A Computer-Controlled SEM-EDX Routine for Characterizing Respirable Coal Mine Dust
Abstract
:1. Introduction
2. Materials and Methods
2.1. Dust Samples
2.1.1. Lab-Generated Dust Samples
2.1.2. Field Samples
2.2. Automated Dust Characterization Routine
2.2.1. Particle Characteristics
2.2.2. Particle Selection and Multi-Frame Sequencing
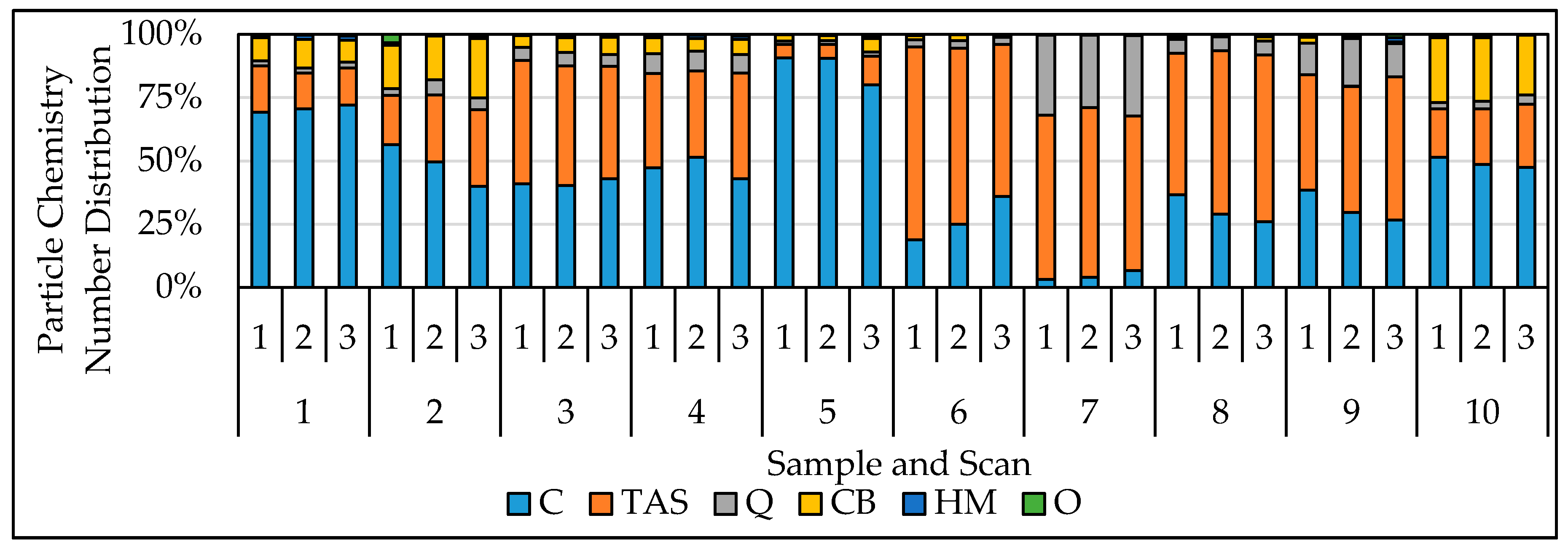
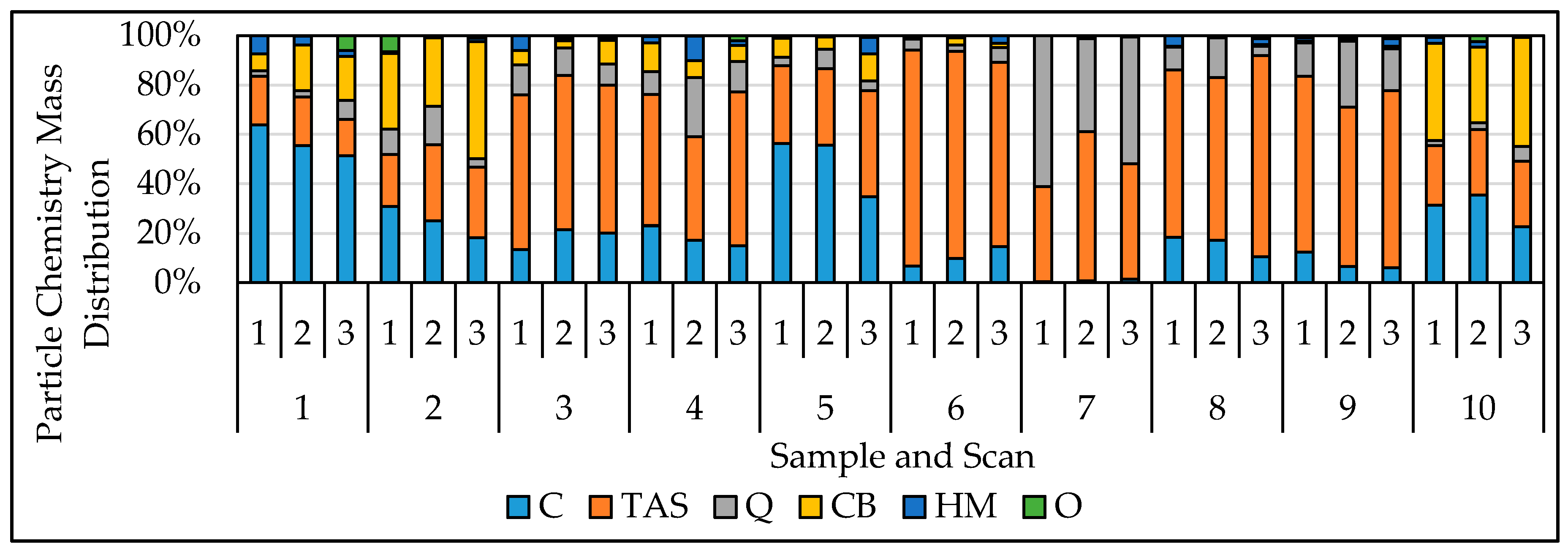
3. Results and Discussion
3.1. Evaluation of Chemical Classification Reliability
3.2. Evaluation of Reproducibility
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Castranova, V.; Vallyathan, V. Silicosis and coal workers’ pneumoconiosis. Environ. Health Perspect. 2000, 108, 675–684. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer (IARC). IARC Monographs on the Evaluation of Carcinogenic Risks to Humans: Silica, Some Silicates, Coal Dust and Para-Aramid Fibrils; IARC Press: Lyon, France, 1997; Volume 68. [Google Scholar]
- International Organization for Standardization (ISO). Air Quality-Particle Size Fraction Definitions for Health Related Sampling; ISO Standard 7708; ISO: Geneva, Switzerland, 1995. [Google Scholar]
- Occupational Safety and Health Administration (OSHA). Occupational Exposure to Respirable Crystalline Silica—Review of Health Effects Literature and Preliminary Quantitative Risk Assessment; OSHA: Washington, DC, USA, 2010.
- World Health Organization (WHO). Hazard Prevention and Control in the Work Environment: Airborne Dust; Report No. WHO/SDE/OEH/99.14; World Health Organization (WHO): Geneva, Switzerland, 1999; pp. 1–246. [Google Scholar]
