Flotability Improvement of Ilmenite Using Attrition-Scrubbing as a Pretreatment Method
Abstract
:1. Introduction
2. Experimental
2.1. Materials and Reagents
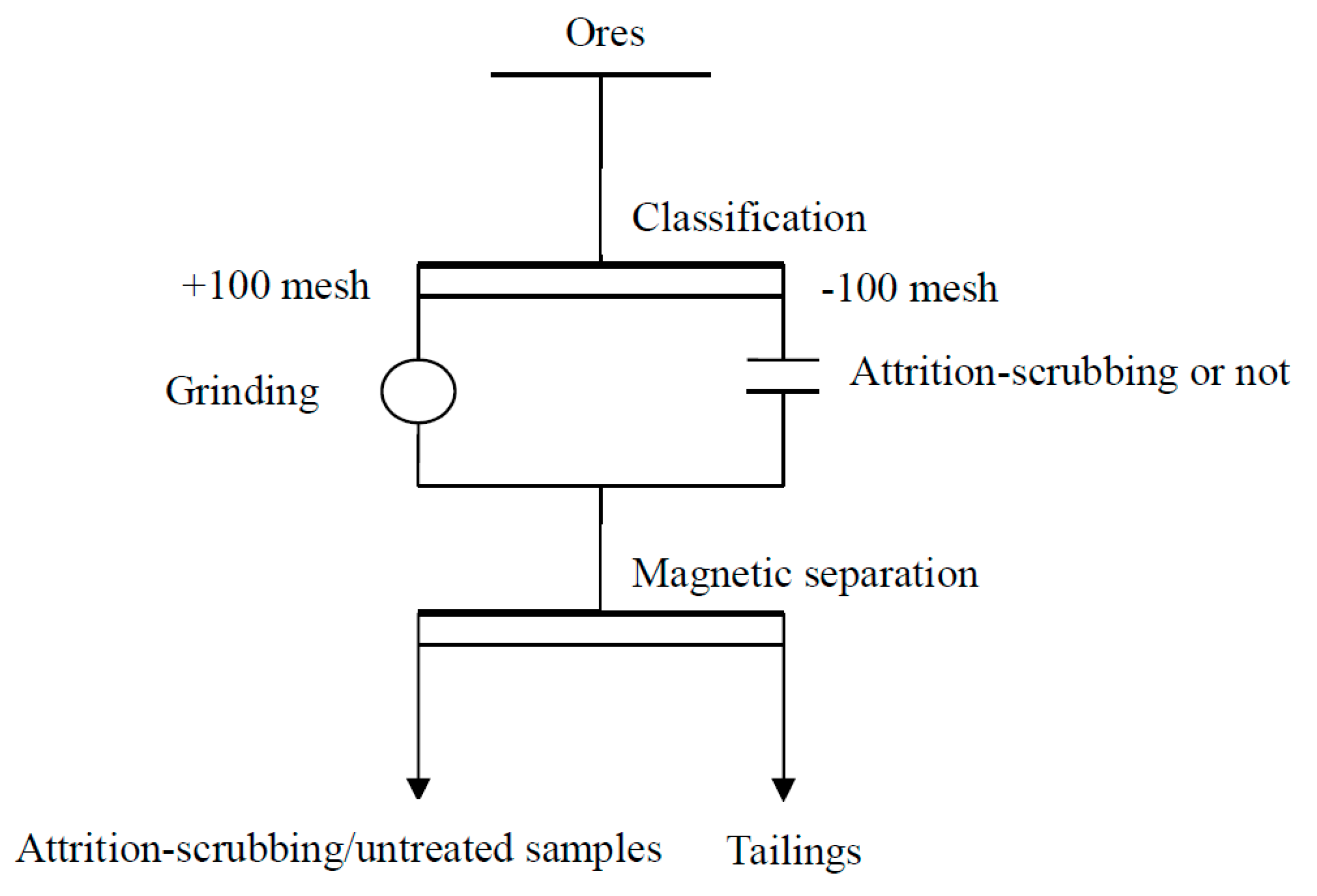
2.2. Pre-Enrichment Methods
2.3. Attrition-Scrubbing Design
2.4. Characterization Methods
2.5. Experimental Procedure
3. Results and Discussions
3.1. Mineralogical Characteristics
3.1.1. Chemical Composition Analysis
3.1.2. Occurrence of Ilmenite
3.1.3. Ilmenite Distribution in Ore Samples
3.2. Flotation Results and Analysis
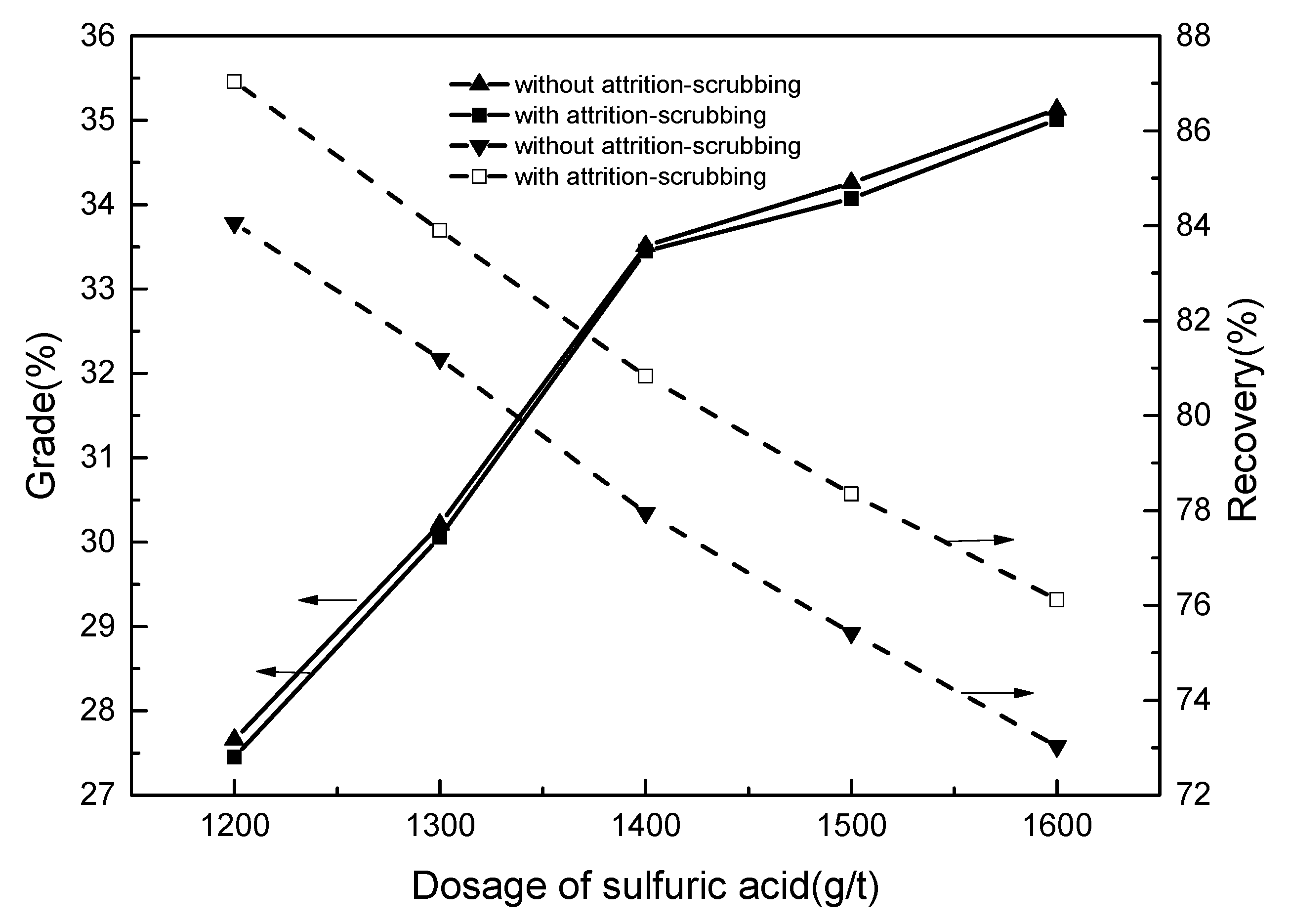
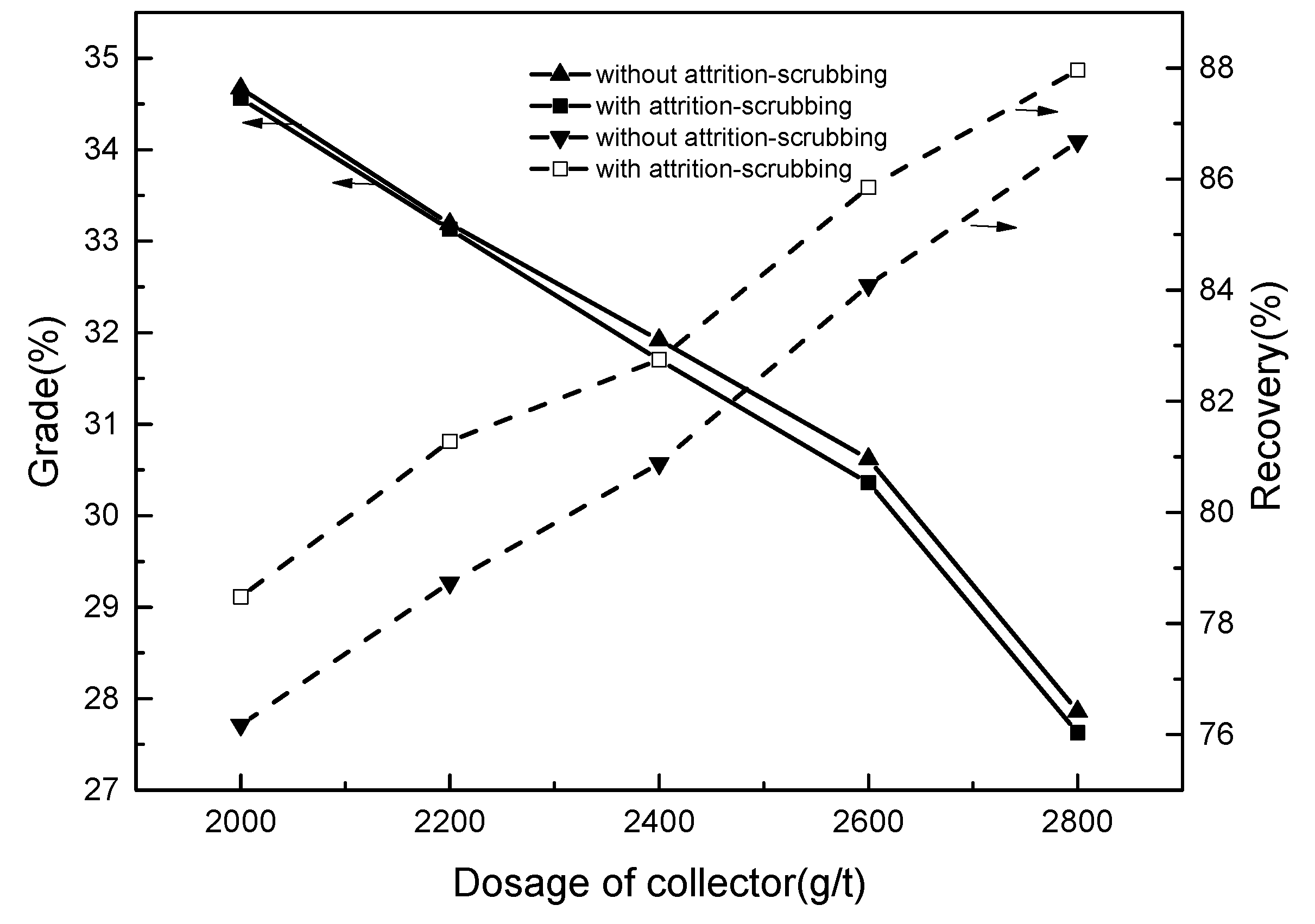
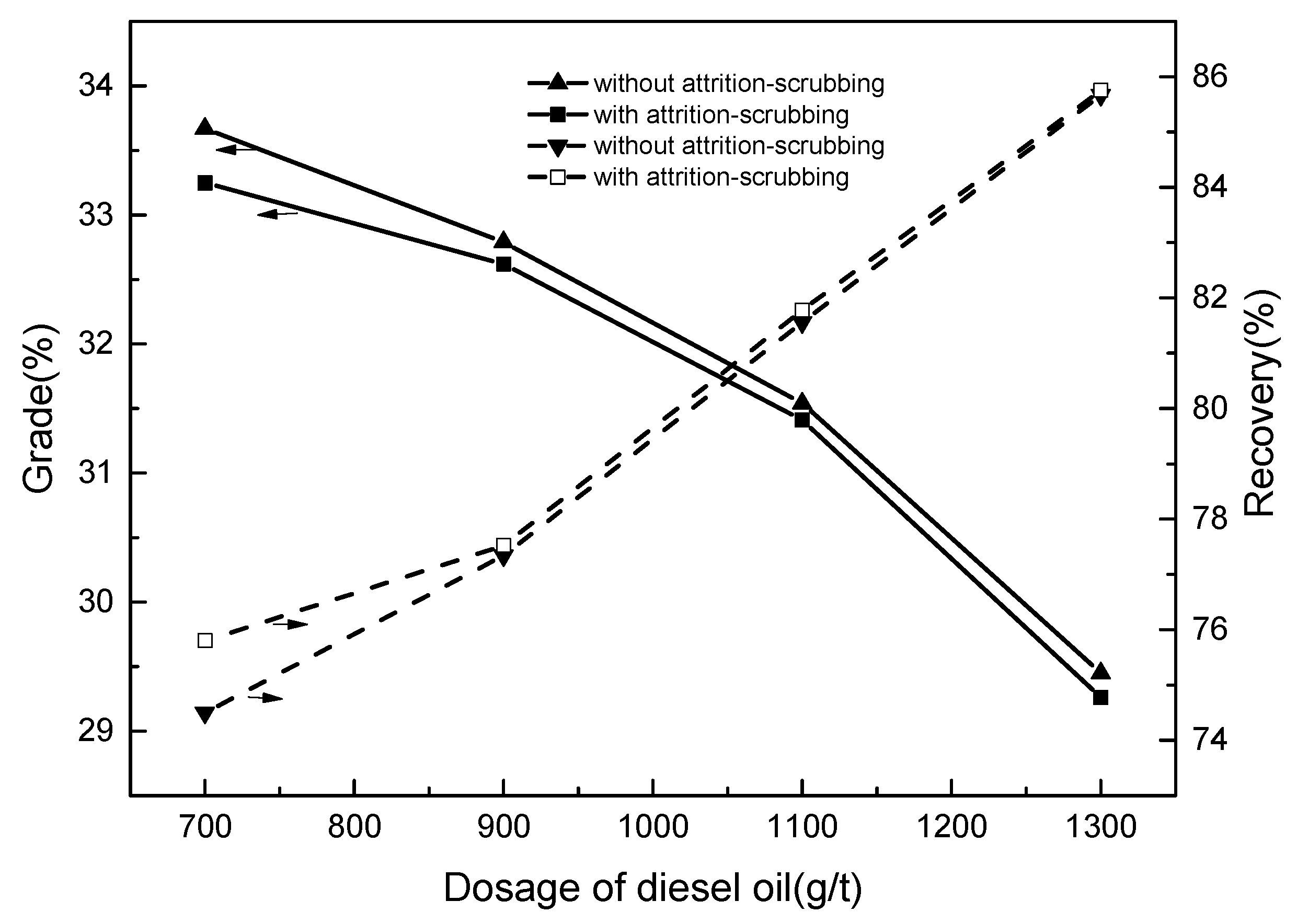
3.2.1. Flotation Condition Tests
3.2.2. Open-Circuit Contrast Tests
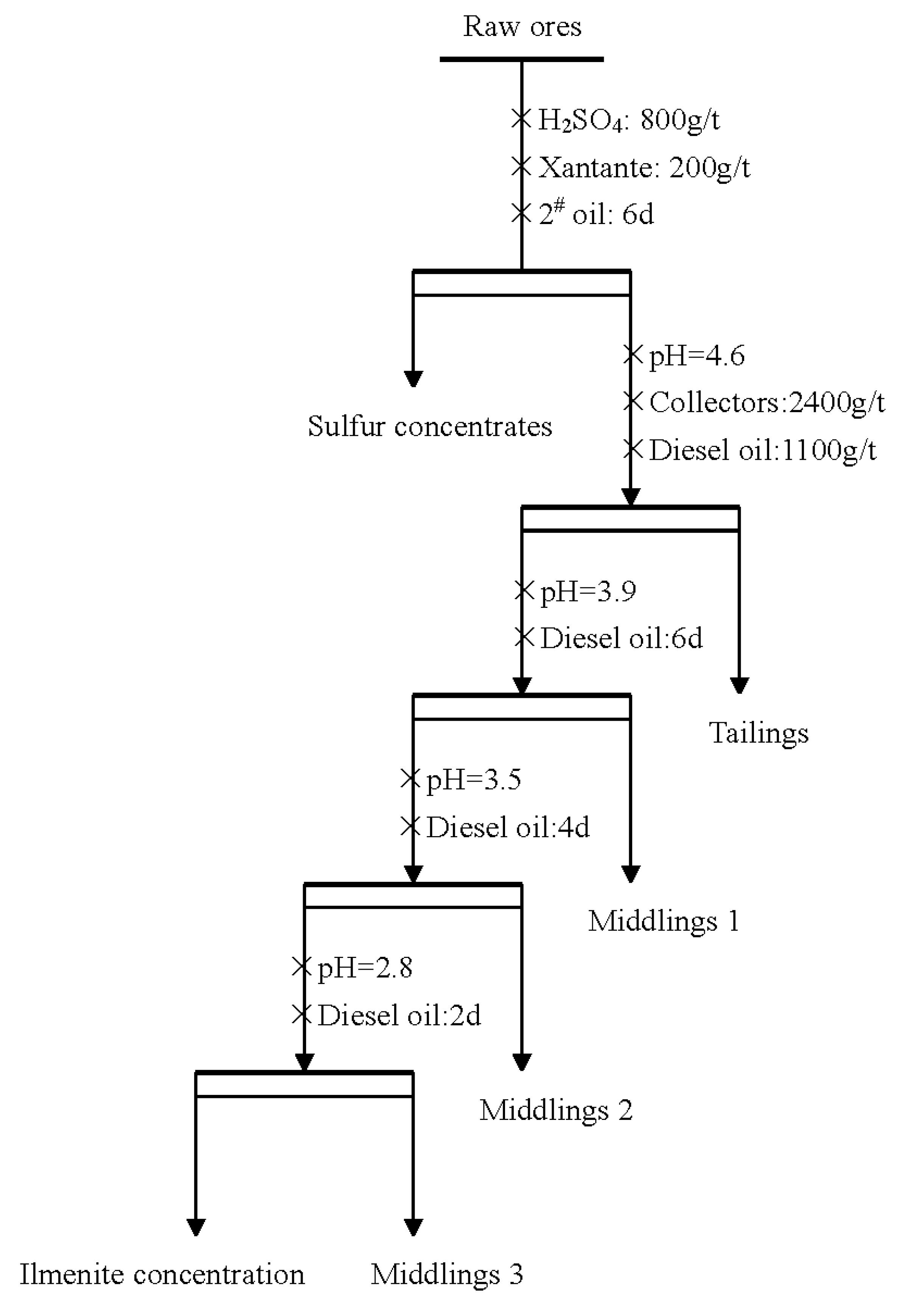
3.2.3. Closed-Circuit Test
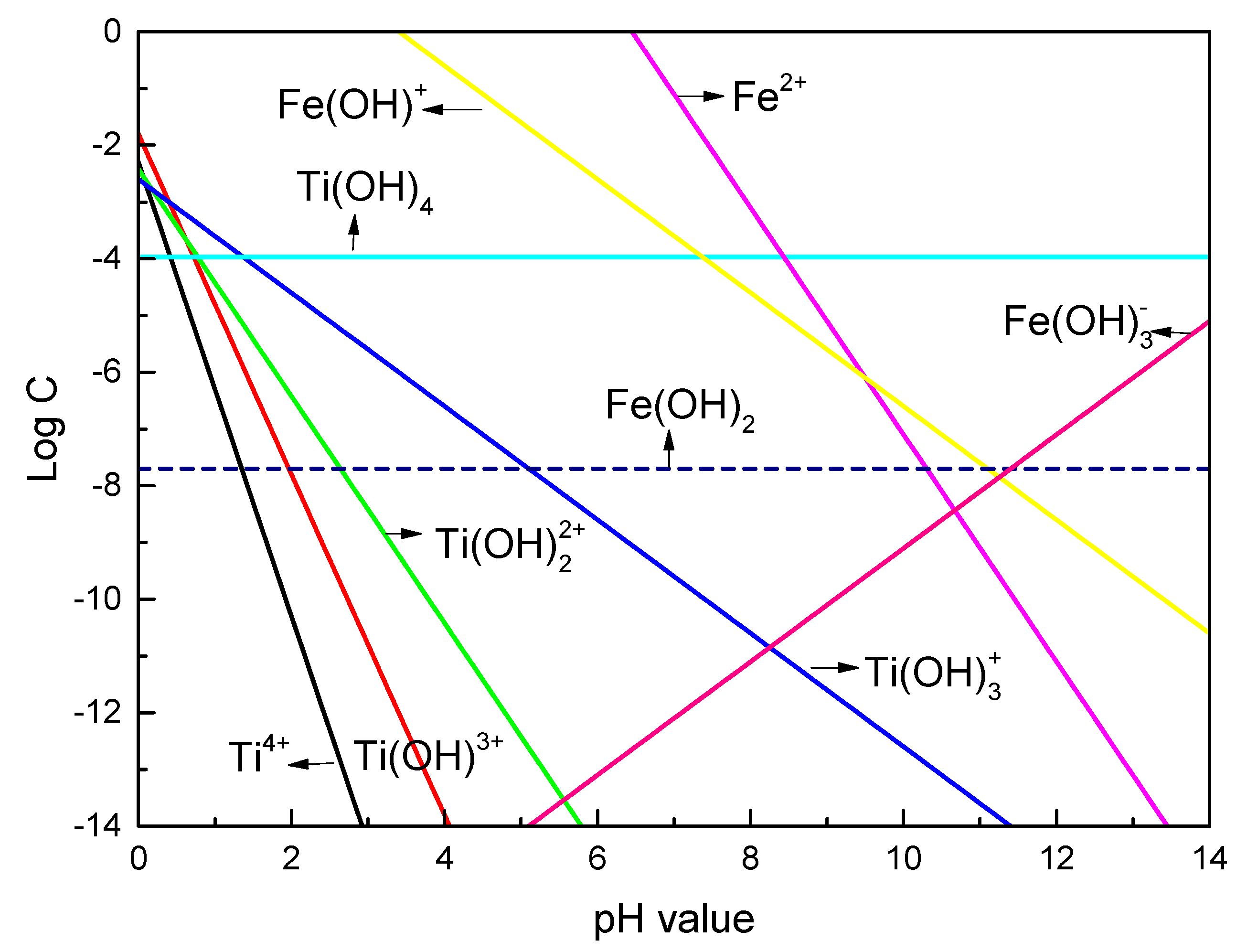
3.3. Discussions
4. Conclusions
- (1)
- The main phase of valuable titanium is ilmenite, which is present in the form of liberated grains of ilmenite and is the primary object for recovering;
- (2)
- During the optimization process of flotation conditions, compared to samples without attrition-scrubbing, attrition-scrubbing samples have superiority with respect to the effective flotation recovery of ilmenite. The results of open-circuit contrast tests further show that attrition-scrubbing can obtain a concentration with