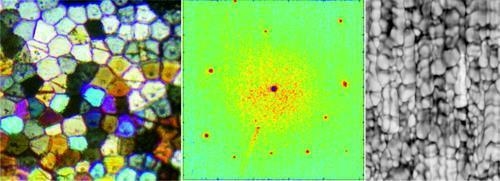
Evidence of a Biological Control over Origin, Growth and End of the Calcite Prisms in the Shells of Pinctada margaritifera (Pelecypod, Pterioidea)
Abstract
:1. Introduction

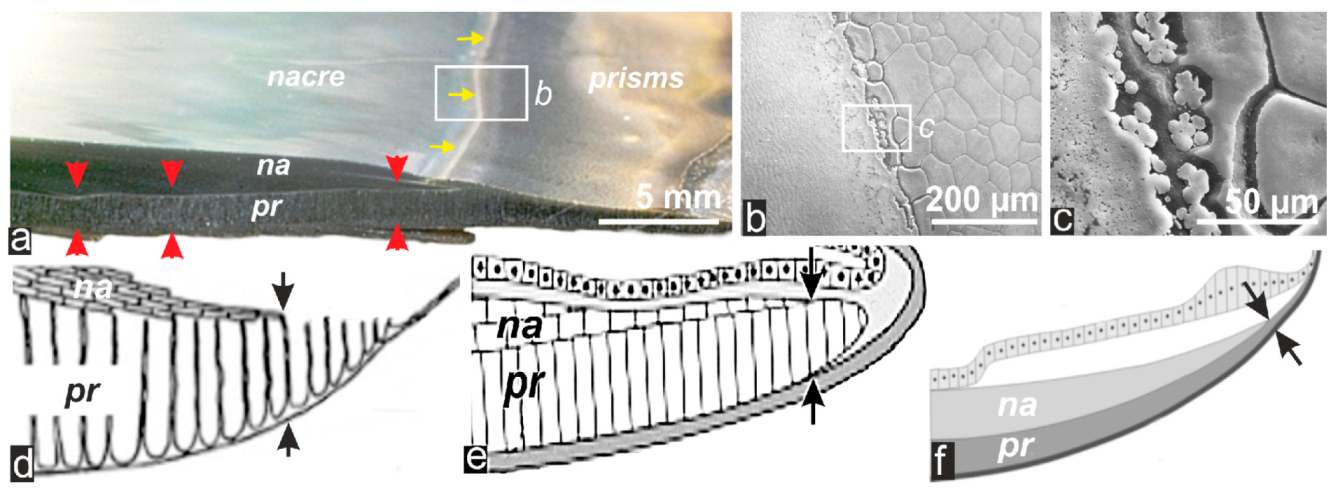
2. Results: The Sequential Growth of the Prisms from Their Early Stage to Their End
- (1)
- Formation of initial calcite disks as isolated and separately growing units;
- (2)
- First prismatic stage exhibiting a single crystal-like behavior and a synchronous layered thickening;
- (3)
- Passage to a polycrystalline prismatic structure;
- (4)
- Progressive degradation of the mineralizing metabolism and final coverage of the prism growing surface by a specifically secreted organic layer, on which aragonite deposition commences.
