Reducing the Entrainment of Gangue Fines in Low Grade Microcrystalline Graphite Ore Flotation Using Multi-Stage Grinding-Flotation Process
Abstract
:1. Introduction
2. Material and Methods
2.1. Test Samples and Reagents
2.2. Methods
2.2.1. Proximate Analysis
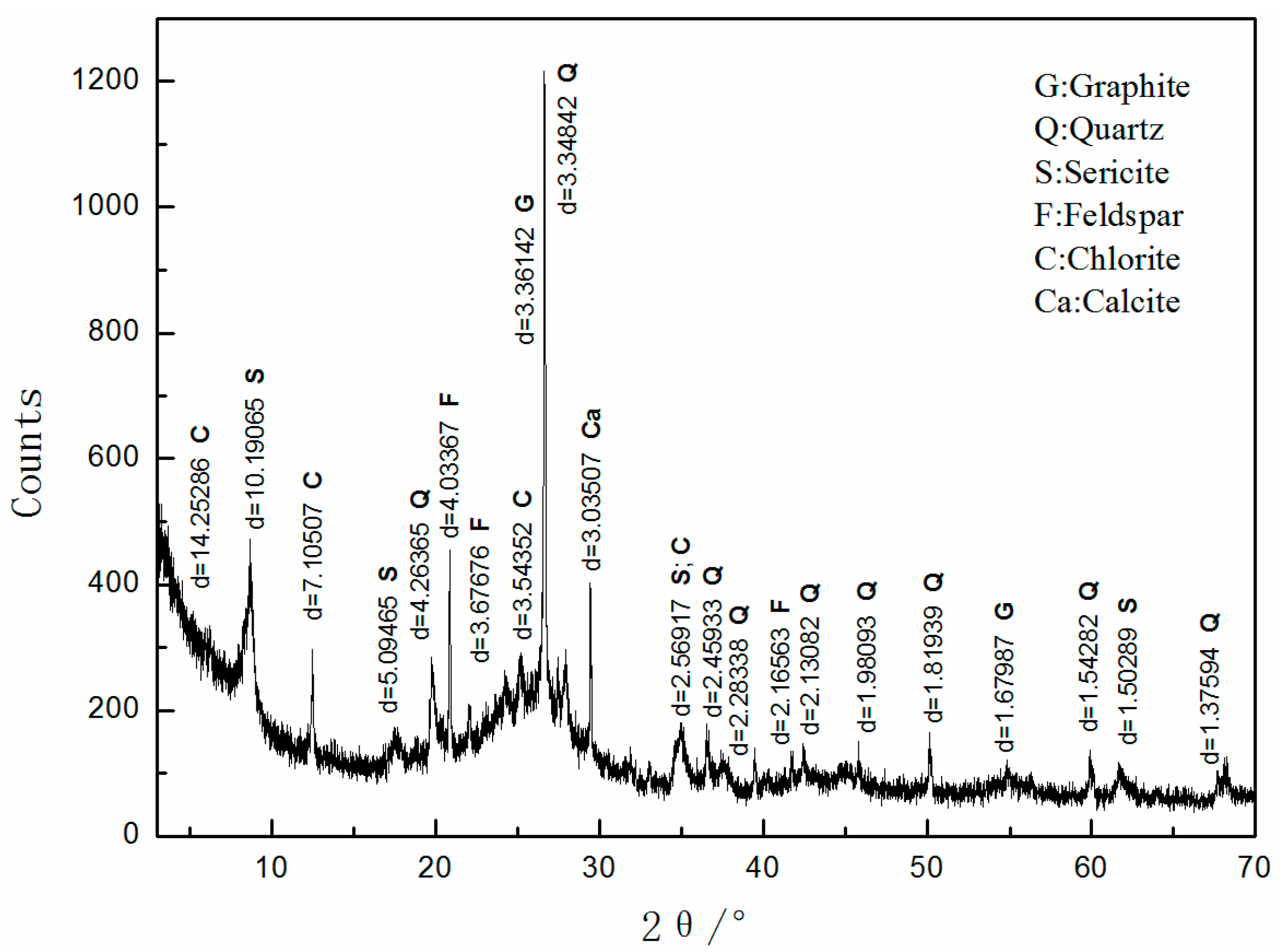
2.2.2. X-Ray Diffraction Analysis
2.2.3. Mineralogical Characteristics Analysis
