Abstract
Large-scale, high-density geochemical explorations entail enormous workloads and high costs for sample analysis, but, for early mineral exploration, absolute concentrations are not essential. Geochemists require ranges, dynamics of variation, and correlations for early explorations rather than absolute accuracy. Thus, higher work efficiency and lower costs for sample analysis are desirable for geochemical exploration. This study comprehensively analyzed the reliability and applicability of portable X-ray fluorescence (pXRF) spectrometry in geochemical exploration. The results show that pXRF can be applied effectively to rock and rock powder samples, and sample preparation and a longer detection time have been shown to increase the precision of the pXRF results. When pXRF is used on rock samples, if less than 30% of the samples are assessed as containing an element, the element is usually undetectable using pXRF when these rock samples are prepared as rock powders, indicating that the data about the detected element are unreliable; thus, it is suggested that some representative samples should be selected for testing before starting to use a pXRF in a geochemical exploration project. In addition, although the extended detection time increased the reliability of the analysis results, an increase in detection time of more than 80 s did not significantly affect the accuracy of the results. For this reason, the recommended detection time for the pXRF analysis of rock powder samples is 80 s for this study. pXRF has the advantages of being low-cost, highly efficient, and stable, and its results are reliable enough to exhibit the spatial distribution of indicator elements (arsenic, nickel, lead, sulfur, titanium, and zinc) in polymetallic mineralization exploration. Therefore, pXRF is recommendable for practical use in geochemical exploration.
1. Introduction
Geochemical exploration is an efficient method of mineral exploration [1,2,3,4]. It is primarily conducted to determine the elemental concentration and spatial distribution of the elements in various environments (e.g., rock, soil, and stream sediment). To maximize the amount of information about the spatial distribution of elements, geologists tend to attempt to collect more samples at considerably high sampling densities; however, it is costly to process and analyze all the samples collected [2]. Consequently, it is difficult to obtain detailed information about the spatial distribution of indicator elements within a large area. Fortunately, the development of science and technology has continued to improve the performance of scientific instruments and reduce their related costs; the portable X-ray fluorescence (pXRF) spectrometer is an example of such advances [5,6,7,8,9]. pXRF allows geologists to qualitatively or semi-quantitatively obtain the concentrations of a variety of elements in only dozens of seconds [10], thus increasing process efficiency and reducing related costs for large-scale explorations. However, pXRF results are known to vary according to the detection time of the instrument and the type and water content of the samples [11,12]. The yielded data are assumed to have poor accuracy, and, for this reason, pXRF spectrometry is not widely applied in large-scale mineral exploration projects. However, the relative (i.e., relative to a specific threshold) rather than the absolute concentration of indicator elements could potentially provide valuable geochemical anomaly information for the practical implementation of mineral exploration projects [13,14,15,16].
One of the obvious features of conventional laboratory analysis programs is that the sample pretreatment and analysis procedures are time-consuming and require a high level of expertise on the part of the operator. In addition, the sample volume is often very large, so it is inevitable that the samples need to be entrusted to more laboratories for analysis, which poses a challenge to data quality control and the comparison of data from different laboratories. Thus, being able to analyze geochemical samples quickly and easily would be helpful in alleviating the above problems. Many studies have shown that pXRF can be successfully applied to the element content analysis of various matrices, such as vegetation [17,18,19,20], soil or sediment [14,21,22,23,24,25,26], rock [4,27,28], or alloys [29,30]. Although some studies state that pXRF can be applied to the element content analysis of rock samples, the effectiveness and shortcomings of its application in actual geochemical exploration projects have not been systematically evaluated. In order to provide reference for efficient work with pXRF in mineral exploration, it is necessary to evaluate the application of pXRF in the actual geochemical project.
In this study, pXRF was used to estimate the concentrations of elements in rocks and rock powders, and these results were compared to those obtained via laboratory analysis. Some factors which may affect the analytical results were discussed, and the applicability of pXRF in actual geochemical exploration projects was also evaluated.
2. Materials and Methods
2.1. Study Area and Sampling Method
The study area is located in the Kalatage ore-concentrated area in the East Tian Shan mountains of Xinjiang, China; it is approximately 130 km west of Hami city. The sampling area was located on the southern margin of the Tulufan–Hami Basin. In recent years, several copper–gold–polymetallic deposits have been discovered in the Kalatage ore-concentrated area, including the Red Sea VMS (volcanic-associated massive sulfide) copper–zinc deposit, the Hongshan epithermal copper–gold deposit, the Hongshi–Meilinge epithermal copper deposit, and the Yudai porphyry copper–gold deposit [31,32] (Figure 1).
