Prediction and Potential Treatment of Underground Contaminated Water Based on Monitoring of pH and Salinity in a Coal Mine Waste Heap, Southern Poland
Abstract
1. Introduction
2. Methodology
2.1. Case Study Description
- Model the hydrodynamic field for the analyzed area under conditions of different evapotranspiration and different precipitation;
- Simulate the distribution of chemicals in the model of the analyzed area;
- Calculate, with the Zone Budget module, the amount of water flowing through the specific area;
- Determine the direction of groundwater flow using the Modpath module;
- Simulate the sorption process for nZVI using the Freundlich and Langmuir isotherm in the MT3DMS module.
2.2. Assessment of the Chemical and Phase Composition of the Deposited Waste
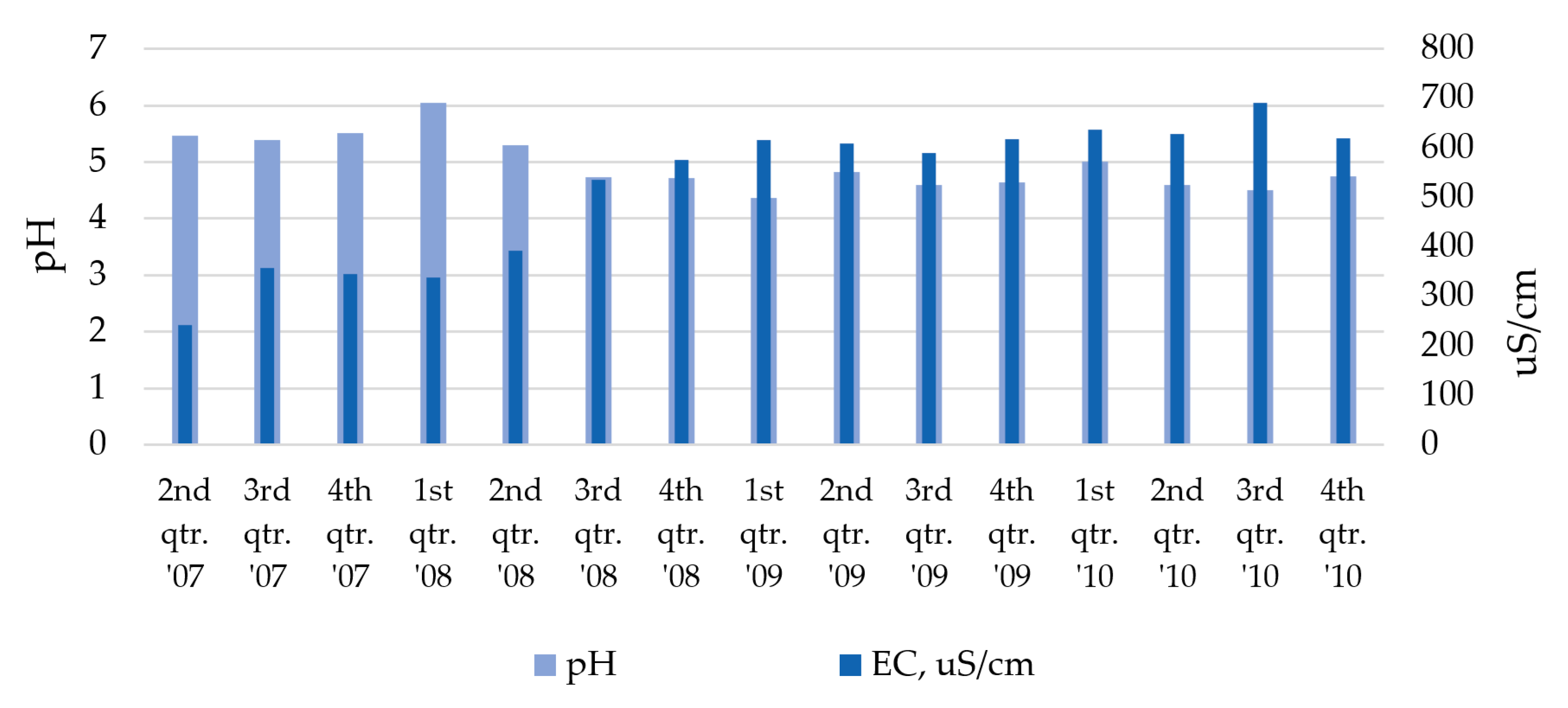
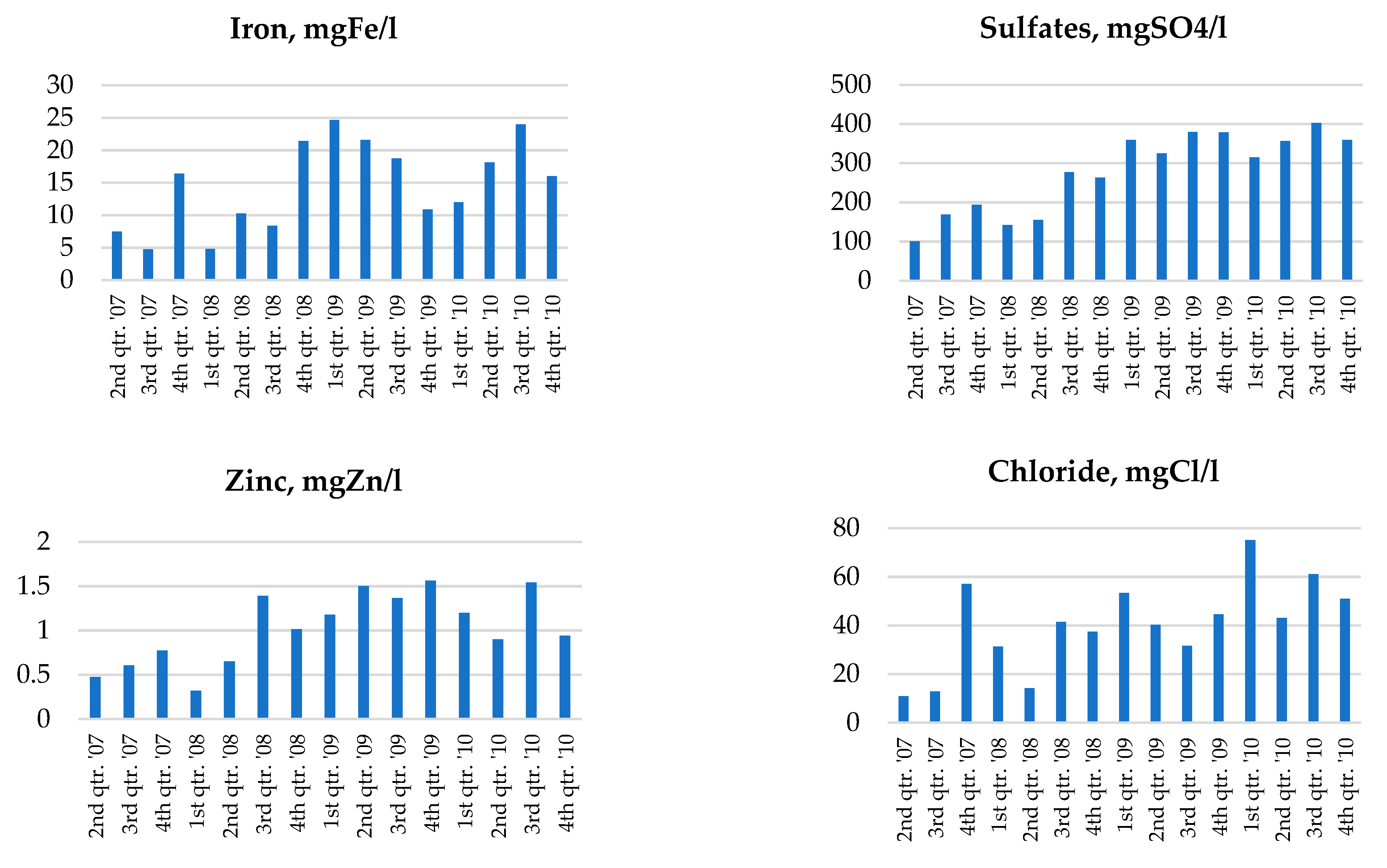
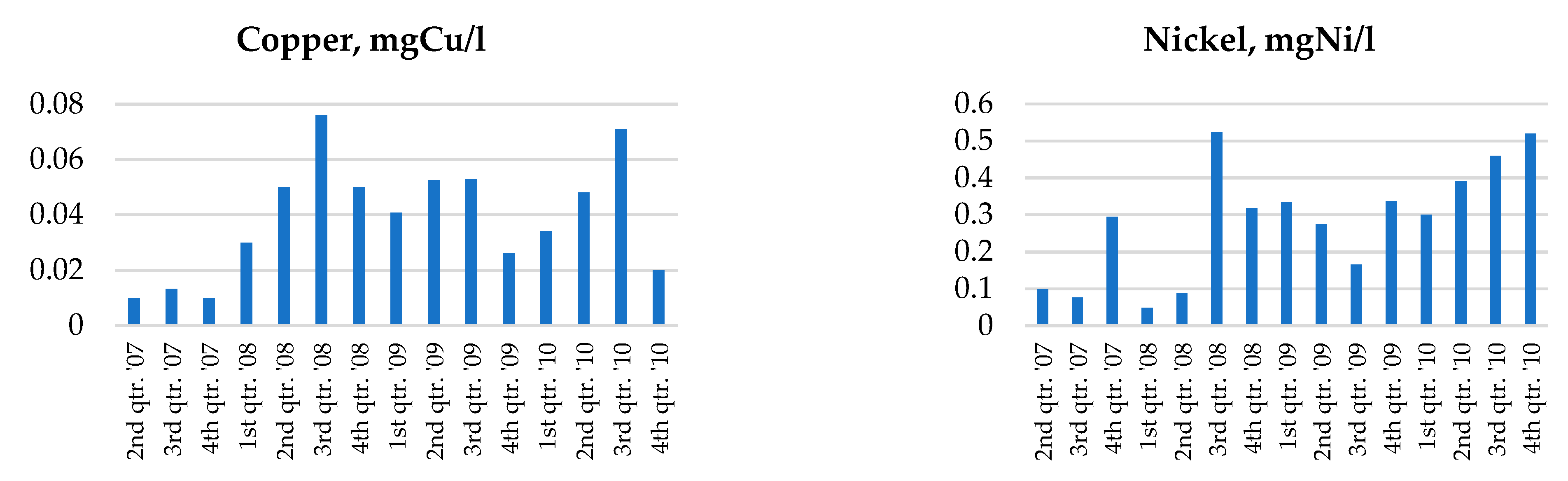