higher recovery and a slight decrease in grade. The results of closed-circuit tests show that a qualified concentrate with the grade of 47.06% and recovery of 60.73% can be produced. The better recovery and utilization of ilmenite can be realized; and
- (3)
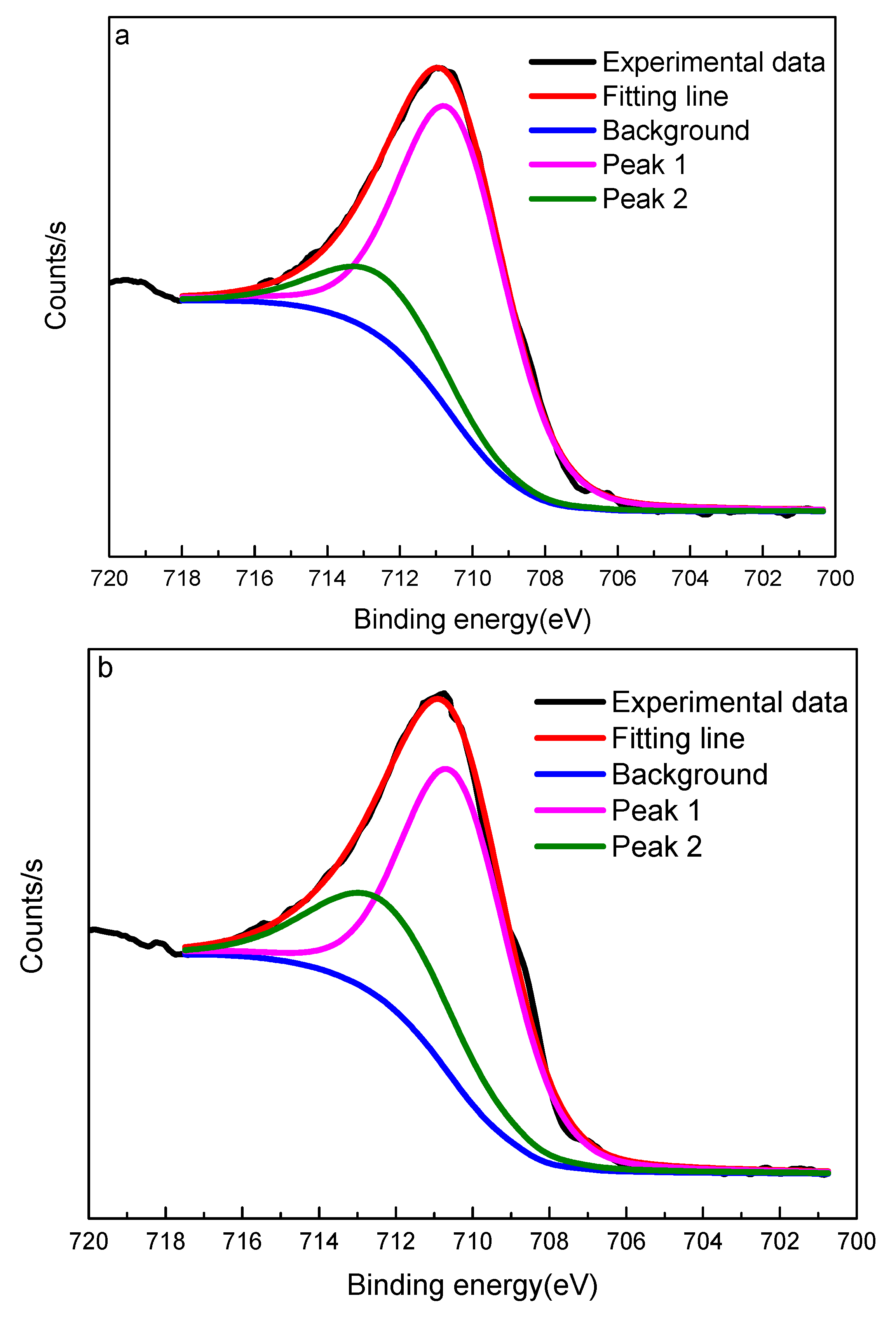
- After discussions, the reasons for the improvement in ilmenite flotability after treatment with attrition-scrubbing are as follows: one is that a fresh surface with more exposed active sites can be produced after attrition-scrubbing; the other is that during the process of attrition-scrubbing, some Fe(II) can convert into Fe(III) and this makes another critical contribution to the improvement in ilmenite flotation.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zorn, G.; Gotman, I.; Gutmanas, E.Y.; Adadi, R.; Salitra, G.; Sukenik, C.N. Surface modification of Ti45Nb alloy with an alkylphosphonic acid self-assembled monolayer. Chem. Mater. 2005, 17, 4218–4226. [Google Scholar] [CrossRef]
- Foster, T.T.; Alexander, M.R.; Leggett, G.J.; McAlpine, E. Friction force microscopy of alkylphosphonic acid and carboxylic acids adsorbed on the native oxide of aluminum. Langmuir 2006, 22, 9254–9259. [Google Scholar] [CrossRef] [PubMed]
- Quiñones, R.; Raman, A.; Gawalt, E.S. Functionalization of nickel oxide using alkylphosphonic acid self-assembled monolayers. Thin Solid Films 2008, 516, 8774–8781. [Google Scholar] [CrossRef]