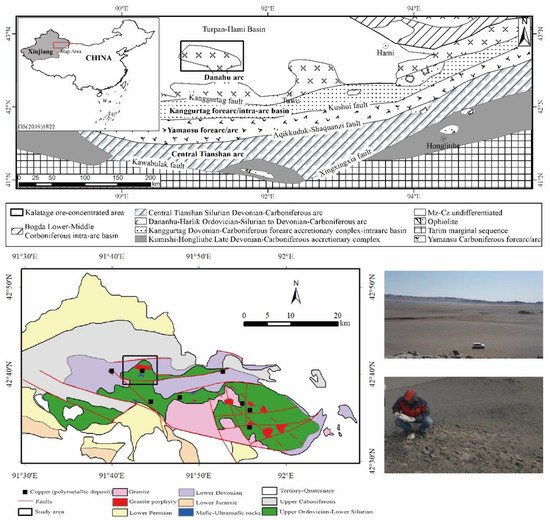
Figure 1.
Geological map of the study area and the typical landscape (modified from Xiao et al., 2004 [33]; Mao et al., 2018 [31]).
The sampling area extended approximately 30 km2 beyond the center of the Yudai porphyry copper–gold deposit, which is located in the Gobi Desert. The wide valleys in the study area are covered by Aeolian sand and alluvium, and the thickness of the cover ranges from several centimeters to meters. Additionally, the rocks on the hillside are weathered away, with irregular patterns.
Most of the materials on the surface are affected by Aeolian sand; thus, the sediment is generally considered to be unsuitable for sampling. The rocks were collected along thirteen north–south routes, each of which is approximately 5 km long, with 200-m intervals between each sampling site and 500-m intervals between each route.
2.2. pXRF Analysis
An energy-dispersive pXRF (Thermo Fisher Scientific Niton XL3t 950 GOLDD+; Waltham, MA, USA) was used to analyze the 316 rock and rock powder samples. To analyze the rock samples, one piece of rock was randomly selected from each sample, the detection time was set to 120 s, and the MINING Cu–Zn mode was used. To analyze the rock powder samples, all rock samples were ground into a powder with a particle diameter of less than 75 μm. Then, the rock powder samples were packed into snap-and-seal polyethylene sample bags (note that a pile of new empty polyethylene bags was tested to make sure they contained no contaminants for the rock powder samples), and each sample was placed on a mobile test stand. Four maximum detection times were applied (40, 80, 120, and 200 s) under the conditions of the MINING Cu–Zn mode. Each of the four built-in exciter filters in the pXRF spectrometer required a quarter of the total detection time for one sample.
2.3. Laboratory Analysis
To determine the concentrations of the elements in the samples accurately, all 316 rock samples were entrusted to a laboratory of the Non-ferrous Geological Exploration Bureau in Xinjiang. Samples were crushed to 75 μm or less, and then the rock powder samples were digested and analyzed according to the methods listed in Table 1.

Table 1.
List of analytical equipment used to measure the concentrations of the 14 elements.
3. Results
3.1. Usability Analysis of pXRF
To investigate the usability of pXRF technology in geochemical exploration, a pXRF was applied to the rocks and corresponding rock powder samples were collected from the study area. The detection time was set to 120 s for the rock samples and 40, 80, 120, or 200 s for the rock powder samples.
A total of 35 elements were identified with pXRF when analyzing the 97 randomly selected rock samples (Figure 2).
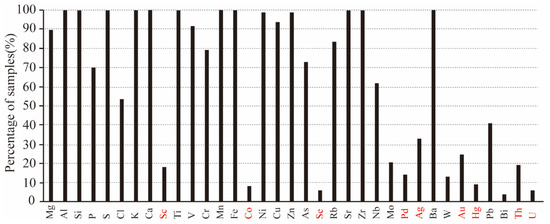
Figure 2.
The percentage of rock samples in which an element could be detected by pXRF.
