Indium Recovery from Jarosite Pb–Ag Tailings Waste (Part 1)
Abstract
1. Introduction
2. Materials and Methods
Analytical Determinations
3. Results and Discussion
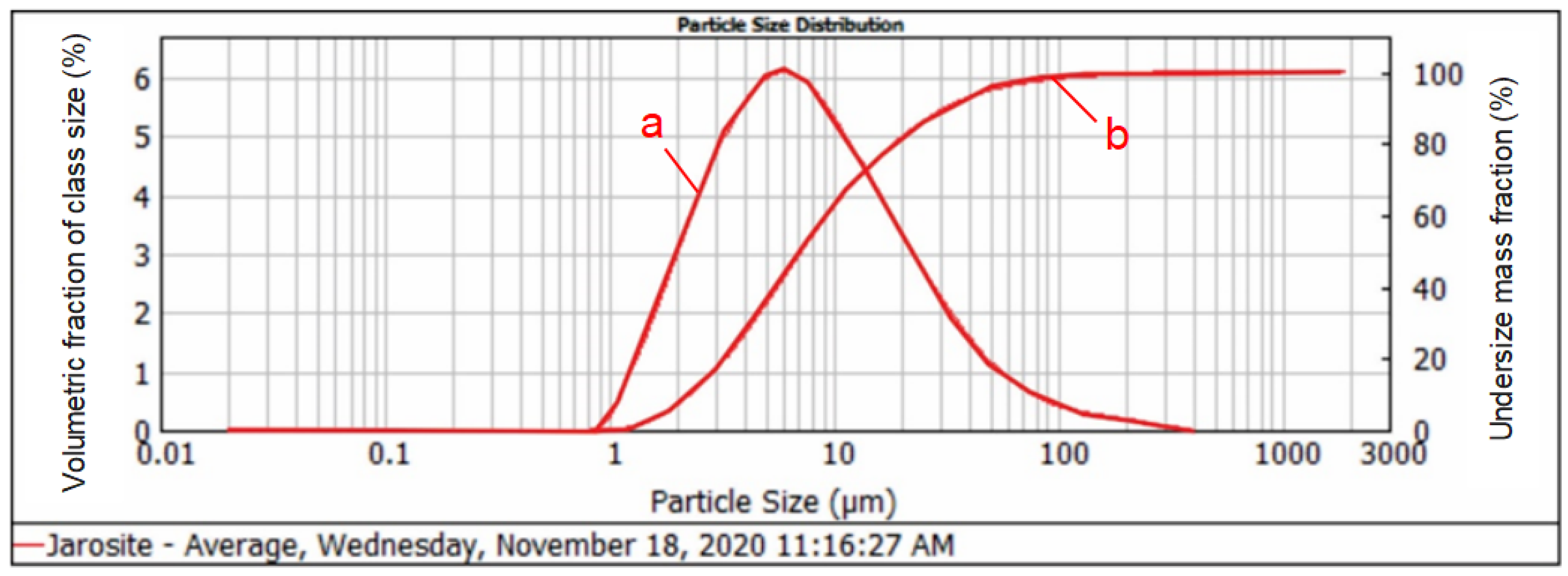
3.1. Grain Size Distribution of the Jarosite Sample
3.2. Mineralogical Composition of the Jarosite Sample
3.3. SEM-EDS Analysis
3.4. Chemical Analyses
3.5. Jarosite Roasting
3.5.1. Roasting Pb–Ag Jarosite Sludge
3.5.2. TGA Analyses of Jarosite Pb–Ag Sludge
- Up to 120 °C (loss of absorbed water)
- 2.
- At 120–260 °C (dihydroxylation, loss of OH−)(M) present in jarosite can be NH4, K, Na, Pb.
- 3.
- At 260–389 °C (loss of ammonia and water)
- 4.