- Merchant, J.A.; Taylor, G.; Hodous, T.K. Coal workers’ pneumoconiosis and exposure to other carbonaceous dusts. In Occupational Respiratory Diseases; Merchant, J.A., Ed.; U.S. Goverment Printing Office: Washington, DC, USA, 1986; pp. 329–384. [Google Scholar]
- Peters, J.M. Silicosis. In Occupational Respiratory Diseases; Merchant, J.A., Ed.; U.S. Goverment Printing Office: Washington, DC, USA, 1986; pp. 219–237. [Google Scholar]
- Antao, V.C.; Petsonk, E.L.; Attfield, M.D. Advanced Cases of Coal Workers’ Pneumoconiosis—Two Counties, Virginia. MMWR 2006, 55, 909–913. Available online: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm5533a1 (accessed on 25 August 2006). [Google Scholar]
- National Institute of Occupational Safety and Health (NIOSH). Criteria for a Recommended Standard—Occupational Exposure to Respirable Coal Mine Dust; DHHS (NIOSH) Publication No. 95-106; NIOSH: Cincinnati, OH, USA, 1995. [Google Scholar]
- Suarthana, E.; Laney, A.S.; Storey, E.; Hale, J.M.; Attfield, M.D. Coal workers’ pneumoconiosis in the United States: Regional differences 40 years after implementation of the 1969 Federal coal mine health and safety act. Occup. Environ. Med. 2011, 68, 908–913. [Google Scholar] [CrossRef] [PubMed]
- Gibson, P. Australian Mining. Available online: https://www.australianmining.com.au/features/black-lungs-back-how-we-became-complacent-with-coal-miners-pneumoconiosis/ (accessed on 26 October 2016).