In total, 26 of the 35 identified elements were measured by operating the pXRF spectrometer in copper–zinc mode to analyze the rock powder samples derived from the above-mentioned 97 rock samples (Figure 3). The results revealed that the detection time could notably affect the results, especially for most of the trace elements. All of the identified elements can be classified into three groups (G1: Mg, Ni, W, and Bi; G2: Al, Ba, Ca, Cl, Si, S, K, V, Ti, Mn, Fe, Rb, Sr, Zn, and Zr; and G3: As, Cr, Cu, Mo, Nb, P, and Pb) according to their different detection times. Very little information was obtained from the G1 group since these elements were only detected in a small percentage of samples. The G2 group was mainly composed of rock-forming elements that could be detected in a detection time as short as 40 s. The G3 group’s results indicated that the detection capability increased as the detection time increased. The G3 group’s results also indicated that G3 contains perhaps three of the most important geochemical pathfinders for hydrothermal ore deposits: Cu, As, and Mo.
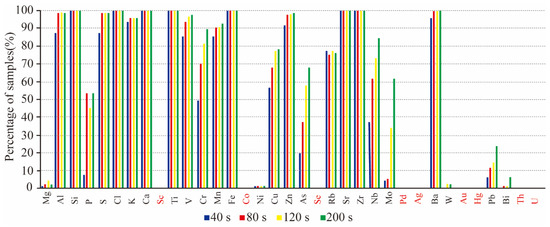
Figure 3.
The percentage of rock powder samples in which an element can be detected by pXRF based on different detection times.
3.2. Influence of Detection Time on Analytical Error
To elucidate the relationship between the detection time and analytical error for each element, four different detection times (40, 80, 120, and 200 s) were applied when the pXRF spectrometer was used to analyze the 97 rock powder samples.
The analytical error results (i.e., two times the standard deviation) were compared based on the detection time for each element detected by the pXRF spectrometer, and the comparative analysis revealed that the analytical error decreased as the detection time increased. The analytical error results for three major elements (aluminum, silicon, and iron) and three trace elements (copper, lead, and zinc) are presented as examples in Figure 4. Table 2 shows that the reduction in the analytical error was most significant when the detection time was increased from 40 s to 80 s, and there is an exponential relationship between the detection time and the analytical error.
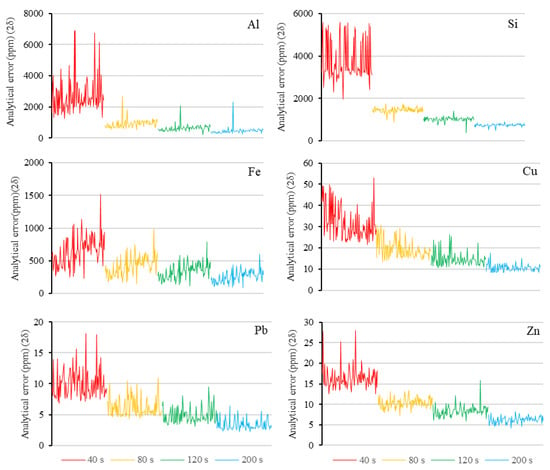
Figure 4.
Analytical error of rock powder samples by pXRF based on different detection times.

Table 2.
The relationship between detection time and mean analytical error.
Increasing the detection time from 40 s to 80 s can result in a relatively significant decrease in error, and the error fluctuation also tended to decrease with the increasing detection time (Figure 4). Therefore, it can be stated that the analytical error and its fluctuation is relatively low and acceptable.
3.3. The Reliability and Stability of pXRF
To verify the results of the pXRF-based analysis method, the rock and rock powder sample results were compared to those obtained via laboratory analysis. The results for eight common indicator elements in mineral exploration (i.e., arsenic, copper, molybdenum, nickel, lead, sulfur, titanium, and zinc) are shown in Figure 5.
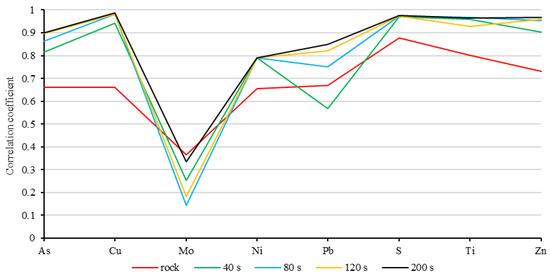
Figure 5.
Similarity between the analysis results of pXRF and laboratory. The 40 s, 80 s, 120 s, and 200 s are the results of rock powder samples reported by pXRF.