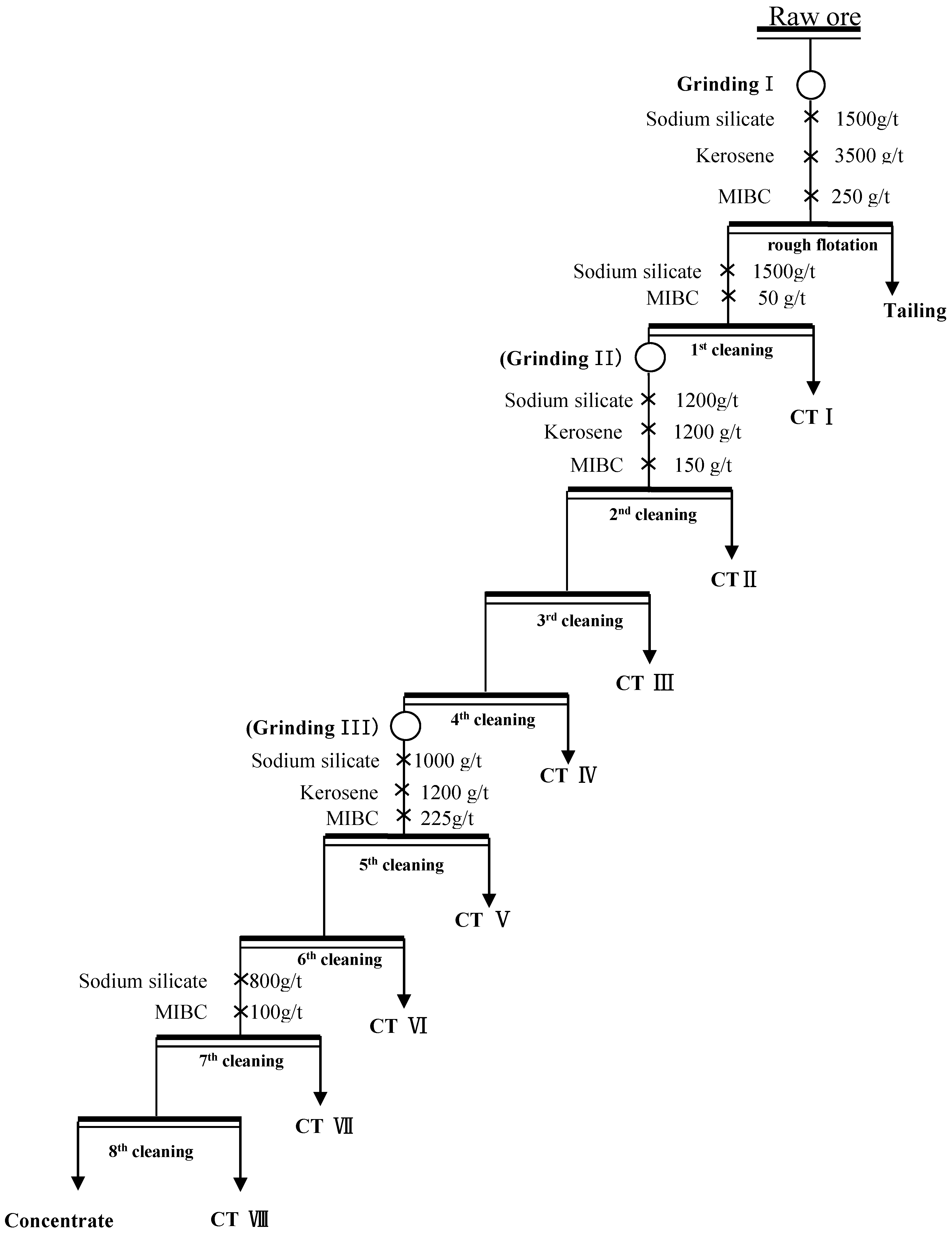
2.2.4. Grinding-Flotation Tests
3. Results and Discussion
3.1. Mineralogical Characteristics
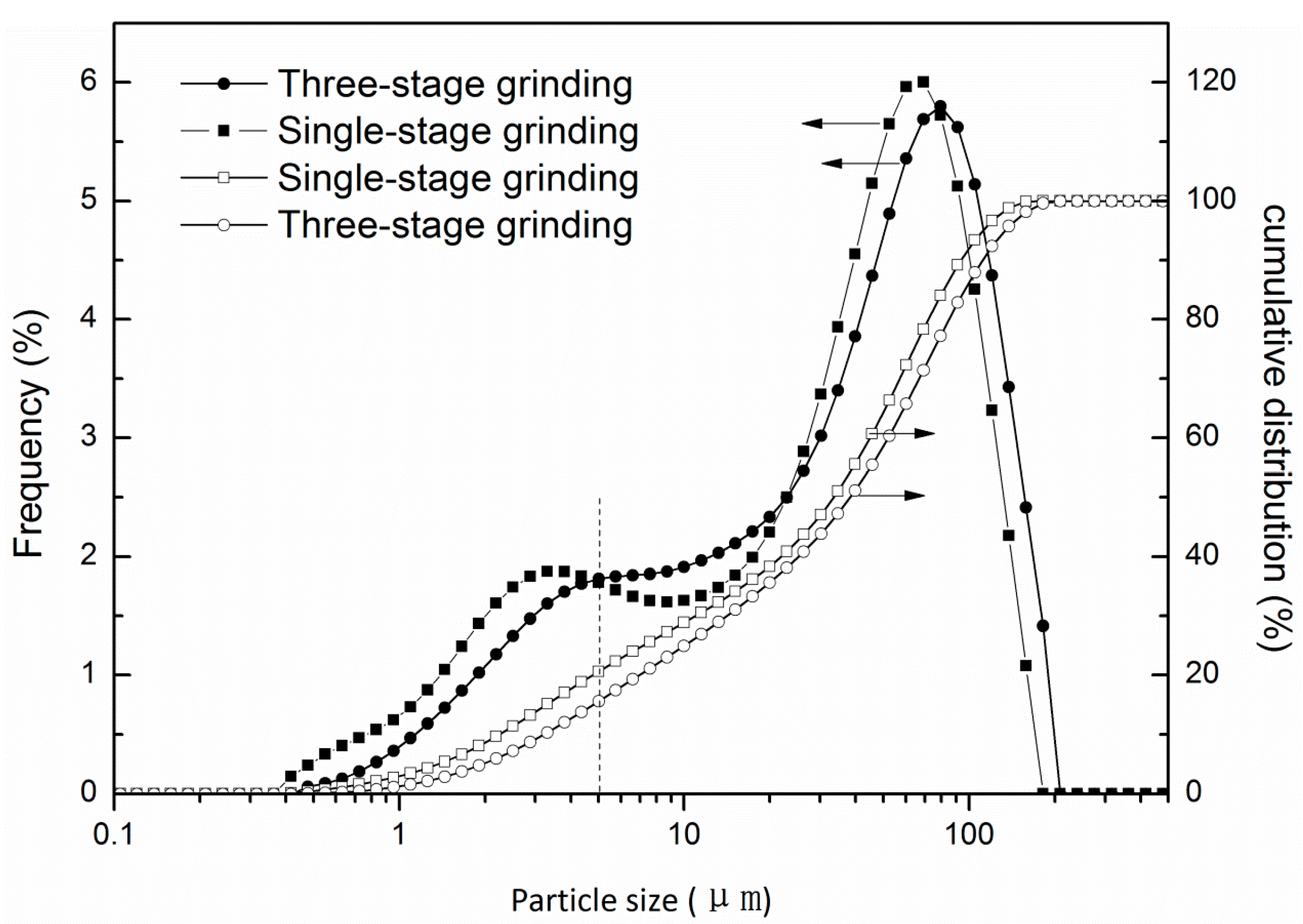
