Geobiological Cycling of Gold: From Fundamental Process Understanding to Exploration Solutions
Abstract
:1. Introduction
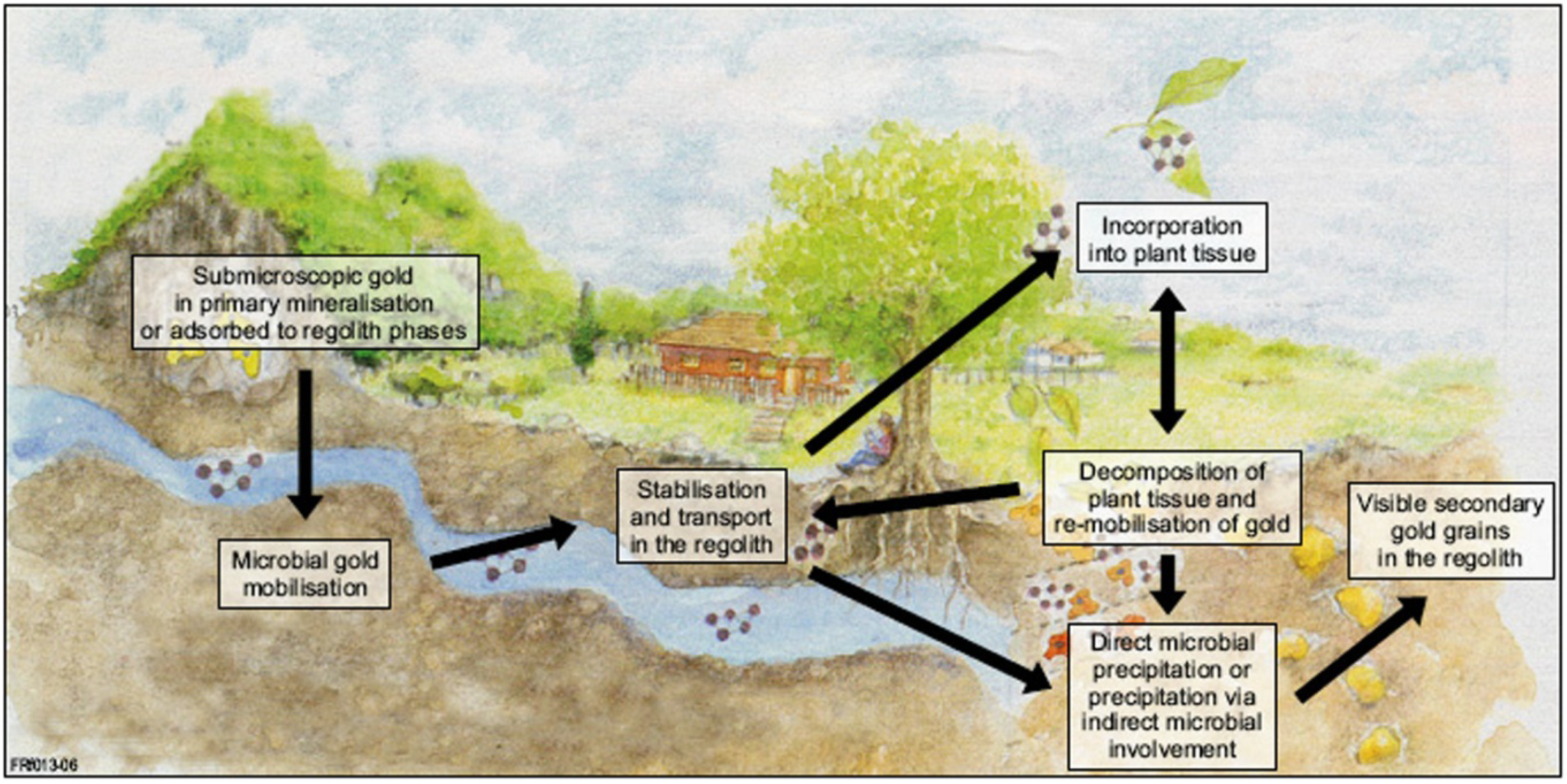
2. Biologically Mediated Transformation of Gold Grains
2.1. Wet Subtropical Environments
2.2. Arid Environments

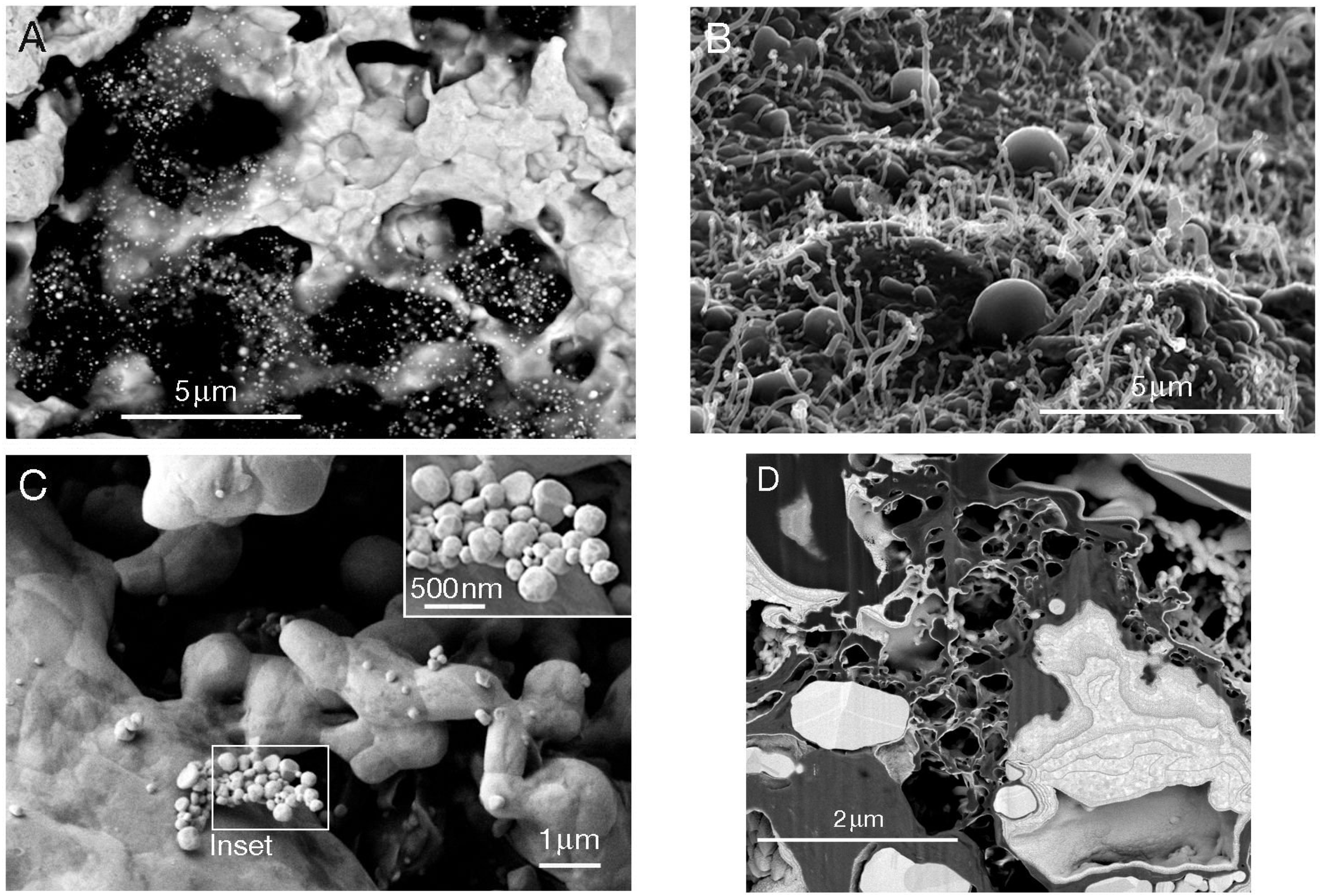
2.3. Temperate and Subarctic Environments
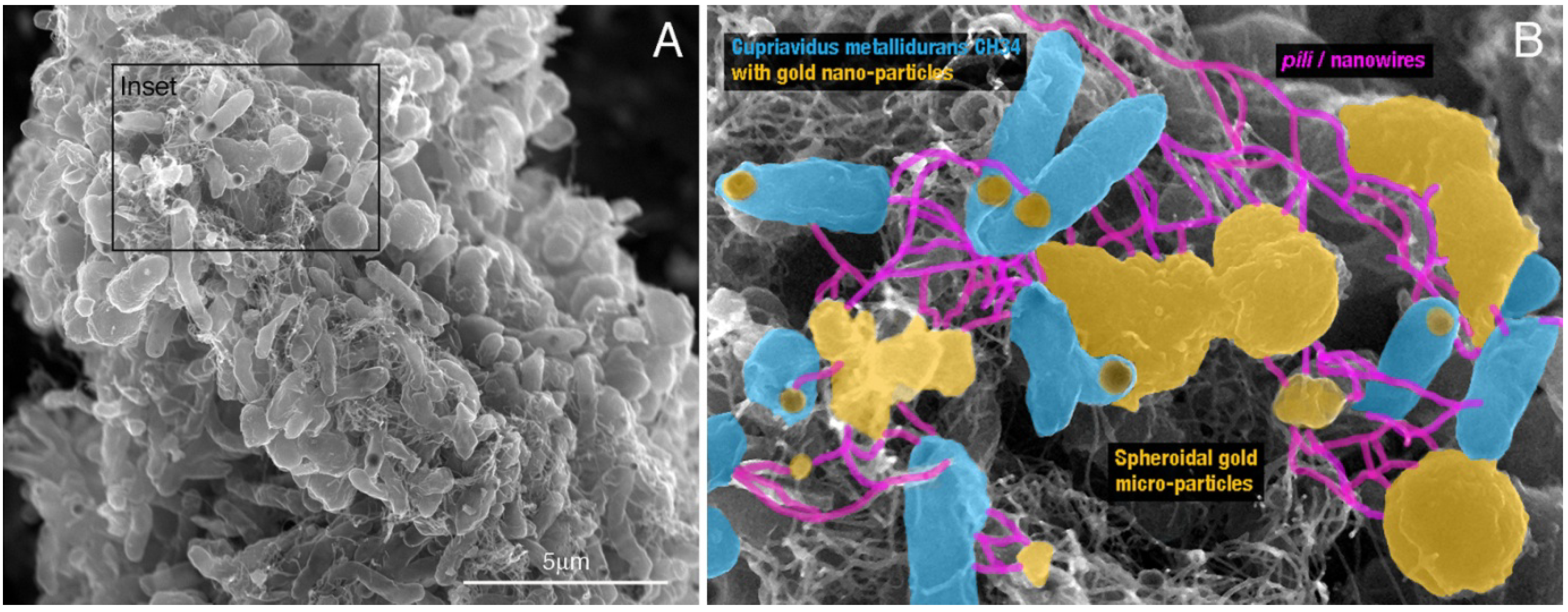
2.4. Laboratory Environments—Formation of Gold Biominerals by C. metallidurans
2.5. A Unified Model for Gold Grain Formation and Implications for Exploration
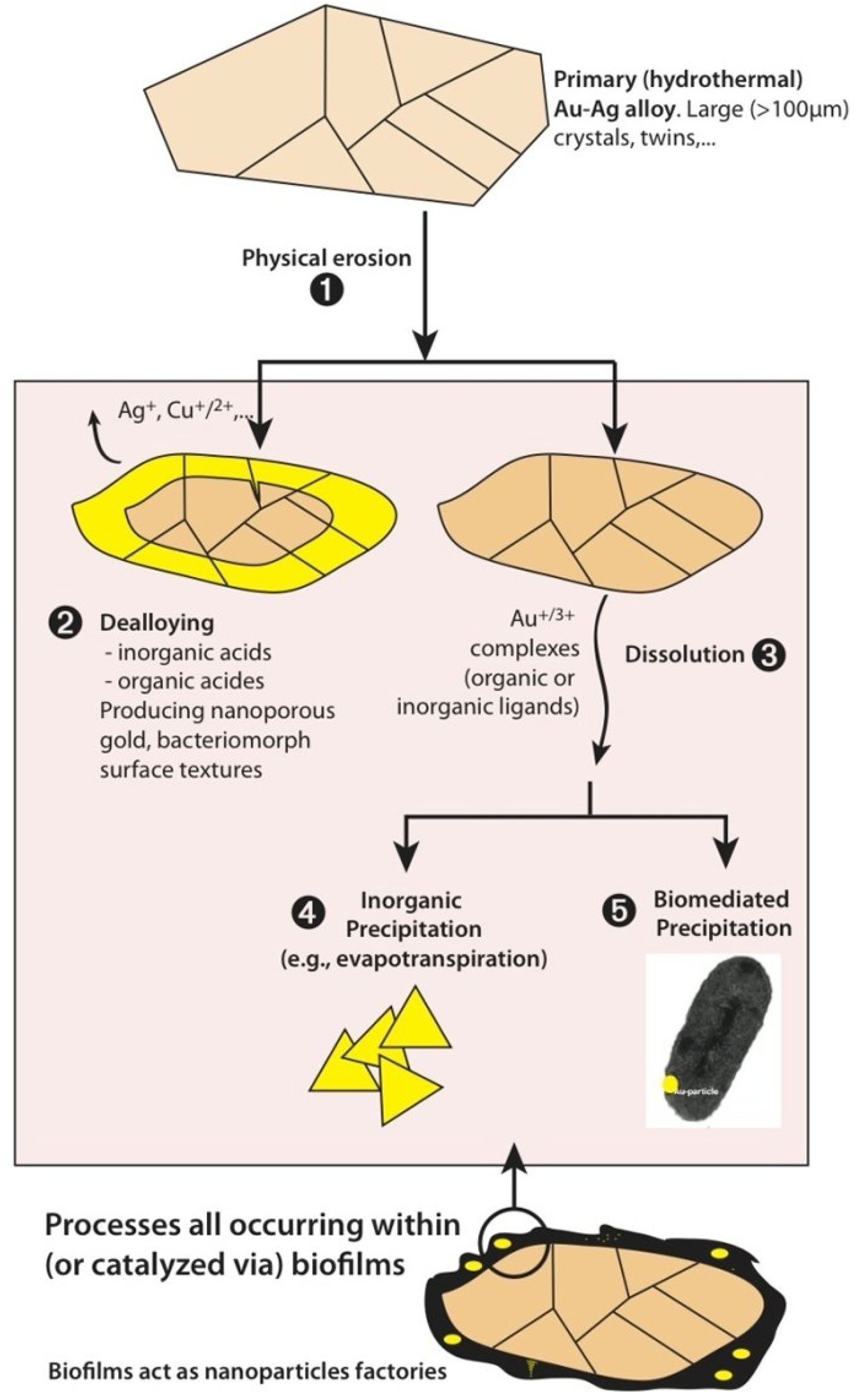
3. Biochemistry of Gold Biomineralization
3.1. Cupriavidus metallidurans CH34—Putative Co-Utilization of Other Metal Resistance Systems for Gold Detoxification
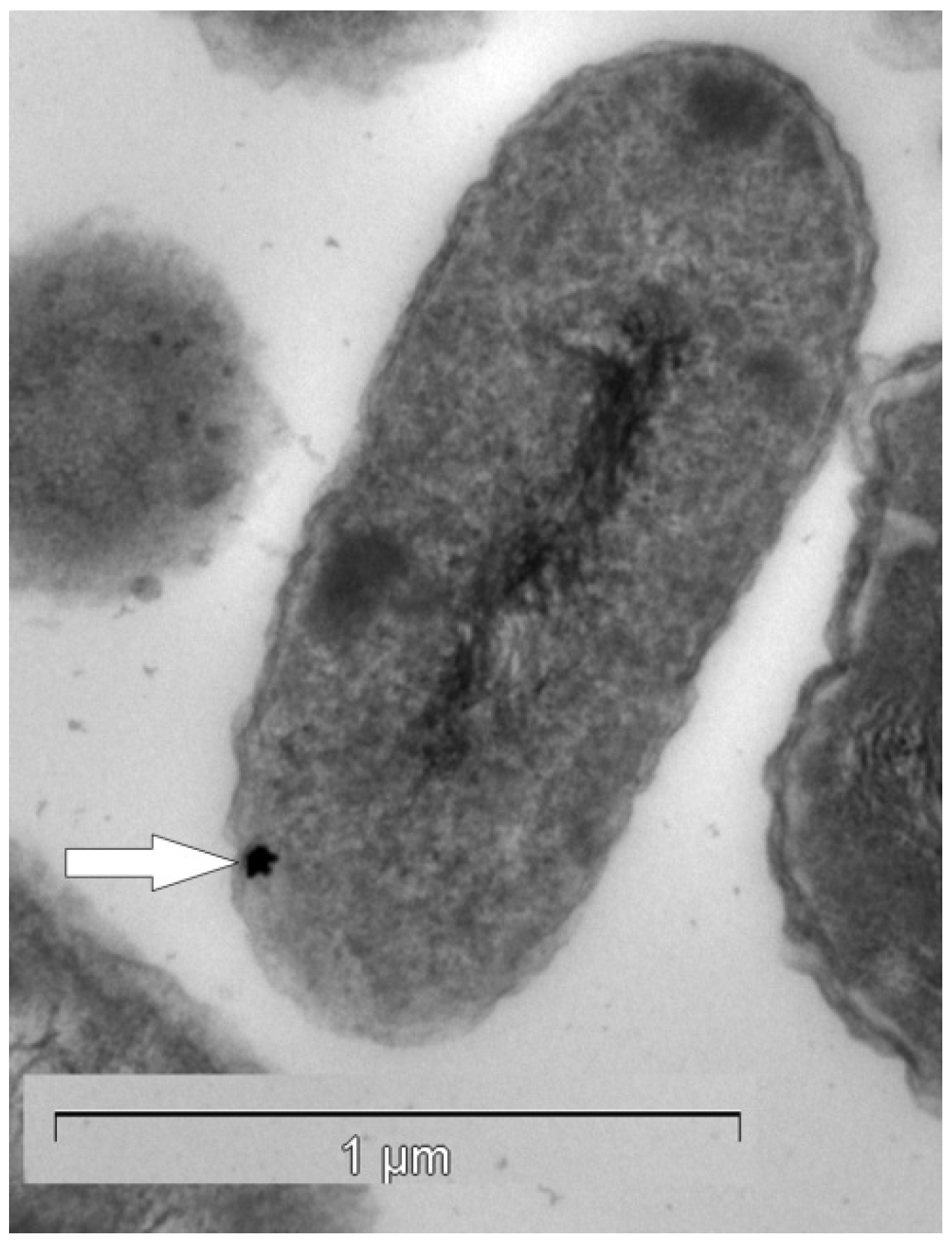
3.2. Salmonella typhimurium Serovar Typhimurium—A Gold Specific Detoxification Pathway
3.3. Extracellular Compounds for Gold Detoxification—Delftia acidovorans and other Organisms
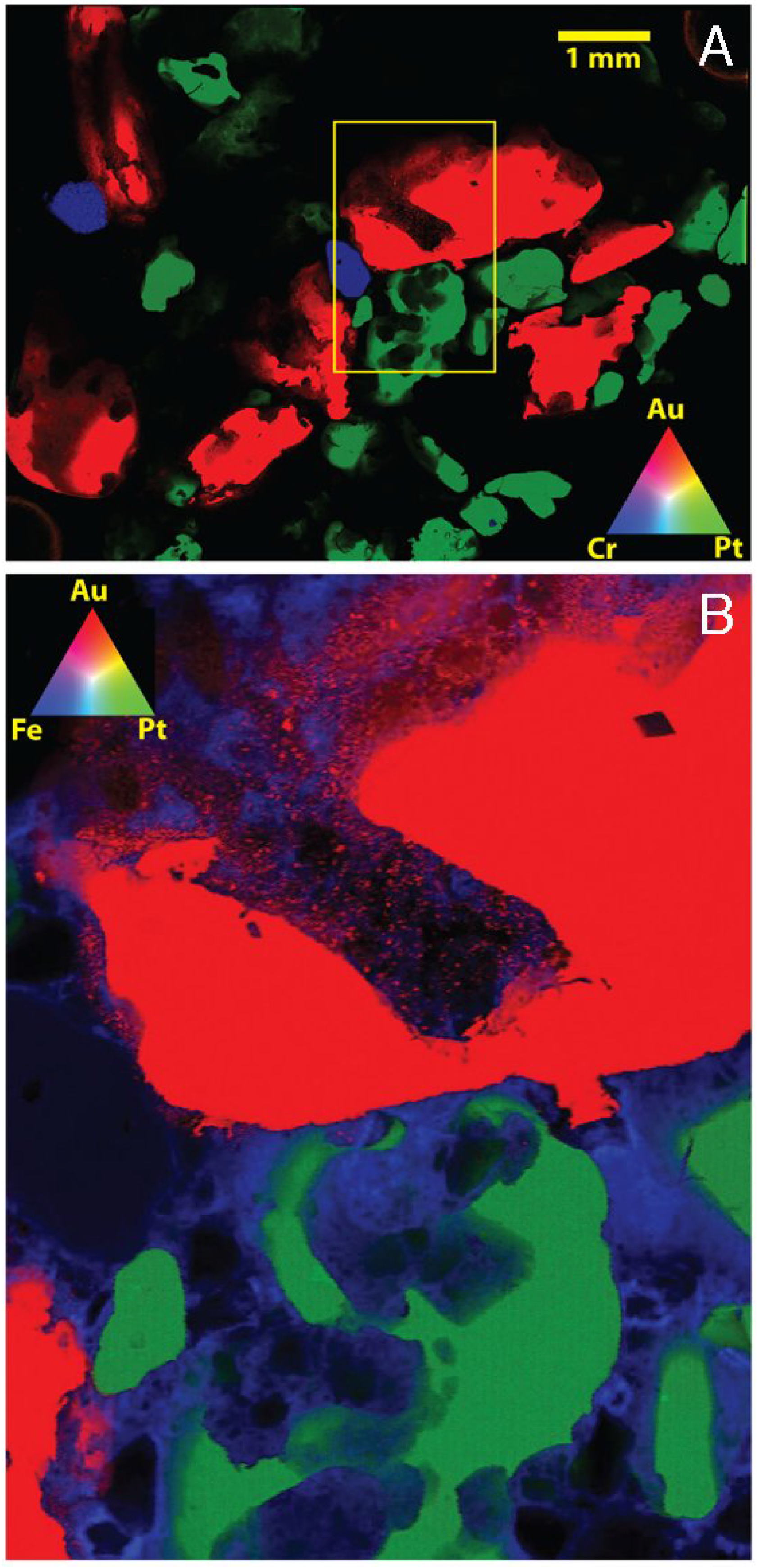
4. Differences in Environmental Mobility of Gold- and Platinum
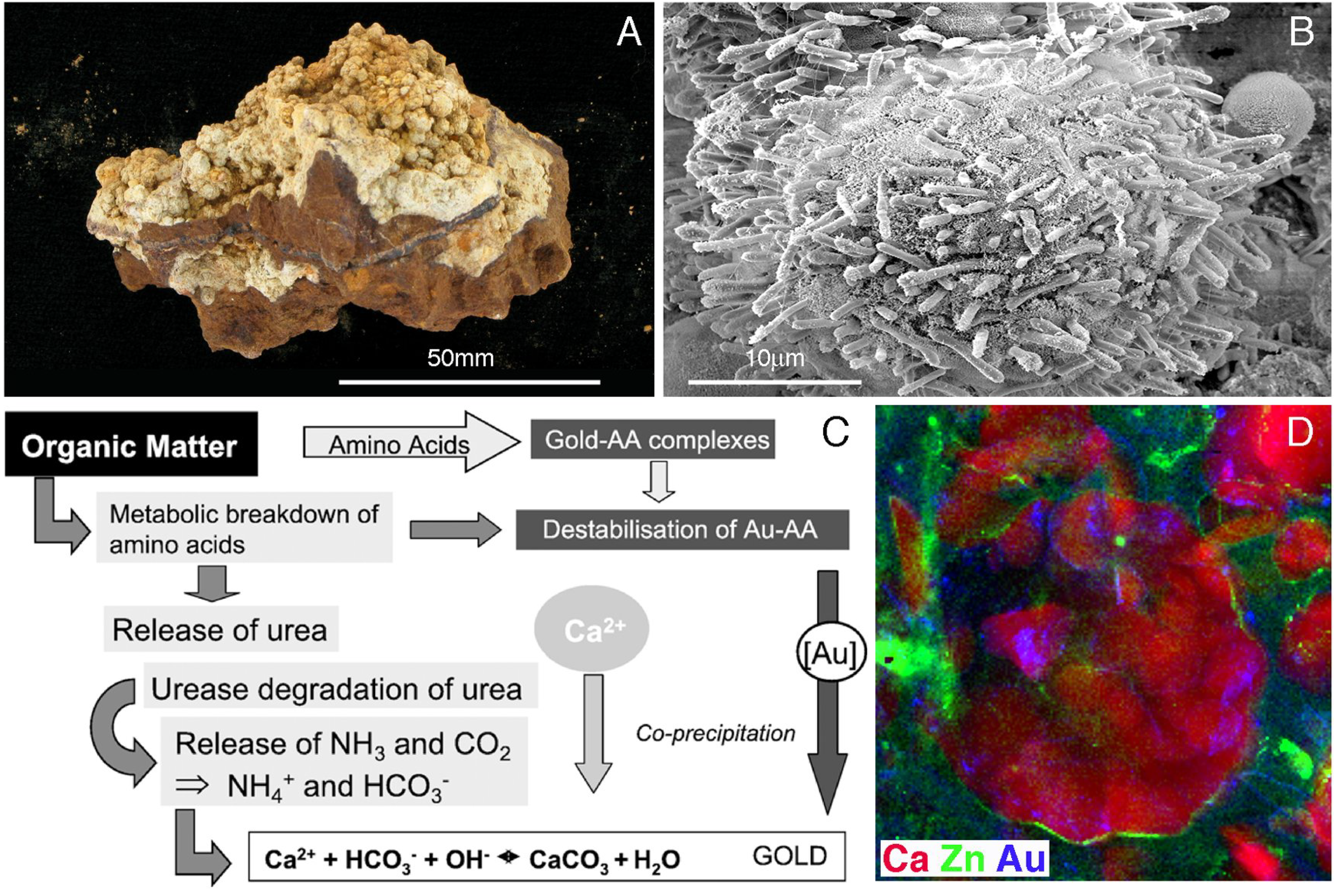
5. Geobiological Formation of Gold Anomalous Calcrete
6. Exploration Geomicrobiology
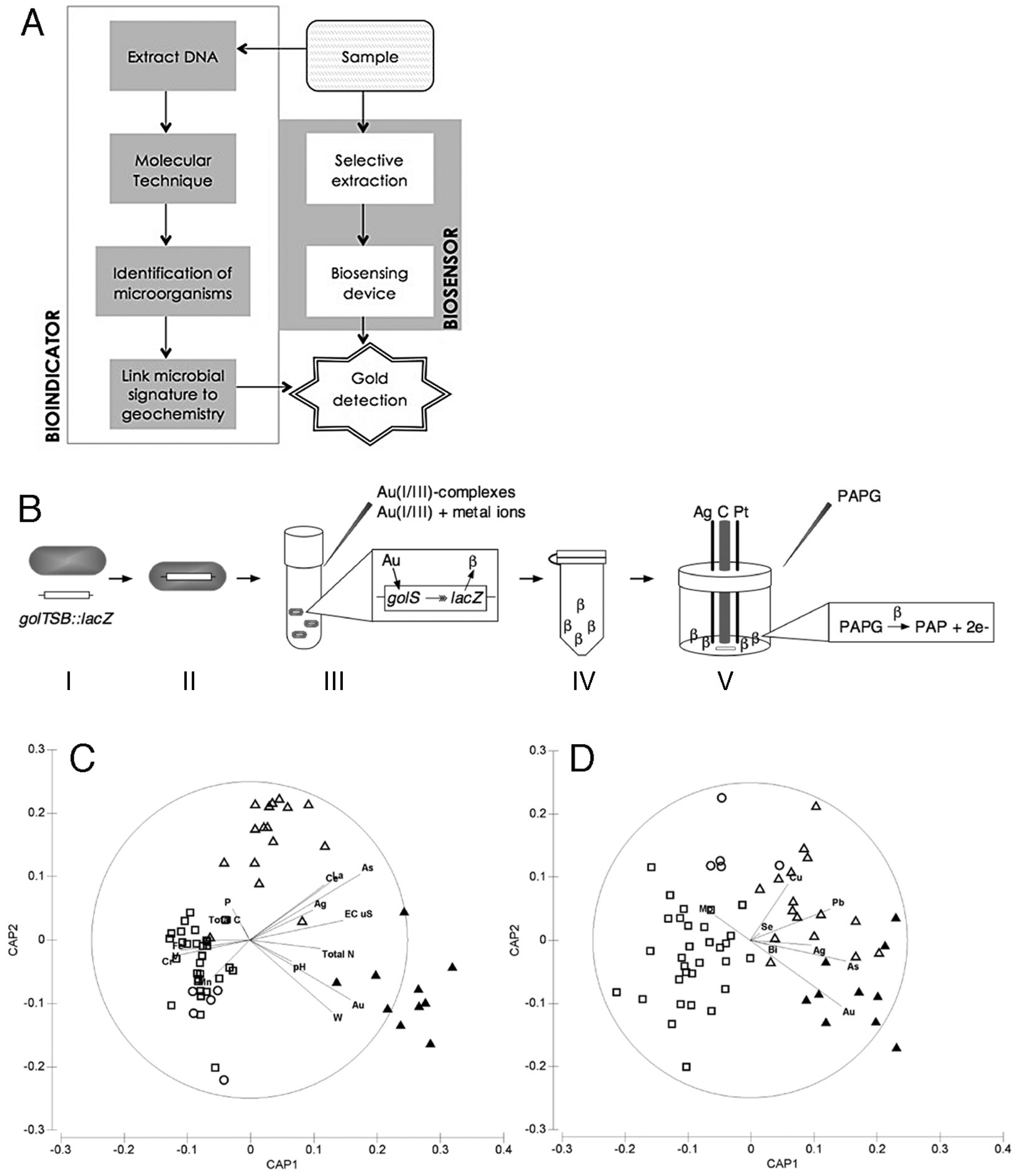
6.1. Bioindicators
6.2. Biosensors
7. Conclusions and Outlook
Acknowledgments
Conflicts of Interest
References
- Ehrlich, H.L. Geomicrobiology: Its significance for geology. Earth Sci. Rev. 1998, 45, 45–60. [Google Scholar] [CrossRef]
- Ehrlich, H.L.; Newman, D.K. Geomicrobiology, 5th ed.; CRC Press: Boca Raton, FL, USA, 2008. [Google Scholar]
- Southam, G.; Saunders, J.A. The geomicrobiology of ore deposits. Econ. Geol. 2005, 100, 1067–1084. [Google Scholar] [CrossRef]
- Reith, F.; Dürr, M.; Welch, C.F.; Rogers, S.L. The Geomicrobiology of the Regolith. In Regolith Science; Scott, K., Pain, C.F., Eds.; CSIRO Press: Melbourne, Australia, 2008; pp. 127–159. [Google Scholar]
- Gadd, G.M. Metals, minerals and microbes: Geomicrobiology and bioremediation. Microbiology 2010, 156, 609–643. [Google Scholar] [CrossRef]
- Gold, T. The deep, hot biosphere. Proc. Natl. Acad. Sci. USA 1992, 89, 6045–6049. [Google Scholar] [CrossRef]
- Fredrickson, J.K.; Balkwill, D.L. Geomicrobial processes and biodiversity in the deep terrestrial subsurface. Geomicrobiol. J. 2006, 23, 345–356. [Google Scholar] [CrossRef]
- Fry, J.C.; Parkes, R.J.; Cragg, B.A.; Weightman, A.J.; Webster, G. Prokaryotic biodiversity and activity in the deep subseafloor biosphere. FEMS Microbiol. Ecol. 2008, 66, 181–196. [Google Scholar] [CrossRef]
- Reith, F. Life in the deep subsurface. Geology 2011, 39, 287–288. [Google Scholar] [CrossRef]