- Zosky, G.R.; Hoy, R.F.; Silverstone, E.J.; Brims, F.J.; Miles, S.; Johnson, A.R.; Gibson, P.G.; Yates, D.H. Coal workers’ pneumoconiosis: An Australian perspective. Med. J. Aust. 2016, 204, 414–418. [Google Scholar] [CrossRef] [PubMed]
- National Institute of Occupational Safety and Health (NIOSH). Coal Workers’ Health Surveillance Program (CWHSP) Data Query System; NIOSH: Morgantown, WV, USA, 2016. [Google Scholar]
- Antao, V.C.; Petsonk, E.L.; Sokolow, L.Z.; Wolfe, A.L.; Pinheiro, G.A.; Hale, J.M.; Attfield, M.D. Rapidly progressive coal workers’ pneumoconiosis in the United States: Geographic clustering and other factors. J. Occup. Environ. Med. 2005, 62, 670–674. [Google Scholar] [CrossRef] [PubMed]
- Laney, A.S.; Attfield, M.D. Coal workers’ pneumoconiosis and progressive massive fibrosis are increasingly more prevalent among workers in small underground coal mines in the United States. J. Occup. Environ. Med. 2010, 67, 428–431. [Google Scholar] [CrossRef] [PubMed]
- Mine Safety and Health Administration (MSHA). All coal mining data table 01. In Number of Coal Operations in the United States, by Primary Activity, 1978–2006, Mining Industry Accident, Injuries, Employment, and Production Statistics; US Department of Labor: Arlington, VA, USA, 2008. [Google Scholar]
- Bennett, J.G.; Dick, J.A.; Kaplan, Y.S.; Shand, P.A.; Shennan, D.H.; Thomas, D.J.; Washington, J.S. The relationship between coal rank and the prevalence of pneumoconiosis. Br. J. Ind. Med. 1979, 36, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Colinet, J.F.; Rider, J.P.; Listak, J.M.; Organiscak, J.A.; Wolfe, A.L. Best Practices for Dust Control in Coal Mining; DHHS (NIOSH) Publication No. 2010-110; NIOSH: Cincinnati, OH, USA, 2010. [Google Scholar]
- Joy, G.J. Evaluation of the approach to respirable quartz exposure control in U.S. Coal mines. J. Occup. Environ. Hyg. 2012, 9, 65–68. [Google Scholar] [CrossRef] [PubMed]
- Landen, D.D.; Wassell, J.T.; McWilliams, L.; Patel, A. Coal dust exposure and mortality from ischemic heart disease among a cohort of U.S. Coal miners. Am. J. Ind. Med. 2011, 54, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Mischler, S.E.; Cauda, E.G.; Di Giuseppe, M.; Ortiz, L.A. A multi-cyclone sampling array for the collection of size-segregated occupational aerosols. J. Occup. Environ. Hyg. 2013, 10, 685–693. [Google Scholar] [CrossRef] [PubMed]
- Page, S.J.; Organiscak, J.A. Suggestion of a cause-and-effect relationship among coal rank, airborne dust, and incidence of workers’ pneumoconiosis. Am. Ind. Hyg. Assoc. J. 2000, 61, 785–787. [Google Scholar]
- Pollock, D.E.; Potts, J.D.; Joy, G.J. Investigation into dust exposures and mining practices in mines in the southern Appalachian region. In Proceedings of the 2009 SME Annual Meeting and Exhibit, Denver, CO, USA, 22–25 February 2009; Society for Mining, Metallurgy, and Exploration, Inc.: Denver, CO, USA, 2009; pp. 1–5. [Google Scholar]
- Agius, R. Occupational and Environmental Lung Disease. Available online: http://www.agius.com/hew/resource/lung.htm (accessed on 1 November 2016).
- Johann, V.; Sarver, E. Considerations for an automated SEM-EDX routine for characterizing respirable coal mine dust. In Proceedings of the 15th North American Mine Ventilation Symposium, Blacksburg, VA, USA, 20–25 June 2015.
- Sellaro, R.; Sarver, E. Preliminary investigation of SEM-EDX as a tool for characterization of coal mine dusts. Miner. Eng. 2014, 66, 16–40. [Google Scholar]
- Sellaro, R.; Sarver, E. Characterization of respirable dust in an underground coal mine in Central Appalachia. Trans. SME 2014, 336, 457–466. [Google Scholar]
- Sellaro, R.; Sarver, E.; Baxter, D. A standard characterization methodology for respirable coal mine dust using SEM-EDX. Resources 2015, 4, 939–957. [Google Scholar] [CrossRef]
- Carpenter, M.; Lifshin, E.; Gauvin, R. SEM-EDS quantative analysis of aerosols ≥80 nm: Impact on atmospheric aerosol characterization campaigns. Microsc. Microanal. 2002, 8, 1482CD–1483CD. [Google Scholar]
- Cvetković, Ž.; Logar, M.; Rosić, A.; Ćirić, A. Mineral composition of the airborne particles in the coal dust and fly ash of the Kolubara basin (Serbia). Period. Mineral. 2012, 81, 205–223. [Google Scholar]
- Deboudt, K.; Flament, P.; Choël, M.; Gloter, A.; Sobanska, S.; Colliex, C. Mixing state of aerosols and direct observation of carbonaceous and marine coatings on African dust by individual particle analysis. J. Geophys. Res. Atmos. 2010, 115, D24207. [Google Scholar] [CrossRef]
- Kasahara, M.; Shinoda, K.; Yoshida, K.; Takahashi, K. Characterization of atmospheric aerosol based on SEM-EDX analysis of individual particles. J. Aerosol Sci. 1993, 24, S585–S586. [Google Scholar] [CrossRef]
- Kasparian, J.; Frejafon, E.; Rambaldi, P.; Yu, J.; Vezin, B.; Wolf, J.P.; Ritter, P.; Viscardi, P. Characterization of urban aerosols using SEM-microscopy, X-ray analysis and lidar measurements. Atmos. Environ. 1998, 32, 2957–2967. [Google Scholar] [CrossRef]
- Micheletti, M.I.; Murruni, L.G.; Debray, M.E.; Rosenbusch, M.; Graf, M.; Ávila Cadena, G.; Vitale, P.; Davidson, J.; Somacal, H. Elemental analysis of aerosols collected at the Pierre Auger cosmic ray observatory with pixe technique complemented with SEM/EDX. Nucl. Instrum. Methods Phys. Res. Sect. B Beam Interact. Mater. Atoms 2012, 288, 10–17. [Google Scholar] [CrossRef]
- Suzuki, K. Characterisation of airborne particulates and associated trace metals deposited on tree bark by ICP-OES, ICP-MS, SEM-EDX and laser ablation ICP-MS. Atmos. Environ. 2006, 40, 2626–2634. [Google Scholar] [CrossRef]
- Wang, A.; Luo, B. Application SEM to analysis formation characteristic of soot aerosol emitted from lump-coal combustion in fixed-bed. In Proceedings of the Asia-Pasific Power and Energy Engineering Conference (APPEEC), Wuhan, China, 28–31 March 2009.