3.2. Determination of the Ultimate Grinding Fineness
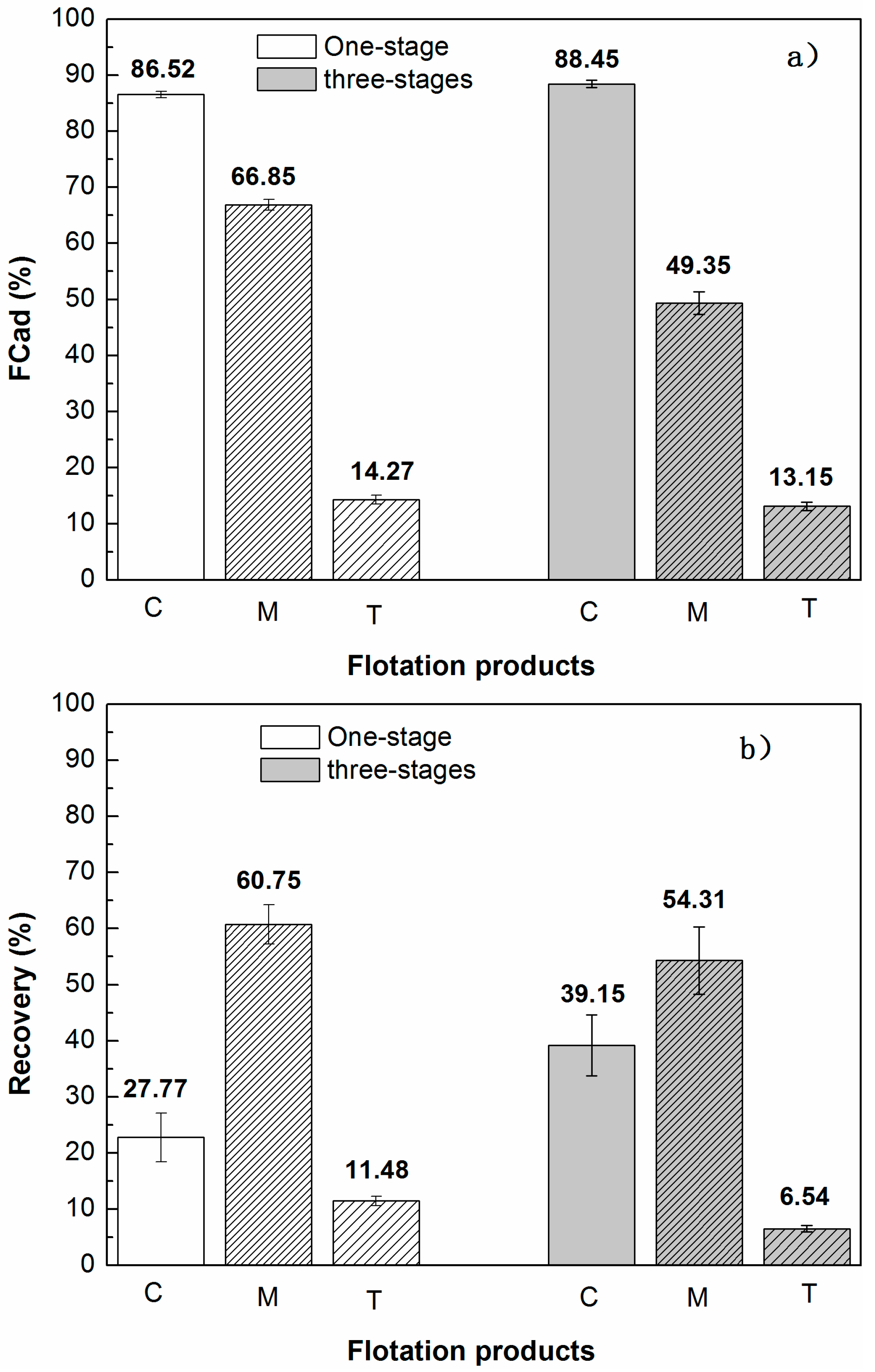
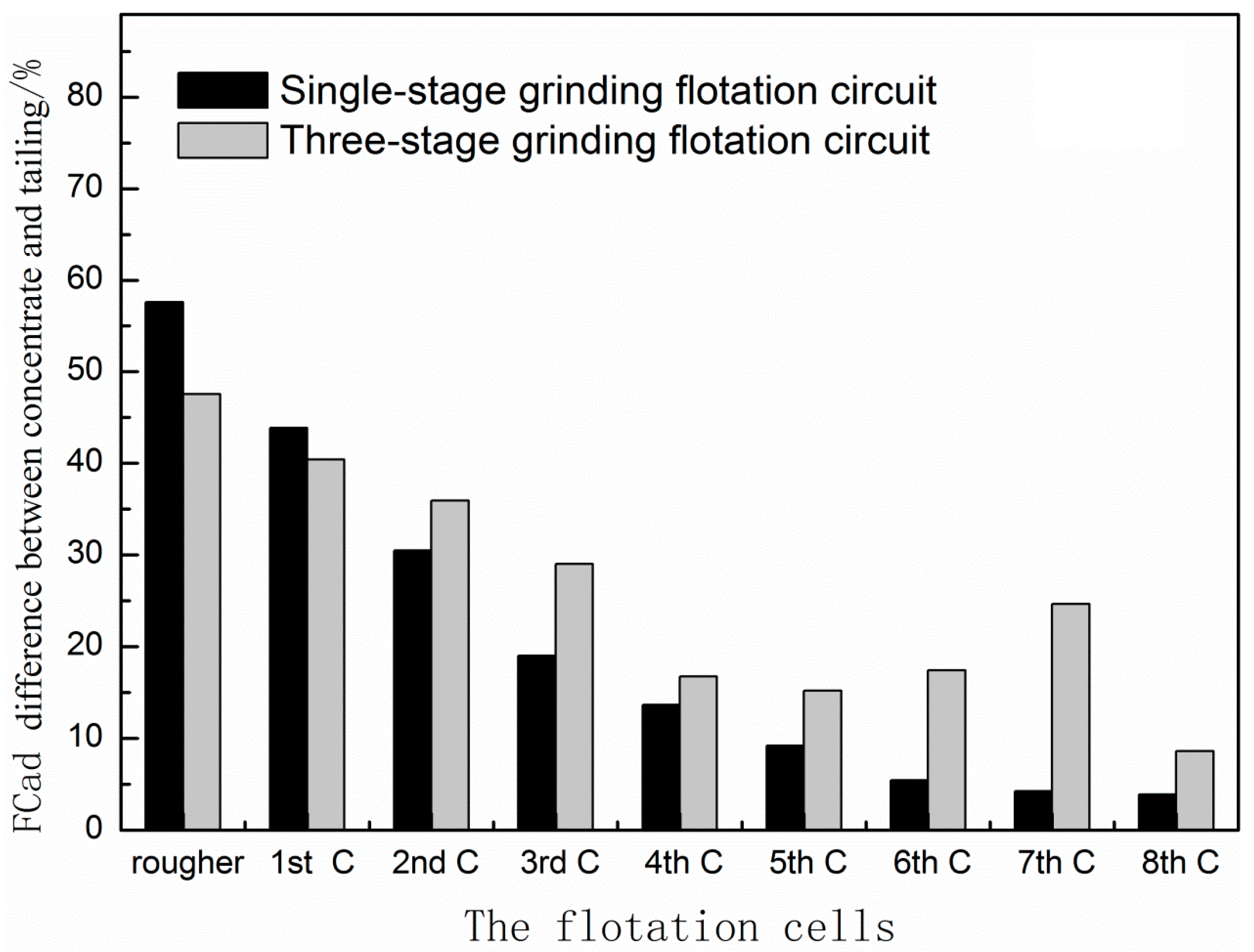