The result in Figure 5 shows that the reliability of the concentrations via pXRF analysis was not consistent across different elements, with the results for molybdenum being the least reliable. This is because the molybdenum concentration determined via pXRF was imprecise regardless of whether a rock or rock powder sample was used. However, the pXRF-derived results for arsenic, copper, nickel, lead, sulfur, titanium, and zinc were relatively reliable, especially for the rock powder samples. The results for most of the elements (e.g., arsenic and lead) were observed to be more accurate when a longer detection time was applied. However, the extent of the increase in accuracy was modest for most of the elements.
To investigate the robustness of the pXRF results, 32 rock powder samples were randomly selected and individually subjected to three successive rounds of pXRF-based analysis. The correlation coefficient results for these repeated measurements are shown in Figure 6. It can be seen that all the correlation coefficients, except Ni and Cr, were close to one.
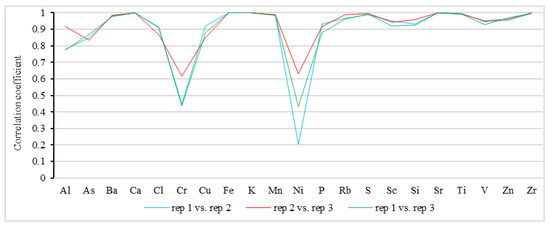
Figure 6.
Correlation between different repeated measurements of rock powder samples by pXRF.
4. Discussion
4.1. The Reliability of pXRF
This study demonstrates that the results of applying pXRF to analyze the prepared samples (e.g., rock powder with an average particle diameter of fewer than 75 μm) can be comparable to those obtained via laboratory analysis, which is consistent with other studies [2,34]. Some previous studies have indicated that other factors may also affect the results of pXRF-based analysis (e.g., the water content, sample size, and roughness on the sample surface) [11,12,35]. However, the effect of these factors on the analysis results can be mitigated by simple pretreatment such as natural drying, screening, and/or crushing. Comparison of the pXRF-derived and laboratory analysis-based results for arsenic, copper, nickel, lead, sulfur, titanium, and zinc revealed that those obtained via pXRF-based analyses were generally reliable when the samples were crushed to 75-μm-sized pieces and the pXRF detection time was at least 40 s. Regarding the influence of detection time, previous studies have shown that a longer detection time corresponded to results with higher accuracy and less errors [36]. However, the results from this study demonstrated that the pXRF detection time does not need to be very long because extending it too much will reduce efficiency. More importantly, and as reported in previous studies, for most of the elements within the detectable pXRF range, the increase in accuracy tends to become less significant as the detection time is increased from 40 s to 200 s [37].
Scholars and geologists should pay attention to the fact pXRF can report some elements (the red labeled elements in Figure 2) when being used on the rock samples that were not reported by pXRF when it was used on the rock powder samples (the red labeled elements in Figure 3). Therefore, it can be stated that although the analytical results of some elements (e.g., As, Cu, Ni, Pb, S, Ti, and Zn) were acceptable in semi-quantitative analysis, the results of the red labeled elements in Figure 2 were inaccurate. Figure 2 also demonstrates that the red labeled elements can only be reported in less than 30% of the total samples, some even less than 20%, and the red labeled elements cannot be reported when the rock samples are prepared as rock powders (Figure 3). Therefore, this study states that the results are unreliable or doubtful when pXRF is used on rock samples if an element can only be reported by pXRF in less than 30% of the total samples.
The various materials (rocks, soils, stream sediments, etc.) taken from the natural environments are usually heterogeneous. In order to obtain more accurate element content, the samples need to be adequately prepared before element content analysis, which is exactly the practice used in conventional geochemical analysis work. Therefore, obtaining accurate analysis results using conventional geochemical analysis is time-consuming and expensive. Unlike conventional methods, pXRF improves efficiency and reduces cost at the expense of the accuracy of the element content analysis. Therefore, pXRF spectrometry is generally considered to be a qualitative or semi-quantitative analytical tool. It should be noted that pXRF analysis results are indeed not as accurate as laboratory analyses, but the application of pXRF makes it possible to analyze more samples, and more data can provide more information, which increases the reliability of the pXRF analysis results.