- United States Environmental Protection Agency (US EPA). Guidelines for the Application of SEM/EDX Analytical Techniques to Particulate Matter Samples; US EPA: Washington, DC, USA, 2002.
- Denee, P.B. Mine dust characterization using the scanning electron microscope. Am. Ind. Hyg. Assoc. J. 1972, 33, 654–660. [Google Scholar] [CrossRef] [PubMed]
- Huggins, C.W.; Meyers, G.T. Particle Size Distribution of Quartz and Other Respirable Dust Particles Collected at Metal Mines, Nonmetal Mines, and Processing Plants; U.S. Department of the Interior, Bureau of Mines: Pittsburgh, PA, USA, 1986.
- Terry, K. Particle size distribution of airborne dusts using a scanning electron microscope. Aerosol Sci. Technol. 1995, 23, 475–478. [Google Scholar] [CrossRef]
- White, E.W.; DeNee, P.B. Characterization of coal mine dust by computer processing of scanning electron microscope information. Ann. N. Y. Acad. Sci. 1972, 200, 666–675. [Google Scholar] [CrossRef] [PubMed]
- Johann, V. Development and Implementation of an Automated Sem-Edx Routine for Characterizing Respirable Coal Mine Dust. Master’s Thesis, Virginia Polytechnic Institute and State University, Blacksburg, VA, USA, 2016. [Google Scholar]
- Ekelund, S.; Werlefors, T. A system for the quantitative characterization of microstructures by combined image analysis and X-ray discrimination in the scanning electron microscope. In Proceedings of the 9th Annual SEM Symposium, Chicago, IL, USA, 5–9 April 1976; IITRI: Chicago, IL, USA, 1976; pp. 419–424. [Google Scholar]
- Hoover, M.F.; White, E.; Lebiedzik, N.J.; Johnson, G.G., Jr. Automated characterization of particulates and inclusions by computer-controlled sem/probe. In Proceedings of the Tenth Annual Conference on Microbeam Analysis Society, Las Vegas, NV, USA, 11–15 August 1975; pp. 54A–54B.
- Fritz, G.; Camus, P.; Rohde, D. Considerations for automated multi-frame particle sizing in the SEM. Microsc. Microanal. 2006, 12, 210–211. [Google Scholar] [CrossRef]
- Ritchie, N.; Filip, V. Semantics for high speed automated particle analysis by SEM/EDX. Microsc. Microanal. 2011, 17, 896–897. [Google Scholar] [CrossRef]
- Worobiec, A.; Potgieter-Vermaak, S.; Brooker, A.; Darchuk, L.; Stefaniak, E.; Van Grieken, R. Interfaced SEM/EDX and micro-raman spectrometry for characterisation of heterogeneous environmental particles—Fundamental and practical challenges. Microchem. J. 2010, 94, 65–72. [Google Scholar] [CrossRef]
- Huffman, G.P.; Shah, N.; Cprek, N.; Huggins, F.E.; Casuccio, G.; Ramer, E.; Hicks, J.B. CCSEM Investigation of Respirable Quartz in Air Samples Collected during Power Plant Maintenance Activities. Fuel 2011, 95, 365–370. [Google Scholar] [CrossRef]
- Cprek, N.; Shah, N.; Huggins, F.E.; Huffman, G.P. Computer-controlled scanning electron microscopy (CCSEM) investigation of quartz in coal fly ash. Fuel Process. Technol. 2007, 88, 1017–1020. [Google Scholar] [CrossRef]
- Code of Federal Regulations (CFR). Title 30—Mineral Resources, Part 70—Mandatory Health Standards-Underground Coal Mines; Office of the Federal Register: Washington, DC, USA, 2014.
- Mine Safety and Health Administration (MSHA). MSHA Handbook Series—Coal Mine Health Inspection Procedures; Mine Safety and Health Administration (MSHA): Washington, DC, USA, 2016.
- Scaggs, M. Development and Implementation of a Standard Methodology for Respirable Coal Mine Dust Characterization with Thermogravimetric Analysis. Master’s Thesis, Virginia Polytechnic Institute and State University, Blacksburg, VA, USA, 2016. [Google Scholar]
- Agresti, A. A survey of exact inference for contingency tables. Stat. Sci. 1992, 7, 131–153. [Google Scholar] [CrossRef]
- Pietrodangelo, A.; Salzano, R.; Bassani, C.; Pareti, S.; Perrino, C. Composition, size distribution, optical properties, and radiative effects of laboratory-resuspended PM10 from geological dust of the Rome area, by electron microscopy and radiative transfer modelling. Atmos. Chem. Phys. 2015, 15, 13177–13194. [Google Scholar] [CrossRef]
- Schatzel, S.J. Identifying sources of respirable quartz and silica dust in underground coal mines in southern west Virginia, western Virginia, and eastern Kentucky. Int. J. Coal Geol. 2009, 78, 110–118. [Google Scholar] [CrossRef]
- Schatzel, S.J. Characterization of source rocks producing respirable quartz and aluminosilicate dust in underground US coal mines. In Proceedings of the 23rd Annual International Pittsburgh Coal Conference, Pittsburgh, PA, USA, 25–28 September 2006; pp. 38–39.