2.1. Formation of Calcareous Disks at the Shell Growth Margin: The Initial Stage in Calcification of the Prismatic Layer
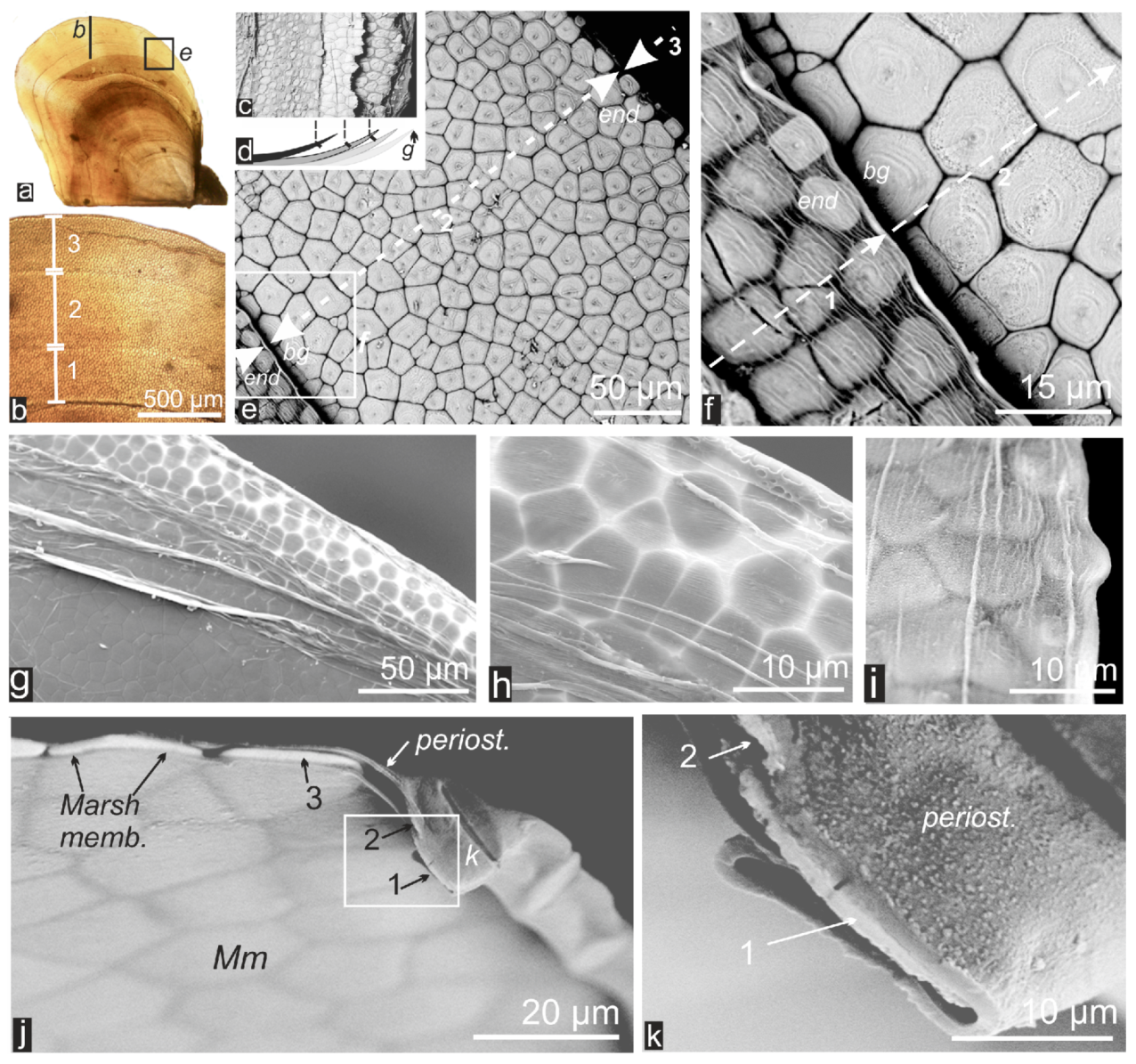
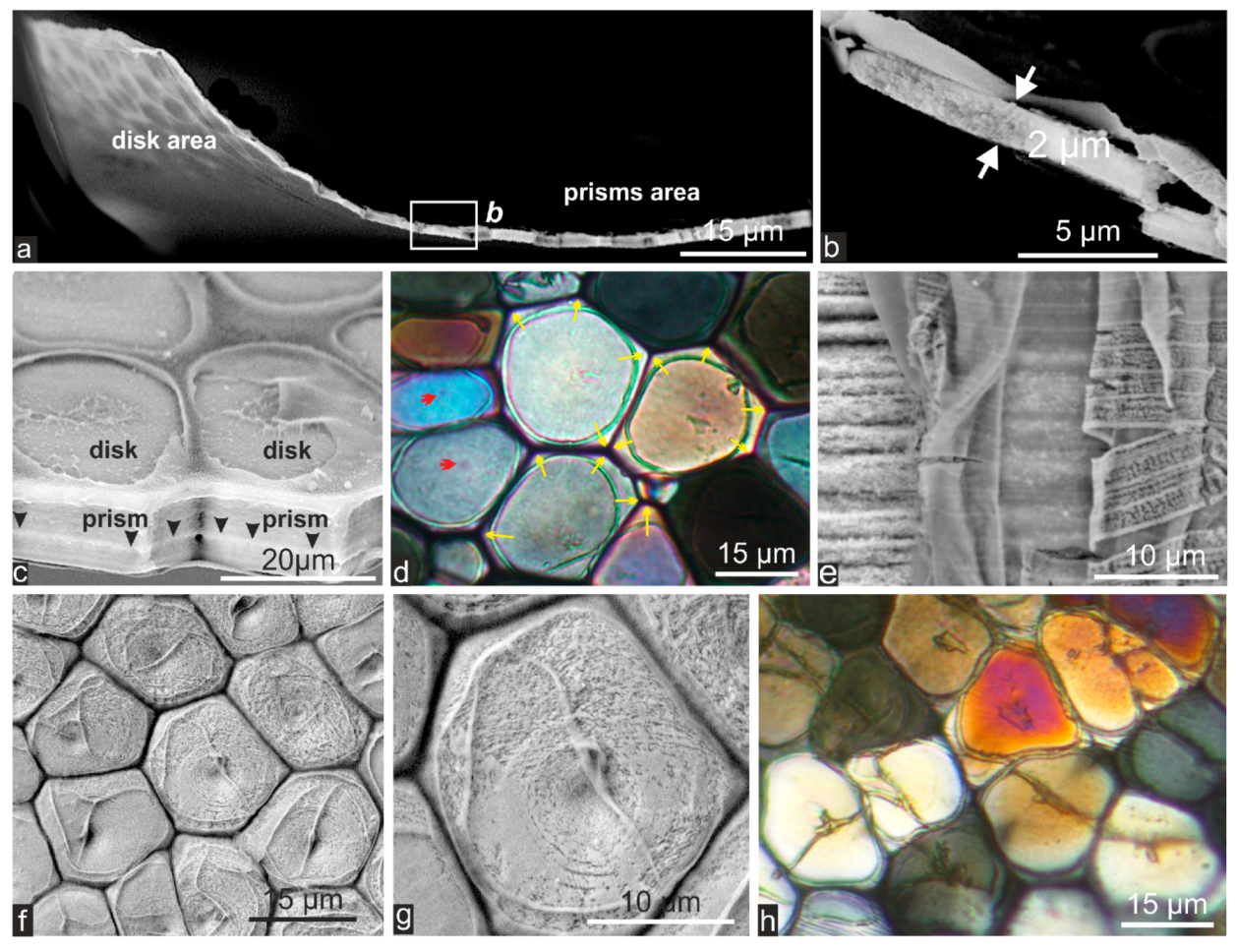
2.2. Passage from Laterally Growing Disks to the Layered Growth Mode of the Prisms
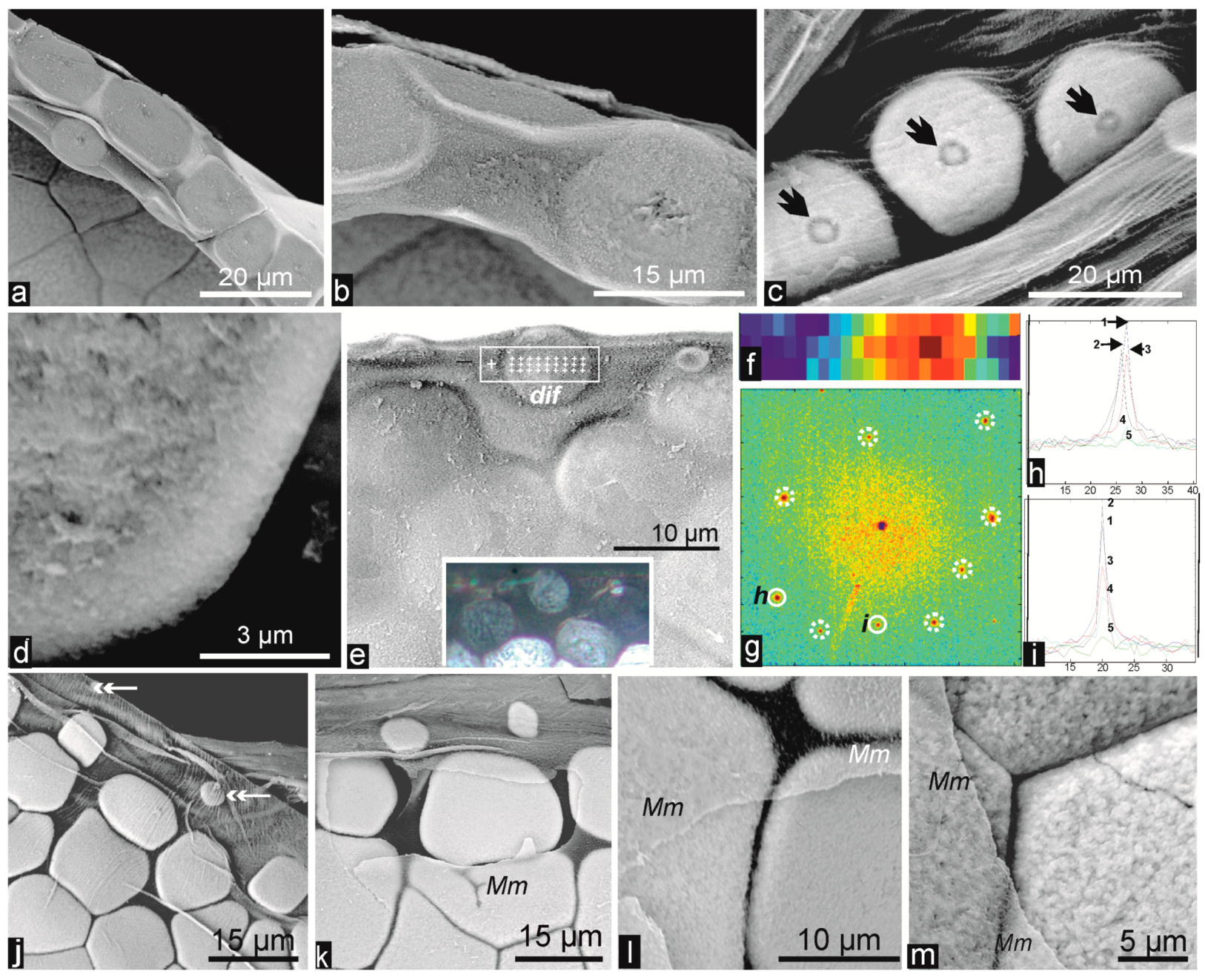
2.3. Microstructural Change during Prism Growth: Passage from a Single-Crystal to Polycrystalline Microstructure
2.4. Structural and Biochemical Changes Occurring at the Distal Growth Stages of the Prisms, Predating Nacre Deposition


3. Discussion
3.1. The Early Calcification Stages
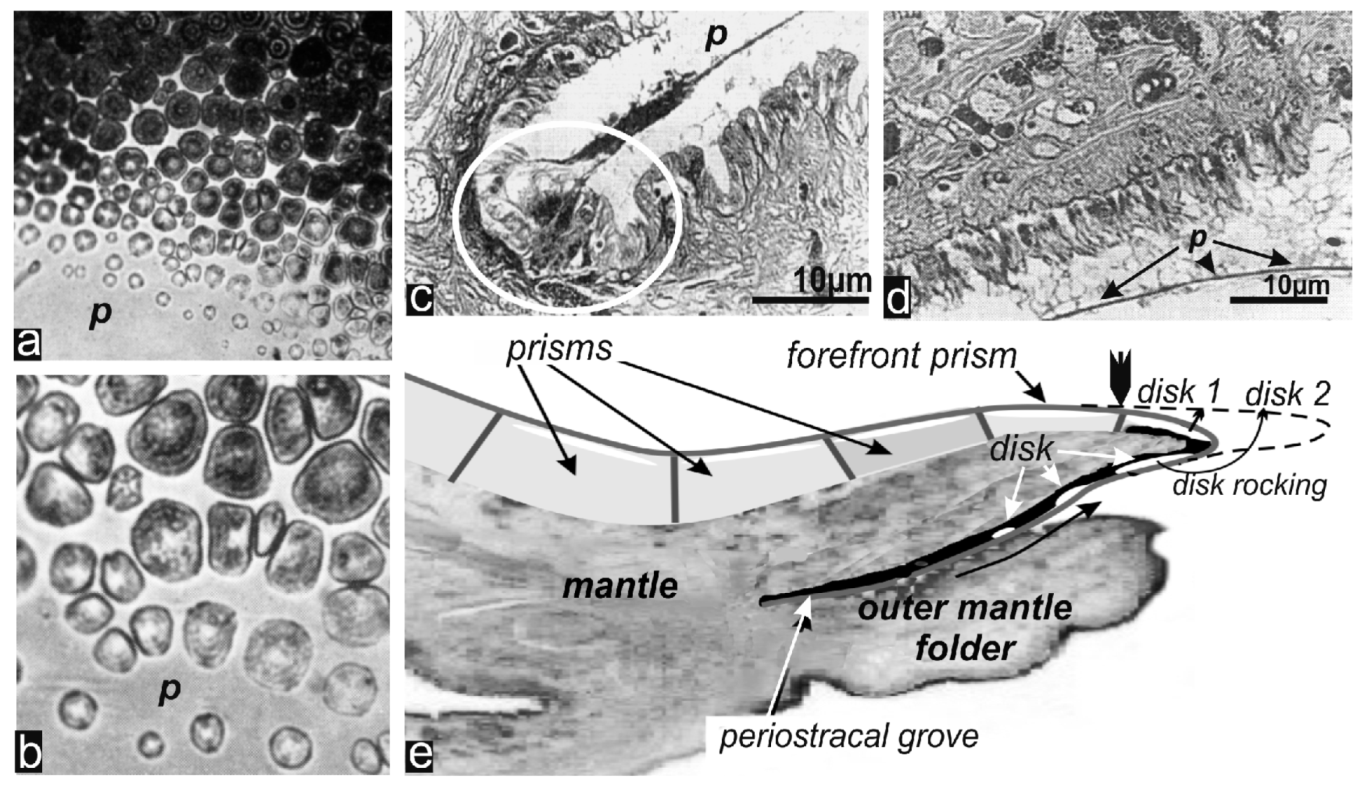

3.2. Insertion of the New Forefront Shell Units into the Common Mineralization Mode of the Mantle Epithelium
3.2.1. Origin of Prism Morphology
3.2.2. Origin of Crystallographic Orientation of the Prisms
3.3. Perturbation of the Mineralizing Mechanism Predating Nacre Deposition at the End of Prism Growth: An Additional Example of Time-Based Metabolic Change in Aging Cells
4 Material and Methods
4.1. Materials
4.2. Methods
5. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Watabe, S.; Maeyama, K.; Nagasawa, H. Recent Advances in Pearl Research—Proceedings of the International Symposium on Pearl Research 2011. TerraPub: Tokyo, Japan, 2013; p. 276. [Google Scholar]
- Schmidt, W.J. Die Bausteine des Tierkörpers in Polarisiertem Lichte; Cohen Verlag: Bonn, Germany, 1924; p. 528. (In German) [Google Scholar]
- Bøggild, O.B. The shell structure of the molluscs. K. Dan. Vidensk. Selsk. Skr. Naturvidensk. Math. Afd. 1930, 9, 231–326. [Google Scholar]
