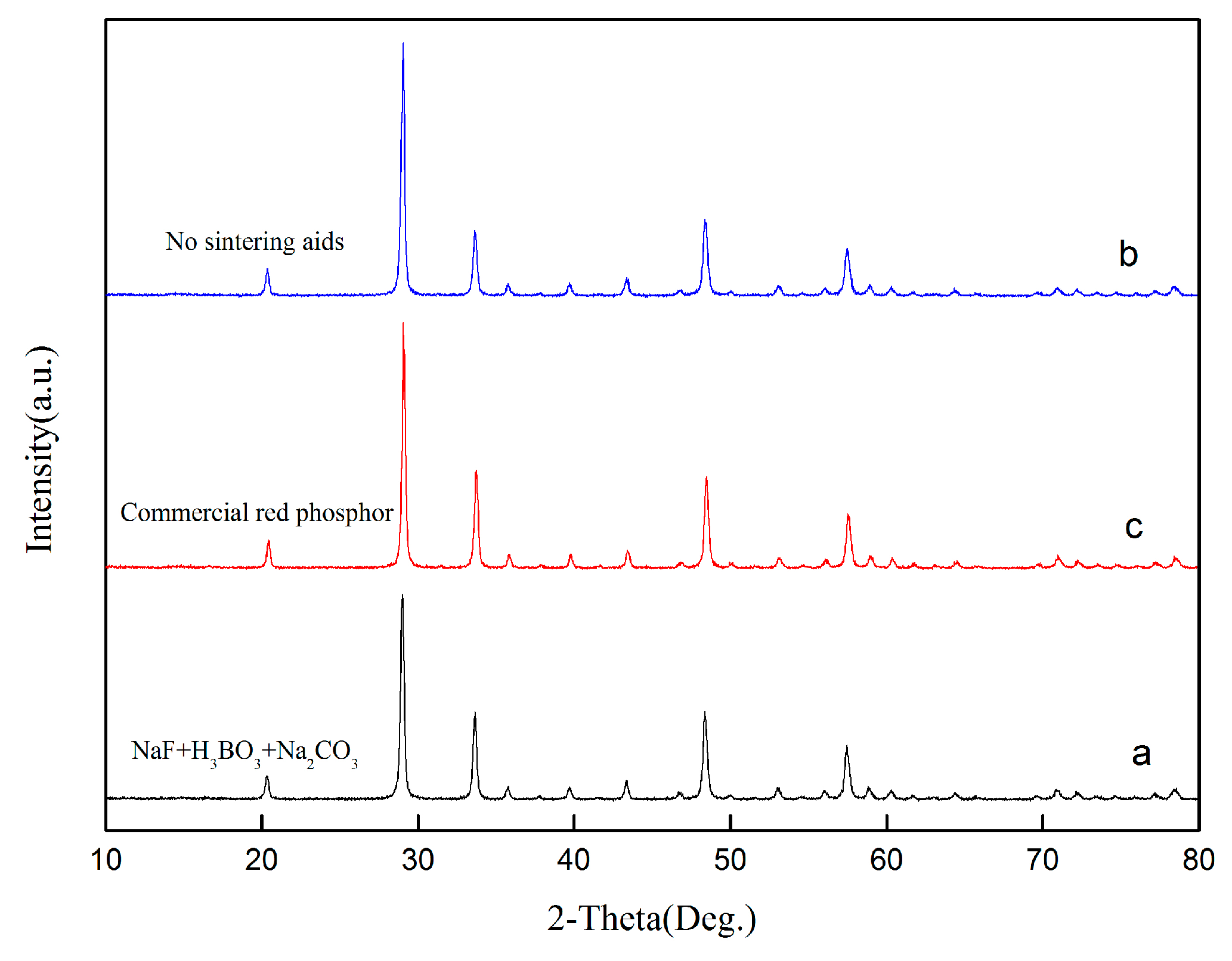
In this paper, recycling and synthesis of high-purity rare earth oxides (Y2O3:Eu) from waste fluorescent powder were studied. Compared to previous work, the innovation of this work is that the purification step was added to the process, with ammonia, sodium sulfide, and Sodium diethyldithiocarbamate trihydrate (DDTC), before precipitation with oxalic acid, which will reduce the amount of oxalic acid. After annealing of the precursor at 1400 °C, we obtained the materials of Y2O3:Eu, which have excellent luminescent properties. The products were characterized by X-ray diffraction (XRD), scanning electron microscope (SEM), and fluorescence spectrometer.