- Colinet, J.F.; Listak, J.M. Silica and Respirable Content in Rock Dust Samples. Coal Age 2012, 117, 48–52. [Google Scholar]
- Mine Safety and Health Administration (MSHA). Lowering Miners’ Exposure to Respirable Coal Mine Dust, Including Continuous Personal Dust Monitors (Final Rule). Fed. Reg. 2014, 79, 24813. [Google Scholar]
- Harris, M.L.; Sapko, M.J.; Zlochower, I.A.; Perera, I.E.; Weiss, E.S. Particle size and surface area effects on explosibility using a 20-L chamber. J. Loss Prev. Process Ind. 2015, 37, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Cohen, R.A.; Petsonk, E.L.; Rose, C.; Young, B.; Regier, M.; Najmuddin, A.; Abraham, J.L.; Churg, A.; Green, F.H.Y. Lung Pathology in U.S. Coal Workers with Rapidly Progressive Pneumoconiosis Implicates Silica and Silicates. Am. J. Respir. Crit. Care Med. 2016, 193, 673–680. [Google Scholar] [CrossRef] [PubMed]
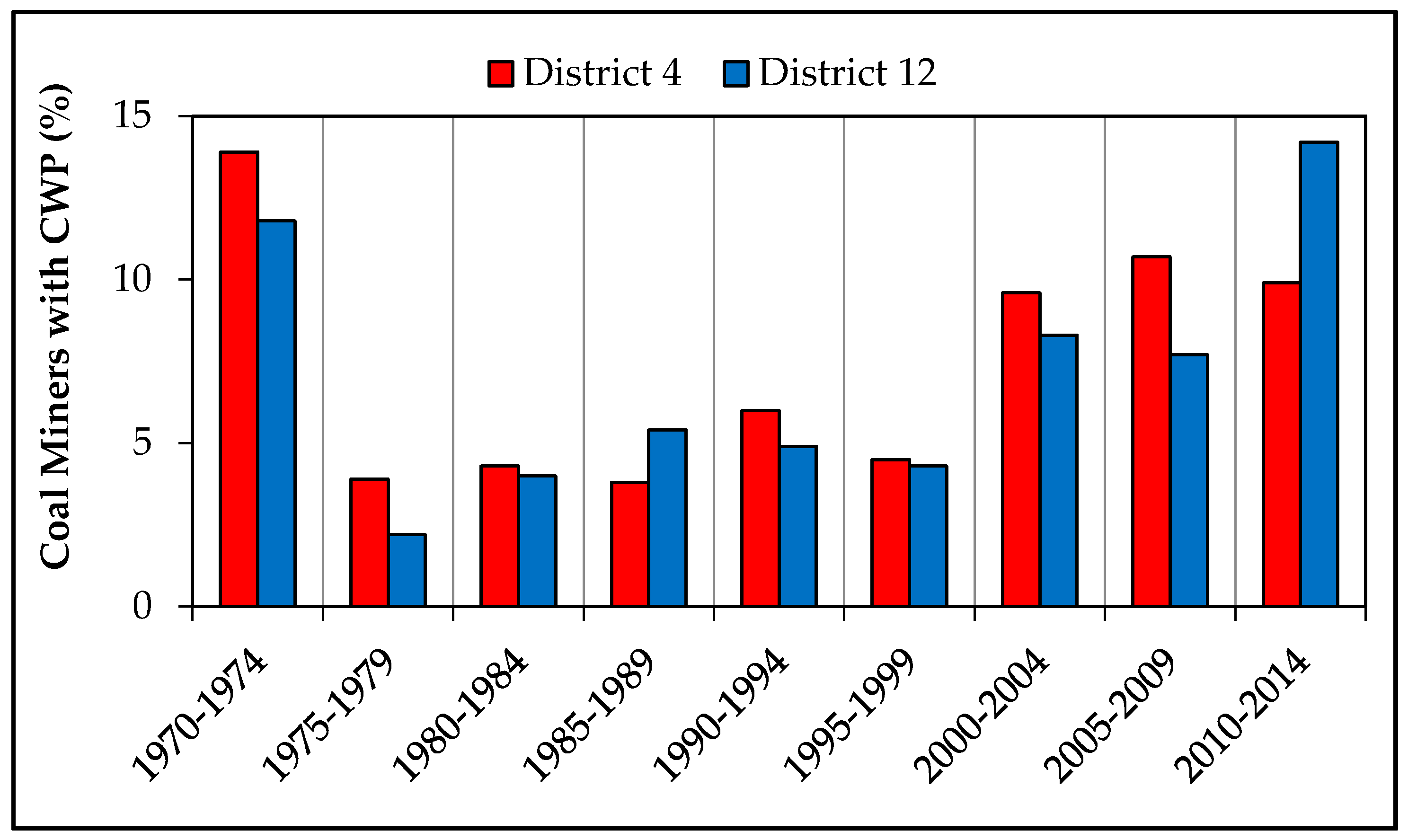
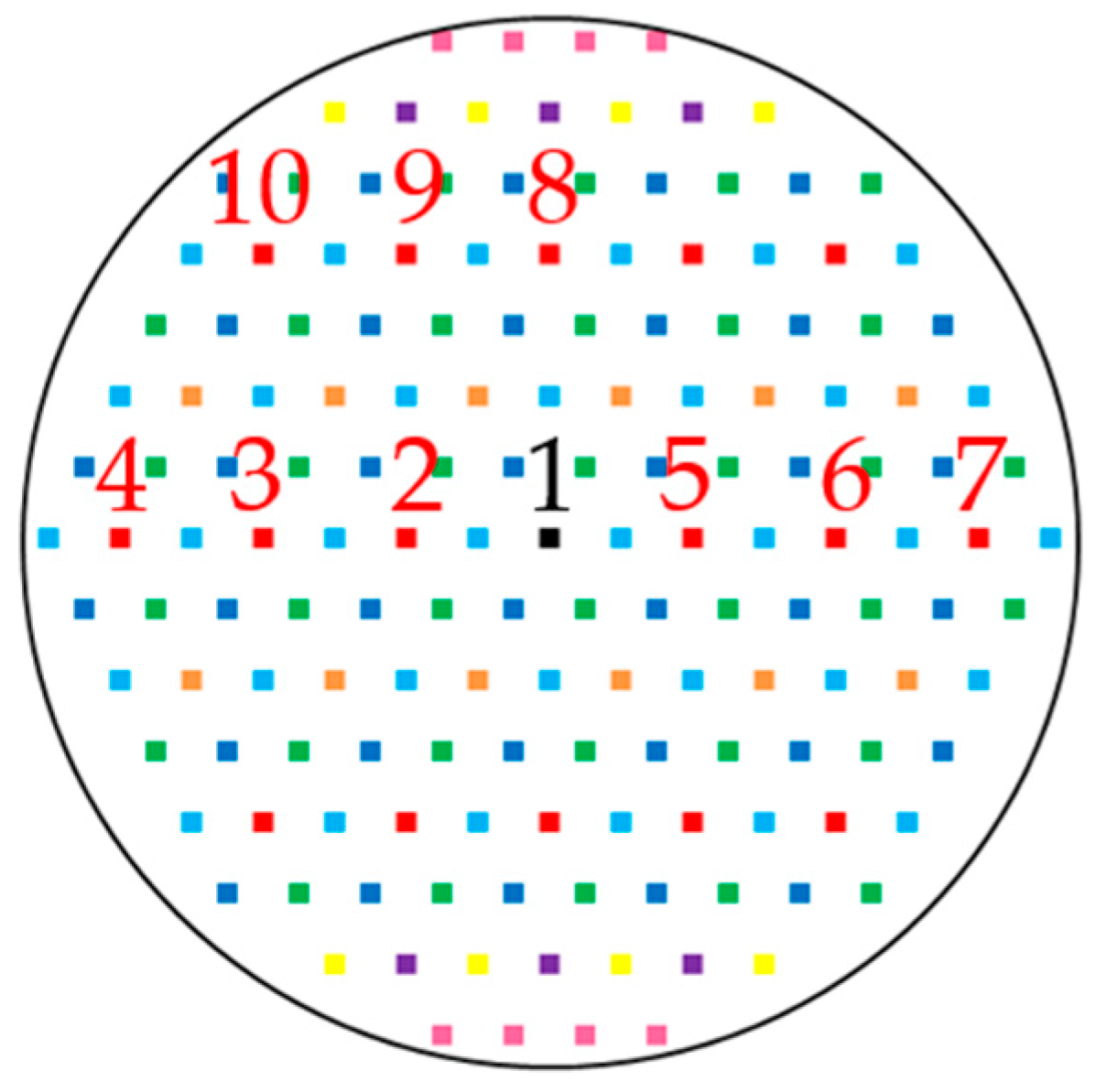

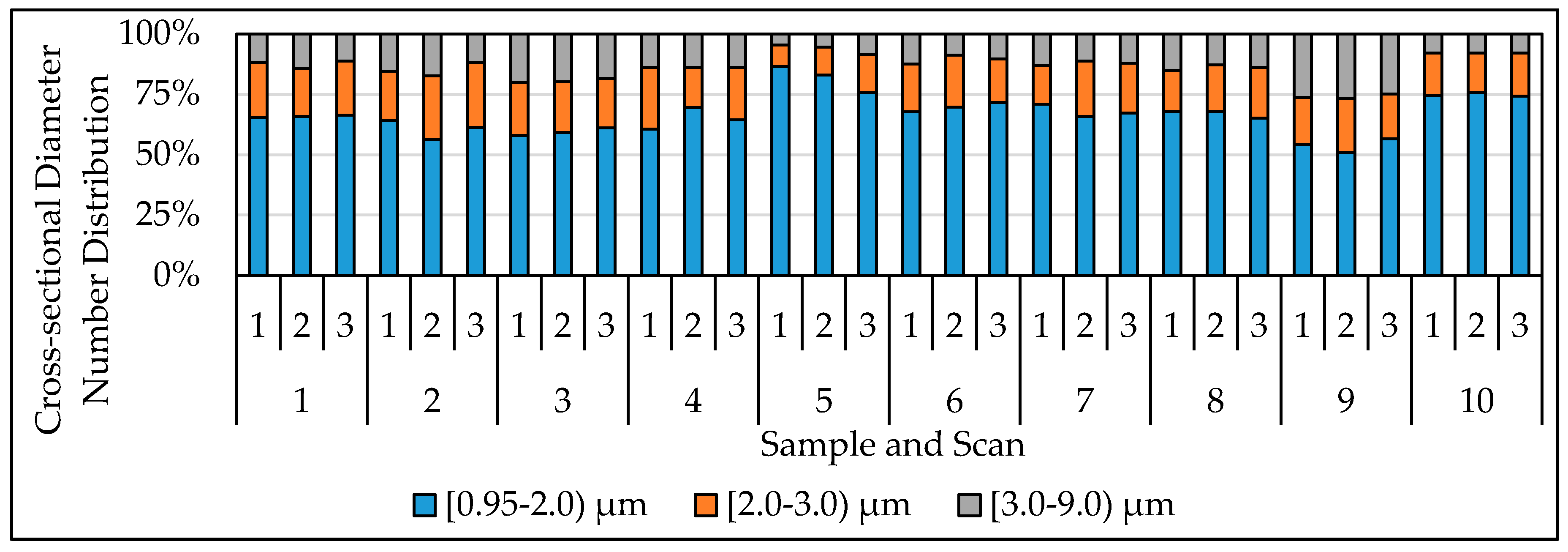
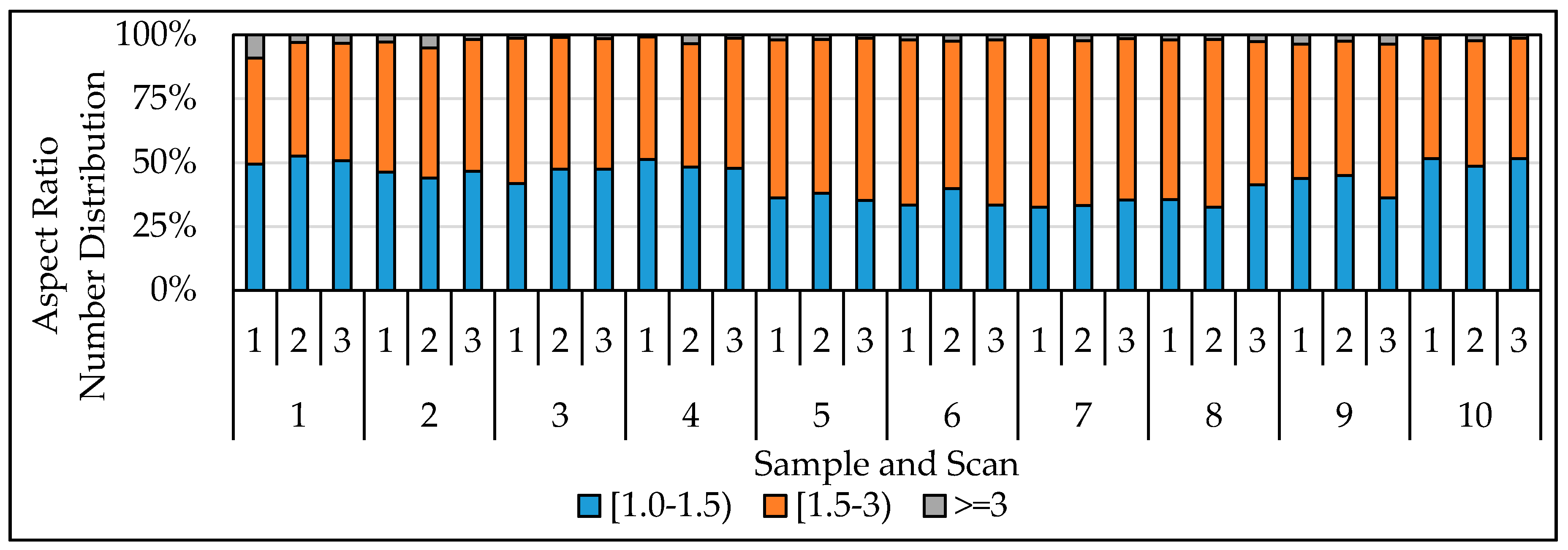
| Sample No. | Mine Region | Roof and/or Floor Rock | Average | Average | Sampling Location |