- Taylor, J.D.; Kennedy, W.J.; Hall, A. The Shell Structure and Mineralogy of the Bivalvia: I. Introduction. Nuculacae-Trigonacae. Bull. Br. Mus. Zool. 1969, 3, 1–125. [Google Scholar]
- Taylor, J.D.; Kennedy, W.J.; Hall, A. The Shell Structure and Mineralogy of the Bivalvia: II. Lucinacea-Clavagellacea, Conclusions. Bull. Br. Mus. Zool. 1973, 22, 253–294. [Google Scholar]
- Carter, J.G. Evolutionary significance of shell microstructures in the Palaeotaxodonta, Pteriomorphia and Isofilibranchia (Bivalvia: Mollusca). In Skeletal Biomineralization: Patterns, Processes and Evolutionary Trends; Carter, J.G., Ed.; Van Nostrand Reinhold: New York, NY, USA, 1990; Volume 1, pp. 135–296. [Google Scholar]
- Wilbur, K.M. Shell formation and regeneration. In Physiology of Mollusca; Wilbur, K.M., Owen, G., Eds.; Academic Press: New York, NY, USA, 1964; Volume 1, pp. 243–282. [Google Scholar]
- Wada, K. Crystal growth of molluscan shells. Bull. Natl. Pearl Res. Lab. 1961, 36, 703–828. [Google Scholar]
- Taylor, J.D.; Kennedy, W.J. The influence of the periostracum on the shell structure of bivalve Molluscs. Calcif. Tissue Res. 1969, 3, 274–283. [Google Scholar]
- Mikkelsen, P.M.; Tëmkin, I.; Bieler, R.; Lyons, W.G. Pinctada longisquamosa (Dunker, 1852) (Bivalvia: Pteriidae), an unrecognized pearl oyster in the Western Atlantic. Malacologia 2004, 46, 473–501. [Google Scholar]
- Saleuddin, A.S.M.; Petit, H. The mode of formation and the structure of the periostracum. In The Mollusca; Saleuddin, A.S.M., Wilbur, K.M., Eds.; Academic Press: New York, NY, USA, 1983; Volume 4, pp. 199–234. [Google Scholar]
- Soldati, A.L.; Jacob, D.E.; Wehrmeister, U.; Hofmeister, W. Structural characterization and chemical composition of aragonite and vaterite in freshwater cultured pearls. Mineral. Mag. 2008, 72, 577–590. [Google Scholar] [CrossRef]
