Thermal Transformation of NH4-Clinoptilolite to Mullite and Silica Polymorphs
Abstract
:1. Introduction
2. Experimental Section
2.1. Starting Material and Beneficiation Process
2.2. Preparation of NH4-Clinoptilolite
2.3. Chemical Analysis
2.4. Thermal Treatments
2.5. X-ray Diffraction (XRD)
2.6. Thermal Analyses
2.7. Scanning Electron Microscope (SEM) Observations
3. Results
4. Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Eyde, T.H.; Holmes, D.A. Zeolites. In Industrial Minerals and Rocks: Commodities, Markets, and Uses, 7th ed.; Kogel, J.E., Trivedi, N.C., Barker, J.M., Krukowski, S.T., Eds.; Society for Mining, Metallurgy, and Exploration, Inc.: Littleton, CO, USA, 2006; pp. 1039–1064. [Google Scholar]
- Pabalan, R.; Bertetti, F.P. Cation-Exchange Properties of Natural Zeolites. In Natural Zeolites: Occurrence, Properties, Applications—Reviews in Mineralogy and Geochemistry, 1st ed.; Bish, D.L., Ming, D.W., Eds.; Mineralogical Society of America: Washington, DC, USA, 2001; Volume 45, pp. 453–518. [Google Scholar]
- Jorgensen, T.C.; Weatherley, L.R. Ammonia removal from wastewater by ion exchange in the presence of organic contaminants. Water Res. 2003, 37, 1723–1738. [Google Scholar] [CrossRef]
- Sprynskyy, M.; Lebedynets, M.; Terzyk, A.P.; Kowalczyk, J.; Namieśnik, J.; Buszewski, B. Ammonium sorption from aqueous solutions by the natural zeolite Transcarpathian clinoptilolite studied under dynamic conditions. J. Colloid Interface Sci. 2005, 284, 408–415. [Google Scholar] [CrossRef] [PubMed]
- Mazloomi, F.; Jalali, M. Ammonium removal from aqueous solutions by natural Iranian zeolite in the presence of organic acids, cations and anions. J. Environ. Chem. Eng. 2016, 4, 240–249. [Google Scholar] [CrossRef]
- Virta, R.L.; Flanagan, D.M. Zeolites. In Minerals Yearbook 2014; U.S. Geological Survey: Reston, VA, USA, 2015; Volume 1, pp. 83.1–83.3. [Google Scholar]