- Wang, S.; Li, J.; Wu, S.; Yan, W.; Huang, W.X.; Miao, L.; Chen, Z. The distribution characteristics of rare metal elements in surface sediments from four coastal bays on the northwestern South China Sea. Estuar. Coast. Shelf Sci. 2015, 169, 106–118. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhang, G.; Feng, Q.; Yan, D.C.; Wang, W.Q. Effect of surface dissolution on flotation separation of fine ilmenite from titanaugite. Trans. Nonferr. Met. Soc. China 2011, 21, 1149–1154. [Google Scholar] [CrossRef]
- Cutmore, N.; Evans, T.; Crnokrak, D.; Middleton, A.; Stoddard, S. Microwave technique for analysis of mineral sands. Miner. Eng. 2000, 13, 729–736. [Google Scholar] [CrossRef]
- Zhang, G.; Ostrovski, O. Effect of preoxidation and sintering on properties of ilmenite concentrates. Int. J. Miner. Process. 2002, 64, 201–218. [Google Scholar] [CrossRef]
- Bryan, C.G.; Hallberg, K.B.; Johnson, D.B. Mobilisation of metals in mineral tailings at the abandoned São Domingos copper mine (Portugal) by indigenous acidophilic bacteria. Hydrometallurgy 2006, 83, 184–194. [Google Scholar] [CrossRef]
- Morin, D.H.R.; D’Hugues, P. Bioleaching of a Cobalt-Containing Pyrite in Stirred Reactors: A Case Study from Laboratory Scale to Industrial Application. In Biomining; Springer: Berlin, Germany, 2007; pp. 35–55. [Google Scholar]