4. Conclusions
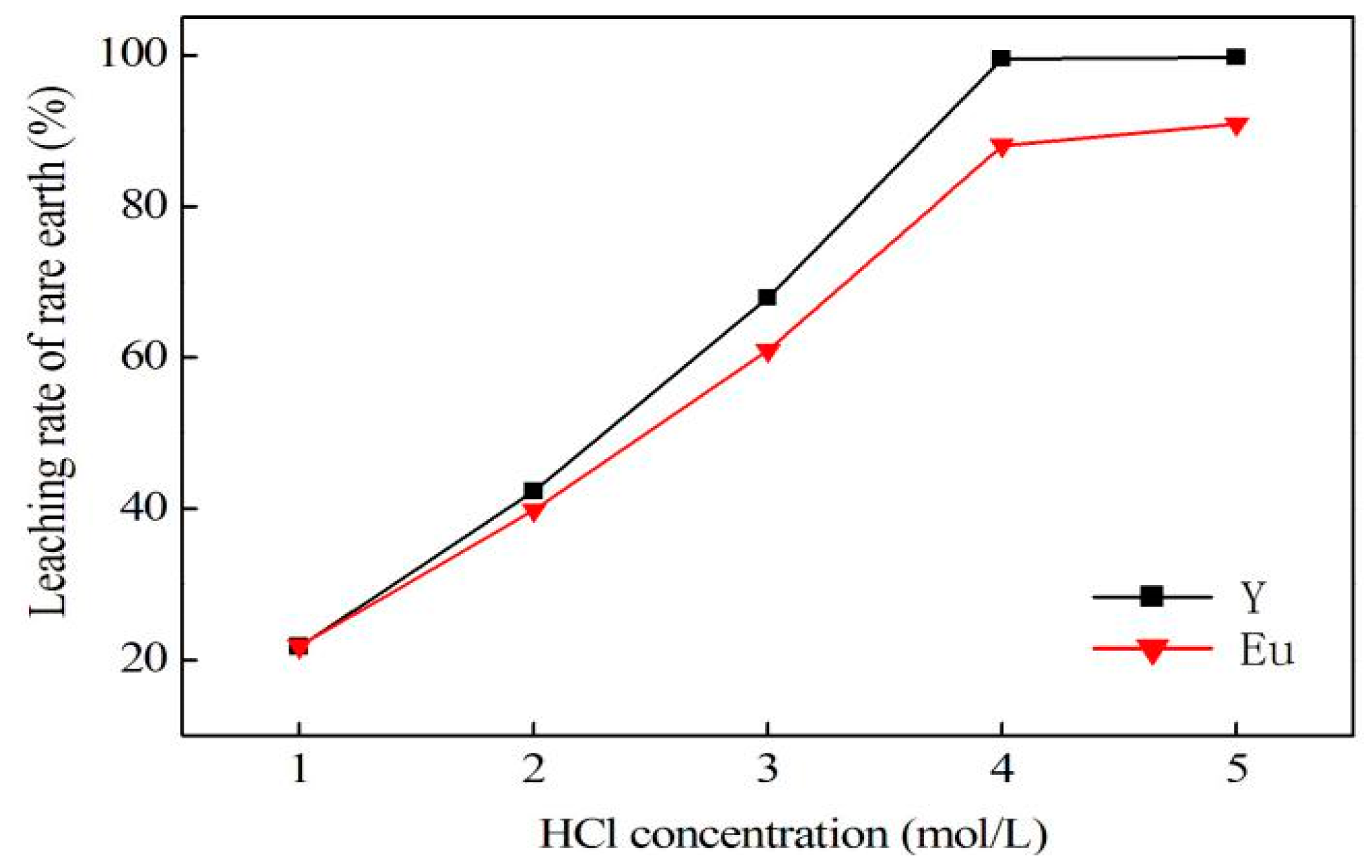
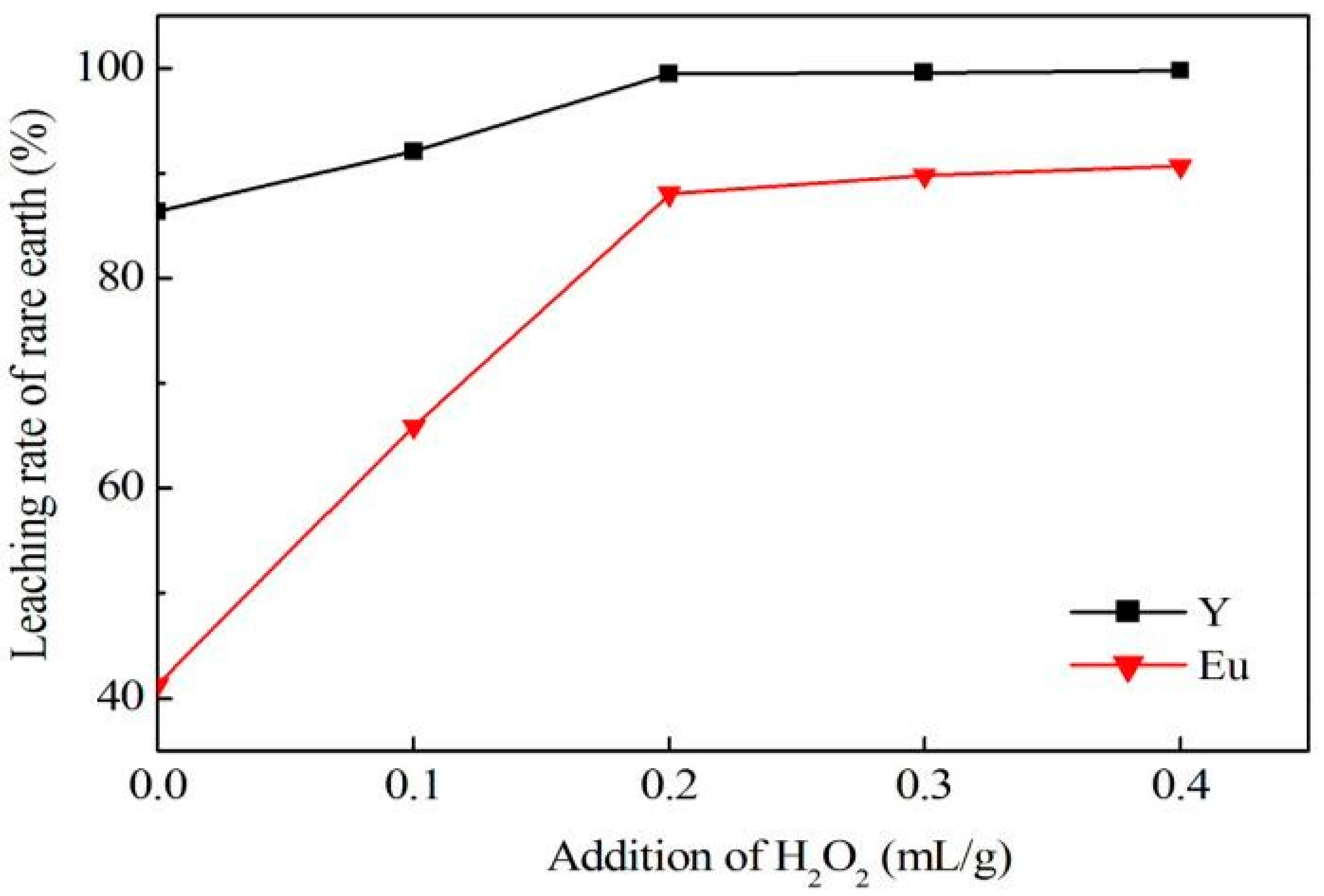
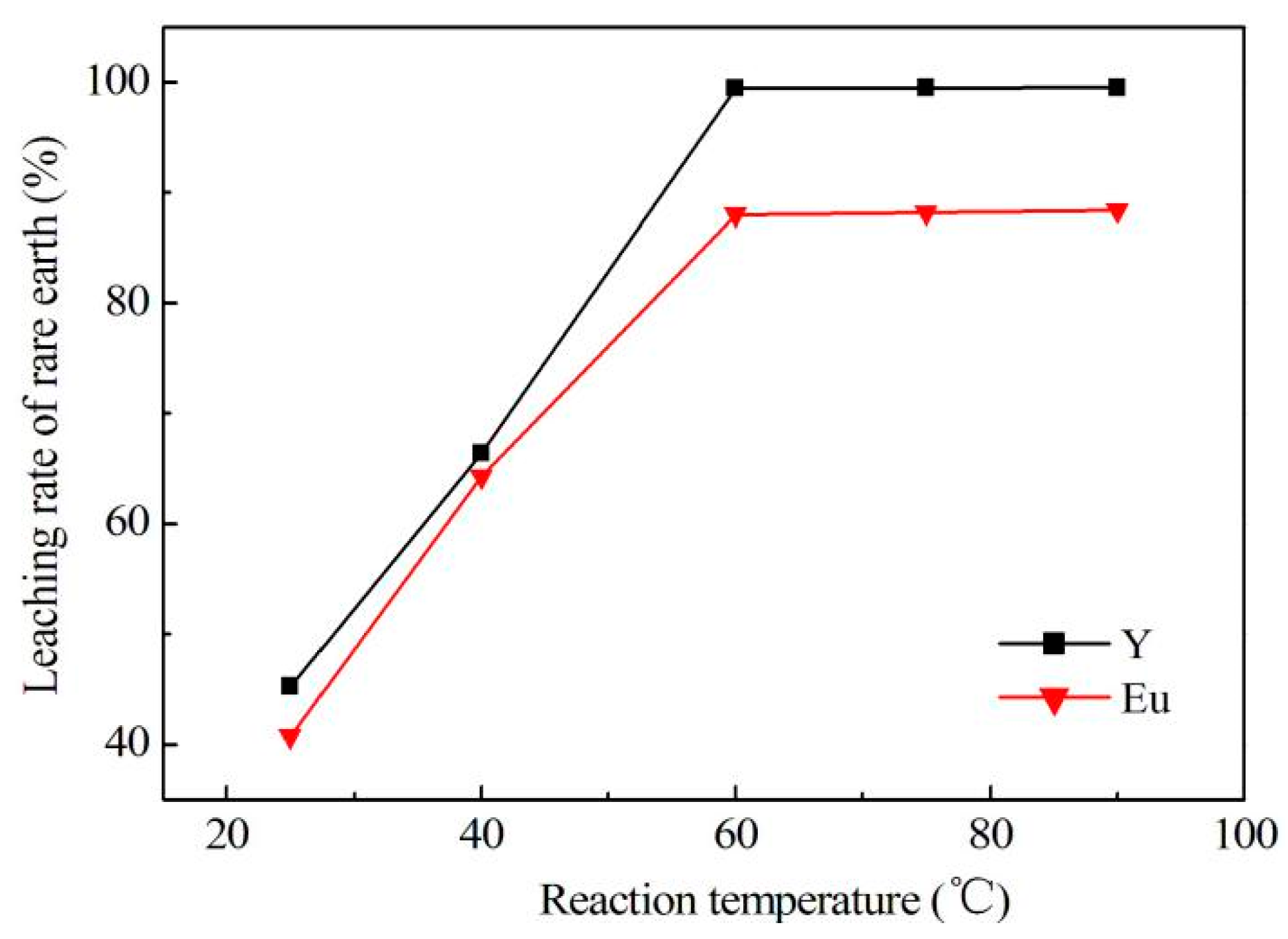
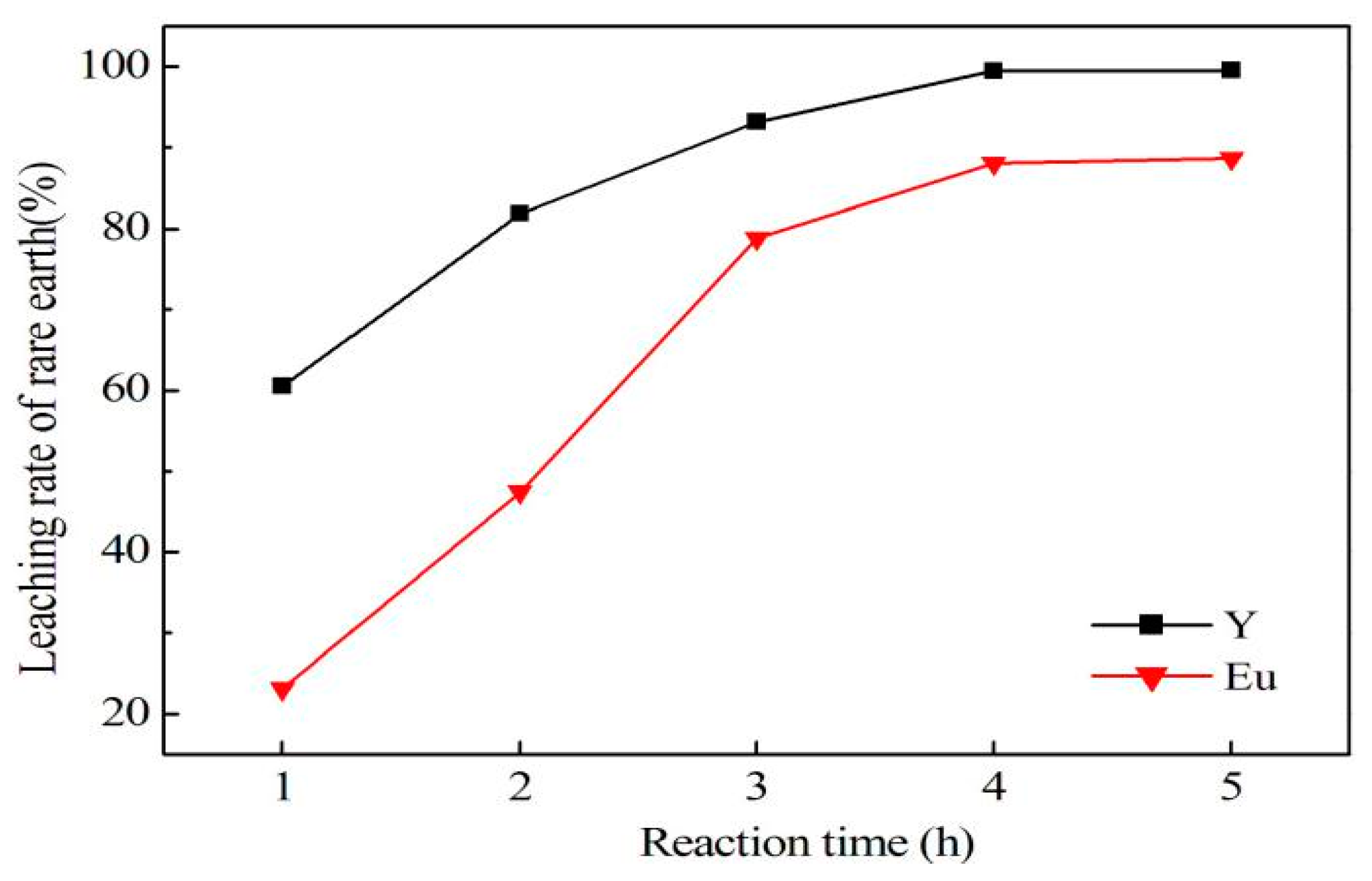
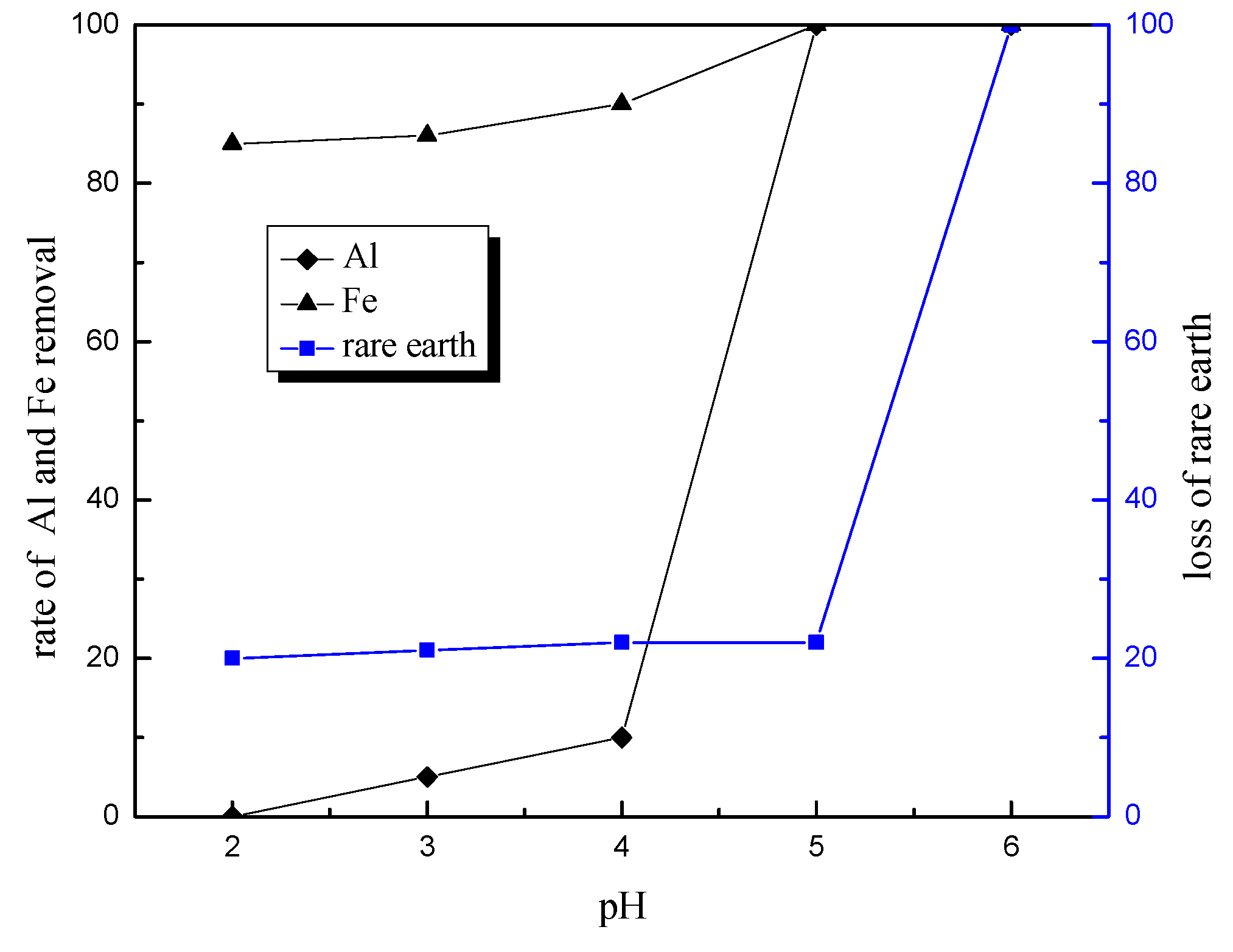
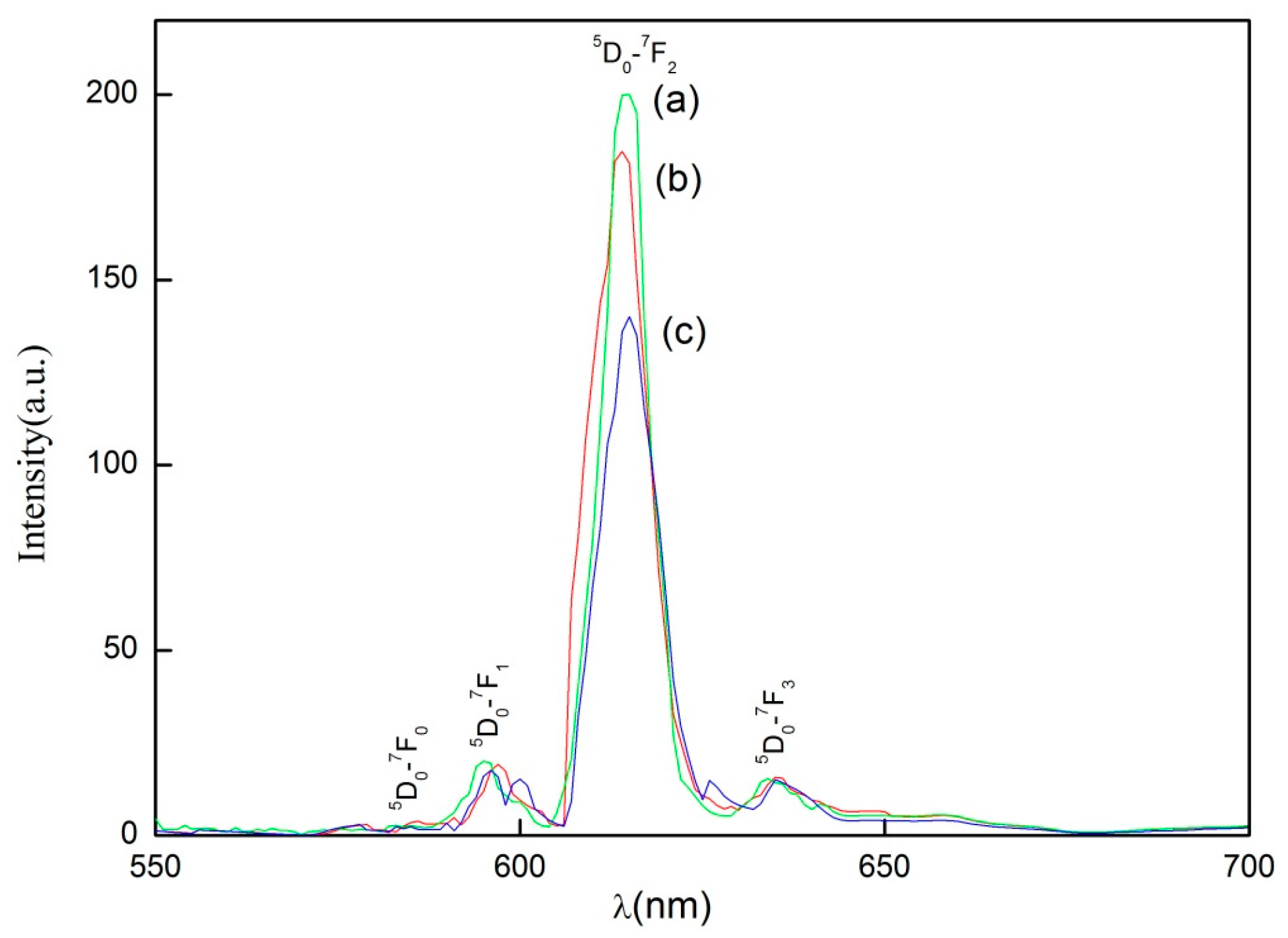
In conclusion, this research describes the recycling and synthesis of Y
2O