- Flanagan, D.M. Zeolites. In Mineral Commodity Summaries 2016; U.S. Geological Survey: Reston, VA, USA, 2016; pp. 190–191. [Google Scholar]
- Armbruster, T. Clinoptilolite-heulandite: Applications and basic research. Stud. Surf. Sci. Catal. 2001, 135, 13–27. [Google Scholar]
- Liberti, L.; Boghetich, G.; Lopez, A.; Petruzzelli, D. Application of microporous materials for the recovery of nutrients from wastewaters. In Natural Microporous Materials in Environmental Technology—Nato Science Series, 1st ed.; Misaelides, P., Macášek, F., Pinnavaia, T.J., Colella, C., Eds.; Springer Science & Business Media, B.V.: Dordrecht, The Netherlands, 1999; Volume 362, pp. 253–270. [Google Scholar]
- Cruciani, G. Zeolites upon heating: Factors governing their thermal stability and structural changes. J. Phys. Chem. Solids 2006, 67, 1973–1994. [Google Scholar] [CrossRef]
- Matsumoto, T.; Goto, Y.; Urabe, K. Formation process of mullite from NH4+-exchanged Zeolite A. J. Ceram. Soc. Jpn. 1995, 103, 93–95. [Google Scholar] [CrossRef]
- Kosanović, C.; Subotić, B.; Smit, I. Thermally induced phase transformations in cation-exchanged zeolites 4A, 13X and synthetic mordenite and their amorphous derivatives obtained by mechanochemical treatment. Thermochim. Acta 1998, 317, 25–37. [Google Scholar] [CrossRef]
- Kosanović, C.; Subotić, B. Preparation of mullite micro-vessels by a combined treatment of zeolite A. Microporous Mesoporous Mater. 2003, 66, 311–319. [Google Scholar] [CrossRef]
- Schneider, H.; Schreuer, J.; Hildmann, B. Structure and properties of mullite—A review. J. Eur. Ceram. Soc. 2008, 28, 329–344. [Google Scholar] [CrossRef]
- De Gennaro, R.; Cappelletti, P.; Cerri, G.; de’ Gennaro, M.; Dondi, M.; Guarini, G.; Langella, A.; Naimo, D. Influence of zeolites on the sintering and technological properties of porcelain stoneware tiles. J. Eur. Ceram. Soc. 2003, 23, 2237–2245. [Google Scholar] [CrossRef]