- Lottermoser, B.G. Recycling, Reuse and Rehabilitation of Mine Wastes. Elements 2011, 7, 405–410. [Google Scholar] [CrossRef]
- Sarquís, P.E.; Menéndez-Aguado, J.M.; Mahamud, M.M.; Dzioba, R. Tannins: The organic depressants alternative in selective flotation of sulfides. J. Clean. Prod. 2014, 84, 723–726. [Google Scholar] [CrossRef]
- Fan, X.; Waters, K.E.; Rowson, N.A.; Paker, D.J. Modification of ilmenite surface chemistry for enhancing surfactants adsorption and bubble attachment. J. Colloid Interface Sci. 2009, 329, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Bulatovic, S.; Wyslouzil, D.M. Process development for treatment of complex perovskite, ilmenite and rutile ores. Miner. Eng. 1999, 12, 1407–1417. [Google Scholar] [CrossRef]
- Li, F.; Zhong, H.; Wang, S.; Liu, G.Y. The activation mechanism of Cu(II) to ilmenite and subsequent flotation response to α-hydroxyoctyl phosphinic acid. J. Ind. Eng. Chem. 2016, 37, 123–130. [Google Scholar] [CrossRef]
- Zhou, M.F.; Chen, W.T.; Wang, C.Y.; Prevec, S.A.; Liu, P.P.P.; Howarth, G.H. Two stages of immiscible liquid separation in the formation of Panzhihua-type Fe-Ti-V oxide deposits, SW China. Geosci. Front. 2013, 4, 481–502. [Google Scholar] [CrossRef]