2.3. Groundwater Monitoring Results
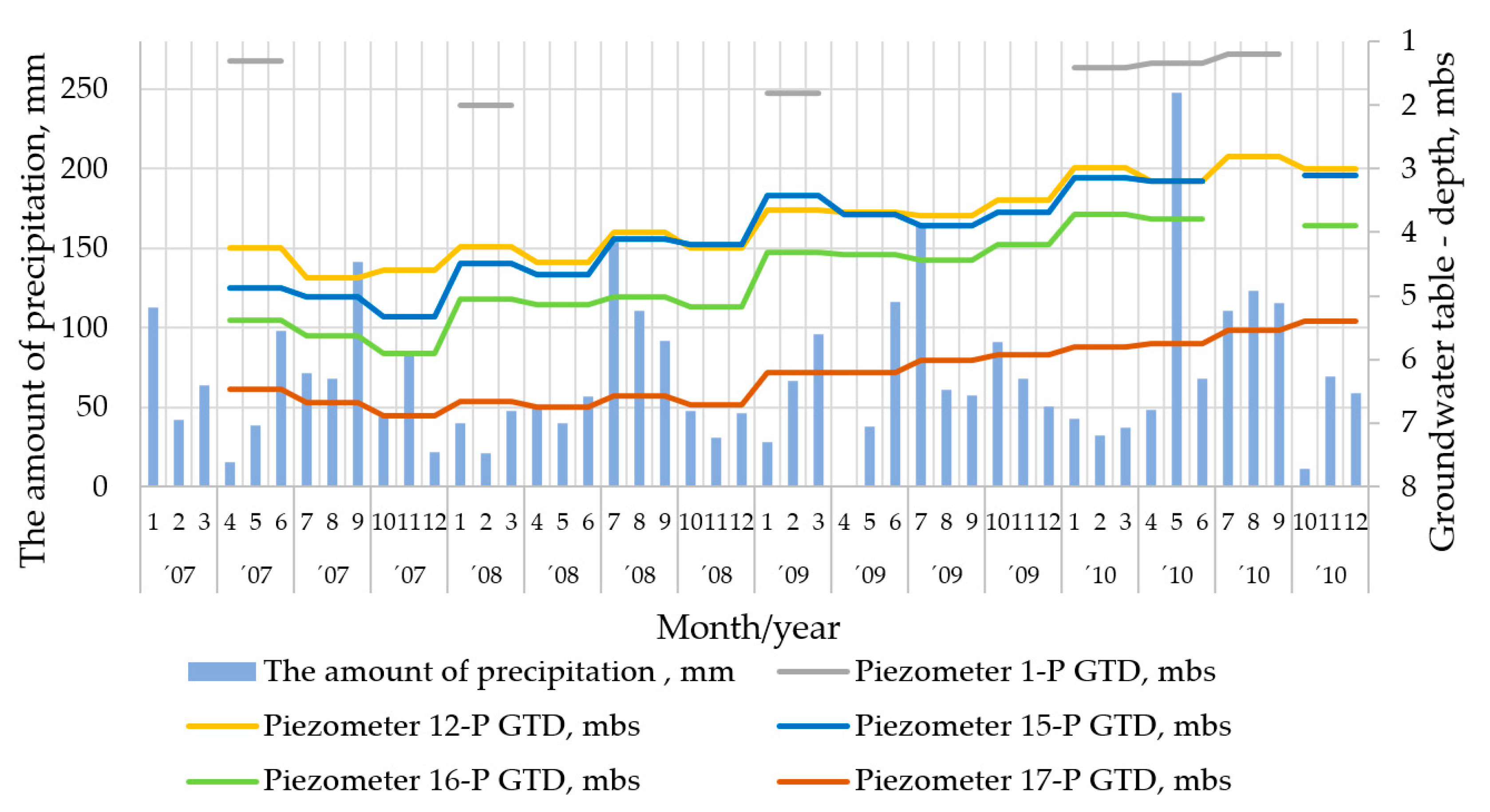
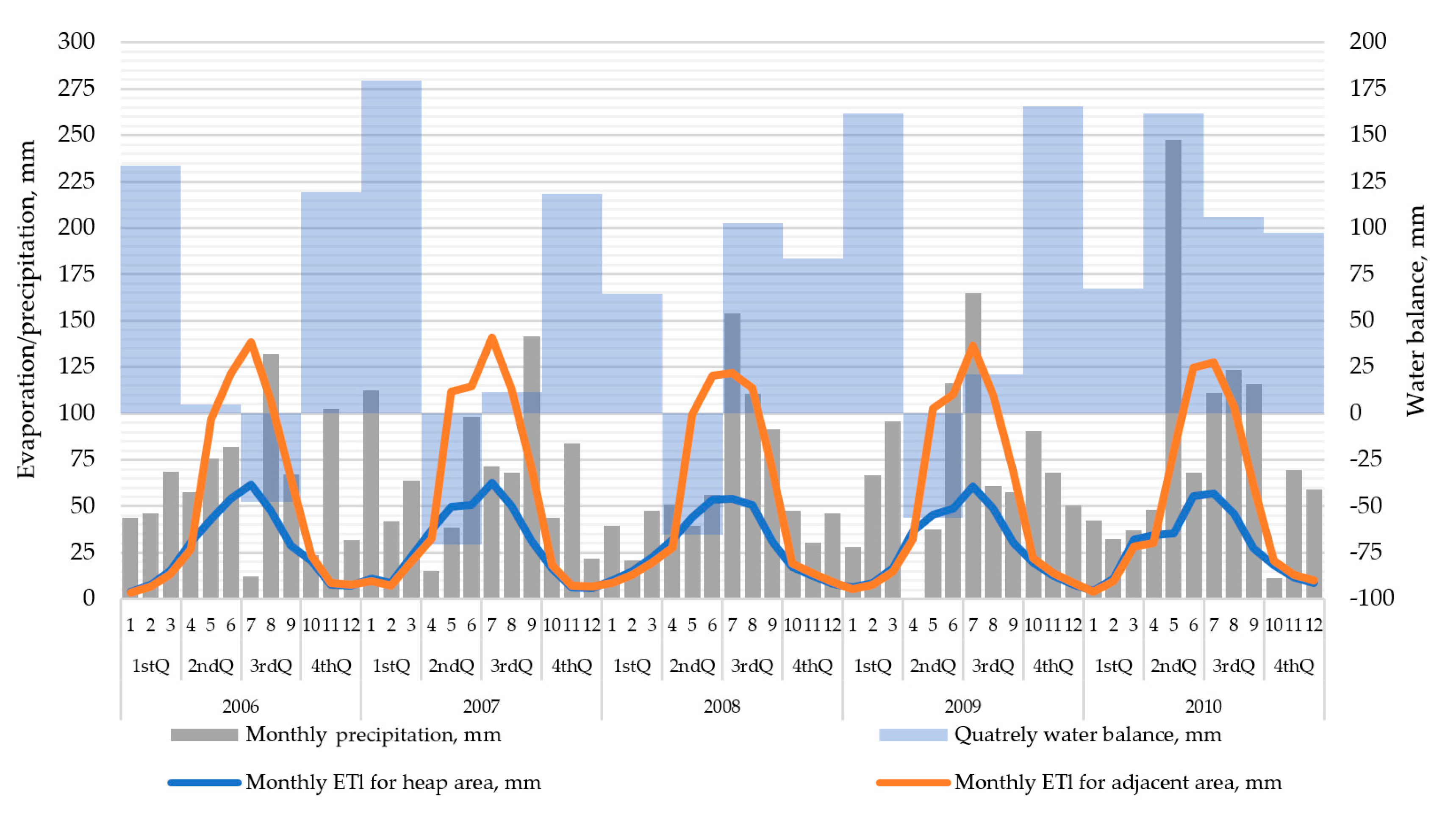
2.4. Geological, Hydrogeological, and Meteorological Conditions
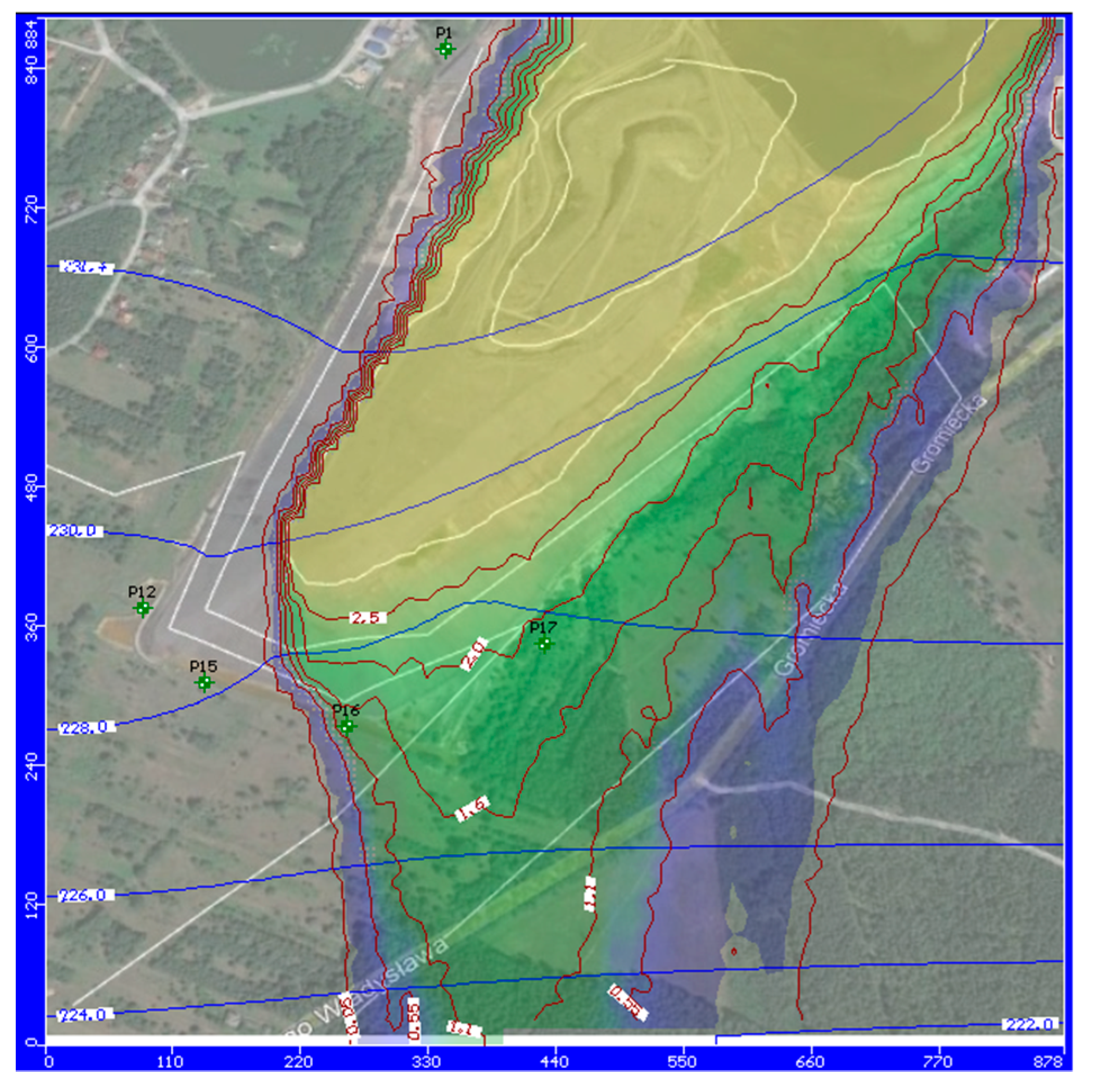
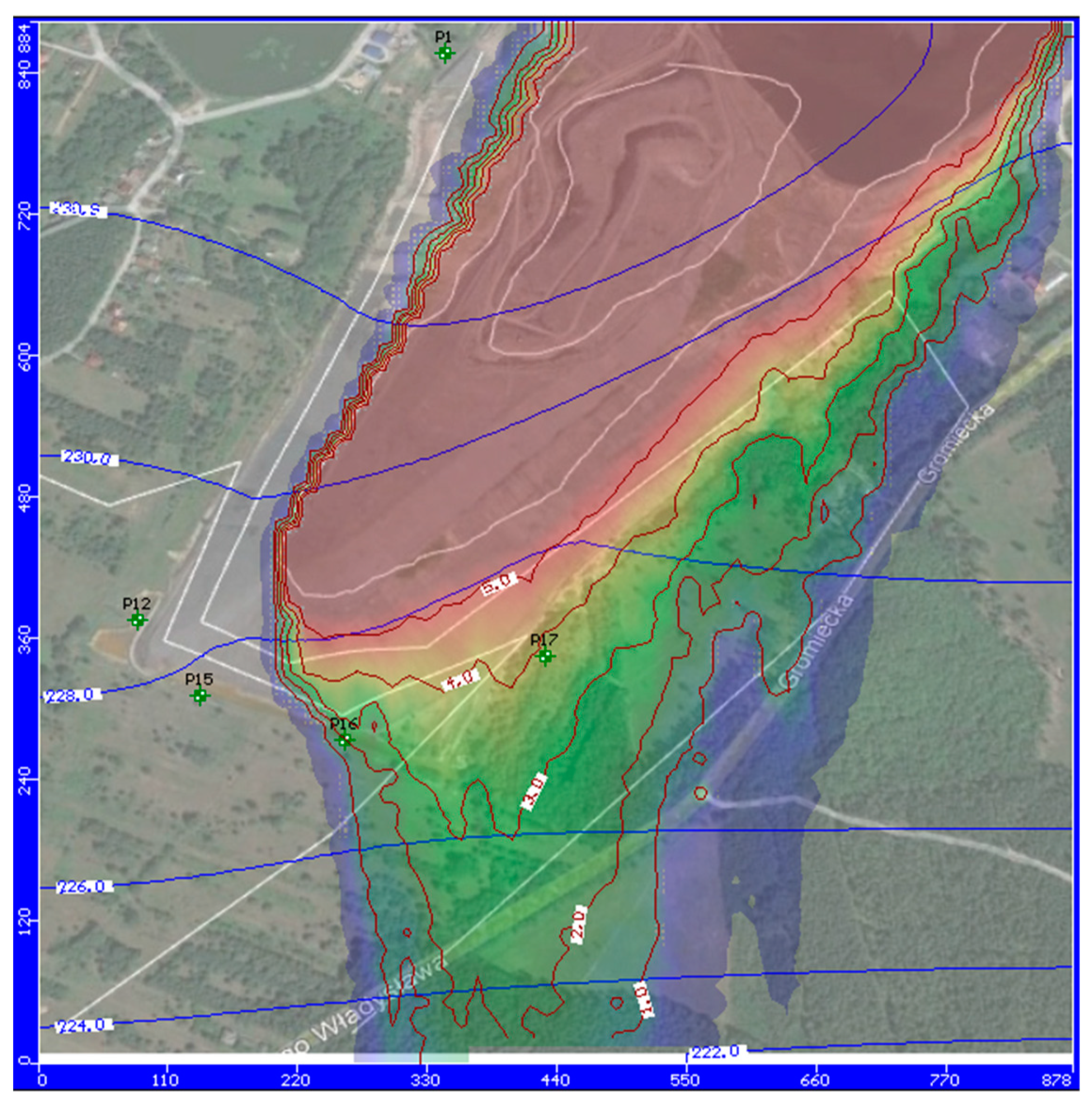
3. Results and Interpretation
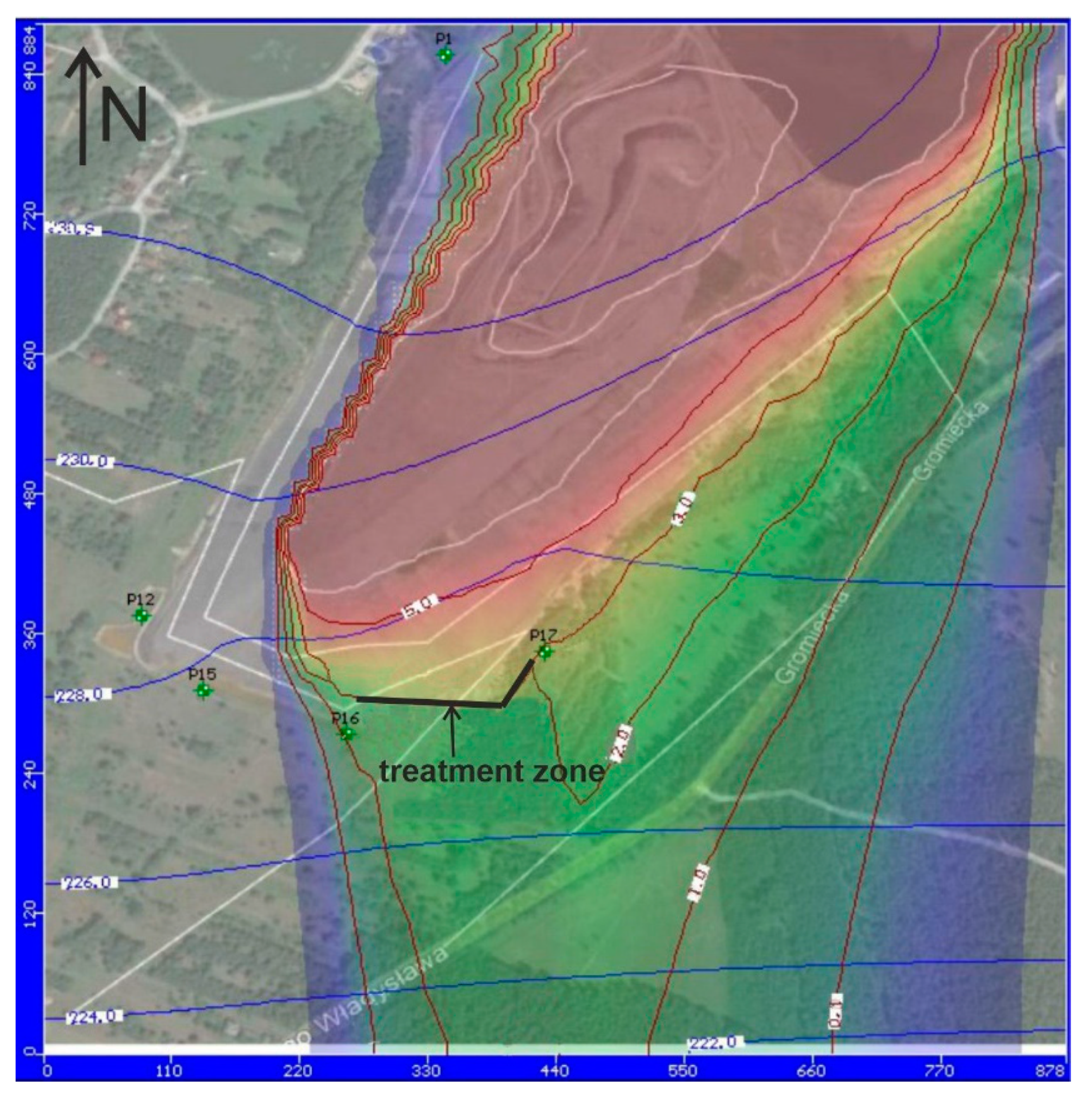
3.1. Modeling the Removal of Pollutants from Groundwater
3.2. General Discussion
4. Conclusions
- Based on geological, hydrogeological, and meteorological data, as well as the monitoring of the groundwater table and the physicochemical parameters of this water and the concentrations of dissolved inorganic substances, models of groundwater flow and migration of pollutants can be prepared by applying the Visual MODFLOW software. Using these models, it is possible to observe detrimental changes in the aquatic environment and use this information to make decisions regarding the need for active remediation of groundwater. In the case of using a reactive material in the form of nZVI, the place and time of injection of this material can be determined. The adverse environmental changes may be related to excessive precipitation, which, after a long period of low precipitation, causes the intensification of leaching of inorganic contaminants from the mine waste.