- Volkmer, D. Biologically inspired crystallization of calcium carbonate beneath monolayers: A critical overview. In Handbook of Biomineralization: Biomimetic and Bioinspired Chemistry; Behrens, P., Baeuerlein, E., Eds.; Wiley: Hoboken, NJ, USA, 2007; pp. 65–87. [Google Scholar]
- Marsh, M.E.; Sass, R.L. Calcium-binding phosphoprotein particles in the extrapallial fluid and innermost shell lamella of clams. J. Exp. Zool. 1983, 226, 193–203. [Google Scholar] [CrossRef]
- Cuif, J.P.; Dauphin, Y.; Nehrke, G.; Nouet, J.; Perez-Huerta, A. Layered growth and crystallization in calcareous biominerals: Impact of structural and chemical evidence on two major concepts in invertebrate biomineralization studies. Minerals 2012, 2, 11–39. [Google Scholar]
- Dauphin, Y.; Cuif, J.P.; Doucet, J.; Salomé, M.; Susini, J.; Williams, C.T. In situ chemical speciation of sulfur in calcitic biominerals and the simple prism concept. J. Struct. Biol. 2003, 142, 272–280. [Google Scholar] [CrossRef]
- Farre, B.; Brunelle, A.; Laprévotte, O.; Cuif, J.P.; Williams, C.T.; Dauphin, Y. Shell layers of the black-lip pearl oyster Pinctada margaritifera: Matching microstructure and composition. Comp. Biochem. Physiol. 2011, 159, 131–139. [Google Scholar] [CrossRef]
- Carter, J.G.; Aller, R.C. Calcification in the bivalve periostracum. Lethaia 1975, 8, 315–320. [Google Scholar] [CrossRef]
- Carter, J.G. Ecology and Evolution of the Gastrochaenacea (Mollusca, Bivalvia) with Notes on the Evolution of the Endolithic Habitat; Peabody Museum of Natural History: New Haven, CT, USA, 1976; pp. 1–92. [Google Scholar]