4.2. The Geological Significance of the Result
Taking S, Zn, As and Cu—which are some of the important indicator elements in mineral exploration—as examples, the inverse distance weighted (IDW) interpolation algorithm was used to exhibit their spatial distribution, and the cell sizes of these raster maps were resampled to approximately 200 m according to the sampling density of this study. The results demonstrate that the contents of various elements detected by pXRF are different from the results of laboratory analysis, but different sample analysis methods have little influence on the spatial distribution characteristics of elements, especially for the S, Zn and Cu in Figure 7, where it is difficult to find the differences between the results obtained by different sample analysis methods without careful comparison. Although it is easy to find the differences between the spatial distributions of arsenic obtained by different sample analysis methods, the high-content areas of As-pXRF and As-lab are similar (Figure 7).
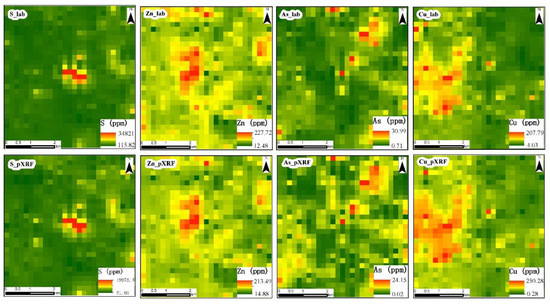
Figure 7.
Spatial distribution of S, Zn, As, and Cu reported by laboratory (S_lab, Zn_lab, As_lab, Cu_lab) and pXRF (S_pXRF, Zn_pXRF, As_pXRF, Cu_pXRF).
A primary goal of geochemical exploration is to identify and delineate anomalous concentrations and spatial distributions of a wide variety of elements in a certain area [2,38]. Therefore, in the case of mineral exploration, it is more important to know the relative concentrations of elements than to obtain very accurate values of the elements in different regions. A pXRF-based analysis can yield reliable results that are adequate to identify the relative elemental concentrations in different regions (Figure 7). Thus, pXRF spectrometry is a reliable tool for the exploration of many kinds of deposits that can be indicated by As, Cu, Ni, Pb, S, Ti, and Zn.
4.3. The Implications for Further Work
The results of a few elements (e.g., molybdenum in Figure 5) reported by pXRF are unreliable, but the analytical error tended to decrease with time. Take molybdenum as an example: its content in most of the samples (98.81%) is lower than their abundance in China’s continental crust [39,40] according to the data reported by the laboratory. Therefore, if the contents of some elements measured by pXRF are lower than their abundances in the continental crust, the analysis data of these elements cannot provide valuable information for mineral exploration, since they state that there is no metallogenic potential in this region for that element, which is valuable information by itself.
Although many countries in the world, including China, have carried out national scale geochemical explorations, the previous work is insufficient for further mineral exploration. First, the sampling density is relatively low in many of the projects that have been carried out, which is sufficient for the identification of large-scale geochemical anomalies but cannot meet the requirements of identifying small-scale geochemical anomalies and locating ore deposits. Second, geochemical surveys have not been carried out in many countries or regions (e.g., some developing countries or the Turpan–Hami basin, Junggar basin, and Tarim basin in Xinjiang, China). Therefore, there is a large amount of geochemical exploration work that still needs to be carried out in the future, and pXRF can be used in the following ways:
Geologists can collect as many samples as possible in the field. If rocks are being collected and reliable element content is expected, the rocks can be crushed into rock powders; if soils or stream sediments are being collected, the samples can be naturally dried and/or screened according to the specific project, and pXRF can be used on the simply prepared samples directly without the need for digestion, laminating, or melting. A relatively small number of samples can also be selected for accurate analysis based on the results of the pXRF analysis, avoiding blindly entrusting all samples to professional laboratories for analysis.
Note that each of the four built-in exciter filters in the pXRF spectrometer required the same amount of time for detection in this study; however, researchers and geologists may only select one, two, or three of the built-in exciter filters as needed to further improve efficiency.
5. Conclusions
- The pXRF can be used to qualitatively analyze rock samples directly for some elements, especially for elements with relatively high abundances in collected samples, but some results are unreliable when the pXRF is used in anisotropic rock samples. This study shows that the pXRF results are unreliable or doubtful when being used on rock samples if an element can only be reported in less than 30% of the total samples.
- Although the absolute precision of the analysis may not be sufficient to be used in conventional petrology, the general trends of the pXRF-derived results were consistent with those observed via laboratory analysis. The pXRF results for prepared samples were comparable to those obtained via laboratory analysis for many elements in the detectable pXRF range. Therefore, scholars and geologists are encouraged to include a pretreatment step (i.e., drying, sieving, and/or milling) to improve data analysis and extend the applicability of pXRF spectrometry.