- Lloyd, J.R. Microbial reduction of metals and radionuclides. FEMS Microbiol. Rev. 2003, 27, 411–425. [Google Scholar] [CrossRef]
- Reith, F.; Lengke, M.F.; Falconer, D.; Craw, D.; Southam, G. The geomicrobiology of gold. ISME J. 2007, 1, 567–584. [Google Scholar] [CrossRef]
- Southam, G.; Lengke, M.F.; Fairbrother, L.; Reith, F. The biogeochemistry of gold. Elements 2009, 5, 303–307. [Google Scholar] [CrossRef]
- Hough, R.M.; Butt, C.R.M.; Reddy, S.M.; Verrall, M. Gold nuggets: Supergene or hypogene? Aust. J. Earth Sci. 2007, 54, 959–964. [Google Scholar] [CrossRef]
- Hough, R.; Noble, R.; Hitchen, G.; Hart, G.; Reddy, S.; Saunders, M.; Clode, P.; Vaughan, D.; Lowe, J.; Gray, D.; et al. Naturally occurring gold nanoparticles and nanoplates. Geology 2008, 36, 571–574. [Google Scholar] [CrossRef]
- Hough, R.M.; Noble, R.R.P.; Reich, M. Natural gold nanoparticles. Ore Geol. Rev. 2011, 42, 55–61. [Google Scholar] [CrossRef]
- Tomkins, A.G. A biogeochemical influence on the secular distribution of orogenic gold. Econ. Geol. 2013, 108, 193–197. [Google Scholar] [CrossRef]
- Lengke, M.; Southam, G. Bioaccumulation of gold by sulfate-reducing bacteria cultured in the presence of gold(I)-thiosulfate complex. Geochim. Cosmochim. Acta 2006, 70, 3646–3661. [Google Scholar] [CrossRef]
- Lengke, M.F.; Southam, G. The deposition of elemental gold from gold(I)-thiosulfate complex mediated by sulfate-reducing bacterial conditions. Econ. Geol. 2007, 102, 109–126. [Google Scholar] [CrossRef]
- Jones, B.; Renault, R.W.; Rosen, M.R. Biogenicity of gold- and silver-bearing siliceous sinters forming in hot (75 °C) anaerobic spring-waters of Champagne Pool, Waiotapu, North Island, New Zealand. J. Geol. Soc. 2001, 158, 895–911. [Google Scholar] [CrossRef]
- Zammit, C.M.; Cook, N.; Brugger, J.; Ciobanu, C.L.; Reith, F. The future of biotechnology for gold exploration and processing. Miner. Eng. 2012, 32, 45–53. [Google Scholar] [CrossRef]
- Etschmann, B.E.; Black, J.R.; Grundler, P.V.; Borg, S.; Brewe, D.; McPhail, D.C.; Spiccia, L.; Brugger, J. Copper(I) speciation in mixed thiosulfate-chloride and ammonia-chloride solutions: XAS and UV-Visible spectroscopic studies. RSC Adv. 2011, 1, 1554–1566. [Google Scholar] [CrossRef]
- Vlassopoulos, D.; Wood, S.A.; Mucci, A. Gold speciation in natural waters: II. The importance of organic complexing—Experiments with some simple model ligands. Geochim. Cosmochim. Acta 1990, 54, 1575–1586. [Google Scholar] [CrossRef]
- Gray, D.J. The Aqueous Chemistry of Gold in the Weathering Environment; Cooperative Research Centre for Landscape Evolution and Mineral Exploration: Wembley, Australia, 1998; Open File Report No 38; p. 57. [Google Scholar]