- At 465–610 °C (loss SO2)
3.6. Leaching of the Roasted Jarosite Sample
3.7. Treatment of the Sulfate Solution
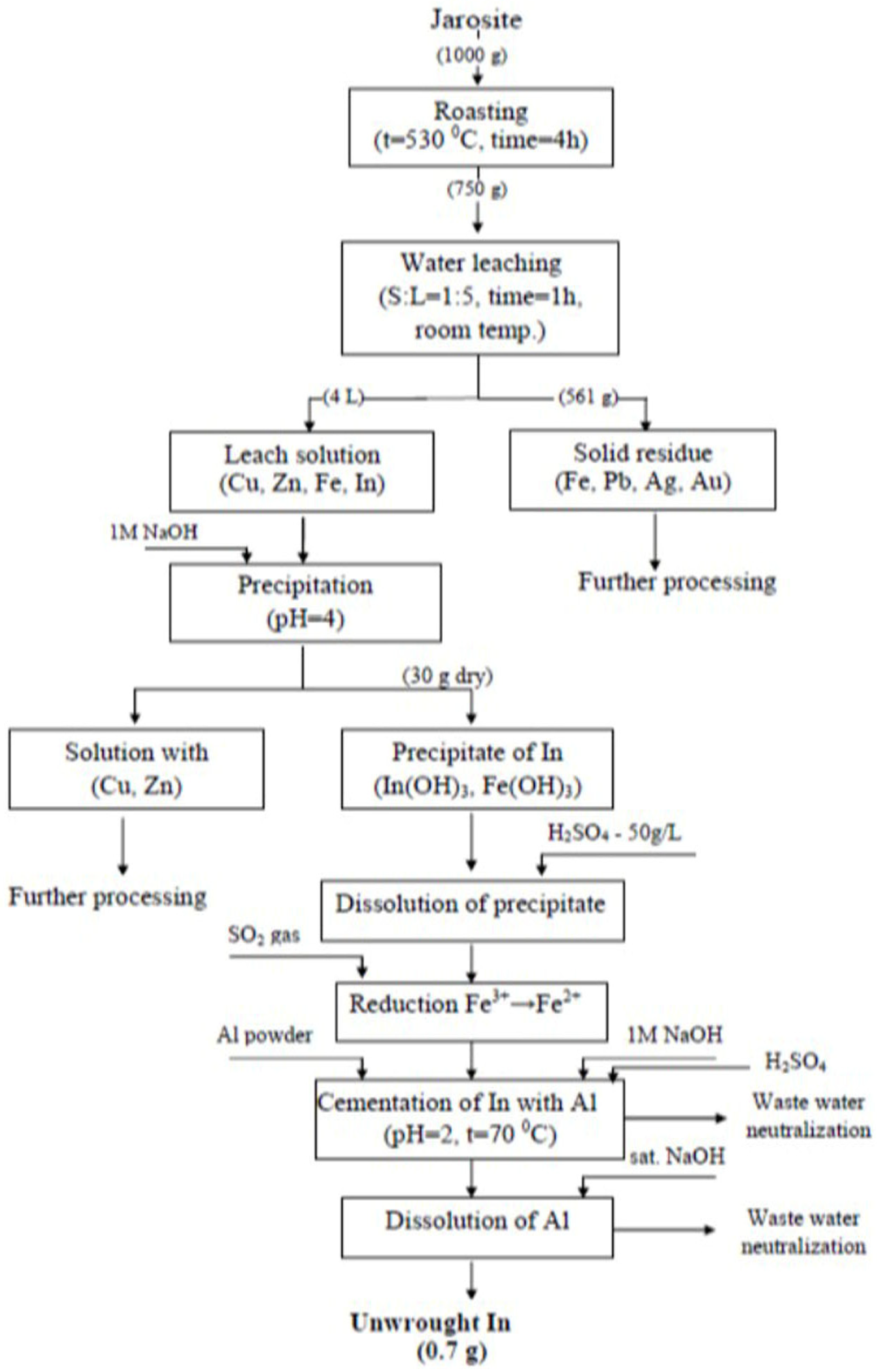
3.8. Indium Recovery Procedure
4. Conclusions
- Jarosite roasting to convert iron sulfate into insoluble hematite;
- Leaching in water in order to leach In;
- The precipitation of In from the leaching solution, 1 M NaOH;
- The dissolution of precipitate with dilute sulfuric acid and the cementation of In from solution with Al;
- The dissolution of Al with saturated NaOH, in which In was isolated as unwrought In.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Reyes, A.I.; Patiño, F.; Flores, U.M.; Pandiyan, T.; Cruz, R.; Gutiérrez, J.E.; Reyes, M.; Flores, H.V. Dissolution rates of jarosite-type compounds in H2SO4 medium: A kinetic analysis and its importance on the recovery of metal values from hydrometallurgical wastes. Hydrometallurgy 2017, 167, 16–29. [Google Scholar] [CrossRef]
- Conić, V.; Rajčić Vujasinović, M.; Trujić, V.; Cvetkovski, V. Copper, Zinc, and Iron Bioleaching from a Polymetallic Sulphide Concentrate. Trans. Nonferrous Met. Soc. China 2014, 24, 3688–3695. [Google Scholar] [CrossRef]
- Zheng, Y.-X.; Lv, J.F.; Liu, W.; Qin, W.-Q.; Wen, S.-M. An innovative technology for recovery of zinc, lead and silver from zinc leaching residue. Physicochem. Probl. Miner. Process. 2016, 52, 943–954. [Google Scholar]
- Wang, Y.; Yang, H.; Zhang, W.; Song, R.; Jiang, B. Study on recovery of lead, zinc, iron from jarosite residues and simultaneous sulfur fixation by direct reduction. Physicochem. Probl. Miner. Process. 2018, 54, 517–526. [Google Scholar]
- Rashchi, F.; Dashti, A.; Arabpour-Yazdi, M.; Abdizadeh, H. Anglesite flotation: A study for lead recovery from zinc leach residue. Miner. Eng. 2005, 18, 205–212. [Google Scholar] [CrossRef]
- Lutandula, M.S.; Maloba, B. Recovery of cobalt and copper through reprocessing of tailings from flotation of oxidized ores. J. Environ. Chem. Eng. 2013, 1, 1085–1090. [Google Scholar] [CrossRef]
- Stanojević, D.; Filipović-Petrović, L. Doprinos integrisanoj valorizaciji metala u hidrometalurgiji cinka. Zaštita Mater. 2014, 55, 11–25. [Google Scholar]
- Sinadinović, D.; Kamberović, Ž.; Šutić, A. Leaching Kinetics of lead from Lead(II) Sulphate in Aqueous Calcium and Magnesium Chloride Solution. Hydrometallurgy 1997, 47, 137–147. [Google Scholar] [CrossRef]
- Kerolli-Mustafa, M.; Ćurković, L.; Fajković, H.; Rončević, S. Ecological Risk Assessment of Jarosite Waste Disposal. Croat. Chem. Acta 2015, 88, 189–196. [Google Scholar] [CrossRef]
- Creedy, S.; Glinin, A.; Matusewics, R.; Hughes, S.; Reuter, M. Outotec Ausmelt Technology for Treating Zinc Residues. World Metall.-Erzmetall 2013, 66, 230–235. [Google Scholar]