- Zhao, J.H.; Zhou, M.F. Geochemistry of Neoproterozoic mafic intrusions in the Panzhihua district (Sichuan Province, SW China): Implications for subduction-related metasomatism in the upper mantle. Precambr. Res. 2007, 152, 27–47. [Google Scholar] [CrossRef]
- Welham, N.J.; Llewellyn, D.J. Mechanical enhancement of the dissolution of ilmenite. Miner. Eng. 1998, 11, 827–841. [Google Scholar] [CrossRef]
- Mehdilo, A.; Irannajad, M.; Rezai, B. Effect of crystal chemistry and surface properties on ilmenite flotation behavior. Int. J. Miner. Process. 2015, 137, 71–81. [Google Scholar] [CrossRef]
- Bayley, R.W.; Biggs, C.A. Characterisation of an attrition scrubber for the removal of high molecular weight contaminants in sand. Chem. Eng. J. 2005, 111, 71–79. [Google Scholar] [CrossRef]
- Petavy, F.; Ruban, V.; Conil, P. Treatment of stormwater sediments: Efficiency of an attrition scrubber–laboratory and pilot-scale studies. Chem. Eng. J. 2009, 145, 475–482. [Google Scholar] [CrossRef]
- Zhong, K.; Cui, L. Influence of Fe2+ ions of ilmenite on its flotability. Int. J. Miner. Process. 1987, 20, 253–265. [Google Scholar] [CrossRef]
- Irannajad, M.; Mehdilo, A.; Nuri, O.S. Influence of microwave irradiation on ilmenite flotation behavior in the presence of different gangue minerals. Sep. Purif. Technol. 2014, 132, 401–412. [Google Scholar] [CrossRef]