- Jabbour-Zahab, J.; Chagot, D.; Blanc, F.; Grizel, H. Mantle histology, histochemistry and ultrastructure of the pearl oyster Pinctada margaritifera (L.). Aquat. Living Res. 1992, 5, 287–298. [Google Scholar]
- Suzuki, M.; Nakayama, S.; Nagasawa, H.; Kogure, T. Initial formation of calcite crystals in the thin prismatic layer with the periostracum of Pinctada fucata. Micron 2013, 45, 136–139. [Google Scholar] [CrossRef]
- Nudelman, F.; Gotliv, B.A.; Addadi, L.; Weiner, S. Mollusk shell formation: mapping the distribution of organic matrix components underlying a single aragonitic tablet in nacre. J. Struct. Biol. 2006, 153, 176–187. [Google Scholar] [CrossRef]
- Dauphin, Y. Nanostructures de la nacre des tests de céphalopodes actuels. Paläontol. Z. 2001, 75, 113–122. (In French) [Google Scholar] [CrossRef]
- Checa, A.G.; Bonarski, J.T.; Willinger, M.G.; Faryna, M.; Berent, K.; Kania, B.; Gonzalez-Segura, A.; Pina, C.M.; Morawiec, J.P.A. Crystallographic orientation inhomogeneity and crystal splitting in biogenic calcite. J. R. Soc. Interface 2013, 10, 20130425. [Google Scholar] [PubMed]
- Cuif, J.P.; Dauphin, Y.; Sorauf, J.E. Biominerals and Fossils through Time; Cambridge University Press: Cambridge, UK, 2011; p. 490. [Google Scholar]
- Griffin, L.E. The Anatomy of the Nautilus Pompilius; Johns Hopkins Press: Baltimore, MD, USA, 1900; pp. 101–197. [Google Scholar]
- Chu, P.K.; Li, L. Characterization of amorphous and nanocrystalline carbon films. Mater. Chem. Phys. 2006, 96, 253–277. [Google Scholar] [CrossRef]
- Yamaguchi, K. Shell structure and behaviour related to cementation in oysters. Mar. Biol. 1994, 118, 89–100. [Google Scholar] [CrossRef]
