A New Direction for Biomining: Extraction of Metals by Reductive Dissolution of Oxidized Ores
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characteristics of the Limonitic Nickel Laterite Ore
| Mineral | Generalized formula | wt % of ore | Nickel deportment (% of total Ni) |
|---|---|---|---|
| Goethite | FeO(OH) | 10 | 56 |
| Quartz | SiO2 | 76 | 19 |
| (Serpentine) | Mg3Si2O5(OH)4 | 2 | 18 |
| Asbolane | Mn3O3(OH)6 | 0.6 | 7 |
| Chromite | FeCr2O4 | 0.8 | 0 |
2.2. Reductive Dissolution of Goethite in the Laterite Ore and Solubilization of Associated Nickel under Controlled Conditions
 ) and nickel (
) and nickel (  ) in bioreactor cultures of At. ferrooxidans containing Ni laterite ore and elemental sulphur, incubated either under anoxic (solid symbols) or aerobic (open symbols) conditions.
) in bioreactor cultures of At. ferrooxidans containing Ni laterite ore and elemental sulphur, incubated either under anoxic (solid symbols) or aerobic (open symbols) conditions.
 ) and nickel (
) and nickel (  ) in bioreactor cultures of At. ferrooxidans containing Ni laterite ore and elemental sulphur, incubated either under anoxic (solid symbols) or aerobic (open symbols) conditions.
) in bioreactor cultures of At. ferrooxidans containing Ni laterite ore and elemental sulphur, incubated either under anoxic (solid symbols) or aerobic (open symbols) conditions.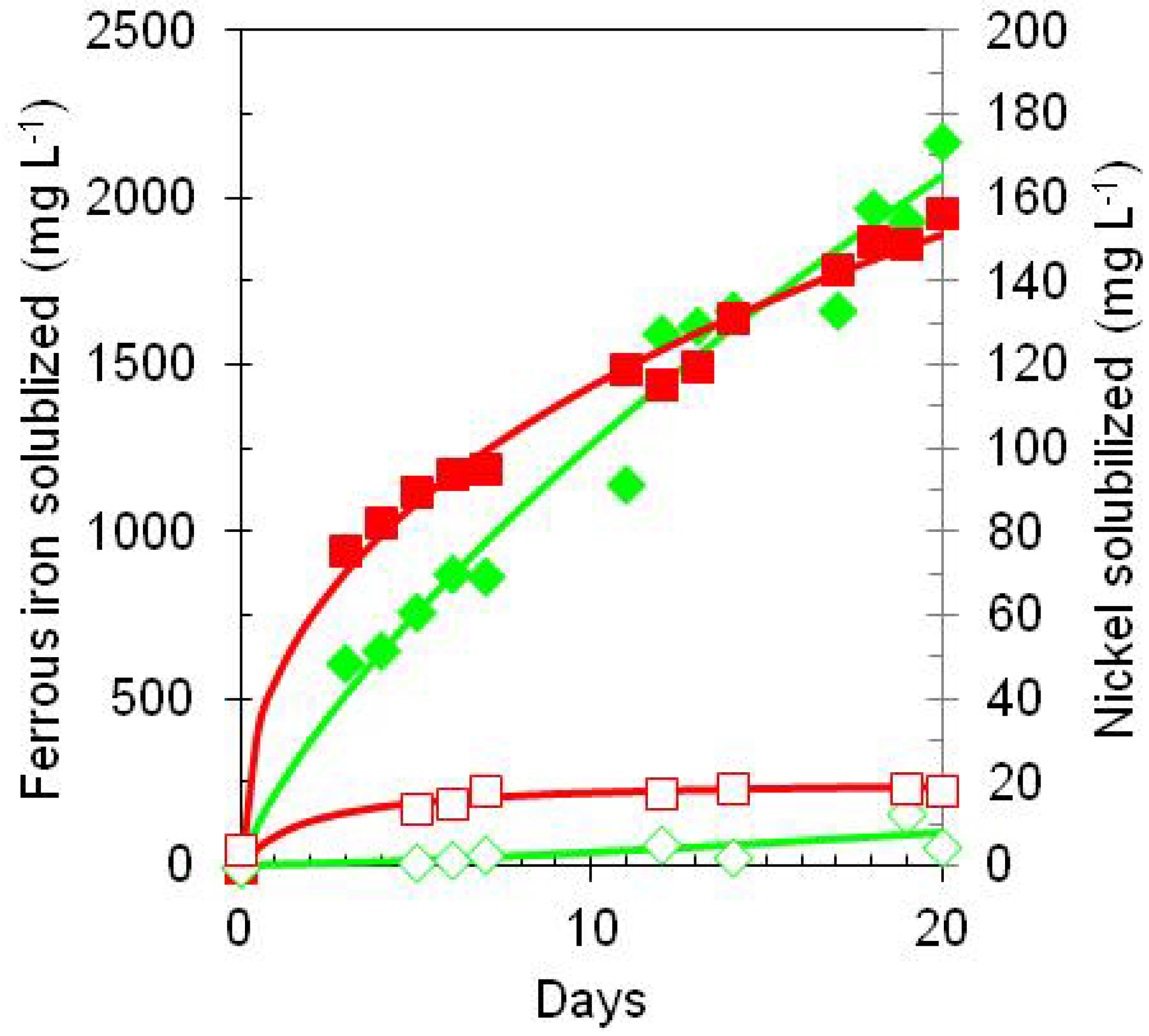
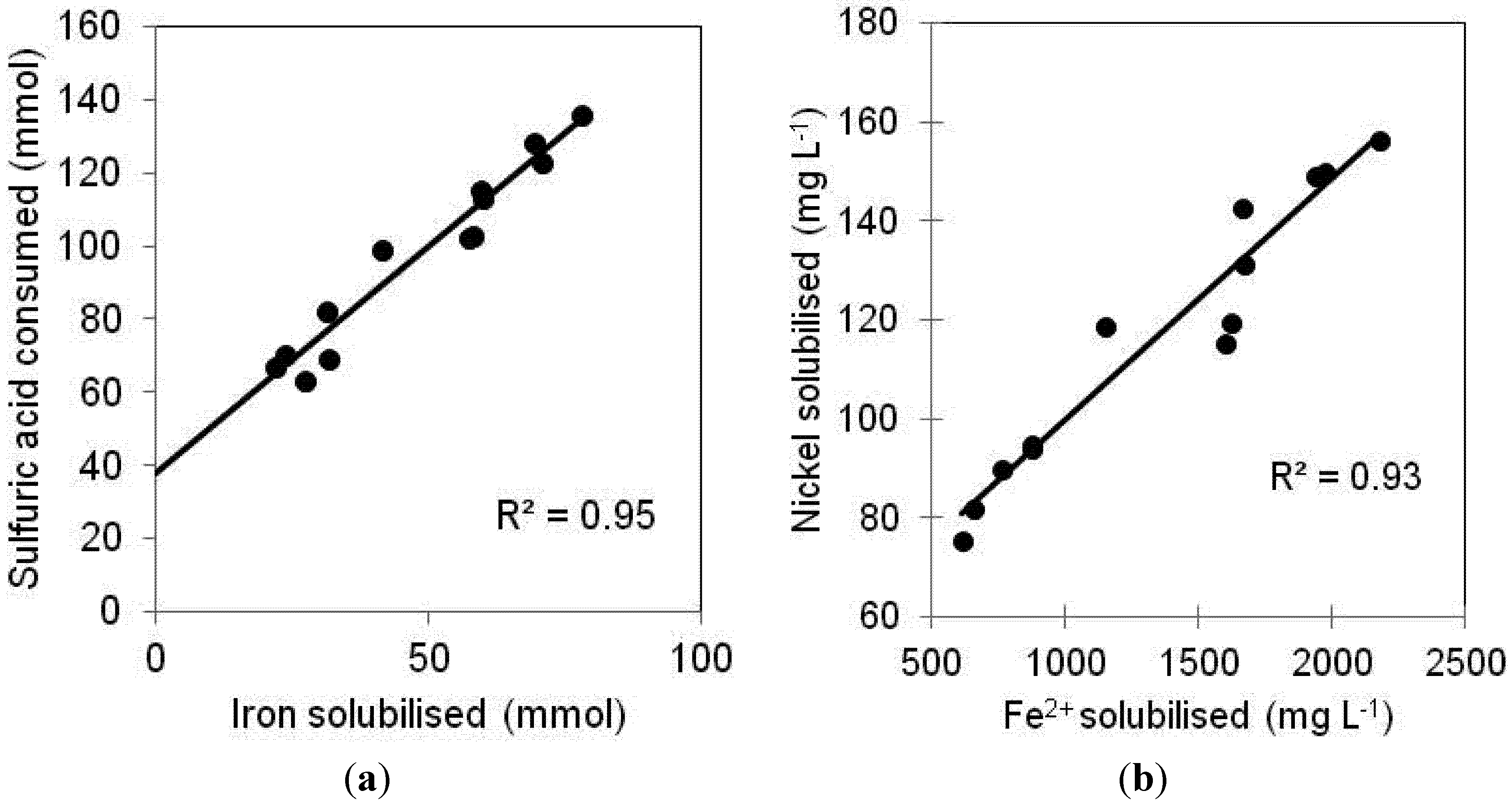
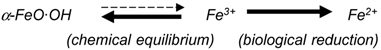
2.3. Solubilization of Other Transition Metals Present in the Laterite Ore
 ) and cobalt (
) and cobalt (  ) in bioreactor cultures of At. ferrooxidans containing Ni laterite ore and elemental sulphur, incubated either under anoxic (solid symbols) or aerobic (open symbols) conditions); (b) correlation between the amounts of manganese and cobalt solubilised under anoxic conditions.
) in bioreactor cultures of At. ferrooxidans containing Ni laterite ore and elemental sulphur, incubated either under anoxic (solid symbols) or aerobic (open symbols) conditions); (b) correlation between the amounts of manganese and cobalt solubilised under anoxic conditions.