- Hughes, S.; Reuter, M.A.; Baxter, R.; Kaye, A.; Hughes, S.; Reuter, M.A.; Baxter, R.; Kaye, A. Ausmelt Technology for Lead and Zinc Processing. Lead Zinc 2008, 2008, 147–162. [Google Scholar]
- Kamberović, Ž.; Gajić, N.; Korać, M.; Jevtić, S.; Sokić, M.; Stojanović, J. Technologically Sustainable Route for Metals Valorization from Jarosite-PbAg Sludge. Minerals 2021, 11, 255. [Google Scholar] [CrossRef]
- Rus, A.; en Sunkar, A.S.; Topkaya, Y.A. Zinc and lead extraction from Cinkur leach residues hydrometallurgical method. Hydrometallurgy 2008, 93, 45–50. [Google Scholar]
- Turan, M.D.; Altundoğan, H.S.; Tümen, F. Recovery of zinc and lead from zinc plant residue. Hydrometallurgy 2004, 75, 169–176. [Google Scholar] [CrossRef]
- Asokan, P.; Saxena, M.; Asolekar, S.R. Hazardous jarosite use in developing nonhazardous product for engineering application. J. Hazard. Mater. 2006, 137, 1589–1599. [Google Scholar] [CrossRef]
- Asokan, P.; Saxena, M.; Asolekar, S.R. Recycling hazardous jarosite waste using coal combustion residues. Mater. Charact. 2010, 61, 1342–1355. [Google Scholar] [CrossRef]
- Pelino, M. Recycling of zinc-hydrometallurgy wastes in glass and glass ceramic materials. Waste Manag. 2000, 20, 561–568. [Google Scholar] [CrossRef]
- Pisciella, P.; Crisucci, S.; Karamanov, A. Chemical durability of glasses obtained by vitrification of industrial wastes. Waste Manag. 2001, 21, 1–9. [Google Scholar] [CrossRef]
- Guler, E.; Seyrankaya, A.; Cöcen, I. Extraction of Lead and Silver from Zinc Leach Residue by Brine Leaching. In Proceedings of the XIIth International Mineral Processing Symposium, Cappadocia Nevsehir, Turkey, 6–8 October 2010. [Google Scholar]
- Yudaev, P.; Chistyakov, E. Chelating Extractants for Metals. Metals 2022, 12, 1275. [Google Scholar] [CrossRef]
- James, S.E.; Watson, J.L.; Peter, J. Zinc Production-a Survey of Existing Smelters and Refineries. Lead-Zinc 2000, 2000, 205–225. [Google Scholar]
- Xu, S.; Wang, G.; Fan, J.; Wang, Z.; Zhang, J.; Chen, J.; Zheng, L.; Pan, J.; Wang, R. Preparation of high purity indium by chemical purification: Focus on removal of Cd, Pb, Sn and removal mechanism. Hydrometallurgy 2020, 200, 105551. [Google Scholar] [CrossRef]
- Burkitbayeva, B.; Argimbayeva, A.; Rakhymbay, G.; Avchukir, K.; Tassibekov, K.; Nauryzbayev, M. Refining of Rough Indium by Method of Reactionary Electrolysis. In Proceedings of the MATEC Web of Conferences REE-2016, Amsterdam, The Netherlands, 23–25 March 2016. [Google Scholar]
- Zelem, J. Application of Amalgam Metallurgy to the Extraction of Indium, Bachelors Theses and Reports, 1928–1970, Summer 6-4-1954, Montana Tech Library, Digital Commons @ Montana Tech. Available online: https://digitalcommons.mtech.edu/bach_theses (accessed on 1 January 2020).