- Fairbrother, L.; Etschmann, B.; Brugger, J.; Shapter, J.; Southam, G.; Reith, F. Biomineralization of gold in biofilms of Cupriavidus metallidurans. Environ. Sci. Technol. 2013, 47, 2628–2635. [Google Scholar] [CrossRef]
- Checa, S.K.; Espariz, M.; Audero, M.E.P.; Botta, P.E.; Spinelli, S.V.; Soncini, F.C. Bacterial sensing of and resistance to gold salts. Mol. Microbiol. 2007, 63, 1307–1318. [Google Scholar] [CrossRef]
- Lengke, M.F.; Fleet, M.E.; Southam, G. Bioaccumulation of gold by filamentous cyanobacteria between 25 and 200 °C. Geomicrobiol. J. 2006, 23, 591–597. [Google Scholar] [CrossRef]
- Reith, F.; Etschmann, B.; Grosse, C.; Moors, H.; Benotmane, M.A.; Monsieurs, P.; Grass, G.; Doonan, C.; Vogt, S.; Lai, B.; et al. Mechanisms of gold biomineralization in the bacterium Cupriavidus metallidurans. Proc. Natl. Acad. Sci. USA 2009, 106, 17757–17762. [Google Scholar] [CrossRef]
- Reith, F.; Rogers, S.L.; McPhail, D.C.; Webb, D. Biomineralization of gold: Biofilms on bacterioform gold. Science 2006, 313, 233–236. [Google Scholar] [CrossRef]
- Lengke, M.F.; Fleet, M.E.; Southam, G. Morphology of gold nanoparticles synthesized by filamentous cyanobacteria from gold(I)-thiosulfate and gold(III)-chloride complexes. Langmuir 2006, 22, 2780–2787. [Google Scholar] [CrossRef]
- Mossman, D.J.; Dyer, B.D. The geochemistry of Witwatersrand-type gold deposits and the possible influence of ancient prokaryotic communities on gold dissolution and precipitation. Precambrian Res. 1985, 30, 303–319. [Google Scholar] [CrossRef]
- Frimmel, H.E.; Le Roex, A.P.; Knight, A.P.; Minter, W.E.L. A case study of the postdepositional alteration of Witatersrand Basal Reef gold placer. Econ. Geol. 1993, 88, 249–265. [Google Scholar] [CrossRef]
- Falconer, D.M.; Craw, D.; Youngson, J.H.; Faure, K. Gold and sulphide minerals in Tertiary quartz pebble conglomerate gold placers, Southland, New Zealand. Ore Geol. Rev. 2006, 28, 525–545. [Google Scholar] [CrossRef]
- Falconer, D.M.; Craw, D. Supergene gold mobility: A textual and geochemical study from gold placers in southern New Zealand. Econ. Heol. Spec. Publ. 2009, 14, 77–93. [Google Scholar]
- Reith, F.; Fairbrother, L.; Nolze, G.; Wilhelmi, O.; Clode, P.L.; Gregg, A.; Parsons, J.E.; Wakelin, S.A.; Pring, A.; Hough, R.; et al. Nanoparticle factories: Biofilms hold the key to gold dispersion and nugget formation. Geology 2010, 38, 843–846. [Google Scholar] [CrossRef]