- De Gennaro, R.; Dondi, M.; Cappelletti, P.; Cerri, G.; de’ Gennaro, M.; Guarini, G.; Langella, A.; Parlato, L.; Zanelli, C. Zeolite-feldspar epiclastic rocks as flux in ceramic tile manufacturing. Microporous Mesoporous Mater. 2007, 105, 273–278. [Google Scholar] [CrossRef]
- Demirkiran, A.Ş.; Artir, R.; Avci, E. Effect of natural zeolite addition on sintering kinetics of porcelain bodies. J. Mater. Process. Tech. 2008, 203, 465–470. [Google Scholar] [CrossRef]
- Ergul, S.; Sappa, G.; Magaldi, D.; Pisciella, P.; Pelino, M. Microstructural and phase transformations during sintering of a phillipsite rich zeolitic tuff. Ceram. Int. 2011, 37, 1843–1850. [Google Scholar] [CrossRef]
- Sokolář, R.; Šveda, M. The use of zeolite as fluxing agent for whitewares. Procedia Eng. 2016, 151, 229–235. [Google Scholar] [CrossRef]
- Brundu, A.; Cerri, G. Thermal transformation of Cs-clinoptilolite to CsAlSi5O12. Microporous Mesoporous Mater. 2015, 208, 44–49. [Google Scholar] [CrossRef]
- Gatta, G.D.; Brundu, A.; Cappelletti, P.; Cerri, G.; de’ Gennaro, B.; Farina, M.; Fumagalli, P.; Guaschino, L.; Lotti, P.; Mercurio, M. New insights on pressure, temperature, and chemical stability of CsAlSi5O12, a potential host for nuclear waste. Phys. Chem. Miner. 2016, 43, 639–647. [Google Scholar] [CrossRef]
- Brundu, A.; Cerri, G. Release of lead from Pb-clinoptilolite: managing the fate of an exhausted exchanger. Int. J. Environ. Sci. Technol. 2016. [Google Scholar] [CrossRef]
- Tomazović, B.; Ćeranić, T.; Sijarić, G. The properties of the NH4-clinoptilolite. Part 2. Zeolites 1996, 16, 309–312. [Google Scholar] [CrossRef]
- Andreola, F.; Barbieri, L.; Karamanova, E.; Lancellotti, I.; Pelino, M. Recycling of CRT panel glass as fluxing agent in the porcelain stoneware tile production. Ceram. Int. 2008, 34, 1289–1295. [Google Scholar] [CrossRef]