3:Eu from waste fluorescent lamps. The process includes leaching tests; impurities removal from leaching solution using ammonia, sodium sulfide, and DDTC; precipitations of rare earth using oxalic acid; and synthesis of Y
2O
3:Eu by high temperature solid-state synthesis method. The leaching tests showed that the optimum condition to leach rare earth was: 4.0 mol/L of hydrochloric acid, 0.2 mL/g of hydrogen peroxide, 7.5 mL/g liquid–solid ratio, 60 °C, and 4 h of reaction. This study is mainly focused on the purification process aimed at optimization of experimental conditions for removal of impurities, as well as reduction of rare earth loss during this procedure. Tests showed that the best way to remove impurities is by adjusting pH to 4.0 using ammonia, and adding sodium sulfide and DDTC to control pH up to 5.0. In addition, Yongsong Liu et al. [
17] found that the morphologies have a great effect on photoluminescence property, and that the microsphere Y
2O
3:Eu
3+ exhibit the strongest red emission intensity among structures obtained in the experiments. M.K. Devaraju et al. [
8] put forward that for successful applications, the phosphor particles should have a spherical shape and high luminescence. Phosphor particles with a spherical shape are capable of minimizing light scattering on their surface. Thus, in this paper, because of the smooth surface and ellipsoid morphology, the materials of Y
2O
3:Eu synthesized by high temperature solid-state synthesis method at 1400 °C with sintering aids exhibited higher PL intensity at 610 nm under the excitation of 254 nm, when compared to those synthesized without sintering aids. This result may provide helpful guidance for the application of phosphors.