- In the case of the presence of a large amount of sulfidic waste in the heap, and a small amount of neutralizing minerals, especially carbonates, it was proposed that three parameters (EC, pH, and the level of the groundwater table) be monitored and used to create a model of pollutant flow in the groundwater. These parameters are to be sent online to the e-cloud, and from there, to the model of groundwater flow and pollution migration. Based on these results it will be possible to obtain quick information regarding the need to carry out remediation works and perform additional environmental analyses, if necessary.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cañedo-Argüelles, M.; Kefford, B.J.; Piscart, C.; Prat, N.; Schafer, R.B.; Schulz, C.J. Salinization of rivers: An urgent ecological issue. Environ. Pollut. 2013, 173, 157–167. [Google Scholar] [CrossRef]
- Van Dam, R.A.; Harford, A.J.; Lunn, S.A.; Gagnon, M.M. Identifying the cause of toxicity of a saline mine water. PLoS ONE 2014, 9, e106857. [Google Scholar] [CrossRef]
- Zhao, Q.; Guo, F.; Zhang, Y.; Ma, S.; Jia, X.; Meng, W. How sulfate-rich mine drainage affected aquatic ecosystem degradation in norteastern China, and potential ecological risk. Sci. Total Environ. 2017, 609, 1093–1102. [Google Scholar] [CrossRef]
- Turunen, K.; Räsänen, T.; Hämäläinen, E.; Hämäläinen, M.; Pajula, P.; Nieminen, S.P. Analysing contaminant mixing and dilution in river waters influenced by mine water discharges. Water Air Soil Pollut. 2020, 231, 317. [Google Scholar] [CrossRef]
- Molenda, T. Impact of a saline mine water discharge on the development of a meromictic pond, the Rontok Wielki Reservoir, Poland. Mine Water Environ. 2018, 37, 807–814. [Google Scholar] [CrossRef]
- Janson, E.; Markowska, M.; Labaj, P.; Wrana, A.; Zawartka, P. A preliminary assessment of climate change impacts: Implications for mining activity in Polish coal regions. Arch. Min. Sci. 2020, 3, 703–717. [Google Scholar] [CrossRef]
- Rusydi, A.F. Correlation between conductivity and total dissolved solid in various type of water: A review. IOP Conf. Ser. Earth Environ. Sci. 2018, 118, 012019. [Google Scholar] [CrossRef]
- Hem, D. Study and Interpretation the Chemical of Natural of Characteristics Natural Water, 3rd ed.; USGS Water Supply Paper; US Government Printing Office: Washington, DC, USA, 1985; Volume 2254, pp. 66–69. [Google Scholar]
- Nordstrom, D.K.; Blowes, D.W.; Ptacek, C.J. Hydrogeochemistry and microbiology of mine drainage: An update. Appl. Geochem. 2015, 57, 3–16. [Google Scholar] [CrossRef]
- Pérez-Ostalé, E.; Grande, J.A.; Valente, T.; De La Torre, M.L.; Santisteban, M.; Fernández, P.; Diaz-Curiel, J. Relationships between sources of acid mine drainage and the hydrochemistry of acid effluents during rainy season in the Iberian Pyrite Belt. Water Sci. Tech. 2016, 73, 345–354. [Google Scholar] [CrossRef]
- Chlot, S.; Widerlund, A.; Husson, A.; Ohlander, B.; Ecke, F. Effects on nutrient regime in two recipients of nitrogen-rich mine effluents in northern Sweden. Appl. Geochem. 2013, 31, 12–24. [Google Scholar] [CrossRef]
- Meays, C.; Nordin, R. Ambient Water Quality Guidelines for Sulphate. Technical Appendix; Water Protection and Sustainability Branch, Environmental Sustainability and Strategic Policy Division, Ministry of Environment: Smithers, BC, Canada, 2013; 55p. [Google Scholar]
- Chandler, L.; Harford, A.J.; Hose, G.C.; Humphrey, C.L.; Chariton, A.; Greenfield, P.; Davis, J. Saline mine-water alters the structure and function of prokaryote communities in shallow groundwater below a tropical stream. Environ. Pollut. 2021, 284, 117318. [Google Scholar] [CrossRef]
- Gray, N.F. Field assessment of acid mine drainage contamination in surface and ground water. Environ. Geol. 1996, 27, 358–361. [Google Scholar] [CrossRef]
- Fytas, K.; Hadjigeorgiou, J. An assessment of acid rock drainage continuous monitoring technology. Environ. Geol. 1995, 25, 36–42. [Google Scholar] [CrossRef]
- Lyew, D.; Sheppard, J. Use of conductivity to monitor the treatment of acid mine drainage by sulphate-reducing bacteria. Water Res. 2001, 35, 2081–2086. [Google Scholar] [CrossRef]
- Suponik, T.; Neculita, C.M. Reactivity of nZVI in the removal of Cu(II) and Zn(II) from synthetic mine drainage. Environ. Prot. Eng. 2021, 47, 93–108. [Google Scholar] [CrossRef]
- Suponik, T. Optmization of the PRB (Permeable Reactive Barriers) Parameters for Selected Area of Dumping Site; Publishing House of the Silesian University of Technology: Gliwice, Poland, 2011; ISBN 978-83-7335-833-1. [Google Scholar]
- Suponik, T.; Franke, D.; Frączek, R.; Nowińska, K.; Pierzyna, P.; Różański, Z.; Wrona, P. The impact of precipitation on the groundwater of coal waste dump. J. Sustain. Min. 2021, 20, 13–19. [Google Scholar] [CrossRef]
- Hydrogeologic, W. Visual MODFLOW Flex 7.0 Integrated Conceptual & Numerical Groundwater Modeling Software, Conceptual Modeling Tutorial. 2021. 47p. Available online: https://www.waterloohydrogeologic.com/wp-content/uploads/2021/01/VMODFlex_ConceptualModeling_Tutorial.pdf (accessed on 24 February 2022).