- Dondi, M.; Guarini, G.; Raimondo, M.; Zanelli, C. Recycling PC and TV waste glass in clay bricks and roof tiles. Waste Manag. 2009, 29, 1945–1951. [Google Scholar] [CrossRef] [PubMed]
- Andreola, F.; Barbieri, L.; Lancellotti, I.; Leonelli, C.; Manfredini, T. Recycling of industrial wastes in ceramic manufacturing: State of art and glass case studies. Ceram. Int. 2016, 42, 13333–13338. [Google Scholar] [CrossRef]
- Cerri, G.; Cappelletti, P.; Langella, A.; de’ Gennaro, M. Zeolitization of Oligo-Miocene volcaniclastic rocks from Logudoro (northern Sardinia, Italy). Contrib. Mineral. Petrol. 2001, 140, 404–421. [Google Scholar] [CrossRef]
- Cerri, G.; de’ Gennaro, M.; Bonferoni, M.C.; Caramella, C. Zeolites in biomedical application: Zn-exchanged clinoptilolite-rich rock as active carrier for antibiotics in anti-acne topical therapy. Appl. Clay Sci. 2004, 27, 141–150. [Google Scholar] [CrossRef]
- Brundu, A.; Cerri, G.; Colella, A.; de Gennaro, M. Effects of thermal treatments on Pb-clinoptilolite. Rend. Online Soc. Geol. Italy 2008, 3, 138–139. [Google Scholar]
- Cerri, G.; Farina, M.; Brundu, A.; Daković, A.; Giunchedi, P.; Gavini, E.; Rassu, G. Natural zeolites for pharmaceutical formulations: Preparation and evaluation of a clinoptilolite-based material. Microporous Mesoporous Mater. 2016, 223, 58–67. [Google Scholar] [CrossRef]
- Kesraoul-Oukl, S.; Cheeseman, C.; Perry, R. Effects of conditioning and treatment of chabazite and clinoptilolite prior to lead and cadmium removal. Environ. Sci. Technol. 1993, 27, 1108–1116. [Google Scholar] [CrossRef]
- Tomazović, B.; Ćeranić, T.; Sijarić, G. The properties of the NH4-clinoptilolite. Part 1. Zeolites 1996, 16, 301–308. [Google Scholar] [CrossRef]
- Jacobs, P.A.; Uytterhoeven, J.B.; Beyer, H.K.; Kiss, A. Preparation and Properties of Hydrogen Form of Stilbite, Heulandite and Clinoptilolite Zeolites. J. Chem. Soc. Faraday Trans. 1 1979, 75, 883–891. [Google Scholar] [CrossRef]