- Fairbrother, L.; Brugger, J.; Shapter, J.; Laird, J.S.; Southam, G.; Reith, F. Supergene gold transformation: Biogenic secondary and nano-particulate gold from arid Australia. Chem. Geol. 2012, 320, 17–31. [Google Scholar]
- Reith, F.; Stewart, L.; Wakelin, S.A. Supergene gold transformation: Secondary and nano-particulate gold from southern New Zealand. Chem. Geol. 2012, 320, 32–45. [Google Scholar] [CrossRef]
- Mann, A.W. Mobility of gold and silver in lateritic weathering profiles: Some observations from Western Australia. Econ. Geol. 1984, 79, 38–49. [Google Scholar] [CrossRef]
- Wilson, A.F. Origin of quartz-free gold nuggets and supergene gold found in laterites and soils—A review and some new observations. Aust. J. Earth Sci. 1984, 31, 303–316. [Google Scholar]
- Bischoff, G.C.O. The biological origin of bacterioform gold from Australia. Neues Jahrb. Geol. Paläontol. Monatshefte 1997, 6, 329–338. [Google Scholar]
- McCready, A.J.; Parnell, J.; Castro, L. Crystalline placer gold from the Rio Neuquen, Argentina: Implications for the gold budget in placer gold formation. Econ. Geol. 2003, 98, 623–633. [Google Scholar]
- Chapman, R.J.; Mortensen, J.K.; LeBarge, W.P. Styles of lode gold mineralization contributing to the placers of the Indian River and Black Hills Creek, Yukon Territory, Canada as deduced from microchemical characterization of placer gold grains. Miner. Deposita 2011, 46, 881–903. [Google Scholar] [CrossRef]
- Knight, J.B.; Morison, S.R.; Mortensen, J.K. The relationship between placer gold particle shape, rimming, and distance of fluvial transport as exemplified by gold from the Klondike District, Yukon Territory, Canada. Econ. Geol. 1999, 94, 635–648. [Google Scholar] [CrossRef]
- Usher, A.; McPhail, D.C.; Brugger, J. A spectrophotometric study of aqueous Au(III) halide–hydroxide complexes at 25–80 °C. Geochim. Cosmochim. Acta 2009, 73, 3359–3380. [Google Scholar] [CrossRef]
- Nies, D.H. Efflux-mediated heavy metal resistance in prokaryotes. FEMS Microbiol. Rev. 2003, 27, 313–339. [Google Scholar] [CrossRef]
- Nam, S.H.; Lee, W.M.; Shin, Y.J.; Yoon, S.J.; Kim, S.W.; Kwak, J.I.; An, Y.J. Derivation of guideline values for gold(III) ion toxicity limits to protect aquatic ecosystems. Water Res. 2013. [Google Scholar] [CrossRef]
- Wiesemann, N.; Mohr, J.; Grosse, C.; Herzberg, M.; Hause, G.; Reith, F.; Nies, D.H. Influence of copper resistance determinants on gold transformation by Cupriavidus metallidurans strain CH34. J. Bacteriol. 2013, 195, 2298–2308. [Google Scholar] [CrossRef]
- Pontel, L.B.; Audero, M.E.; Espariz, M.; Checa, S.K.; Soncini, F.C. GolS controls the response to gold by the hierarchical induction of Salmonella-specific genes that include a CBA efflux-coding operon. Mol. Mircobiol. 2007, 66, 814–825. [Google Scholar] [CrossRef]