 ) and cobalt (
) and cobalt (  ) in bioreactor cultures of At. ferrooxidans containing Ni laterite ore and elemental sulphur, incubated either under anoxic (solid symbols) or aerobic (open symbols) conditions); (b) correlation between the amounts of manganese and cobalt solubilised under anoxic conditions.
) in bioreactor cultures of At. ferrooxidans containing Ni laterite ore and elemental sulphur, incubated either under anoxic (solid symbols) or aerobic (open symbols) conditions); (b) correlation between the amounts of manganese and cobalt solubilised under anoxic conditions.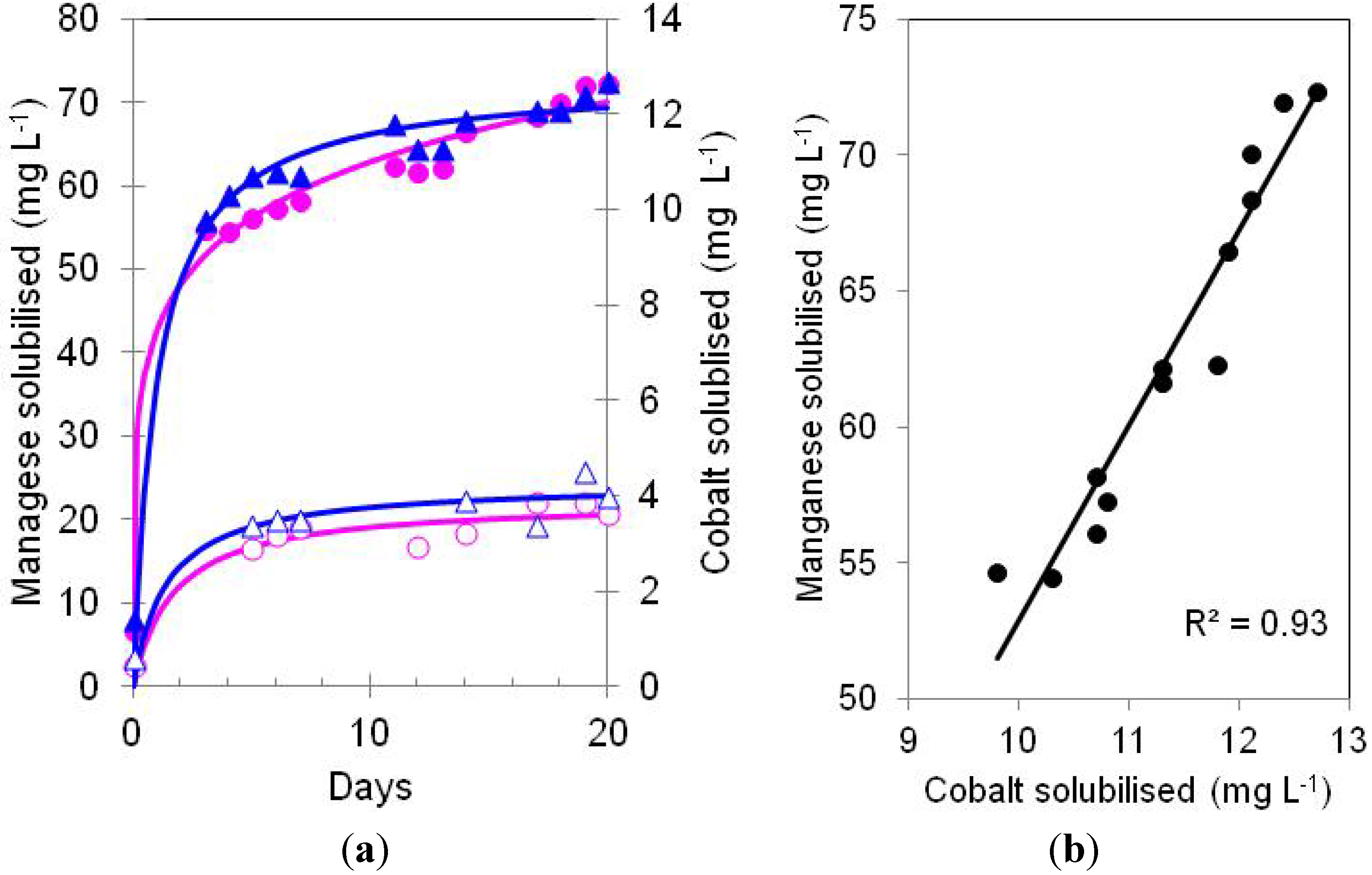
2.4. Mass Balance of Metals in Untreated Ore and Bioleach Residues
| Mass balance based on: | Metal recovery (%) | ||
|---|---|---|---|
| Ni | Co | Mn | |
| XRF analysis | 46 | nd | 75 |
| aqua regia dissolution | 62 | 90 | 94 |
| leachate metal concentrations | 82 | 64 | 116 |
2.5. Implications of Bioreduction of Minerals by Acidophilic Microorganisms
3. Experimental Section
3.1. Ore Material
3.2. Bacteria and Cultivation Conditions
3.3. Reduction of Nickel Laterite ore in Bioreactors
3.4. Abiotic Solubilization of Manganese
3.5. Analytical Techniques
4. Conclusions
Acknowledgments
References
- Rawlings, D.E.; Johnson, D.B. Biomining; Springer-Verlag: Heidelberg, Germany, 2007. [Google Scholar]
- Brierley, C.L. How will biomining be applied in future? Trans. Nonferrous Met. Soc. China 2008, 18, 1302–1310. [Google Scholar] [CrossRef]
- Rawlings, D.E.; Johnson, D.B. The microbiology of biomining: Development and optimization of mineral-oxidizing microbial consortia. Microbiology 2007, 153, 315–324. [Google Scholar] [CrossRef]
- Dalvi, A.D.; Bacon, W.G.; Osborne, R.C. The past and the future of nickel laterites. In Proceedings of Prospectors & Developers Association of Canada (PDAC) 2004 International Convention, Trade Show & Investors Exchange, Toronto, Ontario, Canada, 7-10 March 2004.