- The stability of pXRF spectrometry was found to be excellent when it was applied to the elements with high abundance in prepared samples. For most of the elements in the detectable pXRF range, a longer detection time tended to improve the reliability of the results; however, the improvement was not significant for most of the elements when the pXRF detection time was increased beyond 80 s. Thus, an adequate pXRF detection time is suggested to range between 80 and 120 s for powder samples.
- pXRF spectrometry is a low-cost and efficient technology that can be used to detect the concentrations of dozens of elements in rock powder samples. Thus, pXRF is a practical piece of equipment for geochemical exploration.
Author Contributions
Conceptualization, S.Z. and S.L.; methodology, S.Z.; software, S.Z.; investigation, S.Z. and S.L.; resources, J.W.; project administration, J.W. and W.W. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Natural Science Foundation of China (grant number 41902307), the Open Project of Key Laboratory of Xinjiang Uygur Autonomous Region (grant number 2022D04037), the Third Xinjiang Scientific Expedition Program (grant number 2022xjkk1306), and the Science and Technology Major Project of Xinjiang Uygur Autonomous Region, China (grant number 2021A03001-3).
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to other members of our research team will use the data in the near future.
Acknowledgments
The authors are grateful for the anonymous reviewers’ constructive comments and suggestions, and thanks go to Hami Redstone Mining Limited for the food and accommodation support during the field work.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Arne, D.C.; Mackie, R.A.; Jones, S.A. The use of property-scale portable X-ray fluorescence data in gold exploration: Advantages and limitations. Geochem. Explor. Environ. Anal. 2014, 14, 233–244. [Google Scholar] [CrossRef]
- Sarala, P. Comparison of different portable XRF methods for determining till geochemistry. Geochem. Explor. Environ. Anal. 2016, 16, 181–192. [Google Scholar] [CrossRef]
- Lemiere, B. A review of pXRF (field portable X-ray fluorescence) applications for applied geochemistry. J. Geochem. Explor. 2018, 188, 350–363. [Google Scholar] [CrossRef]
- Ryan, J.G.; Shervais, J.W.; Li, Y.; Reagan, M.K.; Li, H.Y.; Heaton, D.; Godard, M.; Kirchenbaur, M.; Whattam, S.A.; Pearce, J.A.; et al. Application of a handheld X-ray fluorescence spectrometer for real-time, high-density quantitative analysis of drilled igneous rocks and sediments during IODP Expedition 352. Chem. Geol. 2017, 451, 55–66. [Google Scholar] [CrossRef]
- Grunsky, E.C. The interpretation of geochemical survey data. Geochem. Explor. Environ. Anal. 2010, 10, 27–74. [Google Scholar] [CrossRef]
- Turner, A.; Solman, K.R. Analysis of the elemental composition of marine litter by field-portable-XRF. Talanta 2016, 159, 262–271. [Google Scholar] [CrossRef] [PubMed]
- Cohen, D.R.; Cohen, E.J.; Graham, I.T.; Soares, G.G.; Hand, S.J.; Archer, M. Geochemical exploration for vertebrate fossils using field portable XRF. J. Geochem. Explor. 2017, 181, 1–9. [Google Scholar] [CrossRef]
- Winterburn, P.A.; Noble, R.R.P.; Lawie, D. Advances in exploration geochemistry, 2007 to 2017 and beyond. Geochem. Explor. Environ. Anal. 2020, 20, 157–166. [Google Scholar] [CrossRef]
- Vonopartis, L.C.; Booysen, R.; Nex, P.A.M.; Kinnaird, J.A.; Robb, L.J. Combined satellite and portable XRF exploration mapping of the Zaaiplaats tin field, South Africa. S. Afr. J. Geol. 2022, 125, 45–60. [Google Scholar] [CrossRef]