- Von Rozycki, T.; Dietrich, N.H. Cupriavidus metallidurans: Evolution of a metal-resistant bacterium. Antonie Leeuwenhoek 2009, 96, 115–139. [Google Scholar] [CrossRef]
- Janssen, P.J.; van Houdt, R.; Moors, H.; Monsieurs, P.; Morin, N.; Michaux, A.; Benotmane, M.A.; Leys, N.; Vallaeys, T.; Lapidus, A.; et al. The complete genome sequence of Cupriavidus metallidurans strain CH34, a master survivalist in harsh and anthropogenic environments. PLoS One 2010, 5, e10433. [Google Scholar] [CrossRef]
- Mergeay, M.; Nies, D.; Schlegel, H.G.; Gerits, J.; Charles, P.; Van Gijsegem, F. Alcaligenes eutrophus CH34 is a facultative chemolithotroph with plasmid-bound resistance to heavy metals. J. Bacteriol. 1985, 162, 328–334. [Google Scholar]
- Kenney, J.P.L.; Song, Z.; Bunker, B.A.; Fein, J.B. An experimental study of Au removal from solution by non-metabolizing bacterial cells and their exudates. Geochim. Cosmochim. Acta 2012, 87, 51–60. [Google Scholar] [CrossRef]
- Song, Z.; Kenney, J.P.L.; Fein, J.B.; Bunker, B.A. An X-ray absorption fine structure study of Au adsorbed onto the non-metabolizing cells of two soil bacterial species. Geochim. Cosmochim. Acta 2012, 86, 103–117. [Google Scholar] [CrossRef]
- Kabata-Pendias, A.; Pendias, H. Trace Elements in Soils and Plants; CRC Press: Boca Raton, FL, USA, 1984; p. 315. [Google Scholar]
- Reith, F.; Brugger, J.; Zammit, C.M.; Gregg, A.L.; Goldfarb, K.C.; Andersen, G.L.; DeSantis, T.Z.; Piceno, Y.M.; Brodie, E.L.; Lu, Z.; et al. Influence of geogenic factors on microbial communities in metallogenic Australian soils. ISME J. 2012, 6, 2107–2118. [Google Scholar] [CrossRef]
- Reith, F.; McPhail, D.C.; Christy, A.G. Bacillus cereus, gold and associated elements in soil and other regolith samples from Tomakin Park Gold Mine in southeastern New South Wales, Australia. J. Geochem. Explor. 2005, 85, 81–98. [Google Scholar] [CrossRef]
- Magnani, D.; Solioz, M. How Bacteria Handle Copper. In Molecular Microbiology of Heavy Metals; Nies, D., Silver, S., Eds.; Springer: Berlin, Germany, 2007; Volume 6, pp. 259–285. [Google Scholar]
- Todd, E. Epidemiology of foodborne illness: North Amercia. Lancet 1990, 336, 788–790. [Google Scholar] [CrossRef]
- Checa, S.; Soncini, F. Bacterial gold sensing and resistance. BioMetals 2011, 24, 419–427. [Google Scholar] [CrossRef]
- Eswaran, J.; Koronakis, E.; Higgins, M.K.; Hughes, C.; Koronakis, V. Three’s company: Component structures brining a closer view of trpartite drug efflux pumps. Curr. Opin. Struct. Biol. 2004, 14, 741–747. [Google Scholar] [CrossRef]