- Li, D.-S.; Dai, Y.-N.; Yang, B.; Liu, D.-C. Purification of indium by vacuum distillation and its analysis. J. Cent. South Univ. 2013, 20, 337–341. [Google Scholar] [CrossRef]
- Alfantazi, A.M.; Moskalyk, R.R. Processing of indium: A review. Miner. Eng. 2003, 16, 687–694. [Google Scholar] [CrossRef]
- Conić, V.; Božić, D.; Dragulović, S.; Avramović, L.J.; Jonović, R.; Bugarin, M. Research on acid leaching of Cu, Zn and In from Jarosite waste. In Proceedings of the XIV International Mineral Processing and Recycling Conference, Belgrade, Serbia, 12–14 May 2021. [Google Scholar]
- Frost, R.; Wills, A.; Kloprogge, J.; Martens, W. Thermal decomposition of ammonium jarosite (NH4)Fe3(SO4)2(OH)6. J. Therm. Anal. Calorim. 2006, 84, 489–496. [Google Scholar] [CrossRef]
- Hu, B.; Richeys, R.; Baird, J. Chemical Equilibrium and Critical Phenomena: Solubility of Indium Oxide in Isobutyric Acid Water Near the Consolute Point. J. Chem. Eng. 2009, 54, 1537–1540. [Google Scholar] [CrossRef]
- Lokanc, M.; Eggert, R.; Redlinger, M. The Availability of Indium: The Present, Medium Term, and Long Term; National Renewable Energy Laboratory: Golden, CO, USA, 2015; pp. 1–79. Available online: https://www.nrel.gov/docs/fy16osti/62409.pdf (accessed on 7 April 2023).
- Kangas, P.; Lundström, M.; Orko, I.; Koukkari, P. The Jarogain Process for Metals Recovery from Jarosite and Electric Arc Furnace Dust; VTT Technical Research Centre of Finland Ltd.: Espoo, Finland, 2017; ISBN 978-951-38-8596-0. ISSN 2242-1211. [Google Scholar]