- Chen, Z.; Williams, P.N.; Zhang, H. Rapid and nondestructive measurement of labile Mn, Cu, Zn, Pb and As in DGT by using field portable-XRF. Environ. Sci.-Process Impacts 2013, 15, 1768–1774. [Google Scholar] [CrossRef]
- Parsons, C.; Margui Grabulosa, E.; Pili, E.; Floor, G.H.; Roman-Ross, G.; Charlet, L. Quantification of trace arsenic in soils by field-portable X-ray fluorescence spectrometry: Considerations for sample preparation and measurement conditions. J. Hazard. Mater. 2013, 262, 1213–1222. [Google Scholar] [CrossRef]
- Schneider, A.R.; Cances, B.; Breton, C.; Ponthieu, M.; Morvan, X.; Conreux, A.; Marin, B. Comparison of field portable XRF and aqua regia/ICPAES soil analysis and evaluation of soil moisture influence on FPXRF results. J. Soils Sediments 2016, 16, 438–448. [Google Scholar] [CrossRef]
- Reimann, C.; Filzmoser, P.; Garrett, R.G. Background and threshold: Critical comparison of methods of determination. Sci. Total Environ. 2005, 346, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Hall, G.E.; McClenaghan, M.B.; Pagé, L. Application of portable XRF to the direct analysis of till samples from various deposit types in Canada. Geochem. Explor. Environ. Anal. 2016, 16, 62–84. [Google Scholar] [CrossRef]
- Reimann, C.; de Caritat, P. Establishing geochemical background variation and threshold values for 59 elements in Australian surface soil. Sci. Total Environ. 2017, 578, 633–648. [Google Scholar] [CrossRef] [PubMed]
- Tian, M.; Wang, X.; Nie, L.; Liu, H.; Wang, W.; Yan, T. Spatial distributions and the identification of ore-related anomalies of Cu across the boundary area of China and Mongolia. J. Geochem. Explor. 2019, 197, 37–47. [Google Scholar] [CrossRef]
- Asare, M.O.; Hejcman, M. Effect of tree species on the elemental composition of wood ashes and their fertilizer values on agricultural soils. GCB Bioenergy 2022, 14, 1321–1335. [Google Scholar] [CrossRef]
- Zhou, S.; Weindorf, D.C.; Cheng, Q.; Yang, B.; Yuan, Z.; Chakraborty, S. Elemental assessment of vegetation via portable X-ray fluorescence: Sample preparation and methodological considerations. Spectroc. Acta Pt. B Atom. Spectr. 2020, 174, 105999. [Google Scholar] [CrossRef]
- McGladdery, C.; Weindorf, D.C.; Chakraborty, S.; Li, B.; Paulette, L.; Podar, D.; Pearson, D.; Kusi, N.Y.O.; Duda, B. Elemental assessment of vegetation via portable X-ray fluorescence (PXRF) spectrometry. J. Environ. Manag. 2018, 210, 210–225. [Google Scholar] [CrossRef]
- McLaren, T.I.; Guppy, C.N.; Tighe, M.K. A Rapid and Nondestructive Plant Nutrient Analysis using Portable X-Ray Fluorescence. Soil Sci. Soc. Am. J. 2012, 76, 1446–1453. [Google Scholar] [CrossRef]
- Tian, X.; Xie, Q.; Fan, M.; Chai, G.; Li, G. Identification of heavy metal pollutants and their sources in farmland: An integrated approach of risk assessment and X-ray fluorescence spectrometry. Sci. Rep. 2022, 12, 12196. [Google Scholar] [CrossRef] [PubMed]
- Gozukara, G.; Zhang, Y.; Hartemink, A.E. Using pXRF and vis-NIR spectra for predicting properties of soils developed in loess. Pedosphere 2022, 32, 602–615. [Google Scholar] [CrossRef]
- Xue, S.; Korna, R.; Fan, J.; Ke, W.; Lou, W.; Wang, J.; Zhu, F. Spatial distribution, environmental risks, and sources of potentially toxic elements in soils from a typical abandoned antimony smelting site. J. Environ. Sci. 2023, 127, 780–790. [Google Scholar] [CrossRef] [PubMed]
- Nawar, S.; Richard, F.; Kassim, A.M.; Tekin, Y.; Mouazen, A.M. Fusion of Gamma-rays and portable X-ray fluorescence spectral data to measure extractable potassium in soils. Soil Tillage Res. 2022, 223, 105472. [Google Scholar] [CrossRef]
- Andrew, T.; Taylor, A. On site determination of trace metals in estuarine sediments by field-portable-XRF. Talanta 2018, 190, 498–506. [Google Scholar]