- Wen, A.; Fegan, M.; Hayward, C.; Chakraborty, S.; Sly, L.I. Phylogenetic relationships among members of the Comamonadaceae, and description of Delftia acidovorans (den Dooren de Jong 1926 and Tamaoka et al. 1987) gen. nov., comb. nov. Int. J. Syst. Bacteriol. 1999, 49, 567–576. [Google Scholar] [CrossRef]
- Hoffmann, D.; Kleinsteuber, S.; Muller, R.H.; Babel, W. A transposon encoding the complete 2,4-dichlorophenoxyacetic acid degradation pathway in the alkalitolerant strain Delftia acidovorans P4a. Microbiology 2003, 149, 2545–2556. [Google Scholar] [CrossRef]
- Johnston, C.W.; Wyatt, M.A.; Li, X.; Ibrahim, A.; Shuster, J.; Southam, G.; Magarvey, N.A. Gold biomineralization by a metallophore from a gold-associated microbe. Nat. Chem. Biol. 2013, 9, 241–243. [Google Scholar] [CrossRef]
- Schalk, I.J.; Hannauer, M.; Braud, A. New roles for bacterial siderophores in metal transport and tolerance. Environ. Microbiol. 2011, 13, 2844–2854. [Google Scholar] [CrossRef]
- Schroefel, A.; Kratosova, G.; Bohunicka, M.; Dobrocka, E.; Vavra, I. Biosynthesis of gold nanoparticles using diatoms–silica-gold and EPS-gold bionanocomposite formation. J. Nanopart. Res. 2011, 13, 3207–3216. [Google Scholar] [CrossRef]
- Colica, G.; Caparrotta, S.; Bertini, G.; de Philippis, R. Gold biosorption by exopolysaccharide producing cyanobacteria and purple nonsulphur bacteria. J. Appl. Microbiol. 2012, 113, 1380–1388. [Google Scholar] [CrossRef]
- Malhotra, A.; Dolma, K.; Kaur, N.; Rathore, Y.S.; Ashish; Mayilraj, S.; Choudhury, A.R. Biosynthesis of gold and silver nanoparticles using a novel marine strain of Stenotrophomonas. Bioresour. Technol. 2013, 142, 727–731. [Google Scholar] [CrossRef]
- Brugger, J.; Etschmann, B.; Grosse, C.; Plumridge, C.; Kaminski, J.; Paterson, D.; Shar, S.S.; Ta, C.; Howard, D.L.; de Jonge, M.D.; et al. Can biological toxicity drive the contrasting behavior of platinum and gold in surface environments? Chem. Geol. 2013, 343, 99–110. [Google Scholar] [CrossRef]
- Schmidt Mumm, A.; Reith, F. Biomediation of calcrete at the gold anomaly of the Barns prospect, Gawler Craton, South Australia. J. Geochem. Explor. 2007, 92, 13–33. [Google Scholar] [CrossRef]
- Reith, F.; Wakelin, S.A.; Gregg, A.L.; Schmidt Mumm, A. A microbial pathway for the formation of gold-anomalous calcrete. Chem. Geol. 2009, 258, 315–326. [Google Scholar] [CrossRef]
- Reith, F.; Etschmann, B.; Dart, R.C.; Brewe, D.L.; Vogt, S.; Schmidt Mumm, A.; Brugger, J. Distribution and speciation of gold in biogenic and abiogenic calcium carbonates—Implications for the formation of gold anomalous calcrete. Geochim. Cosmochim. Acta 2011, 75, 1942–1956. [Google Scholar] [CrossRef]