- McDonald, R.G.; Whittington, B.I. Atmospheric acid leaching of nickel laterites review. Part I. Sulphuric acid technologies. Hydrometallurgy 2008, 91, 35–55. [Google Scholar] [CrossRef]
- McDonald, R.G.; Whittington, B.I. Atmospheric acid leaching of nickel laterites review. Part II. Chloride and bio-technologies. Hydrometallurgy 2008, 91, 56–69. [Google Scholar]
- Johnson, D.B.; Hallberg, K.B. Carbon, iron and sulphur metabolism in acidophilic micro-organisms. Adv. Microb. Physiol. 2008, 54, 201–255. [Google Scholar] [CrossRef]
- Johnson, D.B.; Kanao, T.; Hedrich, S. Redox transformations of iron at extremely low pH: Fundamental and applied aspects. Front. Microbiol. 2012, 3. [Google Scholar] [CrossRef]
- Bridge, T.A.M.; Johnson, D.B. Reduction of soluble iron and reductive dissolution of ferric iron-containing minerals by moderately thermophilic iron-oxidizing bacteria. Appl. Environ. Microbiol. 1998, 64, 2181–2186. [Google Scholar]
- Lloyd, J.R.; Lovley, D.R.; Macaskie, L.E. Biotechnological applications of metal-reducing microorganisms. Adv. Appl. Microbiol. 2003, 53, 85–128. [Google Scholar] [CrossRef]
- Brandl, H.; Faramarzi, M.A. Microbe-metal-interactions for the biotechnological treatment of metal-containing solid waste. China Particuolog. 2006, 4, 93–97. [Google Scholar] [CrossRef]
- Gadd, G.M. Metals, minerals and microbes: Geomicrobiology and bioremediation. Microbiology 2010, 156, 609–643. [Google Scholar] [CrossRef]
- Wakeman, K.; Auvinen, H.; Johnson, D.B. Microbiological and geochemical dynamics in simulated-heap leaching of a polymetallic sulphide ore. Biotech. Bioeng. 2008, 101, 739–750. [Google Scholar]
- Ñancucheo, I.; Johnson, D.B. Production of glycolic acid by chemolithotrophic iron- and sulphur-oxidizing bacteria and its role in delineating and sustaining acidophilic sulphide mineral-oxidizing consortia. Appl. Environ. Microbiol. 2010, 76, 461–467. [Google Scholar] [CrossRef]
- Lovley, D.R.; Phillips, E.J.P. Rapid assay for microbially reducible ferric iron in aquatic sediments. Appl. Environ. Microbiol. 1987, 53, 1536–1540. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Johnson, D.B.; Grail, B.M.; Hallberg, K.B. A New Direction for Biomining: Extraction of Metals by Reductive Dissolution of Oxidized Ores. Minerals 2013, 3, 49-58. https://doi.org/10.3390/min3010049
Johnson DB, Grail BM, Hallberg KB. A New Direction for Biomining: Extraction of Metals by Reductive Dissolution of Oxidized Ores. Minerals. 2013; 3(1):49-58. https://doi.org/10.3390/min3010049
Chicago/Turabian StyleJohnson, D. Barrie, Barry M. Grail, and Kevin B. Hallberg. 2013. "A New Direction for Biomining: Extraction of Metals by Reductive Dissolution of Oxidized Ores" Minerals 3, no. 1: 49-58. https://doi.org/10.3390/min3010049
APA StyleJohnson, D. B., Grail, B. M., & Hallberg, K. B. (2013). A New Direction for Biomining: Extraction of Metals by Reductive Dissolution of Oxidized Ores. Minerals, 3(1), 49-58. https://doi.org/10.3390/min3010049