3.3. Comparison of Single-Stage Grinding Circuit and Three-Stage Grinding Circuit
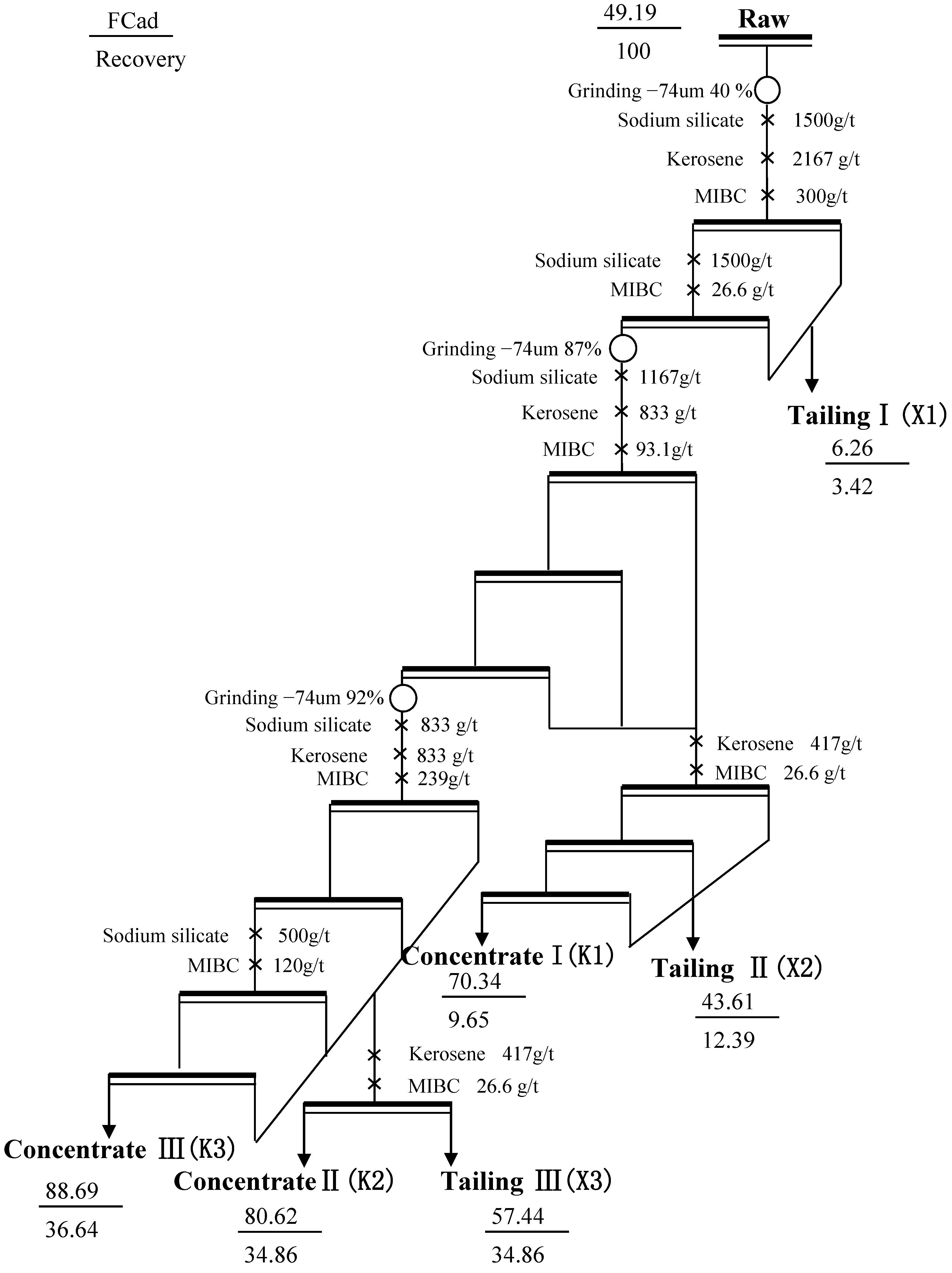
3.4. Application of the Multi-Stage Grinding Flotation Process
4. Conclusions
- (1)
- The results of mineralogical evaluation and grinding flotation tests indicated that a high grade graphite concentrate could be obtained at the fineness of 92% passing 74 μm.