- Castanier, S.; Le Metayer-Levrel, G.; Perthuisot, J.P. Ca-carbonates precipitation and limestone genesis—The microbiogeologist point of view. Sediment. Geol. 1999, 126, 9–23. [Google Scholar] [CrossRef]
- Vogels, G.D.; van der Drift, C. Degradation of purines and pyrimidines by microorganisms. Bacteriol. Rev. 1976, 40, 403–468. [Google Scholar]
- Cunin, R.; Glansdorff, N.; Pierard, A.; Stalon, V. Biosythesis and metabolism of arginine in bacteria. Microbiol. Rev. 1986, 50, 314–352. [Google Scholar]
- Millo, C.; Dupraz, S.; Ader, M.; Guyot, F.; Thaler, C.; Foy, E.; Ménez, B. Carbon isotope fractionation during calcium carbonate precipitation induced by ureolytic bacteria. Geochim. Cosmochim. Acta 2012, 98, 107–124. [Google Scholar] [CrossRef]
- Enders, M.S.; Knickerbocker, C.; Titley, S.R.; Southam, G. The role of bacteria in the supergene environment of the morenci porphyry copper deposit, Greenlee County, Arizona. Econ. Geol. 2006, 101, 59–70. [Google Scholar] [CrossRef]
- Nordstrom, D.K.; Southam, G. Geomicrobiology of sulfide mineral oxidation. Rev. Mineral. Geochem. 1997, 35, 361–390. [Google Scholar]
- Aylmore, M.G.; Muir, D.M. Thiosulfate leaching of gold—A review. Miner. Eng. 2001, 14, 135–174. [Google Scholar] [CrossRef]
- Zammit, C.M.; Quaranta, D.; Gibson, S.; Zaitouna, A.J.; Ta, C.; Brugger, J.; Lai, R.Y.; Grass, G.; Reith, F. A whole-cell biosensor for the detection of gold. PLoS One 2013, 8, e69292. [Google Scholar] [CrossRef]
- Zammit, C.M.; Cook, N.; Brugger, J.; Ciobanu, C.L.; Reith, F. The Future of Biotechnology for Gold Exploration and Processing. In World Gold; AUSIMM Publishing: Carlton, Australia, 2013; pp. 233–246. [Google Scholar]
- Rastogi, G.; Sani, R. Molecular Techniques to Assess Microbial Community Structure, Function, and Dynamics in the Environment. In Microbes and Microbial Technology; Ahmad, I., Ahmad, F., Pichtel, J., Eds.; Springer: Berlin, Germany, 2011; pp. 29–57. [Google Scholar]
- Hu, N.; Zhao, B. Key genes involved in heavy-metal resistance in Pseudomonas putida CD2. FEMS Microbiol. Lett. 2007, 267, 17–22. [Google Scholar] [CrossRef]
- Mergeay, M.; Monchy, S.; Janssen, P.; Houdt, R.; Leys, N. Megaplasmids in Cupriavidus Genus and Metal Resistance. In Microbial Megaplasmids; Schwartz, E., Ed.; Springer: Berlin, Germany, 2009; Volume 11, pp. 209–238. [Google Scholar]
- Woods, E.J.; Cochrane, C.A.; Percival, S.L. Prevalence of silver resistance genes in bacteria isolated from human and horse wounds. Vet. Microbiol. 2009, 138, 325–329. [Google Scholar] [CrossRef]