- Allen, R.G.; Pereira, L.S.; Raes, D.; Smith, M. Crop Evapotranspiration: Guidelines for Computing Crop Water Requirements—FAO Irrigation and Drainage Paper 56; Food and Agriculture Organization of the United Nations: Rome, Italy, 1998; ISBN 92-5-104219-5. [Google Scholar]
- Xu, C.-Y.; Singh, V.P. Evaluation of three complementary relationship evapotranspiration models by water balance approach to estimate actual regional evapotranspiration in different climatic regions. J. Hydrol. 2005, 308, 105–121. [Google Scholar] [CrossRef]
- Matuszko, D.; Celiński-Mysław, D. Warunki solarne krakowa i możliwości ich wykorzystania w helioenergetyce. Acta Sci. Pol. Form. Circumiectus 2016, 15, 103–111. [Google Scholar] [CrossRef]
- Costello, L.R.; Jones, K.S. A Guide to Estimating Irrigation Water Needs of Landscape Plantings in California; University of California Cooperative Extension, California Department of Water Resources: Sacramento, CA, USA, 2000. [Google Scholar]
- Koniarski, M.; Matysiak, B.; Treder, W. Ewapotranspiracja i współczynniki roślinne (k) dla czterech odmian różanecznika przy zastosowaniu regulowanego deficytu nawadniania. Zesz. Nauk. Inst. Ogrod. 2016, 24, 71–80. [Google Scholar]
- Kuźniar, A.; Twardy, S.; Kowalczyk, A.; Kostuch, M. An assessment of the water requirements of a mountain pasture sward in the Polish Western Carpathians. J. Water Land Dev. 2011, 15, 193–208. [Google Scholar] [CrossRef]
- Szwed, M. Projections of changes of areal evapotranspiration for different land-use units in the Wielkopolska Region (Poland). Theor. Appl. Climatol. 2017, 130, 291–304. [Google Scholar] [CrossRef][Green Version]
- Treder, J.; Treder, W.; Klamkowski, K. Determination of irrigation requirements and crop coefficients using weighing lysimeters in perennial plants. Infrastucture Ecol. Rural Areas 2017, 1213–1228. [Google Scholar] [CrossRef]
- Zyromski, A.; Biniak-Pieróg, M.; Szulczewski, W.; Kordas, L.; Kabala, C.; Galka, B. Mathematical Modeling of Evapotranspiration of Selected Energy Crops; Wydawnictwo Uniwersytetu Przyrodniczego We Wroclawiu: Wroclaw, Poland, 2016; ISBN 978-83-7717-253-7. [Google Scholar]
- Allen, R.G.; Pruitt, W.O.; Raes, D.; Smith, M.; Pereira, L.S. Estimating evaporation from bare soil and the crop coefficient for the initial period using common soils information. J. Irrig. Drain. Eng. 2005, 131, 14–23. [Google Scholar] [CrossRef]
- Quinn, R.; Parker, A.; Rushton, K. Evaporation from bare soil: Lysimeter experiments in sand dams interpreted using conceptual and numerical models. J. Hydrol. 2018, 564, 909–915. [Google Scholar] [CrossRef]
- Snyder, R.L.; Geng, S.; Orang, M.; Sarreshteh, S. Calculation and simulation of evapotranspiration of applied water. J. Integr. Agric. 2012, 11, 489–501. [Google Scholar] [CrossRef]
- Lorenc, H. Susze i Opady Maksymalne w Polsce. Prezentacja na Seminarium PK GWP. 16 December. 2011. Available online: http://gwppl.org/data/uploads/prezentacje/Susze%20i%20opady%20maksymalne%20w%20Polsce.pdf (accessed on 6 December 2021).
- Opis Susz w Okresie 1996–2005. Available online: http://posucha.imgw.pl/index.php?option=com_content&view=article&id=25&Itemid=59 (accessed on 6 December 2021).
- Staśko, S.; Tarka, R.; Buczyński, S. Ekstremalne zdarzenia meteorologiczne i ich odzwierciedlenie w stanach wód. Przegląd Geol. 2017, 65, 5. [Google Scholar]
- Lèbre, E.; Corder, G.D.; Golev, A. Sustainable practices in the management of mining waste: A focus on the mineral resource. Miner. Eng. 2017, 107, 34–42. [Google Scholar] [CrossRef]
- Nikitina, N. Geoethics: Theory, Principles, Problems, 2nd ed.; Geoinformmark, Ltd.: Moscow, Russia, 2016; 256p, ISBN 978-5-98877-061-9. [Google Scholar]
- Bondu, R.; Cloutier, V.; Rosa, E.; Benzaazoua, M. A review and evaluation of the impacts of climate change on geogenic arsenic in groundwater from fractured bedrock aquifers. Water Air Soil Pollut. 2016, 227, 296. [Google Scholar] [CrossRef]
- Wolkersdorfer, C.; Nordstrom, D.K.; Beckie, R.D.; Cicerone, D.S.; Elliot, T.; Edraki, M.; Valente, T.; Alves França, S.C.; Kumar, P.; Oyarzún Lucero, R.A.; et al. Guidance for the integrated use of hydrological, geochemical, and isotopic tools in mining operations. Mine Water Environ. 2020, 39, 204–228. [Google Scholar] [CrossRef]
- Kurukulasuriya, D.; Howcroft, W.; Moon, E.; Meredith, K.; Timms, W. Selecting environmental water tracers to understand groundwater around mines: Opportunities and limitations. Mine Water Environ. 2022, 1–13. [Google Scholar] [CrossRef]
- Fytas, K. Développement de la Surveillance en Continu Pour le Drainage Minier Acide. Phase I; Department of Mining and Metallurgy, Laval University: Quebec City, QC, Canada; 180p.