- Checa, A.G.; Salas, C.; Harper, E.M.; de Dios Bueno-Perez, J. Early stage biomineralization in the periostracum of the “living fossil” bivalve Neotrigonia. PLoS One 2014, 9, e90033. [Google Scholar] [PubMed]
- Dauphin, Y.; Cuif, J.P.; Salomé, M. Structure and composition of the aragonitic shell of a living fossil: Neotrigonia (Mollusca, Bivalvia). Eur. J. Mineral. 2014, 26, 485–494. [Google Scholar]
- Voll, G. New work on petrofabrics. Geol. J. 1961, 2, 503–567. [Google Scholar] [CrossRef]
- Ubukata, T. Architectural constraints on the morphogenesis of prismatic structures in Bivalvia. Palaeontology 1994, 37, 241–261. [Google Scholar]
- Ubukata, T. Nucleation and growth of crystals and formation of cellular pattern of prismatic shell microstructure in Bivalve Molluscs. Forma 2001, 16, 141–154. [Google Scholar]
- Checa, A.G.; Rodríguez-Navarro, A.B.; Esteban-Delgado, F.J. The nature and formation of calcitic columnar prismatic shell layers in pteriomorphian bivalves. Biomaterials 2005, 26, 6404–6414. [Google Scholar] [PubMed]
- Checa, A.G.; Okamoto, T.; Ramírez, J. Organization pattern of nacre in Pteriidae (Bivalvia: Mollusca) explained by crystal competition. Proc. Biol. Sci. 2006, 273, 1329–1337. [Google Scholar] [CrossRef]
- Dauphin, Y.; Kervadec, G. Comparaison des diagenèses subies par les phases minérale et protéique soluble des tests de Mollusques Céphalopodes Coléoides. Palaeontographica 1994, A232, 85–98. (In French) [Google Scholar]
- Mutvei, H. Ultrastructure of the mineral and organic components of molluscan nacreous layers. Biomineralization 1970, 2, 48–72. [Google Scholar]