- (2)
- Multi-stage grinding-flotation process can effectively reduce the froth entrainment of gangues; the reduction of gangue slimes plays a vital role in the improvement of the separation effect.
- (3)
- The high grade concentrate which meets the standard of WT88 could be obtained with a recovery of 36.64% using a three-stage grinding flotation process.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Crossley, P. Graphite. Ind. Miner. 1999, 386, 31–47. [Google Scholar]
- Zheng, Z.; Zhang, J.; Huang, J.Y. Observations of microstructure and reflectivity of coal graphites for two locations in China. Int. J. Coal Geol. 1996, 30, 277–284. [Google Scholar] [CrossRef]
- Simandl, G.J.; KenanI, W.M. Microcrystalline Graphite; British Columbia Geological Survey: Vancouver, BC, Canada, 1997; pp. 1–4. [Google Scholar]
- Ji, G.C. Continuous flotation experiment of microcrystalline graphite. Non-Met. Mines 1991, 2, 16–18. [Google Scholar]
- Xia, Y.K.; Ren, Z.M.; Chen, H.X.; Guan, A.F.T. A noval flotation flowsheet of microcrystalline graphite and its industrial application. Non-Met. Mines 1996, 111, 26–28. [Google Scholar]
- Dong, F.Z. A study on flotation of a cryptocrystalline graphite ore. Conserv. Util. Miner. Resour. 1997, 1, 15–17. [Google Scholar]
- Aslan, N.; Cifci, F.; Yan, D. Optimization of process parameters for producing graphite concentrate using response surface methodology. Sep. Purif. Technol. 2008, 59, 9–16. [Google Scholar] [CrossRef]
- Acharya, B.C.; Rao, D.S.; Prakash, S.; Reddy, P.S.R.; Biswal, S.K. Processing of low grade graphite ores of orissa, India. Miner. Eng. 1996, 9, 1165–1169. [Google Scholar] [CrossRef]
- Liu, D.; Peng, Y. Reducing the entrainment of clay minerals in flotation using tap and saline water. Powder Technol. 2014, 253, 216–222. [Google Scholar] [CrossRef]
- Li, H.; Ou, L.; Feng, Q.; Chang, Z. Recovery mechanisms of sericite in microcrystalline graphite flotation. Physicochem. Probl. Miner. Process. 2015, 51, 386–399. [Google Scholar]
- Pugh, R.J. Non-ionic polyethylene oxide frothers in graphite flotation. Miner. Eng. 2000, 13, 151–162. [Google Scholar] [CrossRef]
- Qiu, Y.; Yu, Y.; Zhang, L.; Qian, Y.; Ouyang, Z. An Investigation of Reverse Flotation Separation of Sericite from Graphite by Using a Surfactant: MF. Minerals 2016, 6, 57. [Google Scholar] [CrossRef]
- Gong, J.P.; Bouajila, Y.; Ourriban, A.; Yeung, M.; Liu, Q. Reducing quartz gangue entrainment in sulphide ore flotation by high molecular weight polyethylene oxide. Int. J. Miner. Process. 2010, 97, 44–51. [Google Scholar] [CrossRef]
- Duarte, A.C.P.; Grano, S.R. Mechanism for the recovery of silicate gangue minerals in the flotation of ultrafine sphalerite. Miner. Eng. 2007, 20, 766–775. [Google Scholar] [CrossRef]
- Silvester, E. The recovery of sericite in flotation concentrates. Miner. Process. Extr. Metall. 2011, 120, 10–14. [Google Scholar] [CrossRef]
- Akdemir, Ü.; Taki, G.; Yildiztekin, A.G. Flotation and entrainment behavior of minerals in talc-calcite separation. Scand. J. Metall. 2005, 34, 241–244. [Google Scholar] [CrossRef]
- Kirjavainen, V.M. Review and analysis of factors controlling the mechanical flotation of gangue minerals. Int. J. Miner. Process. 1996, 46, 21–34. [Google Scholar] [CrossRef]