- Garrick, H.; Digges La Touche, G.; Plimmer, B.; Hybert, D. Assessing the hydraulic performance of tailings management facilities. In Proceedings of the 17th Extractive Industry Geology Conference, Ormskirk, UK, 5–8 September 2012; Extractive Industry Geology Conferences Ltd.: Witney, UK, 2014. [Google Scholar]
- Brookfield, A.E.; Blowes, D.W.; Mayer, K.U. Integration of field measurements and reactive transport modelling to evaluate contaminant transport at a sulfide mine tailings impoundment. J. Contam. Hydrol. 2006, 88, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Colombo, L.; Gzyl, G.; Mazzon, P.; Łabaj, P.; Frączek, R.; Alberti, L. Stochastic Particle Tracking Application in Different Urban Areas in Central Europe: The Milano (IT) and Jaworzno (PL) Case Study to Secure the Drinking Water Resources. Sustainability 2021, 13, 10291. [Google Scholar] [CrossRef]
- Anawar, H.M. Impact of climate change on acid mine drainage generation and contaminant transport in water ecosystems of semi-arid and arid mining areas. Phys. Chem. Earth Parts A/B/C 2013, 58–60, 13–21. [Google Scholar] [CrossRef]
- Varadhi, S.N.; Gill, H.; Apoldo, L.J.; Liao, K.; Blackman, R.A.; Wittman, W.K. Full-scale nanoiron injection for treatment of groundwater contaminated with chlorinated hydrocarbons. In Proceedings of the Natural Gas Technologies 2005 Conference, Orlando, FL, USA, 30 January–2 February 2005. [Google Scholar]
- Tiraferri, A.; Chen, K.L.; Sethi, R.; Elimelech, M. Reduced aggregation and sedimentation of zero-valent iron nanoparticles in the presence of guar gum. J. Colloid Interface Sci. 2008, 324, 71–79. [Google Scholar] [CrossRef]

| Element | Unit | Concentration in Sample No. | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | Average | ||
| As | ppm | 16.3 | 28.8 | 9.80 | 14.9 | 4.28 | 19.9 | 0.84 | 13.5 |
| Sn | ppm | 7.17 | 31.8 | 2.63 | 18.8 | 3.80 | 0.49 | 1.41 | 9.44 |
| Cd | ppm | 1.61 | 2.78 | <0.002 | 1.24 | 0.98 | <0.002 | 0.08 | 1.34 |
| Co | ppm | 168 | 59.8 | 19.5 | 89.6 | 4.41 | 0.87 | 2.01 | 49.3 |
| Mo | ppm | 7.07 | 2.87 | 1.11 | 0.87 | <0.006 | 1.89 | 0.14 | 2.33 |
| Hg | ppm | <0.017 | 0.045 | 0.087 | 0.484 | <0.017 | 0.084 | <0.017 | 0.181 |
| Ni | ppm | 52.3 | 58.1 | 0.48 | 5.84 | 78.2 | 55.1 | 10.6 | 37.3 |
| Cu | ppm | 42.4 | 199 | 6.47 | 79.4 | 88.5 | 105 | 59.4 | 83.0 |
| Zn | ppm | 278 | 195 | 31.2 | 423 | 256 | 120 | 155 | 208 |
| Sr | ppm | 101 | 105 | 1.48 | 34.4 | 4.70 | 80.7 | 4.07 | 47.5 |
| Pb | ppm | 160 | 55.8 | 55.5 | 94.7 | 107 | 102 | 163 | 105 |
| Ba | ppm | 288 | 510 | 156 | 4.71 | 0.98 | 12.5 | 25.7 | 142 |
| Cr | ppm | 5.46 | 101 | 1.23 | 97.1 | 4.78 | 45.4 | 33.2 | 41.2 |
| Mn | ppm | 325 | 110 | 124 | 169 | 125 | 189 | 155 | 171 |
| Layers in the Model | Effective Porosity * [-] | Filtration Coefficient ** k [m/s] | Longitudinal Dispersion *** [m2/d] | Transverse Dispersion *** [m2/d] |
|---|---|---|---|---|
| coarse-grained coal waste | 0.55 | 3.82 × 10−5 | 2.5 | 0.25 |
| fine-grained coal waste | 0.35 | 3.42 × 10−7 | 1.0 | 0.1 |
| sand | 0.25 | 2.8 × 10−5 | 5.0 | 0.5 |
| clay | 0.01 | 1.0 × 10−9 | 0.1 | 0.01 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suponik, T.; Franke, D.M.; Neculita, C.M.; Mzyk, T.; Frączek, R. Prediction and Potential Treatment of Underground Contaminated Water Based on Monitoring of pH and Salinity in a Coal Mine Waste Heap, Southern Poland. Minerals 2022, 12, 391. https://doi.org/10.3390/min12040391
Suponik T, Franke DM, Neculita CM, Mzyk T, Frączek R. Prediction and Potential Treatment of Underground Contaminated Water Based on Monitoring of pH and Salinity in a Coal Mine Waste Heap, Southern Poland. Minerals. 2022; 12(4):391. https://doi.org/10.3390/min12040391
Chicago/Turabian StyleSuponik, Tomasz, Dawid M. Franke, Carmen M. Neculita, Tadeusz Mzyk, and Robert Frączek. 2022. "Prediction and Potential Treatment of Underground Contaminated Water Based on Monitoring of pH and Salinity in a Coal Mine Waste Heap, Southern Poland" Minerals 12, no. 4: 391. https://doi.org/10.3390/min12040391
APA StyleSuponik, T., Franke, D. M., Neculita, C. M., Mzyk, T., & Frączek, R. (2022). Prediction and Potential Treatment of Underground Contaminated Water Based on Monitoring of pH and Salinity in a Coal Mine Waste Heap, Southern Poland. Minerals, 12(4), 391. https://doi.org/10.3390/min12040391