- Abou-Shanab, R.A.I.; van Berkum, P.; Angle, J.S. Heavy metal resistance and genotypic analysis of metal resistance genes in gram-positive and gram-negative bacteria present in Ni-rich serpentine soil and in the rhizosphere of Alyssum murale. Chemosphere 2007, 68, 360–367. [Google Scholar] [CrossRef]
- Reith, F.; Rogers, S.L. Assessment of bacterial communities in auriferous and non-auriferous soils using genetic and functional fingerprinting. Geomicrobiol. J. 2008, 25, 203–215. [Google Scholar] [CrossRef]
- Wakelin, S.; Anand, R.R.; Macfarlane, C.; Reith, F.; Noble, R.; Rogers, S. Assessing microbiological surface expression over an overburden-covered VMS deposit. J. Geochem. Explor. 2012, 112, 262–271. [Google Scholar] [CrossRef]
- Wakelin, S.A.; Anand, R.R.; Reith, F.; Gregg, A.L.; Noble, R.R.P.; Goldfarb, K.C.; Andersen, G.L.; DeSantis, T.Z.; Piceno, Y.M.; Brodie, E.L. Bacterial communities associated with a mineral weathering profile at a sulphidic mine tailings dump in arid Western Australia. FEMS Microbiol. Ecol. 2012, 79, 298–311. [Google Scholar] [CrossRef]
- Luong, J.H.T.; Male, K.B.; Glennon, J.D. Biosensor technology: Technology push versus market pull. Biotechnol. Adv. 2008, 26, 492–500. [Google Scholar] [CrossRef]
- Changela, A.; Chen, K.; Xue, Y.; Holschen, J.; Outten, C.E.; O’Halloran, T.V.; Mondragon, A. Molecular basis of metal-ion selectivity and zeptomolar sensitivity by CueR. Science 2003, 301, 1383–1387. [Google Scholar] [CrossRef]
- Cerminati, S.; Soncini, F.C.; Checa, S.K. Selective detection of gold using genetically engineered bacterial reporters. Biotechnol. Bioeng. 2011, 108, 2553–2560. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Reith, F.; Brugger, J.; Zammit, C.M.; Nies, D.H.; Southam, G. Geobiological Cycling of Gold: From Fundamental Process Understanding to Exploration Solutions. Minerals 2013, 3, 367-394. https://doi.org/10.3390/min3040367
Reith F, Brugger J, Zammit CM, Nies DH, Southam G. Geobiological Cycling of Gold: From Fundamental Process Understanding to Exploration Solutions. Minerals. 2013; 3(4):367-394. https://doi.org/10.3390/min3040367
Chicago/Turabian StyleReith, Frank, Joël Brugger, Carla M. Zammit, Dietrich H. Nies, and Gordon Southam. 2013. "Geobiological Cycling of Gold: From Fundamental Process Understanding to Exploration Solutions" Minerals 3, no. 4: 367-394. https://doi.org/10.3390/min3040367
APA StyleReith, F., Brugger, J., Zammit, C. M., Nies, D. H., & Southam, G. (2013). Geobiological Cycling of Gold: From Fundamental Process Understanding to Exploration Solutions. Minerals, 3(4), 367-394. https://doi.org/10.3390/min3040367