- Mulleneers, H.; Koopal, A.L.K.; Bruning, H.; Rulkens, A.W.H. Selective separation of fine particles by a new flotation approach. Sep. Sci. Technol. 2002, 37, 2097–2112. [Google Scholar] [CrossRef]
- Cao, M.L.; Liu, Q. Reexamining the functions of zinc sulfate as a selective depressant in differential sulfide flotation—The role of coagulation. J. Colloid Interface Sci. 2006, 301, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Bergaya, F.; Lagaly, G. Handbook of Clay Science; Newnes: Melbourne, Australia, 2013. [Google Scholar]
- Ata, S.; Ahmed, N.; Jameson, G. The effect of hydrophobicity on the drainage of gangue minerals in flotation froths. Miner. Eng. 2004, 17, 897–901. [Google Scholar] [CrossRef]
- Neethling, S.J.; Cilliers, J.J. The entrainment factor in froth flotation: Model for particle size and other operating parameter effects. Int. J. Miner. Process. 2009, 93, 141–148. [Google Scholar] [CrossRef]
- Newcombe, B.; Bradshaw, D.; Wightman, A.E. Flash flotation and the plight of the coarse particle. Miner. Eng. 2012, 34, 1–10. [Google Scholar] [CrossRef]



| Cell Volume (L) | Air Flow-Rate (L/min) | Impeller Speed (rpm) | Reagent Dosage (g/t) | Solids Weight (g) | pH | ||
|---|---|---|---|---|---|---|---|
| Sodium Silicate | Kerosene | MIBC | |||||
| 1.5 | 4 | 2045 | 1500 | 2000 | 300 | 300 | 8–9 |
| Flow-Sheet in Figure 1 | Grinding I | Grinding II | Grinding III |
|---|---|---|---|
| Single-stage grinding circuit | 92 | ||
| Three-stage grinding circuit | 40 | 83 | 90 |
| Composition | Mad | Aad | Vad | FCad | Sum |
|---|---|---|---|---|---|
| Content | 3.44 | 43.45 | 3.85 | 49.26 | 100.00 |
| Minerals | Graphite | Quartz | Feldspar | Sericite | Kaolinite | Others |
|---|---|---|---|---|---|---|
| Content | 55 | 20 | 5 | 14 | 4 | 2 |
| Product | Yield/% | FCad/% | Recovery/% | Product Number a | Application Field |
|---|---|---|---|---|---|
| K1 | 7.4 | 70.34 | 9.65 | W70 | Foundry, refractory material, dye and electrode. |
| K2 | 23.17 | 80.62 | 34.86 | W80 | |
| K3 | 22.14 | 88.69 | 36.64 | WT88 | Anode, pencil/battery/graphite emulsion, welding rod/bearing |
| Concentrate sum | 52.66 | 81.16 | |||
| Tailing sum | 47.34 | 21.33 | 18.84 | ||
| Feed | 100 | 49.19 | 100 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weng, X.; Li, H.; Song, S.; Liu, Y. Reducing the Entrainment of Gangue Fines in Low Grade Microcrystalline Graphite Ore Flotation Using Multi-Stage Grinding-Flotation Process. Minerals 2017, 7, 38. https://doi.org/10.3390/min7030038
Weng X, Li H, Song S, Liu Y. Reducing the Entrainment of Gangue Fines in Low Grade Microcrystalline Graphite Ore Flotation Using Multi-Stage Grinding-Flotation Process. Minerals. 2017; 7(3):38. https://doi.org/10.3390/min7030038
Chicago/Turabian StyleWeng, Xiaoqing, Hongqiang Li, Shaoxian Song, and Yanyan Liu. 2017. "Reducing the Entrainment of Gangue Fines in Low Grade Microcrystalline Graphite Ore Flotation Using Multi-Stage Grinding-Flotation Process" Minerals 7, no. 3: 38. https://doi.org/10.3390/min7030038
APA StyleWeng, X., Li, H., Song, S., & Liu, Y. (2017). Reducing the Entrainment of Gangue Fines in Low Grade Microcrystalline Graphite Ore Flotation Using Multi-Stage Grinding-Flotation Process. Minerals, 7(3), 38. https://doi.org/10.3390/min7030038