- Langella, A.; Pansini, M.; Cerri, G.; Cappelletti, P.; de Gennaro, M. Thermal behavior of natural and cation-exchanged clinoptilolite from Sardinia (Italy). Clays Clay Min. 2003, 51, 625–633. [Google Scholar] [CrossRef]
- Stoch, L.; Waclawska, I. Phase Transformations in Amorphous Solids. High Temp. Mater. Proc. 1994, 13, 181–201. [Google Scholar] [CrossRef]
- Manning, D.A.C. Introduction to Industrial Minerals, 1st ed.; Chapman & Hall: London, UK, 1995; p. 276. [Google Scholar]
- Coombs, D.S.; Alberti, A.; Armbruster, T.; Artioli, G.; Colella, C.; Galli, E.; Grice, J.D.; Liebau, F.; Mandarino, J.A.; Minato, H.; et al. Recommended nomenclature for zeolite minerals: Report of the Subcommittee on Zeolites of the International Mineralogical Association, Commission on New Minerals and Mineral Names. Can. Miner. 1997, 35, 1571–1606. [Google Scholar]
- Bish, D.L.; Boak, J.M. Clinoptilolite-Heulandite Nomenclature. In Natural Zeolites: Occurrence, Properties, Applications—Reviews in Mineralogy and Geochemistry, 1st ed.; Bish, D.L., Ming, D.W., Eds.; Mineralogical Society of America: Washington, DC, USA, 2001; Volume 45, pp. 207–216. [Google Scholar]
- Radosavljevic-Mihajlovic, A.S.; Kremenovic, A.S.; Dosen, A.M.; Andrejic, J.Z.; Dondur, V.T. Thermally induced phase transformation of Pb-exchanged LTA and FAU-framework zeolite to feldspar phases. Microporous Mesoporous Mater. 2015, 201, 210–218. [Google Scholar] [CrossRef]
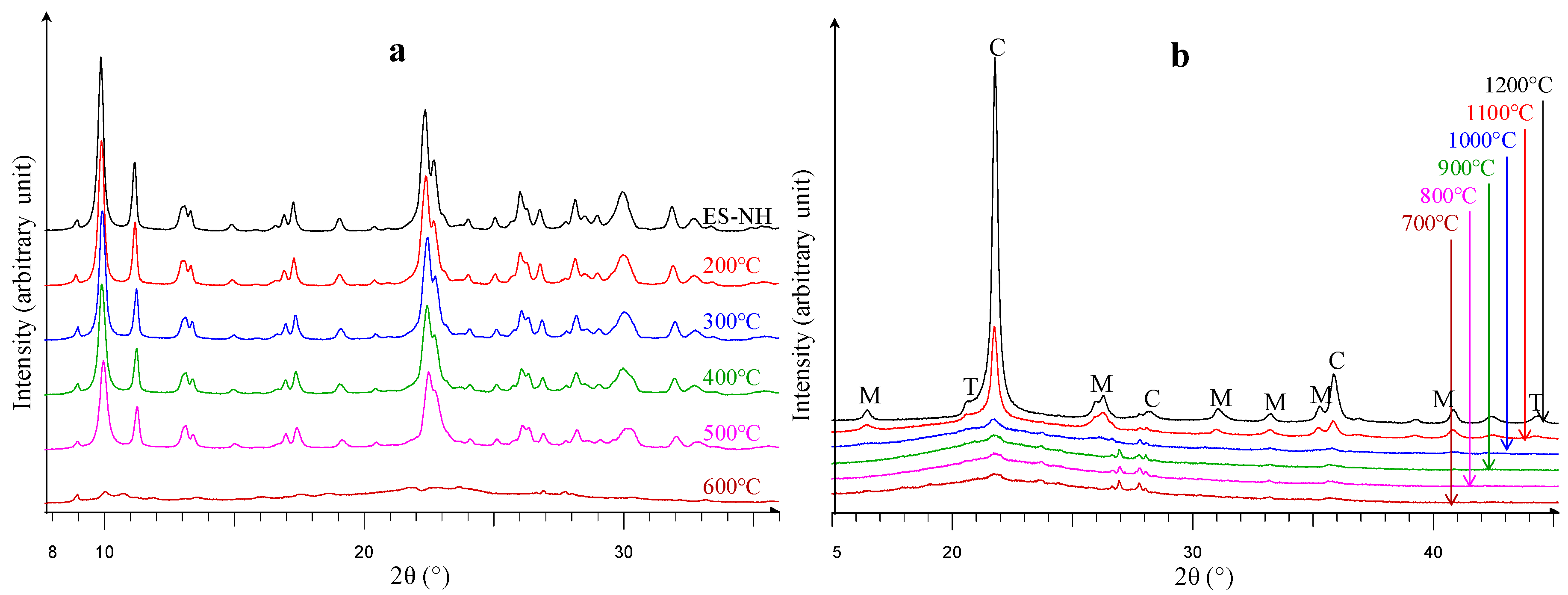

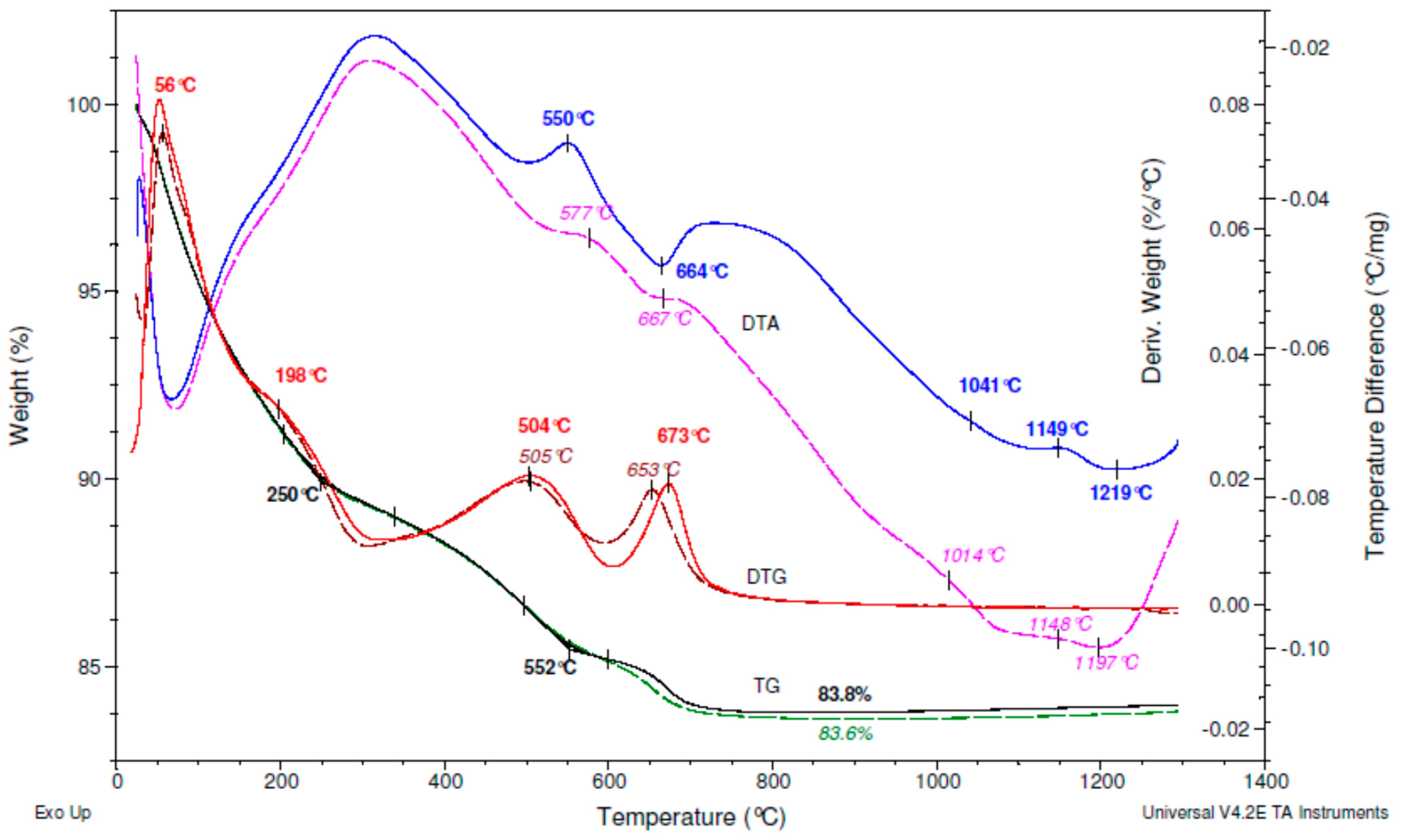

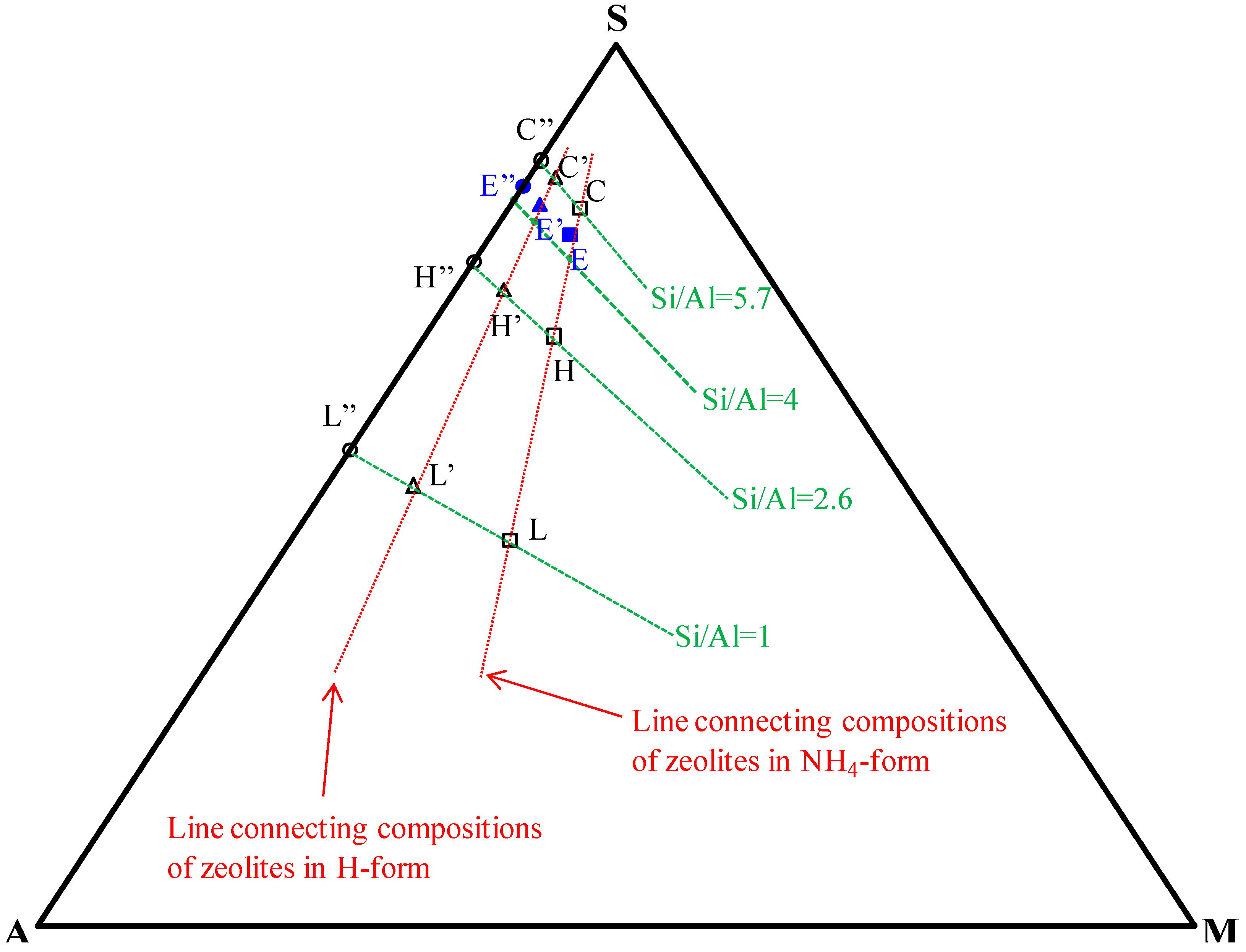
| ES-AR | Clinoptilolite | Feldspars | Quartz | Opal-CT | Biotite | Amorphous | Sum |
|---|---|---|---|---|---|---|---|
| content | 89.3 | 4.0 | 0.7 | 2.1 | 1.0 | 2.9 | 100.0 |
| e.s.d. | ±4.0 | ±1.0 | ±0.2 | ±0.5 | ±0.2 | ±1.2 | - |