- Mehdilo, A.; Irannajad, M. Comparison of microwave irradiation and oxidation roasting as pretreatment methods for modification of ilmenite physicochemical properties. J. Ind. Eng. Chem. 2015, 33, 59–72. [Google Scholar] [CrossRef]

| Particle Size/Mesh | Liberation Degree of Ilmenite (%) |
|---|---|
| +40 | 38.86 |
| +60 | 62.33 |
| +80 | 78.55 |
| +100 | 85.29 |
| +120 | 88.26 |
| +160 | 91.17 |
| +200 | 95.02 |
| −200 | 96.31 |
| Component | TFe | TiO2 | V2O5 | SiO2 | Al2O3 | CaO | MgO | K2O | Na2O | S |
|---|---|---|---|---|---|---|---|---|---|---|
| Content (%) | 19.43 | 17.23 | 0.032 | 30.00 | 6.33 | 8.90 | 12.41 | 0.049 | 0.57 | 0.56 |
| Products | Iron Sulfide | Hematite | Titanomagnetite | Ilmenite | Carbonate Iron | Silicate Iron | Total |
|---|---|---|---|---|---|---|---|
| Content | 0.02 | 5.69 | 0.99 | 9.41 | 0.81 | 2.51 | 19.43 |
| Distribution | 0.10 | 29.28 | 5.10 | 48.43 | 4.17 | 12.92 | 100.00 |
| Products | Titanomagnetite | Ilmenite | Sphene | Total (TiO2) |
|---|---|---|---|---|
| Content | 0.70 | 15.48 | 1.05 | 17.23 |
| Distribution | 4.07 | 89.82 | 6.11 | 100.00 |
| Products | Grain Size (mm) | Yield (%) | Grade (%) | Distribution (%) |
|---|---|---|---|---|
| Untreated Samples | +0.25 | 1.14 | 9.75 | 0.65 |
| +0.18 | 2.27 | 13.86 | 1.83 | |
| +0.154 | 2.99 | 14.86 | 2.58 | |
| +0.125 | 11.56 | 16.03 | 10.77 | |