- Rocchetti, L.; Amato, A.; Beolchini, F. Recovery of indium from liquid crystal displays. J. Clean. Prod. 2016, 116, 299–305. [Google Scholar] [CrossRef]



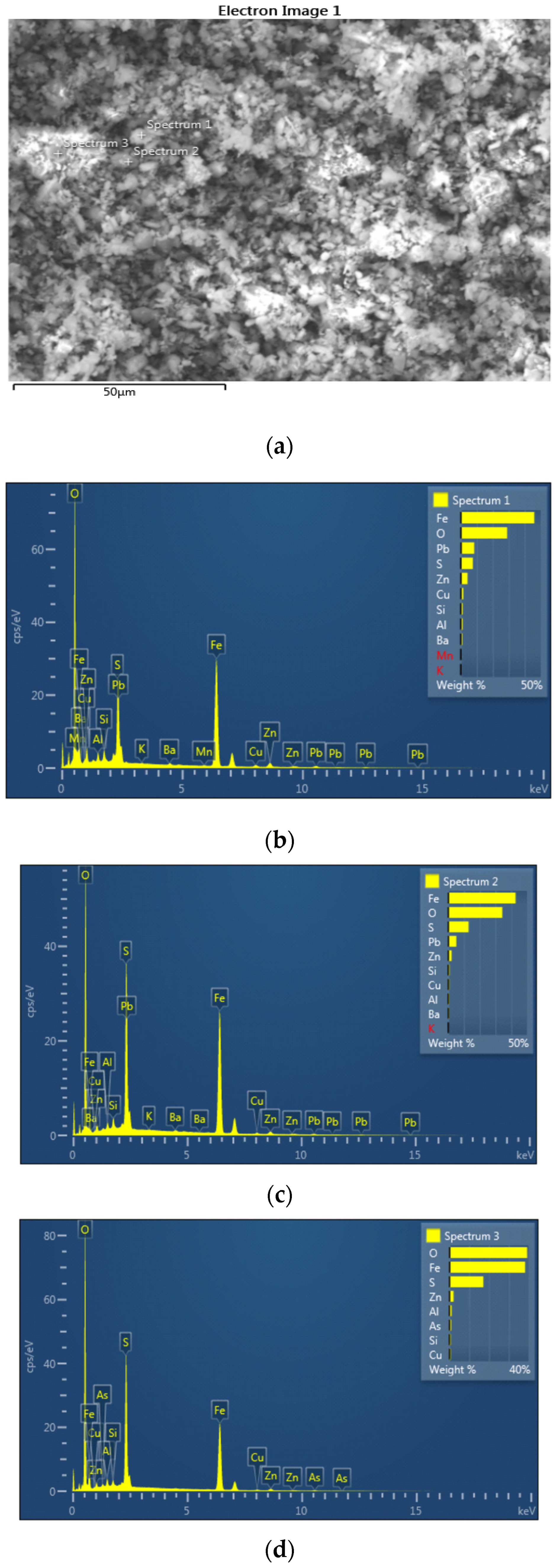
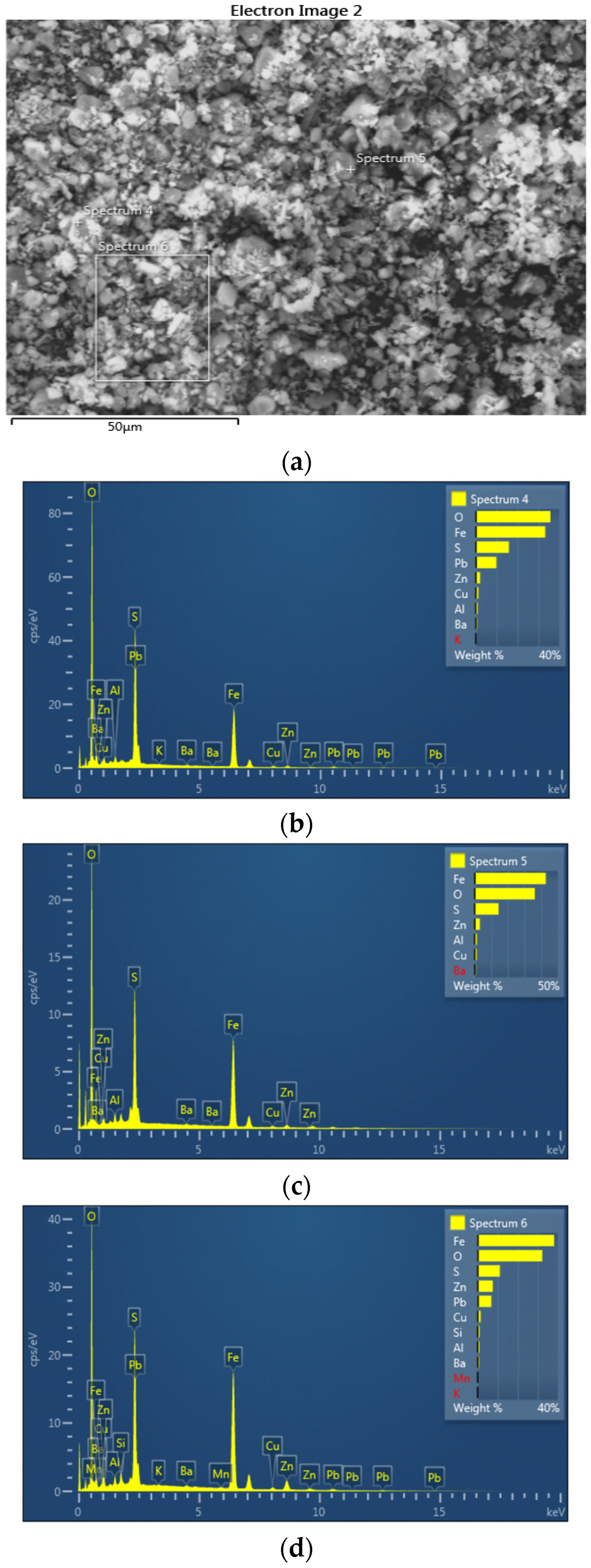


| Spectrum 1 | Spectrum 2 | Spectrum 3 | Spectrum 4 | Spectrum 5 | Spectrum 6 | |
|---|---|---|---|---|---|---|
| O (%) | 28.95 | 34.25 | 39.29 | 35.88 | 36.21 | 32.22 |
| Al (%) | 1.14 | 0.66 | 1.11 | 0.94 | 1.38 | 0.86 |
| Si (%) | 1.14 | 0.81 | 0.65 | - | - | 0.93 |
| S (%) | 7.50 | 12.91 | 17.15 | 15.92 | 14.39 | 11.07 |
| K (%) | 0.11 | 0.07 | - | 0.12 | - | 0.10 |
| Mn (%) | 0.22 | - | - | - | - | 0.29 |
| Fe (%) | 45.97 | 42.72 | 38.24 | 33.35 | 42.81 | 38.19 |
| Cu (%) | 1.48 | 0.74 | 0.65 | 1.19 | 1.31 | 1.43 |
| Zn (%) | 4.22 | 2.09 | 2.08 | 2.04 | 3.15 | 7.44 |
| As (%) | - | - | 0.83 | - | - | - |
| Ba (%) | 0.90 | 0.64 | - | 0.71 | 0.75 | 0.71 |
| Pb (%) | 8.37 | 5.11 | - | 9.85 | - | 6.76 |
| Σ | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 | 100.00 |
| Element | Cu % | Zn % | Fe % | Ag % | Pb % | In % | S % |
|---|---|---|---|---|---|---|---|
| Content | 0.7 | 5.39 | 30.61 | 0.034 | 5.68 | 0.034 | 9.61 |
| Roasting Time 4 h | Leaching with H2O (S:L = 1:5), t = 1 h and pH after Leaching | Extent of Leaching | |||||
|---|---|---|---|---|---|---|---|
| Sample Mass (g) | Roasting Temperature (°C) | pH | Cu % | Zn % | Fe % | In % | |
| 1. | 100 | 530 | 2.25 | 91.07 | 91.97 | 9.60 | 100.00 |