- Nehrke, G.; Nouet, J. Confocal Raman microscope mapping as a tool to describe different mineral and organic phases at high spatial resolution within marine biogenic carbonates: Case study on Nerita undata (Gastropoda, Neritopsina). Biogeosciences 2011, 8, 3761–3769. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cuif, J.-P.; Burghammer, M.; Chamard, V.; Dauphin, Y.; Godard, P.; Moullac, G.L.; Nehrke, G.; Perez-Huerta, A. Evidence of a Biological Control over Origin, Growth and End of the Calcite Prisms in the Shells of Pinctada margaritifera (Pelecypod, Pterioidea). Minerals 2014, 4, 815-834. https://doi.org/10.3390/min4040815
Cuif J-P, Burghammer M, Chamard V, Dauphin Y, Godard P, Moullac GL, Nehrke G, Perez-Huerta A. Evidence of a Biological Control over Origin, Growth and End of the Calcite Prisms in the Shells of Pinctada margaritifera (Pelecypod, Pterioidea). Minerals. 2014; 4(4):815-834. https://doi.org/10.3390/min4040815
Chicago/Turabian StyleCuif, Jean-Pierre, Manfred Burghammer, Virginie Chamard, Yannicke Dauphin, Pierre Godard, Gilles Le Moullac, Gernot Nehrke, and Alberto Perez-Huerta. 2014. "Evidence of a Biological Control over Origin, Growth and End of the Calcite Prisms in the Shells of Pinctada margaritifera (Pelecypod, Pterioidea)" Minerals 4, no. 4: 815-834. https://doi.org/10.3390/min4040815
APA StyleCuif, J.-P., Burghammer, M., Chamard, V., Dauphin, Y., Godard, P., Moullac, G. L., Nehrke, G., & Perez-Huerta, A. (2014). Evidence of a Biological Control over Origin, Growth and End of the Calcite Prisms in the Shells of Pinctada margaritifera (Pelecypod, Pterioidea). Minerals, 4(4), 815-834. https://doi.org/10.3390/min4040815