- Hu, B.; Chen, S.; Hu, J.; Xia, F.; Xu, J.; Li, Y.; Shi, Z. Application of portable XRF and VNIR sensors for rapid assessment of soil heavy metal pollution. PLoS ONE 2017, 12, e0172438. [Google Scholar] [CrossRef]
- Ross, P.S.; Bourke, A.; Fresia, B. Improving lithological discrimination in exploration drill-cores using portable X-ray fluorescence measurements: (1) testing three Olympus Innov-X analysers on unprepared cores. Geochem. Explor. Environ. Anal. 2014, 14, 171–185. [Google Scholar] [CrossRef]
- Gray, C.A.; Van Rythoven, A.D. A Comparative Study of Porphyry-Type Copper Deposit Mineralogies by Portable X-ray Fluorescence and Optical Petrography. Minerals 2020, 10, 431. [Google Scholar] [CrossRef]
- Roxburgh, M.A.; Heeren, S.; Huisman, D.J.; Van Os, B.J.H. Non-Destructive Survey of Early Roman Copper-Alloy Brooches using Portable X-ray Fluorescence Spectrometry. Archaeometry 2019, 61, 55–69. [Google Scholar] [CrossRef]
- Uemoto, M. On-site Analysis and Identification of Aluminum Alloys with a Handheld X-ray Fluorescence Spectrometer for Closed-loop Recycling. Bunseki Kagaku 2019, 68, 457–464. [Google Scholar] [CrossRef]
- Mao, Q.; Yu, M.; Xiao, W.; Windley, B.F.; Li, Y.; Wei, X.; Zhu, J.; Lu, X. Skarn-mineralized porphyry adakites in the Harlik arc at Kalatage, E. Tianshan (NW China): Slab melting in the Devonian-early Carboniferous in the southern Central Asian Orogenic Belt. J. Asian Earth Sci. 2018, 153, 365–378. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, J.; Wang, Y.; Long, L.; Mao, Q.; Yu, M. Ages and origins of granitoids from the Kalatag Cu cluster in Eastern Tianshan, NW China: Constraints on Ordovician-Devonian arc evolution and porphyry Cu fertility in the Southern Central Asian orogenic belt. Lithos 2019, 330, 55–73. [Google Scholar] [CrossRef]
- Xiao, W.J.; Zhang, L.C.; Qin, K.Z.; Sun, S.; Li, J.L. Paleozoic accretionary and collisional tectonics of the Eastern Tianshan (China): Implications for the continental growth of Central Asia. Am. J. Sci. 2004, 304, 370–395. [Google Scholar] [CrossRef]
- Shuttleworth, E.L.; Evans, M.G.; Hutchinson, S.M.; Rothwell, J.J. Assessment of Lead Contamination in Peatlands Using Field Portable XRF. Water Air Soil Pollut. 2014, 225, 1844. [Google Scholar] [CrossRef]
- Yang, G.; Shang, Z.; Li, L.; Ni, X.; Zhang, M. Application of portable-XRF spectrometry for rapid determination of common heavy metals in soil (in Chinese with English abstract). Appl. Chem. Ind. 2016, 45, 1586–1591. [Google Scholar]
- Kilbride, C.; Poole, J.; Hutchings, T.R. A comparison of Cu, Pb, As, Cd, Zn, Fe, Ni and Mn determined by acid extraction/ICP-OES and ex situ field portable X-ray fluorescence analyses. Environ. Pollut. 2006, 143, 16–23. [Google Scholar] [CrossRef]
- Hall, G.E.M.; Bonham-Carter, G.F.; Buchar, A. Evaluation of portable X-ray fluorescence (pXRF) in exploration and mining: Phase 1, control reference materials. Geochem. Explor. Environ. Anal. 2014, 14, 99–123. [Google Scholar] [CrossRef]
- Seyedrahimi-Niaraq, M.; Mahdiyanfar, H. Introducing a new approach of geochemical anomaly intensity index (GAII) for increasing the probability of exploration of shear zone gold mineralization. Geochemistry 2021, 81, 125830. [Google Scholar] [CrossRef]
- Taylor, S.R. Abundance of chemical elements in the continental crust—A new table. Geochim. Cosmochim. Acta 1964, 28, 1273–1285. [Google Scholar] [CrossRef]
- Li, T. Element Abundances of China’s Continental Crust and Its Sedimentary Layer and Upper Continental Crust. Acta Geochim. 1995, 14, 16–32. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).