|---|---|---|---|---|---|
| Seam | Mining | ||||
| Thickness (m) | Height (m) | ||||
| 1 | South-central Appalachia(District 12) | Shale | 0.6 | 1.0 | Belt Drive |
| 2 | Tram | ||||
| 3 | C. Miner 1 | ||||
| 4 | Behind Feeder | ||||
| 5 | Tram | ||||
| 6 | Mid-central Appalachia(District 4) | Sandstone | 0.9–1.5 | 1.5 | Roof Bolter |
| 7 | Sandstone 2 | 0.9–1.2 | 1.7 | Return Entry | |
| 8 | Shale/sandstone | 1.4 | 1.8 | C. Miner 1 | |
| 9 | Sandstone 2 | 0.9–1.2 | 1.7 | C. Miner 1 | |
| 10 | Sandstone | 0.9–1.5 | 1.5 | Behind Feeder |
| Elements | Manual Method Criteria (Raw Peak Heights, Cps/eV) | Automated Method Criteria (Elemental Atomic, %) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C | MC | AS | Q | CB | HM | C | MC | AS | Q | CB | HM | |
| Carbon | ≥80 | ≥80 | - | - | <80 | - | >74 | >78 | <85 | <85 | <85 | - |
| Oxygen | ≤20 | ≤20 | >20 | ≥20 | >20 | >20 | <29 | >13, <20 | >15 | >15 | >15 | >12 |
| Aluminum | - | ≥10, <20 | ≥20 | ≥20 | - | - | <0.3 | >0.2, <0.4 | >0.3 | >0.3 | - | - |
| Silica | - | ≥10, <20 | ≥20 | ≥20 | - | - | <0.3 | >0.2, <0.4 | >0.4 | >0.4 | - | - |
| Ca/Mg 1 | - | - | - | - | ≥20 | - | <0.3 | <0.4 | - | - | >0.5 | - |