| +0.098 | 14.34 | 15.75 | 13.13 | |
| +0.074 | 9.29 | 14.74 | 7.96 | |
| +0.045 | 22.6 | 15.65 | 20.56 | |
| +0.038 | 4.75 | 17.87 | 4.92 | |
| −0.038 | 31.06 | 20.83 | 37.6 | |
| ∑ | 100 | 17.21 | 100 |
| Products | Grain Size (mm) | Yield (%) | Grade (%) | Distribution (%) |
|---|---|---|---|---|
| Attrition-scrubbing samples | +0.25 | 1.21 | 9.83 | 0.69 |
| +0.18 | 2.34 | 13.85 | 1.88 | |
| +0.154 | 3.05 | 14.93 | 2.64 | |
| +0.125 | 10.84 | 15.76 | 9.92 | |
| +0.098 | 14.15 | 15.82 | 12.99 | |
| +0.074 | 9.34 | 14.79 | 8.02 | |
| +0.045 | 22.87 | 15.68 | 20.81 | |
| +0.038 | 4.82 | 17.92 | 5.01 | |
| −0.038 | 31.38 | 20.87 | 38.04 | |
| ∑ | 100 | 17.23 | 100.00 |
| Product | Yield (%) | Grade (%) | Recovery (%) |
|---|---|---|---|
| Sulfur concentrate | 5.09 | 10.53 | 3.12 |
| Concentrate | 17.32 | 47.31 | 47.64 |
| Middling 1 | 12.12 | 15.62 | 11.01 |
| Middling 2 | 7.56 | 24.37 | 10.71 |
| Middling 3 | 5.01 | 34.51 | 10.05 |
| Tailing | 52.9 | 5.68 | 17.47 |
| Raw ore | 100 | 17.2 | 100 |
| Product | Yield (%) | Grade (%) | Recovery (%) |
|---|---|---|---|
| Sulfur concentrate | 5.24 | 10.47 | 3.18 |
| Concentrate | 20.13 | 47.21 | 55.16 |
| Middling 1 | 9.12 | 17.01 | 9 |
| Middling 2 | 6.26 | 23.88 | 8.68 |
| Middling 3 | 3.86 | 32.65 | 7.31 |
| Tailing | 55.39 | 5.18 | 16.67 |
| Raw ore | 100 | 17.23 | 100 |