| 2. | 100 | 570 | 2.73 | 72.74 | 74.23 | 1.30 | 37.91 |
| 3. | 100 | 630 | 4.97 | 42.87 | 71.10 | 0.51 | 0.08 |
| Cu (g/L) | Zn (g/L) | Fe (g/L) | In (g/L) |
|---|---|---|---|
| 0.63 | 4.95 | 2.93 | 0.034 |
| Metals | Fe | In |
|---|---|---|
| Leaching degree, % | 9.60 | 100.00 |
| Percentage of metal content in solution after precipitation, % | 0.61 | 2.10 |
| Concentration in solution, g/L | 2.93 | 0.034 |
| Concentration of metal content in solution after precipitation, g/L | 0.018 | 0.00071 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Janošević, M.; Conić, V.; Božić, D.; Avramović, L.; Jovanović, I.; Kamberović, Ž.; Marjanović, S. Indium Recovery from Jarosite Pb–Ag Tailings Waste (Part 1). Minerals 2023, 13, 540. https://doi.org/10.3390/min13040540
Janošević M, Conić V, Božić D, Avramović L, Jovanović I, Kamberović Ž, Marjanović S. Indium Recovery from Jarosite Pb–Ag Tailings Waste (Part 1). Minerals. 2023; 13(4):540. https://doi.org/10.3390/min13040540
Chicago/Turabian StyleJanošević, Miloš, Vesna Conić, Dragana Božić, Ljiljana Avramović, Ivana Jovanović, Željko Kamberović, and Saša Marjanović. 2023. "Indium Recovery from Jarosite Pb–Ag Tailings Waste (Part 1)" Minerals 13, no. 4: 540. https://doi.org/10.3390/min13040540
APA StyleJanošević, M., Conić, V., Božić, D., Avramović, L., Jovanović, I., Kamberović, Ž., & Marjanović, S. (2023). Indium Recovery from Jarosite Pb–Ag Tailings Waste (Part 1). Minerals, 13(4), 540. https://doi.org/10.3390/min13040540