| Fe/Ti/Al 2 | - | - | - | - | - | ≥20 | - | - | - | - | - | >0.5 |
| Sample Type | Chemical Composition Categories | |||||
|---|---|---|---|---|---|---|
| C | TAS 1 | Q | CB | HM | Other | |
| Coal | 92% | 4% | 1% | 2% | 1% | - |
| Shale | 6% | 88% | 3% | 1% | 2% | - |
| Rock Dust | 3% | 7% | 1% | 88% | 1% | - |
| Quartz | 4% | 4% | 92% | - | - | - |
| Kaolinite | - | 100% | - | - | - | - |
| Calcite | 1% | - | - | 99% | - | - |
| Sample | Size Comparison | Aspect Ratio Comparison | Chemistry Comparison | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 vs. 2 | 1 vs. 3 | 2 vs. 3 | 1 vs. 2 | 1 vs. 3 | 2 vs. 3 | 1 vs. 2 | 1 vs. 3 | 2 vs. 3 | |
| 1 | 0.831 | 1.000 | 0.847 | 0.223 | 0.212 | 0.964 | 0.922 | 0.955 | 0.992 |
| 2 | 0.479 | 0.513 | 0.606 | 0.802 | 1.000 | 0.546 | 0.276 | 0.145 | 0.504 |
| 3 | 1.000 | 0.867 | 0.940 | 0.783 | 0.737 | 1.000 | 0.995 | 0.896 | 0.984 |
| 4 | 0.309 | 0.840 | 0.613 | 0.657 | 0.888 | 0.657 | 0.978 | 0.977 | 0.803 |
| 5 | 0.761 | 0.152 | 0.367 | 0.956 | 0.912 | 0.743 | 1.000 | 0.256 | 0.317 |
| 6 | 0.815 | 0.834 | 0.896 | 0.592 | 1.000 | 0.592 | 0.754 | 0.038 1 | 0.427 |