| Product | Yield (%) | Grade (%) | Recovery (%) |
|---|---|---|---|
| Sulfur concentrate | 5.39 | 10.28 | 3.21 |
| Concentrate | 22.26 | 47.06 | 60.73 |
| Tailing | 72.35 | 8.60 | 36.06 |
| Raw ore | 100 | 17.25 | 100 |
| Sample | Binding Energy (eV) | Percentage in Total Fe (%) | ||
|---|---|---|---|---|
| Fe2+/Peak 1 | Fe3+/Peak 2 | Fe2+/Peak 1 | Fe3+/Peak 2 | |
| Without attrition-scrubbing | 710.5 | 712.5 | 81.33 | 18.67 |
| With attrition-scrubbing | 710.4 | 712.3 | 70.37 | 29.63 |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhai, J.; Chen, P.; Wang, H.; Hu, Y.; Sun, W. Flotability Improvement of Ilmenite Using Attrition-Scrubbing as a Pretreatment Method. Minerals 2017, 7, 13. https://doi.org/10.3390/min7010013
Zhai J, Chen P, Wang H, Hu Y, Sun W. Flotability Improvement of Ilmenite Using Attrition-Scrubbing as a Pretreatment Method. Minerals. 2017; 7(1):13. https://doi.org/10.3390/min7010013
Chicago/Turabian StyleZhai, Jihua, Pan Chen, Hongbin Wang, Yuehua Hu, and Wei Sun. 2017. "Flotability Improvement of Ilmenite Using Attrition-Scrubbing as a Pretreatment Method" Minerals 7, no. 1: 13. https://doi.org/10.3390/min7010013
APA StyleZhai, J., Chen, P., Wang, H., Hu, Y., & Sun, W. (2017). Flotability Improvement of Ilmenite Using Attrition-Scrubbing as a Pretreatment Method. Minerals, 7(1), 13. https://doi.org/10.3390/min7010013