| SiO2 | Al2O3 | Fe2O3 | MnO | MgO | CaO | Na2O | K2O | TiO2 | P2O5 | (NH4)2O | H2O | Sum |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 67.85 | 12.81 | 0.78 | 0.01 | 0.39 | 0.30 | 0.14 | 0.46 | 0.23 | 0.05 | 5.76 | 10.92 | 99.70 |
| Temperature | Time (h) | Cristobalite | Tridymite | Mullite | Amorphous | Sum | Rwp |
|---|---|---|---|---|---|---|---|
| 1000 °C | 32 | 5.1 | 3.5 | 5.2 | 86.2 | 100.0 | 3.91 |
| e.s.d. | - | ±1.0 | ±0.7 | ±1.0 | ±6.0 | - | - |
| 1100 °C | 32 | 59.3 | 10.1 | 21.8 | 8.8 | 100.0 | 7.97 |
| e.s.d. | - | ±2.8 | ±1.6 | ±1.8 | ±2.0 | - | - |
| 1200 °C | 2 | 54.3 | 8.8 | 21.0 | 15.9 | 100.0 | 8.18 |
| e.s.d. | - | ±2.5 | ±1.4 | ±1.8 | ±2.5 | - | - |
| Sample | SiO2 | Al2O3 | Fe2O3 | MnO | MgO | CaO | Na2O | K2O | TiO2 | P2O5 | M2O | Sum |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| D-1 | 76.43 | 14.43 | 0.88 | 0.01 | 0.44 | 0.34 | 0.16 | 0.52 | 0.26 | 0.06 | 6.48 a | 100.00 |
| D-2 | 79.81 | 15.07 | 0.92 | 0.01 | 0.46 | 0.35 | 0.17 | 0.54 | 0.27 | 0.06 | 2.34 b | 100.00 |
| D-3 | 81.73 | 15.43 | 0.94 | 0.01 | 0.47 | 0.36 | 0.17 | 0.56 | 0.27 | 0.06 | - | 100.00 |
| Mul | 28.20 | 71.80 | - | - | - | - | - | - | - | - | - | 100.00 |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brundu, A.; Cerri, G.; Sale, E. Thermal Transformation of NH4-Clinoptilolite to Mullite and Silica Polymorphs. Minerals 2017, 7, 11. https://doi.org/10.3390/min7010011
Brundu A, Cerri G, Sale E. Thermal Transformation of NH4-Clinoptilolite to Mullite and Silica Polymorphs. Minerals. 2017; 7(1):11. https://doi.org/10.3390/min7010011
Chicago/Turabian StyleBrundu, Antonio, Guido Cerri, and Eleonora Sale. 2017. "Thermal Transformation of NH4-Clinoptilolite to Mullite and Silica Polymorphs" Minerals 7, no. 1: 11. https://doi.org/10.3390/min7010011
APA StyleBrundu, A., Cerri, G., & Sale, E. (2017). Thermal Transformation of NH4-Clinoptilolite to Mullite and Silica Polymorphs. Minerals, 7(1), 11. https://doi.org/10.3390/min7010011