| 7 | 0.498 | 0.705 | 0.953 | 1.000 | 0.882 | 0.867 | 0.865 | 0.470 | 0.533 |
| 8 | 0.887 | 0.794 | 0.911 | 1.000 | 0.882 | 0.867 | 0.714 | 0.522 | 0.951 |
| 9 | 0.929 | 0.944 | 0.742 | 0.833 | 0.534 | 0.374 | 0.401 | 0.558 | 0.482 |
| 10 | 0.969 | 1.000 | 0.969 | 0.831 | 1.000 | 0.831 | 0.927 | 0.644 | 0.937 |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Johann-Essex, V.; Keles, C.; Sarver, E. A Computer-Controlled SEM-EDX Routine for Characterizing Respirable Coal Mine Dust. Minerals 2017, 7, 15. https://doi.org/10.3390/min7010015
Johann-Essex V, Keles C, Sarver E. A Computer-Controlled SEM-EDX Routine for Characterizing Respirable Coal Mine Dust. Minerals. 2017; 7(1):15. https://doi.org/10.3390/min7010015
Chicago/Turabian StyleJohann-Essex, Victoria, Cigdem Keles, and Emily Sarver. 2017. "A Computer-Controlled SEM-EDX Routine for Characterizing Respirable Coal Mine Dust" Minerals 7, no. 1: 15. https://doi.org/10.3390/min7010015
APA StyleJohann-Essex, V., Keles, C., & Sarver, E. (2017). A Computer-Controlled SEM-EDX Routine for Characterizing Respirable Coal Mine Dust. Minerals, 7(1), 15. https://doi.org/10.3390/min7010015






